Abstract
The microanatomical features of the intestinal tract mucosa layer in different species of tetrapoda vary according to the type of species, tissue, and function of the targeted cells. In the present study, we have evaluated the histological and histochemical variations of the intestinal tract in four species representing superclass tetrapoda. Bufo regularis (toad), Trachylepis quinquetaeniata (lizard), Columba livia domestica (pigeon) and Mus musculus (mouse) were used as representatives for amphibians, reptilians, avians and mammalians respectively. Histologically, the ileum’s mucosal layer of the lower tetrapods (toad and lizard) was almost similar and consists of elongated finger-like shape villi lined with simple columnar epithelium and goblet cells. Similarly, the microanatomical features in ileum of higher tetrapod representatives (pigeon and mouse) were characterized by the presence of villi lined with simple columnar epithelium and scattered goblet cells as well as intestinal glands (crypts of Lieberkühn) at the bases of the intestinal villi. In the toad rectum, the mucosal layer was similar to that of the ileum but with shorter villi and more numerous goblet cells. However, the mucosal layer of the rectum in the lizard had low numbers of absorptive columnar epithelial cells with abundant goblet basal cells. Comparatively, the pigeon’s rectal mucosa had almost a similar structure to that of ileum but in leaf-like shaped villi. Finally, the rectum of the mouse has narrow rectal pits, instead of villi, lined with goblet cells and absorptive epithelial cells. Histochemically, the ileum in the four studied tetrapod representatives showed varying biodistribution profiles of neutral, sulfated and carboxylated mucins. There are variations encountered in the intestinal brush border and goblet cells of villi in all species as well as the crypts of Lieberkühn in higher tetrapods. Also, the rectum of all tetrapod species showed weak to strong positive signals for the three types of mucins in the brush border and goblet cells of villi in all species and crypts of Lieberkühn in higher tetrapods as well. In addition, the brush border of toad’s rectum was lacking sulfated mucins and that of the lizard did not have any type of mucins. The data of this study will contribute to understand the relationship between the microanatomical features and mucins biodistribution profiles in the mucosal layer of tetrapod intestinal tract and their functions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The digestive system of vertebrates is consisted of many organs performing multiple functions (Duellman and Trueb 1994; Stevens and Hume 2004). In tetrapods the gastrointestinal tract (GIT) is mainly composed of upper GIT and lower GIT. The histological structures of these regions are almost similar to each other in most of tetrapod representatives but with different functions. However, some histological and microanatomical variations in the mucosal layer of each class needs more investigations. Morphologically, the intestinal tract of most amphibians and reptilians is composed mainly of duodenum, ileum, rectum and ends with cloaca (Duellman and Trueb 1994; Vitt and Caldwell 2013; Kardong 2006). However, in avians and mammalians the small intestine is divided into duodenum, jejunum and ileum. While, the large intestine in avians composed form rectal ceca and rectum (Denbow 2015) and the mammalians large intestine consists of cecum, colon and rectum (Kardong 2006).
Histologically, the ileum of most tetrapod representatives is composed of four main layers; serosa, muscularis, submucosa and mucosa (Romer 1970). The mucosa layer showed different histological architectures in each class. For example, the mucosa layer of ileum in amphibians and reptilians is composed of a simple columnar epithelium covered with brush border in its apical portions. These epithelial cells in participation with goblet cells line the intestinal folds as described previously in Boana albopunctata and Boana raniceps (as amphibian models) and Chamaeleon africanus and Uromastyx aegyptiaca (as reptilian models) (Hamdi 2012; Hamdi et al. 2014; Valverde et al. 2019). Comparatively, the avian mucosa layer of ileum showed elongated villi lined with simple columnar epithelium and scattered goblet cells which invaginate at the base between the crypts of Lieberkühn and continuous with the mucosal epithelium (Klasing 1999). Similar to aves, the ileum mucosa of mammals is composed of short cylindrical villi with crypts of Lieberkühn located among its bases (Malewitz 1965).
Microanatomically, the rectum of lower tetrapod (amphibians and reptilians) is similar to ileum but with big increase in goblet cells number, which are important for food digestion and lubrication of undigested food (Wright 1982). In avians, the rectum has the same cell types as those of ileum but the villi are shorter with leaf-like shape and have more goblet cells (Hamdi 2013). However, the rectum of mammals contains a large number of tubular intestinal glands (crypts of Lieberkühn). These glands are lined with goblet cells, Paneth cells, absorptive cells, and a small number of enteroendocrine cells (Mescher 2013).
Histochemically, mucus which is secreted by goblet cells is considered as the first defense line against physical and chemical injuries initiated by ingested food and microbial products. It is well known that, mucins is the main component of the mucous (Hollingsworth and Swanson 2004; Andrianifahanana et al. 2006). Several studies have been done concerning the biodistribution of mucins in the gastrointestinal tract of some amphibian (class: Anura), and reptilian species (Ferri et al. 2001, 1999; Loo and Wong 1975). For example, it was proposed that the small and large intestine of Ceratophrys ornate (as amphibian model) and Laudakia Stellio and Varanus niloticus (as reptilian models) showed a positive signal of acidic and neutral mucins in goblet cells, apical cytoplasmic portion and the apical membrane (Fry and Kaltenbach 1999; Ahmed et al. 2009; Shalaby 2012). In some avian representatives, for example Tyto alba and Gallus gallus domesticus, the goblet cells of intestinal villi and crypts of Lieberkühn in the small and large intestine showed positive signals of acidic and neutral mucins (Suprasert et al. 1987; El-Banhawy et al. 1993; El-Sayyad 1995). While in mammals the small and large intestine showed a positive signal of neutral and mixed mucins in different mammalians species such as Acomys Spinosissimus, Crocidura cyanea and Amblysomus hottentotus (Boonzaier et al. 2013).
Previously we reported the microanatomical and histochemical biodistribution profiles of different types of mucins in the esophagus and stomach of some tetrapod representatives (Awaad et al. 2022). Here in this study we will compare the microanatomical and biodistribution profiles of different types of mucins in mucosa layer of small (ileum) and large intestine (rectum) in some tetrapod representatives. We believe that, the variation in function of different cell types in the mucosa layer of ileum and rectum reflects variances in microanatomy of mucosa layer and the secretions of neutral and acidic mucins. The data obtained from this study will provide us with some information about the function of some microanatomical structures located in the mucosal layer of lower GIT and the localization and biodistribution profiles of different types of mucins.
Materials and methods
Materials
Nuclear fast red, alcian blue, hematoxylin and eosin, basic fuchsin stains and periodic acid were purchased from sigma Aldrich (Ontario, Canada).
Experimental design and study objectives
Using histological and histochemical techniques we will comparatively investigate the morphological, microanatomical, and histochemical biodistribution profiles of the different types of mucins in the mucosal layer of ileum and rectum of tetrapod representatives. To achieves these objectives, the tetrapod representatives were divided into four groups each group contains 5 adult males. The animal groups include toads (Bufo regularis), lizards (Trachylepis quinquetaeniata), pigeons (Columba livia domestica) and mice (Mus musculus) as amphibian, reptilian, avian and mammalian representatives respectively.
Animal artificial habitat design and maintenance
Each animal group was conserved and housed in a special artificial cage and provided with its own food at Animal Facility at Department of Zoology, Faculty of Science, Sohag University (Awaad et al. 2022). The amphibian representatives (male toads) were collected from canals and drains nearby agricultural fields in Sohag Governorate during the period of July to August 2020. The collected toads were bred in an artificial habitat designed to imitate the real toad’s habitat. The toad’s habitat was prepared of a glass cage with dimensions (50 cm × 30 cm × 30 cm). This cage was designed and filled with some wetted soil, water, and some grass in order to stimulate the real habitat in which the toads live. The toads were freely allowed to water, house flies (Musca domestica) and earth worms (Allolobophora caliginosa) as a standard daily food which provided by Department of Zoology at Sohag University. The reptilian representatives (male lizards) were collected from some area located in Nile River banks in Sohag Governorate during the period from June to August 2020. The prepared habitat for lizards is composed mainly of a glass cage similar to toad’s cages in the dimensions but settled with sand instead of wetted soil and medium to large size rocks to stimulate the hard nature habitat of reptiles. The daily food introduced to the lizards was similar to that of toads. The adult males of pigeons were raised in artificial cages prepared specifically for upbringing of birds especially pigeons. Due to large size of pigeons, each pigeon was raised separately in stainless-steel cage with dimensions of (50 cm × 40 cm × 40 cm) bedded with wooden saw dust and settled with water source and food container. Moreover, these cages are composed of stainless-steel cages and settled with mesh covered to allow access of light and ventilation. The daily foods introduced to the pigeons were wheat seeds (Triticum aestivum) and sorghum seeds (Sorghum bicolor). The adult males of mice were breeded and rose in an artificial cages similar to that of pigeons (5 mice/cage). The mice had free access to rodent chow which composed mainly of 23% protein, 5.5% fats and 65% fibers. The detailed information about the animal and food contents were described in Table 1. All animals were kept and housed in the animal facility at standard conditions such as room temperature (25 °C) and 12 h. light:12 h. dark for 2 weeks. The experimental procedures, animal handling and maintenance in this experiment were approved and certified by the local Animal and Experimentation Committee in the Department of Zoology, Faculty of Science, Sohag University.
Animal dissection and organs collection
To achieve the histological and histochemical objectives of this study, animals were anesthetized using an over dose of diethyl ether and dissected carefully at sterilized area. Ileum and rectum of all animals in the four groups were collected and fixed in Carnoy’s fixative for 1 h. Thereafter, organs were dehydrated in ascending series of ethyl alcohols, cleared by methyl benzoate and toluene then infiltrated in paraffin wax. Sections from ileum and rectum were cut at 7 µm by the rotary microtome (RM 2125RTS; Leica Biosystems, China). Then, sections were mounted on clean glass slides and firmly affixed for 24 h at 35 °C using incubator (Venticell 55 Comfort, MMM Medcenter Einrichtungen, Germany).
Histological investigation
For histological investigations, sections were deparaffinized by xylol, rehydrated in series grade of alcohols and then washed in the distilled water. Then sections were stained by hematoxylin for 1 min, washed in tap water for 8 min. and rinsed in distilled water. Later, sections were stained by eosin for 3–5 min, dehydrated through ascending series of ethyl alcohol and cleared in xylol. Stained sections were then mounted by DPX, covered by glass covers and examined under light microscope (Axio Scope.A1, Carl ZEISS, Germany) (Troyer 1980).
Histochemical investigation
For histochemical investigations, we used Preparation of periodic acid Schiff (PAS) and Alcian blue staining protocols to detect neutral and acidic mucins respectively in the mucosal layer of ileum and rectum of each tetrapod representative.
Preparation of periodic acid Schiff (PAS) reagent
According to Troyer (1980), PAS stain constitutes from periodic acid solution (0.5%) for oxidation of neutral mucins. Also, Schiff reagent is composed mainly of 1% basic fuchsin and 1.9% sodium metabisulfite dissolved in hydrochloric acid (0.15 M).
Preparation of Alcian blue solution
Based on Bancroft and Gamble (2008) Alcian blue statin solution were prepared in two different pHs; pH 2.5 and pH 1 for demonstration of carboxylated and sulfated mucins respectively. Alcian blue stain solution (1%) with pH 2.5 was prepared by mixing 1 gm of Alcian blue stain into 100 ml from 3% acetic acid solution. Furthermore, Alcian blue stain (1%) solution with pH 1 was prepared by mixing 1 gm from Alcian blue stain into 100 ml of 0.1 M hydrochloric acid solution.
Neutral mucins detection
To investigate the biodistribution profiles of neutral mucins in the mucosal layer of ileum and rectum, dewaxed sections were rehydrated using descending series of ethyl alcohols and washed in distilled water. Then sections were incubated in periodic acid solution as a weak oxidizing agent of neutral mucins for 5 min. After oxidizing process, sections were washed carefully by distilled water for 2 min and incubated in Schiff reagent in room temperature for 15 min. Subsequentially counter staining was carried out using hematoxylin stain for 10 s, washed in tap water for 5 min and followed by washing again in distilled water. Hereafter, stained sections were dehydrated by ascending series of ethyl alcohols, cleared by xylol and mounted by DPX (Bancroft and Gamble 2008).
Carboxylated mucins detection
Alcian blue staining protocol at pH 2.5 was used to detect carboxylated (non-sulfated) mucins in different tissues (Machado-Santos et al. 2014). In this protocol, sections were deparaffinized in xylene and rehydrated using ethyl alcohol to distilled water. Then hydrated sections were stained for 30 min in Alcian blue stain solution at pH 2.5 at room temperature. Thereafter, sections were washed by tap water for 5 min followed by rinsing in distilled water and counterstained by nuclear fast red stain. Finally, sections were dehydrated, cleared by clearing reagent (xylol) and permanently mounted by DPX (Lai and Lü 2012).
Sulfated mucins detection
For the detection of sulfated mucins in tissues, Alcian blue stain was prepared with pH 1 as mentioned previously (Machado-Santos et al. 2014). Deparaffinized sections from tissues containing sulfated mucins were rehydrated in distilled water and incubated for 30 min in Alcian blue solution (pH 1) at room temperature. Sections then were washed in tap water for 5 min followed by distilled water, then counter stained with nuclear fast red. Thereafter, stained sections were dehydrated by ethyl alcohols, cleared by xylene and mounted by DPX (Bancroft and Gamble 2008).
Semi-quantitative analysis of histochemical stain signals
The staining intensities of sections stained by PAS, Alcian blue (pH 2.5) and Alcian blue (pH 1) stains expressing the amount of neutral, carboxylated and sulfated mucins respectively, were indicated by (+++) for highly positive, (++) for moderately positive, (+) for weakly positive, and (−) for negative signal.
Results
Histological structure of ileum in tetrapod
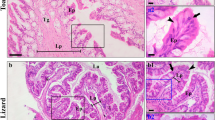
The present study revealed that the ileum of all tetrapod representatives was composed of four main layers; serosa, muscularis, submucosa and mucosa, arranged from outside to inside respectively. There are many histological variations encountered in the intestinal wall including all layers among different studied species. The main variations were observed clearly in the mucosal layer as indicated in Fig. 1A. The mucosal layer of toad ileum is regularly enfolded forming numerous elongated villi that are projecting into the inner lumen. Each villus is lined with columnar epithelium with scattered goblet cells (Fig. 1A1, A2). The lamina propria is composed of loose connective tissue and blood vessels and extending from the submucosa (Fig. 1A). Similarly, the lizard’s ileum mucosa (Fig. 1B) has many elongated villi projecting into the lumen which sound narrower than that of toad’s ileum. Each villus is lined with simple columnar epithelium having numerous goblet cells (Fig. 1B2). Compared to toad’s ileum, the lamina propria of the lizard’s ileum is much reduced with non-intact connective tissue and small numbers of blood vessels (Fig. 1B1, B2).
Photomicrographs of transverse sections of ileum stained with hematoxylin and eosin stains in toad (A) and lizard (B). The mucosal layer of toad’s ileum is composed mainly of longitudinal villi (Vi) extending in the lumen (Lu). Each villus is lined with simple columnar epithelium (Ep) intervened with scattered goblet cells (arrow heads). The core of the villus named as lamina propria (Lp) is composed of loose connective tissue and blood vessels. Similar to toad’s ileum, the mucosa of lizard’s ileum is composed of longitudinal folds extending into the lumen (Lu) and is composed of simple columnar epithelium (Ep) with goblet cells (arrow heads) but with reduced lamina propria (Lp). Scale bar, A and B = 50 µm; A1 and B1 = 10 µm and A2 and B2 = 5 µm
As to the higher tetrapoda representatives (pigeon and mouse), the muscularis layer of the pigeon’s ileum (Fig. 2A) appeared much larger and the submucosal layer much smaller than those of the lower tetrapod representatives (toad and lizard). But similar to the lower tetrapod, the mucosal layer of the pigeon has a large number of villi which are lined with simple columnar epithelia with numerous goblet cells (Fig. 2A2). Microanatomically, intestinal glands (crypts of Lieberkühn) were encountered in the ileum of the pigeon and with a histological structure similar to that of villi (Fig. 2A, A1). Similar to the pigeon, the mucosa of the mouse’s ileum (Fig. 2B) is composed of longitudinal villi with intestinal glands (crypts of Lieberkühn) but it has a wider lumen. In mouse’s ileum, the villi are relatively shorter than those of the pigeon and are formed of simple columnar epithelium with goblet cells (Fig. 2B1, B2). The thickness of the muscularis and submucosa layers of the mouse ileum is relatively small compared to other those of studied tetrapod representatives (Fig. 2B). The lamina propria of the upper tetrapod representatives (pigeon and mouse) is composed of loose connective tissue with blood vessels (Fig. 2A, B).
Photomicrographs of transverse sections of the ileum stained with hematoxylin and eosin stains in pigeon (A) and mouse (B). In pigeon’s ileum the mucosal layer is composed mainly of intestinal glands ((crypts of Lieberkühn) (CL)). These glands open in between the mucosal villi (Vi) rich in its lamina propria (Lp), and they are lined with simple columnar epithelium (Ep) with goblet cells (arrow heads) all facing the ileum’s lumen (Lu). The mucosal layer in mouse’s ileum as in pigeon has Crypts of Lieberkühn (CL), villi (Vi) lined with simple columnar epithelium (Ep) intervened with goblet cells (arrow heads) and surround the lamina propria (Lp), these villi extending into a wide lumen (Lu). Scale bar, A and B = 50 µm; A1 and B1 = 10 µm and A2 and B2 = 5 µm
Histological structure of rectum in tetrapod
The present study revealed a number of variations in the histological structure of the rectum among the different studied tetrapod representative, particularly in the mucosal layer. As in the other GIT regions, the rectal wall consists of the four main layers; the mucosa, submucosa, muscularis and serosa. The mucosa of toad like the case of ileum (Fig. 3A), is folded but it has more numerous and shorter villi than those of ileum. The villi are also lined with simple columnar epithelium rich in goblet cells, however, the goblet cells of the rectal villi are higher in number compared with those of ileum (Fig. 3A1, A2). The muscularis and submucosa layers are thicker in the rectum than in the ileum (Fig. 3A). Comparatively, the mucosa of the lizard’s rectum (Fig. 3B) is composed of few numbers of villi (Fig. 3B1). Each villus is lined with basal cells intervening a large number of goblet cells facing a narrow lumen (Fig. 3B2, A). The epithelium of villi rests on the lamina propria which followed by muscularis mucosa and muscularis.
Photomicrographs of transverse sections of the rectum stained with hematoxylin and eosin stains in toad (A) and lizard (B). The mucosal layer of toad’s rectum is composed of numerous villi with wide lamina propria (Lp), and lined with simple columnar epithelium (Ep), having scattered goblet cells (arrow heads) facing the inner lumen (Lu). The rectal mucosa of the lizard’s is composed of short villi and longitudinal folds lined by columnar epithelium (Ep) with massive number of goblet cells (arrow heads) resting on a relatively thick lamina propria (Lp).Scale bar, A and B = 50 µm; A1 and B1 = 10 µm and A2 and B2 = 5 µm
The mucosa of pigeon’s rectum (Fig. 4A) is composed of a large number of villi in leaf-like shape, each main projection is composed of a few numbers of mini branched villi. The mucosal layer is supported by muscular sublayer (muscularis mucosa) lying at the inner portion of the submucosa followed internally by crypts of Lieberkühn which open into the lumen via the villi. Each villus is lined with simple columnar epithelium having a large number of goblet cells (Fig. 4A1, A2). Comparatively, the mouse’s mucosal layer (Fig. 4B) also has a muscularis mucosa, which is thinner than that of pigeon. The muscularis mucosa was followed internally by lamina propria which support and underlines the lining epithelium which surrounding the crypts of Lieberkühn. The crypts are opened in the rectal lumen forming the pits the wall of which is lined with a simple columnar epithelium and goblet cells (Fig. 4B1, B2).
Photomicrographs of transverse sections of the rectum stained with hematoxylin and eosin stains in pigeon (A) and mouse (B). The mucosa of pigeon’s rectum is composed of muscularis mucosa (Mm) followed by Crypts of Lieberkühn (CL) at the base of the villi. The crypts are surrounded by lamina propria (Lp) and the villi are lined with simple columnar epithelial (Ep) and the goblet cells (arrow heads) facing the inner lumen (Lu). The mucosa of mouse’s rectum is composed also of small muscularis mucosa (Mm) followed by lamina propria (Lp) surrounding Crypts of Lieberkühn (CL) which open in the rectal pits. The rectal pits are lined with simple columnar epithelium with a large number of goblet cells (arrow heads) all are facing the inner lumen (Lu). Scale bar, A and B = 50 µm; A1 and B1 = 10 µm and A2 and B2 = 5 µm
Figure 5 summarizes comparatively the differences between the mucosal layer of the ileum (Fig. 5A–D) and the rectum (Fig. 5E–H) of the four tetrapod representatives. The mucosal layer of the ileum of all representatives is composed of a lining simple columnar epithelium with different number of goblet cells. The lining epithelium evaginates regularly to form numerous villi projecting into the lumen. The lamina propria supports the lining epithelium and surround the villi evenly in all studied tetrapods. Interestingly, a remarkable difference in the rectal mucosal architecture was observed in the different studied tetrapod species. The number of the goblet cells in the lizard’s villus epithelia is higher than those in the other tetrapod representatives. Additionally, the lamina propria in the rectum of all tetrapod representatives is larger than that located in the ileum.
Cartoon drawings showing the mucosal layer with detailed structures in ileum (A–D), rectum (E–H) of tetrapod representatives. The number of the goblet cells in the epithelial layer of rectum in most tetrapod representative is higher than those of the ileum. The lamina propria thickness differs from ileum to rectum and from class to class. Ep epithelium, Lp lamina propria, Gc goblet cells, Bv blood vessel, Bb brush border. Illustrations were drawn by adobe photoshop 2020
Mucins biodistribution in the intestinal tract of the studied tetrapod representatives
The localization of the mucins was mainly detected in the mucosal layer, particularly, in the goblet cells in all tetrapod representatives.
Neutral mucins biodistribution in ileum of tetrapod
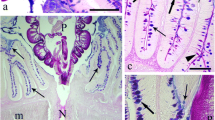
As shown in Fig. 6A, the mucosal layer of toad’s ileum showed a moderately positive signal of neutral mucins in the goblet cells and brush border (Fig. 6A1, A2). However, no signal was seen in the lamina propria, lining epithelium, submucosa and muscularis (Fig. 6A). The mucosal layer of the lizard’s ileum, in turn, showed a strong expression of neutral mucins in the goblet cells and brush border (Fig. 6B1), a moderate expression in lining epithelium (Fig. 6B2) and a weak expression in the muscularis and submucosa layers (Fig. 6B).
Photomicrographs of transverse sections of the ileum stained with PAS stain and counterstained with hematoxylin showing the localization and biodistribution of neutral mucins (purple color) in mucosa of toad (A) and lizard (B). The neutral mucins in the mucosa of toad’s ileum showed a moderately positive signal in the goblet cells of the villi (arrow heads) and brush border (arrow) but they were negative in muscularis, lamina propria (Lp) and lining epithelium (Ep). The mucosa of lizard’s ileum showed a strong positive signal in goblet cells (arrow heads) and brush border (arrow), a moderate positive signal in the lining epithelium (Ep) especially brush border and a weak signal in muscularis layer (Mu). Scale bar, A and B = 50 µm; A1 and B1 = 10 µm and A2 and B2 = 5 µm
The mucosal layer of pigeon’s ileum as shown Fig. 7A, showed a strong positive signal for neutral mucins in the goblet cells of villi and crypts of Lieberkühn as well as in the brush border (Fig. 7A1, A2). However, a weak positive signal of neutral mucins was detected in submucosa and muscularis (Fig. 7A). However, the neutral mucins were not detected neither in the lamina propria nor in the lining epithelial cells (Fig. 7A2). Comparatively, the mucosa of mouse’s ileum as shown in Fig. 7B, showed a moderate positive signal of neutral mucins in the brush border, goblet cells of villi and crypts of Lieberkühn (Fig. 7B2). In contrast, neutral mucins showed a weak positive signal in the muscularis (Fig. 7B), but they were negative signal in the lamina propria, lining epithelium and submucosa (Fig. 7B, B1).
Photomicrographs of transverse sections of the ileum stained with PAS stain and counterstained with hematoxylin showing the localization and biodistribution of neutral mucins (purple color) in mucosa of pigeon (A) and mouse (B). The mucosa of the pigeon’s ileum showed a strong positive expression of neutral mucins in the goblet cells of villi, crypts of Lieberkühn (arrow heads) and brush border (arrow). However, they are weakly positive in the muscularis (Mu), submucosa (SM) and negative in the lamina propria (Lp) as well as the lining epithelium (Ep). The mucosa of the mouse’s ileum showed a moderately positive in the goblet cells (arrow heads), brush border (arrow) of villi and crypts of Lieberkühn (CL). Muscularis (Mu) layer shows a weak positive, while lining epithelium (Ep) and lamina propria (Lp) showed no signals. Scale bar, A and B = 50 µm; A1 and B1 = 10 µm and A2 and B2 = 5 µm
Carboxylated mucins biodistribution in ileum of tetrapod
As shown in Fig. 8A, the mucosa of the toad’s ileum showed a strong positive signal of carboxylated mucins in the brush border and goblet cells (Fig. 8A1, A2). However, carboxylated mucins were not seen in the lamina propria (Fig. 8A1), lining epithelium (Fig. 8A2), submucosa and muscularis (Fig. 8A). The mucosa of lizard’s ileum, in turn, showed a moderate positive signal of carboxylated mucins in the brush border and goblet cells (Fig. 8B2). In contrast, the lamina propria (Fig. 8B1), the submucosa, the longitudinal muscle layer of muscularis and the serosa (Fig. 8B) showed a weak localization of carboxylated mucins. Moreover, no signals of the carboxylated mucins were seen in the lining epithelial cells nor in circular muscle layer of muscularis (Fig. 8B, B1).
Photomicrographs of transverse sections of the ileum stained with Alcian blue stain at pH = 2.5 and counterstained with nuclear fast red showing the localization and biodistribution of carboxylated mucins (blue color) in mucosa of toad (A) and lizard (B). The mucosa of toad’s ileum showes a strong positive signal in the brush border (arrow) and goblet cells (arrow heads), while it has no signals in all other tissuecompartments. The mucosa of the lizard’s ileum showes a moderately positive signal in goblet cells (arrow heads) and brush border (arrow); weakly postive signals in the submucosa (SM), lamina propria (Lp), serosa and longtudinal muscle layer of muscularis (double headed arrow) However, carboxylated mucins are negative in the lining epithelium (EP) and circular muscle layer of muscularis (bold arrow). Scale bar, A and B = 50 µm; A1 and B1 = 10 µm and A2 and B2 = 5 µm
The mucosa of the pigeon’s ileum as shown in Fig. 9A showed a strong expression of carboxylated mucins in the brush border and goblet cells of villi and crypts of Lieberkühn (Fig. 9A1). However, the carboxylated mucins had no localization neither in the lining epithelial cells (Fig. 9A2), nor in the lamina propria (Fig. 9A1) nor in the other tissue compartments (Fig. 9A). The mucosa of mouse’s ileum (Fig. 9B) showed a moderate positive signal in the villi brush border and weak signal in goblet cells of villi and crypts of Lieberkühn (Fig. 9B2). Finally, the carboxylated mucins were negative in the lamina propria, the lining epithelium and the rest of other tissue compartments (Fig. 9B, B1).
Photomicrographs of transverse sections of the ileum stained with Alcian blue stain at pH 2.5 and counterstained with nuclear fast red showing the localization and biodistribution of carboxylated mucins (blue color) in mucosa of pigeon (A) and mouse (B). The mucosa of the pigeon’s ileum shows a strong expression of carboxylated mucins in the brush border (arrow), goblet cells (arrow heads) of villi and crypts of Lieberkühn (CL); while it has no signals in the lining epithelium (Ep), lamina propria (Lp) and all other tissue compartments. The mucosa of the mouse’s ileum shows weakly positive signals in goblet cells (arrow heads) of villi and crypts of lieberkuhn (CL); and a moderatley positive signal in the brush border (arrow). carboxylated mucins are negative in the lining epithelium (Ep), lamina propria (Lp) and all other layers.. Scale bar, A and B = 50 µm; A1 and B1 = 10 µm and A2 and B2 = 5 µm
Sulfated mucins biodistribution in ileum of tetrapod
As shown in Fig. 10A, the mucosa of toad’s ileum reveled a moderate positive signal of sulfated mucins in the brush border and the goblet cells (Fig. 10A2). Sulfated mucins showed a weak positive signal in the lamina propria and submucosa (Fig. 10A1). However, they were negative in the lining epithelium and the muscularis layer (Fig. 10A). In comparison to toad’s ileum, the mucosa of the lizard’s ileum (Fig. 10B) showed a moderate positive signal of sulfated mucins in goblet cells and brush border (Fig. 10B2). However, sulfated mucins were not seen neither in the lining epithelium, nor in the lamina propria nor in the other layers (Fig. 10B, B1).
Photomicrographs of transverse sections of the ileum stained with Alcian blue stain at pH 1 and counterstained with nuclear fast red showing the localization and biodistribution of sulfated mucins (blue color) in mucosa of toad (A) and lizard (B). The mucosa of the toad’s ileum shows a moderately positive signal in the goblet cells (arrow heads) and brush border (arrow); a weakly positive in the submucosa (SM) and lamina propria (Ep) but no signals in the lining epithelium (Ep) and the other remaining tissue compartments were seen. The mucosa of the lizard’s ileum shows a moderately positive signal in the brush border (arrow) and goblet cells (arrow heads) but no signals are seen in the lining epithelium (Ep), lamina propria (Lp) and all other tissue compartments. Scale bar, A and B = 50 µm; A1 and B1 = 10 µm and A2 and B2 = 5 µm
The mucosa of the pigeon’s ileum (Fig. 11A) showed a strong expression of sulfated mucins in the brush border and goblet cells of villi and crypts of Lieberkühn (Fig. 11A2). However, the other layers in the pigeon’s ileum were negative (Fig. 11A, A1). In a similar manner, the mucosa of the mouse’s ileum (Fig. 11B) showed a strong positive signal of the sulfated mucins in goblet cells of villi and crypts of Lieberkühn (Fig. 11B1). Sulfated mucins also showed a moderate signal in the brush border (Fig. 11B2), but they were negative in the lining epithelium, lamina propria and the rest of tissue compartments (Fig. 11B).
Photomicrographs of transverse sections of the ileum stained with Alcian blue stain at pH 1 and counterstained with nuclear fast red showing the localization and biodistribution of sulfated mucins (blue color) in mucosa of pigeon (A) and mouse (B). The mucosa of the pigeon’s ileum shows a strong signal of sulfated mucins in the brush border (arrow) and goblet cells (arrow heads) of crypts of Lieberkühn (CL) and villi, but no signals are seen in the lining epithelium (Ep), lamina propria (Lp) and the rest of mucosal compartments. The mucosa of the mouse’s ileum shows a strong positive signal in the goblet cells (arrow heads) of villi and crypts of Lieberkühn; and a moderately positive in the brush border (arrows); while it has no signals in the lamina propria (Lp) and lining epithelium (Ep) as well as the remaining of the tissue compartments. Scale bar, A and B = 50 µm; A1 and B1 = 10 µm and A2 and B2 = 5 µm
Neutral mucins biodistribution in rectum of tetrapod
As shown in Fig. 12A, the rectal mucosa of the toad showed a high localization of the neutral mucins in the goblet cells and brush border (Fig. 12A2), a moderate localization in the muscularis (Fig. 12A) and a weak localization in the lamina propria (Fig. 12A1). However, neutral mucins were absent in lining epithelium and submucosa layers of the toad’s rectum. The mucosa of the lizard’s rectum (Fig. 12B) showed similar localization profiles of neutral mucins as shown in the toad’s rectum. Interestingly, the neutral mucins showed a strong positive signal associated with massive number of goblet cells (Fig. 12B2). Additionally, the lamina propria and submucosa showed a moderate positive signal of neutral mucins. However, the muscularis layer showed a weak signal of neutral mucins (Fig. 12B, B1).
Photomicrographs of transverse sections of the rectum stained with PAS stain and counterstained with hematoxylin showing the localization and biodistribution of neutral mucins (purple color) in the mucosa layer of toad (A) and lizard (B). The mucosa of the toad’s rectum shows a strong positive signal in the goblet cells (arrow heads) and brush border (arrow); a moderate signal in muscularis (Mu); and a weak signal in the lamina propria (Lp) but it has no signals in the lining epithelium (Ep) and submucosa (SM). The mucosa of the lizard’s rectum shows moderate positive signals in the goblet cells (arrow heads), lamina propria (Lp) and submucosa (SM) and a weak positive signal in the muscularis (Mu). Scale bar, A and B = 50 µm; A1 and B1 = 10 µm and A2 and B2 = 5 µm
Comparatively, the mucosa of the pigeon’s rectum (Fig. 13A) showed a strong positive signal of neutral mucins in the brush border and goblet cells of mucosal villi and crypts of Lieberkühn (Fig. 13A1, A2) and a weak signal in the lamina propria, muscularis mucosa, muscularis and submucosa layers (Fig. 13A, A1). The neutral mucins were, however, negative in the lining epithelium (Fig. 13A2). In the mouse’s rectum, the neutral mucins were heavily localized in the goblet cells of rectal pits and crypts of Lieberkühn. As well as in the brush border (Fig. 13B1). However, muscularis mucosa, submucosa and muscularis layers showed a weak positive signal of neutral mucins (Fig. 13B). Finally, the lining epithelium was negative from neutral mucins (Fig. 13B2).
Photomicrographs of transverse sections of the rectum stained with PAS stain and counterstained with hematoxylin showing the localization and biodistribution of neutral mucins (purple color) in mucosa of pigeon (A) and mouse (B). The mucosa of the pigeon’s rectum shows a strong positive signal in the brush border (arrows) and goblet cells of villi and crypts of Lieberkühn; weak signals in lamina propria (Lp), submucosa (SM) and muscularis (Mu) but a negative signal in the lining epithelium (Ep). The mucosa of the mouse’s rectum shows a strong positive signal of the neutral mucins in the brush border (arrows) and goblet cells of villi and crypts of Lieberkühn; weak positive signals in the submucosa (SM), muscularis mucosa (Mm) and muscularis (Mu) and negative signals in lamina propria (Lp) and lining epithelium (Ep). Scale bar, A and B = 50 µm; A1 and B1 = 10 µm and A2 and B2 = 5 µm
Carboxylated mucins biodistribution in rectum of tetrapod
As shown in Fig. 14A, the mucosa of the toad’s rectum showed only a strong positive signal of carboxylated mucins in the goblet cells of the mucosal villi (Fig. 14A2) but a weak signal in the submucosa and lamina propria layers (Fig. 14A1). No signals of carboxylated mucins were seen in the rest of all layers compartment. The goblet cells of mucosal fold in lizard’s mucosa were heavily loaded (Fig. 14B2), while submucosa was slightly loaded with carboxylated mucins (Fig. 14B). Unlike the toad’s mucosa, the lamina propria of lizard mucosa was negative for carboxylated mucins, in addition to the other layer compartments (Fig. 14B, B1).
Photomicrographs of transverse sections of the rectum stained with Alcian blue stain at pH 2.5 and counterstained with nuclear fast red showing the localization and biodistribution of carboxylated mucins (blue color) in mucosa of toad (A) and lizard (B). The mucosa of the toad’s rectum shows a strong expression of carboxylated mucins in the goblet cells (arrow heads) of villi; a weak positive signal in the lamina propria (Lp) and negative signals in the brush border (arrow), lining epithelium (Ep) and all other tissue compartments. The mucosa of the lizard’s rectum shows a strong positive signal in the goblet cells (arrow heads), a weak positive signal in the submucosa (SM) but no signal in the lamina propria (Lp), lining epithelium (Ep) and muscularis (Mu). Scale bar, A and B = 50 µm; A1 and B1 = 10 µm and A2 and B2 = 5 µm
The pigeon’s rectal mucosa showed a strong positive signal of carboxylated mucins in the brush border and goblet cells of mucosal villi and crypts of Lieberkühn (Fig. 15A1, A2). Also, a weak positive signal of carboxylated mucins was seen in the serosa, however, the other layers compartment were negative (Fig. 15A). In the rectum of the mouse (Fig. 15B), positive signals of carboxylated mucins were only seen in the brush border and goblet cells of the rectal villi and crypts of Lieberkühn (Fig. 15B1, B2).
Photomicrographs of transverse sections of the rectum stained with Alcian blue stain at pH 2.5 and counterstained with nuclear fast red showing localization and biodistribution of carboxylated mucins (blue color) in mucosa of pigeon (A) and mouse (B). The mucosa of the pigeon’s rectum shows a strong positive signal in the brush border (arrow) and goblet cells (arrow heads) of villi and crypts of Lieberkühn; a weak positive signal in the serosa (Se) and it has no signal in the lining epithelium (Ep) lamina propria (Lp) and all other tissue compartments. The mucosa of the mouse’s rectum shows a strong expression of carboxylated mucins in the brush border (arrow) and goblet cells (arrow heads) of rectal pits and crypts of Lieberkühn (CL) but it is negative in the lining epithelium (Ep), lamina propria (Lp) and all the remaining tissue compartments. Scale bar, A and B = 50 µm; A1 and B1 = 10 µm and A2 and B2 = 5 µm
Sulfated mucins biodistribution in rectum of tetrapod
In the toad’s mucosa (Fig. 16A) only the goblet cells of mucosal villi and the brush border had strong positive signals of sulfated mucins (Fig. 16A2) while all other layers were negative. Similarly, the mucosa of lizard’s rectum showed a strong positive signal of sulfated mucins only in a large number of goblet cells of mucosal villi (Fig. 16B1, B2) but all other layers were negative of sulfated mucins (Fig. 16B).
Photomicrographs of transverse sections of the rectum stained with Alcian blue stain at pH 1 and counterstained with nuclear fast red showing the localization and biodistribution of sulfated mucins (blue color) in mucosa of toad (A) and lizard (B). The mucosa of the toad’s rectum shows a strong positive signal in the brush border (arrow) and goblet cells (arrow heads) of the rectal villi but it has no signals in the lining epithelium (Ep), lamina propria (Lp) and all other tissue compartments. The mucosa of the lizard’s rectum shows a strong positive signal of sulfated mucins in the goblet cells (arrow heads but it has no signal in all epithelial cells and other tissue compartments. Scale bar, A and B = 50 µm; A1 and B1 = 10 µm and A2 and B2 = 5 µm
The sulfated mucins in the pigeon’s mucosa were heavily localized only in the brush border and the goblet cells of crypts of Lieberkühn and the mucosal villi (Fig. 17A1, 2). However, no signals of sulfated mucins were seen in the other rectal layers (Fig. 17A). The biodistribution profiles of the sulfated mucins in the mucosa of mouse’s rectum were nearly similar to those in the pigeon but in a lower level. Specifically, the mucosa of the mouse’s rectum showed a moderate positive signal of sulfated mucins in the goblet cells of mucosal pits and crypts of Lieberkühn (Fig. 17B2). While the brush border of the mucosal villi showed a weak signal (Fig. 17B1) the other layers were negative of sulfated mucins (Fig. 17B).
Photomicrographs of transverse sections of the rectum stained with Alcian blue stain at pH 1 and counterstained with nuclear fast red showing localization and biodistribution of sulfated mucins (blue color) in mucosa of pigeon (A) and mouse (B). The mucosa of the pigeon’s rectum shows a strong positive signal in the brush border (arrows) and goblet cells (arrow heads) of rectal villi and crypts of Lieberkühn (CL) but it has no signals in the other rectal compartments. The mucosa of the mouse’s rectum shows moderate positive signal in the goblet cells (arrow heads) of rectal villi and crypts of Lieberkühn (CL); a weak signal in the brush border (arrow) but it has no signals in the lamina propria (Lp), lining epithelium (Ep) as well as other rectal compartments. Scale bar, A and B = 50 µm; A1 and B1 = 10 µm and A2 and B2 = 5 µm
Figure 18 and Table 2 summarizing the localization and biodistribution profiles of neutral, carboxylated and sulfated mucins in both ileum and rectum mucosae of the four studied tetrapod representatives. In general, the goblet cells were loaded with different types of mucins in the ileum and the rectum of all studied tetrapod representatives with varied degrees of mucins level. The lining columnar epithelial cells were negative from all different types of mucins both in ileum and rectum. On the other hand, the brush border had positive expressions for all mucins with varying staining intensities in all ileal and rectal mucosae of lizard, pigeon, and mouse. Contradictory, the brush border in the villi of toad’s rectum showed a negative localization of sulfated mucins (Fig. 18F–I). However, the brush border of the lizard had a negative expression of all mucin types (Fig. 18D–F(II)).
Cartoon drawings of epithelial cell layer of the ileum (A–C) and rectum (D–F) of tetrapod representatives. The biodistribution of neutral mucins (A and D), carboxylated mucins (B and E) and sulfated mucins (C and F) was comparatively concluded in mucosa layer of ileum and rectum of toad (I), lizard (II), pigeon (III) and mouse (IV). Goblet cells are loaded with all three types of mucins in the mucosal layer of ileum and rectum in all tetrapod representatives. The enterocytes, however, are free of all types of mucins in all tetrapod representatives except apical cytoplasmic portions of the lizard’s ileum which are positive for neutral mucins. The brush border of ileum and rectum mucosae show positivity for all mucins in all tetrapod representatives. The toad’s is free of carboxylated mucins. Illustrations were drawn by adobe photoshop 2020
Discussion
The microanatomical features of the intestinal tract specially the mucosal layer showed variations among tetrapoda classes or even between two species in the same class. These variations are related to regional difference in the intestinal tract (duodenum, jejunum, ileum, rectum…. etc.) and the function of each region (Stevens and Hume 2004). Excitingly, the main differences were observed in the mucosal layer as compared to other layers. In amphibians such as Bufo regularis investigated in this study and Boana albopunctata and Boana raniceps in previous studies, the mucosal layer of the small intestine is composed of elongated villi lined with simple columnar epithelium and scattered goblet cells between absorptive cells (Valverde et al. 2019). Similarly, the lizard (Trachylepis quinquetaeniata manipulated by this study) has a mucosal layer similar to that of amphibians with many longitudinal folds facing the inner lumen but showing a reduction in the lamina propria. The same results were observed in many other reptilian models such as Chamaeleon africanus, Uromastyx aegyptiaca and Laudakia stellio (Hamdi 2012; Shalaby 2012; Hamdi et al. 2014). Interestingly, in our study, the lower tetrapod’s ileum didn’t show any presence of intestinal glands. However, the intestinal glands were found in the midgut of salamander and some reptiles (Luppa 1976), although, those of reptiles were less developed than those of birds and mammals (Stevens and Hume 2004).
The mucosal layer of ileum in the avians such as Columba livia domestica at the present study and Elanus caeruleus and Coturnix coturnix is composed of intestinal glands called crypts of Lieberkühn. Also, numerous longitudinal villi are lined with simple columnar epithelium and goblet cells (Ziswiler 1986; Hamdi 2013; Zaher et al. 2012). In mammals (Mus musculus in this study) the ileum showed thin muscularis layer in comparable to that of birds. However, the mucosal layer of mouse’s intestine is characterized with short cylindrical villi, and crypts of Lieberkühn which are surrounded by lamina propria and composed of several types of cells such as absorptive, Paneth cells and goblet cells. The results of this study are matching the previous study of three marsupials species (Didelphis albiventris, Monodelphis dimidiate, Lestodelphys halli and Atelerix albiventris) (Andrini et al. 2019; Jaji et al. 2019). The main difference between the mucosal layer of the lower tetrapod (toad and lizard) and the higher tetrapod (pigeon and mouse) is the presence of the crypts of Lieberkühn. Microanatomically, the structure of ileum is largely appropriate to its function and regional peculiarity. The ileal numerous villi are helping in increasing the luminal absorption surface, also its mucous goblet cells, act as a digestive, absorptive and protective functions as well as the lubrication of the undigested food passed into the large intestine (rectum) (Johnson et al. 2006; Valverde et al. 2019).
Similar to the ileum, the microanatomy of the rectal mucosa in the toad (bufo regularis as a model of the current study), is composed of elongated villi similar to those of the ileum but with shorter length and more numerous goblet cells (Claypole 1897). These observations agreed with those of Bufo melanostictus, Boana albopunctata, Boana raniceps (Loo and Wong 1975; Valverde et al. 2019). Otherwise in the reptilian model of this study (Trachylepis quinquetaeniata), the rectal mucosal layer is composed of massive number of goblet cells with basal cells interposed between them. These results agree with some extent of a previously published study using Uromastyx aegyptiaca, which has a rectal mucosa lined with columnar epithelium, basal cells and large number of goblet cells (Hamdi 2012). However, these data disagree with what was previously published using chameleon africanus and Laudakia Stellio which have showed rectal mucosae composed of simple columnar epithelium with few goblet cells (Shalaby 2012; Hamdi et al. 2014).
Comparatively, the rectal mucosal layer of the pigeon as a model for aves in the present study has a leaf-like shaped villi with a composition similar to those of ileum but shorter in length. These results agreed with those published previously using an avian model (Coturnix coturnix) (Klasing 1999; Zaher et al. 2012). Unlike the ileum, the mucosa of the mouse’s rectum showed no villi but instead there were rectal pits which are characterized with the presence of enterocytes and goblet cells with crypts of Lieberkühn among the pits. Recently, similar result were reported in some mammalian representatives such as Rhinolophus ferrumequinum, Lestodelphys halli and Monodelphis dimidiate (Suganuma et al. 1981). However, few smaller villi were observed in the rectum of Didelphis albiventris as a mammalian model (Scillitani et al. 2007; Andrini et al. 2019). The histological microanatomical features of rectum are appropriated and accommodate to its function as it reabsorbs fluids and electrolytes and collects the undigested food to pass it to the cloaca as reported previously (Martini and Ober 2006).
Histochemically, mucins are categorized into neutral (carrying no charges) and acidic mucins (carrying a carboxyl or sulfuric acid group) (Filipe and MI 1979). Generally, mucins play a role in lubricating food along the intestinal tract, protecting epithelium form abrasive movements of food, pathogens and toxins (Laux et al. 2005; Rose and Voynow 2006). It has been proposed that sulfated mucins play a vital role in chemical defense of epithelia against enzymatic degradation of the mucus barrier by bacterial glycosidase (Eggert-Kruse et al. 2000). The present study revealed that the ileum of amphibians exhibited a positive signal for neutral as well as acidic mucins in the goblet cells and brush border of toad’s epithelium. These data are consistent with those of a previously published study using two species of amphibians such as, salamander (Proteus anguinus) and the frog Rana aurora aurora (Ferri et al. 2001; Bizjak Mali and Bulog 2004). Also, the present study revealed that the lizard’s columnar epithelium located in the mucosal layer displayed a positive signal of neutral mucins only in its apical cytoplasmic portion. However, the goblet cells and brush border showed a positive for the three types of mucins. These findings agreed with those of Hamdi (2012) and Hamdi et al. (2014) who reported the same observations in Chamaeleon africanus and Uromastyx aegyptiaca.
Comparatively, in higher tetrapod representatives (pigeon and mouse), the ileum showed a localization with varying degrees of neutral, carboxylated and sulfated mucins in the intestinal villi, brush border and crypts of Lieberkühn. Similar biodistribution profiles were observed in the black winged kite (Elanus caeruleus) and chickens (Gallus gallus domesticus) digestive tract (Pastor et al. 1988; Hamdi 2013). The mammalian model in our study showed a moderate to strong localization of neutral mucins in the goblet cells and brush border of epithelial cells of ileum and rectum. In correspondence to the data observed in mouse, similar findings were seen in other models such as Amblysomus hottentotus, Acomys Spinosissimus and Rhinolophus ferrumequinum (Scillitani et al. 2007; Boonzaier et al. 2013).
The rectum of lower tetrapod representatives showed a localization of the three types of mucins in the goblet cells of the mucosal villi and brush border of ileum and rectum. Interestingly, the brush border of absorptive cells in toad’s rectal mucosa didn’t show any signal for carboxylated mucins. Absence of carboxylated mucins indicates the absence of sialomucins and presence of neutral and sulfomucins as reported previously in some mammalian species as (Cricetinae, Rattus and Mus musculus) (Sheahan and Jervis 1976). However, Triturus carnifex and Bufo melanostictus as amphibian models showed positive signals for both neutral and acidic mucins in the goblet cells of the large intestine (Loo and Wong 1975; Liquori et al. 2007). Similar to the data of the present study the goblet cells and brush border of the reptilian models, Uromastyx aegyptiaca, Chamaeleon africanus and Varanus salvator revealed positive signals for neutral and acidic mucins. In contrast, the columnar epithelial cells in these species showed a variation in amount of their content of neutral mucins (Hamdi 2012; Hamdi et al. 2014; Widyaningsih 2020). However, our data showed that most of absorptive endothelial cells of the ileum and rectum in all studied tetrapod representatives lack neutral and acidic mucins except those of reptiles. This may be due to the type of food intake and its carbohydrate contents.
On the other side, the higher tetrapod representatives of the present study (pigeon and mouse) showed a positive localization of neutral, carboxylated and sulfated mucins in most of mucosal layer compartments. Similar results were observed in Elanus caeruleus and Coturnix coturnix (avaian models) where a weak positive signal for all mucins was observed on the surface of epithelial cells in the former model and a stronger signal in the latter one. In contrast, the large intestine of the chicken (Gallus gallus domesticus) showed strong positive signals for the three types of mucins but with variation in strength between the goblet cells of villi and crypts of Lieberkühn (Suprasert et al. 1987; Zaher et al. 2012; Hamdi 2013). Interestingly, the biodistribution profiles of all mucins in our study mammalian model (mouse) was similar to a number of previously published studies using different mammalian models (Sheahan and Jervis 1976; Boonzaier et al. 2013; Johnson et al. 2016).
In conclusion the present study showed the microanatomical and histochemical variations in the mucosal layer of the intestinal tract (ileum and rectum) in some tetrapod representatives. These variations may be due to the different function of each region in the intestinal tract. The different mucins biodistribution profiles in the intestinal tract mucosal layer may indicate the different regional functions and its relation with different mucin types in the intestinal tract. Finally, the data of this study will help us to understand the relationship between the microanatomical features of the intestinal mucosa and their functions especially those related to mucins production.
Abbreviations
- GIT:
-
Gastrointestinal tract
- PAS:
-
Periodic acid Schiff
References
Ahmed Y, Ea E, Ae Z (2009) Histological and histochemical studies on the esophagus, stomach and small intestines of vara-nus niloticus. J Vet Anat 2:35–48. https://doi.org/10.21608/jva.2009.45136
Andrianifahanana M, Moniaux N, Batra SK (2006) Regulation of mucin expression: mechanistic aspects and implications for cancer and inflammatory diseases. Biochim Biophys Acta (BBA) 1765:189–222. https://doi.org/10.1016/j.bbcan.2006.01.002
Andrini LB, García MN, Inda AM et al (2019) Comparative histology of the digestive tract in three new world marsupials. Acta Zool 100:153–159. https://doi.org/10.1111/azo.12241
Awaad A, Rushdy A, Adly MA (2022) Comparative microanatomical and histochemical biodistribution profiles of different types of mucins in oesophageal gastric tract mucosa of some tetrapod representative. Histochem Cell Biol. https://doi.org/10.1007/s00418-021-02049-x
Bancroft JD, Gamble M (2008) Theory and practice of histological techniques, 6th edn. Elsevier Health Sciences, Amsterdam
Bizjak Mali L, Bulog B (2004) Histology and ultrastructure of the gut epithelium of the neotenic cave salamander, Proteus anguinus (Amphibia, Caudata). J Morphol 259:82–89. https://doi.org/10.1002/jmor.10171
Boonzaier J, Van der Merwe EL, Bennett NC, Kotzé SH (2013) A comparative histochemical study of the distribution of mucins in the gastrointestinal tracts of three insectivorous mammals. Acta Histochem 115:549–556. https://doi.org/10.1016/j.acthis.2012.12.003
Claypole EJ (1897) The comparative histology of the digestive tract. Trans Am Microsc Soc 19:83–92. https://doi.org/10.2307/3221131
Denbow DM (2015) Gastrointestinal anatomy and physiology. Sturkie’s avian physiology. Elsevier, Amsterdam, pp 337–366
Duellman WE, Trueb L (1994) Biology of amphibians. JHU Press, Baltimore
Eggert-Kruse W, Botz I, Pohl S et al (2000) Antimicrobial activity of human cervical mucus. Hum Reprod 15:778–784. https://doi.org/10.1093/humrep/15.4.778
El-Banhawy M, Mohallal ME, Rahmy TR, Moawad TI (1993) A comparative histochemical study on the proventriculus and ileum of two birds with different feeding habits. J Egypt Ger Soc Zool 11:155–174
El-Sayyad HIH (1995) Structural analysis of the alimentary canal of hatching youngs of the owl Tyto alba alba. J Egypt Ger Soc Zool 16:185–202
Ferri D, Liquori GE, Natale L et al (2001) Mucin histochemistry of the digestive tract of the red-legged frog Rana aurora aurora. Acta Histochem 103:225–237. https://doi.org/10.1078/0065-1281-00582
Ferri D, Liquori GE, Scillitani G (1999) Morphological and histochemical variations of mucous and oxynticopeptic cells in the stomach of the seps, Chalcides chalcides. J Anat 194(Pt 1):71–77. https://doi.org/10.1046/j.1469-7580.1999.19410071.x
Filipe MI (1979) Mucins in the human gastrointestinal epithelium: a review. Investig Cell Pathol 2(3):195–216
Fry AE, Kaltenbach JC (1999) Histology and lectin-binding patterns in the digestive tract of the carnivorous larvae of the Anuran, Ceratophrys ornata. J Morphol 241:19–32. https://doi.org/10.1002/(SICI)1097-4687(199907)241:1%3c19::AID-JMOR2%3e3.0.CO;2-4
Hamdi H (2012) Anatomical, histological and histochemical adaptations of the reptilian alimentary canal to their food habits: I. Uromastyx aegyptiaca. Life Sci 9:84–104
Hamdi H (2013) Anatomical, histological and histochemical adaptations of the avian alimentary canal to their food habits: II- Elanus caeruleus. Int J Sci Eng Res 4:1355–1364
Hamdi H, El-Ghareeb A, Zaher M et al (2014) Anatomical, histological and histochemical adaptations of the reptilian alimentary canal to their food habits: II-Chamaeleon africanus. World Appl Sci J 30:1306–1316
Hollingsworth MA, Swanson BJ (2004) Mucins in cancer: protection and control of the cell surface. Nat Rev Cancer 4:45–60. https://doi.org/10.1038/nrc1251
Jaji AZ, Akano JJ, Atabo SM et al (2019) Morphometry and histology of the gastrointestinal organs of the African White Breasted Hedgehog (Atelerix albiventris). J Vet Anat 12:1–14. https://doi.org/10.21608/JVA.2019.98374
Johnson LR, Barret KE, Gishan FK et al (2006) Physiology of the gastrointestinal tract. Elsevier, Amsterdam
Johnson O, Marais S, Walters J et al (2016) The distribution of mucous secreting cells in the gastrointestinal tracts of three small rodents from Saudi Arabia: Acomys dimidiatus, Meriones rex and Meriones libycus. Acta Histochem 118:118–128. https://doi.org/10.1016/j.acthis.2015.12.003
Kardong KV (2006) Vertebrates: comparative, anatomy, function, evolution. Washington State University, Washington
Klasing KC (1999) Avian gastrointestinal anatomy and physiology. Seminars in avian and exotic pet medicine. Elsevier, Amsterdam, pp 42–50
Lai M, Lü B (2012) 304 - tissue preparation for microscopy and histology. In: Pawliszyn JBT-CS and SP (ed). Academic Press, Oxford, pp 53–93
Laux DC, Cohen PS, Conway T (2005) Role of the mucus layer in bacterial colonization of the intestine. In: Jataro JP (ed) Colon mucosal surfaces. ASM Press, Washington, pp 199–212
Liquori GE, Mastrodonato M, Zizza S, Ferri D (2007) Glycoconjugate histochemistry of the digestive tract of Triturus carnifex (Amphibia, Caudata). J Mol Histol 38:191–199. https://doi.org/10.1007/s10735-007-9087-4
Loo SK, Wong WC (1975) Histochemical observations on the mucins of the gastrointestinal tract in the toad (Bufo melanostictus). Acta Anat 91:97–103. https://doi.org/10.1159/000144374
Luppa H (1976) Histology of the digestive tract. Morphol E, pp 225–313
Machado-Santos C, Pelli-Martins AA, Abidu-Figueiredo M, de Brito-Gitirana L (2014) Histochemical and immunohistochemical analysis of the stomach of Rhinella icterica (Anura, Bufonidae). J Histol 2014:872795. https://doi.org/10.1155/2014/872795
Malewitz TD (1965) Normal histology of the digestive tract of the mouse. Okajimas Folia Anat Jpn 41:21–47. https://doi.org/10.2535/ofaj1936.41.1_21
Martini F, Ober WC (2006) Martini’s atlas of the human body, 7th edn. Pearson/Benjamin Cummings, San Francisco
Mescher AL (2013) Junqueira’s basic histology: text and atlas, 13th edn. McGraw-Hill Medical, New York
Pastor LM, Ballesta J, Madrid JF et al (1988) A histochemical study of the mucins in the digestive tract of the chicken. Acta Histochem 83:91–97. https://doi.org/10.1016/S0065-1281(88)80076-X
Romer AS (1970) Vertebrate body. Saunders, Philadelphia
Rose MC, Voynow JA (2006) Respiratory tract mucin genes and mucin glycoproteins in health and disease. Physiol Rev 86:245–278. https://doi.org/10.1152/physrev.00010.2005
Scillitani G, Zizza S, Liquori GE, Ferri D (2007) Lectin histochemistry of gastrointestinal glycoconjugates in the greater horseshoe bat, Rhinolophus ferrumequinum (Schreber, 1774). Acta Histochem 109:347–357. https://doi.org/10.1016/j.acthis.2007.02.010
Shalaby SY (2012) Anatomical, histological, and scanning electron microscopic studies of the alimentary canal of Laudakia stellio (agamidae). Egypt J Exp Biol 8:1–7
Sheahan DG, Jervis HR (1976) Comparative histochemistry of gastrointestinal mucosubstances. Am J Anat 146:103–131. https://doi.org/10.1002/aja.1001460202
Stevens CE, Hume ID (2004) Comparative physiology of the vertebrate digestive system, 2nd edn. Cambridge University Press, Cambridge
Suganuma T, Katsuyama T, Tsukahara M et al (1981) Comparative histochemical study of alimentary tracts with special reference to the mucous neck cells of the stomach. Am J Anat 161:219–238
Suprasert A, Fujioka T, Yamada K (1987) The histochemistry of glycoconjugates in the colonic epithelium of the chicken. Histochemistry 86:491–497. https://doi.org/10.1007/BF00500622
Troyer H (1980) Principles and techniques of histochemistry. Wiley, Hoboken
Valverde BSL, Fanali LZ, Franco-Belussi L, de Oliveira C (2019) Comparative morphology of the digestive tract of two Neotropical tree frogs (Genus Boana). Zool Anz 281:44–52. https://doi.org/10.1016/j.jcz.2019.05.002
Vitt LJ, Caldwell JP (2013) Herpetology: an introductory biology of amphibians and reptiles. Academic Press, Cambridge
Widyaningsih R (2020) Histological structure of varanus salvator intestine. Proc Int Conf Sci Eng. https://doi.org/10.14421/icse.v3.482
Wright L (1982) The IUCN amphibia-reptilia red data book. IUCN
Zaher M, El-Ghareeb AW, Hamdi H, AbuAmod F (2012) Anatomical, histological and histochemical adaptations of the avian alimentary canal to their food habits: I-Coturnix coturnix. Life Sci J 9:253–275
Ziswiler V (1986) Comparative morphology of the avian digestive tract. In: Congress of International Ornithology XIX. pp 2436–2444
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Awaad, A., Rushdy, A. & Adly, M.A. Comparative microanatomical and histochemical biodistribution profiles of different types of mucins in the intestinal mucosa of some tetrapod representatives. J Mol Histol 53, 449–472 (2022). https://doi.org/10.1007/s10735-022-10071-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-022-10071-z