Abstract
Modifications to riverine systems that alter freshwater inflow to downstream estuarine habitats have resulted in altered patterns of nekton distribution and abundance. To examine how nekton assemblages respond to variable hydrologic patterns, we used trawl and seine survey data to compare the seasonal trends (dry vs. wet season) expected of a natural system to those of a river with regulated flow discharges that often magnify high flow events. Nekton assemblages differed between seasons in a representative natural system, similar to other estuaries of the region. For example, assemblage differences were characterized by significantly higher abundance and richness in trawl surveys, and significantly higher richness in seine surveys in the wet relative to the dry season. These seasonal trends were dampened in the altered system. Species important in defining seasonal dissimilarities in both systems were characterized as estuarine resident species, including Anchoa mitchilli, Menidia spp., Cynoscion arenarius, and Trinectes maculatus, yet were observed largely to have opposing seasonal trends in abundance between the two rivers. Our comparison provides evidence that flow modifications result in a loss of natural seasonal variability in estuarine nekton assemblages, but additional investigations of flow-altered systems are needed to confirm these findings.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Freshwater flow is known to be an influential factor structuring nekton communities of estuarine reaches within tidal rivers (Peterson & Ross, 1991; Sklar & Browder, 1998), with the nekton assemblages changing most rapidly at the oligohaline–mesohaline boundary (Greenwood et al., 2007). Because many estuarine species have evolved life history strategies in response to natural seasonal flow regimes (Bunn & Arthington, 2002; Lytle & Poff, 2004), alterations to the magnitude and timing of flow can be detrimental (Drinkwater & Frank, 1994; Gillson, 2011). Multiple effects including a reduction in species growth rates (Rypel & Layman, 2008) and recruitment dynamics (Jenkins et al., 2010), and changes to the overall structure of estuarine food webs (Adams et al., 2009) have been documented in response to altered flow regimes.
Seasonal fluctuations in estuarine nekton assemblages are common across estuaries worldwide and are important determinants of community structure and dynamics (Idelberger & Greenwood, 2005; Greenwood et al., 2007; Rozas et al., 2007; Sheaves et al., 2010). However, extreme flow events where frequency or severity becomes too great can reduce system complexity through lower species diversity and abundance. Natural extreme flow disturbances such as drought and storms in stream (Walters & Post, 2011), estuarine (Livingston et al., 1997, Greenwood et al., 2006; Baptista et al., 2010), and coastal marine (Byrnes et al., 2011) communities provide common examples of these effects. More specifically in estuarine environments, lower diversity of estuarine resident, and marine nekton and macrofaunal species, have been associated with prolonged periods of freshwater inflow resulting from human alteration (Rutger & Wing, 2006; McLeod & Wing, 2008). Similarly, decreases in seagrasses, oyster beds, and juvenile fish abundance and richness have been observed, partly in response to rapidly changing salinities and sediment loads as a result of heavy freshwater flows (Chamberlain & Doering, 1998). Consequences of altered flow for the complexity of estuarine communities, however, can be unpredictable. For example, Kimmerer (2002) observed that lower trophic levels (i.e., plankton) responded negatively to high flow (i.e., lower abundance), whereas higher trophic levels (i.e., fishes) responded positively to flow (i.e., higher abundance). Nevertheless, lower species diversity and abundance following extreme flow events have the potential to destabilize food webs (Rooney et al., 2006).
For relatively natural rivers, strong seasonal changes in nekton assemblages with increases in nekton community metrics (e.g., species richness) and distinct changes in assemblage structure—with seasonal progression of dry to wet seasons—have been well documented (Tsou & Matheson, 2002; Idelberger & Greenwood, 2005). Seasonal high flow events have been shown to alter resources available to nekton consumers and subsequently the flow of energy through estuarine food webs (Olin et al., 2013). The aim of the present study was to evaluate differences between nekton assemblages during seasonal periods of freshwater inflow in two riverine systems that experience different inflow regimes; one that has undergone major human development and experiences altered flow regimes, and one that serves as the most proximate example of a river experiencing relative natural flows. Given the magnitude of high flow events in the altered system we predict nekton assemblages in this system to have reduced connectivity and exhibit different seasonal responses compared with a more natural system. Specifically, we predict that nekton density, richness, and overall community structure will remain similar across seasons in the altered system, while the natural system should exhibit different seasonal patterns.
Materials and methods
Study areas
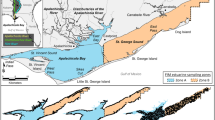
The Caloosahatchee River (26°30′ N, 81°54′ W) is a major tributary of the Charlotte Harbor estuary, a large (~700 km2) relatively shallow estuary on the southwest coast of Florida, USA (Fig. 1). The artificial connection of Lake Okeechobee to the Caloosahatchee River represents a unique anthropogenic manipulation of hydrology (Doering & Chamberlain, 1998), whereby substantial seasonal discharge from Lake Okeechobee occurs for flood control and water supply, as well as to flush algal blooms and prevent salt water intrusion (Flaig & Capece, 1998). Major modifications to the hydrology, along with land-use transformations and dredging for navigation (e.g., ~70% of shoreline is hardened with seawalls and rip-rap) have resulted in large-scale alterations within the estuary (Barnes, 2005). The volume of the estuarine portion of the Caloosahatchee River is approximately 105 × 106 m3, while the median annual discharge is 870 × 106 m3 (Flaig & Capece, 1998). During periods of low freshwater discharge (i.e., during winter/spring months), salt water regularly intrudes to the Franklin Lock, the most downstream water control structure, often exceeding 10 psu (Fig. 2). High freshwater discharge (i.e., during summer/autumn months) can cause salinity to drop below 5 psu at the mouth and the transition between the two states can be rapid, sometimes occurring in less than a week (Doering et al., 2002; Fig. 2). Fluctuations observed at the head and mouth of the river, exceed salinity tolerances of most oligohaline and marine species (Barnes, 2005). These alterations to flow patterns of the Caloosahatchee River are particularly relevant, in light of implications for the Comprehensive Everglades Restoration Plan, thereby creating an ideal system to document the effects of altered flow on community dynamics (Perry, 2004).
Mean daily discharge recorded in the Myakka (gray) and Caloosahatchee (black) Rivers from 2004 to 2009. River discharge data were obtained from the U.S. Geological Survey (http://water.usgs.gov/data.html) for the Myakka River at Myakka River near Sarasota (Station 02298830) and from the South Florida Water Management District (http://my.sfwmd.gov) for the Caloosahatchee River at the Cape Coral Bridge (Station CCORAL)
Trends in abundances of nekton can be influenced by a myriad of factors, including recruitment and/or stochastic climactic events (Greenwood et al., 2007). To minimize variability associated with these factors, we chose the Myakka River (82°12′ W, 26°57′ N) for comparison with the Caloosahatchee River, as it is proximately located (<100 km; Fig. 1), and therefore, is accessible to fishes of Charlotte Harbor. The Myakka River experiences relatively natural flow periods, despite several small dams and canal diversions (Fig. 2). Additionally, its’ shoreline has been subjected to comparatively minor anthropogenic modification [i.e., ~40% of shoreline area is hardened; estimated from 2007 Digital Ortho Quad County Mosaic, USDA, Geospatial Data Gateway in ArcGIS (ESRI ArcGIS version 9)]. Based on similar trends in fish and macrofaunal abundance among proximate estuaries in Chesapeake Bay (Kraus & Secor, 2004), and among three river-estuaries along the Texas coast (Palmer et al., 2011), the Myakka River provides a reference by which comparisons of nekton community dynamics to the Caloosahatchee River can be made.
Nekton communities
Data on nekton assemblages in the Myakka and Caloosahatchee Rivers were obtained from a long-term fisheries-independent monitoring (FIM) program in the Charlotte Harbor estuary. Between 2004 and 2009, monthly stratified-random sampling was conducted in the estuarine reaches of the Myakka and Caloosahatchee Rivers using a 6.1-m trawl (38-mm stretch mesh, 3.2-mm stretch mesh liner) and a 21.3-m seine (3.2-mm stretchmesh, center-bag). The 6.1-m otter trawl targets deeper areas, while the 21.3-m seine targets shoreline areas. Sampling gear types were selected to provide a representative sample of the nekton assemblage. Sampling locations were chosen randomly each month from all possible sites that contained adequate depth for trawling (1.8–7.6 m) and seining (0.3–1.8 m). The sampling effort that fell within the areas used in this study was 3–5 trawls and 3–4 seines/month for the Myakka River and 4–5 trawls and 10–13 seines/month for the Caloosahatchee River. The trawl was towed for 5 min at 0.6 m s−1, providing a tow length of ~180 m. Trawl width averaged ~4 m, providing an approximate area of 720 m2 sampled by a typical tow. The seine was deployed from a boat in a shallow arc parallel to shore and hauled directly along the shoreline. The two ends of the seine were pulled together, sampling an area of ~68 m2.
During each sampling event, environmental parameters—including temperature (°C), salinity (psu), and dissolved oxygen (mg l−1)—were profiled with a Hydrolab water quality datasonde (measurements taken at 0.2 m, every 1.0 m if applicable, and at the bottom). Fishes and select invertebrates collected during each sampling event were identified to the lowest practical taxonomic level (nomenclature for fishes follows Nelson et al., 2004), measured [standard length (SL) for fishes and carapace width (CW) for crabs], counted, and released. Representative subsamples of organisms were retained for laboratory verification. For specific details on site selection and sampling technique refer to Idelberger & Greenwood (2005) and Idelberger et al. (2011).
Statistical analysis
In southwest Florida, many rivers are categorized as having the southern river flow pattern, i.e., a significant proportion of riverine annual flow (~60%) is concentrated in the wet season, which occurs during the months of July–October (Kelly & Gore, 2008). In the case of the Caloosahatchee River, a fundamental premise in our analysis is that the wet season is further exaggerated by discharges from Lake Okeechobee, while the Myakka River experiences a relatively natural hydrological cycle. For both rivers, data for all trawl and seine surveys from dry and wet seasons were, therefore, grouped for the months of May–June (dry) and August–September (wet), respectively, from all years of sampling. These years differ in magnitude of freshwater flow (Fig. 2) providing the opportunity to characterize the River’s assemblages during seasonal periods of varying freshwater inflow.
Mean environmental conditions (averaged for the entire water column) from the two rivers and flow data (daily flow recordings averaged by month for each season) were explored using principal component analysis (PCA) in R (R Core Development Team, 2011). This approach was used to resolve four environmental variables (temperature, salinity, dissolved oxygen and flow) into orthogonal components based on the correlation matrix. The principal components (PC) were rotated using the varimax option to facilitate interpretability of each component. PC scores were then used to calculate centroids for rivers, seasons, and survey method. Confidence intervals (±2 SE) around these centroids were estimated as the mean standard errors on the three component axes for each river, season, and survey method. Environmental variables were tested for normality using Shapiro–Wilk tests and quantile–quantile probability plots. Where the assumptions of normality and equal variance were not met, the data were log-transformed.
Linear mixed-effect models fit using restricted maximum likelihood in the lme4 package in R (Bates & Maechler, 2010), were constructed to investigate the effects of season (dry vs. wet) on trawl and seine assemblages within the two rivers separately. Linear mixed-effect models, with year as the random effect, were applied to test for differences in the dependent variables (i.e., density and species richness) derived for trawl and seine assemblages between seasons in each river. This was based on the premise that we were testing for the effects of altered flow on the assemblages, not annual variation in the magnitude of flow. For each mixed-effects model, we applied one degree of freedom orthogonal linear contrasts between seasons in each river, evaluated using t-tests. An examination of the probability plots of residuals from linear mixed-effect models indicated that models fit adequately, and quantile–quantile plots showed data to be generally described by normally distributed errors for all comparisons.
Seasonal differences in assemblage structure were analyzed for seine and trawl surveys separately using multivariate techniques performed using the vegan package in R (Oksanen, 2011). To reduce the influence of highly abundant species, density estimates for each species were square-root transformed. An analysis of similarity (ANOSIM) was used to compare assemblages between seasons (dry and wet) for each river, separately. Before ANOSIM was performed, Bray–Curtis similarity matrices were calculated for data averaged by sampling event (river, year, and month). Similarity percentage analysis (SIMPER) was used to identify species representative of dissimilarities between seasons determined from ANOSIM. Species that were considered distinguishing were those that contributed >2% to the total average dissimilarity between seasons. To illustrate the magnitude of difference between seasons for the distinguishing species, output from SIMPER was displayed graphically after subtracting the average abundance of each species (square-root transformed) in the wet season from that of the dry season. These species were then grouped by life history characteristics, for example, as estuarine resident species (i.e., those capable of completing their entire life cycle within the estuarine environment following Elliott et al. 2007; Stevens et al., 2013) and Welch two -sample t-tests were applied to determine seasonal differences. Non-metric multidimensional scaling (NMDS) ordination was used to graphically coordinate the patterns in community structure and composition between seasons within river for each sampling method on data averaged by river, year, and month. Data from a Bray–Curtis similarity matrix were used to construct the ordination plots. We predicted that if nekton assemblages were affected by high flow, then the results from these analyses would show non-significant differences among seasons.
Results
Environmental conditions varied between survey methods and between seasons in each of the two rivers (Table 1). The PCA identified three major axes (eigenvalues >1) that cumulatively explained 92.5% of the total environmental variability (Table 2), with clear separation between seasons (Fig. 3). PC1 indicated a separation of season and river for both survey methods with the Myakka River tending to have higher salinity and lower flow during both seasons relative to the Caloosahatchee River (Table 2; Fig. 3). PC2 indicated a gradient in DO with the dry season having higher values for this variable relative to the wet season in both rivers (Table 2; Fig. 3). PC3 indicated a gradient in water temperature with warmer temperatures observed in the wet season in both rivers (Table 2; Fig. 3).
Flow estimates and environmental conditions from trawl and seine surveys between rivers (Caloosahatchee and Myakka) and among seasons [dry (spring—May and June); wet (autumn—August and September)] plotted in 3-dimensional principal components space. Balloons indicate location of centroids with radii representing two standard errors around the means
A total of 144,354 individuals constituting 100 species were included in this study (Table 3), with the most abundant species being bay anchovy Anchoa mitchilli, silverside species Membras martinica and Menidia spp., and mojarra species Eucinostomus spp. (Table 3). Trawl assemblages in the Myakka River exhibited significantly higher density (t = −4.36, P = 0.001) and richness (t = −3.63, P = 0.000) during the wet season [density: 59.3 ± 1.9 (mean ± SE); richness: 8.5 ± 0.4] compared with the dry season (density: 19.8 ± 1.7; richness: 6.3 ± 0.4). Seine assemblages in the Myakka River showed significantly higher richness (t = −2.73, P = 0.006) in the wet compared with the dry season but lower density (377.9 ± 89.9 to 219.2 ± 41.8), although not significant (t = 1.34, P = 0.179). In the Caloosahatchee River, there were no statistically significant trends for density (trawl: t = −0.49, P = 0.620; seine: t = −0.34, P = 0.728) or richness (trawl: t = −0.41, P = 0.681; seine: t = −0.53, P = 0.601) between seasons, although lower density was observed in trawl assemblages (9.4 ± 3.1 to 8.7 ± 2.1) and higher density in seine assemblages (558.5 ± 289.8 to 634.2 ± 420.1) in the wet compared with the dry season. Lower richness was observed for both trawl (5.8 ± 0.3 to 5.6 ± 0.3) and seine (6.2 ± 0.3 to 5.9 ± 0.4) assemblages in the wet compared with the dry season.
Nekton varied seasonally within each river for both trawl and the seine assemblages (Fig. 4). Assemblages differed in their degree of similarity based on R values from ANOSIM that ranged from 0.16 to 0.42. Seasonal differences in assemblages of the Myakka River were more pronounced than those of the Caloosahatchee River for each gear type (trawls: R = 0.42 vs. R = 0.33; seines: R = 0.37 vs. R = 0.16; Fig. 4). The NMDS plots of Bray–Curtis similarity values of assemblages showed clear differentiation between seasons in the Myakka River (Fig. 4) for both assemblages, whereas seasonal differences in the Caloosahatchee River were apparent only for the trawl assemblage, supporting ANOSIM results (Fig. 4).
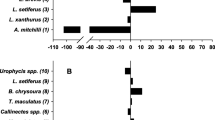
Species distinguishing the dry from wet season were similar between the two rivers, based on SIMPER (Fig. 5), but followed different patterns. For trawl assemblages in the Myakka River, higher abundances were generally observed in the wet relative to the dry season (Fig. 5), whereas, this trend was largely not observed in the Caloosahatchee River for the same species (Fig. 5). In the Myakka River, seine assemblage differences were driven by lower densities of A. mitchilli and M. martinica and higher densities of Menidia spp., Eucinostomus spp. and E. harengulus in the wet compared with the dry season (Fig. 5). In the Caloosahatchee River seine assemblage, densities of Menidia spp. and Eucinostomus spp. followed the same tends as populations in the Myakka River. However, opposite trends in densities of A. mitchilli and E. harengulus were observed (Fig. 5). The distinguishing species driving seasonal differences in trawl assemblages of the Myakka River were largely represented by estuarine resident species (see species designations in Table 4). For example, Cynoscion arenarius, Trinectes maculatus, A. mitchilli, and Menticirrhus americanus contributed to differences in assemblage structure between seasons (44%; Table 4) with cumulative densities higher in the wet relative to the dry season (t = 6.38, P = 0.001). Estuarine resident species also contributed to seasonal differences in the trawl assemblages of the Caloosahatchee River. However, their cumulative density remained unchanged between seasons (t = 1.39, P = 0.165; Table 4). Similarly, estuarine resident species contributed to the seasonal differences among seine assemblages in both rivers, but significant changes in overall density among seasons in either river were not observed (Myakka River: t = −0.26, P = 0.796; Caloosahatchee River: t = 0.35, P = 0.728; Table 4).
Differences in average square-root densities between nekton species (dry–wet) identified from SIMPER analyses that distinguish seasonal relationships in trawl and seine assemblages within the Myakka and Caloosahatchee Rivers. For example, negative values indicate that the distinguishing species is more abundant in the wet season. Species include those that contributed ≥2% to the total average dissimilarity between seasons
Discussion
Given the global scale of anthropogenic alteration across riverine systems (Nilsson et al., 2005), understanding the effects of high flow disturbance on estuarine nekton assemblage structure is important for developing successful management strategies aimed at maintaining productive habitats. High flow events are known to affect estuarine ecosystems causing, for example, declines in the catches of estuarine and coastal fisheries (Drinkwater & Frank, 1994; Gillson et al., 2012) and decreases in abundances of estuarine fishes and invertebrates (Costa et al., 2007; McLeod & Wing, 2008). Our comparison of seasonal nekton assemblage dynamics in two rivers that experience contrasting hydrological flow regimes provides evidence for significant reductions in community variability under high flow conditions. In the Myakka River, there were significant changes in the nekton assemblage between dry and wet seasons with marked increases in the density and species richness of deeper water trawl-surveyed species observed during the wet season in addition to increased richness of shoreline seine-surveyed species. These observations are consistent with those predicted for minimally impacted natural river systems (Greenwood et al., 2007; Sheaves et al., 2010). In contrast, such seasonal differences were less defined in the Caloosahatchee River with lower densities and lower species richness observed in the trawl-surveyed assemblages and lower richness in seine-surveyed assemblages during the altered high flow season. These findings are consistent with the premise that modified inflow to rivers results in less complex communities (Livingston et al., 1997; Greenwood et al., 2006; Baptista et al., 2010).
Seasonal changes observed in the nekton communities of both rivers were largely driven by estuarine species, including A. mitchilli and C. arenarius. However, these patterns were largely divergent, whereby abundances of these species generally increased in the wet season in the Myakka River and either remained stable or decreased in the Caloosahatchee River in the wet season, specifically evident in the trawl assemblage. For these influential species in the Myakka River, the increase in abundance is likely a result of natural recruitment dynamics in tidal rivers (Greenwood et al., 2007; Sheaves et al., 2010). Seasonal variation in estuarine fish assemblages is strongly influenced by biological factors including the spawning and recruitment patterns of the individual species (Sheaves et al., 2010). However, estuarine species are likely more subject to local conditions than marine migrants whose recruitment patterns into estuaries include broad-scale oceanic and meteorological processes to a larger degree (Elliott et al. 2007). Idelberger & Greenwood (2005) observed recruitment of the majority of estuarine fish species (e.g., A. mitchilli and C. arenarius), into the Myakka River during the period of June through October, suggesting that these species take advantage of such factors as abundant food resources and the shelter provided by complex shoreline habitat and enhanced turbidity as associated with increased river discharge. Moreover, Purtlebaugh & Allen (2010) demonstrated a positive relationship between relative abundance and river flow for juveniles of estuarine species (i.e., age-0 C. nebulosus and C. arenarius) in the lower Suwannee River and that these fishes experienced increased growth rates during the wetter years (i.e., period of increased flow). This suggests that some species, especially estuarine resident species that exhibit marked responses to changes in freshwater inflow, might be good indicators of the potential effects associated with flow alteration. Understanding such changes in nekton communities will be useful as escalating human water demand and climate change is predicted to lead to increased frequency of extreme flow events (Vörösmarty et al., 2000).
Characterizing the influences of freshwater flow regimes on nekton communities represent a unique challenge. Some of the difficulty arises from distinguishing direct effects of altered flow regimes from indirect effects associated with land-use change that often accompany urbanization and water resource development. To isolate if flow is the factor-driving nekton assemblage differences it is necessary to conduct cross-system comparisons (Mayer & Galatowitsch, 2001; Tsou & Matheson, 2002), such as those conducted in the present study. Similar trends in fish abundance and assemblage composition have been observed in proximate estuaries (Kraus & Secor, 2004; Idelberger & Greenwood, 2005), lending support to our selection of proximate river systems. However, there are always inherent differences between systems that should be taken into consideration in cross-system comparisons. In this case, greater urbanization in the Caloosahatchee River relative to the Myakka River is exemplified by the presence of major cities, artificial connections to adjacent inland systems, and greater proportion of hardened shoreline (70 vs. 40%). Fish assemblages respond to urbanization gradients with sensitive species disappearing as urbanization increases and heterogeneity of habitat decreases (Walters et al., 2003; Morgan & Cushman, 2005). We acknowledge that the present study design is limited to a single river comparison, and as a result, direct comparisons of the observed patterns to other systems warrant caution given the magnitude of anthropogenic modifications across these rivers. The similarity in distinguishing species contributing to the seasonal assemblages in these rivers provides confidence that altered flow was likely a major cause for the divergence of nekton community metrics that occurred during the wet season. We recommend additional studies using a similar approach. If a pattern of dampened seasonality is apparent with altered river flow in multiple studies, then mounting evidence would suggest a homogenization of river fauna.
The geomorphology of a river system is also an important factor to consider when examining the effects of altered flow on nekton assemblages (Visintainer et al., 2006; Allen et al., 2007; Stevens et al., 2010). It is possible that the effects of altered high flow on community structure of the Caloosahatchee River were dampened through geomorphological characteristics that create a balance between individuals entering and leaving the system, particularly for species sampled via trawl. The relatively long mixing zone in the Caloosahatchee River, combined with the high flow event, could result in clearly defined isohalines within the estuarine reach of the river that would not necessarily be so apparent under natural flow regimes. With distinct isohalines, the distribution of species and their centers of abundance would be expected to shift (e.g., freshwater species move downstream with freshwater flow; Kimmerer, 2002; Greenwood et al., 2007; Guenther & MacDonald, 2012). Such distributional responses following high inflow events could account for the lack of significant change in density observed from the trawl and seine surveys in the Caloosahatchee River, as nekton had more area to find preferable habitat and not be displaced out of the system. Given the different dynamics in estuaries, it is important to reiterate that there was a little change in the density of nekton species and overall community structure, which conflicts with the results of the natural dynamics of tidal rivers.
Reduction in abundance and richness of species within a community can result in a loss or overall homogenization of energy flow pathways and ultimately a less stable and more simplified food web structure (Layman et al., 2007). This is an important consideration for the Caloosahatchee River, given the lower density and richness of species observed in the wet seasonal relative to the dry season. What remains to be better understood, is how the observed changes in community composition and the response of particular species can alter energy flow and food web interactions within these systems (Piazza & La Peyre, 2011; Olin et al., 2013). For example, Piazza & La Peyre (2012) documented an increase in density and biomass of marsh surface-associated fishes following a river pulse event in Brenton Sound, Louisiana, suggesting the potential for export of these resources to subtidal habitats. Such dynamics may be important considerations for rivers with large floodplains. To advance our understanding of community level effects, future studies should consider the application of food web models to explore the effects of altered flow regimes on community assembly.
References
Adams, A. J., R. K. Wolfe & C. A. Layman, 2009. Preliminary examination of how human-driven freshwater flow alteration affects trophic ecology of juvenile snook (Centropomus undecimalis) in estuarine creeks. Estuaries and Coasts 32: 819–828.
Allen, D. M., S. S. Haertel-Borer, B. J. Milan, D. Bushek & R. F. Dame, 2007. Geomorphological determinants of nekton use of intertidal salt marsh creeks. Marine Ecology Progress Series 329: 57–71.
Baptista, J., F. Martinho, M. Dolbeth, I. Viegas, H. Cabral, et al., 2010. Effects of freshwater flow on the fish assemblage of the Mondego estuary (Portugal): comparison between drought and non-drought years. Marine and Freshwater Research 61: 490–501.
Barnes, T., 2005. Caloosahatchee Estuary conceptual ecological model. Wetlands 25: 884–897.
Bates, D. & M. Maechler, 2010. Linear mixed-effects models using S4 class in R. package version
Bunn, S. E. & A. H. Arthington, 2002. Basic principles and ecological consequences of altered flow regimes for aquatic biodiversity. Environmental Management 30: 492–507.
Byrnes, J. E., D. C. Reed, B. J. Cardinale, K. C. Cavanaugh, S. J. Holbrook, et al., 2011. Climate-driven increases in storm frequency simplify kelp forest food webs. Global Change Biology 17: 2513–2524.
Chamberlain, R. H. & P.H. Doering, 1998. Freshwater inflow to the Caloosahatchee estuary and the resource-based method for evaluation. In S.F. Treat (ed.), Proceedings of the 1997 Charlotte Harbor Public Conference and Technical Symposium, South Florida Water Management District and Charlotte Harbor National Estuary Program, Technical Report No. 98-02. Washington, D.C.: 81–90
Costa, M. J., R. Vasconcelos, J. L. Costa & H. N. Cabral, 2007. River flow influence on the fish community of the Tagus estuary (Portugal). Hydrobiologia 587: 113–123.
Doering, P.H. & R.H. Chamberlain, 1998. Water quality in the Caloosahatchee Estuary, San Carlos Bay and Pine Island Sound. In: Proceedings of the Charlotte Harbor Public Conference and Technical Symposium, Technical Report No. 98-02, Charlotte Harbor National Estuary Program, Punta Gorda, FL: 229–240.
Doering, P. H., R. H. Chamberlain & D. E. Haunert, 2002. Using submerged aquatic vegetation to establish minimum and maximum freshwater inflows to the Caloosahatchee estuary, Florida. Estuaries 25: 1343–1354.
Drinkwater, K. F. & K. T. Frank, 1994. Effect of river regulation and diversion on marine fish and invertebrates. Aquatic Conservation-Marine & Freshwater Ecosystems 4: 135–151.
Elliott, M., A. K. Whitfield, I. C. Potter, S. J. M. Blaber, D. P. Cyrus, et al., 2007. The guild approach to categorizing estuarine fish assemblages: a global review. Fish and Fisheries 8: 241–268.
Flaig, E.G. & J. Capece, 1998. Water use and runoff in the Caloosahatchee watershed. In: Proceedings of the Charlotte Harbor Public Conference and Technical Symposium, Technical Report No. 98-02, Charlotte Harbor National Estuary Program, Punta Gorda, FL, 73–80.
Gillson, J., I. Suthers & J. Scandol, 2012. Effects of flood and drought events on multi-species, multi-method estuarine and coastal fisheries in eastern Australia. Fisheries Management and Ecology 19: 54–68.
Gillson, J., 2011. Freshwater flow and fisheries production in estuarine and coastal systems: where a drop of rain is not lost. Reviews in Fisheries Science 19: 168–186.
Greenwood, M. F. D., R. E. Matheson, R. H. McMichael & T. C. MacDonald, 2007. Community structure of shoreline nekton in the estuarine portion of the Alafia River, Florida: differences along a salinity gradient and inflow-related changes. Estuarine, Coastal Shelf Science 74: 23–238.
Greenwood, M. F. D., P. W. Stevens & R. E. Matheson, 2006. Effects of the 2004 hurricanes on the fish assemblages in two proximate southwest Florida estuaries: changes in the context of inter-annual variability. Estuaries and Coasts 29: 985–996.
Guenther, C. B. & T. C. MacDonald, 2012. Comparison of estuarine salinity gradients and associated nekton community change in the lower St. Johns River Estuary. Estuaries and Coasts 35: 1443–1452.
Idelberger, C. F. & M. F. D. Greenwood, 2005. Seasonal variation in fish assemblages within the estuarine portions of the Myakka and Peace Rivers, Southwest Florida. Gulf of Mexico Science 2: 224–240.
Idelberger, C. F., C. J. Stafford & S. E. Erickson, 2011. Distribution and abundance of introduced fishes in Florida’s Charlotte Harbor Estuary. Gulf and Caribbean Research 23: 13–22.
Jenkins, G. P., S. D. Conron & A. K. Morison, 2010. Highly variable recruitment in an estuarine fish is determined by salinity stratification and freshwater flow: implications of a changing climate. Marine Ecology Progress Series 417: 246–261.
Kelly, M. H. & J. A. Gore, 2008. Florida river flow patterns and the Atlantic multi-decadal oscillation. River Research and Applications 24: 598–616.
Kimmerer, W. J., 2002. Effects of freshwater flow on abundance of estuarine organisms: physical effects or trophic linkages? Marine Ecology Progress Series 243: 39–55.
Kraus, R. & D. H. Secor, 2004. Dynamics of white perch (Morone americana) population contingents in the Patuxent River estuary, Maryland USA. Marine Ecology Progress Series 279: 247–259.
Layman, C. A., J. P. Quattrochi, C. M. Peyer & J. E. Allgeier, 2007. Niche width collapse in a resilient top predator following ecosystem fragmentation. Ecology Letters 10: 937–944.
Livingston, R. J., X. Niu, G. Lewis & G. C. Woodsum, 1997. Freshwater input to a gulf estuary: long-term control of trophic organization. Ecological Applications 7: 277–299.
Lytle, D. A. & N. L. Poff, 2004. Adaptation to natural flow regime. Trends in Ecology and Evolution 19: 94–100.
Mayer, P. M. & S. M. Galatowitsch, 2001. Assessing ecosystem integrity of restored prairie wetlands from species production-diversity relationships. Hydrobiologia 443: 177–185.
McLeod, R. J. & S. R. Wing, 2008. Influence of altered salinity regime on the population structure of two infaunal bivalve species. Estuarine, Coastal and Shelf Science 78: 529–540.
Morgan, R. P. & S. F. Cushman, 2005. Urbanization effects on stream fish assemblages in Maryland, USA. Journal of North American Benthological Society 24: 643–655.
Nelson, J. S., E. J. Crossman, H. Espinosa-Pérez, L. T. Findley, C. R. Gilbert, et al., 2004. Common and Scientific Names of Fishes from the United States, Canada, and Mexico. Committee on Names of Fishes, 6th ed. American Fisheries Society, Bethesda, MD.
Nilsson, C., C. A. Reidy, M. Dynesius & C. Revenga, 2005. Fragmentation and flow regulation of the world’s large river systems. Science 308: 405–408.
Oksanen, J., 2011. Multivariate analysis of ecological communities in R: vegan tutorial.
Olin, J. A., N. E. Hussey, S. A. Rush, G. R. Poulakis, C. A. Simpfendorfer, et al., 2013. Seasonal variability in stable isotopes of estuarine consumers under different freshwater flow regimes. Marine Ecology Progress Series 487: 55–69.
Palmer, T. A., P. A. Montagna, J. B. Pollack, R. D. Kalke & H. R. DeYoe, 2011. The role of freshwater inflow in lagoons, rivers and bays. Hydrobiologia 667: 49–67.
Perry, W., 2004. Elements of South Florida’s comprehensive everglades restoration plan. Ecotoxicology 13: 185–194.
Peterson, M. S. & S. T. Ross, 1991. Dynamics of littoral fishes and decapods along a coastal river–estuarine gradient. Estuarine, Coastal and Shelf Science 33: 467–483.
Piazza, B. P. & M. K. La Peyre, 2011. Nekton community response to a large-scale Mississippi River discharge: examining spatial and temporal response to river management. Estuarine, Coastal and Shelf Science 91: 379–387.
Piazza, B. P. & M. K. La Peyre, 2012. Measuring changes in consumer resource availability to riverine pulsing in Breton Sound, Louisiana, USA. PLoS ONE 7(5): e37536.
Purtlebaugh, C. H. & M. S. Allen, 2010. Relative abundance, growth, and mortality of five age-0 estuarine fishes in relation to discharge of the Suwannee River, Florida. Transactions of the American Fisheries Society 139: 1233–1246.
R Development Core Team, 2011. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria.
Rooney, N., K. McCann, G. Gellner & J. C. Moore, 2006. Structural asymmetry and the stability of diverse food webs. Nature 442: 265–269.
Rozas, L. P., T. J. Minello, R. J. Zimmerman & P. Caldwell, 2007. Nekton populations, long-term wetland loss, and the effect of recent habitat restoration in Galveston Bay, Texas, USA. Marine Ecology Progress Series 344: 119–130.
Rutger, S. M. & S. R. Wing, 2006. Effects of freshwater input on shallow-water infaunal communities in Doubtful Sound, New Zealand. Marine Ecology Progress Series 314: 35–47.
Rypel, A. L. & C. A. Layman, 2008. Degree of aquatic ecosystem fragmentation predicts population characteristics of gray snapper in Caribbean tidal creeks. Canadian Journal of Fisheries and Aquatic Sciences 65: 335–339.
Sheaves, M., R. Johnston & R. M. Connolly, 2010. Temporal dynamics of fish assemblages of natural and artificial tropical estuaries. Marine Ecology Progress Series 410: 143–157.
Sklar, F. H. & J. A. Browder, 1998. Coastal environmental impacts brought about by alterations to freshwater flow in the Gulf of Mexico. Environmental Management 22: 547–562.
Stevens, P. W., M. F. D. Greenwood, C. F. Idelberger & D. A. Blewett, 2010. Mainstem and backwater fish assemblages in the tidal Caloosahatchee River: implications for freshwater inflow studies. Estuaries and Coasts 5: 1216–1224.
Stevens, P. W., M. F. D. Greenwood & D. A. Blewett, 2013. Fish assemblages in the oligohaline stretch of a southwest Florida river during periods of extreme freshwater inflow variation. Transactions of the American Fisheries Society 142: 1644–1658.
Tsou, T. S. & R. E. Matheson, 2002. Seasonal changes in the nekton community of the Suwannee River estuary and the potential impacts of freshwater withdrawal. Estuaries 25: 1372–1381.
Visintainer, T. A., S. M. Bollens & C. S. Simenstad, 2006. Community composition and diet of fishes as a function of tidal channel geomorphology. Marine Ecology Progress Series 321: 227–243.
Vörösmarty, C. J., P. Green, J. Salisbury & R. B. Lammers, 2000. Global water resources: vulnerability from climate change and population growth. Science 289: 284–288.
Walters, A. W. & D. M. Post, 2011. How low can you go? Impacts of low-flow disturbance on aquatic insect communities. Ecological Applications 21: 163–174.
Walters, D. M., D. S. Leigh & A. B. Bearden, 2003. Urbanization, sedimentation, and the homogenization of fish assemblages in the Etowah River Basin, USA. Hydrobiologia 494: 5–10.
Acknowledgements
The authors thank the staff of the Florida Fish & Wildlife Conservation Commission’s Charlotte Harbor Field Laboratory for sample collection and logistics. Comments from two anonymous reviewers improved the quality of this manuscript. Field sampling was supported by the South Florida Water Management District (CP040063), the Florida Department of Environmental Protection Coastal Management Program (CZ906), funds collected from the state of Florida saltwater fishing license sales, and the Department of the Interior, US Fish & Wildlife Service Federal Aid for Sport Fish Restoration Grant Number F-43. Additional support was provided through University of Windsor scholarships to J.A. Olin and Natural Sciences & Engineering Research Council (NSERC) of Canada Discovery grant funds to A.T. Fisk.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: M. Power
Rights and permissions
About this article
Cite this article
Olin, J.A., Stevens, P.W., Rush, S.A. et al. Loss of seasonal variability in nekton community structure in a tidal river: evidence for homogenization in a flow-altered system. Hydrobiologia 744, 271–286 (2015). https://doi.org/10.1007/s10750-014-2083-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-014-2083-2