Abstract
Strobel Plateau hosts more than 1,500 shallow lakes displaying different water regimes, which constitute the habitat for many species. Although the region is naturally fishless, many of the lakes were stocked with trout, bearing uncertainty about the possible effects on the ecosystem. The main objective of this study was to analyze the characteristics of planktonic autotrophic communities of lakes differing in regime (phytoplankton turbid, clear vegetated, and clear unvegetated) and presence/absence of fish. During late spring and summer, 14 water bodies were sampled in 2011 and 2013 considering different regimes and presence/absence of fish. Besides limnological variables, the autotrophic communities, from pico to microplankton, were also analyzed. Differences in physical and chemical characteristics observed among the lakes corresponded to their current regime and the presence/absence of trouts. Autotrophic picoplankton and phytoplankton > 20 µm abundances differed among lake types being highest in fish-stocked lakes. Although the three type of lakes presented phycoerythrin-rich picocyanobacteria and picoeukaryotes, only fish-stocked lakes hosted phycocyanin-rich picocyanobacteria. Moreover, fish-stocked lakes were dominated by cyanobacteria, while chlorophytes abounded in fishless systems. Evidences that lake regime and fish occurrence influence planktonic autotrophic communities of Strobel plateau is here provided, highlighting the intra- and interannual dynamism of the aquatic systems.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Phytoplankton from Patagonian lakes has been addressed in several ecological and taxonomical studies encompassing different types of aquatic environments (e.g., Thomasson, 1959, 1963; Izaguirre et al., 1990; Díaz et al., 2000; Queimaliños & Díaz, 2014 and cites therein). The lakes from Patagonian plateau have received comparatively less attention than those in the Andean region, probably due to the remoteness of the environments, which in some cases are of difficult accessibility (Izaguirre & Saad, 2014).
Wetlands (shallow lakes, “mallines” and peat-bogs) covering almost 5% of Patagonian territory represent essential habitats to sustain biodiversity and wildlife in the extra-andean and the Patagonian desert (Perotti et al., 2005). These environments are threatened for multiples causes (Sala et al., 2000; Brinson & Malvárez, 2002), and therefore the need to enhance the knowledge of the functioning and ecological state of water bodies in this region is paramount.
Most lakes of the arid Patagonia are located in basaltic plateaus formed by tectonic processes and subsequent deflations of the basaltic mantle (Pereyra et al., 2002). These lakes present well and deeply mixed water columns due to the incidence of strong winds, and due to this permanent mixing are currently considered shallow from a dynamic point of view (Lancelotti, 2009). The first limnological characterization of these water bodies and their contextualization in the frame of stable alternative states model (Scheffer et al., 1993) was also made by Lancelotti (2009).
According to this widely known model, shallow lakes may have two equilibrium states, one of them “clear” with high water transparency, low phytoplankton biomass, and dominance of submerged macrophytes, and another “turbid” with low water transparency, high biomass of phytoplankton, and without submerged macrophytes. These two states were proposed in this model as “alternative states,” since they exhibit homeostatic tendencies due to positive feedbacks. Increasing nutrients in shallow water bodies drive to phytoplankton biomass enhancement, with a consequent decrease in water transparency. In a turbid state, the penetration of light limits the development of submerged macrophytes, which in turn generates wind-driven sediment resuspension, thus increasing turbidity. In the opposite scenario, under great development of submerged macrophytes, resuspension is lessened and sedimentation is favored. On the other hand, some macrophytes may produce allelopathic substances that might limit the development of phytoplankton and also provide shelter for zooplankton that preys on phytoplankton (Schallenberg & Sorrell, 2009 and citations therein). Despite that these two states can coexist in a wide range of nutrient contents, certain disturbances can trigger the rapid passage from one state to another. Since it is not always possible to establish the occurrence of alternative equilibrium states in natural lakes (i.e., registering the change from one state to another over time), the term “regime” is now currently used (Scheffer et al., 2009) and, thus, sudden changes between the above-mentioned lake conditions can be considered as shift regimes.
The food web structure in aquatic systems is determined primarily by controls “top-down” and “bottom-up,” where the regulation of a trophic level occurs through predation by a higher level and by nutrient availability for primary producers, respectively. The theory of trophic cascades is focused on the interactions predator–prey as drivers of changes in biomass or productivity of aquatic communities (Pace et al., 1999), whereas the bottom-up controls are those that define the potential productivity of a lake; changes in this potential are due to top-down interactions. Thus, trophic cascades can explain differences in productivity in lakes with equal nutrient input (Carpenter et al., 1985). In this sense, the introduction of omnivorous fish in shallow water environments is associated with direct and indirect effects on aquatic communities: on one hand, predation on zooplankton can generate an intense effect top-down that fosters the development of phytoplankton (Blanco et al., 2008), and on the other hand, feeding on benthic organisms generates a sediment removal that causes resuspension of nutrients (i.e., bottom-up effect which also promotes the development of phytoplankton) (Schaus et al., 2002). Both processes promote the shift and/or permanence in turbid state of water bodies (Blanco, 2001).
Introductions of species, both of flora and fauna, have been common practices for more than a century in Patagonia. The introduction of salmonids and also the translocation of native fish species have been reported for many water systems (Perotti et al., 2005). Particularly, in naturally fishless lakes from the Strobel Plateau (Santa Cruz), the introduction of rainbow trout (Oncorhynchus mykiss Walbaum) began around 1940, but over the last 20 years there has been an increase in this activity as a productive alternative to the traditional sheep raising (Lancelotti et al., 2009).
Comparative studies of fishless and stocked lakes of the Lake Strobel plateau showed significant changes in the community structure and zooplankton size spectrum due to trout predation (Lancelotti et al., 2015), suggesting that these changes could be projected over the lower trophic levels. However, there is uncertainty about the occurrence of such changes in the affected lakes. Preliminary studies suggested that fish introduction in the area was associated to changes in micro- and nano-phytoplankton (Izaguirre & Saad, 2014). This evidence needs to be validated for a larger number of lakes with different regimes and further exploring the effect of fish stocking on picoplankton. In this context, the numerous and diverse aquatic environments of the Strobel plateau, where different degrees of impact of introduced trout are recorded, represent an extraordinary scenario to analyze changes at community and ecosystem level due to the introduction of a top predator under different lake scenarios.
The main objective of this study was to analyze comparatively the planktonic autotrophic communities among lakes of Strobel Plateau differing in regime (clear vegetated, clear unvegetated, and phytoplankton turbid) and presence/absence of introduced fish. The working hypothesis of this study is that both fish introduction (through trophic cascade) and lake regime (mainly determined by macrophyte cover) influence the abundance and composition of planktonic autotrophic communities of the different size fractions (micro-, nano-, and picoplankton).
Methods
Study area
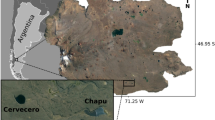
Lake Strobel Plateau is located in the center-west of the Province of Santa Cruz (Argentina). From the geological prospect, it consists of a bed of alkaline basalt with a 50 m thickness that was caused by tectonic activity during the Tertiary and Quaternary (Panza & Franchi, 2002). The plateau has a flat surface of 3,000 km2 with a slight slope from west to east, reaching altitudes of 1,200 m in the west and 700 m in the east. The natural depressions that receive water from melting winter snow form a system of shallow lakes, many of them temporary. The number of water bodies reaches up to 2,000 during wet years, and approximately 700 correspond to permanent lakes (Lancelotti, 2009).
The region has a cold and dry weather, with average maximum temperatures of 2.9 and 14°C in winter and summer, respectively, and annual rainfalls ranging between 200 and 400 mm spread throughout the year, being in form of snow in winter. The region is windy, with prevailing winds from the west with greater intensity in summer than in winter. This area is characterized by a high cloudiness with an average of 60% overcast sky. The weather information was taken from the station located in Gobernador Gregores, which is the closest city to the Strobel Plateau (40 km southeastern boundary of the plateau). Given the inclement weather, the plateau remains covered by snow and ice from late April to mid-November (Lancelotti, 2009).
Lancelotti et al. (2009) originally classified the water bodies of this region based on their limnological, topographical, and geographical variables, differentiating among vegetated (with submerged macrophytes), unvegetated, and turbid lakes. In vegetated water bodies, the coverage of the dominant submerged macrophyte Myriophyllum quitense Kunth shows great seasonal variation, reaching in some cases 95% coverage of the total area of the lake.
Although the water bodies of the region are considered shallow, there exists a range of lake depths that were classified by Izaguirre et al. (under review), following the criteria of Abell et al. (2012) in shallow (maximum depth < 10 m) and deep lakes (maximum depth > 10 m), including very shallow lakes (maximum depth < 1.5 m) as an additional category.
The Strobel plateau was recognized as priority area of waterbird conservation in Argentina (Imberti, 2005), representing the primary foraging and reproductive habitat for numerous species, including several endemisms and threatened species. In particular, this plateau has historically represented the core of the Hooded grebe (Podiceps gallardoi Rumboll) distribution, which was categorized as critically endangered species (IUCN Red List of Threatened Species). The effects derived from trout introduction have been proposed as one of the main causes of the critical situation of the Hooded grebe.
For this study, 14 water bodies were sampled considering differences in their regimes and the presence/absence of fish: fishless lakes including clear-unvegetated (Rod 18 and Pif) and clear-vegetated (Justita, Las Coloradas, S94, S64, Nieve) water bodies, and fish-stocked lakes referred as phytoplankton-turbid systems (Herradura, 9, 8, Temp, Campamento, Potrerillo, Verde). The geographical position of water bodies was obtained with GPS (positioning system geographical, Garmin, KS, USA) and it is shown in Fig. 1. All stocked lakes analyzed in this study were originally fishless, and stocked with Rainbow trout before 2001 (but lake Temp in 2006). Lakes were stocked with 60 alevins/0.01 km2. However, the rate of stocking and removal of trout differed among lakes.
Two sampling campaigns were conducted, one during December 2011 and the other during February and March 2013 (late spring and summer, respectively). All phytoplankton-turbid lakes and clear-vegetated lakes were sampled on both occasions. Clear-unvegetated lakes were sampled only in 2013 due to logistical and climatic difficulties.
Physical and chemical analyses
Samples and measurements were obtained from the pelagic zone at the upper water layer, integrating waters from the surface to 50 cm. Temperature, pH, and conductivity were measured using a Horiba D-54 sensor, dissolved oxygen (DO) with a HI 9146 Hanna Hatch device or a HQ30d portable meter. The photosynthetically active radiation (PAR) was also measured in situ and at 10-cm depth intervals with a LI-COR radiometer equipped with a submersible spherical quantum sensor (Li-Cor 3 SA Li-Cor PAR, NE, USA). The vertical attenuation coefficient of PAR (Kd) was calculated as the slope of the relationship between ln (irradiance) and depth (Kirk, 1994).
Samples for nutrient analysis, chlorophyll a (Chl a), and dissolved organic carbon (DOC) were transported refrigerated and filtered through Whatman GF/F filters upon returning from field to avoid deterioration. Dissolved nutrient analysis (nitrate, ammonium, and soluble reactive phosphorus—SRP) was performed on the filtered water with a Hach TMDR/2800 spectrophotometer and its corresponding reagent kits (detection limit for all nutrients 0.01 mg l−1). Dissolved inorganic nitrogen (DIN) was obtained from the sum of the concentrations of nitrate + nitrite and ammonium. The concentration of Chl a (corrected by pheopigments) was estimated from the material retained in the filter, which was preserved at − 20°C and darkness until its processing. Chl a extractions were performed using acetone as solvent; filters were stored at 4°C overnight and then concentrations of Chl a were determined by spectrophotometry at 665 and 750 nm absorbances. The correction by phaeopigments was performed by the addition of 1 N HCl, and the equations given by Marker et al. (1980) were used. DOC was determined from filtered and acidified water (pH < 2) with sulfuric acid and using the high temperature Pt catalytic oxidation method (Shimadzu 5000A TOC analyzer) following the recommendations of Sharp et al. (1993); determinations were carried out by the technicians of the National Water Institute (INA) (Buenos Aires, Argentina).
Phytoplankton analysis
Samples for the qualitative analysis of phytoplankton (nano- and microplankton fractions) were taken with a 15 μm pore net, in order to concentrate the organisms and were preserved with 2–3% formaldehyde. The observation was performed under binocular optical microscope (Olympus CX31) at ×1000 magnification, and identifications were carried out following specialized literature for each group of algae. Samples for quantitative phytoplankton analysis were preserved with a 1% acidified Lugol solution. Phytoplankton counts were performed using an inverted Olympus CKX41 microscope following the technique of Utermöhl (1958), and counting error was estimated according to Venrick (1978). In all cases, the individual algae were considered as units (unicellular, colony, conglomerate, or filament). In order to characterize the functional groups in the phytoplankton communities, we classified the entire list of phytoplankton species following the classification of Reynolds et al. (2002) updated by Padisák et al. (2009).
The autotrophic picoplankton was quantified through the use of flow cytometry technique. For this, the samples of each water body were fixed with 10% cold glutaraldehyde to a final concentration of 1% and stored in cryovials in liquid nitrogen (during the campaigns) and then at − 80°C. To the thawed fixed samples, a known volume was added with a suspension of beads (Fluospheres ® carboxylate microspheres of 1 μm diameter = beads, yellow-green fluorescent 505/515, Molecular Probes) and then analyzed with the flow cytometer FACSAria II, which is equipped with a solid state blue laser (488 nm > 20mW) and a red laser of Gaseous state (633 nm > 18mW). For the analysis of the populations, four parameters were used: FL2 (PE, 488 nm excitation, 585/42 nm emission BP), FL3 (PERCP, 488 nm excitation, 670 nm LP emission), FL4 (APC 635 nm excitation, 661/61 nm BP emission) and SSC (light scattered by the particle at an angle of 90°). The samples were generally run for 300 s and with the information of the set of detectors different cytometric populations were differentiated. Cytometric populations are conformed by single cells that express the same combination of parameters (SSC, PERCP, APC, and PE), but not necessarily conform a homogeneous taxonomical group; they share similar size/shape and pigment composition, thus conferring similar ecological properties. Data analysis was carried out with the FlowJo program (Tree Star).
Statistical analyses
In order to corroborate the homogeneity of the group of lakes, a discriminant analysis (DA) was performed, followed by multivariate analysis of variance (MANOVA), including DIN, SRP, Conductivity, pH, DOC, and Chla as proxies of the trophic state of the lakes. A logarithmic transformation was applied to the variables to meet the assumptions of multivariate normality and equality of covariance.
In order to evaluate the differences between the abundances of the autotrophic communities among the different types of lakes, a Kruskall–Wallis non-parametric variance analysis was performed. All of these analyses were carried out with IBM SPSS Statistics 20 software.
Results
The surveyed shallow lakes from Strobel Plateau here classified according to their different regimes and to the presence or absence of trouts showed differences both in the physical and chemical characteristics (Table 1), and in the structure of their autotrophic planktonic communities. The discriminant analysis based on physical and chemical variables allowed the ordination of the water bodies according to the proposed classification (Fig. 2), thus corroborating the homogeneity of the established groups: Axis 1 explained 64.3% of the variability with Conductivity, DOC, and DIN as the main variables; Axis 2 explained 21.1% mainly associated to Conductivity and SRP. The cross-classification analysis showed a correct classification of the lakes with an error of 4.17%. The multivariate analysis of variance evidenced that lakes in each sampling campaign differed significantly (Table 2). In 2011, clear-vegetated and phytoplankton-turbid lakes differed from each other, whereas in 2013 clear-unvegetated lakes differed from the former groups.
Biplot of discriminant analysis (DA) based on physical and chemical variables from lakes of Strobel Plateau: DIN dissolved inorganic nitrogen, SRP soluble reactive phosphorus, DOC dissolved organic carbon, Chl a chlorophyll a, pH and Cond conductivity. Natural logarithm transformation was conducted in the variables. Lakes were classified according to the regime and presence/absence of fish. Lines indicate the loading of each variable on the two first axes
Clear-vegetated lakes did not differ significantly between the two sampling dates, and showed intermediate values of Kd, DOC, and Chl a. By contrast, samples of fish-stocked lakes (phytoplankton-turbid) of both years ordinated towards higher Chl a values, and differed between 2011 and 2013, displaying a decline in Kd, Chl a, and DOC values due to higher water level (as a consequence of the occurrence of a dilution process) in most of these shallow lakes. Clear-unvegetated lakes showed the highest values of Chl a and smallest Kd and DOC values with respect to the other two groups of lakes in 2013. Regarding dissolved nutrients, clear-unvegetated lakes displayed the lowest concentrations of DIN and the highest SRP values of the dataset (Table 1). From 2011 to 2013, SRP and DIN values decreased in clear-vegetated and phytoplankton-turbid lakes, with the exception of DIN in phytoplankton-turbid that increased in this period.
Autotrophic picoplankton
Autotrophic picoplankton abundances differed significantly among lake types (H = 7.08; P = 0.03). Phytoplankton-turbid lakes had on average higher abundances of picoalgae (8.4 × 105 ind ml−1) than clear-vegetated water bodies (5.5 × 104 ind ml−1). Clear-unvegetated lakes, with intermediate abundances (3.5 × 105 ind ml−1), did not differ from the other types of lakes (Fig. 3A).
Picoplankton abundance (A) and composition (B) in the three types of studied lakes from Strobel Plateau. Different letters indicate significant differences (P < 0.05) among lake types. Cyano cyanobacteria; Peuk. picoeukaryotes, PC-rich Pcy phycocyanin-rich picocyanobacteria, PE-rich Pcy phycoerythrin-rich picocyanobacteria. Error bars indicate standard error
The analysis of the cytometric populations revealed differences in the composition of the groups of autotrophic picoplankton among the types of shallow lakes (Fig. 3B). The three types of lakes presented picoeukaryotes and picocyanobacteria populations, and also showed the presence of cyanobacterial microcolonies. Both clear type lakes presented picoeukaryotes and phycoerythrin-rich picocyanobacteria; a small population of colonial cyanobacteria was also observed in a vegetated shallow lake in 2013. Phytoplankton-turbid lakes showed phycocyanin-rich picocyanobacteria besides the mentioned populations; colonial cyanobacterial microcolonies were also observed in samples of both sampling dates.
When grouping the lakes in fish-stocked and fishless categories, higher abundances of picocyanobacteria (Pcy) were observed in the former lakes, especially phycocyanin rich (PC-rich Pcy), which were completely absent in fishless ones (Fig. 4). Regarding the number of cytometric populations, the picoeukaryotes (Peuk) were the most frequent picoplanktonic algal group in all the studied lakes, usually represented by one or two populations almost in all water bodies, and achieving three populations in Rod 18, a clear-unvegetated lake. Pcy, when present, never achieved more than one cytometric population in fishless lakes and two populations in fish-stocked lakes.
A Abundance and maximum number of cytometric populations of each type of picoplankton group assessed in fish-stocked/fishless lakes from Strobel Plateau. B Representative cytograms of two studied lakes. Picoeuk. picoeukaryotes, PC-rich Pcy phycocyanin-rich picocyanobacteria, PE-rich Pcy, phycoerythrin-rich picocyanobacteria. PerCP-H, green fluorescence (488 nm excitation, 670 nm LP emission), PE-H orange fluorescence (488 nm excitation, 585/42 nm BP emission), APC-H red fluorescence (635 nm excitation, 661/61 nm BP emission)
Phytoplankton
Total phytoplankton abundance differed among lakes with different regimes (H = 6.32, P = 0.04). In clear-vegetated lakes, the mean phytoplankton abundance (1.9 × 103 ind ml−1) was significantly lower than in phytoplankton-turbid lakes (fish-stocked) (1.2 × 104 ind ml−1) (Fig. 5A). In clear-unvegetated lakes, the average phytoplankton density was 5.4 × 103 ind ml−1, with no significant differences with the other types of lakes.
Phytoplankton (> 20 µm) abundance (A) and composition (B) in the three types of studied lakes from Strobel Plateau. Different letters indicate significant differences among lake types. Cyano. cyanobacteria, Bacillario. bacillariophyceae, Crypto. cryptophyceae, Chloro. chlorophyceae, Chryso. chrysophyceae
The composition of phytoplankton groups differed among the studied systems: both clear systems showed a dominance of Chlorophyceae (clear vegetated = 42.6% and clear unvegetated = 68.5%) where Oocystis lacustris Chod., Monoraphidium circinale (Nyg.) Nyg., Chlamydomonas spp., and Botryococcus braunii Kütz. prevailed among others. On the other hand, phytoplankton-turbid lakes had a high proportion of Cyanobacteria in both sampling dates (64.5%), mainly represented by Aphanocapsa delicatissima W. & G. S. West and Aphanothece caldariorum Richt.. In clear-vegetated lakes, the sub-dominant group in terms of abundance was Cryptophyceae (21.8%), with Plagioselmis nannoplanctica (Skuja) Novar., Lucas et Morr., and Cryptomonas marssonii Skuja as the best represented species, whereas in clear-unvegetated water bodies Chrysophyceae (Chromulina sp. forming palmelloid aggregates) thrived (Fig. 5B).
Considering Reynolds functional groups classification, clear differences in the best represented coda were observed between fish-stocked and fishless lakes. Fishless systems showed higher proportion of nanoplanktonic groups, including some typically mixotrophic species; by contrast fish-stocked lakes presented higher proportion of species > 20 µm and contribution of cyanobacteria (Table 3).
Discussion
Our study showed that the three main types of water bodies recognized in the Strobel Plateau differ in their physical and chemical variables, as well as in the composition of their planktonic autotrophic communities; both phytoplankton–macrophyte interaction and fish introduction influenced community structuring.
Shallow lakes in this region are typically stressed by persistent winds that washout nutrients from soil and by resuspension from the bottom, thus conferring trophic states that vary from meso- to eutrophic (sensu Vollenweider & Kerekes, 1982). Notwithstanding, lakes are differentiated by the concentration of Chlorophyll a, dissolved nutrients, and light penetration. These variables are central to the conceptual model of alternative equilibria proposed by Scheffer et al. (1993); nevertheless, it is interesting to discuss to what extent this scheme is appropriate to describe the differences between the studied systems.
Most phytoplankton-turbid lakes here surveyed are connected by the snowmelt Moro stream that has a variable discharge with torrential characteristics, strongly affecting their water level; hence, limnological characteristics and phytoplankton biomass are influenced by these hydrological changes. Accordingly, a recent study involving a great number of shallow lakes from two Patagonian plateaus (Strobel and Buenos Aires) showed that water level is an important factor associated to the shift from clear to turbid states in arheic systems of the region (Izaguirre et al., 2018). Several studies performed in shallow lakes elsewhere (e.g., Blindow et al., 1993; Medina-Sánchez et al., 2004; de Tezanos Pinto & O’Farrell, 2014 and cites therein) also showed that water-level variation constituted an important factor associated with changes in the regime alternation of shallow lakes.
Water-level variation affects macrophyte development in the shallow lakes of the region, as the strong hydrometric decrease evidenced a demise of the community (Izaguirre et al., 2018). Besides, other environmental variables, such as sediment type, nutrient concentration, direction and wind intensity, sediment type (Feldmann & Nõges, 2007), and slope and depth of the basin (Duarte & Kalff, 1986) were also indicated to influence aquatic vegetation. In our study, lake morphometry was the main determinant for macrophyte cover. The lakes of the Strobel Plateau with considerable depth only allowed for the development of macrophytes arranged in rings in the perimeter or outer area of the water body, whereas in shallower ones a much larger surface was prone to colonization, reaching 100% coverage in some of them.
The differences in phytoplankton structure among the lakes seem also to be related with fish introduction, probably due to a top-down effect. In our study, the higher phytoplankton biomass observed in fish-stocked lakes supports this assumption. Likewise, Blanco (2001) showed that fish introduction generates direct or indirect effects on aquatic food webs, leading to an increase in algal growth, higher turbidity, and nutrients, which generates a decrease in the density of submerged macrophytes and epiphytic algae. For the lakes of the Strobel Plateau, previous studies confirmed the effect of trouts on both the structure of species and sizes of zooplankton (Lancelotti et al., 2017; Izaguirre et al., 2018) and consequently on the diet of trout themselves (Lancelotti et al., 2015): feeding trout significantly affected the abundance and composition of the different groups of zooplankton. The decrease in biomass and individual size of zooplankton registered (Lancelotti et al. op cit) is in accordance with the phytoplankton enhancement observed in fish-stocked lakes in our study.
Our results cannot support a lower transparency in fish-stocked lakes attributed solely to the presence of fish; Mormul et al. (2012) using a long-term dataset proved that the resuspension of sediments by benthic fish in low-water phase mainly drives the transition from a macrophyte-dominated state to a turbid state in a floodplain pond. However, this statement should be verified with further investigations in the area as other factors not addressed until now, like lake size, spatial heterogeneity, and internal connectivity, also deeply contribute in the transition between states (Janssen et al., 2014).
Additionally, our analysis showed that fish introduction influences the composition of autotrophic planktonic groups. As observed for pico-, nano-, and microplankton, the composition of the fish-stocked lakes is different from the two studied fishless systems; the presence of fish was associated to higher abundances of microplanktonic cyanobacteria and also to the unique presence of PC-rich Pcy. These results are in agreement with exploratory studies performed by Izaguirre & Saad (2014) that showed differences among dominant groups in a limited dataset of lakes from Strobel Plateau with different regimes and presence/absence of fish. In this sense, phytoplankton composition shifts were also demonstrated in experimental studies that aimed to assess the impact of fish on food webs after trout removal via rotenone treatment (Duggan et al., 2015), and after nutrients and fish biomass manipulation (Romo et al., 2004).
The application of the phytoplankton functional classification (Reynolds et al., 2002, updated by Padisák et al., 2009) to our data displayed marked differences between fishless and fish-stocked lakes. Coda grouping small unicellular species, a characteristic of variable trophic state systems (X1, X2, X3, and J), abounded in fishless systems most of them vegetated, which may be explained by the selective predation of zooplankton over higher size classes of phytoplankton. As Borics et al. (2012) and citations therein described, the presence of macrophytes (by generating light attenuation, rising sedimentation, and bringing habitat for grazers) favors the dominance of these small flagellated algae as well as the development of chlorococcalean green algae in the mixed open waters areas of shallow lakes. By contrast, the dominance of colonial cyanobacteria and chlorophytes (K, Lm, F) and bigger cells like diatoms (P) in fish-stocked lakes evidenced the higher trophic state of this type of lakes as these groups typically develop in more enriched systems. These results are in line with Bartozek et al. (2016) who reported the dominance of these groups during an experimental fish culture under high-nutrient and good light conditions. It would be interesting to confirm by an experimental approach in the Strobel shallow lakes to what extent shifts on phytoplankton composition are an effect of fish-driven top-down and bottom-up effects. In this sense, an experimental study of fish removal in a lake of the region was conducted in order to analyze its evolution over time (Lancelotti, com. pers.). Despite differences among functional groups, fish-stocked and fishless systems share groups X1 and X2, both distinguished by their opportunistic characteristics and the high surface/volume that confers a high efficiency in nutrient acquisition. These characteristics are suitable to succeed in highly seasonal and dynamic environments as Patagonic plateaus lakes.
Among the unvegetated lakes of the Strobel Plateau, a marked difference of the dominant algal groups was observed, prevailing green algae in fishless lakes and cyanobacteria in fish-stocked lakes. This result is consistent with observations of Reissig et al. (2006) for other lakes in the north-Patagonian plateau. Phytoplankton compositional changes and in particular cyanobacteria dominance can be driven by changes in nutrient recycling caused by alterations in the food web structure such as the presence of fish in a water body (Elser, 1999). In fact, Horppila et al. (1998) presented the effect of large-scale fish removal in a Finish lake, where it triggered the depletion in total phosphorus followed by a marked reduction in cyanobacterial biomass. This group is composed by organisms that tolerate a wide environmental variability, most of them possess the ability to utilize atmospheric nitrogen for growth and are thus selectively promoted by high-phosphorus–low-nitrogen conditions (Suikkanen et al., 2013). Also cyanobacteria are known to be adapted to low-light environments (Reynolds, 1984).
As for the composition of autotrophic picoplankton, fish-stocked lakes were the only ones showing PC-rich Pcy assemblies, together with PE-rich Pcy and Peuk, which were also found in the other shallow lakes. PC-rich Pcy are adapted to environments with limited light as this group is more efficient at absorbing red light that is prevalent in turbid waters (Stomp et al., 2007). The same pattern was described in different water bodies in Argentina and elsewhere (e.g., Schiaffino et al., 2013; Izaguirre et al., 2014; Liu et al., 2014).
The occurrence of cyanobacteria due to the presence of trout could represent a potential threat for waterbirds using those lakes. This result is particularly relevant for the conservation of the Hooded grebe, since large vegetated lakes (including stocked ones) represent the only reproductive habitat for this species, and the presence of cyanobacteria in lakes with trouts could reduce their reproductive success and cause mortalities in adults, as it was observed in other species of grebes (Carmichael & Li, 2006). Likewise, the occurrence of toxic cyanobacteria was proposed as a potential cause of the declination Eared grebe (Podiceps nigricollis) in the Salton Sea (Anderson et al., 2007).
As the hydrology of the shallow lakes of this region is highly dynamic (Lancelotti, 2009; Lancelotti et al., 2010a, b; Izaguirre et al., 2018), changes in water level surely also affect the structure of the microbial autotrophic communities, and thus further field surveys, complemented with experimental studies, are needed to encompass contrasting hydrological conditions.
These results provide evidences that lake regime (mainly determined by the aquatic vegetation) and fish introduction exert a marked influence on planktonic autotrophic communities in arid Patagonia. The differences found both in the phytoplankton fraction > 20 μm and in the autotrophic picoplankton seem to be associated with the higher turbidity in lakes with fish introduction or without vegetation, as well as to a trophic cascade effect in fish-stocked systems. The marked effect of fish introduction on the composition of phytoplankton provokes changes in the food webs of the studied lakes, among which the proliferation of unwanted phytoplanktonic species may threaten the conservation of these sensitive environments.
Change history
05 December 2018
Due to an unfortunate turn of events, the first- and surnames of all authors were transposed in the original publication. The original article has been corrected and the proper representation of the authors’ names and their affiliation is also listed here.
References
Abell, J. M., D. Özkundakci, D. P. Hamilton & J. R. Jones, 2012. Latitudinal variation in nutrient stoichiometry and chlorophyll-nutrient relationships in lakes: a global study. Fundamental and Applied Limnology 181: 1–14.
Anderson, T. W., M. A. Tiffany & S. H. Hurlbert, 2007. Stratification, sulfide, worms, and decline of the Eared Grebe (Podiceps nigricollis) at the Salton Sea, California. Lake and Reservoir Management 23(5): 500–517.
Bartozek, E. C. R., N. C. Bueno, A. Feiden & L. C. Rodrigues, 2016. Response of phytoplankton to an experimental fish culture in net cages in a subtropical reservoir. Brazilian Journal of Biology 76(4): 824–833.
Blanco, S., 2001. Estudio Experimental sobre la Influencia de los Nutrientes en la Ecología Trófica de los Peces de una Laguna Somera. M Sc Thesis, Universidad de León.
Blanco, S., M. Fernández-Aláez & E. Bécares, 2008. Efficiency of top-down control depends on nutrient concentration in a Mediterranean shallow lake: a mesocosm study. Marine and Freshwater Research 59: 922–930.
Blindow, I., G. Andersson, A. Hargeby & F. Johansson, 1993. Long-term patterns of alternative stable states in two shallow eutrophic lakes. Freshwater Biology 30: 159–167.
Borics, G., B. Tóthmérész, B. A. Lukács & G. Várbíró, 2012. Functional groups of phytoplankton shaping diversity of shallow lake ecosystems. Hydrobiologia 698: 251–262.
Brinson, M. M. & A. I. Malvárez, 2002. Temperate freshwater wetlands: types, status and threats. Wetlands 29(2): 115–133.
Carpenter, S. R., J. F. Kitchell & J. R. Hodgsdon, 1985. Cascading trophic interaction and lake ecosystem productivity. BioScience 35: 635–639.
Carmichael, W. W. & R. H. Li, 2006. Cyanobacteria toxins in the Salton Sea. Saline Systems 2: 5.
de Tezanos Pinto, P. & I. O’Farrell, 2014. Regime shifts between free-floating plants and phytoplankton: a review. Hydrobiologia 740: 13–24.
Díaz, M., F. Pedrozo & N. Baccala, 2000. Summer classification of southern hemisphere temperate lakes (Patagonia, Argentina). Lakes and Reservoirs: Research and Management 5: 213–229.
Duarte, C. M. & L. Kalff, 1986. Littoral slope as a predictor of the maximum biomass of submerged macrophyte communities. Limnology and Oceanography 31(5): 1072–1080.
Duggan, I. C., S. A. Wood & D. W. West, 2015. Brown trout (Salmo trutta) removal by rotenone alters zooplankton and phytoplankton community composition in a shallow mesotrophic reservoir. N Z J Marine and Freshwater Research 49: 356–365.
Elser, J. J., 1999. The pathway to noxious cyanobacteria blooms in lakes: the food web as the final turn. Freshwater Biology 42(3): 537–543.
Feldmann, T. & P. Nõges, 2007. Factors controlling macrophyte distribution in large shallow Lake Võrtsjärv. Aquatic Botany 87: 15–21.
Horppila, J., H. Peltonen, T. Malinen, E. Luokkanen & T. Kairesalo, 1998. Top-down or bottom-up effects by fish: issues of concern in biomanipulation of lakes. Restoration Ecology 6: 20–28.
Imberti S., 2005. Meseta Lago Strobel. In Áreas importantes para la conservación de las aves en Argentina. Sitios prioritarios para la conservación de la biodiversidad. Di Giacomo A. S. (ed.). Aves Argentinas/Asociación Ornitológica del Plata: Buenos Aires: 415–416.
Izaguirre, I. & J. F. Saad, 2014. Phytoplankton from natural water bodies of the Patagonian Plateau. Advances in Limnology 65: 309–319.
Izaguirre, I., P. del Giorgio, I. O’Farrell & G. Tell, 1990. Clasificación de 20 cuerpos de agua andinopatogónicos (Argentina) en base a la estructura del fitoplancton estival. Cryptogamie Algologie 11: 31–46.
Izaguirre, I., F. Unrein, B. Modenutti & L. Allende, 2014. Photosynthetic picoplankton in Argentina lakes. Advances in Limnology 65: 343–357.
Izaguirre, I., J. Lancelotti, J. F. Saad, S. Porcel, I. O’Farrell, M. C. Marinone, I. Roesler & M. C. Dieguez, 2018. Influence of fish introduction and water level decrease on lakes of the arid Patagonian plateaus with importance for biodiversity conservation. Global Ecology and Conservation 14: e00391.
Janssen, A. B. G., S. Teurlincx, S. An, J. H. Janse, H. W. Paerl & W. M. Mooij, 2014. Alternative stable states in large shallow lakes? Journal of Great Lakes Research 40: 813–826.
Kirk, J. T. O., 1994. Light and Photosynthesis in Aquatic Ecosystems, 2nd ed. Cambridge University Press, Cambridge: 509.
Lancelotti, J. L., 2009. Caracterización limnológica de lagunas de la Provincia de Santa Cruz y efectos de la introducción de trucha arco iris: (Oncorhynchus mykiss) sobre las comunidades receptoras. Ph. D. Thesis, Universidad del Comahue: 122.
Lancelotti, J. L., L. M. Pozzi, P. M. Yorio, M. C. Diéguez & M. A. Pascual, 2009. Fishless shallow lakes of Southern Patagonia as habitat for waterbirds at the onset of trout aquaculture. Aquatic Conservation: Marine and Freshwater Ecosystems 19: 497–505.
Lancelotti, J. L., M. A. Pascual & A. Gagliardini, 2010a. A dynamic perspective of shallow lakes of arid Patagonia as habitat for waterbirds. In Meyer, P. L. (ed.), Ponds: Formation Characteristics and Uses. Nova Science Publishers Inc, New York: 187.
Lancelotti, J. L., L. M. Pozzi, P. M. Yorio, M. C. Diéguez & M. A. Pascual, 2010b. Precautionary rules for exotic trout aqua-culture in fishless shallow lakes of Patagonia: minimizing impacts on the threatened Hooded Grebe (Podiceps gallardoi). Aquatic Conservation: Marine and Freshwater Ecosystems 20: 1–8.
Lancelotti, J. L., L. M. B. Bandieri & M. A. Pascual, 2015. Diet of the exotic Rainbow Trout in the critical habitat of the threatened Hooded Grebe. Knowledge and Management of Aquatic Ecosystems 416: 1–11.
Lancelotti, J., M. C. Marinone & I. Roesler, 2017. Rainbow trout effects on zooplankton in the reproductive area of the critically endangered hooded grebe. Aquatic conservation 27: 128–136.
Liu, H., H. Jing, T. H. Wong & B. Chen, 2014. Co-occurrence of phycocyanin- and phycoerythrin-rich Synechococcus in subtropical estuarine and coastal waters of Hong Kong. Environmental Microbiology Reports 6: 90–99.
Marker, A. F. H., A. Nusch, H. Rai & B. Riemann, 1980. The measurement of photosynthetic pigments in freshwater and standardization of methods: conclusions and recommendations. Archiv für Hydrobiologie Beihefte. Ergebnisse der Limnologie. 14: 91–106.
Medina-Sánchez, J. M., M. Villar-argaiz & P. Carrillo, 2004. Neither with nor without you: a complex algal control on bacterioplankton in a high mountain lake. Limnology and Oceanography 49(5): 1722–1733.
Mormul, R. P., S. Thomaz, A. A. Agostinho, C. C. Bonecker & N. Mazzeo, 2012. Migratory benthic fishes may induce regime shifts in a tropical floodplain pond. Freshwater biology 57: 1592–1602.
Pace, M. L., J. J. Cole, S. R. Carpenter & J. F. Kitchell, 1999. Trophic cascades revealed in diverse ecosystems. Trends in Ecology and Evolution 14: 483–488.
Padisák, J., L. O. Crossetti & L. Naselli-Flores, 2009. Use and misuse in the application of the phytoplankton functional classification: a critical review with updates. Hydrobiologia 621(1): 1–19.
Panza, J. L. & M. R. Franchi, 2002. Magmatismo Basáltico Cenozoico Extrandino. In Haller, M. J. (ed.), Geología y Recursos Naturales de Santa Cruz. Relatorio del XV congreso Geológico Argentino, El Calafate: 201–236.
Pereyra, F. X., L. Fauqué & E. F. González Díaz, 2002. Geomorfología. In Haller, M. J. (ed.), Geología y Recursos Naturales de Santa Cruz. Relatorio del XV Congreso Geológico Argentino, El Calafate: 325–352.
Perotti, M. G., M. C. Diéguez & F. G. Jara, 2005. Estado del conocimiento de humedales del norte patagónico (Argentina): aspectos relevantes e importancia para la conservación de la biodiversidad regional. Revista Chilena de Historia Natural 78: 179–200.
Queimaliños, C. & M. Diaz, 2014. Phytoplankton of Andean Patagonian lakes. Advances in Limnology 65: 235–256.
Reissig, M., C. Trochine, C. Queimaliños, E. Balseiro & B. Modenutti, 2006. Impact of fish introduction on planktonic food webs in lakes of the Patagonian Plateau. Biological Conservation 132: 437–447.
Reynolds, C. S., 1984. The Ecology of Freshwater Phytoplankton. Cambridge University Press, Cambridge.
Reynolds, C. S., V. Huszar, C. Kruk, L. Naselli-Flores & S. Melo, 2002. Towards a functional classification of the freshwater phytoplankton. Journal of Plankton Research 24(5): 417–428.
Romo, S., M. R. Miracle, M. J. Villena, J. Rueda, F. Cerriol & E. Vicente, 2004. Mesocosm experiments on nutrient and fish effects on shallow lake food webs in a Mediterranean climate. Freshwater Biology 49(12): 1593–1607.
Sala, O. E., F. S. Chapin, J. J. Armesto, E. Berlow, J. Bloomfield, R. Dirzo, E. Huber-Sanwald, L. F. Huenneke, R. B. Jackson, A. Kinzig, R. Leemans, D. M. Lodge, H. A. Mooney, M. Oesterheld, N. LeRoy Poff, M. T. Sykes, B. H. Walker, M. Walker & D. H. Wall, 2000. Global biodiversity scenarios for the year 2100. Science 287(5459): 1770–1774.
Schallenberg, M. & B. Sorrell, 2009. Regime shifts between clear and turbid water in New Zealand lakes: environmental correlates and implications for management and restoration. N Z J Marine and Freshwater Research 43(3): 701–712.
Schaus, M. H., M. J. Vanni & T. E. Wissing, 2002. Biomass-dependent diet shifts in omnivorous gizzard shad: implications for growth, food web, and ecosystem effects. Transactions of the American Fisheries Society 131(1): 40–54.
Scheffer, M., S. H. Hosper, M. L. Meijer, B. Moss & E. Jeppesen, 1993. Alternative equilibria in shallow lakes. Trends in Ecology and Evolution 8: 275–279.
Scheffer, M., J. Bascompte, W. A. Brock, V. Brovkin, S. R. Carpenter, V. Dakos, H. Held, E. H. Van Nes, M. Rietkerk & G. Sugihara, 2009. Early-warning signals for critical transitions. Nature 461(7260): 53–59.
Schiaffino, M. R., J. M. Gasol, I. Izaguirre & F. Unrein, 2013. Picoplankton abundance and cytometric group diversity along a trophic and latitudinal lake gradient. Aquatic Microbial Ecology 68: 231–250.
Sharp, J. H., E. T. Peltzer, M. J. Alperin, G. Cauwet, J. W. Farrington, B. Fry, D. M. Karl, J. H. Martin, A. Spitzy, S. Tugrul & C. A. Carlson, 1993. Procedures subgroup report. Marine Chemistry 41: 37–49.
Stomp, M., J. Huisman, L. Vörös, F. R. Pick, M. Laamanen, T. Haverkamp & L. J. Stal, 2007. Colourful coexistence of red and green picocyanobacteria in lakes and seas. Ecology Letters 10: 290–298.
Suikkanen, S., S. Pulina, J. Engström-Öst, M. Lehtiniemi, S. Lehtinen & A. Brutemark, 2013. Climate change and eutrophication induced shifts in northern summer plankton communities. PLoS ONE 8: e66475.
The IUCN Red List of Threatened Species. (n.d.). Podiceps gallardoi. BirdLife International. 2016. Podiceps gallardoi. The IUCN Red List of Threatened Species 2016: e.T22696628A93574702. http://dx.doi.org/10.2305/IUCN.UK.2016-3.RLTS.T22696628A93574702.en. Downloaded on 27 February 2018.
Thomasson, K., 1959. Nahuel Huapi. Plankton of some lakes in an argentine National Park, with notes on terrestrial vegetation. Acta Phytogeografica Suecica 42: 1–83.
Thomasson, K., 1963. Araucanian Lakes. Plankton studies in North Patagonia, with notes on terrestrial vegetation. Acta Pytogeografica Suecica 47: 1–39.
Utermöhl, H., 1958. Zur Vervollkomnung der quantitativen Phytoplankton- methodik. Mitteilung Internationale Vereinigung fuer Theoretische unde Amgewandte Limnologie 9: 1–38.
Venrick, E. L., 1978. How many cells to count? In Sournia, A. (ed.), Phytoplankton Manual. UNESCO, Paris: 167–180.
Vollenweider, R. A. & J. Kerekes, 1982. Eutrophication of Waters. Monitoring, Assessment and Control. Organization for Economic Co-Operation and Development (OECD), Paris.
Acknowledgements
We thank Lic. Cristina Marinone, Dr. Ignacio Roesler, and Dr. Rodrigo Sinistro for their collaboration during the field campaigns. Also special thanks to the people of “Estancia Laguna Verde” for the lodging and collaboration on the logistics during the campaigns. This investigation was supported by a grant of the Argentinean Fund for Technical and Scientific Investigation (FONCYT, PICT 0794).
Author information
Authors and Affiliations
Corresponding author
Additional information
The original version of this article was revised: the first- and surnames of all authors were transposed.
Guest editors: Hugo Sarmento, Irina Izaguirre, Vanessa Becker & Vera L. M. Huszar / Phytoplankton and its Biotic Interactions
Rights and permissions
About this article
Cite this article
Saad, J.F., Porcel, S., Lancelotti, J. et al. Both lake regime and fish introduction shape autotrophic planktonic communities of lakes from the Patagonian Plateau (Argentina). Hydrobiologia 831, 133–145 (2019). https://doi.org/10.1007/s10750-018-3660-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-018-3660-6