Abstract
This work studies benthic CPOM in two streams of Ecuador: the Atacames stream, located in a developed watershed, and the Súa stream, located in a rural watershed and used as a reference. It is tested whether the amount, composition and timing of benthic CPOM will differ between them as a function of watershed and riparian land uses. Benthic CPOM was collected at five study sites on each stream with a Surber net and classified into four categories: leaves, twigs and bark, flowers and fruits and debris. Leaves were further identified to genus or species. There were no significant differences in the amount, composition and timing of benthic CPOM between the streams. CPOM storage showed strong seasonality linked to seasonal rainfall and a weak relation with land uses, channel width and stream order. Diversity of the benthic CPOM was high and 30 species contributed to the benthic leaf pool. Presence or absence of Ficus species with heavy leaves that are easily retained in the streambed explained the spatial distribution of benthic CPOM, so spatial differences in the composition of the riparian vegetation in these tropical streams seem to be more important to explain CPOM distribution than in their temperate counterparts.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Coarse particulate organic matter (CPOM) in rivers is composed of particles with a diameter larger than 1 mm (Pozo et al., 2009). CPOM in forest streams is mainly composed of plant materials including leaves, twigs, reproductive structures and fragmented materials that originate in the riparian vegetation that borders the channel (Webster et al., 1990; Pozo et al., 1997). CPOM enters the stream as a direct input from the forest canopy, as a lateral input when the materials on the banks slide down into the channel or as an upstream input when materials are transported by the water flow (Molinero & Pozo, 2004, 2006) and is trapped in channel obstacles such as rocks, boulders, branches and debris damns (Díez et al., 2000; Larrañaga et al., 2003). Benthic CPOM that is retained in the streambed plays a major role in the ecological functioning of forest rivers and streams because it contributes to channel heterogeneity (Webster et al., 1994) and it is an energy source for microorganisms and benthic macroinvertebrates (Wallace et al., 1997; Baldy et al., 2007; Woodward et al., 2012). In addition to be an energetic resource in streams, terrestrial CPOM inputs to fluvial systems can be considered a fraction of lateral carbon fluxes which transport organic carbon away from the areas where it was originally photosynthesized, contributing to regional CO2 sinks and balancing the carbon budget of terrestrial ecosystems (Ciais et al., 2008; Aufdenkampe et al., 2011). Transport of carbon by riverine systems into coastal waters is dominated by dissolved inorganic carbon and CPOM represents only a minor fraction (Aufdekampe et al., 2011; Stackpoole et al., 2012). However, CPOM is temporally retained within the drainage network, which provides many opportunities for its biological degradation and may represent an important source of atmospheric CO2 (Battin et al., 2008; Stackpoole et al., 2012).
Land use changes near the stream modify the amount, quality and timing of CPOM inputs and benthic CPOM storage (Delong & Brusven, 1994; Karlsson et al., 2005; Carroll & Jackson, 2009). The loss of the riparian vegetation because of land use changes or because of forestry practices reduces the benthic CPOM stocks in the channel (Delong & Brusven, 1993; Reid et al., 2008). When the riparian vegetation is removed, the amount of leaves in the benthic CPOM diminishes, although the residues of the clearing activities may be accumulated in the channel for different periods of time (Haggerty et al., 2004; Santiago et al., 2011). In monoculture forestry plantations, the riparian vegetation is substituted by fast-growing species of commercial interest which largely modifies the stream CPOM budget (Molinero & Pozo, 2006). These changes are considered deleterious and the quantity and quality of the benthic CPOM is a good indicator of the conservation status of these stream ecosystems (Epstein et al., 2016).
Studies about the CPOM in the tropical rivers of South America are scarce, although there is information from some biomes: in Ecuador, there are studies on the benthic CPOM in Andean streams (Ríos-Touma et al., 2009; Vanegas, 2016); in Brazil, in the savannah and the Atlantic rainforest (Gonçalves et al., 2006; Gonçalves & Callisto, 2013; Gonçalves et al., 2014; Bambi et al., 2017; Rezende et al., 2017) and in Colombia, in the rainforest of the Chocó (Valencia et al., 2009). These works have shown that benthic CPOM storage is controlled by the seasonality of rainfall and discharge and that its composition is characterized by a high diversity of plant species. However, none of these studies have addressed the impact of land use changes on the benthic CPOM storage.
This work compares benthic CPOM in two coastal tropical streams of Ecuador: one of them, the Atacames stream, is located in a developed watershed with paved roads, established town centers and two weirs for recreational use, and the other, the Súa streams, is located in a watershed mostly occupied by dispersed farms and is used as a reference. The objectives of this work are: (1) to compare the amount, composition and temporal variability of benthic CPOM in two streams with different land uses and (2) to identify the factors that control storage and composition of benthic CPOM. It is hypothesized that the amount, composition and temporal variability of benthic CPOM will differ between the two streams as a function of the land uses in the riparian area and in the watershed.
Materials and methods
Study area
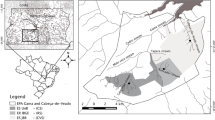
The Atacames and Súa streams are in the Ecuadorian Chocó region, South of Esmeraldas city (Fig. 1). Mean monthly temperature varies between 24 and 26°C and annual precipitation, between 500 mm in the lower parts of the Atacames watershed and 4000 mm in the South-western edge of the Súa watershed. Precipitation is strongly seasonal, with a wet season from January to June and a dry season from July to December (GADMA, 2014; GADPS, 2015; Fig. S1). Potential vegetation in both watersheds ranges from tropical evergreen forest in the headwaters and in the middle part of the watershed to tropical semideciduous forest in the lower part of the watershed (MAE, 2005).
The Atacames, a 4th order stream, is 42.5 km long, springs in the North-western side of the Cordillera Costanera inside the Mache-Chindul Natural Reserve and ends at the Atacames city (Fig. 1). The Atacames watershed has 117.6 km2 with a 34.8% cover of native forest and 17.6% of pasture land, although if we exclude the Reserva Natural Mache-Chindul, land uses become 25.5% of native forest and 23.7% of pasture land (MAGAP, 2006; MAG, 2016). The Súa, a 3rd order stream, is 27.6 km long, also springs inside the Mache-Chindul Natural Reserve and ends at Súa town (Fig. 1). The Súa watershed has 63.7 km2 with a 25.8% cover of native forest and 4.3% of pasture land (MAGAP, 2006; MAG, 2016). Other parts of the watersheds are occupied by a mosaic of villages, agricultural land, forest patches and forestry plantations. Both streams have a low gradient channel (1.0–2.3%) that forms a sequence of riffles and pools with a streambed composed by gravel and sand and a mean depth than ranges between 30 and 70 cm during the dry season. Because the Atacames watershed is close to the touristic town of Atacames with more than 20,000 habitants, it is more developed and has asphalt roads, regular town centers, two large weirs that are used for recreation and the banks in the Atacames stream are more modified than in the Súa stream.
Field work
We selected five sampling sites on each river as a function of stream order. Study sites were further characterized by measuring channel width, canopy angle (angle that is formed by the highest points of the riparian vegetation on each side and the middle point of the stream channel) and the percentage of pasture land and native forest cover in the riparian area (a 30-m-width buffer strip on each side of the channels draining into the sampling point) and in the watershed (Fig. 1, Table 1). At each site, we established study reaches of 25 m (sites with a channel width of 5 m or more) or 50 m (sites with a channel width less than 5 m) and we collected five random samples of benthic CPOM with a Surber sampler (30 × 30 cm) and 0.5 mm mesh size. Sites were sampled bimonthly from July 2016 to May 2017. The collected samples were stored in labeled plastic bags that were transported to the laboratory and frozen until analyzed.
Laboratory work
In the laboratory, samples were unfrozen and air dried for 48 h in paper pans. The CPOM was sorted into four categories: leaves, twigs and bark (only branches with less than 1 cm diameter), flowers and fruits and debris (particles over 0.5 mm diameter that we couldn’t sort into the other categories). Additionally, the leaves were sorted into morphotypes and identified to genus or species with field guides (Little & Dixon, 1983; Palacios, 2011) and the use of digital herbariums from the Missouri Botanical Garden (2018), Arizona State University (2018) and the Field Museum (2018). The identified species were further classified into three groups: species that are common on the margins of rivers (riparian); species that are cultivated (cultivated) and species that are not cultivated (non-cultivated). The sorted CPOM was dried in an Elos Heat oven at 65°C for 24 h and combusted in a furnace at 450°C for 5 h and weighted in a Mettler Toledo MS104S scale to the nearest 0.0001 g. All CPOM data were expressed as ash free dry weight in g m−2.
Statistical analysis
To test spatial and temporal differences between the two rivers, data of total CPOM density and the categories (g m−2) were transformed with the square root function to reduce the impact of extreme values (Greenacre & Primicerio, 2014) and were compared by nested four-way ANOVA (River × Season × Site × Month) with Site as a random factor nested into the River factor and Month as a non-random factor nested into the Season factor (Table S1). Contributions of categories to total CPOM (%) were transformed with the logit function (Warton & Hui, 2011) and compared by nested four-way ANOVA in the same way as the CPOM density data (Table S2).
To identify which factors are controlling the quantity and composition of the benthic CPOM, a principal component analysis (PCA) was performed on the variables used to describe the study sites (Table 1). Significance of the principal components and the variable loadings was assessed by the broken-stick method following Peres-Neto et al. (2003). Then, linear correlations between the amount and composition of the benthic CPOM and the first and second principal components obtained in the PCA were performed. Dependent variables were transformed as explained above before the correlation analyses. All statistical analyses were done in R (R Development Core Team, 2015).
Results
Benthic CPOM storage
Mean total CPOM storage varied between 28.2 and 86.6 g m−2 among the sites in the Atacames river and between 16.8 and 44.2 g m−2 among the sites in the Súa river (Table 2), but there were no significant differences between the two rivers (four-way ANOVA, F1,8 = 0.97, P > 0.05). There were neither significant differences between the two rivers in the CPOM categories (four-way ANOVA: leaves, F1,8 = 0.11, P > 0.05; twigs and bark, F1,8 = 2.36, P > 0.05; flowers and fruits, F1,8 = 1.12, n. s.; debris, F1,8 = 0.11, n. s.), although the average amount of twigs and bark in the Atacames river (18.3 g m−2) was more than double than in the Súa river (7.0 g m−2).
There was a strong seasonality in the benthic CPOM storage in both rivers (Fig. 2). CPOM storage peaked in the middle (September) or at the end (November) of the dry season and diminished after January during the wet season. Differences in CPOM storage between the dry and wet season were significant (four-way ANOVA, F1,8 = 19.53, p < 0.01). This temporal pattern was clearly observed at sites A1, A2, A5, S1, S3, and S4, but slightly differed in the other study sites (Fig. 2). The categories showed a temporal variability that was similar to that of total CPOM (not shown). Differences between the dry and wet season were significant for the leaves, twigs and bark and debris categories (four-way ANOVA: leaves, F1,8 = 37.47, P< 0.001; twigs and bark: F1,8 = 6.50, P < 0.05; debris: F1,8 = 21.46, P < 0.01) and were not significant for the flowers and fruits category (four-way ANOVA, F1,8 = 2.65, p > 0.05).
Benthic CPOM composition
Leaves and twigs and bark categories represented more than 50% of the total benthic CPOM in both streams (Table 2). The debris category was also abundant and represented more than 30% of the total benthic CPOM. The flower and fruits category contributed with the lowest percentage to the total benthic CPOM in both rivers. The percentage of twigs and bark in the Atacames stream was significantly larger than in the Súa stream (four-way ANOVA, F1,8 = 6.08, P < 0.05), but the other categories showed no significant differences between the two rivers (four-way ANOVA: leaves, F1,8 = 0.71, P > 0.05; fruits and flowers, F1,8 = 0.10, P > 0.05; debris, F1,8 = 0.11, P > 0.05).
Benthic CPOM composition varied along time but there was no a consistent temporal pattern among the study sites (Fig. 3). There were significant differences between the wet and dry season in the percentage of twigs and bark (four-way ANOVA, F1,8 = 5.62, P < 0.05), but the other categories showed no significant differences between the two seasons (four-way ANOVA: leaves, F1,8 = 0.26, p > 0.05; flowers and fruits, F1,8 = 0.43, P> 0.05; debris, F1,8 = 0.76, P> 0.05).
Contribution of riparian species
Diversity of leaves in the benthic CPOM of these streams was high and 30 morphotypes were identified and classified to genus and species (Table S3 and Table 3). The number of species was similar in both streams and 17 were shared between them (Table S3). Seventeen families were identified and Moraceae and Fabaceae together contributed to 64% of the benthic leaf stock in the Atacames stream and 86% in the Súa stream. Other families that contributed with a percentage of 5% or higher to the benthic leaf stock were Lauraceae, Malvaceae, Poaceae and Sapotaceae.
The most abundant species in the benthic leaf stock were Zygia longifolia (Humb. & Bonpl. Ex Willd.) Britton & Rose and an undetermined Ficus species (Ficus sp1, Table 3). Mean benthic storage of Z. longifolia was similar in both streams, and there were no significant differences between them (four-way ANOVA, F1,8 = 0.15, P > 0.05). Mean benthic storage of Ficus sp1 in the Atacames stream was lower than in the Súa stream but the differences were not significant (four-way ANOVA, F1,8 = 1.86, P> 0.05). Z. longifolia was abundant in all the sites located in third- and four-order reaches, excepting site A5 (Fig. 4), while Ficus sp1 was abundant in the lower part of the Súa stream (sites S4 and S5, Fig. 4). Benthic storage of Z. longifolia peaked during the dry season, while Ficus sp1 peaked during the dry season, but it was also found in the benthos during the wet season in four sites (S2, S3, S4 and S5, Fig. 4). The seasonal differences were only significant for Z. longifolia (four-way ANOVA: Z. longifolia, F1,8 = 8.07, P < 0.05; Ficus sp1, F1,8 = 0.57, P > 0.05).
Contribution of Z. longifolia and Ficus sp1 to the benthic leaf stock in the Atacames stream was lower than in the Súa stream (Table 3). However, there were no significant differences between the two streams (four-way ANOVA: Z. longifolia, F1,8 = 0.15, P > 0.05; Ficus sp1, F1,8 = 1.86, P > 0.05). There were neither significant differences between the dry and wet seasons in the contribution of these species to the benthic leaf stock (four-way ANOVA: Z. longifolia, F1,8 = 0.40, P > 0.05; Ficus sp1, F1,8 = 0.07, P > 0.05). Other species that represented a 5% or more of the benthic leaf stock: Castilla tunu Hemsl., Chrysophyllum argenteum Jacq., Theobroma cacao L., Guadua angustifolia Kunth, Inga edulis Mart., Ficus sp2, Terminalia catappa L., Erythrina sp. and Mezilaurus sp. (Table 3).
Neither the amount of leaves from riparian species, cultivated species and non-cultivated species in the benthos (Table 3, four-way ANOVA: riparian: F1,8 = 0.22, P > 0.05; non-cultivated, F1,8 = 1.20, P > 0.05; cultivated, F1,8 = 3.32, P > 0.05) nor their contribution to the benthic leaf stock as percentages (Table 3, four-way ANOVA, riparian: F1,8 = 0.42, P > 0.05; non-cultivated, F1,8 = 1.53, P > 0.05; cultivated, F1,8 = 2.21, P > 0.05) showed significant differences. However, the amount of cultivated species in the Atacames stream was twofold higher than in the Súa stream (Table 3). Only the amount of riparian and cultivated species in the benthos showed significant seasonal differences (four-way ANOVA: riparian, F1,8 = 8.07, P < 0.05; non-cultivated, F1,8 = 0.95, P > 0.05; cultivated, F1,8 = 5.71, P < 0.05). However, there were no significant seasonal differences in the contribution of these categories to the benthic leaf stock as percentages (Table 3, four-way ANOVA: riparian, F1,8 = 0.14, P > 0.05; non-cultivated, F1,8 = 0.28, P > 0.05; cultivated, F1,8 = 0.02, P > 0.05).
Factors controlling CPOM distribution
The PCA selected two components that explained 82% of the variance of physical variables and land use indicators used to describe the study sites (Table 4, Fig. S2). The first component (PC1) was positively correlated with drainage area, the canopy angle, stream order and width and the percentage of riparian forest. The second component (PC2) was positively correlated with stream order and negatively correlated with the percentage of forest in the watershed (Table 4). Study sites from each river were segregated in two groups along the PC1 and, excepting A1, study sites of the Atacames river had higher scores than sites of the Súa river (Fig. S2). On the contrary, there was no segregation of the study sites of each river along the PC2.
The storage and composition of the benthic CPOM showed low correlations with the two principal components identified in the PCA (Table 5). In the dry season, only the storage and the percentage of the debris category were inversely correlated with the PC1, which indicated that both diminished as stream width and order and the percentage of riparian forest increased. In the wet season, only the storage of leaves was correlated with the PC1, which indicated that it diminished as the stream size and the percentage of riparian forest increased. The storage of leaves, leaves of Ficus sp1 and leaves of non-cultivated species and the percentages of Ficus sp1 and non-cultivated species in the leaf stock were correlated with the PC2 during the dry season, but only the percentages of Ficus sp1 and non-cultivated species in the leaf stock were significantly correlated with the PC2 during the wet season. These correlations indicated that the storage of leaves, leaves of Ficus sp1 and leaves of non-cultivated species and the percentages of Ficus sp1 and non-cultivated species in the leaf stock increased as stream order increased and the percentage of forest in the watershed diminished.
Discussion
Mean benthic CPOM stocks in this study, between 23.3 and 142.6 g m−2, were like those observed in streams of the Colombian Chocó (between 9.4 and 110.9 g m−2; Valencia et al., 2009). They were also like values observed in other tropical streams elsewhere (between 6.9 and 281 g m−2; Dobson et al., 2002; Colón-Gaud et al., 2008; Li & Dudgeon, 2008; Tonin et al., 2017). It seems that there is a higher variability in the benthic CPOM stocks of temperate rivers (as an example, CPOM data compiled by Jones (1997) showed a variation between 40 and 970 g m−2), which could be a result of the higher seasonality of leaf litter inputs to streams draining temperate deciduous forests that leads to high CPOM accumulations in autumn (Delong & Brusven, 1993; Molinero & Pozo, 2004) or a consequence of having less observations on the benthic CPOM of tropical rivers and streams.
Temporal variability of benthic CPOM stocks is controlled by the timing of inputs from the riparian vegetation and discharge variability (Molinero & Pozo, 2004; Tonin et al., 2017). In the studied streams, rainfall seasonality controls the annual cycle of the benthic CPOM, with peaks at the end of the dry season (September–November) and minimums along the wet season (January–May), as observed in other tropical streams (Ríos-Touma et al., 2009; Bambi et al., 2017). Obstacles such as boulders and branches enhance CPOM retention in the channel (Díez et al., 2000; Larrañaga et al., 2003), but the studied streams have uniform streambeds composed of gravel and sand (Montaño, 2018) and low densities of woody debris (Cuenca, 2018). In these two streams, benthic CPOM rests on the streambed during the dry season and the lack of retention structures favors the downstream transport of CPOM at the beginning of the wet season.
Leaves and woody materials make the most abundant fraction of the benthic CPOM in temperate rivers (Delong & Brusven, 1993; Molinero & Pozo, 2004), but debris was the most abundant category in the studied streams. The high abundance of the debris category suggests a high CPOM decomposition rate, which might be a consequence of high water temperatures (23–28°C, unpublished data) because temperature is one of the main factors that regulate CPOM decomposition (Martínez et al., 2014; Follstad et al., 2017). In low-latitude streams, leaf litter is more recalcitrant than in temperate streams (Boyero et al., 2017) and its decomposition is mostly mediated by microorganisms due to the scarcity of shredders (Boyero et al., 2016). Shredder action converts CPOM to fine particulate matter, so the benthic accumulations of debris could be an indication of low shredder density or activity. In addition to the lower contribution of leaves to the benthic CPOM stock, leaves rarely formed leaf packs in the studied streams. Leaf packs provide both food and refuge to benthic macroinvertebrates (Richardson, 1992) and the lack of suitable habitats could have led to low shredder densities in these streams.
A high number of species contributed to the benthic CPOM in the studied streams, as observed in other tropical streams (França et al., 2009; Valencia et al., 2009; Gonçalves et al., 2014; Lisboa et al., 2015), but two species, the “chíparo” (Zygia longifolia) and Ficus sp1, represented up to 75% of the leaf stock in the benthos. This dominance of a few riparian species in the CPOM pool is usually observed in temperate streams (Webster et al., 1990; Molinero & Pozo, 2006). Functioning of forest streams is linked to the composition of riparian vegetation as it determines timing, composition and quantity of CPOM inputs and benthic CPOM, and ultimately CPOM breakdown rates and usage by shredders (Cummins et al. 1989). More recently, the effect of leaf mixtures on leaf decomposition rates has been addressed with variable results. Decomposition of some species is independent of being alone or in a mixture with others, while other species have shown additive and synergistic effects (Lecerf et al., 2007; Santschi et al., 2018). However, it is still premature to drive any conclusion about the functioning of the studied streams because these experiments use leaf mixtures that do not resemble the high diversity observed in the benthic CPOM pool of tropical streams.
Channel width and stream order also determines CPOM inputs and the amount of benthic CPOM in streams (Delong & Brusven, 1993, 1994; González & Pozo, 1996), but there was no a clear CPOM storage longitudinal gradient along the studied streams. The presence of Ficus sp1 on the banks seemed to have a big impact on the spatial distribution of benthic CPOM. Ficus sp1 produces leaves that are thick and heavy, up to 30 cm length and easily retained in the benthos. The correlation between the benthic leaf stock and the PC2 suggest that the amount of leaves in the benthos was greater in study sites of higher order with low forest cover in the associated watershed. However, this correlation is driven by the presence of Ficus sp1 leaves on the benthos, which was more abundant on the lower reaches of these streams. So, species composition of the riparian vegetation in the studied streams was more important to determine CPOM storage than in temperate streams. Many tropical tree species produce gigantic leaves and the presence or absence of these species might be an important driver of spatial differences in the benthic CPOM storage of tropical streams.
Land use changes, especially those that modify the density and composition of the riparian vegetation, modify benthic CPOM quantity and quality in fluvial systems (Delong & Brusven, 1993; Molinero & Pozo, 2004). However, there were neither significant differences between the two streams in benthic CPOM storage nor a strong relationship between benthic CPOM storage and the percentage of forest in the watershed or in the riparian area. A low-intensity arboriculture is common in the study area. Productive species are mixed with riparian species and with other non-productive species that are grown to provide shadow or enhance the quality of soils, which seems to be enough to maintain similar benthic CPOM stocks in forested and agriculturally impacted streams. Also, the composition of the benthic leaf pool is a better indicator of riparian alteration in the Atacames stream than the quantity and timing of benthic CPOM.
References
Arizona State University, 2018. Neotropical Flora. Arizona State University. https://hasbrouck.asu.edu/neotrop/plantae/index.php. Accessed on: 29 April 2019.
Aufdenkampe, A. K., E. Mayorga, P. A. Raymond, J. M. Melack, S. C. Doney, S. R. Alin, R. E. Aalto & K. Yoo, 2011. Riverine coupling of biogeochemical cycles between land, oceans, and atmosphere. Frontiers in Ecology and the Environment 9: 53–60.
Baldy, V., V. Gobert, F. Guérold, E. Chauvet, D. Lambrigot & J. Charcosset, 2007. Leaf litter breakdown budgets in streams of various trophic status: effects of dissolved inorganic nutrients on microorganisms and invertebrates. Freshwater Biology 52: 1322–1335.
Bambi, P., R. de Souza, M. J. Feio, G. F. M. Leite, E. Alvin, J. M. B. Quintão, F. Araújo & J. F. Gonçalves, 2017. Temporal and spatial patterns in inputs and stock of organic matter in savannah streams of central Brazil. Ecosystems 20: 757–768.
Battin, T. J., L. A. Kaplan, S. Findlay, C. S. Hopkinson, E. Marti, A. I. Packman, J. D. Newbold & F. Sabater, 2008. Biophysical controls on organic carbon fluxes in fluvial networks. Nature Geoscience 1: 95–100.
Boyero, L., R. G. Pearson, C. Hui, M. O. Gessner, J. Pérez, M. A. Alexandrou, M. A. Graça, B. J. Cardinale, R. J. Albarico, M. Arunachalam, L. A. Barmuta, A. J. Boulton, A. Bruder, M. Callisto, E. Chauvet, R. G. Death, D. Dudgeon, A. C. Encalada, V. Ferreira, R. Figueroa, A. Flecker, J. F. Gonçalves, J. Helson, T. Iwata, T. Jinggut, J. Mathooko, C. Mathuriau, C. M’Erimba, M. S. Moretti, C. P. Pringle, A. Ramírez, L. Ratnarajah, J. Rincón & C. M. Yule, 2016. Biotic and abiotic variables influencing plant litter breakdown in streams: a global study. Proceedings of the Royal Society B: Biological Sciences 283: 20152664.
Boyero, L., M. A. Graca, A. M. Tonin, J. Pérez, A. J. Swafford, V. Ferreira, A. Landeira-Dabarca, M. A. Alexandrou, M. O. Gessner, B. G. McKie, R. J. Albariño, L. A. Barmuta, M. Callisto, J. Chará, E. Chauvet, C. Colón-Gaud, D. Dudgeon, A. C. Encalada, R. Figueroa, A. S. Flecker, T. Fleituch, A. Frainer, J. E. Helson, T. Iwata, J. Mathooko, C. M’Erimba, M. Pringle, A. Ramírez, C. M. Swan, C. M. Yule & R. G. Pearson, 2017. Riparian plant litter quality increases with latitude. Scientific Reports 7: 10562.
Carroll, D. & G. R. Jackson, 2009. Observed relationships between urbanization and riparian cover, shredder abundance, and stream leaf litter standing crops. Fundamental and Applied Limnology 173: 213–225.
Ciais, P., A. V. Borges, G. Abril, M. Meybeck, G. Folberth, D. Hauglustaine & I. A. Janssens, 2008. The impact of lateral carbon fluxes on the European carbon balance. Biogeosciences 5: 1259–1271.
Colón-Gaud, C., S. Peterson, M. R. Whiles, S. S. Kilham, K. R. Lips & C. M. Pringle, 2008. Allochthonous litter inputs, organic matter standing stocks, and organic seaton dynamics in upland Panamanian streams: potential effects of larval amphibians on organic matter dynamics. Hydrobiologia 603: 301–312.
Cuenca, C., 2018. Comparación de la acumulación de detritos leñosos en la cuenca de los ríos Atacames y Súa: influencia del uso de cambio de suelo. Graduation Thesis. Pontificia Universidad Católica del Ecuador Sede Esmeraldas, Esmeraldas, Ecuador.
Cummins, K. W., M. A. Wilzbach, D. M. Gates, J. B. Perry & W. B. Taliaferro, 1989. Shredders and riparian vegetation. BioScience 39: 24–30.
Delong, M. D. & M. A. Brusven, 1993. Storage and decomposition of particulate organic matter along the longitudinal gradient of an agriculturally-impacted stream. Hydrobiologia 262: 77–88.
Delong, M. D. & M. A. Brusven, 1994. Allochthonous input of organic matter from different riparian habitats of an agriculturally impacted stream. Environmental Management 18: 59–71.
Dobson, M., A. Magana, J. M. Mathooko & F. K. Ndegwa, 2002. Detritivores in Kenyan highland streams: more evidence for the paucity of shredders in the tropics? Freshwater Biology 47: 909–919.
Díez, J. R., S. Larrañaga, A. Elosegi & J. Pozo, 2000. Effect of removal of wood on streambed stability and retention of organic matter. Journal of The North American Benthological Society 19: 621–632.
Epstein, D. M., J. E. Kelso & M. A. Baker, 2016. Beyond the urban stream syndrome: organic matter budget for diagnostics and restoration of an impaired urban river. Urban Ecosystems 19: 1041–1061.
Follstad, J. J., J. S. Kominoski, M. Ardyn, W. K. Dodds, M. O. Gessner, N. A. Griffiths, C. P. Hawkins, S. L. Johnson, A. Lecerf, C. J. LeRoy, D. W. P. Manning, A. D. Rosemond, R. L. Sinsabaugh, C. M. Swan, J. R. Webster & L. H. Zeglin, 2017. Global synthesis of the temperature sensitivity of leaf litter breakdown in streams and rivers. Global Change Biology 23: 3064–3075.
França, J. S., R. S. Gregório, J. D. de Paula, J. F. Gonçalves, F. A. Ferreira & M. Callisto, 2009. Composition and dynamics of allochthonous organic matter inputs and benthic stock in a Brazilian stream. Marine and Freshwater Research 60: 990–998.
GADMA, 2014. Plan de ordenamiento y desarrollo territorial del cantón Atacames 2014-2019. Gobierno Autónomo Descentralizado Municipal de Atacames, Esmeraldas.
GADPS, 2015. Plan de ordenamiento y desarrollo territorial de la parroquia de Súa 2015. Gobierno Autónomo Descentralizado Parroquial de Súa, Esmeraldas.
Gonçalves, J. F. & M. Callisto, 2013. Organic-matter dynamics in the riparian zone of a tropical headwater stream in Southern Brasil. Aquatic Botany 109: 8–13.
Gonçalves, J. F., J. S. Franca & M. Callisto, 2006. Dynamics of allochthonous organic matter in a tropical Brazilian headstream. Brazilian Archives of Biology and Technology 49: 967–973.
Gonçalves, J. F., R. de Souza, R. S. Gregório & G. C. Valentin, 2014. Relationship between dynamics of litterfall and riparian plant species in a tropical stream. Limnologica 44: 40–48.
González, E. & J. Pozo, 1996. Longitudinal and temporal patterns of benthic coarse particulate organic matter in the Agüera stream (northern Spain). Aquatic Sciences 58: 355–366.
Greenacre, M. & R. Primicerio, 2014. Multivariate analysis of ecological data. Fundación BBVA, Bilbao.
Haggerty, S., D. Batzer & C. Jackson, 2004. Macroinvertebrate response to logging in coastal headwater streams of Washington, USA. Canadian Journal of Fisheries and Aquatic Sciences 61: 529–537.
Jones, J. B., 1997. Benthic organic matter storage in streams: influence of detrital import and export, retention mechanisms, and climate. Journal of The North American Benthological Society 16: 109–119.
Karlsson, O. M., J. S. Richardson & P. M. Kiffney, 2005. Modelling organic matter dynamics in headwater streams of south-western British Columbia, Canada. Ecological Modelling 183: 463–476.
Larrañaga, S., J. R. Diez, A. Elosegi & J. Pozo, 2003. Leaf retention in streams of the Agüera basin (northern Spain). Aquatic Sciences 65: 158–166.
Lecerf, A., G. Risnoveanu, C. Popescu, M. O. Gessner & E. Chauvet, 2007. Decomposition of diverse litter mixtures in streams. Ecology 88: 219–227.
Li, A. O. & D. Dudgeon, 2008. Food resources of shredders and other benthic macroinvertebrates in relation to shading conditions in tropical Hong Kong streams. Freshwater Biology 53: 2011–2025.
Lisboa, L. K., A. L. L. da Silva, A. E. Siegloch, J. F. Gonçalves & M. M. Petrucio, 2015. Temporal dynamics of allochthonous coarse particulate organic matter in a subtropical Atlantic rainforest Brazilian stream. Marine and Freshwater Research 66: 674–680.
Little, E. L. & R. G. Dixon, 1983. Arboles comunes de la provincia de Esmeraldas. Naciones Unidas, Roma. https://babel.hathitrust.org/cgi/pt?id=txu.059173004452938. Accessed on 29 April 2019.
MAE, 2005. Plan de manejo y gestión participativa de la reserva ecológica Mache-Chindul. Ministerio del Medio Ambiente, Quito.
MAGAP, 2006. Mapa de usos y cobertura de la tierra a escala 1:250000. Ministerio de Agricultura, Ganadería, Acuicultura y Pesca, Quito.
MAG, 2016. Mapa de cobertura de uso del suelo a escala provincial de los años 2013-14. Ministerio de Agricultura y Ganadería, Quito.
Martínez, A., A. Larrañaga, J. Pérez, E. Descals & J. Pozo, 2014. Temperature affects leaf litter decomposition in low-order forest streams: field and microcosm approaches. FEMS Microbiology Ecology 87: 257–267.
Missouri Botanical Garden, 2018. Trópicos. http://www.tropicos.org/. Accessed on 29 April 2019.
Molinero, J. & J. Pozo, 2004. Impact of a eucalyptus (Eucalyptus globulus Labill.) plantation on the nutrient content and dynamics of coarse particulate organic matter (CPOM) in a small stream. Hydrobiologia 528: 143–165.
Molinero, J. & J. Pozo, 2006. Organic matter, nitrogen and phosphorus fluxes associated with leaf litter in two small streams with different riparian vegetation: a budget approach. Archiv für Hydrobiologie 166: 363–385.
Montaño, H., 2018. Comparación de características geomorfológicas de las cuencas del río Atacames y Súa. Graduation Thesis. Pontificia Universidad Católica del Ecuador Sede Esmeraldas, Esmeraldas, Ecuador.
Palacios, W. A., 2011. Arboles del Ecuador. Ministerio del Medio Ambiente, Quito.
Peres-Neto, P. R., D. A. Jackson & K. M. Somers, 2003. Giving meaningful interpretation to ordination axes: assessing loading significance in principal component analysis. Ecology 84: 2347–2363.
Pozo, J., E. González, J. Díez, J. Molinero & A. Elósegui, 1997. Inputs of particulate organic matter to streams with different riparian vegetation. Journal of The North American Benthological Society 16: 602–611.
Pozo, J., A. Elosegi, J. Díez & J. Molinero, 2009. Dinámica y relevancia de la materia orgánica. In Elosegi, A. & S. Sabater, Conceptos y técnicas en ecología fluvial. Fundación BBVA, Barcelona: 141–148.
R Development Core Team, 2015. R: A Language and Environment for Statistical Computing. Vienna, Austria. http://www.R-project.org.
Reid, D., P. Lake, G. Quinn & P. Reich, 2008. Association of reduced riparian vegetation cover in agricultural landscapes with coarse detritus dynamics in lowland streams. Marine and Freshwater Research 59: 998–1014.
Rezende, R. S., M. A. Sales, F. Hurbath, N. Roque, J. F. Gonçalves & A. O. Medeiros, 2017. Effect of plant richness on the dynamics of coarse particulate organic matter in a Brazilian Savannah stream. Limnologica 63: 57–64.
Richardson, J., 1992. Food, microhabitat, or both? Macroinvertebrate use of leaf accumulations in a montane stream. Freshwater Biology 27: 169–176.
Ríos-Touma, B., A. C. Encalada & N. P. Fornells, 2009. Leaf litter dynamics and its use by invertebrates in a high-altitude tropical Andean stream. International Review of Hydrobiology 94: 357–371.
Santiago, J., J. Molinero & J. Pozo, 2011. Impact of timber harvesting on litterfall inputs and benthic coarse particulate organic matter (CPOM) storage in a small stream draining a eucalyptus plantation. Forest Ecology and Management 262: 1146–1156.
Santschi, F., I. Gounand, E. Harvey & F. Altermatt, 2018. Leaf litter diversity and structure of microbial decomposer communities modulate litter decomposition in aquatic systems. Functional Ecology 32: 522–532.
Stackpoole, S. M., D. Butman, D. W. Clow, C. P. McDonald, E. G. Stets & R. G. Striegl, 2012. Baseline carbon sequestration, transport, and emission from inland aquatic ecosystems in the Western United States. In Zhu, Z. & B. C. Reed (eds), Baseline and projected future carbon storage and greenhouse-gas fluxes in ecosystems of the Western United States, U.S. Geological Survey, Reston, Virginia: 1–18.
The Field Museum, 2018. Tropical plant guides. https://plantidtools.fieldmuseum.org/. Accessed on 29 April 2019.
Tonin, A. M., J. F. Gonçalves, P. Bambi, S. R. Couceiro, L. A. Feitoza, L. E. Fontana, N. Hamada, L. U. Hepp, V. G. Lezan-Kowalczuk, G. F. Leite, A. L. Lemes-Silva, L. K. Lisboa, R. C. Loureiro, R. T. Martins, A. O. Medeiros, P. B. Morais, Y. Moretto, P. C. A. Oliveira, E. B. Pereira, L. P. Ferreira, J. Pérez, M. M. Petrucio, D. F. Reis, R. S. Rezende, N. Roque, L. E. P. Santos, A. E. Siegloch, G. Tonello & L. Boyero, 2017. Plant litter dynamics in the forest-stream interface: precipitation is a major control across tropical biomes. Scientific Reports 7: 10799.
Valencia, S. M., G. A. Pérez, P. X. Lizarazo & J. F. Blanco, 2009. Patrones espacio-temporales de la estructura y composición de la hojarasca en las quebradas del Parque Nacional Natural Gorgona. Actualidades Biológicas 31: 197–211.
Vanegas, R. M., 2016. Disponibilidad de materia orgánica bentónica y su potencial relación con la estructura física de cauces en microcuencas prioritarias del cantón Loja. Universidad Nacional de Loja, Loja.
Wallace, J. B., S. L. Eggert, J. L. Meyer & J. R. Webster, 1997. Multiple trophic levels of a forest stream linked to terrestrial litter inputs. Science 277: 102–104.
Warton, D. I. & F. K. Hui, 2011. The arcsine is asinine: the analysis of proportions in ecology. Ecology 92: 3–10.
Webster, J., S. Golladay, E. Benfield, D. D’Angelo & G. Peters, 1990. Effects of forest disturbance on particulate organic matter budgets of small streams. Journal of The North American Benthological Society 9: 120–140.
Webster, J., A. Covich, J. Tank & T. Crockett, 1994. Retention of coarse organic particles in streams in the southern Appalachian Mountains. Journal of The North American Benthological Society 13: 140–150.
Woodward, G., M. O. Gessner, P. S. Giller, V. Gulis, S. Hladyz, A. Lecerf, B. Malmqvist, B. G. McKie, S. D. Tiegs, H. Cariss, M. Dobson, A. Elosegi, V. Ferreira, M. A. S. Graça, T. Fleituch, J. O. Lacoursière, M. Nistorescu, J. Pozo, G. Risnoveanu, M. Schindler, A. Vandieanu, L. B. M. Vought & E. Chauvet, 2012. Continental-scale effects of nutrient pollution on stream ecosystem functioning. Science 336: 1438–1440.
Acknowledgements
This work was financed by the 2016 research call of the Pontificia Universidad Católica del Ecuador Sede Esmeraldas. The author wants to thank N. Jijón and P. Jiménez for their assistance in the field and laboratory work and F. Quiroz for helping with the identification of the morphotypes to genus and species. The Instituto Geográfico Militar of Ecuador (IGM) provided 1:50,000 cartography of the study area. The Instituto Nacional de Meteorología e Hidrología (INAMHI) and the Coronel Carlos Concha Torres airport provided meteorological and hydrological data at the city of Esmeraldas. The author also thanks meteorologist E. Bastidas from the Instituto Nacional Oceanográfico de la Armada (INOCAR) for providing data from the meteorological station at Esmeraldas harbor. J. Molinero is a member of the IBEPECOR network (http://www.ibepecor.csic.es/).
Data availability
The data that support the findings of this study are available from the corresponding author J. Molinero upon reasonable request.
Funding
This study was funded by the Pontificia Universidad Católica del Ecuador Sede Esmeraldas.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that he has no conflict of interests.
Additional information
Handling editor: Verónica Ferreira
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Molinero, J. Seasonality and composition of benthic coarse particulate organic matter in two coastal tropical streams with different land uses. Hydrobiologia 838, 29–43 (2019). https://doi.org/10.1007/s10750-019-03974-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-019-03974-8