Abstract
Fish respond to predation threat by size/cohort-dependent presence in particular habitats and this may contribute to sexual segregation between habitats in species with sexual size dimorphism (SSD). The present study examines the validity of the “predation risk hypothesis” and importance of SSD on habitat (pelagic/inshore) segregation and dietary differences between sexes in three cyprinids with various magnitudes of SSD-roach (Rutilus rutilus), freshwater bream (Abramis brama) and bleak (Alburnus alburnus). Fish were sampled using gillnets over five consecutive years in the Římov Reservoir. Habitat segregation with female overrepresentation in the pelagic habitat was found in the most sexually dimorphic species with 26% bigger females—the roach. When analysis of habitat segregation was size-controlled, this segregation ceased to confirm the importance of SSD. Freshwater bream sexes (2% SSD) differed in diet but did not differ in habitat occupation. Bleak sexes were not segregated. Larger roach individuals (predominantly females) are less threatened by gape-limited predators and consequently they can occupy the risky but optimal (for zooplankton acquisition) pelagic habitat. Our results demonstrate that habitat segregation is present in the species with the most pronounced SSD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sexual segregation is defined as at least partial spatial segregation of sexes outside the reproductive season (Conradt, 2005). One of the factors contributing to sexual segregation is sexual size dimorphism-SSD (Main et al., 1996; Conradt, 1998; Ruckstuhl & Neuhaus, 2000; Croft et al., 2006; MacFarlane & Coulson, 2006) which is usually caused and maintained by different selective pressures on body size between sexes (Slatkin, 1984; Parker, 1992). For example the “fecundity selection hypothesis” explains SSD in species where larger body size of one sex is related to increased fecundity (Andersson, 1994). The “niche divergence hypothesis” states that each sex occupies a different niche which comes with the benefit of better resource exploitation (Shine, 1989). It was observed, that sexual segregation occurs in taxa where SSD is more than a threshold of 20% difference in size between sexes (Main et al., 1996; Ruckstuhl & Neuhaus, 2002; MacFarlane & Coulson, 2006). Several hypotheses explaining sexual segregation are often complementary and more than a single hypothesis can be supported for each species (Conradt, 2005).
The “predation risk hypothesis” explains habitat segregation when the more vulnerable sex prefers safer habitats than the less vulnerable sex (Bowyer, 1984; Ruckstuhl & Neuhaus, 2000). Habitat segregation by body size with larger individuals prevailing in predation risky habitat is a common coping mechanism with predation threat in fish (Werner et al., 1983; Brabrand & Faafeng, 1993; Hölker et al., 2002). For example, males of Trinidadian guppies Poecilia reticulata Peters, 1859 are under higher predation risk because they are smaller and more brightly coloured than females and hence they prefer shallower habitats with fewer predatory fish (Croft & Krause, 2004; Croft et al., 2006). Despite the guppy males’ attempts to hide from predators, they are still predated more than females, which leads to female-biased populations (Arendt et al., 2014). Also, only large individuals of roach Rutilus rutilus (Linnaeus, 1758) use open water when there is predation risk but all size cohorts use open water when there are no predators (Brabrand & Faafeng, 1993; Hölker et al., 2002). Roach females tend to be bigger than males (Tarkan, 2006) which in cases when the predator avoidance hypothesis applies can lead to habitat segregation of sexes (Croft et al., 2005). Unfortunately, to our knowledge it has never been tested in roach. In general, the body size assortment has the potential to be strong habitat segregation driver especially in sexually dimorphic fish species (Croft et al., 2005).
The “Forage selection hypothesis” explains sexual segregation caused by different diet requirements of the sexes (Beier, 1987). Diet can differ qualitatively (Nagy, 1987) and/or quantitatively (Sano, 1993). Size dimorphism is a strong prerequisite for sex-specific diet (Magurran & Maciás Garcia, 2000; Conradt, 2005). A large body has fitness advantages such as higher fecundity (Barneche et al., 2018) or higher competitive ability (Parker, 1992), but comes with the cost of higher absolute metabolic rate (Geist, 1974; Holker, 2003). Hence, the larger sex is strongly motivated to find optimal foraging habitats and often takes more risks when food gathering (Gross et al., 1995; Croft et al., 2006). For example, in Nubian ibex Capra ibex nubiana F. C. Cuvier, 1825 males are larger and consume a higher mass of food; thus, males seek resource-rich habitats where they can satisfy their higher forage requirements (Gross et al., 1995). Also, some monomorphic species have sex-specific diets: e.g. female sterlet Acipenser ruthenus Linnaeus, 1758 have a more diverse diet and forage on more Oligochaetes than males (Nagy, 1987). A common explanation for sex-specific diet is the higher energy demands of females for gamete production and/or spatial separation of sexes (Nagy, 1987; Gross et al., 1995; Magurran & Maciás Garcia, 2000).
Surprisingly, sexual segregation is understudied in the most diversified vertebrate group—freshwater bony fishes. Several existing studies were performed on model organisms for behavioural studies such as Trinidadian guppy, Eurasian minnow Phoxinus phoxinus (Linnaeus, 1758), threespine stickleback Gasterosteus aculeatus Linnaeus, 1758 and economically important brown trout Salmo trutta Linnaeus, 1758 (Haraldstad & Jonsson, 1983; Croft et al., 2006; Griffiths et al., 2014; Rystrom et al., 2018). In addition, only studies performed on Trinidadian guppy and brown trout were done in the wild, and hence there is a knowledge gap in sexual segregation of wild freshwater fish populations. It is noteworthy that common and widespread freshwater species, such as roach, freshwater bream Abramis brama (Linnaeus, 1758) and bleak Alburnus alburnus (Linnaeus, 1758)—have, to our knowledge, never been studied from the perspective of sexual segregation so far.
Roach, freshwater bream and bleak are common cyprinid species of European waters (Kottelat & Freyhof, 2007). These species have undistinguishable sex-specific characteristics outside the reproductive season but females are often larger and mature one year later than males (Backiel & Zawisza, 1968; Mann, 1973; Vollestad & L’Abée-Lund, 1987). The roach is a total spawner with a single spawning per year but freshwater bream and bleak are batch spawners with the possibility of multiple spawnings within a year (Backiel & Zawisza, 1968; Nunn et al., 2007). In accordance with the income-capital breeder continuum, dividing reproductive output into multiple batches potentially reduces the need for higher long-term energy storage (McBride et al., 2015). Reproductive mode has the potential to influence SSD (Parker, 1992) and consequently it can affect the degree of sexual segregation (Croft et al., 2005). Therefore, the various degrees of SSD make the studied species optimal models for testing the factors responsible for sexual segregation.
In the present study, we investigated sexual segregation in three European cyprinid species with various levels of SSD—roach, freshwater bream and bleak. For that purpose, we gathered fish samples using gillnets installed into inshore and pelagic habitat during five consecutive seasons in the Římov Reservoir, Czech Republic. We determined individual habitat, sex, body mass, age and gut content on large subsample of fish. Also we collected data on the occurrence of large predatory fish and zooplankton abundance. From these data, we estimated habitat segregation by comparing sex ratio between pelagic and inshore habitat and how sex ratio is influenced by habitat-specific predator and/or zooplankton abundance. We also determined SSD, habitat-specific fish-size structure, sex-specific mortality and we compared diets between sexes by gut content analysis. We predicted that habitat segregation will occur in species with pronounced SSD and that the smaller sex (under higher predation risk) would occupy safer inshore habitats and will be underrepresented in older age cohorts (predator avoidance hypothesis). We also predicted that spatial segregation, SSD and/or sex-specific investment in the gonads would contribute to sex-specific diet (forage selection hypothesis). We interpret our results in the light of sexual segregation in relation to predation avoidance and occupation of optimal feeding habitat.
Materials and methods
Study area and fish sampling design
The study was carried out at the canyon-shaped Římov Reservoir (48°50′ N; 14°30′ E, max. area 210 ha, max. depth 45 m) in the Czech Republic. The mesotrophic to eutrophic reservoir is used as a drinking water supply and commercial or recreational fishery is prohibited.
Sampling was conducted annually in the first half of August 2012–2016, long after the cessation of spawning in all studied species. European standard gillnets (benthic gillnet: 1.5 m height × 30 m length; pelagic gillnet: 3 m height x 30 m length, (CEN & EN14757, 2005) were used for sampling (except 2012 when pelagic nets with height of 4.5 m were used) at four localities (tributary, upper, middle, dam, Fig. 1) equally distributed along the reservoir. Standard gillnets (12 mesh sizes) were supplemented by four large mesh size gillnets to capture usually underestimated large fish individuals (Šmejkal et al., 2015). For the detection of habitat-specific sex ratios (i.e. habitat segregation of sexes), a triplicate of benthic gillnets was deployed inshore at two depth layers (0–3 m, 3–6 m) and a triplicate of pelagic gillnets was deployed offshore (pelagic habitat) to one depth layer (0–5 m). Inshore habitat was considered to be areas with maximal depth of 6 m and no further from the bank than 1/4 of the relative width of the reservoir profile at each sampling point. Pelagic habitat was considered to be areas with minimal depth of 4 m and further from the shore than 1/3 of the relative width of the reservoir profile. Reduced sampling design with sampling down to depth of 6 m produces corresponding results to sampling at all depth strata (Blabolil et al., 2017). Total sampling effort was 48 inshore gillnets (2160 m2) and 24 pelagic gillnets (2160 m2, except 2012) per season. All gillnets were set overnight which catches fish mainly during sunset and sunrise, the peak of activity (Prchalová et al., 2010). We agree that such sampling reduces the possibility of distinguishing separate groups of fish which occupy the location of the gillnet installation but, on the other hand, it provides information about sex ratio occurring over a longer period of time in the respective habitat. For statistical analysis, the overall catch to 6 m depth from both gillnet types (standard and large mesh gillnets = 12 nets per deployment inshore and 6 nets in pelagic habitat) was pooled separately for inshore and separately for pelagic habitat at each locality.
To estimate the predation threat of studied cyprinids, data about the abundance (CPUE, ind × 1000 m−2 of gillnet) of predatory fish species were collected from the same gillnet sampling as for studied cyprinids. Pikeperch Sander lucioperca (Linnaeus, 1758), asp Leuciscus aspius (Linnaeus, 1758), pike Esox lucius Linnaeus, 1758 and catfish Silurus glanis Linnaeus, 1758 were summed as large predatory fish sum. Perch Perca fluviatilis Linnaeus, 1758 was omitted as a predator due to its predominant planktivory and its orientation on juveniles in the Římov Reservoir (Kubečka et al., 1998).
Sampling was part of a commercial contract for the Vltava River Authority (http://www.pvl.cz/en) contract number 300/7225. All sampling procedures were approved by the Ethics Committee of Czech Academy of Sciences, Vltava River Authority and Environmental Department of the Municipal Authority of the Town of České Budějovice. The study was conducted in accordance with all legal requirements of the Czech Republic.
Determination of size, sex and age
Sexual size dimorphism affects the habitat choice of each sex by way of their reaction to predation risk (Brabrand & Faafeng, 1993; Croft et al., 2005). To test SSD, we had to determine size, sex and age of the fish. Each fish was measured to the nearest 5 mm standard length (SL) and weighed to the nearest 1 g using scales (Kern EMB 500, Germany and WH-A03L, China). Sex was determined by visual inspection of gonads in the field using a random subsample, in total we sex-determined 72% of the total catch of studied cyprinids during five seasons (average 762 fish per sampling year). Female gonads were recognized according to their ocher-pink colour and apparent presence of granulation. Testes lack granulation and have a pale white or light-grey colour. None of the fish were in spawning condition with ripened eggs in ovaries or releasing sperm.
For individual age estimates, scales from the dorsal part of the left side of fish were taken in seasons 2012–2014 and their annuli were read in the laboratory (Holčík & Hensel, 1972). At least 10 scales were taken to prevent age determination from regenerates. Otoliths (sagitta) were taken from large individuals who could be older than 5 years for more accurate age determination in the laboratory (Stevenson & Campana, 1992).
Food availability sampling
To reveal the potential role of food availability on sexual segregation (“forage selection hypothesis”), the inshore and pelagic zooplankton communities were sampled at close proximity to installed gillnets in 2012, 2013 and 2014. Zooplankton is the most important food resource in canyon-shaped reservoirs (Duncan & Kubečka, 1995). In addition, zooplankton is present in both pelagic and inshore habitats, and hence it is more relevant than zoobenthos which is present inshore only. Planktonic communities were sampled in five water layers from 0 to 5 m where the highest zooplankton abundance occurs (Seďa et al., 2007). Zooplankton in the pelagic habitat was sampled by 5 m vertical haul with a cone-shaped planktonic net with opening diameter of 30 cm and mesh size of 200 μm. The zooplankton in the inshore habitat was sampled using 30 L plexiglass collector (height 75 cm, equipped with a 200 μm net) directly above the bottom at five depths from 1 to 5 m. Zooplankton communities were estimated as the number of individuals per litre of water volume and the overall mean from all depth layers was used in the analysis.
Diet analysis
To compare differences in diet composition between sexes (“forage selection hypothesis”), we performed diet analysis from gut contents of captured fish in the years 2012–2014. The gut from pharyngeal teeth to anus was removed from fresh fish after sex determination and stored in ethanol fixative for further laboratory analysis. For diet analysis, three subsamples were taken from three different segments of each gut separated by an intestinal loop. Diet items were determined under a stereomicroscope (× 40 magnification) and categorized into 11 groups (plant detritus and mineral particles, Ephemeroptera, Chironomidae, Trichoptera, Heteroptera, non-aquatic insect, Daphnia spp., Leptodora sp., other Cladocera, Copepoda, other). The volumetric proportion (%) of each food item was visually estimated for each subsample and then the average value from three subsamples was computed. The dominant food item (the highest average of the estimated volume from three samples weighted by index of gut fullness) was compared between sexes.
The diet richness was estimated as the number of different food items found in the whole gut from three samples. The gut fullness index was represented by an ordinal scale from 0 to 5 (0—empty gut, 0.5—only a few food items; 1: 1–25% of gut filled; 1.5: exactly 25% of gut filled; 2: 26–50%; 2.5: 50%; 3: 51–75%; 3.5: 75%; 4: 75–100%; 4.5: 100% and 5: extremely full gut leading to extreme gut widening). Gut fullness was determined for each part of the gut, and thereafter the overall mean was computed as a single variable per individual used for analysis.
Statistical analysis
Testing SSD and indirect estimate of sex-specific mortality
We checked the degree of differences in average size between sexes and the outcome of different predation threats to sexes (estimated as age-related mortality) as significant predictors of habitat segregation (Croft et al., 2005). The difference in average body mass of females and males was tested by species-specific ANCOVA with log-transformed fish body mass as a response variable, fish sex as an explanatory variable and year of sampling (factor, 2012–2016) as covariate. To control for the effect of the different age of sexes on sexual size dimorphism, additional ANCOVA (with a smaller dataset 2012–2014 where age was determined) with the addition of age (continuous, exact determined age) as a covariate was carried out. The SSDindex was computed using the amended SSDindex; SSDindex = (Fbm − Mbm)/Mbm (Ruckstuhl & Neuhaus, 2000) for each species from average body masses obtained by mentioned ANCOVAs. Fbm is mean female body mass and Mbm is mean male body mass. This SSDindex was used for comparison with other studies examining the role of SSD in sexual segregation.
With data from 2012 to 2016, we compared average fish body mass between habitats, by species-specific ANCOVAs with log-transformed body mass as a response variable and sex in interaction with habitat as an explanatory variable. The factor year was used as a categorical covariate. Indirect evidence of different mortality between the sexes was compared as species-specific differences in male proportion between age cohorts (reduced to categories 2 + , 3 + , 4 + , and older in seasons 2012–2014) by binomial Generalized linear model with sex (binomial) as a response variable and age cohort (categorical) in interaction with year (categorical) as explanatory variables.
Examination of habitat segregation
Sexual segregation was tested in three steps:
-
1.
The first step determined if there is a difference in the ratio of males and females between habitats (habitat segregation) or along the longitudinal axis of the reservoir. Analysis without biotic variables was performed on a larger dataset (capture data from 2012 to 2016) to reduce potential Type II error which could occur with a smaller dataset. Species-specific binomial (Bernoulli) Generalized Linear Mixed Model (GLMM) with log-link function was used. The response variable was fish sex provided as raw binomial data from gillnet capture. Locality (tributary, upper, middle, dam) in interaction with habitat (pelagic, inshore) were added as explanatory variables and year (levels 2012–2016) was added as a random factor (Table S1).
The following steps (2 and 3) were performed to reveal the most important factor causing habitat segregation in our system. The role of abundance of predators (“predation risk hypothesis”), zooplankton abundance (“forage selection hypothesis”) and SSD (influences both hypotheses) were tested.
-
2.
The second step was performed in the case of significant segregation (differences in sex ratio) by locality or habitat in Step 1. In Step 2, we included data from 2012 to 2014 only, when biotic parameters: zooplankton abundance (ind × l−1) and large predatory fish sum (all size cohorts, CPUE, ind × 1000 m−2 of gillnet, Table S2) were measured and we added them to the binomial GLMM model structure from Step 1. These parameters should provide additional insights into the relationships of habitat segregation to the “predation risk hypothesis” and/or “forage selection hypothesis” for examined species.
-
3.
The third step was the addition of fish body mass as a covariate to minimal adequate models from Step 1 and Step 2 to control for the size difference between sexes and to see if this covariate would influence habitat segregation. Additional analysis with the addition of SL (instead body mass) was done to confirm the consistency of results.
All minimal adequate models were selected using backward selection by comparing the Akaike information criterion (AIC) between the full model and reduced model.
Diet segregation
The sex-specific proportion of dominant food items was compared by species-specific Multinomial regression (MR) with sex as a response variable and the covariates body mass, habitat, locality and year of sampling added. Sex*Habitat interaction was included in all models to reveal any potential interaction effect on the proportion of dominant diet items but it was removed due to worse model fit (based on ΔAIC > 2). Only dominant items, which dominated at least in five individuals in one species in total, were analysed. Diet richness and gut fullness of the whole gut were compared between the sexes (“forage selection hypotheses”). Gut fullness [permutation test with 5000 iterations (Wheeler & Torchiano, 2016)] and diet richness (Poisson GLM) were response variables and sex was the explanatory variable. The variables body mass, locality, habitat and year of sampling (factor) were added.
All statistical procedures were performed in the R environment v 3.4.3 (R Core Team, 2018). All models were checked for multicollinearity by variance inflation factor (vif function in the car package (Fox & Weisberg, 2011)) and model residuals were visually checked via diagnostic plots. All unimportant interactions in models were omitted if ΔAIC > 2. Multiple comparisons were carried out with Tukey method from emmeans package (Lenth, 2016). The level of statistical significance was set at α = 0.05.
Results
Sexual size dimorphism and indirect evidence of sex-specific mortality
With data from five seasons (2012–2016), a size comparison between sexes of each species and the SSDindex were tested for 1032 roach, 433 freshwater bream and 1642 bleak. With age determined fish from three seasons (2012–2014), a size comparison between sexes of each species, SSDindex and mortality analysis were completed for 535 roach, 193 freshwater bream and 406 bleak.
Roach was the most sexually dimorphic species in terms of body mass out of the studied cyprinids with SSDindex 83% and 26%, respectively (age-uncontrolled value and age-controlled value, respectively) with larger females than males (Table 1, see Table S3 for statistics of body mass comparison). Bleak had larger females than males with SSDindex 30% and 8%, respectively (Table S4). Surprisingly, males and females in freshwater bream were of similar sizes, SSDindex 12% and 2%, respectively (Tables 1, S5). Significantly bigger female and male roach were caught in the pelagic habitat (F1,1025 = 118.48, P < 0.001; Fig. 2). Pelagic roach males had similar body mass to inshore roach females (t1025 = 2.25, P = 0.112). There was no significant size difference between fish captured in both habitats in freshwater bream (Fig. 2). In bleak, significantly larger bleak were caught inshore (F1,1632 = 7.65, P = 0.006).
Sex- and habitat-dependent body mass of a roach, b freshwater bream and c bleak, in the Římov Reservoir, 2012–2016. Bold lines represent the median, white squares are the mean, boxes are interquartile ranges and whiskers are no more than 1.5 times the interquartile range. F female, M male, INS the inshore habitat, PEL the pelagic habitat
The proportion of males decreased with increasing age cohort in roach and bleak (Table 2, Fig. 3) indicating higher mortality of males with increasing age. Freshwater bream sex ratio was consistent in all age cohorts, Fig. 3. This pattern was consistent among years because cohort*year interaction was not significant (Table 2).
Age-dependent male proportion from the Římov Reservoir for three cyprinid species (roach—circle, freshwater bream—square, bleak—triangle). Filled symbols are mean proportion of males from three seasons (2012–2014). Error bars are 95% confidence intervals. Age cohort 2 + was not analysed for bream due to small sample of aged individuals. The grey horizontal line represents the equal sex ratio
Habitat segregation of sexes
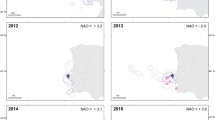
Habitat segregation was tested on the dataset from five seasons (Step 1) for 1239 roach, 572 freshwater bream and 1937 bleak. The roach was sexually segregated between the pelagic and inshore habitat with a higher proportion of females in the pelagic habitat (χ2 = 21.46, DF = 1, P < 0.001, Fig. 4a). There was no overlap in female proportion between habitats in any year (Fig. 4a). An analysis including zooplankton and predator abundance (Step 2) provided the same result as without them (Table S6, habitat-specific biotic parameters are shown in Fig. S1). The SSD was probably responsible for a different habitat choice between sexes because the addition of the fish body mass (χ2 = 82.36, DF = 1, P < 0.001) covariate to the model eliminated the occurrence of habitat-specific sex ratio (i.e. habitat segregation) (χ2 = 0.09, DF = 1, P = 0.765, Fig. 4b). The same results were obtained when we ran the analysis on standard length rather than body mass (Table S7). The sex ratios between inshore and pelagic habitat were similar in bleak and freshwater bream. (Table S8, S9).
Sexual segregation between inshore and pelagic habitats (habitat-dependent female proportion) of roach, in the Římov Reservoir. a Size-uncontrolled analysis of segregation, b size-controlled analysis of segregation. The size-controlled plot is from binomial GLMM with body mass as a covariate which controls for sexual dimorphism in body mass. The points represent female proportions in each sampling year (2012–2016). The bold horizontal line represents mean value. Error bars are 95% confidence intervals
Sex-specific diet composition
Diet analysis was completed on a subsample of 244 roach, 355 bleak and 146 freshwater bream from seasons 2012–2014. The female roach’s diet was more frequently dominated by detritus and mineral particles and the male roach’s diet was more frequently dominated by Daphnia sp. (sex: χ2 = 8.17, DF = 2, P = 0.017, Table 3). Body mass was not the significant covariate (χ2 = 4.22, DF = 2, P = 0.121). The same model revealed that the diet of pelagic roach was dominated by Daphnia spp. (t = − 6.17, DF = 18, P < 0.001) and the diet of inshore roach by detritus and mineral particles (t = 5.75, DF = 18, P < 0.001). Locality (χ2 = 55.11, DF = 6, P < 0.001) and year of sampling (χ2 = 62.07, DF = 4, P < 0.001) were significant factors. Freshwater bream females had more frequently chironomid larvae as a dominant food item and less frequently Daphnia spp. than males (sex: χ2 = 12.9, DF = 3, P = 0.005, Table 3). Body mass (χ2 = 31.08, DF = 3, P < 0.001), locality (χ2 = 44.38, DF = 9, P < 0.001), habitat (χ2 = 19.49, DF = 3, P < 0.001) and year (χ2 = 13.87, DF = 6, P = 0.031) were important factors. Dominant food items were similar in both sexes in bleak (sex: χ2 = 1.50, DF = 3, P = 0.682, Table 3); locality (χ2 = 112.34, DF = 9, P < 0.001) and year (χ2 = 99.74, DF = 6, P < 0.001) were both important factors. Both sexes had equally filled guts in all species (Table S10). Gut fullness was habitat-dependent in roach, with fuller guts in the pelagic habitat (Fig. S2, Table S10) and also in freshwater bream with fuller guts inshore (Fig. S2, Table S10). There was no habitat-dependent gut fullness in bleak (Fig. S2, Table S10). Diet richness did not differ between sexes in all species (Poisson GLM; roach: χ2 = 0.43, P = 0.513, bleak: χ2 = 0.14, P = 0.712, freshwater bream: χ2 = 0.14, P = 0.707).
Discussion
One of the major consequences of behavioural and life history differences between the sexes is sexual segregation in terms of at least partial spatial separation of sexes during the non-reproductive season (Ruckstuhl & Neuhaus, 2005; Ruckstuhl, 2007; Wearmouth & Sims, 2008). In the present study, habitat segregation with a higher proportion of females in the pelagic habitat than inshore was confirmed in roach, the species with the most pronounced SSD and sex-specific mortality. This segregation was present in all five consecutive seasons. Sexual differences in dominant dietary items were found in roach and freshwater bream. The female freshwater breams’ diet was more often dominated by chironomid larvae and the males’ diet by Daphnia sp. This study provides evidence for how sexual differences manifest in sexual segregation in three common European cyprinids.
Sexual segregation is often related to SSD (Ruckstuhl & Neuhaus, 2000; MacFarlane & Coulson, 2006; Ruckstuhl, 2007). In roach, females were much larger than males even when the analysis of body mass difference between the sexes was controlled for age. Observed SSDindex in roach was above the previously suggested threshold of 20% that significantly contributes to sexual segregation in ungulates (Main et al., 1996; Ruckstuhl & Neuhaus, 2002) and marsupials (MacFarlane & Coulson, 2006). The importance of SSD in roach habitat segregation was supported by a model with body mass as a covariate which meant that habitat factor was no longer a significant predictor. An alternative explanation for the observed differential habitat use by sex dimorphic roach is that ecological differentiation of each sex contributes to the occurrence of SSD (e.g. Shine, 1989). Nonetheless, more research is needed to disentangle the habitat segregation–SSD relationship in roach. On the other hand, the relevance of the SSDindex threshold level for sexual segregation in reservoir cyprinids was supported by the data from bleak and bream which did not overcome the 20% age-controlled SSDindex threshold and were not sexually segregated between habitats. We speculate that the 30% SSDindex in bleak from age-uncontrolled analysis is probably too small to have an antipredatory effect for a small cyprinid in a system with large predators. Bream adults are probably large enough to be protected from the majority of predators in the Římov Reservoir (Nilsson & Brönmark, 2000; Vašek et al., 2018). In general, habitat segregation between inshore and pelagic habitat was found only in species with the most pronounced SSDindex.
Sex-specific mortality risk results in overrepresentation of one sex in older age cohorts (Garcia et al., 1998; Tamate & Maekawa, 2004; Arendt et al., 2014). We identified a decreasing proportion of males with increasing age cohort in roach and bleak, which contributed to overall size differences between sexes. The observed decrease in proportion of males with increasing age cohort was stable over all three examined years (non-significant cohort*year interaction) and was also found in other studies (Mackay & Mann, 1969), suggesting the wide occurrence of this phenomenon. One of the contributing factors to the female bias in older roach and bleak age cohorts could be higher predation on males because they grow more slowly than females, and are susceptible for a longer time to gape-limited predators. Regardless of the higher proportion of roach males in safer inshore habitat, they can suffer higher predation rates, which was also observed in Trinidadian guppies where males are also smaller and choose safer habitats (Croft et al., 2006; Arendt et al., 2014). Hence, roach males as smaller individuals in a population would be pushed to occupy inshore habitats where they have a chance to hide (Werner et al., 1983; Hölker et al., 2002).
Size-specific habitat preference and size-assortative shoaling are important driving factors of habitat segregation in fish and often occurs as an antipredatory strategy (Croft et al., 2005). The present study was performed in a system with a similar abundance of predators in inshore and pelagic habitat, but the pelagic habitat is more predator-risky due to its lower complexity (Werner et al., 1983; Lammens et al., 1992). Larger roach individuals occupy open water in lakes with the presence of predators, while in a predator-free lake or with low predator abundance more sizes are present in open water (Lammens et al., 1992; Brabrand & Faafeng, 1993; Hölker et al., 2002). This study broadens existing mentioned evidence and provides additional information about sex bias in size-related use of habitat resulting in habitat segregation of sexes. A similar dominance of the larger sex in open water was found in brown trout (Haraldstad & Jonsson, 1983) and Trinidadian guppy (Croft et al., 2006). The presented evidence suggests that the “predation risk hypothesis” explains segregation between habitats in roach.
The larger sex often favours habitats providing higher food resources (Werner et al., 1983; Jakimchuk et al., 1987; Croft & Krause, 2004). Roach females as bigger individuals need to satisfy the higher energetic demands of a large body and gamete production (Trivers, 1972; Holker, 2003). Hence, females are pushed to occupy habitats with good food access such as the pelagic habitat where roach is an effective zooplankton predator (Winfield, 1986; Vašek et al., 2003). In the case of the Římov Reservoir, both sexes of roach captured in the pelagic habitat had fuller guts, mainly filled by zooplankton indicating that it is an important habitat for feeding (Vašek et al., 2003). We speculate that foraging could be the main reason why some males [potentially bolder individuals (Chapman et al., 2011)] take the risk of entering the pelagic habitat. Consumption of zooplankton has a profound effect on the growth and fecundity of roach (Perrow et al., 1990; Hölker et al., 2002). On the other hand, analysis controlled for habitat and body mass revealed that the diet of roach females was more often dominated by detritus and the diet of roach males by Daphnia spp. Naturally occurring SSD in roach prevents the detection of sex-specific diet because this difference emerges only when the analysis is controlled for body mass. Our results suggest that despite the evident higher proportion of roach males in the inshore habitat they rather consume zooplankton than other diet items. The aforementioned evidence implies that each sex evaluates trade-offs in predation risk and exploitation of optimal foraging habitat differently.
The diet of freshwater bream females was more often dominated by chironomid larvae and the diet of freshwater bream males by Daphnia spp. Because diet differences are not dependent on SSD in freshwater bream, we suggest that it is probably adaptive. Females should maximize energy intake due to higher investment in gamete production (Trivers, 1972; McBride et al., 2015). Zoobenthos alone is more nutritious than zooplankton (Wissing & Hasler, 1971; Hölker et al., 2002), but consumption of a benthic organism is usually related to larger effort and relatively high consumption of less nutritious detritus. On the other hand, zooplankton are usually captured in open water (Winfield, 1986; Vašek et al., 2008; Jarolím et al., 2010) without detritus intake, and hence the total energy gain can be higher in case of strict zooplankton consumption. The sex differences in diet and no apparent spatial segregation in freshwater bream can be caused by sex-specific dietary preferences within the same habitat or by limitation of our sampling design. In general, we can see an apparent preference for benthic feeding in females and planktonic feeding in males in both roach and freshwater bream. Diet differences between sexes can be related to the forage selection hypothesis (Beier, 1987) but our results are not conclusive.
The major caveat of the present study is the long soaking time of gillnets which reduces the distinction of separate fish groups captured at different times. On the other hand, it provides the sex ratio in each respective habitat over relatively long time interval and on a large sample size. To our knowledge, there is only a single study which studied habitat segregation in a large freshwater body which also used gillnets with a soaking time up to 24 h (Haraldstad & Jonsson, 1983). The disadvantage of night gillnet sampling is that it samples a more equally dispersed (in terms of fish abundance) fish community between inshore and pelagic habitat because smaller fish from inshore move to pelagic habitat due to horizontal diurnal migrations (Vašek et al., 2009). On the other hand, twilight periods of the day during fish intensively move are the key periods of capture for passive gears as gillnets (Prchalová et al., 2010). However, our result has validity for overnight data, as we were able to detect habitat segregation in roach. It is noteworthy that a previous study in the Římov Reservoir found that the occurrence of larger roach in pelagic habitat is stronger during daylight (Vašek et al., 2009). This suggests that females can dominate the pelagic habitat during the day even more than during the night. We acknowledge that the method used cannot provide sufficient information about the potential role of social factors in habitat segregation which is common in many terrestrial species (Ruckstuhl & Neuhaus, 2005).
The presence of sexual segregation between habitats suggests that males and females select living areas according to different criteria. In the present study, we have demonstrated that sexual differences in SSD, mortality risk, and diet contribute to habitat segregation of wild roach. Our results suggest that habitat segregation in roach is related to the “predation risk hypothesis” via SSD with larger females occupying higher predation risk unstructured pelagic habitat. In addition, pelagic roach had a fuller gut which suggests that the pelagic habitat is important for feeding, and therefore the “forging selection hypothesis” cannot be excluded. There was some evidence for freshwater bream sex-specific diet which was not related to SSD but is likely related to sex-specific energy allotment to gonads. In bleak, there was no sexual segregation despite the sex-specific mortality and weak SSD. The bleaks’ SSD did not overcome the previously suggested threshold for SSD and was not segregated. The present study is in accordance with increasing attention to sex-specific ecology and behaviour of freshwater fish (Hanson et al., 2008) and widens knowledge of sexual segregation in freshwater fish which can be useful in sex-aimed management of the studied species. We believe that a major benefit of the present study is evidence of sexual segregation in cyprinids from the wild and that it can become a starting point for future studies using more precise and/or individual-based methods (e.g. telemetry) for disentangling sexual segregation in reservoir cyprinids.
References
Andersson, M. B., 1994. Sexual selection. Princeton University Press.
Arendt, J. D., D. N. Reznick & A. López-Sepulcre, 2014. Replicated origin of female-biased adult sex ratio in introduced populations of the Trinidadian Guppy (Poecilia reticulata). Evolution 68: 2343–2356.
Backiel, T. & J. Zawisza, 1968. Synopsis of biological data on the bream. FAO Fisheries Synopsis 36: 121.
Barneche, D. R., C. R. White & D. J. Marshall, 2018. Fish reproductive-energy output increases disproportionately with body size. Science 360: 642–645.
Beier, P., 1987. Sex differences in quality of White-tailed deer diets. Journal of Mammalogy 68: 323–329.
Blabolil, P., D. S. Boukal, D. Ricard, J. Kubečka, M. Říha, M. Vašek, M. Prchalová, M. Čech, J. Frouzová, T. Jůza, M. Muška, M. Tušer, V. Draštík, M. Šmejkal, L. Vejřík & J. Peterka, 2017. Optimal gillnet sampling design for the estimation of fish community indicators in heterogeneous freshwater ecosystems. Ecological Indicators 77: 368–376.
Bowyer, T., 1984. Sexual segregation in Southern mule deer. Journal of Mammalogy 65: 410–417.
Brabrand, Å. & B. Faafeng, 1993. Habitat shift in roach (Rutilus rutilus) induced by pikeperch (Stizostedion lucioperca) introduction: predation risk versus pelagic behaviour. Oecologia 95: 38–46.
CEN EN14757, 2005. Water quality: sampling of fish with multi-mesh gillnets. En 14757:2005 3: 29.
Chapman, B. B., K. Hulthén, D. R. Blomqvist, L. A. Hansson, J. Å. Nilsson, J. Brodersen, P. Anders Nilsson, C. Skov & C. Brönmark, 2011. To boldly go: individual differences in boldness influence migratory tendency. Ecology Letters 14: 871–876.
Conradt, L., 1998. Could asynchrony in activity between the sexes cause intersexual social segregation in ruminants? Proceedings of the Royal Society B: Biological Sciences 265: 1359–1368.
Conradt, L., 2005. Definitions, hypotheses, models and measures in the study of animal segregation. Sexual segregation in vertebrates. Cambridge University Press, Cambridge: 11–35.
Croft, D. P. & M. Krause, 2004. Is sexual segregation in the guppy, Poecilia reticulata, consistent with the predation risk hypothesis? Environmental Biology of Fishes 71: 127–133.
Croft, D. P., J. Krause & R. James, 2005. Predation risk as a driving factor for size assortative shoaling and its implications for sexual segregation in fish. In Ruckstuhl, K. E. & P. Neuhaus (eds), Sexual segregation in vertebrates. Cambridge University Press, Cambridge: 115–126.
Croft, D. P., L. J. Morrell, A. S. Wade, C. Piyapong, C. C. Ioannou, J. R. G. Dyer, B. B. Chapman, Y. Wong & J. Krause, 2006. Predation risk as a driving force for sexual segregation: a cross-population comparison. The American Naturalist 167: 867–878.
Duncan, A. & J. Kubečka, 1995. Land/water ecotone effects in reservoirs on the fish fauna. Hydrobiologia 303: 11–30.
Fox, J. & S. Weisberg, 2011. An R companion to applied regression. Sage, Thousand Oaks, CA.
Garcia, C. M., E. Saborio & C. Berea, 1998. Does male-biased predation lead to male scarcity in viviparous fish? Journal of Fish Biology 53: 104–117.
Geist, V., 1974. On the relationship of social evolution and ecology in ungulates. Integrative and Comparative Biology 14: 205–220.
Griffiths, S. W., J. E. Orpwood, A. F. Ojanguren, J. D. Armstrong & A. E. Magurran, 2014. Sexual segregation in monomorphic minnows. Animal Behaviour 88: 7–12.
Gross, J. E., P. U. Alkon & M. W. Demment, 1995. Grouping patterns and spatial segregation by Nubian ibex. Journal of Arid Environments 30: 423–439.
Hanson, K. C., M. A. Gravel, A. Graham, A. Shoji & S. J. Cooke, 2008. Sexual variation in fisheries research and management: when does sex matter? Reviews in Fisheries Science 16: 421–436.
Haraldstad, O. & B. Jonsson, 1983. Age and sex segregation in habitat utilization by brown trout Salmo trutta in a norwegian lake. Transactions of the American Fisheries Society 112: 27–37.
Holčík, J. & K. Hensel, 1972. Ichtyologická príručka. Vydavaťelstvo Obzor, Bratislava.
Holker, F., 2003. The metabolic rate of roach in relation to body size and temperature. Journal of Fish Biology 62: 565–579.
Hölker, F., S. S. Haertel, S. Steiner & T. Mehner, 2002. Effects of piscivore-mediated habitat use on growth, diet and zooplankton consumption of roach: an individual-based modelling approach. Freshwater Biology 47: 2345–2358.
Jakimchuk, R. D., S. H. Ferguson & L. G. Sopuck, 1987. Differential habitat use and sexual segregation in the Central Arctic caribou herd. Canadian Journal of Zoology 65: 534–541.
Jarolím, O., J. Kubečka, M. Čech, M. Vašek, J. Peterka & J. Matěna, 2010. Sinusoidal swimming in fishes: the role of season, density of large zooplankton, fish length, time of the day, weather condition and solar radiation. Hydrobiologia 654: 253–265.
Kottelat, M. & J. Freyhof, 2007. Handbook of European freshwater fishes. Recherche, Berlin.
Kubečka, J., J. Seďa & J. Matěna, 1998. Fish-zooplankton interactions during spring in a deep reservoir. International Review of Hydrobiology 83: 431–441.
Lammens, E. H. R. R., A. Frank-Landman, P. J. McGillavry & B. Vlink, 1992. The role of predation and competition in determining the distribution of common bream, roach and white bream in Dutch eutrophic lakes. Environmental Biology of Fishes 33: 195–205.
Lenth, R. V., 2016. Least-squares means: the R package lsmeans. Journal of Statistical Software 69: 1–33.
MacFarlane, A. M. & G. Coulson, 2006. Sexual segregation in Australian marsupials. Sexual segregation in vertebrates: ecology of the two sexes. Cambridge University Press, Cambridge: 254–279.
Mackay, I. & K. H. Mann, 1969. Fecundity of two cyprinid fishes in the River Thames, Reading, England. Journal of the Fisheries Research Board of Canada 26: 2795–2805.
Magurran, A. E. & C. Maciás Garcia, 2000. Sex differences in behaviour as an indirect consequence of mating system. Journal of Fish Biology 57: 839–857.
Main, M. B., F. W. Weckerly & V. C. Bleich, 1996. Sexual segregation in ungulates: new directions for research. Journal of Mammalogy 77: 449–461.
Mann, R. H. K., 1973. Observations on the age, growth, reproduction and food of the roach Rutilus rutilus (L.) in two rivers in southern England. Journal of Fish Biology 5: 707–736.
McBride, R. S., S. Somarakis, G. R. Fitzhugh, A. Albert, N. A. Yaragina, M. J. Wuenschel, A. Alonso-Fernández & G. Basilone, 2015. Energy acquisition and allocation to egg production in relation to fish reproductive strategies. Fish and Fisheries 16: 23–57.
Nagy, S., 1987. The food of sterlet (Acipenser ruthenus) in the Czechoslovak–Hungarian section of the Danube. Folia Zoologica 36: 281–287.
Nilsson, P. A. & C. Brönmark, 2000. Prey vulnerability to a gape-size limited predator: behavioural and morphological impacts on northern pike piscivory. Oikos 88: 539–546.
Nunn, A. D., J. P. Harvey & I. G. Cowx, 2007. Variations in the spawning periodicity of eight fish species in three English lowland rivers over a 6 year period, inferred from 0 + year fish length distributions. Journal of Fish Biology 70: 1254–1267.
Parker, G., 1992. The evolution of sexual size dimorphism in fish. Journal of Fish Biology 41: 1–20.
Perrow, M. R., G. Peirson & C. R. Townsend, 1990. The dynamics of a population of roach Rutilus rutilus L. in a shallow lake. Is there a two-year cycle in recruitment? Hydrobiologia 191: 67–74.
Prchalová, M., T. Mrkvička, J. Kubečka, J. Peterka, M. Čech, M. Muška, M. Kratochvíl & M. Vašek, 2010. Fish activity as determined by gillnet catch: a comparison of two reservoirs of different turbidity. Fisheries Research 102: 291–296.
R Core Team, 2018. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria, https://www.r-project.org/.
Ruckstuhl, K. E., 2007. Sexual segregation in vertebrates: proximate and ultimate causes. Integrative and Comparative Biology 47: 245–257.
Ruckstuhl, K. E. & P. Neuhaus, 2000. Sexual segregation in Ungulates: a new approach. Behaviour 137: 361–377.
Ruckstuhl, K. E. & P. Neuhaus, 2002. Sexual segregation in ungulates: a comparative test of three hypotheses. Biological Reviews of the Cambridge Philosophical Society 77: 77–96.
Ruckstuhl, K. E. & P. Neuhaus (eds), 2005. Sexual segregation in vertebrates. Cambridge University Press, Cambridge.
Rystrom, T. L., V. F. Clement, I. P. Rick, T. C. M. Bakker & M. Mehlis, 2018. Shoal sex composition and predation risk influence sub-adult threespine stickleback shoaling decisions. Behavioural processes. Elsevier BV, New York.
Sano, M., 1993. Foraging activities and diets of males and females in a haremic sandperch (Pisces: pinguipedidae). Marine Ecology Progress Series 98: 55–59.
Seďa, J., A. Petrusek, J. Macháček & P. Smilauer, 2007. Spatial distribution of the Daphnia longispina species complex and other planktonic crustaceans in the heterogeneous environment of canyon-shaped reservoirs. Journal of Plankton Research 29: 619–628.
Shine, R., 1989. Ecological causes for the evolution of sexual dimorphism: A review of the evidence. The Quarterly Review of Biology 64: 419–461.
Slatkin, M., 1984. Ecological causes of sexual dimorphism. Evolution 38: 622–630.
Šmejkal, M., D. Ricard, M. Prchalová, M. Říha, M. Muška, P. Blabolil, M. Čech, M. Vašek, T. Jůza, A. M. Herreras, L. Encina, J. Peterka & J. Kubečka, 2015. Biomass and abundance biases in European standard gillnet sampling. PLoS ONE 10: 1–15.
Stevenson, D. K. & S. E. Campana (eds), 1992. Otolith microstructure examination and analysis. Department of Fisheries and Oceans, Ottawa.
Tamate, T. & K. Maekawa, 2004. Female-biased mortality rate and sexual size dimorphism of migratory masu salmon, Oncorhynchus masou. Ecology of Freshwater Fish 13: 96–103.
Tarkan, A. S., 2006. Reproductive ecology of two cyprinid fishes in an oligotrophic lake near the southern limits of their distributions range. Ecology of Freshwater Fish 15: 131–138.
Trivers, R. L., 1972. Parental investment and sexual selection. In Campbell, T. (ed.), Sexual selection & the descent of man. Aldine Publishing Company, Chicago: 136–179.
Vašek, M., J. Kubečka & J. Seda, 2003. Cyprinid predation on zooplankton along the longitudinal profile of a canyon-shaped reservoir. Archiv Fur Hydrobiologie 156: 535–550.
Vašek, M., O. Jarolím, M. Čech, J. Kubečka, J. Peterka & M. Prchalová, 2008. The use of pelagic habitat by cyprinids in a deep riverine impoundment: rímov Reservoir, Czech Republic. Folia Zoologica 57: 324–336.
Vašek, M., J. Kubečka, M. Čech, V. Draštík, J. Matěna, T. Mrkvička, J. Peterka & M. Prchalová, 2009. Diel variation in gillnet catches and vertical distribution of pelagic fishes in a stratified European reservoir. Fisheries Research 96: 64–69.
Vašek, M., A. P. Eloranta, I. Vejříková, P. Blabolil, M. Říha, T. Jůza, M. Šmejkal, J. Matěna, J. Kubečka & J. Peterka, 2018. Stable isotopes and gut contents indicate differential resource use by coexisting asp (Leuciscus aspius) and pikeperch (Sander lucioperca). Ecology of Freshwater Fish 27: 1054–1065.
Vollestad, L. A. & J. H. L’Abée-Lund, 1987. Reproductive biology of stream-spawning roach, Rutilus rutilus. Environmental Biology of Fishes 18: 219–227.
Wearmouth, V. J. & D. W. Sims, 2008. Sexual segregation in marine fish, reptiles, birds and mammals. Behaviour patterns, mechanisms and conservation implications. Advances in Marine Biology 54: 107–170.
Werner, E. E., J. F. Gilliam, D. J. Hall & G. G. Mittelbach, 1983. An experimental test of the effects of predation risk on habitat use in fish. Ecology 64: 1540–1548.
Wheeler, B., & M. Torchiano, 2016. lmPerm: permutation tests for linear models. R package version 2.1.0, https://cran.r-project.org/package=lmPerm.
Winfield, I. J., 1986. The influence of simulated aquatic macrophytes on the zooplankton consumption rate of juvenile roach, Rutilus rutilus, rudd, Scardinius erythrophthalmus, and perch, Perca fluviatilis. Journal of Fish Biology 29: 37–48.
Wissing, T. E. & A. D. Hasler, 1971. Intraseasonal change in caloric content of some freshwater invertebrates. Ecology 52: 371–373.
Acknowledgements
We thank Martin Čech, Tomáš Jůza, Michal Tušer and Daniel Bartoň for fish-sex determination, Kateřina Soukalová for age determination, Vilém Děd for data processing and all other FishEcU members who helped with data collection http://www.fishecu.cz. We thank Ingrid Steenbergen for English correction and three anonymous reviewers for their insightful comments. The work was supported from ERDF/ESF project Biomanipulation as a tool for improving the water quality of dam reservoirs (No. CZ.02.1.01/0.0/0.0/16_025/0007417) and by Czech Science Foundation P505-12-P647. The work of Jakub Žák from the Department of Zoology, Charles University was partially supported by SVV 260 434/2020.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflict of interest.
Additional information
Handling editor: Grethe Robertsen
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Žák, J., Prchalová, M., Šmejkal, M. et al. Sexual segregation in European cyprinids: consequence of response to predation risk influenced by sexual size dimorphism. Hydrobiologia 847, 1439–1451 (2020). https://doi.org/10.1007/s10750-020-04198-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-020-04198-x