Abstract
Arbovirus diseases are caused by viruses transmitted by arthropods, mainly by hematophagous mosquitoes. Dengue is an important arbovirus caused by the dengue virus. This study analyzed the epidemiological profile and spatial distribution of dengue cases in the municipality of Araguaína, Tocantins, between 2011 and 2020. This was an ecological study of time series and trends in dengue cases and spatial analysis of the incidence of cases by neighborhood in the Araguaína municipality. It was found that the prevalence of dengue was generally predominant in the economically active population (20–39 years) and in pardos. Dengue cases showed a stationary trend with a high incidence in the Araguaína municipality. Therefore, to improve the efficiency of these methods, it is necessary to apply a unique health approach to the development of public policies that include interdisciplinary action to combat dengue.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Arboviruses represent a global health concern. They are diseases caused by viruses transmitted by arthropods, and are transmitted mainly by hematophagous mosquitoes. The main arboviral families are Bunyaviridae, Togaviridae, Reoviridae, Flaviviridae, and Rhabdoviridae (Avelino-Silva & Ramos, 2017). This group of viruses has a high capacity to adapt to vertebrate and invertebrate hosts, and is easily disseminated by the population (Lopes et al., 2014).
Dengue, an important arbovirus, is caused by the dengue virus (DENV), which can be divided into four serotypes: DENV-1, DENV-2, DENV-3, and DENV-4 (Donalisio et al., 2017). Transmission occurs through Culicidae mosquitoes belonging to the Aedes genus. Currently, Aedes aegypti is mainly responsible for the transmission of dengue strains in humans that circulate in domestic and peridomestic environments; however, other species of Aedes can act as secondary vectors. The most common clinical manifestations are sudden fever, arthralgia, myalgia, eye pain, maculopapular rashes, and leukopenia. In more severe cases, bleeding may occur, requiring hospitalization (Bezerra et al., 2021).
The factors that influence the spread of the disease are important. The proliferation of the vector is associated with changes in air temperature and rainfall levels, which favor the formation of breeding sites (Chen & Vasilakis, 2011). In addition, unsanitary conditions, inadequate water storage, and unfavorable economic conditions have facilitated the development of breeding sites (). It is worth noting that disorderly population growth and population migration between regions or even countries also contributes to this increase, given that an individual carrying an undetected virus can contaminate other non-endemic areas (Carvalho et al., 2017).
Globally, dengue is present in more than 110 countries with an estimated 100 million cases every year. In the Americas, Brazil accounts for approximately 70% of dengue cases each year, with significant transmission of the disease (Lima-Camara, 2016).
During the wetter and warmer periods, Brazil registered several epidemic cycles in several regions. In the State of Tocantins, dengue is considered an endemic disease because of its insertion into the Legal Amazon and characteristics that favor the development of the vector (Lima Neto et al., 2016).
Interdisciplinary and transdisciplinary approaches are essential, because social, economic, climatic, and environmental changes influence the transmission of diseases in different areas of the world, presenting an interrelationship between animals, humans, and the environment. Therefore, what stands out is the approach proposed by one health in the control of various diseases offering a more comprehensive view of the various public health issues associated with the different factors that may influence public and environmental health (Limongi & Oliveira, 2020; Valadares et al., 2013); Zinsstag et al., 2011).
Given this scenario, this study analyzed the spatial and temporal distribution of reported cases of dengue virus infection in the Araguaína municipality, in the State of Tocantins, from 2011 to 2020.
Materials and methods
This was an analytical study of the ecological time series and trends of confirmed dengue cases in the municipality of Araguaína, State of Tocantins, Brazil, from 2011 to 2020.
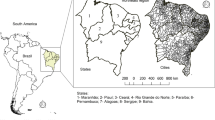
The Araguaína municipality is located in the northern region of the State of Tocantins, Brazil, and has an area of 4000.42 km2 with an estimated population of 160.000 people (IBGE, 2023).
The Araguaína municipality is located in the north of the state of Tocantins, a region belonging to the Legal Amazon. It is characterized by being an important economic reference for several surrounding municipalities, with a growing industrial and commercial center (IBGE, 2023).
It has a well-structured public health network with all levels of healthcare and is a sophisticated university hospital specializing in tropical medicine (HDT/UFNT). It is a reference for the regional health center in Central North Araguaia, supporting several municipalities in different states.
Araguaína has a tropical climate and is an economic reference in the region, with neighborhoods in a situation of social vulnerability, being hyperendemic for arboviruses and several other neglected diseases.
Trend analysis was performed for the municipality of Araguaína and other regions of Brazil using the Prais–Winsten model, which considers the rates as the dependent variable (Y) and the year studied as the independent variable (X). This model is suitable for trend analysis because it corrects the temporal autocorrelation of residues based on the ecological assumption that incidences can be influenced by each other in the years of the time series (Antunes & Cardoso, 2015).
The rates for the time series were smoothed using a third-order moving average. The analysis of the scatterplots of the incidences and autocorrelation of the residues allowed us to identify the behavior of the trend: stable (p > 0.05), decreasing (if p < 0.05 and regression coefficient (β1) was negative), and increasing (if p < 0.05 and regression coefficient (β1) was positive). The regression coefficient of the Prais-Winsten model and the annual variation in the incidences in the period (in %) were estimated using the formula (− 1 + 10-^b) × 100, since the regression uses the logarithm of the rates (10^b) (Antunes & Cardoso, 2015).
For trend analysis, annual dengue incidence coefficients were calculated, where dengue incidence = number of cases/population × 100,000.
To calculate the average prevalence for the period, the total average was extracted and the prevalence rates for dengue were calculated.
Variables
The study was conducted by punctual identification according to the geolocated addresses of confirmed cases of dengue in the Araguaína municipality from 2011 to 2020. In this study, the building infestation index (BII) of the municipality, which measures the relationship between the number of properties where Ae. aegypti larvae were found (Ae. Aegypti) and the number of properties surveyed, varied from 0 to 1% (low-risk), 1–3.99% (alert), and > 3.99% (high-risk). The analyzed variables were arranged according to the average of the indices during the study period.
The geolocation of point data was conducted according to the address of each notified case superimposed on the BII of each neighborhood in the year with the highest incidence of dengue in the municipality (2019) for the period studied; we analyzed the annual total and by four month intervals.
All information on dengue arboviruses and BII results was obtained from the DATASUS, state, and municipal health departments.
Mapping
The addresses of dengue cases were geocoded by an application programming interface (API) of Google Maps (Google®), based on the cartographic map of streets in the Araguaína municipality. Lower geocoding scores were topologically adjusted to ensure the accuracy of georeferencing. Locations not identified by the application were collected in the field at the locations of occurrence. The point features were plotted in a Geographic Information System (GIS) ArcGIS 10.2.2 (ESRI, Image) and Quantum GIS (QGIS) 0.16.3.
Statistical analysis
Cluster analysis was used to detect significant concentrations of absolute cases of dengue and infestation by Ae. aegypti as measured by the BII within a 95% confidence interval (CI). Clusters were calculated using the local Moran’s I analysis, which identifies features with high or low spatial cluster values. This pattern can be expressed using clustered, dispersed, or random features that represent measurable spatial aggregation units. To identify the pattern of distribution and presence of punctual concentrations of cases locally in the municipality, the kernel estimator was represented by the statistical method of estimating density curves.
Stata ® Program version 14.0 was used. QGIS software was used for spatial analysis and choropleth mapping (Fig. 1).
The municipality of Araguaína, State of Tocantins, Brazil. Source: Own elaboration based on IBGE data (2023)
Results
The disease had similar characteristics to those described in several other regions of Brazil and the world, it was more prevalent in females in the economically active age group, the predominant color in the region was brown, and three deaths were confirmed during the study period, two in 2013 and one in 2019, which in turn was characterized by a major epidemic in the municipality (Table 1).
Regarding the trend of cases, Araguaína showed a stationarity trend in its series, a fact that concerns the health surveillance services, as the municipality is in a high-risk incidence region for the disease, with an average annual rate of 457 cases per 100,000 inhabitants, and in some years it has reached incidence values above 900 cases, as was the case in 2012 and 2019 (Table 2).
When referring to the spatial distribution and concentration of the BII, it is worth noting the presence of low rates in the southwest of the municipality, characterized by neighborhoods of high economic status, areas with high rates in the northwest, and regions with high levels of social vulnerability (Fig. 2C).
Although the municipality has a high prevalence in a large part of the territory, some neighborhoods are considered risk areas with hot spots (areas with a high density of cases), mainly in peripheral regions, which are conducive to the spread of Ae. aegypti and/or due to social constraints (Figs. 2, 3).
It is worth noting that despite the higher level of prevalence clusters located in peripheral regions, the vast majority of neighborhoods had a coefficient greater than 300 cases per 100,000 inhabitants, an index considered to be at high-risk for dengue epidemics, some of which had values close to 2000 cases per 100,000 inhabitants, further demonstrating the level of urgency for this problem (OPAS, 2019) (Fig. 4).
Point analysis of absolute dengue cases, according to the level of building infestation (BII) analyzed in 2019 and by quarters (1°–2°–3°–4°). Source: Own elaboration based on IBGE data (2023)
Discussion
The Araguaína municipality has experienced the three largest dengue epidemics recorded in the last two decades, with the greatest impact in 2012, followed by 2017 and 2019 (Moraes et al., 2019). The most important factor may be a reflection of a multifactorial set, which has the potential to be related to the transmission of different serotypes in the same area. In addition, because fluctuations in rainfall and air temperature interfere with vector development, the large number of cases in 2012 may be linked, in part, to the La Niña episode that caused cooling in the Pacific; consequently, areas that are influenced by the Amazon rainforest, which is the case in the Araguaína municipality, experience more flooding than normal (Chen & Vasilakis, 2011).
In several studies, females showed a higher prevalence, which is due to the fact that women generally spend more time at home, a place of great potential for vector propagation, and because of the greater demand for health services compared to men (Andrioli et al., 2020; Cardoso et al., 2012).
Regarding age, a significant discrepancy in the number of cases present in the age group between 20 and 39 years was noticeable in relation to other age groups in all years studied. This can be explained by the fact that the designated age group corresponds to a group with more individuals belonging to the economically active population. This leads to a greater displacement of these people from performing their work activities, which increases the probability of transmission of the dengue virus (Gomes et al., 2023).
Several factors contribute to the high rates of infection in Brazil, including climatic, environmental, and socioeconomic conditions. There was a higher incidence of infections during periods of higher temperatures as there was an acceleration in the vector's reproductive cycle. In contrast, colder climates showed a reduced reproductive cycle. The climate, precarious socioeconomic aspects, such as lack of basic sanitation, unplanned urban expansion, increase in population density, and inadequate garbage collection, are factors that facilitate the formation of breeding sites and dissemination of the mosquito Ae. aegypti, in addition to compromising vector control methods (Brazilian, 2022).
Araguaína, the second most populous municipality in the State of Tocantins, is a border area with other states and is interconnected by federal highways that connect the Amazon region to the Midwest and Northeast regions. Moreover, the city is also a reference, both as an economic center and a health center for surrounding municipalities, which are important constraints for the dissemination of vectors and/or diseases imported into the municipality (Handschumacher et al., 2022).
Thus, the implementation of public policies for vector control is effective. Home visits to search for breeding sites, the use of larvicides, health education, and other actions are effective measures when conducted in a prolonged and standardized manner and require the joint participation of community health agents, Combat Endemic Diseases (ACE), and the population ().
However, in addition to reduced resources, these strategies do not prevent long-term transmission and do not address the particularities of each territory, demonstrating effectiveness by not showing a tendency to increase cases, but by remaining at a stationary level even at high rates (Zara et al., 2016).
Several factors contribute to the endemicity of dengue in the city of Araguaína, with an emphasis on socioeconomic characteristics. Thus, it is worth noting that the municipality is an economic hub, where there is a large circulation of people and goods, which generates large amounts of materials, predisposing the maintenance and increase in breeding sites that maintain the transmission cycle (Adnan et al., 2021).
It is important to note the years 2013, 2014, 2018, and 2020, which showed a significant reduction in incidence compared to other periods. This fact is consistent with several studies that have shown a drastic reduction in cases in different parts of the world, usually up to two years after the great dengue and Zika epidemics, due to the existence of cross-immunity conferred by infection with one of the DENV serotypes against other serotypes or by previous Zika virus infection (Ribeiro et al., 2018).
Geographical characteristics of the city, such as the humid and hot climate and the areas of vegetation found in several places, are favorable habitats and reproductive conditions for the vectors Ae. aegypti. The presence of a lake close to the high prevalence points suggests its role as a natural breeding ground. Studies have shown that neighborhoods close to water reservoirs, such as lakes, streams and puddles, were the places most affected by dengue (Costa et al., 2011).
It is worth highlighting the triad of human, animal, and environmental health, as dengue transmission occurs in both urban and wild environments and requires mechanical, biological, and chemical control. It is worth highlighting a method of biological control of the vector, with the use of predatory species to parasitize or destroy mosquitoes at several stages, among which the most promising is the use of the Wolbachia bacterium, which induces two effects: reduction of replication of DENV and manipulation of reproduction in a way that increases the spread of the symbiont in the vector (Dutra et al., 2021).
The BII presents important data to relate to the prediction and incidence of these cases, since the larval indices can be closely related to the manifestation of dengue epidemics and other arboviruses (Ribeiro et al., 2021).
It is important to highlight that some studies suggest the need to invest in the development of complementary surveillance methodologies that allow reliable measurement of the risk of transmission in the territory owing to the low performance of the rapid survey of indices for Ae. aegypti (LIRAa) to predict epidemics. Highlighting the multicausal component in the temporal chain for the occurrence of outbreaks modified by socioeconomic, demographic, and environmental factors, host susceptibility, and etiology of the circulating agent (Ribeiro et al., 2021).
The Araguaína municipality had a high concentration of absolute cases with the presence of clusters in the central region and a positive spatial correlation for cases of the disease in neighborhoods with a high population density and high flow of people.
When analyzing the prevalence, cases were concentrated in peripheral areas with characteristics of greater social vulnerability Building infestation rates with high-risk characteristics and a positive spatial correlation between areas of high social vulnerability and low-risk levels with a similar correlation in high-income neighborhoods.
Despite having a health system with well-established levels of complexity, recurrent epidemics and outbreaks of communicable diseases are part of everyday life in the municipality.
This reality may be related to climatic conditions, characteristics of precarious basic sanitation in areas with a high degree of social vulnerability, and low investment in research aimed at preventing outbreaks in the region.
Conclusion
The implementation of basic care for the control of dengue should not be disregarded, as preventive measures are essential for better control of the disease. Urban areas with high social vulnerability should be assessed using the process of reverse equity, since the state's inefficiency in providing minimum health conditions and quality of life associated with health issues makes this population extremely susceptible to public health problems.
The municipality of Araguaína has several factors that contribute to the high risk of dengue. In addition, the recommended strategies for disease control are adequate to a certain extent, but are not effective in the long-term. This is because of the scarcity of available resources and because they do not consider the geographic and environmental characteristics of each location.
Therefore, strengthening actions with a single health focus is extremely important for the development of more robust, multi-sectoral, and multi-professional public policies to combat dengue. Thus, it strengthens the disease prevention, control, and mitigation measures already implemented in the region.
The implementation of actions and measures to effectively reduce morbidity and mortality from dengue is a great challenge in several countries worldwide. The recent approval by health bodies in endemic countries, such as Brazil and Indonesia, for the use of the recently launched Qdenga vaccine from the Japanese laboratory Takeda represents an important advancement in the process of attempting to reduce disease rates (Brazil, 2023; Mallapaty, 2022; Rivera et al., 2022).
References
Adnan, R. A., Ramli, M. F., Othman, H. F., Asha’ri, Z. H. S., Ismail, S. N., & Samsudin, S. (2021). The Impact of Sociological and Environmental Factors for Dengue Infection in Kuala Lumpur. Malaysia. Minutes Tropica, 216, 105834. https://doi.org/10.1016/j.actatropica.2021.105834. ISSN 0001-706X.
Andrioli, D. C., Busato, M. A., & Lutinski, J. A. (2020). Characteristics of the dengue epidemic in Pinhalzinho, Santa Catarina, 2015–2016. Epidemiology and Health Services. https://doi.org/10.5123/S1679-49742020000400007. Accessed 2 August 2022
Antunes, J. L. F., & Cardoso, M. R. A. (2015). Use of time series analysis in epidemiological studies. Epidemiology and Health Services, 24(3), 565–576. https://doi.org/10.5123/S1679-49742015000300024
Avelino-Silva, V. L., & Ramos, J. F. (2017). Arboviruses and public policies in Brazil/Arboviruses and public policies in Brazil. Health Sciences Magazine, 7(3), 1–2. https://doi.org/10.21876/rcsfmit.v7i3.675
Bezerra, J. M. T., de Sousa, S. C., Tauil, P. L., Carneiro, M., & Barbosa, D. S. (2021). Entry of dengue virus serotypes and their geographic distribution in Brazilian federative units: a systematic review. Brazilian Journal of Epidemiology. https://doi.org/10.1590/1980-549720210020. Retrieved August 2, 2022, from https://www.scielo.br/j/rbepid/a/z96xfD3n9nhNTqRqqPfwSYR/?lang=en#
Brazil. (2023). Anvisa approves new vaccine for dengue. Retrieved March 10, 2023, from https://www.gov.br/anvisa/pt-br/assuntos/noticias-anvisa/2023/anvisa-aprova-nova-vacina-para-a-dengue
Brazilian Institute of Geography and Statistics. (2022). Gender Statistics. Retrieved August 3, 2022, from https://www.ibge.gov.br/apps/snig/v1/?loc=0&cat=-1,1,2,-2,99,100,101,60,8,128&ind=4726
Cardoso, F. D. P., Batista, H. L., Araújo, B. M., & Nunes, R. M. (2012). Observations on the epidemiology of dengue in Araguaína, Tocantins. Health Sciences Review. Retrieved August 2, 2022, from http://periodicoseletronicos.ufma.br/index.php/rcisaude/article/view/1276
Carvalho, S., de Magalhães, M. A. F. M., & de Medronho R. A. (2017). Analysis of the spatial distribution of dengue cases in the city of Rio de Janeiro, 2011 and 2012. The Annual Review of Public Health, 510, 79. https://doi.org/10.11606/s1518-8787.2017051006239. Retrieved August 2, 2022, from https://www.revistas.usp.br/rsp/article/view/138329
Chen, R., & Vasilakis, N. (2011). Dengue—Quo tu et quo vadis? Virus, 3(9), 1562–1608. https://doi.org/10.3390/v3091562. Accessed August 2, 2022.
da Costa, A. G., dos Santos, J. D., da Conceição, J. K. T., Alecrim, P. H., Casseb, A. A., Batista, W. C., & Heckmann, M. I. O. (2011). Dengue: epidemiological aspects and the first outbreak in the Middle Solimões region, Coari, State of Amazonas, from 2008 to 2009. Journal of the Brazilian Society of Tropical Medicine, 44(4), 471–474. https://doi.org/10.1590/S0037-86822011000400014. Accessed 2 August 2022.
de Moraes, B. C., de Souza, E. B., Sodré, G. R. C., Ferreira, D. B. S., & Ribeiro, J. B. M. (2019). Seasonality in dengue notifications in Amazonian capitals and the impacts of El Niño/La Niña. Public Health Notebooks. https://doi.org/10.1590/0102-311X00123417. Accessed 2 August 2022.
Donalisio, M. R., Freitas, A. R. R., Zuben, A. P. B. V. (2017). Arboviruses emerging in Brazil: challenges for clinical and implications for public health. Journal of Public Health, 51, 30. https://doi.org/10.1590/S1518-8787.2017051006889. Retrieved August 2, 2022, from https://www.scielo.br/j/rsp/a/Nym8DKdvfL8B3XzmWZB7hJH/?lang=en#
Dutra, H. L., Ford, S. A., Allen, S. L., Bordenstein, S. R., Chenoweth, S. F., Bordenstein, S. R., & McGraw, E. A. (2021). The impact of artificial selection for Wolbachia-mediated dengue virus blocking on phage WO. Plos Neglected Tropical Diseases, 15(7), e0009637. https://doi.org/10.1371/journal.pntd.0009637. Accessed 2 August 2022.
Gomes, H., de Jesus, A. G., & Quaresma, J. A. S. (2023). Identification of risk areas for arboviruses transmitted by Aedes aegypti in northern Brazil: A One Health analysis. One Health, 16, 100499. https://doi.org/10.1016/j.onehlt.2023.100499. PMID: 36844974; PMCID: PMC9945760.
Handschumacher, P., Fournet, F., & Ramalho, W. (2022). Space, territories and vector diseases: lessons from Southern countries to fight the threat of arboviruses in Brazil? Confis. https://doi.org/10.4000/confins.45060. Retrieved August 2, 2022, from http://journals.openedition.org/confins/45060
IBGE. (2023). Brazilian Institute of Geography and Statistics. Araguaína in: Cities and States. Retrieved January 6, 2023, from https://www.ibge.gov.br/cidades-e-estados/to/araguaina.html
Lima-Camara, T. N. (2016). Emerging arboviruses and public health challenges in Brazil. Journal of Public Health, 50, 36. https://doi.org/10.1590/S1518-8787.2016050006791. Accessed 2 August 2022.
Limongi, J. E., & de Oliveira, S. V. (2020). COVID-19 and the One Health approach: A systematic review. Vigil Toilet Debate, 8(3), 139–144. https://doi.org/10.22239/2317-269X.01610. Retrieved August 3, 2022, from https://www.redalyc.org/journal/5705/570566811016/html/
Lopes, N., Nozawa, C., & Linhares, R. E. C. (2014). General characteristics and epidemiology of emerging arboviruses in Brazil. Rev Pan-Amaz Cheers, 5(3): 55–64. https://doi.org/10.5123/s2176-62232014000300007. Retrieved August 2, 2022, from http://scielo.iec.gov.br/scielo.php?script=sci_arttext&pid=S2176-62232014000300055&lng=en
Mallapaty, S. (2022). Dengue Vaccine poised for roll-out but safety concerns linger. Nature (london), 611, 7936. https://doi.org/10.1038/d41586-022-03546-2
Neto, A. S. L., do Nascimento, O. J., de Sousa, G. D. S., & de Oliveira Lima, J. W. (2016). Dengue, zika and chikungunya—vector control challenges facing the occurrence of the three arboviruses—Part I. Revista Brasileira em Promocao da Saude, 29(3), 305–312. https://doi.org/10.5020/18061230.2016.p305. Retrieved August 2, 2022, from https://periodicos.unifor.br/RBPS/article/view/5782
OPAS. (2019). Organização Pan-Americana da Saúde. Documento técnico para a implementação de intervenções baseado em cenários operacionais genéricos para o controle do Aedes aegypti. OPAS.
Ribeiro, M. S., Ferreira, D. F., Azevedo, R. C., Santos, G. B. G. D., & Medronho, R. D. A. (2021). Larval indices of Aedes aegypti and dengue incidence: An ecological study in the State of Rio de Janeiro, Brazil. Public Health, 37(7), e00263320. https://doi.org/10.1590/0102-311X00263320. Accessed July 16, 2022. Epub 19 Jul 2021. ISSN 1678-4464.
Ribeiro, G. S., Kikuti, M., Tauro, L. B., Nascimento, L. C. J., Cardoso, C. W., Campos, G. S., Ko, A. I., Weaver, S. C., Reis, M. G., Kitron, U., Paploski, I. A., & Salvador Arboviral Research Group. (2018). Does immunity after Zika virus infection cross-protect against dengue? The Lancet Glob Health, 6(2), e140–e141. https://doi.org/10.1016/S2214-109X(17)30496-5. Retrieved August 2, 2022, from https://www.thelancet.com/journals/langlo/article/PIIS2214-109X(17)30496-5/fulltext
Rivera, L., Biswal, S., Sáez-Llorens, X., Reynales, H., López-Medina, E., Borja-Tabora, C., Bravo, L., Sirivichayakul, C., Kosalaraksa, P., Martinez Vargas, L., & Yu, D. (2022). Three-year efficacy and safety of Takeda’s dengue vaccine candidate (TAK-003). Clinical Infectious Diseases, 75, 107–117.
Santos, G. V. D., Arduino, M. D. B., & Serpa, L. L. N. (2022b). Aedes aegypti queenslandensis: First geographic occurrence in Brazil and epidemiological implications. Brazilian Journal of Entomology. https://doi.org/10.1590/1806-9665-RBENT-2021-0112
Santos, J. P. C. D., Albuquerque, H. G., Siqueira, A. S. P., Praça, H. L. F., Pereira, L. V., Tavares, A. D. M., Gusmão, E. V. V., Bruno, P. R. D. A., Barcellos, C., Carvalho, M. D. S., & Sabroza, P. C. (2022a). ARBOALVO: Territorial stratification to define areas of prompt response for surveillance and control of urban arboviruses in a timely manner. Public Health. https://doi.org/10.1590/0102-311X00110121. Accessed 2 August 2022a
Valadares, A. F., Rodrigues, C. F. J., & Peluzio, J. M. (2013). Impact of dengue in two main cities in the State of Tocantins: Infestation and environmental factor (2000 to 2010). Epidemiology and Health Services, 22(1), 59–66. https://doi.org/10.5123/S1679-49742013000100006. Cited 2022 Aug 02.
Zara, A. L. S. A., dos Santos, S. M., Fernandes-Oliveira, E. S., Carvalho, R. G., & Coelho, G. E. (2016). Aedes aegypti control strategies: A review. Epidemiology and Health Services, 25(2), 391–404. https://doi.org/10.5123/S1679-49742016000200017. Cited 2022 Aug 02.
Zinsstag, J., Schelling, E., Waltner-Toews, D., & Tanner, M. (2011). From "one medicine" to "one health" and systemic approaches to health and well-being. Preventive Veterinary Medicine, 101(3–4), 148–56. https://doi.org/10.1016/j.predicted.2010.07.003. Retrieved August 2, 2022, from https://pubmed.ncbi.nlm.nih.gov/20832879/
Acknowledgements
The authors would like to thank the scientific initiation program of the Federal University of Norte do Tocantins (PIBIC/UFNT). State University of Pará; and the health department of the municipality of Araguaína Tocantins, Brazil.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gomes, H., Kihara, P.M., Nunes, M.H.S. et al. Risk of Dengue and tendency map based on geographic localization of cases and vectorial infestation in the North of Brazil. GeoJournal 88, 5259–5269 (2023). https://doi.org/10.1007/s10708-023-10892-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10708-023-10892-9