Abstract
The intensification of agriculture has caused severe environmental damage affecting numerous ecosystem services, such as freshwater quality. Understanding and monitoring its impact on the aquatic biota is fundamental to improve the water quality. In this paper, we investigated the influence of environmental condition established by an agropastoral landscape on the local and regional diversity of rotifers. For this, the local and regional diversity were modelled with generalized additive mixed models and canonical correspondence analysis, respectively. Our results indicated that both the local and regional structures of the rotifer assemblage are affected by high nutrient concentrations and other environmental variables. Spatial distance also influences the regional structure, accounting for 23% of the variation in the beta-diversity. The environmental variables are responsible for 9% of the variation in the beta-diversity and the surrounding matrix for 5%. Together, these components and the interactions among them, account for 54% of the regional structure of the rotifer assemblage. Based on our study, we concluded that agriculture lands, primarily citrus crops, adversely influence the rotifer assemblage both locally and regionally. That negative impact, in turn, results in a cascade effect in the aquatic ecosystem, leading mainly to the loss of species-poor taxa.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
As part of the zooplankton community, rotifers play a key role in tropical freshwaters. They are dominant in many aquatic ecosystems (Roth, 2009; Rico-Martínez et al., 2016), including many eutrophic environments (Liang et al., 2020), so that their high abundance results from their mode of reproduction and short regeneration time (Wallace et al., 2006). Rotifera also represents one of the richest components of the zooplankton community in freshwaters (Sampaio et al., 2002; Lucena et al., 2015; Rosińska et al., 2019; Karpowicz et al., 2020; Li & Chen, 2020). Moreover, they play an essential link between the phytoplankton and the carnivorous fish larvae and invertebrates (Roth, 2009; Wallace et al., 2015), and respond to changes in the trophic state and aquatic environment (Sládecek, 1983; May & O’Hare, 2005; Wen et al., 2011; Gopko & Telesh, 2013; Ismail & Adnan, 2016), reflecting their useful role in the monitoring and management of the ecosystem (Sládecek, 1983; Saksena, 1987; Jeppesen et al., 2011; Perbiche-Neves et al., 2013; Li & Chen, 2020; Liang et al., 2020).
Small and shallow reservoirs are natural or artificial lakes used as a source of water supply, which are characterized and permanently influence by the watersheds (Bucci et al., 2015). They are vital to humanity because they provide water for human and animal consumption, irrigation, industrial activities, energy generation, transport, recreation and the dilution of waste (Postel & Carpenter, 1997; Lanna & Braga, 2015). But the reservoir ecosystem is susceptible to anthropogenic impact that can alter the patterns of water quality and consequently, affect its biota and restrict its use for humans (Naiman et al., 1995; Rhodes et al., 2001; Sperling, 2012; Tundisi et al., 2015; Hoffmann et al., 2019). In tropical countries, many farms have small reservoirs or ponds built by damming streams and used for human and animal water consumption and agricultural irrigation (Postel & Carpenter, 1997; Mioduszewski, 2012; Tundisi et al., 2015).
Due to their utility and location close to cultivated areas and urban centres, these small reservoirs typically contain an ecosystem very susceptible to anthropogenic impact that can alter water quality (Alsharif & Fouad, 2012; Zhang et al., 2015; Giri & Qiu, 2016; Winton et al., 2019) and affect its biota (Naiman et al., 1995; Norton et al., 2013; Turcotte et al., 2017). Erosion, pollution and contamination are among the primary anthropogenic impacts that affect aquatic ecosystems and lead to the loss of biodiversity (Janse et al., 2015; Tundisi et al., 2015; Bashir et al., 2020). Consequently, water quality and productivity are modulated by the quantity and the type of external nutrient loadings (Rockström et al., 2017; Withers et al., 2018; Hoffmann et al., 2019). Those nutrient inputs reflect the climatic regime, soil type and adjacent land use (Kennedy & Walker, 1990).
The use of fertilizer is one of the agricultural practices closely associated with a considerable loss of biodiversity (Foley et al., 2005; Isbell et al., 2013). When applied to the soil, many fertilizers, especially nitrogen and phosphorus, are transported to the freshwater ecosystem via stormwater runoff, causing pollution of water bodies (Liu et al., 2019) and, consequently, their eutrophication (Tilman et al., 2001; Huang et al., 2017). As result of the eutrophication, cyanobacteria populations grow excessively triggering a cascade effect that alters the aquatic food web (McCarthy et al., 2007; Du et al., 2015; Li & Chen, 2020), and lead to loss of biodiversity and reduced ecosystem functions (Schoumans et al., 2014). Since some species are very sensible to eutrophication, it is possible to observe changes in the rotifer species composition (Sládecek, 1983) which can be measured by beta diversity.
The negative effect of the high concentrations of nutrients on the alfa diversity from aquatic environments has been reported by different studies (Pinto-Coelho, 1998; Leibold, 1999; Matsumura-Tundisi & Tundisi, 2005; Brito et al., 2011; Jeppesen et al., 2011; Wen et al., 2011; Major et al., 2017; Li & Chen, 2020). Other studies have reported the influence of nutrients on the beta diversity (Soininen et al., 2007; Ji et al., 2013; Symons et al., 2014; Mantovano et al., 2015; Oh et al., 2017; Li & Chen, 2020). However, the great majority of these studies did not identify the causal relationships between predictor and response variables. They also did not integrate both local (alpha) and regional (beta) diversity. Mokany et al. (2011) highlight that combining alpha and beta diversity represents a powerful way to help fill gaps in knowledge of the structure of ecological communities, especially for that including highly diverse and poorly studied taxa. Furthermore, those studies did not analyze how different agropastoral landscapes influence the zooplankton diversity. Besides that, approaches that investigate the influence of different types of crops on zooplankton diversity are vital for understanding the dynamics of modified landscapes and for supporting decision-making in effective nature conservation (Scholz et al., 2012; Hritonenko & Yatsenko, 2013).
The influence of the nutrients in the beta diversity has been also reported by several studies (Soininen et al., 2007; Ji et al., 2013; Symons et al., 2014; Mantovano et al., 2015; Oh et al., 2017; Li & Chen, 2020), but no study has effectively analyzed how agropastoral landscapes influence the beta diversity of zooplankton. Considering that different plants show different nutritional requirements (FAO, 2004) and their crops may be conditioned to different tillage and management practices (Pradhan et al., 2011), it is expected that different types of crops influence the diversity of reservoirs in different ways. The loss of biodiversity and reduced ecosystem functions in the aquatic ecosystem are among the adverse environmental consequences of eutrophication (Schoumans et al., 2014). However, some aspects of the zooplankton community structure, like phylogenetic diversity, are not well explored and only superficially understood.
Thus, this study aimed to investigate whether special variables associated with agropastoral landscape influence the local and regional diversity of rotifers in shallow reservoirs. Based on an integrative and effective data analysis, our investigation sought to understand the relationships between the differences found for pasture and sugarcane and citrus crops with species sharing, species richness and taxonomic distinctness. Moreover, we searched the direct influence of an agropastoral landscape on beta-diversity, discriminating the types of landscape components and relating the environmental and geographical spaces. Through this approach, we could understand the regulatory and structuring processes being influenced by agriculture.
Materials and methods
Study area
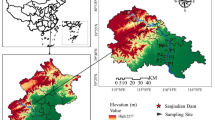
We sampled eight reservoirs built by damming of small rivers in three watersheds in the northwestern State of São Paulo-Brazil: (1) São José dos Dourados Basin, (2) Baixo-Tietê Basin and (3) Turvo-Grande Basin (Fig. 1). These reservoirs are located in an area of transition between the Atlantic Forest and Cerrado (Brazilian Savanna), with an agropastoral landscape composed of cattle pasture, sugarcane and oranges (Fig. 1). The sampled reservoirs are located in a region with a temperate climate that is characterized by dry winters and hot summers, with temperatures typically higher than 22 °C, according to Köppen–Geiger (Peel et al., 2007).
Sampling design
Rotifer sampling was conducted during the rainy season of 2008 and 2009. Samples were taken by vertical hauls, from the bottom to the surface, with a 45 μm plankton mesh net in the littoral and pelagic zones of all reservoirs. The depth of the sampling sites varied from 0.45 to 5.1 m, with the littoral zones presenting average depth of 1.45 m (± 0.35) and pelagic zones with average depth of 3.3 m (± 0.34). A minimum of 300 l of water was filtered in each of the samples. The littoral zones of some reservoirs were sampled twice because one of their margins was covered by native vegetation and the other one by an agropastoral practice (pasture, citrus crop or sugar-cane crop). Eight samples from the pelagic zone and 10 from the littoral zone were collected. The collected rotifers were anaesthetized with carbonated water and posteriorly fixed with 4% formalin. In addition, we measured temperature, pH, dissolved oxygen (DO), total nitrogen (TN) and phosphorus (TP) concentrations. The thermal profile, dissolved oxygen and pH were measured using a multiparameter Horiba U-10 in three depth (surface, middle and bottom); averages of the three depths were used. Moreover, 500 ml of water was collected from all sampling sites to estimate the TN and TP concentration, following methods proposed by Mackereth et al. (1978), and Strickland & Parsons (1972), respectively.
Statistical analyses
Alpha diversity was characterized by species richness and taxonomic distinctness. The total species richness was estimated using the Chao1 index. Taxonomic distinctness was calculated using the average taxonomic distinctness (Δ+) and the variation in the taxonomic distinctness (Λ+) proposed by Clarke & Warwick (1998, 2001) and Warwick & Clarke (1998). Both the species richness and taxonomic distinctness indices (Δ+ and Λ+) were modelled as dependent variables with TP, TN, DO, temperature and pH (independent variables). For this, generalized additive mixed models (GAMM) were used for modelling. We specified a Poisson error distribution for species richness and a Gaussian error distribution for Δ+ and Λ+. Moreover, we applied the penalized smoothing term thin plate regression spline (s) for independent variables (TP, TN and Temperature) and the tensor product smoothing term (te) for the independent covariables (DO and pH). The pH was also treated as a dependent variable and modelled with TN and TP concentrations (independent variables) based on the Gaussian distribution. For the models with pH as dependent variables, we applied the tensor product smoothing term for both TN and TP. The TN was logarithmic to decrease the sizable value discrepancy and allow the detection of possible non-linear relationships. All models included the limnologic zones and the type of agropastoral landscape as random effects, along with spatial autocorrelation. Five structures of spatial autocorrelations were tested (exponential, Gaussian, linear, spherical, and quadratic rational). The models with the best fit were chosen based on the Akaike Information Criterion (AIC) and residual variance. For this, we calculated the ΔAIC and multiplied the residual variance ((residual standard deviation)2) with ΔAIC + 1, so that for species richness models it was considered the Pearson residuals. In this way, the best fitted model should present the lowest value of that selection criterion. Alfa diversity was estimated using functions of the R package vegan version 2.0-8 (Oksanen et al., 2020), and modelled with functions of the package mgcv version 1.8-31 (Wood, 2020).
Beta diversity (variation in species composition among sites) was analyzed following Legendre et al. (2005). For this, the partial canonical correspondence analysis (pCCA; ter Braak, 1988) was applied incorporating the same independent variables used to model the alpha diversity and the limnologic zones as a conditional factor. Legendre & Gallagher (2001) emphasise that CCA should only be applied when it is needed to give high weight to rare species under particular environmental conditions. We then performed the pCCA, since the surrounding agropastoral landscape could affect water quality (Schoumans et al., 2014; Mockler et al., 2017; Nhiwatiwa et al., 2017; Liu et al., 2019; Paula-Filho et al., 2019). Water quality, in turn, can influence the assemblage structure of rotifers (Sládecek, 1983; Saksena, 1987; Jeppesen et al., 2011; Perbiche-Neves et al., 2013; Li & Chen, 2020; Liang et al., 2020) because species richness is negatively affected by nutrient concentration recorded in the study area. In addition, CCA was chosen because it employs unimodal associations (ter Braak & Verdonschot, 1995; Legendre & Legendre, 1998) like those expected for some of the environmental variables, like TN, pH and temperature.
The pCCA was performed without species exclusively recorded in a single sample site and with data transformed using the chi-square distance (Legendre & Gallagher, 2001). Significant relationships of the environmental variables were estimated with ANOVA based on 100,000 permutations. The significance of the variation explained by the three first axes was also estimated by ANOVA with 100,000 replications. Moreover, a variance partitioning analysis was conducted to check the contribution of spatial distances, environmental variables and the surrounding matrix on the beta diversity variation. The adjusted coefficient of determination for pCCA was calculated from permutation processes and, considering that such processes can present some volatility for low number of permutation (Oksanen et al., 2020), the variance partitioning analysis was performed with 1,000,000 permutations (Fig. 2).
Relationships between the species richness (S) and the explanatory variables that compound the best-fitted model (R2 = 0.662; F4,8 = 477.685; P < 0.0001; Tables 1, S1)
The spatial distance was computed using the Principal Coordinates of Neighbourhood Matrix (PCNM). The significance of the relationship between spatial distances and the species sharing among reservoirs was also checked with a pCCA and subsequent ANOVA based on 100,000 permutations. This analytical procedure was also conducted to check the significant influence of the agropastoral landscape on the beta diversity of rotifers. Furthermore, a spatial partitioning and a permutations test were performed to check the spatial independence of the pCCA residuals. Beta diversity was analyzed using functions of the R package vegan version 2.0-8 (Oksanen et al., 2020).
All statistical analyses were performed using the R environment version 4.0.0 (R Core Team, 2020).
Results
Environmental factors
The water temperature varied between 24.53 and 28 °C (mean 25.76 °C; s.d. 0.88 °C), the dissolved oxygen ranged between 1.14 and 7.70 mg l−1 (mean 4.565 mg l−1; s.d. 1.60 mg l−1) and the pH varied from 5.02 to 8.35 (mean 6.496; s.d. 0.82). As for nutrients, the TN concentration recorded in the sampling points varied between 70.5 and 4,273.33 µg l−1 (mean 590.705 µg l−1; s.d. 1,063.237) and the TP concentration ranged from 0.06 to 55.94 µg l−1 (mean 14.419 µg l−1; s.d. 18.317 µg l−1). The pH responded to the TP and TN concentrations, decreasing linearly and non-linearly with increasing of TP and TN, respectively (Adjusted R2 = 0.486; F3,9 = 615.741; P < 0.0001; Table 1; Fig. S1).
Alpha diversity
In total, 112 species of rotifers were collected in 7 reservoirs, with an average of 28.37 species (± 14.23) per reservoir. The species richness (S) estimated by Chao1 for each sample was equal to the sampled species number, and TN concentration plus temperature and pH explain 66.2% of its variation modelled by GAMM with a Gaussian spatial correlation structure (F4,8 = 477.685; P < 0.0001; Tables 1, S1). Species richness decreased with TN concentration and increasing temperature and was higher in reservoirs with neutral pH (Fig. 2).
The Δ+ ranged from 50.182 to 76.923 (mean = 65.129; s.d. = 8.061), so that the lowest Δ+ is based on 11 species, 6 genera, 4 families and 1 order, while the highest Δ+ is represented by 13 species, 11 genera, 9 families, 3 orders and 2 superorders. About 29.8% of the variation of Δ+ was explained by the TP concentration modelled by GAMM with Rational correlation structure (F2,10 = 680.198; P < 0.0001; Tables 1, S1), from a negative relationship (Fig. 3A). We found the prevalence of the species-richest Order Ploima, with a great number of species of Lecane Nitzsch in reservoir with the high nutrient concentrations. On the other hand, species-poor taxa, such as belong to the Order Flosculariaceae and Collothecacea, were absent in the most reservoirs with high nutrient concentrations.
Taxonomic distinctness of Rotifera in reservoirs in an agropastoral region in the southeastern Brazil. A Relationship between average taxonomic distinctness (Δ+) and the total phosphorus (R2 = 0.298; F2,10 = 680.198; P < 0.0001; Tables 1, S1). B Relationship between variation in taxonomic distinctness (Λ+) and dissolved oxygen (R2 = 0.413; F2,10 = 147.974; P < 0.0001; Tables 1, S1)
The Λ+ ranged between 99.556 and 670.014 (mean = 493.286; s.d. = 162.215), with highest Λ+ represented by 36 species, 15 genera, 11 families, 2 orders and 2 suborders, while the lowest Λ+ value was determined based on 12 species of 8 genera, 6 families and a single order. Its variation was best explained by the DO concentration modelled by GAMM with Gaussian correlation structure (F2,10 = 147.974; P < 0.0001; Tables 1, S1), with a positive relationship and an asymptote between 5.8 and 7.7 mg l−1 of DO (Fig. 3B).
Beta diversity
According to the pCCA, beta diversity is influenced by TP, TN, DO and pH (Table 2). The considerable variations observed for these explanatory variables are mainly associated with orange crops (Fig. 4A, B). The temperature showed no significant relationship with the sharing of rotifer fauna (Table 2). The variation partition analysis indicated that 9% of species sharing is explained by the four environmental variables above (F5,10 = 1.753; P < 0.0001), 23% by the relative distance among the sample sites (F6,11 = 2.471; P < 0.0001) and 5% by the agropastoral landscape (F2,15 = 2.136; P < 0.0001) (Fig. 4C).
Beta diversity analysed by partial Canonical Correspondence Analysis (pCCA; see Table 2 for details); A relationships among the sampling sites and environmental vectors, B relationships between species and environmental vectors in the agropastoral landscape (see Table S2 for species codes), and C variation Partition based on CCA and 1,000,000 replications. TP total phosphorus, logTN total nitrogen in logarithmic scale, DO dissolved oxygen, ResAPel Pelagic sample of the reservoir A, ResALitM Littoral sample of the reservoir A bordered by surrounding matrix, ResALitF Littoral sample of the reservoir A bordered by forest, ResBPel Pelagic sample of the reservoir B, ResBLit Littoral sample of the reservoir B bordered by surrounding matrix, ResCPel Pelagic sample of the reservoir C, ResCLitM Littoral sample of the reservoir C bordered by surrounding matrix, ResCLitF Littoral sample of the reservoir C bordered by forest, ResDPel Pelagic sample of the reservoir D, ResDLitM Littoral sample of the reservoir D bordered by surrounding matrix, ResEPel Pelagic sample of the reservoir E, ResELitM Littoral sample of the reservoir E bordered by surrounding matrix, ResFPel Pelagic sample of the reservoir F, ResFLitM Littoral sample of the reservoir F bordered by surrounding matrix, ResGPel Pelagic sample of the reservoir G, ResGLitM Littoral sample of the reservoir G bordered by surrounding matrix, ResHPel Pelagic sample of the reservoir H, ResHLitM Littoral sample of the reservoir H bordered by surrounding matrix. Yellow polygon orange crop matrix, blue polygon sugarcane crop matrix, green polygon pasture matrix
The pCCA indicated two distinct groups of reservoirs, one surrounded by a predominantly citrus agricultural landscape and the other surrounded by a pasture and sugarcane agricultural landscape (Fig. 4A, B). These groups are represented along the first pCCA axis, which showed 32.99% of the beta-diversity variation representing the eutrophication gradient. On the other hand, the rotifer fauna in the reservoirs bordered by pasture and cane sugar were reflected in the second pCCA axis (Fig. 4A, B). In the reservoirs surrounded by the citrus matrix, six species are at the extreme of the first pCCA axis: Anuraeopsis navicula Rousselet, Brachionus havanaensis Rousselet, Kellicottia bostoniensis (Rousselet), Keratella americana Carlin, Keratella tecta (Gosse) and Trichocerca pusilla (Jennings). Our findings suggest those species are commonly found in eutrophic environments. On the other hand, three species, Brachionus dolabra Harring, Filinia pejleri Hutchinson and Microdon clavus Ehrbg, are located at the opposite extreme of the first axis (Fig. 4B), in those reservoirs surrounded by the sugarcane matrix and characterised by relatively low nutrient concentrations.
Discussion
Understanding the interference of agricultural land management with the local and regional aquatic communities is essential to maintaining biodiversity and ecosystem services (Brauman et al., 2007; Hoffmann et al., 2019). This study indicates that both local (alpha) and regional (beta) rotifer diversities are influenced by nutrients that are linked primarily to land management. The intensification of agricultural uses affected the quality of surface waters and modified the biogeochemical cycles of nutrients, altering ecohydrological processes in areas where fertilizer use was intensified (Schoumans et al., 2014; Mockler et al., 2017; Nhiwatiwa et al., 2017; Liu et al., 2019; Paula-Filho et al., 2019).
Castilho-Noll et al. (2012) reported that most of the reservoirs in the rural areas of São Paulo suffer from eutrophication, especially in areas of excessive fertilizer use. Those fertilizers are a source of nutrient loading, primarily nitrogen and phosphorus. Filoso et al. (2003) pointed out that TN export from citrus and sugar cane crops is among the primary sources contributing to the concentration of this nutrient in the reservoirs of southeastern Brazil. However, the highest values of TP and TN were recorded in reservoirs surrounded by citrus crops, despite this type plantation requires, respectively, lower and similar rates of phosphorus and nitrogen, respectively, compared to those required by sugar cane crops.
The impressive high concentrations of TP and TN in reservoirs associate with citrus crops suggested this type of plantation is over-fertilized. In contrast, reservoirs surrounded by sugar-cane and pasture areas showed relatively low concentrations of TP and greater faunistic similarities among them than with citrus areas (Fig. 4A, B). Zeni et al. (2017) also reported no taxonomic and functional differences in fish fauna of basins with pasture and sugar-cane landscapes. In the last two decades, the fertilizer use in Brazil has increased much more than the average rate of cropland expansion (Withers et al., 2018), and intensive agricultural production consequently has resulted in over-fertilization (Francia-Martínez et al., 2006; Oelofse et al., 2010).
Local characteristics, spatial distance and the agropastoral landscape can influence the regional structure of the tropical Rotifera assemblage (Fig. 4C), such as found by Kuczyńska-Kippen & Basińka (2014) for a European region with intense anthropogenic interference. The TP concentration did not influence the species richness but negatively affected the Δ+, selecting closer related species from the regional species pool. In contrast, TN adversely affected the species richness, along with temperature and extremes in pH (Fig. 2). Vilmi et al. (2016) reported different relationships between Δ+ and nutrients for different taxa and emphasized that values of this index may be dependent on the organismal group.
Both TN and TP are the determinants of eutrophication (Conley et al., 2009). In general, phosphorous is the main limiting factor for freshwater systems (Conley et al., 2009; Schoumans et al., 2014). Stamou et al. (2017) and Jiang et al. (1994) also reported negative relationships between freshwater eutrophication and Δ+ of rotifers and fish, respectively. The negative relationship between Δ+ and disturbed habitats has been reported as result of the imbalanced loss of species-poor higher taxa (Clarke & Warwick, 2001; Stamou et al., 2017; Jiang et al., 1994). As also reported by Kuczynska-Kippen & Pronin (2018) and Smaoune et al. (2020), we found prevalence of the species-richest Order Ploima, mainly species of Brachionus, Keratella, Trichocerca and Lecane Nitzsch in reservoir with the high nutrient concentrations. In contrast, the species-poor taxa, such as Flosculariaceae and Collothecacea, are more frequent in oligotrophic reservoirs (Wærvågen & Andersen, 2018).
The eutrophication caused by high levels of N and P results in dominance of some phytoplanktonic species (Ho et al., 2019; GrIffith & Gobler, 2020), mainly of Cyanobacteria (Rigosi et al., 2014; Josué et al., 2019; Amorim & Moura, 2020). Usually, Cyanobacteria blooms interrupt the grazing of zooplankton on phytoplankton (Moustaka-Gouni & Sommer, 2020) and block the energy transfer from producers to consumers (Filstrup et al., 2014; Tian et al., 2017). Consequently, this situation results in a bottom-up effect and a decreasing in the diversity of freshwater zooplankton (Amorim et al., 2020; Moustaka-Gouni & Sommer, 2020). Our results show that this effect is more accentuated in reservoirs surrounded by citrus crops. Top-down and bottom-up biotic effects are considered one of the most critical regulating factors of the zooplankton community structure (Symons et al., 2014; Du et al., 2015; Rosińska et al., 2019; Li & Chen, 2020). Communities of rotifers are more affected by bottom-up than top-down effects (Yoshida et al., 2003; Sinistro, 2010; Rosińska et al., 2019).
Nhiwatiwa et al. (2017) pointed out that nutrient runoff from agricultural areas can also change pH levels in water systems, what would explain the relationship found between pH and TP plus TN in the present study (Table 1). Neutral pH favours the occurrence of most zooplankton species so that the highest species richness of zooplankton is found in waters with a pH between 6 and 8.5 (Kalff, 2002). This range in pH levels was corroborated in our study. According to Cremona et al. (2020), pH can have direct and indirect effects on zooplankton. The direct effects are the physiological changes in the organisms, and the indirect effects are on those organisms used as food by the zooplankton, such as algae. The variation in pH can cause physiological stress on diatoms and also bias other aspects of the chemistry of the aquatic ecosystem (Dalu et al., 2014). Consequently, this physiological stress on phytoplankton influences the rotifer assemblage via trophic cascade by decreasing their food resources (Ji et al., 2013).
The low or non-sensitiveness of Λ+ to environmental degradation has been reported by other studies (Bevilacqua et al., 2011; Alahuhta et al., 2017; Stamou et al., 2017; Jiang et al., 1994). The relationship found between Λ+ and DO (Fig. 3B) indicates that low DO concentrations support evolutionarily closer species, because high values of Λ+ represent great taxonomic evenness of the higher levels in the hierarchical classification (Clarke & Warwick, 2001). Karpowicz et al. (2020) found that a large number of rotifer species are tolerant of oxygen deficits, which would confirm the absence of a relationship between species richness and dissolved oxygen under these conditions. Supposedly species tolerant to low DO are more related among them than to species that prefer high DO concentrations. In this way, high Λ+ values can represent direct physiological diversity of rotifers or indirectly indicate high diversity feeding traits, once the rotifer feeding habits are related to their life history (Wallace & Snell, 2001). A prerequisite for the coexistence of different taxa is selectivity for different food types (Walz, 1997). Obertegger & Flaim (2018) found that feeding traits explain the extensive variety of rotifers in temperate lakes. Rotifers can be bacterivorous, detritivores, phytoplanktivorous or predatory (Walz, 1997; Wallace et al., 2015; Oh et al., 2017; Rosińska et al., 2019).
Although the highest Λ+ is a reservoir surrounded by citrus crop, most of the sites with high Λ+ is in reservoir surrounded mainly by pasture lands. In a general way, high Λ+ values were determined by the presence of rotifers of the Superorder Gnesiotrocha, mainly of the Order Flosculariaceae. Most reservoir surrounded by citrus crops showed from low to medium Λ+ values. However, the variation in taxonomic distinctness presented the strongest spatial correlation of the three alpha diversity measures (Table 1), indicating that variation in the rotifer fauna is conditioned by geographical distance and dispersion events among water bodies.
Several studies have highlighted the need to adopt mitigating measures to minimize the loss of nutrients in the aquatic environment (Cherry et al., 2008; Withers et al., 2011; Schoumans et al., 2014; Liu et al., 2019) and ensure diverse ecosystem services (Swinton et al., 2007; Hoffmann et al., 2019). Schoumans et al. (2014) and Liu et al. (2019) concluded that controlling agricultural drainage is the most critical action to avoid nutrient runoff and accumulation and protect water quality. However, in the past two years, the Brazilian government released hundreds of pesticides, demonstrating a disregard for that issue. If there were no concerns over the release of highly toxic pesticides for use in Brazilian agriculture, there are unlikely to be concerns about controlling nutrient loss unless the over-application of pesticides and eutrophication of reservoirs results in a financial loss for growers and operators. Based on Withers et al. (2018), nearly 70% of the money spent on fertilisers could be saved with improved agricultural practices.
In conclusion, our study indicated that, when subjected to intensive agricultural practices, the rotifer is negatively affected by nutrients at both local and regional scales. We found that TN and TP concentrations affected the structure of the rotifer assemblage in different ways so that their combined effect can magnify the loss of rotifer diversity. This effect applies especially to reservoirs adjacent to land managed for citrus crops. Acting as herbivores or consumers of debris, rotifers form an essential link in aquatic food chains, connecting the sources of organic matter with the upper tiers of the aquatic food webs. The loss of species diversity in rotifers is likely to result in a cascade effect causing damage to the upper trophic levels. Considering such problems, we believe this study can draw attention to and raise awareness among Brazilian authorities and farmers about the influence of misguided agricultural policies, primarily those associated with citrus plantations.
References
Alahuhta, J., M. Toivanen, J. Hjort, F. Ecke, L. B. Johnson, L. Sass & J. Heino, 2017. Species richness and taxonomic distinctness of lake macrophytes along environmental gradients in two continents. Freshwater Biology 62: 1194-1206.
Alsharif, K. A. & G. Fouad, 2012. Lake performance differences in response to land use and water quality: data envelopment analysis. Lake and Reservoir Management 28: 130-141.
Amorim, C. A. & A. N. Moura, 2020. Ecological impacts of freshwater algal blooms on water quality, plankton biodiversity, structure, and ecosystem functioning. Science of the Total Environment. https://doi.org/https://doi.org/10.1016/j.scitotenv.2020.143605
Bashir, I., Lone, F. A., Bhat, R. A., Mir, S. A., Dar, Z. A. & S. A. Dar, 2020. Concerns and threats of contamination on aquatic ecosystems. In Hakeem, K., Bhat, R. & H. Quadri (eds), Bioremediation and Biotechnology. Springer, Cham, pp 1-26.
Bevilacqua, S., S. Fraschetti, L. Musco, G. Guarnieri & A. Terlizzi, 2011. Low sensitiveness of taxonomic distinctness indices to human impacts: evidences across marine benthic organisms and habitat types. Ecological Indicators 11: 448-455.
Brauman, K. A., G. C. Daily, T. K. Duarte & H. A. Mooney, 2007. The nature and value of ecosystem services: an overview highlighting hydrologic services. Annual Review of Environment and Resources 32: 67-98.
Brito, S. L., P. M. Maia-Barbosa & R. M. Pinto-Coelho, 2011. Zooplankton as an indicator of trophic conditions in two large reservoirs in Brazil. Lakes and Reservoirs: Research and Management 16: 253-264.
Bucci, M. M. H. S., F. E. F. Delgado & L. F. C. Oliveira, 2015. Water quality and trophic state of a tropical urban reservoir for drinking water supply (Juiz de Fora, Brazil). Lake and Reservoir Management 31: 134-144.
Castilho-Noll, M. S. M., C. F. Câmara, M. F. Chicone, E. H. Shibata & L. R. Stephan, 2012. Copepods (Crustacea, Maxillopoda) from shallow reservoirs. Acta Limnologica Brasiliensia, 24: 149-159.
Cherry, K. A., M. Shpherd, P. J. A. Withers, & S. J. Mooney, 2008. Assessing the effectiveness of actions to mitigate nutrient loss from agriculture: a review of methods. A review of methods. Science of the Total Environment 406: 1-23.
Clarke, K. R. & R. M. Warwick, 1998. A taxonomic distinctness index and its statistical properties. Journal of Applied Ecology 35: 523-531.
Clarke, K. R. & R. M. Warwick, 2001. A further biodiversity index applicable to species lists: variation in taxonomic distinctness. Marine Ecology Progress Series 216: 265-278.
Conley, D. J., H. W. Peerl, R. W. Howarth, D. F. Boesch, S. P. Seitzinger, K. E. Havens, C. Lancelot & G. E. Likens, 2009. Controlling eutrophication: nitrogen and phosphorus. Science 323: 1014-1015.
Cremona, F., H. Agasild, J. Haberman, P. Zingel, P. Nõges, T. Nõges, & A. Laas, 2020. How warming and other stressors affect zooplankton abundance, biomass and community composition in shallow eutrophic lakes. Climatic Change 159: 565-580.
Dalu, T., P. W. Froneman, L. D. Chari & N. B. Richoux, 2014. Colonization and community structure of benthic diatoms on artificial substrates following a major flood event: a case of the Kowie River (Eastern Cape, South Africa). Water SA 40: 471-480.
Du, X., E. García-Berthou, Q. Wang, J. Liu, T. Zhang & Z. Li, 2015. Analyzing the importance of top-down and bottom-up controls in food webs of Chinese lakes through structural equation modeling. Aquatic Ecology 49:199-210.
Filoso, S., L. A. Martinelli, M. R. Williams, L. B. Lara, A. Krusche, M. V. Ballester, R. Victoria & P. B. Camargo, 2003. Land use and nitrogen export in the Piracicaba River Basin, Southeast Brazil. Biogeochemistry 65: 275-294.
Filstrup, C. T., H. Hillebrand, A. J. Heathcote, W. S. Harpole & J. A. Downing, 2014. Cyanobacteria dominance influences resource use efficiency and community turnover in phytoplankton and zooplankton communities. Ecology Letters 17: 464-474.
Foley, J. A., R. DeFries, G. P. Asner, C. Barford, G. Bonan, S. R. Carpenter, F. S. Chapin, M. T. Coe, G. C. Daily, H. K. Gribbs, J. H. Helkowski, T. Holloway, E. A. Horward, C. J. Kucharik, C. Monfreda, J. A. Patz, L. C. Prentice, N. Ramankutty & P. K. Snyder, 2005. Global consequences of land use. Science 309: 570-574.
Food and Agriculture Organization of the United Nations (FAO), 2004. Fertilizer Use by Crop in Brazil. Land and Plant Nutrition Management Service and Land and Water Development Division [available on internet at www.fao.org/3/y5376e/y5376e00.htm]. Accessed 2 February 2021.
Francia-Martínez, J. R., V. H. D. Zuazo & A. M. Raya, 2006. Environmental impact from mountainous olive orchards under different soil-management systems (SE Spain). Science of the Total Environment 358: 46-60.
Giri, S. & Z. Qiu, 2016. Understanding the relationship of land uses and water quality in Twenty First Century: a review. Journal of Environmental Management 173: 41-48.
Gopko, M. & I. V. Telesh, 2013. Estuarine trophic state assessment: new plankton index based on morphology of Keratella rotifers. Estuarine, Coastal and Shelf Science 130: 222-230.
Griffith, A. W. & C. J. Glober, 2020. Harmful algal blooms: a climate change costressor in marine and freshwater ecosystems. Harmful Algae 91: 101590.
Ho, J. C., A. M. Michalak & N. Pahlevan, 2019. Widespread global increase in intense lake phytoplankton blooms since the 1980s. Nature 574: 667-670.
Hoffmann, H., S. Schomers, C. Meyer, K. Sander, V. Hickey & A. Feuerbacher, 2019. Agriculture and ecosystem services. In Ferranti, P., E. M. Berry & J. R. Anderson (eds), Encyclopedia of Food Security and Sustainability: Sustainable Food Systems and Agriculture, Vol 3. Elsevier, Amsterdam: pp 9-13.
Hritonenko, N. & Y. Yatsenko. 2013. Mathematical Modelling in Economics and the Environment, 2nd edn. Springer, New York.
Huang, J., C. Xu, B. G. Ridoutt, X. Wang & P. Ren, 2017. Nitrogen and phosphorus losses and eutrophication potential associated with fertilizer application to cropland in China. Journal of Cleaner Production 159: 171-179.
Isbell, F., P. B. Reich, D. Tilman, S. E. Hobbie, S. Polasky & S. Binder, 2013. Nutrient enrichment, biodiversity loss, and consequent declines in ecosystem productivity. Proceedings of the National Academy of Sciences of USA 110: 11911-11916.
Ismail, A. H. & A. A. Adnan, 2016. Zooplankton composition and abundance as indicators of eutrophication in two small man-made lakes. Tropical Life Sciences Research 27: 31-38.
Janse, J. H., Kuiper, J. J., Weijers, M. J., Westerbeek, E. P., Jeuken, M. H. J. L., Bakkenes, M., Alkemade, R., Mooij, W. M. & J. T. A. Verhoeven, 2015. GLOBIO-Aquatic, a global model of human impact on the biodiversity of inland aquatic ecosystems. Environmental Science and Policy 48: 99-114.
Jeppesen, E., P. Nöges, T. Davidson, J. Haberman, T. Nöges, K. Blank, T. L. Lauridsen, M. Søndergaard, C. Sayer, R. Laugaste, L. S. Johansson, R. Bjerring & S. L. Amsinck, 2011. Zooplankton as indicators in lakes: a scientific-based plea including zooplankton in the ecological quality assessment of lakes according to the European Water Framework Directive (WFD). Hydrobiologia 676: 279-297.
Ji, G., X. Wang & L. Wang, 2013. Planktonic rotifers in a subtropical shallow lake: succession, relationship to environmental factors, and use as bioindicators. The Scientific World Journal 2013: 702942.
Jiang, X., B. Pan, Z. Sun, L. Cao & Y. Lu., 1994. Application of taxonomic distinctness indices of fish assemblages for assessing effects of river-lake disconnection and eutrophication in floodplain lakes. Ecological Indicators 110: 105955.
Josué, I. I. P., S. J. Cardoso, M. Miranda, M. Mucci, K. A. Ger, F. Roland & M. M. Marinho, 2019. Cyanobacteria dominance drives zooplankton functional dispersion. Hydrobiologia 831: 149-161.
Kalff, J., 2002. Limnology: Inland Water Ecosystems. Prentice Hall, Upper Saddle River.
Karpowicz, M., J. Ejsmont-Karabin, J. Koslowska, I. Feniova & A. R. Dzialowski, 2020. Zooplankton community responses to oxygen stress. Water 12: 706.
Kennedy, R. H. & W. W. Walker, 1990. Reservoir nutrient dynamics. In Thornton, K. W., B. L. Kimmel & F. E. Payne (eds), Reservoir Limnology: Ecological Perspectives. Wiley-Interscience Publication, New York: pp 109-132.
Kuczyńska-Kippen, N. & A. Basińska, 2014. Habitat as the most important influencing factor for the rotifer community structure at landscape level. International Review of Hydrobiology 99: 58-64.
Kuczyńska-Kippen, N. & M. Pronin, 2018. Diversity and zooplankton species associated with certain hydroperiods and fish state in field ponds. Ecological Indicators 90: 171-178.
Lanna, A. E. L. & B. Braga, 2015. Hidroeconomia, In Braga, B., J. G. Tundisi, T. Matsumura-Tundisi & V. S. T. Ciminelli (eds), Águas doces no Brasil: Capital ecológico, uso e conservação, Escrituras Editora, São Paulo: pp 607-638.
Legendre, P., D. Borcard & P. R. Peres-Neto, 2005. Analyzing beta diversity: partitioning the spatial variation of community composition data. Ecological Monographs 75: 435-450.
Legendre, P. & E. D. Gallagher, 2001. Ecologically meaningful transformations for ordination of species data. Oecologia 129: 271-180.
Legendre, P. & L. Legendre, 1998. Numerical Ecology. Elsevier, Amsterdam.
Leibold, M. A., 1999. Biodiversity and nutrient enrichment in pond plankton communities. Evolutionary Ecology Research 1: 73-95.
Li, Y. & F. Chen, 2020. Are zooplankton useful indicators of water quality in subtropical lakes with high human impacts? Ecological Indicators 113: 106167.
Liang, D., Q. Wang, N. Wei, C. Tang, X. Sun & Y. Yang, 2020. Biological indicators of ecological quality in typical urban river–lake ecosystems: the planktonic rotifer community and its response to environmental factors. Ecological Indicators 112: 106127.
Liu, X., G. Zhang, G. Sun, Y. Wu & Y. Chen, 2019. Assessment of lake water quality and eutrophication risk in an agricultural irrigation area: a case study of the Chagan Lake in northeast China. Water 11: 2380.
Lucena, L. C. A., T. X. Melo & E. S. F. Medeiros, 2015. Zooplankton community of Parnaíba River, northeastern Brazil. Acta Limnologica Brasiliensia 27: 118-129.
Mackereth, F. J. H., J. Heron & J. F. Talling, 1978. Water Analysis: Some Revised Methods for Limnologists. Freshwater Biological Association, Windermere.
Major, Y., D. Kifle, G. H. Niedrist & R. Sommaruga, 2017. An isotopic analysis of the phytoplankton–zooplankton link in a highly eutrophic tropical reservoir dominated by cyanobacteria. Journal of Plankton Research 39: 220-231.
Mantovano, T., R. L. Arrieira, L. T. F. Schwind, C. C. Bonecker & F. A. Lansac-Tôha. 2015. Rotifer community structure along a stretch under the influence of dams in the Upper Paraná River Floodplain. Acta Scientiarum 37: 281-289.
Matsumura-Tundisi, T. & J. G. Tundisi, 2005. Plankton richness in a eutrophic reservoir (Barra Bonita Reservoir, SP, Brazil). Hydrobiologia 542: 367-378.
May, L. & M. O’Hare, 2005. Changes in rotifer species composition and abundance along a trophic gradient in Loch Lomond, Scotland, UK. Hydrobiologia 546: 397-404.
McCarthy, M. J., P. J. Lavrentyev, L. Yang, L. Zhang, Y. Chen, Q. Boqiang & S. G. Wayne, 2007. Nitrogen dynamics and microbial food web structure during a summer cyanobacterial bloom in a subtropical, shallow, well-mixed, eutrophic lake (Lake Taihu, China). Hydrobiologia 581: 195-207.
Mioduszewski, W., 2012. Small water reservoirs – their function and construction. Journal of Water and Land Development 17: 45-52.
Mockler, E. M., J. Deakin, M. Archbold, L. Gill, D. Daly & M. Bruen, 2017. Sources of nitrogen and phosphorus emissions to Irish rivers and coastal waters: estimates from a nutrient load apportionment framework. Science of the Total Environment 601–602: 326-339.
Mokany, K., T. D. Harwood, J. M. Overton, G. M. Barker & S. Ferrier, 2011. Combining α- and β-diversity model to fill gaps in our knowledge of biodiversity. Ecology Letters 14: 1043-1051.
Moustaka-Gouni, M. & U. Sommer, 2020. Effects of harmful blooms of large-sized and colonial Cyanobacteria on aquatic food webs. Water 12: 1587.
Naiman, R. J., J. J. Magnuson, D. M. Mcknight & J. A. Stanford, 1995. The Freshwater Imperative. Island Press, Washington, DC.
Nhiwatiwa T., T. Dalu & L. Brendonck, 2017. Impact of irrigation based sugarcane cultivation on the Chiredzi and Runde Rivers quality, Zimbabwe. Science of the Total Environment 587–588: 316-325.
Norton, D. A., N. Reid & L. Young, 2013. Ultimate drivers of native biodiversity change in agricultural systems. F1000Research 2: 214.
Obertegger, U. & G. Flaim, 2018. Taxonomic and functional diversity of rotifers, what do they tell us about community assembly? Hydrobiologia 823: 79-91.
Oelofse, M., H. Høgh-Jensen, L. S. Abreu, G. F. Almeida, A. El-Araby, Q. Y. Hui & A. Neergaard, 2010. A comparative study of farm nutrient budgets and nutrient flows of certified organic and non-organic farms in China, Brazil and Egypt. Nutrient Cycling in Agroecosystems 87: 455-470.
Oh, H. -J., H. -G. Jeong, G. -S. Nam, Y. Oda, W. Dai, E. -H. Lee, D. Kong, S. -J. Hwang & K. -H. Chang, 2017. Comparison of taxon-based and trophi-based response patterns of rotifer community to water quality: applicability of the rotifer functional group as an indicator of water quality. Animal Cells and Systems 21: 133-140.
Oksanen, J., F. G. Blanchet, R. Kindt, P. Legendre, P. R. Minchin, R. B. O’Hara, G. L. Simpson, P. Solymos, M. H. H. Stevens, E. Szoecs & H. Wagner, 2020. Package Vegan Version 2.5-6: Community Ecology Package [available on internet at https://cran.r-project.org/web/packages/vegan/vegan.pdf]. Accessed 2 February 2020.
Paula-Filho, F. J., A. D. S. Sampaio, J. M. C. Menezes, C. T. F. Costa & M. O. Santiago, 2019. Land uses, nitrogen and phosphorus estimated fluxes in a Brazilian semiarid watershed. Journal of Arid Environments 163: 41-49.
Peel, M. C., B. L. Finlayson & T. A. McMahon, 2007. Updated world map of the Köppen–Geiger climate classification. Hydrology and Earth System Sciences 11: 1633-1644.
Perbiche-Neves, G., C. Fileto, J. Laço-Portinho, A. Troguer & M. Serafim-Junior, 2013. Relations among planktonic rotifers, cyclopoid copepods, and water quality in two Brazilian reservoirs. Latin American Journal of Aquatic Research 41: 138-149.
Pinto-Coelho, R. M., 1998. Effects of eutrophication on seasonal patterns of mesozooplankton in a tropical reservoir: a 4-year study in Pampulha Lake, Brazil. Freshwater Biology 40: 159-173.
Postel, S. & S. R. Carpenter, 1997. Freshwater ecosystem services, in: Daily, G. (Ed), Nature’s Services: Societal Dependence on Natural Ecosystems. Island Press, Washington, DC, pp 195-214.
Pradhan, P. R., R. N. Pandey, U. K. Behera, A. Swarup, S. C. Datta. & B. S. Dwivedi, 2011. Tillage and crop residue management practices on crop productivity, phosphorus uptake and forms in wheat (Triticum aestivum)-based cropping systems. The Indian Journal of Agricultural Sciences 81: 1168-1173.
R Core Team, 2020. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna [available on internet at https://www.R-project.org/].
Rhodes, A. L., R. M. Newton & A. Pufall, 2001. Influences of land use on water quality of a diverse New England Watershed. Environment Science Technology 35: 3640-3645.
Rico-Martínez, R., M. A. Arzate-Cárdenas, D. Robles-Vargas, I. A. Pérez-Legaspi, A.-F. Jesús & G. E. Santos-Medrano, 2016. Rotifers as models in toxicity screening of chemicals and environmental samples. In Larramendy, M. & S. Soloneski (eds), Invertebrates: Experimental Models in Toxicity Screening. IntechOpen, London, pp 57-99.
Rigosi, A., C. C. Carey, B. W. Ibelings & J. D. Brookes, 2014. The interaction between climate warming and eutrophication to promote cyanobacteria is dependent on trophic state and varies among taxa. Limnology and Oceanography 59: 99-114.
Rockström, J., J. Williams, G. Daily, A. Noble, N. Matthews, L. Gordon, H. Wtterstrand, F. DeClerck, M. Shah, P. Steduto, C. de Fraiture, N. Hatibu, O. Unver, J. Bird, L. Sibanda & J. Smith, 2017. Sustainable intensification of agriculture for human prosperity and global sustainability. Ambio 46: 4-17.
Rosińska, J., W. Romanowicz-Brzozowska, A. Kozak & R. Goldyn, 2019. Zooplankton changes during bottom-up and top-down control due to sustainable restoration in a shallow urban lake. Environmental Science and Pollution Research 26: 19575-19587.
Roth, R. A., 2009. Freshwater Aquatic Biomes. Greenwood Press, Westport.
Saksena, D. N., 1987. Rotifers as indicators of water quality. Acta Hydrochimica et Hydrobiologica 15: 481-485.
Sampaio, E. V., O. Rocha, T. Matsumura-Tundisi & J. G. Tundisi, 2002. Composition and abundance of zooplankton in the limnetic zone of seven reservoirs of the Paranapanema River, Brazil. Brazilian Journal of Biology 62: 525-545.
Scholz, M., Å. Hedmark & W. Hartley, 2012. Recent advances in sustainable multifunctional land and urban management in Europe: a review. Journal of Environmental Planning and Management 55: 833-854.
Schoumans, O. F., W. J. Chardon, M. E. Bechmann, C. Gascuel-Odoux, G. Hofman, B. Kronvang, G. H. Rubaek, B. Ulén & J.-M. Dorioz, 2014. Mitigation options to reduce phosphorus losses from the agricultural sector and improve surface water quality: a review. Science of the Total Environment 468–469: 1255-1266.
Sinistro, R., 2010. Top-down and bottom-up regulation of planktonic communities in a warm temperate wetland. Journal of Plankton Research. 32: 209-220.
Sládecek, V., 1983. Rotifer as indicators of water quality. Hydrobiologia 100: 169-201.
Smaoune, G., D. Bouchelouche, A. Taleb & A. Arab, 2020. Evaluation of the trophic status in three reservoirs in Algeria (northwest) using physicochemical analysis and rotifers structure. Environmental Science and Pollution Research. https://doi.org/https://doi.org/10.1007/s11356-020-11233-w
Soininen, J., M. Kokocinski, S. Estlander, J. Kotanen & J. Heino, 2007. Neutrality, niches, and determinants of plankton metacommunity structure across boreal wetland ponds. Ecoscience 14: 146-154.
Sperling, E., 2012. Hydropower in Brazil: overview of positive and negative environmental aspects. Energy Procedia 18: 110-118.
Stamou, G., C. Polyzou, A. Karagianni & E. Michaloudi, 2017. Taxonomic distinctness índices for discriminating patterns in freshwater rotifer assemblages. Hydrobiologia 796: 319-331.
Strickland, J. D. H. & T. R. A. Parsons, 1972. A Practical Handbook of Seawater Analysis, Bulletin 167. Fisheries Research Board of Canada, Ottawa.
Swinton, S. M., F. Lupi, G. P. Robertson & S. K. Hamilton, 2007. Ecosystem services and agriculture: cultivating agricultural ecosystems for diverse benefits. Ecological Economics 64: 245-252.
Symons, C. C., M. T. Pedruski, S. E. Arnott & J. N. Sweetman, 2014. Zooplankton community composition in subarctic lakes and ponds in Wapusk National Park, Canada. Arctic, Antarctic, and Alpine Research 46: 159-190.
ter Braak, C. J. F., 1988. Partial canonical correspondence analysis. In Bock, H. H. (ed), Classification and Related Methods of Data Analysis. Elsevier, Amsterdam, pp 551-558.
ter Braak, C. J. F. & P. F. M. Verdonschot, 1995. Canonical correspondence analysis and related multivariate methods in aquatic ecology. Aquatic Sciences 57: 255-289.
Tian, W., H. Zhang, J. Zhang, L. Zhao, M. Miao, H. Huang, 2017. Biodiversity effects on resource use efficiency and community turnover of plankton in Lake Nansihu, China. Environmental Science and Pollution Research 24: 11279-11288.
Tilman, D., J. Fargione, B. Wolff, C. D’Antonio, A. Dobson, R. Howarth, D. Schindler, W. H. Schlesinger, D. Simberloff & D. Swackhamer, 2001. Forecasting agriculturally driven global environmental change. Science 292: 281-284.
Tundisi, J. G., T. Matsumura-Tundisi & O. Rocha, 2015. Ecossistemas de águas interiores. In Braga, B., J. G. Tundisi, T. Matsumura-Tundisi & V. S. T. Ciminelli (eds), Águas doces no Brasil: Capital ecológico, uso e conservação, 4th edn. Escrituras Editora, São Paulo.
Turcotte, M. M., H. Araki, D. S. Karp, K. Poveda & S. R. Whitehead, 2017. The ecoevolutionary impacts of domestication and agricultural practices on wild species. Philosophical Transactions of the Royal Society B: Biological Sciences 372: 20160033.
Vilmi, A., S. M. Karjalainen, M. Kuoppala, K. T. Tolonen & J. Heino, 2016. Taxonomic distinctness along nutrient gradients: more diverse, less diverse or not different from random? Ecological Indicators 61: 1033-1041.
Wærvågen, S. B. & T. Andersen, 2018. Seasonal quantitative dynamics and ecology of pelagic rotifers in an acidified boreal lake. Journal of Limnology 77: 147-163.
Wallace, R. L. & T. W. Snell, 2001. Phylum Rotifera. In Thorp, J. P. & A. P. Covich (eds), Ecology and Classification of North American Freshwater Invertebrates, 2nd edn. Academic, San Diego, pp 195-254.
Wallace, R. L., T. W. Snell, C. Ricci & T. Nogrady, 2006. Rotifera: biology, ecology and systematics. In Seger, H. (ed), Guides to the Identification of the Micro-invertebrates of the Continental Waters of the World, Vol. 23, 2nd edn. Backhuys Publishers, Leiden: 1–299.
Wallace, R. L., T. W. Snell & H. A. Smith, 2015. Phylum Rotifera. In Thorp, J. H. & D. C. Rogers (eds) Thorp and Covich’s Freshwater Invertebrates: Ecology and General Biology, vol I. Elsevier, Amsterdam, pp 225-271.
Walz, N., 1997. Rotifer life history strategies and evolution in freshwater plankton communities. In Streit, B., T. Städler & C. M. Lively (eds) Evolutionary Ecology of Freshwater Animals: Concepts and Case Studies. Springer, Basel, pp 119-149.
Warwick, R. M. & K. R. Clarke, 1998. Taxonomic distinctness and environmental assessment. Journal of Applied Ecology 35: 532-543.
Wen, X. -L., Y. -L. Xi, F. -P. Qian, G. Zhang & X. -L. Xiang, 2011. Comparative analysis of rotifer community structure in five subtropical shallow lakes in east China: role of physical and chemical conditions. Hydrobiologia 661: 303-316.
Winton, R. S., E. Calamita & B. Wehrli, 2019. Reviews and syntheses: dams, water quality and tropical reservoir stratification. Biogeosciences. 16, 1657-1671.
Withers, P. J. A., H. P. Jarvie & C. Stoate, 2011. Quantifying the impact of septic tank systems on eutrophication risk in rural headwaters. Environmental International 37: 644-653.
Withers, J. P. A., M. Rodrigues, A. Soltangheisi, T. S. de Carvalho, L. R. G. Guilherme, V. M. Benites, L. C. Gatiboni, D. M. G. Sousa, R. S. Nunes, C. A. Rosolem, F. D. Andreote, A. Oliveira-Junior, E. L. M. Coutinho & P. S. Pavinato, 2018. Transitions to sustainable management of phosphorus in Brazilian agriculture. Scientific Reports 8: 2537.
Wood, S., 2020. Package mgcv: Mixed GAM computation vehicle with automatic smoothness estimation [available on internet at https://cran.r-project.org/web/packages/mgcv/mgcv.pdf]. Accessed 2 February 2021.
Yoshida, T., J. Urabe & J. J. Elser, 2003. Assessment of ‘top-down’ and ‘bottom-up’ forces as determinants of rotifer distribution among lakes in Ontario, Canada. Ecological Research 18: 639-650.
Zeni, J. O., D. J. Hoeinghaus & L. Casatti, 2017. Effects of pasture conversion to sugarcane for biofuel production on stream fish assemblages in tropical agroecosystems. Freshwater Biology 62: 2026-2038.
Zhang, T., J. Ni & D. Xie, 2015. Severe situation of rural nonpoint source pollution and efficient utilization of agricultural wastes in the Three Gorges Reservoir Area. Environ. Environmental Science and Pollution Research 22: 16453-1462.
Acknowledgements
We are grateful to Dr. R. Henry and J. P. Maiorano for their assistance with sampling. This study was supported by the São Paulo Research Foundation (FAPESP: process numbers: 2004/04820-3 and 2007/05134-4). E. F. dos Santos was supported by PNPD-CAPES Fellowship for Graduate Program in Animal Biology at the IBILCE-UNESP 2018-2020.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Bernadette Pinel-Alloul.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
dos Santos, E.F., Abra, J. & Castilho-Noll, M.S.M. Does land use influence the local and regional structure of the rotifer assemblage?. Hydrobiologia 848, 1059–1072 (2021). https://doi.org/10.1007/s10750-020-04513-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-020-04513-6