Abstract
Cardiac magnetic resonance spectroscopy (MRS) is a noninvasive method to assess by-products of myocardial metabolism. Recent developments in shorter scan protocols and more powerful field strengths have created interest in utilizing this technology in studying and characterizing the metabolic derangements in heart failure patients. Our lack of understanding in heart failure could be greatly enhanced by identifying the metabolic changes and eventually modifying metabolic substrate to achieve improved cardiac mechanics with the aid of this technology. However, there are several impediments for the widespread applicability of this technology. This review discusses the principals of human cardiac MRS and literature pertaining to use of MRS in patients with cardiomyopathy.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Magnetic resonance cardiac spectroscopy (MRS) has recently emerged as a novel tool that can be used to noninvasively study the metabolism of myocardium. Conceptually, it is similar to other spectroscopic methods that are used to either determine the molecular structure of compounds or the presence of a compound in a well-defined sample. Another advantage of this method is to the acquisition of the data can be performed repetitively without destroying the sample. In a nutshell, it is based on the phenomenon that certain atoms have a magnetic moment. Nuclei with an odd number of protons and neutrons such as 1H, 31P, 13C, 23Na, and 87Rb have a magnetic moment. These are involved in high-energy phosphate metabolism, Kerb’s cycle (tricarboxylic acid cycle), creatine kinase metabolism, and fatty acid metabolism, all of which are essential for viable myocardium. An electromagnetic wave is directed on these molecules that leads to change in magnetic momentum, and hence, energy is released that could be measured and interpreted as a spectrum. Compounds containing these nuclei can be identified and quantified. Compounds such as adenosine triphosphate (ATP) and phosphocreatine (PCr) form the basis of several metabolic processes in myocardium. This was first observed by George Radda using 31P-MRS while studying skeletal muscles [1]. Later on, Garlick et al. first used it to study isolated perfused heart in 1977 [2]. Since then, this technique has become much advanced. Some of the principals of this technique are given below.
Principals
While in principal it appears to be a simple procedure however technically it is challenging. This is particularly because heart is constantly in motion and is located behind the chest wall. In addition, the sensitivity of 31P and 13C is much lower relative to 1H (6.6 and 0.018% of 1H sensitivity, respectively) [3]. Therefore, there is low signal to noise ratio, and thus, there is necessity for use of surface coils to enhance the signal. Linearly polarized surface coils used mainly for animal studies become useless in humans and thus the advent of quadrature detection coils are essential. ECG-gating and breath-holding are the other important requirements. In comparison to 31P-MRS, 1H-MRS does not require special broad band radiofrequency (RF) amplifiers and 31P tuned RF coils. Therefore, 1H-MRS is more widely available. In addition, 31P-MRS requires some fine tuning to obtain good quality spectra. However, information obtained from 1H-MRS differs from the information obtained from 31P-MRS. 13C-MRS has limited application in myocardium due to its low sensitivity but potentially can be used to study metabolites of Kreb’s cycle, beta oxidation of fatty acids, and pyruvate dehydrogenase flux. The first report of 13C-MRS in human heart showed its feasibility in four human subjects [4]. 23Na-MRS is also not well established but can be used to investigate and identify myocardial scar. The degree of post infarct scar has shown to correlate with 23Na signal. 87Rb is a K+ analogue and can help identify function of Na+/K+ ATPase pump by using 87Rb-MRS. This review will focus on 31P and 1H-MRS only.
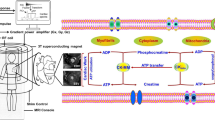
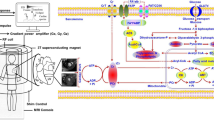
Typically, an MR scout image using spin-echo or single state free precession (SSFP) MR sequence is obtained initially. There are two main methods of localization pulse sequences for 1H-MRS. The first, stimulated echo acquisition mode (STEAM) uses three 90 degree pulses and uses TE in the range of 12–20 ms and the second, point resolved spectroscopy (PRESS) uses two slice selective spin-echo sequence and a slice selective excitation pulse to define the volume and uses TE in the range of 25–35 ms. These have been described in more detail elsewhere [5, 6]. Other sequences include depth-resolved surface coil spectroscopy (DRESS), image-selected in vivo spectroscopy (ISIS), and three-dimensional chemical shift imaging (3D-CSI). Signals obtained are corrected for T2 relaxation. End-systolic images using ECG-gating and breath-hold or end expiratory phase using respiratory gating are utilized to obtain good quality spectra. A small volume is selected in the interventricular septum, since it is free of pericardial fat. The volume is typically placed using four-chamber and four short axis images. To ensure that the volume stays on the septum, cine images are used to determine the delay time. Once the spectra are obtained, using a priori spectroscopic knowledge and signal modeling, signals are quantified. Since with 31P-MRS absolute values of ATP and PCr are difficult to obtain, so only PCr to ATP ratio is usually reported. Normal PCr-to-ATP ratios of healthy subjects range between 1.1 and 2.5. 1H-MRS can measure creatine as well as triglyceride content. The triglyceride content is usually expressed in ratio of triglyceride to water (%). Normal ratio is <0.9%. The dominant water related signal and blood contamination are suppressed by using correction factors in order to obtain reliable spectra. Prone position of subjects is favored to minimize the distance between the surface coil and myocardium however studies have studied patients in supine position as well.
Once spectra are obtained, it is essential to follow a standardized data processing methodology. A typical 31P spectrum from a healthy subject shows six resonances (Fig. 1): 3 31P atoms (α, β, and γ) of ATP, PCr, 2,3-diphosphoglycerate (from erythrocytes), and phosphodiesters (from membrane and serum phospholipids). A typical 1H spectrum from a healthy subject shows a tall tissue water peak around 5 ppm, a prominent triglyceride peak (composed of methylenes; –CH2 and –CH3 of fatty acids) and several other peaks of 1H rich metabolites (creatine, lactate, choline, and taurine). The first peak is of triglyceride (methylenes of fatty acids) which usually ranges from 1.0 to 2.0 ppm (Fig. 2) Spectra are processed using various softwares, e.g., NUTS software (Acorn NMR, Fremont, Calif) [7], java Magnetic Resonance User Interface (jMRUI) [8] and Advanced Method for Accurate, Robust and Efficient Spectral fitting of MRS data (AMARES) [9]. In summary, there are many phases of MRS that require a certain level of mastery to collect and optimize data signals since it is not an automated process. Consistence of user methods is probably more important than individualizing the settings for obtaining MRS data. A brief review of MRS use in myocardial diseases is discussed below.
MR spectroscopy and myocardial diseases
MRS use in myocardial disease started to gain momentum in 1990s in the early era of modern revascularization and risk stratification. Initially, in vivo correlation of myocardial metabolism and perfusion was studied [10]. Later on, MRS helped in providing insight into metabolic response of inotropic myocardial stress [11]. A decrease in PCr/ATP ratio or increase in triglyceride to water ratio provided information of myocardial metabolic health. In healthy athlete volunteers, under normal and supranormal cardiac stimulation, the PCr/ATP ratio remained constant. It was not until under extreme inotropic stimulation that PCr/ATP started dropping [12]. Therefore, researchers thought of this as a direct measurement of myocardial bioenergetics and an era of human research experiments began. Table 1 shows the potential changes in metabolites as detected by MRS techniques in various cardiomyopathies. A schematic diagram of myocardial energy metabolism and various metabolites that can be detected by 1H, 31P, and 13C MRS techniques is shown in Fig. 3.
Ischemic heart disease
With ischemia PCr levels drop within seconds of decrease in perfusion [13]. For the preservation of myocardial funciton, maintenance of high levels of high energy phosphates is required. Decrease in PCr/ATP ratio with isometric exercise (>30% increase in work load) in coronary artery disease (CAD) patients was observed in a study of 16 patients with CAD [14]. This was not observed in normal controls (N = 11) and nonischemic patients (N = 9). The levels of PCr/ATP returned to normal after revascularization. These findings were further confirmed by other studies with longer follow-ups that studied 31P-MRS stress test and its prognostic value [15, 16]. However, despite its value as a biomarker test, the lack of spatial and temporal resolution led to its poor clinical acceptance. It cannot identify coronary artery territory better than other much cheaper and less labor-intensive stress tests. A potential role of 31P-MRS to identify viable myocardium was investigated and showed promise [17]. In hibernating or stunned myocardium, the ATP levels remain near normal while PCr decrease. On the other hand, in the scarred myocardium both PCr and ATP decrease, so the ratio remained near normal. Due to large variation in the normal values of PCr/ATP, this also remained an unattractive use of 31P-MRS. In addition, the abnormalities of PCr/ATP are not specific for ischemic damage or nonviability. The approaches used in the prior studies only involved left anterior wall and apex so the inferior and posterior walls cannot be studied. A potential solution is to use higher field strength as 4.1 T has shown to improve voxel size to ∼8 ml with much improved spatial resolution [18].
Due to higher sensitivity of 1H compared with 31P, 1H has been studied to examined and shown promise in identifying nonviable myocardium by showing regional creatine depletion [19]. In patients with dilated cardiomyopathy secondary to ischemic heart disease, average PCr/ATP ratio was 1.53 ± 0.07 which was significantly lower than healthy normals 1.80 ± 0.06 [20]. However, the average PCr/ATP ratio of patients with ischemic cardiomyopathy was not different from average PCr/ATP ratio of patients with idiopathic cardiomyopathy and thus questioned the specificity of these ratios for ischemic cardiomyopathy. Similar to infarcted myocardium where PCr/ATP ratio is not useful as both absolute values of PCr and ATP are low, the absolute levels of PCr and ATP in patients with systolic dysfunction and may underestimate the bioenergetics in failing myocardium. Alteration in the preference of glucose versus free fatty acid utilization is controversial and may be studied using methods to assess TCA cycle in conjunction with PCr/ATP ratio. However, modification of myocardial substrate utilization is an area of active investigation and therapeutics needed to alter or improve myocardial bioenergetics may utilize MRS as an intermediate outcome to show efficacy. Creatine is also decreased in ischemic cardiomyopathy and 1H-MRS is a much more sensitive method to detect the creatine depletion [19, 21]. Lastly, choline has been shown to be associated with presence and severity of CAD [22]. It can be detected by 1H-MRS, but literature is limited regarding its role in myocardial metabolism. MRS has an edge over the other noninvasive testing modalities as it does not require injection of any contrast agent or radiation and has validated data available regarding its utility in providing information for ischemia and viability in ischemic heart disease. However, due to the large voxel size and poor spatial resolution its progress is impeded until a later day when this issue could be resolved.
Nonischemic heart disease
There are two questions that are still unanswered: (1) are changes in myocardial bioenergetics in ischemic and nonischemic myocardium similar or different; (2) do the metabolic changes occur as a bystander or are causal in nature. Even though these have not been definitively answered in literature, there is evidence that these changes differ between ischemic and nonischemic myocardium. Also, diabetic patients have shown to have increased triglyceride content that precedes the symptoms of heart. These findings may suggest that metabolic derangements may be causal in nature. Furthermore, reversal of intramyocardial fat with bariatric surgery in severe obese individuals improves cardiomyopathy and cardiac mechanics as well as metabolic peaks, suggesting a possible causal relationship. We have classified nonischemic heart disease into (a) metabolic heart disease, (b) valvular heart disease, and (c) inherited cardiomyopathies, and a brief discussion of MRS is included below:
-
A.
Metabolic heart disease
Metabolic heart disease is a heterogenous term, but understandably, MRS will have high yield in this group of myocardial diseases. Changes in myocardium of diabetics could be detected as early as 1 year after onset of diabetes [23]. Intramyocardial triglyceride accumulation (cardiac steatosis) is common among patients with diabetes type I and type II [24–27]. The reversal of cardiac steatosis has been observed with caloric restriction [28, 29]. A study that used pioglitazone showed improvement in diastolic function and myocardial glucose uptake without affecting the myocardial triglyceride content, suggesting some signal that it might be of use among patients with diabetic cardiomyopathy [30]. Unfortunately, pioglitazone has a US black box warning regarding its use in heart failure patients and may lead to heart failure in up to 8% of the patients due to unknown reasons. Patients with dilated cardiomyopathy have 51% lower absolute levels of PCr, 35% lower absolute levels of ATP, and 25% lower levels of PCr/ATP ratio [31]. This has been shown by using spectral localization with optimum point spread function (SLOOP) 31P-MRS technique that can measure absolute levels of PCr and ATP [31]. Whether lower levels of PCr and ATP precede contractile dysfunction in diabetic cardiomyopathy or are just a compensatory mechanism remains unclear. To answer this, a longitudinal study of diabetes patients would be required. However, patients without CAD but with diabetes are found to have low PCr/ATP, suggesting that PCr/ATP may occur prior to subclinical or clinical dysfunction [27]. Cardiomyopathy that results due to pressure overload such as hypertension has also lower levels of PCr/ATP. In these patients, diastolic dysfunction correlated with PCr/ATP ratios [32]. Once systolic dysfunction sets in, the PCr/ATP levels further decrease [33]. Dilated cardiomyopathy is usually a final common pathway of most of the metabolic cardiomyopathies. Figure 4 shows the differences in the 31P-MRS spectra of normal and dilated cardiomyopathy patients.
-
B.
Valvular heart disease
Among patients with valvular heart disease, PCr/ATP ratio has shown to be reduced in heart failure patients with mitral regurgitation or aortic stenosis [34–36]. Presence of myocardial hypertrophy was common in these patients. The levels of PCr/ATP ratio improved significantly from 1.28 ± 0.17 to 1.47 ± 0.14, 9 months after aortic valve surgery [37]. These changes have even been observed in patients with severe aortic stenosis and subclinical left ventricular dysfunction. Both perfusion reserve and oxygenation as measured by blood-oxygen level dependent (BOLD) signal showed improvement in left ventricular strain and energetics 8 months after aortic valve replacement [38]. With the advent of transcutaneous therapies, it would be interesting to study bioenergetics after transcutaneous aortic valve implantation. Currently, the timing for valve surgery is based on echocardiographic measures. It might be possible that in the future, measurements of cellular level biomolecular changes could guide these valvular replacement procedures before the development of ventricular dysfunction.
-
C.
Inherited cardiomyopathies
Symptomatic and asymptomatic patients with hypertrophic cardiomyopathy have shown to have decreased PCr/ATP ratio [39–41]. This is also shown in hypertrophic cardiomyopathy of genetic origin [40, 42, 43]. This preexisting energy deficit is exacerbated with exercise stress as measured by 31P-MRS [44]. This might explain the exercise-induced diastolic dysfunction in these patients. Female careers and patients of Becker’s muscular dystrophy also have low levels of PCr and ATP when compared with normal volunteers [45]. Patients with familial hypercholesterolemia [46] and hemochromatosis [47] also have low levels of PCr/ATP ratio. Statins have shown to improve PCr/ATP ratio in familial hypercholesterolemia [46]. Myocardial fat depots in Anderson-Fabry’s disease, an inherited X-linked lysosomal storage disease could be studied using 1H-MRS [48]. This study observed that in advanced Fabry’s disease, the triglyceride content was found to be severely elevated. This suggests a possible role in monitoring progression of disease in these patients.
Cardiac transplant
31P-MRS can detect post-transplant acute rejection by showing low PCr/ATP ratio. The ratio of PCr/ATP is lower early post-transplant but improves with time. However, these ratios did not correlate with the cardiac biopsy findings. Another study evaluated 31P-MRS stress test and showed that 40% of the transplant recipients had >25% decrease in PCr/ATP ratio. The significance of this finding is not clear. Early noninvasive detection of transplant rejection could become an important discovery and could help target immunosuppressive therapy. Use of 13C-hyperpolarized MRS might help unravel many of the myocardial changes that occur with immunosuppressive therapy and could lead to chronic allograft vasculopathy and rejection.
Response to heart failure treatment
Patients treated with diuretics, beta-blockers, and angiotensin-converting enzyme inhibitors showed an improvement in PCr/ATP ratio from 1.51 ± 0.32 to 2.15 ± 0.27 within 3 months of therapy that paralleled the clinical improvement [49]. Myocardial substrate modifiers such as trimetazidine have also been examined. Trimetazidine inhibits fatty acid oxidation and switches myocardial metabolism to glycolysis. There was improvement in PCr/ATP ratio by 33% in trimetazidine-treated patients compared to placebo (1.35 ± 0.33 vs. 1.80 ± 0.50, respectively) [50]. Another trial studying the effect of supervised exercise training in patients with dilated cardiomyopathy observed that PCr/ATP levels did not change, but there was an improvement in LV function, suggesting that exercise does not adversely affect the myocardial metabolism. Such designs can be used to assess therapies in clinical heart failure trials [51]. More recently, beta-blocker use in heart failure showed favorable cardiac energetics. [52] In a study of patients with dilated cardiomyopathy, treatment with recombinant human growth hormone led to increase in PCr/ATP ratio and myocardial mass. However, improvement in myocardial function was not demonstrated [53]. More recently, using a combination of 1H-MRS, 31P-MRS, and MR imaging, researchers showed that short-term caloric restriction led to increase in myocardial triglyceride levels without affecting PCr/ATP ratio [54]. Another dietary study of high-fat diet in 16 volunteers showed impaired PCr/ATP ratios without altering cardiac function [55]. These studies suggest that dietary interventions and their effects on myocardium can be studied by using MRS. Despite MRS showing promise in determining the effects of some of the heart failure therapies, more work is still needed to define MRS role in monitoring and determining the effect of these therapies. A possible use of MRS to show improvement or reversal of diabetic cardiomyopathy after the use of advanced glycation end-product inhibitor could be its important contribution to the field of science.
Future directions
Even though the field has existed for >3 decades, the penetration of this technology into clinical realm is still not fully realized. The future awaits the technological advances with increase in magnetic field signal strength to a level where spatial and temporal resolutions become meaningful. Three tesla is better than 1.5 T, and 7 T is better than 3 T [56, 57]. Seven tesla can provide a resolution of <5 ml. A comparison of spectra obtained at 3 and 7 T is given in Fig. 5. Scan timings have been decreasing, and it has been shown that ∼1.5 min 1H-MRS scan can provide reliable information about triglycerides, creatine, and choline myocardial content [58]. However, spectral separation of creatine and choline spectral peaks at 3.0 and 3.2 ppm is reduced when fewer 1H-MRS signals are averaged in a shorter period of acquisition. Regional temporal changes in biochemical processes in real time have not been well studied, but with enhanced temporal resolution, it could become one of the possibilities. In addition to field strength, advanced coil design and sequence development might enhance the quality of spectral signals and provide more granular data regarding the metabolites. The reproducibility of signals is another hurdle which also needs to be addressed by using well-defined protocols. Part of this is due to poor signal to noise ratio, which can be boosted by using dynamic nuclear hyperpolarization. Direct estimation of myocardial TCA flux in vivo has been demonstrated using hyperpolarized 13C-MRS [59]. This field is rapidly expanding with several experiences noted in the literature [60, 61].
a Graph shows comparison of spectra in a typical patient (57-year-old woman) at 3 and 7 T. These spectra have had a matched filter applied and have been normalized to mean baseline noise, so the PCr peak height is, by definition, the PCr SNR. Increase in SNR at 7 T is readily apparent. b Corresponding mid-short axis localizer image acquired at 7 T. c Corresponding four-chamber localizer image acquired at 7 T. The spectroscopy matrix is overlaid in red, and the voxel plotted in a is highlighted. The yellow-shaded region denotes the regional saturation slab used to suppress signal from overlying skeletal muscle
Conclusions
In conclusion, MRS is still limited to research settings where it provides noninvasive fundamental insight into myocardial metabolism. Technical advances in developing techniques to enhance spatial and temporal resolution will dramatically expand its application to clinical realm. Since the epidemiology of cardiac diseases is changing, with disproportionate increase in metabolic diseases when compared with ischemic heart disease, the utility of MRS will expand. Future studies should focus on designing larger cohort studies that employ MRS to study the fundamental changes in cardiac metabolism.
References
Ackerman JJ, Grove TH, Wong GG, Gadian DG, Radda GK (1980) Mapping of metabolites in whole animals by 31P NMR using surface coils. Nature 283:167–170
Garlick PB, Radda GK, Seeley PJ, Chance B (1977) Phosphorus NMR studies on perfused heart. Biochem Biophys Res Commun 74:1256–1262
Ackerman JJ, Gadian DG, Radda GK, Wong GG (1981) Observation of 1H NMR signals with receiver coils tuned for other nuclides. J Magn Reson (1969) 42:498–500
Cunningham CH, Lau JY, Chen AP, Geraghty BJ, Perks WJ, Roifman I, Wright GA, Connelly KA (2016) Hyperpolarized 13C metabolic MRI of the human heart: novelty and significance. Circ Res 119:1177–1182
Granot J (1986) Selected volume excitation using stimulated echoes (VEST). Applications to spatially localized spectroscopy and imaging. J Magn Reson (1969) 70:488–492
Hamilton G, Middleton MS, Bydder M, Yokoo T, Schwimmer JB, Kono Y, Patton HM, Lavine JE, Sirlin CB (2009) Effect of press and steam sequences on magnetic resonance spectroscopic liver fat quantification. J Magn Reson Imaging 30:145–152
Acorn N (2000) NUTS NMR utility transform software. Acorn NMR, Livermore
Naressi A, Couturier C, Devos J, Janssen M, Mangeat C, De Beer R, Graveron-Demilly D (2001) Java-based graphical user interface for the MRUI quantitation package. MAGMA 12:141–152
Turner CE, Russell BR, Gant N (2015) Comparative quantification of dietary supplemented neural creatine concentrations with 1 H-MRS peak fitting and basis spectrum methods. Magn Reson Imaging 33:1163–1167
Osbakken M, Mitchell MD, Zhang D, Mayevsky A, Chance B (1991) In vivo correlation of myocardial metabolism, perfusion, and mechanical function during increased cardiac work. Cardiovasc Res 25:749–756
Lamb HJ, Beyerbacht HP, Ouwerkerk R, Doornbos J, Pluim BM, van der Wall EE, van der Laarse A, de Roos A (1997) Metabolic response of normal human myocardium to high-dose atropine-dobutamine stress studied by 31P-MRS. Circulation 96:2969–2977
Pluim BM, Lamb HJ, Kayser HW, Leujes F, Beyerbacht HP, Zwinderman AH, van der Laarse A, Vliegen HW, de Roos A, van der Wall EE (1998) Functional and metabolic evaluation of the athlete’s heart by magnetic resonance imaging and dobutamine stress magnetic resonance spectroscopy. Circulation 97:666–672
Clarke K, O’Connor AJ, Willis RJ (1987) Temporal relation between energy metabolism and myocardial function during ischemia and reperfusion. Am J Phys 253:H412–H421
Weiss RG, Bottomley PA, Hardy CJ, Gerstenblith G (1990) Regional myocardial metabolism of high-energy phosphates during isometric exercise in patients with coronary artery disease. N Engl J Med 323:1593–1600
Johnson BD, Shaw LJ, Buchthal SD, Merz CNB, Kim H-W, Scott KN, Doyle M, Olson MB, Pepine CJ, den Hollander J (2004) Prognosis in women with myocardial ischemia in the absence of obstructive coronary disease results from the National Institutes of Health–National Heart, Lung, and Blood Institute–Sponsored Women’s Ischemia Syndrome Evaluation (WISE). Circulation 109:2993–2999
Buchthal SD, den Hollander JA, Merz CNB, Rogers WJ, Pepine CJ, Reichek N, Sharaf BL, Reis S, Kelsey SF, Pohost GM (2000) Abnormal myocardial phosphorus-31 nuclear magnetic resonance spectroscopy in women with chest pain but normal coronary angiograms. N Engl J Med 342:829–835
Yabe T, Mitsunami K, Inubushi T, Kinoshita M (1995) Quantitative measurements of cardiac phosphorus metabolites in coronary artery disease by 31P magnetic resonance spectroscopy. Circulation 92:15–23
Hetherington HP, Luney DJ, Thomas Vaughan J, Pan JW, Ponder SL, Tschendel O, Twieg DB, Pohost GM (1995) 3D 31P spectroscopic imaging of the human heart at 4.1 T. Magn Reson Med 33:427–431
Bottomley PA, Weiss RG (1998) Non-invasive magnetic-resonance detection of creatine depletion in non-viable infarcted myocardium. Lancet 351:714–718
Hardy CJ, Weiss RG, Bottomley PA, Gerstenblith G (1991) Altered myocardial high-energy phosphate metabolites in patients with dilated cardiomyopathy. Am Heart J 122:795–801
Nakae I, Mitsunami K, Matsuo S, Matsumoto T, Morikawa S, Inubushi T, Koh T, Horie M (2004) Assessment of myocardial creatine concentration in dysfunctional human heart by proton magnetic resonance spectroscopy. Magnetic resonance in medical sciences : MRMS : an official journal of Japan Society of Magnetic Resonance in Medicine 3:19–25
Senturk T, Tutuncu A, Ozdemir B, Ozdabakoglu O, Aydin S, Baran I, Gullulu S, Savci V, Aydinlar A (2010) Serum choline levels in patients with stable angina and acute coronary syndromes: relation to the severity of coronary artery disease. Coron Artery Dis 21:466–471
Levelt E, Ntusi N, Mahmod M, Wainwright C, Piechnik S, Francis J, Davis A, Schneider J, Leeson P, Karamitsos T (2014) 127 early manifestations of diabetic cardiomyopathy assessed by cardiac magnetic resonance imaging and spectroscopy. Heart 100:A73–A74
Metzler B, Schocke MF, Steinboeck P, Wolf C, Judmaier W, Lechleitner M, Lukas P, Pachinger O (2002) Decreased high-energy phosphate ratios in the myocardium of men with diabetes mellitus type I. Journal of cardiovascular magnetic resonance : official journal of the Society for Cardiovascular Magnetic Resonance 4:493–502
van der Meer R, Rijzewijk L, Smit J, Diamant M, Bax J, Hammer S, Romijn J, de Roos A, Lamb H (2008) Myocardial steatosis is an independent predictor of diastolic dysfunction in type 2 diabetes mellitus. Myocardial Steatosis and Left Ventricular Function in Type 2 Diabetes Mellitus 137
McGavock JM, Lingvay I, Zib I, Tillery T, Salas N, Unger R, Levine BD, Raskin P, Victor RG, Szczepaniak LS (2007) Cardiac steatosis in diabetes mellitus: a 1H-magnetic resonance spectroscopy study. Circulation 116:1170–1175
Scheuermann-Freestone M, Madsen PL, Manners D, Blamire AM, Buckingham RE, Styles P, Radda GK, Neubauer S, Clarke K (2003) Abnormal cardiac and skeletal muscle energy metabolism in patients with type 2 diabetes. Circulation 107:3040–3046
Snel M, Hammer S, Lamb HJ, Jazet IM, Van der Meer RW, Pijl H, Meinders AE, Romijn JA (2011) Prolonged caloric restriction in obese patients with type 2 diabetes mellitus decreases myocardial triglyceride content and improves myocardial function. The Effects of a Very Low Calorie Diet and Exercise in Obese Type 2 Diabetes Mellitus Patients 52:107
Hammer S, Snel M, Lamb HJ, Jazet IM, van der Meer RW, Pijl H, Meinders EA, Romijn JA, de Roos A, Smit JW (2008) Prolonged caloric restriction in obese patients with type 2 diabetes mellitus decreases myocardial triglyceride content and improves myocardial function. J Am Coll Cardiol 52:1006–1012
van der Meer RW, Rijzewijk LJ, de Jong HW, Lamb HJ, Lubberink M, Romijn JA, Bax JJ, de Roos A, Kamp O, Paulus WJ (2009) Pioglitazone improves cardiac function and alters myocardial substrate metabolism without affecting cardiac triglyceride accumulation and high-energy phosphate metabolism in patients with well-controlled type 2 diabetes mellitus. Circulation 119:2069–2077
Beer M, Seyfarth T, Sandstede J, Landschütz W, Lipke C, Köstler H, von Kienlin M, Harre K, Hahn D, Neubauer S (2002) Absolute concentrations of high-energy phosphate metabolites in normal, hypertrophied, and failing human myocardium measured noninvasively with 31P-SLOOP magnetic resonance spectroscopy. J Am Coll Cardiol 40:1267–1274
Heyne J-P, Rzanny R, Hansch A, Leder U, Reichenbach J, Kaiser W (2006) 31P-MR spectroscopic imaging in hypertensive heart disease. Eur Radiol 16:1796–1802
Lamb HJ, Beyerbacht HP, Van Der Laarse A, Stoel BC, Doornbos J, van der Wall EE, de Roos A (1999) Diastolic dysfunction in hypertensive heart disease is associated with altered myocardial metabolism. Circulation 99:2261–2267
Conway MA, Bottomley PA, Ouwerkerk R, Radda GK, Rajagopalan B (1998) Mitral regurgitation impaired systolic function, eccentric hypertrophy, and increased severity are linked to lower phosphocreatine/ATP ratios in humans. Circulation 97:1716–1723
Neubauer S, Horn M, Pabst T, Harre K, Stromer H, Bertsch G, Sandstede J, Ertl G, Hahn D, Kochsiek K (1997) Cardiac high-energy phosphate metabolism in patients with aortic valve disease assessed by 31P-magnetic resonance spectroscopy. Journal of investigative medicine : the official publication of the American Federation for Clinical Research 45:453–462
Conway MA, Allis J, Ouwerkerk R, Niioka T, Rajagopalan B, Radda GK (1991) Detection of low phosphocreatine to ATP ratio in failing hypertrophied human myocardium by 31P magnetic resonance spectroscopy. Lancet 338:973–976
Beyerbacht HP, Lamb HJ, van Der Laarse A, Vliegen HW, Leujes F, Hazekamp MG, de Roos A, van Der Wall EE (2001) Aortic valve replacement in patients with aortic valve stenosis improves myocardial metabolism and diastolic function. Radiology 219:637–643
Mahmod M, Francis JM, Pal N, Lewis A, Dass S, De Silva R, Petrou M, Sayeed R, Westaby S, Robson MD (2014) Myocardial perfusion and oxygenation are impaired during stress in severe aortic stenosis and correlate with impaired energetics and subclinical left ventricular dysfunction. J Cardiovasc Magn Reson 16:1
Ormerod JO, Frenneaux MP (2016) Sherrid MV. Myocardial energy depletion and dynamic systolic dysfunction in hypertrophic cardiomyopathy, Nature Reviews Cardiology
Crilley JG, Boehm EA, Blair E, Rajagopalan B, Blamire AM, Styles P, McKenna WJ, Östman-Smith I, Clarke K, Watkins H (2003) Hypertrophic cardiomyopathy due to sarcomeric gene mutations is characterized by impaired energy metabolism irrespective of the degree of hypertrophy. J Am Coll Cardiol 41:1776–1782
Jung W-I, Sieverding L, Breuer J, Hoess T, Widmaier S, Schmidt O, Bunse M, van Erckelens F, Apitz J, Lutz O (1998) 31P NMR spectroscopy detects metabolic abnormalities in asymptomatic patients with hypertrophic cardiomyopathy. Circulation 97:2536–2542
Abraham MR, Bottomley PA, Dimaano VL, Pinheiro A, Steinberg A, Traill TA, Abraham TP, Weiss RG (2013) Creatine kinase adenosine triphosphate and phosphocreatine energy supply in a single kindred of patients with hypertrophic cardiomyopathy. Am J Cardiol 112:861–866
Ewijk PA, Schrauwen-Hinderling VB, Bekkers SC, Glatz JF, Wildberger JE, Kooi ME (2015) MRS: a noninvasive window into cardiac metabolism. NMR Biomed 28:747–766
Dass S, Cochlin LE, Suttie JJ, Holloway CJ, Rider OJ, Carden L, Tyler DJ, Karamitsos TD, Clarke K, Neubauer S (2015) Exacerbation of cardiac energetic impairment during exercise in hypertrophic cardiomyopathy: a potential mechanism for diastolic dysfunction. Eur Heart J ehv120
Crilley JG, Boehm EA, Rajagopalan B, Blamire AM, Styles P, Muntoni F, Hilton-Jones D, Clarke K (2000) Magnetic resonance spectroscopy evidence of abnormal cardiac energetics in Xp21 muscular dystrophy. J Am Coll Cardiol 36:1953–1958
Schocke MF, Martinek M, Kremser C, Wolf C, Steinboeck P, Lechleitner M, Jaschke W, Pachinger O, Metzler B (2003) 3-Hydroxy-3-methylglutaryl coenzyme A reductase inhibitors improve myocardial high-energy phosphate metabolism in men. J Cardiovasc Magn Reson 5:595–602
Schocke MF, Zoller H, Vogel W, Wolf C, Kremser C, Steinboeck P, Poelzl G, Pachinger O, Jaschke WR, Metzler B (2004) Cardiac phosphorus-31 two-dimensional chemical shift imaging in patients with hereditary hemochromatosis. Magn Reson Imaging 22:515–521
Petritsch B, Köstler H, Machann W, Horn M, Weng A, Goltz J, Hahn D, Niemann M, Weidemann F, Wanner C (2012) Non-invasive determination of myocardial lipid content in Fabry disease by 1H-MR spectroscopy. RöFo-Fortschritte auf dem Gebiet der Röntgenstrahlen und der bildgebenden Verfahren 184:1020–1025
Neubauer S, Krahe T, Schindler R, Horn M, Hillenbrand H, Entzeroth C, Mader H, Kromer EP, Riegger GA, Lackner K et al (1992) 31P magnetic resonance spectroscopy in dilated cardiomyopathy and coronary artery disease. Altered cardiac high-energy phosphate metabolism in heart failure. Circulation 86:1810–1818
Fragasso G, Perseghin G, De Cobelli F, Esposito A, Palloshi A, Lattuada G, Scifo P, Calori G, Del Maschio A, Margonato A (2006) Effects of metabolic modulation by trimetazidine on left ventricular function and phosphocreatine/adenosine triphosphate ratio in patients with heart failure. Eur Heart J 27:942–948
Beer M, Wagner D, Myers J, Sandstede J, Köstler H, Hahn D, Neubauer S, Dubach P (2008) Effects of exercise training on myocardial energy metabolism and ventricular function assessed by quantitative phosphorus-31 magnetic resonance spectroscopy and magnetic resonance imaging in dilated cardiomyopathy. J Am Coll Cardiol 51:1883–1891
Spoladore R, Fragasso G, Perseghin G, De Cobelli F, Esposito A, Maranta F, Calori G, Locatelli M, Lattuada G, Scifo P (2013) Beneficial effects of beta-blockers on left ventricular function and cellular energy reserve in patients with heart failure. Fundam Clin Pharmacol 27:455–464
Wittlinger T, Voigtlander T, Kreitner KF, Kalden P, Genth-Zotz S, Darius H, Thelen M, Meyer J (1998) 31P-MR spectroscopy in human end-stage heart failure during therapy with recombinant human growth hormone. MAGMA 6:171–172
van der Meer RW, Hammer S, Smit JW, Frölich M, Bax JJ, Diamant M, Rijzewijk LJ, de Roos A, Romijn JA, Lamb HJ (2007) Short-term caloric restriction induces accumulation of myocardial triglycerides and decreases left ventricular diastolic function in healthy subjects. Diabetes 56:2849–2853
Holloway CJ, Cochlin LE, Emmanuel Y, Murray A, Codreanu I, Edwards LM, Szmigielski C, Tyler DJ, Knight NS, Saxby BK (2011) A high-fat diet impairs cardiac high-energy phosphate metabolism and cognitive function in healthy human subjects. Am J Clin Nutr 93:748–755
Tyler DJ, Hudsmith LE, Clarke K, Neubauer S, Robson MD (2008) A comparison of cardiac 31P MRS at 1.5 and 3 T. NMR Biomed 21:793–798
Clarke WT, Robson MD, Rodgers CT (2015) Bloch-Siegert B1+-mapping for human cardiac 31P-MRS at 7 Tesla. Magn Reson Med
Gillinder L, Goo SY, Cowin G, Strudwick M, van der Geest RJ, Wang WY, Ng AC (2015) Quantification of intramyocardial metabolites by proton magnetic resonance spectroscopy. Front Cardiovasc Med 2
Bastiaansen JA, Cheng T, Lei H, Gruetter R, Comment A (2015) Direct noninvasive estimation of myocardial tricarboxylic acid cycle flux in vivo using hyperpolarized 13 C magnetic resonance. J Mol Cell Cardiol 87:129–137
Cunningham CH, Lau JY, Chen AP, Geraghty BJ, Perks WJ, Roifman I, Wright GA, Connelly KA (2016) Hyperpolarized 13C metabolic MRI of the human heart: initial experience. Circ Res CIRCRESAHA.116.309769
Schroeder MA, Lau AZ, Chen AP, Gu Y, Nagendran J, Barry J, Hu X, Dyck JR, Tyler DJ, Clarke K (2013) Hyperpolarized 13C magnetic resonance reveals early- and late-onset changes to in vivo pyruvate metabolism in the failing heart. Eur J Heart Fail 15:130–140
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Dr. Qureshi reports receiving honorarium/consultation fee from Medicure.
Rights and permissions
About this article
Cite this article
Qureshi, W.T., Nasir, U.b. Principals and clinical applications of magnetic resonance cardiac spectroscopy in heart failure. Heart Fail Rev 22, 491–499 (2017). https://doi.org/10.1007/s10741-017-9611-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10741-017-9611-x