Abstract
Testosterone (T) deficiency is prevalent particularly in elderly men and lead to physical and sexual morbidities. Although low levels of T are associated with low urinary tract symptoms, the correlation between T deficiency and bladder dysfunction is not clearly identified. The aim of this study was to investigate the effect of high dose testosterone replacement therapy (TRT) on the histological structure of the UB in castrated rats. Twenty-five adult male rats were divided into three groups: control, castrated and castrated + TRT. T was administrated in high dose (100 mg/kg) two intramuscular injections/week for 60 days. UB sections were prepared and stained with H&E, Masson’s trichrome and immunohistochemical detection of Cytokeratin 20 (Ck20). All data were morphometrically and statistically analyzed. In castrated group, significant atrophy of the urothelium (P < 0.001) accompanied with widening of the corium were observed. The smooth muscle appeared thin with marked increase in the collagen fibers. On treating the castrated group with TRT, atypical Ck20 expression as well as significant increase in urothelial thickness (P < 0.05) and smooth muscle/collagen ratio (P < 0.001) were detected. In castrated rat model, high dose TRT has a positive effect on the UB smooth muscle rather than the urothelium which acquired atypical patterns.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Testosterone (T) deficiency is highly prevalent particularly in elderly men and frequently lead to physical and sexual morbidities (Corona et al. 2011). Symptomatic late-onset hypogonadism (SLOH) becomes manifested when declining testosterone levels give rise to unwanted symptoms such as erectile dysfunction, reduced bone density and muscle strength, and the metabolic syndrome (Yassin et al. 2014). Although low levels of T are associated with low urinary tract symptoms (LUTS), the correlation between T deficiency and bladder dysfunction is not clearly identidied (Litman et al. 2007).
Besides its role in improving the metabolic syndrome, obesity parameters and other health-related quality of life (Yassin et al. 2014), testosterone replacement therapy (TRT) can improve bladder function and reversing its wall alterations by increasing urinary bladder (UB) smooth muscle in late-onset hypogonadal men with urogenital dysfunction (Tek et al. 2010). TRT acts on androgen receptors (AR) which are widely expressed in the bladder, including urothelium, muscularis propria and neurons (Salmi et al. 2001; Celayir et al. 2002). However, it is not known whether these beneficial effects of TRT can be continued in the long term (Yassin et al. 2014). Moreover, the significance of low T as well as the exact mechanism of TRT-induced improvement of LUTS are not well-understood (Kalinchenko et al. 2008).
Testosterone forms such as testosterone enanthate (TE) and testosterone undecanoate (TU) are frequently used in hypogonadal men. TU is given in less frequent dosage than TE (Jockenhovel et al. 2009). Therefore when fertility is not desired, TRT is the gold standard well tolerated medication in hypogonadal men (Corona et al. 2011). However other studies demonstrated that TRT is not very satisfactory for hypogonadic patients because the available T preparations weren’t satisfactory to mimic the exact physiological pattern of T secretion in men (Giagulli et al. 2011b).
A single injection of TRT (TU 100 mg/kg) is effective in inducing physiological testosterone levels in castrated (CAST) rats for about 4 weeks which can be extended to 6 weeks by higher doses of TRT (TU 500 mg/kg) given as a single injection resulting in supraphysiological testosterone levels (Callies et al. 2003). However, the effect of high dose TRT on the histological structure of the UB specifically regarding TRT effect on the urothelium of hypogonadic CAST model are not frequently manipulated.
Cytokeratins are a family of intermediate-filament polypeptides present in epithelial cells (Moll 1991). In non-neoplastic urothelium, the expression of Ck20 is restricted to the superficial umbrella cells and occasionally the intermediate cells (Mallofre et al. 2003). Atypical Ck20 expression is a reliable, positive marker and indicator of dysplastic changes in urothelial mucosa. Thus, it could be used to distinguish dysplastic changes from reactive atypia (Kunju et al. 2005).
The aim of this study was to investigate the effect of high dose TRT on the histological structure of the UB, including the expression of Ck20, in an experimental model of hypogonadism induced by castration in mature male rats.
Experimental design
Twenty-five adult male albino rats weighing 180–200 gm were provided by the Center of Animal Housing of the Faculty of Pharmacy, Mansoura University, Egypt. The animals were placed in standard plastic cages under 12/12 h light/dark cycle with 23–25 °C room temperature. All animals were received standard laboratory animal’s chow and water ad labitum during the whole period of experiment. All surgical and experimental procedures were approved by the Institutional Laboratory Animal Care and Use Committee of Mansoura Faculty of Medicine and were conducted in accordance with their guidelines.
The animals were divided into three groups: control group (five animals), castrated group (CAST group, ten animals) and castrated group followed by TRT (CAST + TRT group, ten animals). Testosterone (Cidoteston, 250 mg Testosterone enanthate, Cid, Egypt) was administrated after castration in a high dose of 100 mg/kg two intramuscular injections every week for 60 days.
Surgical procedure (CAST rat models)
Rats were anesthetized by intraperitoneal injection with sodium pentobarbital (50 mg/kg of body weight). A single dose of ciprofloxacin (20 mg/kg) was used for prophylactic antibiotic therapy. After disinfecting the skin of the surgical site, the scrotum was then opened through a midline incision until the testes were fully exposed. The spermatic cords were ligated and both testes were removed (Gallo et al. 2012).
Histological and immunohistochemical assessments
After 60 days, all rats were sacrificed by cervical dislocation, both ureters were ligated, the UB was filled with 10 % formalin solution through a catheter transurethrally and kept distended overnight. Fixed bladder tissues were rinsed in PBS, dehydrated through graded ethanol washes, and embedded in paraffin. Bladder Sections (3–4 µm thickness) were cut, prepared and stained with H&E for histopathological changes, Masson’s trichrome for evaluation of smooth muscles and collagen contents (Bancroft and Layton 2013) and immunohistochemical detection of Ck20 (Kunju et al. 2005).
Immunohistochemistry
Immunohistochemistry was performed using refined labeled streptavidin–biotin technique (LSAB + Kit, HRP, DakoCytomation, Carpinteria, CA, USA) for Ck20. After paraffin removal and hydration, slides were treated with protease 1 enzyme for 16 min (for Ck20) for optimal antigen retrieval before immunostaining. Four µm-thick sections were stained with monoclonal antibodies against Ck20 (DakoCytomation) at 1:25 dilution for 32 min at 42 °C. A Ventana Basic DAB Detection Kit was used according to manufacturer’s specifications for Ck20 and staining was performed on the Ventana ES autostainer (Ventana Medical Systems, Tucson, AZ, USA). Sections were then counterstained with Gills hematoxylin (Kunju et al. 2005).
Image capture
Tissue sections were examined and images were digitally captured using a hardware consisting of a high-resolution color digital camera mounted on an Olympus microscope (Olympus, Japan), connected to a computer.
Morphometric studies
Thickness of the urothelium
Thickness of the urothelium was measured from images captured from bladder sections stained with H&E at a magnification of 1000 in each animal. The linear distance (in µm) between basement membrane of the urothelium to its luminal surface was measured by Image-J software version 1.43 (NIH, Bethesda, Maryland). Ten measurements were estimated per field, and 5 fields were evaluated per section.
Ratio between the smooth muscle and collagen
In bladder sections stained by Masson’s trichrome, the areas of collagen and smooth muscle were semi-quantitatively measured by Image-Pro Plus 6.0 software (Media Cybernetics, Bethesda, MD) based on color segmentation technique. The ratio between the area percentage of the relative contents of smooth muscle and collagen were calculated, taking the Masson’s trichrome positive area as an assessment parameter. For each animal, five sections were acquired, and ten fields were evaluated per section.
Assessment of serum testosterone levels
Blood samples from every animal of each group were collected for measurement of serum testosterone concentration using ELISA reader (BIOLINE BPR08) at 1, 30 and 60 days from the start of the experiment. Samples were centrifuged at 2000×g for 20 min and T concentration was measured according to the guidelines supplied with the kit (UBI Magiwel Total Testosterone kit, United Biotech Inc. Nahata and Dixit 2012).
Statistical analysis
All statistical data analysis and graphs were carried out using SPSS 16.0 (SPSS, Chicago, IL, USA) and Microsoft Excel 13. Data were presented in mean (±) standard deviations (SD) by using ANOVA test for comparison between groups. The differences were considered significant when probability (P) was <0.05.
Histological results
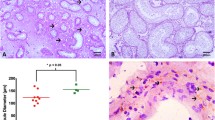
Histological examination of the UB of the control group stained with H&E showed normal folded thick mucosa which was formed of transitional epithelium separated from the corium by a basement membrane (Fig. 1a, b). The urothelium was formed of superficial umbrella cells with underlying inverted flask shaped cells lying on intermediate cells and basal cell layers. The cytoplasm of most of these cells was vacuolated and empty due to its glycogen content. In addition, the nuclei appeared central and vesicular and occasionally two nuclei were seen in some cells (Fig. 2a). The underlying connective tissue corium was thin separating the mucosa from thick well developed musculosa (Fig. 1a, b).
Photomicrographs in the UB of all groups stained with H&E. Intact folded mucosa showing arrangement of urothelium separated from the well-developed musculosa by thin corium of connective tissue, is detected both in the control (a, b). Atrophy of the urothelium of the mucosa of the UB of CAST group (c, d) compared with apparently normal mucosa of the CAST + TRT group which shows increased thickness of the urothelium (e, f). The widened corium of CAST group displays increased number of connective tissue cells (arrowhead) separated by abundant intercellular matrix (asterisk) containing elastic fibers (arrow). (H & E, a, c, e ×200, scale bar 100 µm; b, d, f ×400, scale bar 50 µm)
Photomicrographs in the UB of all groups stained with H&E. UB of the control group shows normal architecture of the urothelium formed of top umbrella shaped cells (thick arrow), inverted flask shaped cells (arrow), underlying intermediate cells and basal cells. The nuclei of the urothelium are rounded central and vesicular, and sometimes binucleated (arrow head) (a). The UB of CAST group shows thinner urothelium with atrophy of the whole layers (b) compared with the CAST + TRT group which has a relatively thicker urothelium (c). The cytoplasm of urothelium of CAST appears deep acidophilic and containing dark nuclei with indistinct underlying basement membrane, unlike that of the control & CAST + TRT groups where the urothelium appeared vacuolated and lying on distinct basement membrane. (H & E, ×1000, scale bar 10 µm)
On the other hand in the CAST group, marked atrophy and thinning of the urothelium with disruption of the basement membrane were observed (Fig. 1c, d). Many abnormal forms as well as encysted hyaline masses were detected in the urothelium (Fig. 3b, d). The urothelial cells had deep acidophilic cytoplasm and darkened nuclei (Fig. 2b). The underlying corium became very wide showing increased density of connective tissue cells and excessive deposition of the intercellular matrix containing occasional elastic fibers (Figs. 1d, 3a, b). The smooth muscle layer appeared thin and disrupted (Fig. 3a, c).
Photomicrographs in the UB of CAST group. It displays widened corium underlying the abnormally appearing atrophied urothelium which has occasionally abnormal epithelial patterns including acidophilic hyaline masses lying inside cystic spaces (a, b, d). The bundles of smooth muscle fibers appeared disrupted and separated by wide areas of connective tissue (c). (H & E, a ×200, scale bar 100 µm; b, c, d ×400, scale bar 50 µm)
Meanwhile, in UB of the CAST + TRT group, the histological changes were reversed by the effect of T in the form of restoration of the normal appearance and thickness of the urothelium. Its cytoplasm appeared empty vacuolated and separated from the underlying narrow CT corium by distinct basement membrane. The underlying CT corium was thinner in comparison with the CAST group (Fig. 1e, f, 2c), while the smooth muscle layer was thick and well-developed (Fig. 1e).
By Masson’s trichrome stain, there was few collagen fibers stained blue lying mainly in the corium and to less extent between the smooth muscle fibers in the UB of the control group (Fig. 4a, b). On the other hand, there was a marked increase of the collagen fibers deposition in both the corium and between the muscle fibers of the CAST group compared with the control group (Fig. 4c, d). However, in the CAST group treated by TRT, few fine collagen was deposited (Fig. 4e, f).
Photomicrographs in the UB of all groups stained with Masson’s trichrome. Marked increase of the collagen fibers deposited in the corium and in between the smooth muscle fibers in the UB of CAST rats (c, d), compared with either control (a, b) or the CAST + TRT group (e, f) where less abundant fine collagen fibers were detected. (Masson’s trichrome, a, b, c ×200, scale bar 100 µm; b, c, d ×400, scale bar 50 µm)
The expression of the Ck20 in the control group was mainly detected in the top umbrella shaped cells (Fig. 5a). Meanwhile, the urothelium of the CAST group did not show any Ck20 expression in almost all fields (Fig. 5b). On treating the CAST group with TRT, the expression of Ck20 was abnormally seen not restricted to the top superficial layer but it extended to involve the whole urothelium (Fig. 5c).
Photomicrographs in the UB of all groups immunostained with Ck20. The whole urothelium does not show any positive ck20 expression in the CAST group (b) unlike the CAST + TRT group (c) where the urothelium displays abnormally increased ck20 expression not restricted to the top umbrella layers, like the control (a), but extends to involve the whole thickness of the urothelium. (Ck20, ×1000, scale bar 10 µm)
Morphometric and statistical results
The urothelial height was significantly decreased in the CAST group in comparison with the control group (P < 0.001). However, there was a significant increase in the urothelial height of the CAST + TRT group compared to the CAST group (P < 0.05) (Fig. 6a).
Moreover, in the CAST group there was a significant decrease in the smooth muscle/collagen ratio in comparison with the control group (P < 0.001). This ratio significantly increased in the CAST + TRT group when compared with the CAST group (P < 0.001) (Fig. 6b).
Serum testosterone levels
A highly significant decrease in the serum concentration of T was observed 30 days after castration compared with the control group. On the other hand, in the CAST + TRT group the serum concentration of T was restored back to the normal control level 30 days from the start of TRT. Then significant increase in this concentration was observed at the end of the experiment compared with the control group (Table 1).
Discussion
Low T levels are associated with metabolic syndrome, erectile dysfunction as well as LUTS. Many of these symptoms are reversed by restoring T to physiological concentrations (Yassin et al. 2014). Although the exact relationship between T and LUTS is not yet well established, TRT could also improve LUTS and UB function associating T deficiency (Gallo et al. 2012). This improvement in men with SLOH is physiologically attributed to the increase in compliance and bladder capacity, as well as to a decrease in detrusor pressure to the maximum flow rate (Karazindiyanoglu and Cayan 2008; Koritsiadis et al. 2008; Haider et al. 2009).
In the present study, histological examination of the UB of CAST group revealed that there was a significant atrophy of both the urothelium and musculosa unlike the corium which was abnormally increased in thickness. The urothelium showed many abnormal forms including encysted hyaline masses. Additionally, there was a significant increase in the collagen both in the corium and in the smooth muscle layer.
In the CAST + TRT group, the urothelium preserved its normal thickness and shape. Also, there was a significant increase in the thickness the muscle layer compared with the CAST group. This was in contrary to the study conducted by (Gallo et al. 2012) who showed that there is no differences in the epithelial height after TRT. However, castration is previously reported to decrease the amount of smooth muscle and to increase the amount of collagen and connective tissue in the UB (Das et al. 2002; Magari et al. 2014). Studies have demonstrated noticeable decrease of the elastic fibers in the castrated animals (Gallo et al. 2012) which might be attributed to their replacement by collagenous fibers, as occurring in smooth muscle fibers, which is not only confined to the UB but could involve the corpus cavernosum of the penis (Shen et al. 2003). Other studies showed that the number of vessels as well as the quantity of muscle fibers and the urothelium thickness are increased by TRT in castrated rat UB (Madeiro et al. 2002). Sulfated glycosaminoglycans and hyaluronic acid content are decreased in the UB of CAST rats, their quantity and quality could be altered by TRT (de Deus et al. 2003; Gallo et al. 2012). This might explain the widening of the UB corium in the CAST group in the present investigation.
Recent studies showed that TRT in SLOH men with urogenital dysfunction may have a positive role by increasing quantity of smooth muscle fibers that leads to improvement of bladder capacity and function (Cayan et al. 2008; Tek et al. 2010; Magari et al. 2014). Androgen deficiency induces bladder fibrosis and decreases the bladder maximal volume and compliance which could be effectively prevented by TRT (Gallo et al. 2012; Zhang et al. 2012; de Barros et al. 2013). The expression of TGF-beta, a common profibrotic factor, is upregulated by androgen deficiency, which induces procollagen production and consequently UB fibrosis and dysfunction (Matsuoka and Tsukamoto 1990; Wen et al. 2010; Lang et al. 2011; Samarakoon et al. 2012).
Many molecular- and neurogenic-based mechanisms for the improvement of LUTS and bladder function by TRT have been proposed. At the molecular level, UB contraction is essentially dependant on the adenosine triphosphate (ATP) concentration in the bladder wall, as well as mitochondrial oxidative enzymes activity for energy production (Juan et al. 2007). Castration decreases noticeably the activities of the mitochondria specific enzyme, citrate synthase of the UB muscle and mucosa (Juan et al. 2007) which could be corrected by TRT. Nitric oxide (NO) is also one of the mediators of dilatation of the bladder neck and urethra (McVary 2006) by its smooth muscle relaxing effect in the urogenital system (Pradidarcheep 2008). It was suggested that TRT could improve the urinary symptoms by increasing NO production by modulatory effect on NO synthase (Chamness et al. 1995) or phosphodiesterase which might be linked to TRT effect on the penile tissues in hypogonadal men (Kalinchenko et al. 2008). In addition, as increased rho-kinase activity coincides with the development of LUTS in elderly men, therefore TRT-induced inhibition of rho-kinase is thought to decrease smooth muscle cell proliferation and to decrease adrenergic contractions (Rees et al. 2003).
Moreover, T can affect UB detrusor muscle contraction by neuronal regulation (Keast 1999; Hall et al. 2002; Purves-Tyson et al. 2007). Androgen deprivation apparently down-regulated the activity and expression of enzymes controlling the cholinergic as well as non-cholinergic nerve functions (Filippi et al. 2007; Juan et al. 2007). Morita et al. (1992) stated that α 1-adrenergic and muscarinic cholinergic receptor densities considerably decreased after castration, and TRT restored them back to normal conditions level (Takyu 1993) by affecting the relative expression of their subtypes (Longhurst 2003).
In the present investigation, the expression of Ck20 was increased in the UB of CAST + TRT compared with the CAST group. There was atypical urothelium regarding both the morphology and expression of the Ck20 which was detected in the whole epithelial thickness and not restricted to the superficial umbrella shaped cells. This abnormal distribution of expression might be attributed to administration of high dosage of TRT. Ck20 expression is typically restricted to superficial umbrella cells and occasionally intermediate cells in normal conditions, even in the presence of severe UB inflammation (Harnden et al. 1996). In almost all cases of dysplasia, complete loss of this restriction was seen at least focally with positive expression in all layers of the urothelium (McKenney et al. 2001). The abnormal expression of Ck20 is considered a reliable marker of urothelial dysplasia in the UB, therefore it can be a useful tool to distinguish dysplastic changes from reactive atypia (Mallofre et al. 2003).
At cellular level, T exerts its action by binding to AR. AR have been widely identified in UB including its urothelium, smooth muscle cells and neurons (Pradidarcheep 2008). Activation of AR by androgen may stimulate stromal precursor cells to differentiate into smooth muscle cells (Traish et al. 2007). This could be attributed to alteration of chromatin remodeling as well as DNA transcription (Rahman et al. 2004). Studies has demonstrated that the length of polymorphism Cytosine-Adenine-Guanine (CAG) trinucleotide repeats present in AR gene might co-regulate the effectiveness of TRT (Giagulli et al. 2011a; Tirabassi et al. 2014). Shorter length AR gene CAG repeat number is associated with better recovery of sexual function after TRT in hypogonadism (Tirabassi et al. 2014). In addition, AR showed stimulatory behavoiur, within many hormone-related tumors such as in the UB, as well as in kidney and lung metastases (Chang et al. 2014). These stimulatory effects of androgens on tumorigenesis via the AR pathway may offer great potential for androgen deprivation as a therapeutic or chemopreventive option in patients with bladder malignancy (Li et al. 2012).
This study clearly showed that high dose TRT has a preferential positive effect on the UB smooth muscle cells rather than on its urothelium, however further research is recommended to adjust the proper dose of TRT to be a gold standard regimen in alleviating hypogonadism-induced urinary manifestations without causing urothelial atypic changes.
References
Bancroft JD, Layton C (2013) The Hematoxylin and eosin. In: Suvarna SK, Layton C and Bancroft JD (eds), Theory & Practice of histological techniques. 7th ed., Churchill Livingstone of El Sevier, Philadelphia, Ch.10 and 11, pp 173–214
Callies F, Kollenkirchen U, von zur Muhlen C et al (2003) Testosterone undecanoate: a useful tool for testosterone administration in rats. Exp Clin Endocrinol Diabetes 111:203–208
Cayan F, Tek M, Balli E et al (2008) The effect of testosterone alone and testosterone + estradiol therapy on bladder functions and smooth muscle/collagen content in surgically menopause induced rats. Maturitas 60:248–252
Celayir S, Ilce Z, Dervisoglu S (2002) The sex hormone receptors in the bladder in childhood - I: preliminary report in male subjects. Eur J Pediatr Surg 12:312–317
Chamness SL, Ricker DD, Crone JK et al (1995) The effect of androgen on nitric oxide synthase in the male reproductive tract of the rat. Fertil Steril 63:1101–1107
Chang C, Lee SO, Yeh S et al (2014) Androgen receptor (AR) differential roles in hormone-related tumors including prostate, bladder, kidney, lung, breast and liver. Oncogene 33:3225–3234
Corona G, Rastrelli G, Forti G et al (2011) Update in testosterone therapy for men. J Sex Med 8: 639-654; quiz 655
Das AK, Leggett RE, Whitbeck C et al (2002) Effect of doxazosin on rat urinary bladder function after partial outlet obstruction. Neurourol Urodyn 21:160–166
de Barros CA, Lorenzetti F, Ortiz V et al (2013) Testosterone supplementation’s effects on age-related bladder remodeling-experimental study in rats. Aging Male 16:102–107
de Deus JM, Girao MJ, Sartori MG et al (2003) Glycosaminoglycan profile in bladder and urethra of castrated rats treated with estrogen, progestogen, and raloxifene. Am J Obstet Gynecol 189:1654–1659
Filippi S, Morelli A, Sandner P et al (2007) Characterization and functional role of androgen-dependent PDE5 activity in the bladder. Endocrinology 148:1019–1029
Gallo CB, Miranda AF, Felix-Patricio B et al (2012) Effects of castration and hormone replacement in the urinary bladder of rats: structural, ultrastructural, and biochemical analysis. J Androl 33:684–690
Giagulli VA, Triggiani V, Carbone MD et al (2011a) The role of long-acting parenteral testosterone undecanoate compound in the induction of secondary sexual characteristics in males with hypogonadotropic hypogonadism. J Sex Med 8:3471–3478
Giagulli VA, Triggiani V, Corona G et al (2011b) Evidence-based medicine update on testosterone replacement therapy (TRT) in male hypogonadism: focus on new formulations. Curr Pharm Des 17:1500–1511
Haider A, Gooren LJ, Padungtod P et al (2009) Concurrent improvement of the metabolic syndrome and lower urinary tract symptoms upon normalisation of plasma testosterone levels in hypogonadal elderly men. Andrologia 41:7–13
Hall R, Andrews PLR, Hoyle CHV (2002) Effects of testosterone on neuromuscular transmission in rat isolated urinary bladder. Eur J Pharmacol 449:301–309
Harnden P, Eardley I, Joyce AD et al (1996) Cytokeratin 20 as an objective marker of urothelial dysplasia. Br J Urol 78:870–875
Jockenhovel F, Minnemann T, Schubert M et al (2009) Comparison of long-acting testosterone undecanoate formulation versus testosterone enanthate on sexual function and mood in hypogonadal men. Eur J Endocrinol 160:815–819
Juan Y-S, Onal B, Broadaway S et al (2007) Effect of castration on male rabbit lower urinary tract tissue enzymes. Mol Cell Biochem 301:227–233
Kalinchenko S, Vishnevskiy EL, Koval AN et al (2008) Beneficial effects of testosterone administration on symptoms of the lower urinary tract in men with late-onset hypogonadism: a pilot study. Aging Male 11:57–61
Karazindiyanoglu S, Cayan S (2008) The effect of testosterone therapy on lower urinary tract symptoms/bladder and sexual functions in men with symptomatic late-onset hypogonadism. Aging Male 11:146–149
Keast JR (1999) The autonomic nerve supply of male sex organs–an important target of circulating androgens. Behav Brain Res 105:81–92
Koritsiadis G, Stravodimos K, Mitropoulos D et al (2008) Androgens and bladder outlet obstruction: a correlation with pressure-flow variables in a preliminary study. BJU Int 101:1542–1546
Kunju LP, Lee CT, Montie J et al (2005) Utility of cytokeratin 20 and Ki-67 as markers of urothelial dysplasia. Pathol Int 55:248–254
Lang Q, Liu Q, Xu N et al (2011) The antifibrotic effects of TGF-β1 siRNA on hepatic fibrosis in rats. Biochem Biophys Res Commun 409:448–453
Li Y, Izumi K, Miyamoto H (2012) The role of the androgen receptor in the development and progression of bladder cancer. Jpn J Clin Oncol 42:569–577
Litman HJ, Bhasin S, O’Leary MP et al (2007) An investigation of the relationship between sex-steroid levels and urological symptoms: results from the Boston Area Community Health survey. BJU Int 100:321–326
Longhurst PA (2003) Functional development of the rat urinary bladder after pre- or postpubertal castration. Pharmacology 67:72–82
Madeiro A, Girao M, Sartori M et al (2002) Effects of the association of androgen/estrogen on the bladder and urethra of castrated rats. Clin Exp Obstet Gynecol 29:117–120
Magari T, Shibata Y, Arai S et al (2014) Time-dependent effects of castration on the bladder function and histological changes in the bladder and blood vessels. Asian J Androl 16:457–460
Mallofre C, Castillo M, Morente V et al (2003) Immunohistochemical expression of CK20, p53, and Ki-67 as objective markers of urothelial dysplasia. Mod pathol 16:187–191
Matsuoka M, Tsukamoto H (1990) Stimulation of hepatic lipocyte collagen production by Kupffer cell-derived transforming growth factor beta: implication for a pathogenetic role in alcoholic liver fibrogenesis. Hepatology 11:599–605
McKenney JK, Desai S, Cohen C et al (2001) Discriminatory immunohistochemical staining of urothelial carcinoma in situ and non-neoplastic urothelium: an analysis of cytokeratin 20, p53, and CD44 antigens. Am J Surg Pathol 25:1074–1078
McVary K (2006) Lower urinary tract symptoms and sexual dysfunction: epidemiology and pathophysiology. BJU Int 97 Suppl 2: 23–28; discussion 44–45
Moll R (1991) Molecular diversity of cytokeratins: significance for cell and tumor differentiation. Acta Histochemica Supplementband 41:117–127
Morita T, Tsuchiya N, Tsujii T et al (1992) Changes of autonomic receptors following castration and estrogen administration in the male rabbit urethral smooth muscle. Tohoku J Exp Med 166:403–405
Nahata A, Dixit VK (2012) Ameliorative effects of stinging nettle (Urtica dioica) on testosterone-induced prostatic hyperplasia in rats. Andrologia 44(Suppl 1):396–409
Pradidarcheep W (2008) Lower urinary tract symptoms and its potential relation with late-onset hypogonadism. Aging Male 11:51–55
Purves-Tyson TD, Arshi MS, Handelsman DJ et al (2007) Androgen and estrogen receptor-mediated mechanisms of testosterone action in male rat pelvic autonomic ganglia. Neuroscience 148:92–9104
Rahman M, Miyamoto H, Chang C (2004) Androgen receptor coregulators in prostate cancer: mechanisms and clinical implications. Clin Cancer Res 10:2208–2219
Rees RW, Foxwell NA, Ralph DJ et al (2003) Y-27632, a Rho-kinase inhibitor, inhibits proliferation and adrenergic contraction of prostatic smooth muscle cells. J Urol 170:2517–2522
Salmi S, Santti R, Gustafsson JA et al (2001) Co-localization of androgen receptor with estrogen receptor beta in the lower urinary tract of the male rat. J Urol 166:674–677
Samarakoon R, Overstreet JM, Higgins SP et al (2012) TGF-β1 → SMAD/p53/USF2 → PAI-1 transcriptional axis in ureteral obstruction-induced renal fibrosis. Cell Tissue Res 347:117–128
Shen Z-J, Zhou X-L, Lu Y-L et al (2003) Effect of androgen deprivation on penile ultrastructure. Asian J Androl 5:33–36
Takyu S (1993) Effects of testosterone on the autonomic receptor-mediated function in lower urinary tract from male rabbits. Nihon Hinyokika Gakkai Zasshi 84:330–338
Tek M, Balli E, Cimen B et al (2010) The effect of testosterone replacement therapy on bladder functions and histology in orchiectomized mature male rats. Urology 75:886–890
Tirabassi G, Delli Muti N, Corona G et al (2014) Androgen receptor gene CAG repeat polymorphism independently influences recovery of male sexual function after testosterone replacement therapy in postsurgical hypogonadotropic hypogonadism. J Sex Med 11:1302–1308
Traish AM, Goldstein I, Kim NN (2007) Testosterone and erectile function: from basic research to a new clinical paradigm for managing men with androgen insufficiency and erectile dysfunction. Eur Urol 52:54–70
Wen J, Jiang X, Dai Y et al (2010) Increased adenosine contributes to penile fibrosis, a dangerous feature of priapism, via A2B adenosine receptor signaling. FASEB J 24:740–749
Yassin DJ, Doros G, Hammerer PG et al (2014) Long-term testosterone treatment in elderly men with hypogonadism and erectile dysfunction reduces obesity parameters and improves metabolic syndrome and health-related quality of life. J Sex Med 11:1567–1576
Zhang Y, Chen J, Hu L et al (2012) Androgen deprivation induces bladder histological abnormalities and dysfunction via TGF-beta in orchiectomized mature rats. Tohoku J Exp Med 226:121–128
Acknowledgments
The authors have not made any acknowledgments.
Conflict of interest
There is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abdel-Hamid, A.A.M., Ali, E.M.T. Effect of testosterone therapy on the urinary bladder in experimental hypogonadism of rats. J Mol Hist 46, 263–272 (2015). https://doi.org/10.1007/s10735-015-9617-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-015-9617-4