Abstract
Exercise-based cardiac rehabilitation (CR) is an evidence-based recommendation for patients with stable heart failure (HF). Less clear is how effective exercise-based CR is for women with HF. The aim of this review was to synthesize the evidence for the effects of exercise-based CR on mortality, hospitalizations, exercise capacity, and quality of life (QOL) among women with HF. We identified 18 studies comprising 4917 patients, of which 1714 were women. The interventions evaluated consisted of various combinations of supervised in-hospital and out-patient sessions as well as home-based programs that included aerobic (walking, treadmill, bicycle) and resistance training. The interventions ranged from 12 to 54 weeks, with a frequency of 2–7 sessions per week, lasting from 30 to 105 min per session. Because of a paucity of sex-specific analyses of the outcomes, it was not possible to draw conclusions for women. There was limited evidence for mortality benefit for men or women participating in exercise-based CR. There was more substantial evidence for reductions in hospitalizations for the participants. Generally, exercise training improved exercise capacity. The effects of exercise-based CR on QOL were mixed with most studies favoring CR at 3 months but not at 4, 5, and 12 months. Moreover, generally, the physical dimension of QOL but not the mental dimensions improved. Recommendations for future research to reduce the gap in knowledge about the effects of exercise-based CR for women are offered.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Heart failure (HF) is a persistent health challenge affecting 6.7 million Americans [1]. Prevalence of HF is predicted to exceed 8 million Americans by 2030, owing to an aging population and contemporary therapeutic advances that have improved survival [2, 3]. The economic burden of HF is projected to increase to $69.8 billion in 2030 largely due to unplanned hospitalizations [1]. Between 2015 and 2018, HF was the leading cause of hospitalization, morbidity, and mortality in women accounting for 3 million women living with HF; 44,958 deaths among women were attributed to HF in 2020 [1]. Heart failure subtypes include HF with reduced EF (HFrEF, EF ≤ 40%), mildly reduced EF (HFmrEF, 41–49%), and preserved EF (HFpEF, ≥ 50%) [1, 2]. Among 335,735 patients in the American Heart Association (AHA) Get with the Guidelines HF registry (GWTG-HF) across three time periods (2005–2009, 2010–2014, and 2015–2018), 49.3%, 48.5%, and 46.6% of patients, respectively, were women, of which 68% were White and 19% Black [4]. Although the incidence of HF is similar for women and men, there are phenotypic sex differences with more women than men presenting with HFpEF particularly at older ages [5,6,7,8,9]. Moreover, Black postmenopausal women have the worst prognosis after HF develops [1, 10]. Sex differences across the HF trajectory include risk factors, etiology, guideline-based therapies, receipt of secondary prevention services, and clinical outcomes [8, 11].
Exercise-based cardiac rehabilitation (CR) provides comprehensive secondary prevention services and improves outcomes among patients with cardiovascular disease including a small reduction in all-cause mortality, substantial reduction in all-cause hospitalization, and improved quality of life (QOL) with up to 12 months’ follow-up [12]. However, women represent less than 15% of participants in randomized clinical trials (RCTs) of exercise-based CR [12]. Evidence-based guidelines endorse CR and exercise training as safe and effective for medically stable patients, including women with HFrEF, to improve exercise capacity, clinical outcomes, and QOL [2, 13,14,15]. Furthermore, the most functionally impaired patients often achieve the greatest improvements with CR [16, 17].
Despite the undisputed benefits of CR, only 25% of older patients eligible for CR participate [18, 19]; less than 10% of eligible HF patients receive a CR referral [20]. Inequity in access and quality of healthcare in women is well documented in the USA [21]. Referral, participation, and completion rates are very low in older women especially among ethnic minorities and economically constrained subgroups [22,23,24,25,26,27]. Women participate less often than men across all ages but because women have a longer life trajectory than men, these gaps tend to worsen among older adults [28]. Barriers to CR attendance include the lack of understanding among patients and clinicians of the value of CR, prohibitive logistics particularly because many older women cannot drive, unaffordable copayments for CR for patients, and inflexible delivery formats to achieve personalized CR interventions tailored to the varied needs and circumstances of women [29].
Several studies examining the recruitment of women in HF RCTs reported that enrollment of women varied between 21 and 29% [30,31,32]. A review found that only 25% of participants in RCTs of HFrEF were women; women were under-enrolled relative to the population sex distribution of HF in more than 70% of the RCTs, with no significant improvement over time [33]. The underrepresentation of women in HF RCTs limits the ability to analyze sex-specific efficacy and safety of secondary prevention interventions. The aim of this review was to evaluate the effect of exercise-based CR on the outcomes of mortality, hospitalizations, exercise capacity, and QOL among women with HF. We also examined the referral, enrollment, and adherence of women with HF to CR in observational studies. Finally, we offer recommendations for future research.
Methods
Search strategy and study selection
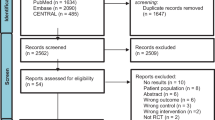
We utilized the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [34] when obtaining relevant literature regarding the health outcomes among women with HF participating in RCTs of exercise-based CR (Fig. 1). Our search strategy included a systematic review of the literature across three databases (PubMed, CINAHL, and Web of Science). Within each database, a string of Medical Subject Heading (MeSH) terms was utilized (Supplemental Material Table SM1). The complete database of retrieved articles was screened initially for relevance by title and abstract. Potentially relevant articles were further screened via full-text review. The resulting search retrieved n = 380 articles for review. After screening for relevant titles and abstracts, n = 82 records were reviewed by full text. After full-text review, 18 studies were included in the final review. A summary of the included studies is provided in Supplemental Material Table SM2 (Table SM2).
Inclusion criteria included English language publications between 2009 and 2022, specifically addressing RCTs that evaluated outcomes or adherence either in the primary outcomes or in subgroup analyses of women diagnosed with all HF subtypes in exercise training and exercise-based CR. Outcomes evaluated included mortality (all-cause and cardiovascular), hospitalizations (all-cause and cardiovascular), exercise capacity, and QOL. We excluded studies that did not include women with HF, pilot studies, case reports, review articles, and expert opinion editorials. We sought to exclude studies without sex-specific primary or subgroup analyses on the outcome variables reported, but there were so few such studies that we retained all studies that included women.
Results
Study characteristics
Of the 18 studies included in this review, 6 were conducted in the USA [35,36,37,38,39,40]; 3 in Australia [41,42,43]; 2 each in Germany [44, 45], Italy [46, 47], and the UK [48, 49]; and 1 each in Norway [50], Greece [51], and Spain [52]. Nine studies were multi-center trials [35, 39, 40, 42, 44, 45, 47, 49, 50]. Follow-up of outcomes ranged from upon completion of the intervention at 3 months to up to 10 years.
Participant characteristics
The 18 RCTs enrolled 4917 patients, of which 1714 were women. The number of participants in each of the studies ranged from 25 to 2331 while the number of women ranged from 15 to 661. Women comprised between 21% (n = 15) [51] and 81% (n = 81) [38] of study participants. One large multi-center RCT, Heart Failure: A Controlled Trial Investigating Outcomes of exercise Training (HF-ACTION), recruited not only the largest proportion of HF participants (51%) but also the greatest number of women (n = 661) [35]. In seven studies, ≥ 50% of the participants were women [36,37,38, 40, 44, 45, 52]. Only 2 trials conducted pre-specified subgroup analyses of women [40, 53]. Participant age ranged from about 59 to 83 years. Nine studies included patients with HFrEF [35, 39, 41, 46,47,48,49,50,51] and twelve studies included patients with HFpEF [36,37,38,39,40,41,42,43,44,45, 47, 52]. In general, enrollment of women in HFpEF trials was greater than that in HFrEF trials reflecting the preponderance of HFpEF among women.
Interventions
Eight studies described testing a comprehensive multidisciplinary CR intervention [38,39,40,41,42, 46, 48, 49] and all studies implemented various exercise training protocols (see Table SM2 for details). Six studies implemented both aerobic and resistance exercise training [39,40,41,42, 44, 48]. Several interventions included a home-based CR component [35, 39,40,41, 47] and two studies implemented entirely home-based interventions [48, 49]. Davidson et al. incorporated a structured HF-specific disease management component into CR [41]. Similarly, Mudge et al. provided study participants in both groups with a HF disease management program and a home exercise program whereas the CR participants (experimental group) were offered an additional 24 weeks of supervised center-based exercise training [42]. Three studies implemented high-intensity interval training (HITT) [45, 50, 51] while Palau et al. implemented inspiratory muscle training over 3 months [52]. Kitzman et al. examined the independent effects of a calorie restricted diet or aerobic exercise training as well as the combined effect of a diet and exercise intervention [38]. Kitzman and colleagues subsequently implemented an early, transitional, tailored progressive CR intervention focused on multiple physical function domains for older patients who were hospitalized for acute decompensated HF [40]. The dose of aerobic exercise training, and thus, the total volume of exercise, varied widely across the studies with single session durations of 30–105 min, session frequencies of 2–7 sessions per week, and total program durations of 12–54 weeks.
Outcomes
Composite mortality and hospitalization
Four studies reported data on the composite outcome of all-cause mortality and hospitalization, all of which were not statistically significantly different between groups (Table 1): O’Connor et al. [35] (HR 0.93, 95% CI 0.84–1.02, p = 0.13), Jolly et al. [48] (HR 1.45, 95% CI 0.43–4.86), Mudge et al. [42] (OR 0.80 95% CI 0.49–1.30), and Kitzman et al. [40] (rate ratio 0.93, 95% CI 0.77–1.12). Only one study reported a HR for the primary endpoint of all-cause mortality and hospitalization by sex: women (HR 0.83, 95% CI 0.68–1.00), men (HR 0.97, 95% CI 0.871.09), with a nonsignificant treatment-by-sex interaction (p = 0.17) [35]. After adjustment for significant covariates, women in the exercise training group had a significant 26% reduction in all-cause mortality or all-cause hospitalization (HR 0.74, 95% CI 0.59–0.91) compared to no reduction in men (HR 0.99, 95% CI 0.86–1.13) with a significant treatment-by-sex interaction (p = 0.027) [53].
All-cause mortality
Five studies provided data for all-cause mortality with follow-up periods from 6 to 120 months [35, 40,41,42, 49]. After 30 months, the HF-ACTION trial reported nonsignificant reductions in mortality in the CR group versus the UC group (HR 0.96, 95% CI 0.79–1.17, p = 0.70) [35]. Dahal et al. reported no between-group differences in mortality (HR 1.01, 95% CI 0.26–3.92) [49] nor did Kitzman and colleagues (rate ratio, 1.17; 95% CI 0.61–2.27, p = 0.63). Conversely, Mudge et al. reported significantly fewer deaths at 12 months in the CR group compared with the UC group (p = 0.04) [42]. Moreover, Davidson and colleagues [41] reported that 79% of the UC group and 93% of the CR group were alive at 12 months (OR 3.85, 95% CI 1.03–14.21, p = 0.004) [41].
Cardiovascular mortality
Two studies provided data for cardiovascular mortality with no sex-specific analyses. After 120 months of follow-up, Belardinelli et al. reported that cardiovascular mortality was higher in the UC group compared with the CR group (10 vs 4, HR: 0.68, 95% CI 0.30–0.82, p < 0.001) [46]. Moreover, gender did not predict the outcome. The HF-ACTION trial reported a nonsignificant reduction in the combined endpoint of cardiovascular mortality or cardiovascular hospitalizations in the CR group versus the UC group (HR 0.92; 95% CI 0.83–1.03, p = 0.14). There was also a nonsignificant reduction in cardiovascular mortality or HF hospitalization in the CR group (HR 0.87, 95% CI 0.75–1.00. p = 0.06) [35].
Hospitalizations
All-cause hospitalizations were reported in eight studies [35, 40,41,42, 46,47,48,49]; three studies reported fewer hospitalizations with exercise-based CR [41, 46, 47]. Belardinelli et al. reported 8 hospitalizations in the CR group and 25 in the UC group (HR 0.64, 95% CI 0.34–0.81, p < 0.001) [46]. Another study reported that among participants aged 76.9 ± 5.67, over 6 months, all-cause hospitalizations occurred in 25 (15.2%) in the CR group versus 60 (36.8%) in the UC group (HR 2.91, 95% CI 1.70–4.97, p < 0.001). After adjustment for covariates, CR reduced all-cause hospitalizations by 44.2% (β = 0.558, 95% CI 0.326–0.954, p = 0.033). Participant sex had no effect on hospitalization (β = 0.129, HR 1.138, 95% CI 0.652–1.985, p = 0.650) [47]. Davidson and colleagues reported that at 12 months, compared with the UC group, fewer participants in the CR group were hospitalized (44% vs 69%, p = 0.01). Those in the CR group had lower odds of having any hospital admission (OR 0.20, 95% CI 0.07–0.58, p = 0.01) or having a cardiovascular hospitalization over 12 months (OR 0.12, 95% CI 0.04–0.37) [41]. Dalal and colleagues reported no significant between-group differences in hospitalizations during 12 months of follow-up (OR 0.72, 95% CI 0.35–1.51, p = 0.386) [49]. Mudge and colleagues also found no significant between-group differences at 12 months in all-cause readmissions or HF readmissions [42]. Finally, Kitzman et al. reported that the rate of all-cause rehospitalization at 6 months was not different between groups (rate ratio, 0.93; 95% CI 0.66–1.19) [40].
Cardiopulmonary exercise capacity
Ten studies reported data on cardiopulmonary exercise capacity (VO2peak ml/min/kg) [35,36,37,38, 43,44,45,46, 50, 52]. Most of these (7/10) studies showed an increased exercise capacity resulting from exercise training (Table 2). One study (19% women) showed no benefit in VO2peak of HIIT compared to moderate continuous exercise (MCT); however, 51% of the HIIT group exercised below the prescribed intensity whereas 80% in the MCT group trained at a higher intensity [50]. Moreover, at baseline, the UC group had the highest mean VO2peak. In a second study (50% women), after 4 months of exercise training, VO2peak increased 24.6% (p = 0.02) in HFpEF patients compared with no change in the UC group (5.1%, p = 0.19). However, there were no significant between-group differences (p = 0.06) [43]. Similarly, Mueller and colleagues found no significant change in VO2peak in patients with HFpEF (66.5% women) at 3 or 12 months between the HIIT and MCT groups and neither group met the minimal clinically important difference compared with the UC group [45].
Twelve studies provided data on the 6-min walk test (6MWT) [35,36,37,38,39,40,41,42, 44, 47, 51, 52, 57]. Two studies measuring exercise capacity with the incremental shuttle walk test (ISWT) found no significant between-group differences at 3 or 12 months after CR [48, 49]. Of the 12 studies measuring the 6MWT, five studies found significant improvement with CR over UC at 3 months [35, 44, 47, 51, 52], two studies at 4 months [36, 37], one study at 5 months [38], and one study at 6 months [47]. Three studies that measured the 6MWT at 12 months found no significant between-group differences [35, 39, 41, 57]. Pozehl and colleagues found significant improvement in the 6MWT among the HFpEF subgroup but not the HFrEF subgroup and only at the 18-month follow-up; there were no between-group differences in this subgroup between CR and UC at 6 and 12 months [39, 57].
Quality of life
Most studies measured QOL with either the Kansas City Cardiomyopathy Questionnaire (KCCQ) which measures 5 domains (physical limitation, symptoms, self-efficacy, QOL, social limitations) and generates 3 summary scores (overall summary, clinical summary, total symptom score) [54] or the Minnesota Living with Heart Failure Questionnaire (MLHFQ) [55]. Of the twelve studies measuring QOL with the MLHFQ, six assessed QOL at 3 months [41, 43, 44, 47, 51, 52] and two of these studies found no significant between-group differences [43, 47] (Table 3). Three studies reported MLHFQ scores at 4 months [36,37,38] and only one study found a significant improvement in MLHFQ physical score and no change in the mental score or the total score between groups [36]. Of the two studies that administered the MLHFQ at 6 months, one found significant between-group differences [47] and one did not [48]. Three studies administered the MLHFQ at 12 months, one of which found significant between-group differences [49] and two did not [41, 48]. One Italian study measured QOL using the MLHFQ over 120 months and found significant between-group differences which were sustained over the study period [46].
Six studies measured QOL with the KCCQ, four of which measured this outcome at 3 months [40, 45, 50, 56] and two of which found significant between-group differences [40, 56]. The HF-ACTION trial administered the KCCQ every 3 months for the first year and then annually thereafter. They found significant between-group differences at 3 months in the exercise training group that was sustained throughout the study [56]. One study administered the KCCQ at 4 months and found that only the diet intervention improved scores [38]. Finally, one study administered the KCCQ at 6, 12, and 18 months and found significant between-group differences over time in the HFpEF subgroup only [57].
Adherence to interventions in randomized controlled trials
The dose–response relationship between exercise-based CR participation and outcomes has been well established [58, 59]. Adherence to the CR interventions in the RCTs described in Table SM2, where reported, is difficult to synthesize given the wide range of methods of measuring the dose of exercise received or the number of sessions attended. Understanding the dose of the intervention received is important for interpreting the results and for understanding the acceptability of the intervention by participants. In the HF-ACTION trial, participants exercised a median of 76 min per week at 3 months with the goal of 90 min per week. By 12 months, exercise time decreased to about 74 min per week [35]. Moreover, 50% of the UC group were dissatisfied with their group assignment with the possibility of crossover to exercise. The Heart Camp (HC) trial reported that the intervention group had significantly greater adherence to exercise at 12 months (42%) and 18 months (35%) compared with the enhanced UC group (28% and 19%, respectively) with no significant between-group differences found at 6 months [39]. Mudge et al. reported that there was no association between mortality and rehospitalizations and exercise training attendance (< 8 sessions 21/30 [70%], 8–15 sessions 28/43 [65%], 16–23 sessions 10/30 [49%], ≥ 24 sessions 16/28 [57%]; p = 0.27) [42]. Moreover, they reported that center-based CR adherence was poor at 43%. Conversely, nine studies reported over 80% adherence to their interventions [37, 38, 40, 43,44,45,46, 48, 50].
Cardiac rehabilitation referral and participation in observational studies
Alongside the RCTs reviewed, we identified additional observational studies that conducted sex-specific subgroup analyses of referral to and participation in CR. Among 105,619 participants in the GWG-HF registry, there was no significant difference in referral between the sexes after adjusting for clinically relevant covariates [20]. Conversely, a study from Denmark found that among 33,257 participants, men with HF were significantly less likely to be referred to CR than women with HF (OR 0.85, 95% CI 0.80–0.89) [60]. These findings are consistent with prior findings that Italian women were significantly more likely to be enrolled in CR than men following decompensated HF [61]. In a more contemporary Australian study, women and older patients were significantly less likely to be referred to CR than men (OR: 0.65, 95% CI 0.42–1.02, p = 0.06) [62]. Notably, only women with HFrEF were significantly less likely to be referred to CR than men (OR 0.47, 95% CI 0.21–1.04, p = 0.06), and not women with HFpEF (p = 0.21). Similarly, an investigation found that among 243,208 Medicare beneficiaries, women with HF were less likely to participate in CR than men (1.8% vs 3.7%, p < 0.001) but there was no sex difference among 66,710 veterans [63]. Among Medicare beneficiaries with HF from 2015 to 2016, only 611 (4.3%) of 11,696 HF patients eligible for CR participated within 6 months of HF hospitalization, with lower participation in women versus men (3.3% vs 5%; p < 0.001) and Black versus White patients (2.9% vs 3.7%; p < 0.001) [26]. This sex disparity in CR referral, enrollment, and adherence is consistent with trends established over the past decade [64, 65]. While these contrasting results provide additional insight into sex-specific differences in referral to and participation in CR, the synthesis of these studies should be taken together with the RCTs reviewed herein when drawing generalizations.
Discussion
We reviewed 18 RCTs that included women as participants and that examined the effects of exercise-based cardiac rehabilitation on mortality, hospitalizations, exercise capacity, and QOL among patients with HFpEF and HFrEF. A systematic review was not feasible because only two studies provided any type of sex-specific analyses. Despite this, we found limited evidence for mortality benefit for men or women participating in exercise-based CR. Plausible explanations for limited prognostic effects include underpowered studies with small sample sizes and less than optimal adherence to the exercise training protocol. In the only study reporting sex-specific data, after adjustment for significant covariates, women in the exercise training group in the HF-ACTION trial had a significant 26% reduction of all-cause death or all-cause hospitalization compared with no reduction in men. Our review of RCTs found that three of eight studies reported reductions in rehospitalizations among the intervention group. Generally, exercise training improved cardiopulmonary exercise capacity as measured by VO2peak and the 6MWT. The influence of exercise-based CR on QOL was mixed with most studies favoring CR at 3 months but generally not at 4, 6, and 12 months which is typical of the fading effects when interventions are withdrawn. Moreover, generally, the physical dimension of QOL but not the mental dimensions improved.
A previous review of 44 trials of exercise-based CR for HF patients (n = 5783) reported that 21 trials (n = 2182) demonstrated reduction in overall hospital admissions at 12 months of follow-up (CR 180/1093 [16.5%] versus UC 258/1089 [23.7%]; RR 0.70, 95% CI 0.60–0.83) but no difference in all-cause mortality [66]. Long and colleagues also reported that 14 trials (n = 1114) demonstrated reductions in HF-specific hospitalization (CR 40/562 [7.1%] versus UC 61/552 [11.1%], RR 0.59, 95% CI 0.42–0.84). Improved QOL (MLFHQ) was reported in 17 trials (n = 1995) (mean difference (MD) − 7.11 points, 95% CI − 10.49 to − 3.73) [66]. Another review of 25 RCTs (n = 4481) that included only HFrEF patients reported no benefit for hospitalizations or mortality with exercise-based CR [67]. One systematic review found that exercise training improved physical performance and cardiorespiratory outcomes in patients with HFpEF [68]. Similarly, a meta-analysis of 8 RCTs with 436 HFpEF patients found that exercise training improved VO2peak (weighted MD, 95% CI, 1.660 [0.973–2.348] ml/kg/min), 6MWD (33.883 [12.384–55.381] m), and MLHFQ total score (9.059 [3.083–15.035] points) compared with UC [69]. They also observed that exercise training did not significantly improve mental or emotional dimensions of QOL in HFpEF patients. Without exception, these reviews were unable to report on sex-specific outcomes.
The most glaring deficiency of the studies reviewed was the lack of sex-specific data analyses. Despite the stipulation of the National Institutes of Health Revitalization Act to include women and men in RCTs proportionate to the sex-specific prevalence of the disease, women are under-represented in HF RCTs [8, 11, 70]. Morgan and colleagues identified 146 HF RCTs including 248,620 patients, of which only 25.8% were women. Women were least likely to be enrolled in trials recruiting patients with ischemic cardiomyopathy (17.9%) and severe systolic dysfunction (EF < 35%) (21.4%), and those involving an invasive procedure (21.1%). The highest proportion of women was enrolled in RCTs assessing HFpEF (51.6%) and trials including older participants (40.5%) [70]. This underrepresentation has been partly attributed to age biases because HF is predominately present in older women. Others point to the numerous barriers women encounter when approached to participate in RCTs including transportation, caregiving responsibilities, lack of perceived benefit, and poor health literacy about HF trials [71]. Notably, RCTs led by women are more likely to enroll representative samples of women [72, 73]. Given the paucity of data on the effects of exercise-based CR among women with HF, we offer recommendations for future research (Table 4).
Recommendations for research
Under-enrollment of women with HF in secondary prevention RCTs deprives them of the benefits of trial participation, including guideline-directed medical therapy, close clinical follow-up, and better health outcomes. Women should be included in RCTs in proportion to the sex-specific distribution of HF. Strategies for including more women in RCTs include removing upper and lower age limits in exclusion criteria for enrollment in trials, training clinicians and research personnel on the importance of enrolling women, targeting important stakeholders in the community to increase awareness and importance of enrolling women, and removing barriers to enrollment to make participation convenient [8, 71]. Additionally, given that racially and ethnically diverse women present with unique biological and social determinants of health, studies that evaluate the intersection between race and sex warrant further investigation as well as the possible role of both implicit and explicit biases in CR enrollment and participation [8, 71]. The health literacy and cognitive function of participants should be evaluated during recruitment and intervention implementation so that research materials and instructions can be tailored to their needs [17]. Medical education and training on sex and gender diverse cardiovascular needs will better prepare our future scientists. The sex-specific manifestations of HF and response to treatments among cis- and gender diverse individuals warrant additional research [74]. Finally, study participants should be included as equal partners and stakeholders in designing the recruitment process and messaging, the informed consenting process, the research questions, the interventions, and the selection of meaningful and relevant patient-reported outcomes.
Many of the RCTs reviewed herein primarily examined the effects of the exercise training aspect of secondary prevention. Cardiac rehabilitation for patients with HF is a complex, multidisciplinary intervention for which a standardized dose or delivery format may not be feasible or acceptable given the unique needs, skills, capacities, competencies, exercise capacity, motivation, and preferences of women compared to men [29, 75]. Given that many individuals with HF are elderly and frail and have multiple comorbidities such as diabetes, depression, and cognitive impairment, and limited or no social support, there is a need for more holistic approaches to designing rehabilitation interventions that address these issues among women for improved uptake and adherence. The mechanisms by which varied exercise-based secondary prevention interventions work for whom, when, and why are unclear, and more nuanced approaches for women are needed for this to be elucidated [76]. Research on effective multifaceted interventions to address the sex and gender gaps in HF outcomes is needed. Mixed methods research should be used not only to measure intervention outcomes but also to understand the complexities of what intervention components work for whom, when, and why.
Studies of secondary prevention interventions for women with HF should be co-designed with diverse patients so that they are tailored to their needs in terms of essential components, mode of delivery, frequency, and setting, and then rigorously tested in RCTs. While exercise training is central to secondary prevention for patients with HF, other components are equally essential including education, diet and nutrition counseling, and psychosocial counseling [77]. Theory-based behavior change techniques, which consider the multiple factors that influence health behaviors, are important for improving adherence to interventions. The perceptions, motivations, skills, and the social environment are key influences on the behavior of participants in our studies. Knowledge of behavior change theory and the processes by which behavior change occurs helps us design interventions that lead to meaningful improvement in health behaviors. Delivering comprehensive CR (e.g., education, counseling, support, skill building) has been found to be superior to providing only selected core components such as exercise training [78]. A systematic review found that exercise training, psychosocial management, and risk factor modification each contributed directly to the effectiveness of CR [79]. Women-focused CR interventions may better engage women [80], might result in better psychosocial outcomes and QOL than traditional programs [29, 81,82,83], and should be tested in RCTS with women with HF. Ghisi and colleagues provide a clinical practice guideline that offers 15 recommendations for improving referral of women to CR, for adjustments to the mode of exercise training delivery in CR, and for women-focused education and counseling. This guideline specifically recommends that women are provided with greater choice of their preferences for modes of exercise training and the setting in which it is delivered [81]. Additional research is needed to examine the efficacy of such interventions for women with HF.
Many examples of new delivery models of CR are emerging; some of which might be most beneficial for women with HF. Dalal and colleagues tested a novel, tailored, home-based HF self-management program for patients with HF and their caregivers [49]. However, only 22% of the participants were women and it would be beneficial to examine such an intervention in a study with a greater proportion of women using sex-specific analyses. Home-based and technology-enhanced, patient-centered models of CR with appropriate quality assurance as an alternative or adjunct to traditional, center-based programs are needed to improve access to CR [27, 84]. A home-based strategy to CR may overcome various barriers contributing to underutilization [85] especially for women with HF. A Cochrane review of 23 trials of 2890 participants in CR demonstrated that home-based programs had similar efficacy as center-based programs with no significant group differences in mortality, exercise capacity, or QOL [86]. Increasingly, people 65 years of age and older are using smartphones which may enhance CR with features such as messaging and two-way video capabilities as well as accelerometers to track steps [27]. However, caution is warranted because individuals with poor health and technology literacy and who are non-English speaking, women, and older adults especially in rural areas may have difficulty engaging in technology-enhanced CR [87].
For research findings to be maximally interpretable and generalizable, sex-specific analyses in research and RCT design should be the standard, not the exception. Future HF clinical trials should be powered to investigate the substantial heterogeneity of the female HFpEF population in terms of age, ejection fraction, comorbidities, and frailty. For example, in a study of 295 patients with HFpEF (59% women), women had worse peripheral oxygen extraction with exercise, worse right ventricular and left ventricular systolic reserve, and worse diastolic reserve compared with men [88]. Scientific journals could also require authors to address sex and gender differences to publish manuscripts.
Limitations
We intended to conduct a systematic review of the effects of exercise-based cardiac rehabilitation on relevant health outcomes among women with HF. Due to the paucity of sex-specific data in the studies reviewed, this goal was not realized. Moreover, most of the studies included were small. We also included studies of patients with all subtypes of HF and with heterogenous characteristics, comorbidities, and cardiorespiratory fitness. Finally, study interventions were diverse with variable adherence by participants which likely influenced the outcomes evaluated.
Conclusion
The review of RCTs examining the effects of exercise-based cardiac rehabilitation of patients with HF provided very little sex-specific information for which to conclude exercise-based CR is effective for women with HF. There remain significant gaps in our knowledge about the best strategies for recruiting more women to clinical trials, the appropriate intervention components most effective for women with HF, and how the health outcomes of women differ from men after these interventions. More creative, flexible, and individualized, preference-based interventions that exploit the appropriate use of digital technologies may fill the existing gaps and lead to sex-specific clinical practice guidelines. Significantly more research is warranted.
Availability of data and materials
This declaration is not relevant to the content of the submitted work.
References
Tsao CW, Aday AW, Almarzooq ZI, Anderson CAM, Arora P, Avery CL, Baker-Smith CM, Beaton AZ, Boehme AK, Buxton AE, Commodore-Mensah Y, Elkind MSV, Evenson KR, Eze-Nliam C, Fugar S, Generoso G, Heard DG, Hiremath S, Ho JE, Kalani R, Kazi DS, Ko D, Levine DA, Liu J, Ma J, Magnani JW, Michos ED, Mussolino ME, Navaneethan SD, Parikh NI, Poudel R, Rezk-Hanna M, Roth GA, Shah NS, St-Onge MP, Thacker EL, Virani SS, Voeks JH, Wang NY, Wong ND, Wong SS, Yaffe K, Martin SS, American Heart Association Council on E, Prevention Statistics C, Stroke Statistics S (2023) Heart disease and stroke statistics-2023 update: a report from the American Heart Association. Circulation. https://doi.org/10.1161/CIR.0000000000001123
Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM, Deswal A, Drazner MH, Dunlay SM, Evers LR, Fang JC, Fedson SE, Fonarow GC, Hayek SS, Hernandez AF, Khazanie P, Kittleson MM, Lee CS, Link MS, Milano CA, Nnacheta LC, Sandhu AT, Stevenson LW, Vardeny O, Vest AR, Yancy CW (2022) 2022 AHA/ACC/HFSA guideline for the management of heart failure: executive summary: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol 79:1757–1780. https://doi.org/10.1016/j.jacc.2021.12.011
Urbich M, Globe G, Pantiri K, Heisen M, Bennison C, Wirtz HS, Di Tanna GL (2020) A systematic review of medical costs associated with heart failure in the USA (2014–2020). Pharmacoeconomics 38:1219–1236. https://doi.org/10.1007/s40273-020-00952-0
Hamo CE, Fonarow GC, Greene SJ, Vaduganathan M, Yancy CW, Heidenreich P, Lu D, Matsouaka RA, DeVore AD, Butler J (2021) Temporal trends in risk profiles among patients hospitalized for heart failure. Am Heart J 232:154–163. https://doi.org/10.1016/j.ahj.2020.11.015
Ho JE, Enserro D, Brouwers FP, Kizer JR, Shah SJ, Psaty BM, Bartz TM, Santhanakrishnan R, Lee DS, Chan C, Liu K, Blaha MJ, Hillege HL, van der Harst P, van Gilst WH, Kop WJ, Gansevoort RT, Vasan RS, Gardin JM, Levy D, Gottdiener JS, de Boer RA, Larson MG (2016) Predicting heart failure with preserved and reduced ejection fraction: the international collaboration on heart failure subtypes. Circ Heart Fail 9. https://doi.org/10.1161/CIRCHEARTFAILURE.115.003116
Lala A, Tayal U, Hamo CE, Youmans Q, Al-Khatib SM, Bozkurt B, Davis MB, Januzzi J, Mentz R, Sauer A, Walsh MN, Yancy C, Gulati M (2022) Sex differences in heart failure. J Card Fail 28:477–498. https://doi.org/10.1016/j.cardfail.2021.10.006
van Ommen A, Canto ED, Cramer MJ, Rutten FH, Onland-Moret NC, Ruijter HMD (2022) Diastolic dysfunction and sex-specific progression to HFpEF: current gaps in knowledge and future directions. BMC Med 20:496. https://doi.org/10.1186/s12916-022-02650-4
Khan SS, Beach LB, Yancy CW (2022) Sex-based differences in heart failure: JACC Focus Seminar 7/7. J Am Coll Cardiol 79:1530–1541. https://doi.org/10.1016/j.jacc.2022.02.013
Pepine CJ, Merz CNB, El Hajj S, Ferdinand KC, Hamilton MA, Lindley KJ, Nelson MD, Quesada O, Wenger NK, Fleg JL, College A, of Cardiology Committee on Cardiovascular Disease in W, (2020) Heart failure with preserved ejection fraction: similarities and differences between women and men. Int J Cardiol 304:101–108. https://doi.org/10.1016/j.ijcard.2020.01.003
Carnethon MR, Pu J, Howard G, Albert MA, Anderson CAM, Bertoni AG, Mujahid MS, Palaniappan L, Taylor HA Jr, Willis M, Yancy CW, Council AHA, on E, Prevention, Council on Cardiovascular Disease in the Y, Council on C, Stroke N, Council on Clinical C, Council on Functional G, Translational B, Stroke C, (2017) Cardiovascular health in African Americans: a scientific statement from the American Heart Association. Circulation 136:e393–e423. https://doi.org/10.1161/CIR.0000000000000534
Sullivan K, Doumouras BS, Santema BT, Walsh MN, Douglas PS, Voors AA, Van Spall HGC (2021) Sex-specific differences in heart failure: pathophysiology, risk factors, management, and outcomes. Can J Cardiol 37:560–571. https://doi.org/10.1016/j.cjca.2020.12.025
Dibben G, Faulkner J, Oldridge N, Rees K, Thompson DR, Zwisler AD, Taylor RS (2021) Exercise-based cardiac rehabilitation for coronary heart disease. Cochrane Database Syst Rev 11:CD001800. https://doi.org/10.1002/14651858.CD001800.pub4
Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE Jr, Drazner MH, Fonarow GC, Geraci SA, Horwich T, Januzzi JL, Johnson MR, Kasper EK, Levy WC, Masoudi FA, McBride PE, McMurray JJ, Mitchell JE, Peterson PN, Riegel B, Sam F, Stevenson LW, Tang WH, Tsai EJ, Wilkoff BL, College A, of Cardiology F, American Heart Association Task Force on Practice G, (2013) 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 62:e147-239. https://doi.org/10.1016/j.jacc.2013.05.019
Mosca L, Benjamin EJ, Berra K, Bezanson JL, Dolor RJ, Lloyd-Jones DM, Newby LK, Pina IL, Roger VL, Shaw LJ, Zhao D, Beckie TM, Bushnell C, D’Armiento J, Kris-Etherton PM, Fang J, Ganiats TG, Gomes AS, Gracia CR, Haan CK, Jackson EA, Judelson DR, Kelepouris E, Lavie CJ, Moore A, Nussmeier NA, Ofili E, Oparil S, Ouyang P, Pinn VW, Sherif K, Smith SC Jr, Sopko G, Chandra-Strobos N, Urbina EM, Vaccarino V, Wenger NK (2011) Effectiveness-based guidelines for the prevention of cardiovascular disease in women–2011 update: a guideline from the American Heart Association. Circulation 123:1243–1262. https://doi.org/10.1161/CIR.0b013e31820faaf8
McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Bohm M, Burri H, Butler J, Celutkiene J, Chioncel O, Cleland JGF, Coats AJS, Crespo-Leiro MG, Farmakis D, Gilard M, Heymans S, Hoes AW, Jaarsma T, Jankowska EA, Lainscak M, Lam CSP, Lyon AR, McMurray JJV, Mebazaa A, Mindham R, Muneretto C, Francesco Piepoli M, Price S, Rosano GMC, Ruschitzka F, Kathrine Skibelund A, Group ESCSD (2021) 2021 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J 42:3599–3726. https://doi.org/10.1093/eurheartj/ehab368
Baldasseroni S, Pratesi A, Francini S, Pallante R, Barucci R, Orso F, Burgisser C, Marchionni N, Fattirolli F (2016) Cardiac rehabilitation in very old adults: effect of baseline functional capacity on treatment effectiveness. J Am Geriatr Soc 64:1640–1645. https://doi.org/10.1111/jgs.14239
Yoo BW, Wenger NK (2019) Gender disparities in cardiac rehabilitation among older women: key opportunities to improve care. Clin Geriatr Med 35:587–594. https://doi.org/10.1016/j.cger.2019.07.012
Ritchey MD, Maresh S, McNeely J, Shaffer T, Jackson SL, Keteyian SJ, Brawner CA, Whooley MA, Chang T, Stolp H (2020) Tracking cardiac rehabilitation participation and completion among Medicare beneficiaries to inform the efforts of a national initiative. Circ Cardiovasc Qual Outcomes 13:e005902
de Araujo S, Pio C, Beckie TM, Varnfield M, Sarrafzadegan N, Babu AS, Baidya S, Buckley J, Chen SY, Gagliardi A, Heine M, Khiong JS, Mola A, Radi B, Supervia M, Trani MR, Abreu A, Sawdon JA, Moffatt PD, Grace SL (2020) Promoting patient utilization of outpatient cardiac rehabilitation: a joint International Council and Canadian Association of Cardiovascular Prevention and Rehabilitation position statement. Int J Cardiol 298:1–7. https://doi.org/10.1016/j.ijcard.2019.06.064
Golwala H, Pandey A, Ju C, Butler J, Yancy C, Bhatt DL, Hernandez AF, Fonarow GC (2015) Temporal trends and factors associated with cardiac rehabilitation referral among patients hospitalized with heart failure: findings from Get With The Guidelines-Heart Failure registry. J Am Coll Cardiol 66:917–926. https://doi.org/10.1016/j.jacc.2015.06.1089
Vogel B, Acevedo M, Appelman Y, Bairey Merz CN, Chieffo A, Figtree GA, Guerrero M, Kunadian V, Lam CSP, Maas A, Mihailidou AS, Olszanecka A, Poole JE, Saldarriaga C, Saw J, Zuhlke L, Mehran R (2021) The Lancet women and cardiovascular disease Commission: reducing the global burden by 2030. Lancet 397:2385–2438. https://doi.org/10.1016/S0140-6736(21)00684-X
Thomas RJ, Balady G, Banka G, Beckie TM, Chiu J, Gokak S, Ho PM, Keteyian SJ, King M, Lui K, Pack Q, Sanderson BK, Wang TY (2018) 2018 ACC/AHA clinical performance and quality measures for cardiac rehabilitation: a report of the American College of Cardiology/American Heart Association Task Force on Performance Measures. Circ Cardiovasc Qual Outcomes 11:e000037. https://doi.org/10.1161/HCQ.0000000000000037
Peters AE, Keeley EC (2017) Trends and predictors of participation in cardiac rehabilitation following acute myocardial infarction: data from the Behavioral Risk Factor Surveillance System. J Am Heart Assoc 7. https://doi.org/10.1161/JAHA.117.007664
Sun EY, Jadotte YT, Halperin W (2017) Disparities in cardiac rehabilitation participation in the United States: a systematic review and meta-analysis. J Cardiopulm Rehabil Prev 37:2–10. https://doi.org/10.1097/HCR.0000000000000203
Oosenbrug E, Marinho RP, Zhang J, Marzolini S, Colella TJ, Pakosh M, Grace SL (2016) Sex differences in cardiac rehabilitation adherence: a meta-analysis. Can J Cardiol 32:1316–1324. https://doi.org/10.1016/j.cjca.2016.01.036
Pandey A, Keshvani N, Zhong L, Mentz RJ, Pina IL, DeVore AD, Yancy C, Kitzman DW, Fonarow GC (2021) Temporal trends and factors associated with cardiac rehabilitation participation among Medicare beneficiaries with heart failure. JACC Heart Fail 9:471–481. https://doi.org/10.1016/j.jchf.2021.02.006
Beatty AL, Beckie TM, Dodson J, Goldstein CM, Hughes JW, Kraus WE, Martin SS, Olson TP, Pack QR, Stolp H, Thomas RJ, Wu WC, Franklin BA (2023) A new era in cardiac rehabilitation delivery: research gaps, questions, strategies, and priorities. Circulation 147:254–266. https://doi.org/10.1161/CIRCULATIONAHA.122.061046
Supervia M, Medina-Inojosa JR, Yeung C, Lopez-Jimenez F, Squires RW, Perez-Terzic CM, Brewer LC, Leth SE, Thomas RJ (2017) Cardiac rehabilitation for women: a systematic review of barriers and solutions. Mayo Clin Proc. https://doi.org/10.1016/j.mayocp.2017.01.002
Mamataz T, Ghisi GLM, Pakosh M, Grace SL (2021) Nature, availability, and utilization of women-focused cardiac rehabilitation: a systematic review. BMC Cardiovasc Disord 21:459. https://doi.org/10.1186/s12872-021-02267-0
Reza N, Gruen J, Bozkurt B (2022) Representation of women in heart failure clinical trials: barriers to enrollment and strategies to close the gap. Am Heart J Plus 13. https://doi.org/10.1016/j.ahjo.2022.100093
Tahhan AS, Vaduganathan M, Greene SJ, Fonarow GC, Fiuzat M, Jessup M, Lindenfeld J, O’Connor CM, Butler J (2018) Enrollment of older patients, women, and racial and ethnic minorities in contemporary heart failure clinical trials: a systematic review. JAMA Cardiol 3:1011–1019. https://doi.org/10.1001/jamacardio.2018.2559
Jin X, Chandramouli C, Allocco B, Gong E, Lam CSP, Yan LL (2020) Women’s participation in cardiovascular clinical trials from 2010 to 2017. Circulation 141:540–548. https://doi.org/10.1161/CIRCULATIONAHA.119.043594
Whitelaw S, Sullivan K, Eliya Y, Alruwayeh M, Thabane L, Yancy CW, Mehran R, Mamas MA, Van Spall HGC (2021) Trial characteristics associated with under-enrolment of females in randomized controlled trials of heart failure with reduced ejection fraction: a systematic review. Eur J Heart Fail 23:15–24. https://doi.org/10.1002/ejhf.2034
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J, Moher D (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ 339:b2700. https://doi.org/10.1136/bmj.b2700
O’Connor CM, Whellan DJ, Lee KL, Keteyian SJ, Cooper LS, Ellis SJ, Leifer ES, Kraus WE, Kitzman DW, Blumenthal JA, Rendall DS, Miller NH, Fleg JL, Schulman KA, McKelvie RS, Zannad F, Pina IL, Investigators H-A (2009) Efficacy and safety of exercise training in patients with chronic heart failure: HF-ACTION randomized controlled trial. JAMA 301:1439–1450. https://doi.org/10.1001/jama.2009.454
Kitzman DW, Brubaker PH, Morgan TM, Stewart KP, Little WC (2010) Exercise training in older patients with heart failure and preserved ejection fraction: a randomized, controlled, single-blind trial. Circ Heart Fail 3:659–667. https://doi.org/10.1161/CIRCHEARTFAILURE.110.958785
Kitzman DW, Brubaker PH, Herrington DM, Morgan TM, Stewart KP, Hundley WG, Abdelhamed A, Haykowsky MJ (2013) Effect of endurance exercise training on endothelial function and arterial stiffness in older patients with heart failure and preserved ejection fraction: a randomized, controlled, single-blind trial. J Am Coll Cardiol 62:584–592. https://doi.org/10.1016/j.jacc.2013.04.033
Kitzman DW, Brubaker P, Morgan T, Haykowsky M, Hundley G, Kraus WE, Eggebeen J, Nicklas BJ (2016) Effect of caloric restriction or aerobic exercise training on peak oxygen consumption and quality of life in obese older patients with heart failure with preserved ejection fraction: a randomized clinical trial. JAMA 315:36–46. https://doi.org/10.1001/jama.2015.17346
Pozehl BJ, McGuire R, Duncan K, Kupzyk K, Norman J, Artinian NT, Deka P, Krueger SK, Saval MA, Keteyian SJ (2018) Effects of the HEART Camp trial on adherence to exercise in patients with heart failure. J Card Fail 24:654–660. https://doi.org/10.1016/j.cardfail.2018.06.007
Kitzman DW, Whellan DJ, Duncan P, Pastva AM, Mentz RJ, Reeves GR, Nelson MB, Chen H, Upadhya B, Reed SD, Espeland MA, Hewston L, O’Connor CM (2021) Physical rehabilitation for older patients hospitalized for heart failure. N Engl J Med 385:203–216. https://doi.org/10.1056/NEJMoa2026141
Davidson PM, Cockburn J, Newton PJ, Webster JK, Betihavas V, Howes L, Owensby DO (2010) Can a heart failure-specific cardiac rehabilitation program decrease hospitalizations and improve outcomes in high-risk patients? Eur J Cardiovasc Prev Rehabil 17:393–402. https://doi.org/10.1097/HJR.0b013e328334ea56
Mudge AM, Denaro CP, Scott AC, Meyers D, Adsett JA, Mullins RW, Suna JM, Atherton JJ, Marwick TH, Scuffham P, O’Rourke P (2018) Addition of supervised exercise training to a post-hospital disease management program for patients recently hospitalized with acute heart failure: the EJECTION-HF randomized phase 4 trial. JACC Heart Fail 6:143–152. https://doi.org/10.1016/j.jchf.2017.11.016
Smart NA, Haluska B, Jeffriess L, Leung D (2012) Exercise training in heart failure with preserved systolic function: a randomized controlled trial of the effects on cardiac function and functional capacity. Congest Heart Fail 18:295–301. https://doi.org/10.1111/j.1751-7133.2012.00295.x
Edelmann F, Gelbrich G, Dungen HD, Frohling S, Wachter R, Stahrenberg R, Binder L, Topper A, Lashki DJ, Schwarz S, Herrmann-Lingen C, Loffler M, Hasenfuss G, Halle M, Pieske B (2011) Exercise training improves exercise capacity and diastolic function in patients with heart failure with preserved ejection fraction: results of the Ex-DHF (Exercise training in Diastolic Heart Failure) pilot study. J Am Coll Cardiol 58:1780–1791. https://doi.org/10.1016/j.jacc.2011.06.054
Mueller S, Winzer EB, Duvinage A, Gevaert AB, Edelmann F, Haller B, Pieske-Kraigher E, Beckers P, Bobenko A, Hommel J, Van de Heyning CM, Esefeld K, von Korn P, Christle JW, Haykowsky MJ, Linke A, Wisloff U, Adams V, Pieske B, van Craenenbroeck EM, Halle M, OptimEx-Clin Study G (2021) Effect of high-intensity interval training, moderate continuous training, or guideline-based physical activity advice on peak oxygen consumption in patients with heart failure with preserved ejection fraction: a randomized clinical trial. JAMA 325:542–551. https://doi.org/10.1001/jama.2020.26812
Belardinelli R, Georgiou D, Cianci G, Purcaro A (2012) 10-year exercise training in chronic heart failure: a randomized controlled trial. J Am Coll Cardiol 60:1521–1528. https://doi.org/10.1016/j.jacc.2012.06.036
Antonicelli R, Spazzafumo L, Scalvini S, Olivieri F, Matassini MV, Parati G, Del Sindaco D, Gallo R, Lattanzio F (2016) Exercise: a “new drug” for elderly patients with chronic heart failure. Aging (Albany NY) 8:860–872. https://doi.org/10.18632/aging.100901
Jolly K, Taylor RS, Lip GY, Davies M, Davis R, Mant J, Singh S, Greenfield S, Ingram J, Stubley J, Bryan S, Stevens A (2009) A randomized trial of the addition of home-based exercise to specialist heart failure nurse care: the Birmingham Rehabilitation Uptake Maximisation study for patients with Congestive Heart Failure (BRUM-CHF) study. Eur J Heart Fail 11:205–213. https://doi.org/10.1093/eurjhf/hfn029
Dalal HM, Taylor RS, Jolly K, Davis RC, Doherty P, Miles J, van Lingen R, Warren FC, Green C, Wingham J, Greaves C, Sadler S, Hillsdon M, Abraham C, Britten N, Frost J, Singh S, Hayward C, Eyre V, Paul K, Lang CC, Smith K (2019) The effects and costs of home-based rehabilitation for heart failure with reduced ejection fraction: the REACH-HF multicentre randomized controlled trial. Eur J Prev Cardiol 26:262–272. https://doi.org/10.1177/2047487318806358
Ellingsen O, Halle M, Conraads V, Stoylen A, Dalen H, Delagardelle C, Larsen AI, Hole T, Mezzani A, Van Craenenbroeck EM, Videm V, Beckers P, Christle JW, Winzer E, Mangner N, Woitek F, Hollriegel R, Pressler A, Monk-Hansen T, Snoer M, Feiereisen P, Valborgland T, Kjekshus J, Hambrecht R, Gielen S, Karlsen T, Prescott E, Linke A, Group SHFS (2017) High-intensity interval training in patients with heart failure with reduced ejection fraction. Circulation 135:839–849. https://doi.org/10.1161/CIRCULATIONAHA.116.022924
Chrysohoou C, Tsitsinakis G, Vogiatzis I, Cherouveim E, Antoniou C, Tsiantilas A, Tsiachris D, Dimopoulos D, Panagiotakos DB, Pitsavos C, Koulouris NG, Stefanadis C (2014) High intensity, interval exercise improves quality of life of patients with chronic heart failure: a randomized controlled trial. QJM 107:25–32. https://doi.org/10.1093/qjmed/hct194
Palau P, Dominguez E, Nunez E, Schmid JP, Vergara P, Ramon JM, Mascarell B, Sanchis J, Chorro FJ, Nunez J (2014) Effects of inspiratory muscle training in patients with heart failure with preserved ejection fraction. Eur J Prev Cardiol 21:1465–1473. https://doi.org/10.1177/2047487313498832
Pina IL, Bittner V, Clare RM, Swank A, Kao A, Safford R, Nigam A, Barnard D, Walsh MN, Ellis SJ, Keteyian SJ, Investigators H-A (2014) Effects of exercise training on outcomes in women with heart failure: analysis of HF-ACTION (Heart Failure-A Controlled Trial Investigating Outcomes of Exercise TraiNing) by sex. JACC Heart Fail 2:180–186. https://doi.org/10.1016/j.jchf.2013.10.007
Green CP, Porter CB, Bresnahan DR, Spertus JA (2000) Development and evaluation of the Kansas City Cardiomyopathy Questionnaire: a new health status measure for heart failure. J Am Coll Cardiol 35:1245–1255. https://doi.org/10.1016/s0735-1097(00)00531-3
Rector TS, Cohn JN (1992) Assessment of patient outcome with the Minnesota Living with Heart Failure questionnaire: reliability and validity during a randomized, double-blind, placebo-controlled trial of pimobendan. Pimobendan Multicenter Research Group. Am Heart J 124:1017–1025. https://doi.org/10.1016/0002-8703(92)90986-6
Flynn KE, Pina IL, Whellan DJ, Lin L, Blumenthal JA, Ellis SJ, Fine LJ, Howlett JG, Keteyian SJ, Kitzman DW, Kraus WE, Miller NH, Schulman KA, Spertus JA, O’Connor CM, Weinfurt KP, Investigators H-A (2009) Effects of exercise training on health status in patients with chronic heart failure: HF-ACTION randomized controlled trial. JAMA 301:1451–1459. https://doi.org/10.1001/jama.2009.457
Alonso WW, Kupzyk KA, Norman JF, Lundgren SW, Fisher A, Lindsey ML, Keteyian SJ, Pozehl BJ (2022) The HEART Camp exercise intervention improves exercise adherence, physical function, and patient-reported outcomes in adults with preserved ejection fraction heart failure. J Card Fail 28:431–442. https://doi.org/10.1016/j.cardfail.2021.09.003
Hammill BG, Curtis LH, Schulman KA, Whellan DJ (2010) Relationship between cardiac rehabilitation and long-term risks of death and myocardial infarction among elderly Medicare beneficiaries. Circulation 121:63–70. https://doi.org/10.1161/CIRCULATIONAHA.109.876383
Suaya JA, Stason WB, Ades PA, Normand SL, Shepard DS (2009) Cardiac rehabilitation and survival in older coronary patients. J Am Coll Cardiol 54:25–33. https://doi.org/10.1016/j.jacc.2009.01.078
Thygesen LC, Zinckernagel L, Dalal H, Egstrup K, Glumer C, Gronbaek M, Holmberg T, Kober L, la Cour K, Nakano A, Nielsen CV, Sibilitz KL, Tolstrup JS, Zwisler AD, Taylor RS (2022) Cardiac rehabilitation for patients with heart failure: association with readmission and mortality risk. Eur Heart J Qual Care Clin Outcomes 8:830–839. https://doi.org/10.1093/ehjqcco/qcab086
De Feo S, Tramarin R, Ambrosetti M, Riccio C, Temporelli PL, Favretto G, Furgi G, Griffo R (2012) Gender differences in cardiac rehabilitation programs from the Italian survey on cardiac rehabilitation (ISYDE-2008). Int J Cardiol 160:133–139. https://doi.org/10.1016/j.ijcard.2011.04.011
Giuliano C, Vicendese D, Vogrin S, Lane R, Driscoll A, Dinh D, Palmer K, Levinger I, Neil C (2022) Predictors of referral to cardiac rehabilitation in patients following hospitalisation with heart failure: a multivariate regression analysis. J Clin Med 11. https://doi.org/10.3390/jcm11051232
Park LG, Schopfer DW, Zhang N, Shen H, Whooley MA (2017) Participation in cardiac rehabilitation among patients with heart failure. J Card Fail 23:427–431. https://doi.org/10.1016/j.cardfail.2017.02.003
Colella TJ, Gravely S, Marzolini S, Grace SL, Francis JA, Oh P, Scott LB (2015) Sex bias in referral of women to outpatient cardiac rehabilitation? A meta-analysis. Eur J Prev Cardiol 22:423–441. https://doi.org/10.1177/2047487314520783
Samayoa L, Grace SL, Gravely S, Scott LB, Marzolini S, Colella TJ (2014) Sex differences in cardiac rehabilitation enrollment: a meta-analysis. Can J Cardiol 30:793–800. https://doi.org/10.1016/j.cjca.2013.11.007
Long L, Mordi IR, Bridges C, Sagar VA, Davies EJ, Coats AJ, Dalal H, Rees K, Singh SJ, Taylor RS (2019) Exercise-based cardiac rehabilitation for adults with heart failure. Cochrane Database Syst Rev 1:CD003331. https://doi.org/10.1002/14651858.CD003331.pub5
Bjarnason-Wehrens B, Nebel R, Jensen K, Hackbusch M, Grilli M, Gielen S, Schwaab B, Rauch B, German Society of Cardiovascular P, Rehabilitation (2020) Exercise-based cardiac rehabilitation in patients with reduced left ventricular ejection fraction: the Cardiac Rehabilitation Outcome Study in Heart Failure (CROS-HF): a systematic review and meta-analysis. Eur J Prev Cardiol 27:929–952. https://doi.org/10.1177/2047487319854140
Leggio M, Fusco A, Loreti C, Limongelli G, Bendini MG, Mazza A, Coraci D, Padua L (2020) Effects of exercise training in heart failure with preserved ejection fraction: an updated systematic literature review. Heart Fail Rev 25:703–711. https://doi.org/10.1007/s10741-019-09841-x
Fukuta H, Goto T, Wakami K, Kamiya T, Ohte N (2019) Effects of exercise training on cardiac function, exercise capacity, and quality of life in heart failure with preserved ejection fraction: a meta-analysis of randomized controlled trials. Heart Fail Rev 24:535–547. https://doi.org/10.1007/s10741-019-09774-5
Morgan H, Sinha A, McEntegart M, Hardman SM, Perera D (2022) Evaluation of the causes of sex disparity in heart failure trials. Heart 108:1547–1552. https://doi.org/10.1136/heartjnl-2021-320696
van Diemen J, Verdonk P, Chieffo A, Regar E, Mauri F, Kunadian V, Sharma G, Mehran R, Appelman Y (2021) The importance of achieving sex- and gender-based equity in clinical trials: a call to action. Eur Heart J 42:2990–2994. https://doi.org/10.1093/eurheartj/ehab457
Eliya Y, Whitelaw S, Thabane L, Voors AA, Douglas PS, Van Spall HGC (2021) Temporal trends and clinical trial characteristics associated with the inclusion of women in heart failure trial steering committees: a systematic review. Circ Heart Fail 14:e008064. https://doi.org/10.1161/CIRCHEARTFAILURE.120.008064
Reza N, Tahhan AS, Mahmud N, DeFilippis EM, Alrohaibani A, Vaduganathan M, Greene SJ, Ho AH, Fonarow GC, Butler J, O’Connor C, Fiuzat M, Vardeny O, Pina IL, Lindenfeld J, Jessup M (2020) Representation of women authors in international heart failure guidelines and contemporary clinical trials. Circ Heart Fail 13:e006605. https://doi.org/10.1161/CIRCHEARTFAILURE.119.006605
Streed C Jr, Caceres B, Mukherjee M (2021) Preventing cardiovascular disease among sexual and gender minority persons. Heart. https://doi.org/10.1136/heartjnl-2021-319069
Thompson DR, Clark AM (2020) Heart failure disease management interventions: time for a reappraisal. Eur J Heart Fail 22:578–580. https://doi.org/10.1002/ejhf.1777
Thompson DR, Ski CF, Clark AM (2019) Transitional care interventions for heart failure: what are the mechanisms? Am J Med 132:278–280. https://doi.org/10.1016/j.amjmed.2018.09.028
Bozkurt B, Fonarow GC, Goldberg LR, Guglin M, Josephson RA, Forman DE, Lin G, Lindenfeld J, O’Connor C, Panjrath G, Pina IL, Shah T, Sinha SS, Wolfel E, Failure ACsH, Transplant S, Leadership C (2021) Cardiac rehabilitation for patients with heart failure: JACC Expert Panel. J Am Coll Cardiol 77:1454–1469. https://doi.org/10.1016/j.jacc.2021.01.030
van Halewijn G, Deckers J, Tay HY, van Domburg R, Kotseva K, Wood D (2017) Lessons from contemporary trials of cardiovascular prevention and rehabilitation: a systematic review and meta-analysis. Int J Cardiol 232:294–303. https://doi.org/10.1016/j.ijcard.2016.12.125
Francis T, Kabboul N, Rac V, Mitsakakis N, Pechlivanoglou P, Bielecki J, Alter D, Krahn M (2019) The effect of cardiac rehabilitation on health-related quality of life in patients with coronary artery disease: a meta-analysis. Can J Cardiol 35:352–364. https://doi.org/10.1016/j.cjca.2018.11.013
Beckie TM, Beckstead JW (2010) Predicting cardiac rehabilitation attendance in a gender-tailored randomized clinical trial. J Cardiopulm Rehabil Prev 30:147–156. https://doi.org/10.1097/HCR.0b013e3181d0c2ce
Ghisi GLM, Kin SMR, Price J, Beckie TM, Mamataz T, Naheed A, Grace SL (2022) Women-focused cardiovascular rehabilitation: an International Council of Cardiovascular Prevention and Rehabilitation clinical practice guideline. Can J Cardiol 38:1786–1798. https://doi.org/10.1016/j.cjca.2022.06.021
Beckie TM, Beckstead JW, Schocken DD, Evans ME, Fletcher GF (2011) The effects of a tailored cardiac rehabilitation program on depressive symptoms in women: a randomized clinical trial. Int J Nurs Stud 48:3–12. https://doi.org/10.1016/j.ijnurstu.2010.06.005
Beckie TM, Beckstead JW (2010) The effects of a cardiac rehabilitation program tailored for women on global quality of life: a randomized clinical trial. J Womens Health (Larchmt) 19:1977–1985. https://doi.org/10.1089/jwh.2010.1937
Taylor RS, Dalal HM, McDonagh STJ (2022) The role of cardiac rehabilitation in improving cardiovascular outcomes. Nat Rev Cardiol 19:180–194. https://doi.org/10.1038/s41569-021-00611-7
Thomas RJ, Beatty AL, Beckie TM, Brewer LC, Brown TM, Forman DE, Franklin BA, Keteyian SJ, Kitzman DW, Regensteiner JG, Sanderson BK, Whooley MA (2019) Home-based cardiac rehabilitation: a scientific statement from the American Association of Cardiovascular and Pulmonary Rehabilitation, the American Heart Association, and the American College of Cardiology. Circulation 140:e69–e89. https://doi.org/10.1161/CIR.0000000000000663
Anderson L, Sharp GA, Norton RJ, Dalal H, Dean SG, Jolly K, Cowie A, Zawada A, Taylor RS (2017) Home-based versus centre-based cardiac rehabilitation. Cochrane Database Syst Rev 6:CD007130. https://doi.org/10.1002/14651858.CD007130.pub4
Beatty AL, Brown TM, Corbett M, Diersing D, Keteyian SJ, Mola A, Stolp H, Wall HK, Sperling LS (2021) Million Hearts Cardiac Rehabilitation Think Tank: accelerating new care models. Circ Cardiovasc Qual Outcomes 14:e008215. https://doi.org/10.1161/CIRCOUTCOMES.121.008215
Lau ES, Cunningham T, Hardin KM, Liu E, Malhotra R, Nayor M, Lewis GD, Ho JE (2020) Sex differences in cardiometabolic traits and determinants of exercise capacity in heart failure with preserved ejection fraction. JAMA Cardiol 5:30–37. https://doi.org/10.1001/jamacardio.2019.4150
Author information
Authors and Affiliations
Contributions
Each author contributed equally to the conceptualization, data collection, data synthesis, and writing and reviewing of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
This declaration is not relevant to the content of the submitted work.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wills, W.B., Athilingam, P. & Beckie, T.M. Exercise-based cardiac rehabilitation in women with heart failure: a review of enrollment, adherence, and outcomes. Heart Fail Rev 28, 1251–1266 (2023). https://doi.org/10.1007/s10741-023-10306-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10741-023-10306-5