Abstract
Nitrogen use efficiency is a key factor for the economic and environmental sustainability of farms. It comprises the ability for crops to recover applied fertilizer N in their aboveground biomass, called fertilizer N uptake efficiency (NupE). Sweet pearl millet [Pennisetum glaucum (L.) R.BR.] and sweet sorghum [Sorghum bicolor (L.) Moench] are C4 annual crops known for their capacity to produce high yield under N-limiting conditions, suggesting high NupE. A field study was conducted for 2 years on sandy loam soils in eastern Canada. The NupE was determined using a 15N-tracing approach. Comparisons were made for both species in regards to their response to (1) increasing mineral N rate (0–160 kg N ha−1), (2) mineral versus organic N (liquid swine and dairy cattle manures), and (3) single versus split N application. For mineral N treatments, NupE ranged from 54 to 82 %, which is greater than values generally reported for conventional crops such as corn. Moreover, NupE increased with N rates. These findings suggest than both species expressed N luxury consumption. The NupE was lower with organic than with mineral N, and a larger proportion of N remained in the soil, suggesting that N immobilization occurred. The presumed losses (unrecovered 15N) were <24 kg N ha−1 in all mineral and most organic N treatments. Splitting N application had little effect on NupE. Our results confirm that sweet pearl millet and sweet sorghum are highly efficient at recovering fertilizer N and, with adequate residue management, may represent a low environmental risk.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Sweet pearl millet [Pennisetum glaucum (L.) R.BR.] and sweet sorghum [Sorghum bicolor (L.) Moench] are annual C4 grasses that have been studied in eastern Canada for the last decade because of their potential to replace or complement grain corn (Zea Mays L.) for ethanol production. These species can be exploited similarly to sugarcane, by extracting the sugar-rich juice from their stalks and fermenting it into ethanol (Yu et al. 2012).
In previous experiments under the wet and cool conditions of eastern Canada, sweet pearl millet and sweet sorghum produced aboveground biomass up to 19 and 20 Mg DM ha−1, respectively (Dos Passos Bernardes et al. 2015; Thivierge et al. 2015). The mineral N rate (ammonium nitrate) that maximized ethanol yields for sweet pearl millet and sweet sorghum in eastern Canada varied from 78 to 91 kg N ha−1 (Leblanc et al. 2012; Thivierge et al. 2015). In comparison, grain corn, the current feedstock used for ethanol production in eastern Canada, yielded an average of 6.9 Mg DM ha−1 from 2008 to 2012 (FADQ 2015). Nitrogen rates (ammonium nitrate) required to maximize corn grain and ethanol yield in the same area varied from 128 to 285 kg N ha−1 (Liang et al. 1996; Gagnon and Ziadi 2010). Therefore, the high DM yields obtained from sweet pearl millet and sweet sorghum with relatively low N rates suggest that these species may have high fertilizer N uptake efficiency (NupE; proportion of applied fertilizer N recovered in the aboveground biomass at harvest).
Using 15N-labelled fertilizers, several authors in eastern Canada and the United States demonstrated that for corn, NupE (1) varied from 28 to 60 % (Reddy and Reddy 1993; Liang and MacKenzie 1994; Tran et al. 1997; Stevens et al. 2005; Nyiraneza et al. 2010), (2) decreased with increasing mineral N rate (Liang and MacKenzie 1994; Tran et al. 1997), and (3) was lower with animal manure than with mineral N (Carter et al. 2010). We are not aware of any study on sweet pearl millet and sweet sorghum using this technique.
The aims of the present study were (1) to determine NupE in sweet pearl millet and sweet sorghum, and (2) to compare the response of these species to (i) increasing rates of mineral N, (ii) mineral versus organic N sources (liquid swine and dairy manures), and (iii) to single versus split application of mineral N. Work was carried out at two sites under wet and cool conditions in eastern Canada.
Materials and methods
Sites and species description
Crops were grown at Sainte-Anne-de-Bellevue (Québec, Canada, 45°26′N, 73°56′W), located in the Mixedwood Plains (MWP) ecozone, in 2011 and 2012, and at Saint-Augustin-de-Desmaures (Québec, Canada, 46°44′N, 71°31′W), located in the Boreal Shield (BS) ecozone, in 2010 and 2011. A complete description of ecozones of Canada is found in the National ecological framework for Canada (ESWG 1995). The two ecozones were chosen because they provide the possibility to grow sweet pearl millet and sweet sorghum as bioenergy crops using their sweet juice, or as forage crops, under two contrasting temperature regimes. Mean daily temperatures from seeding to harvest were 21.5 °C in 2011 and 21.9 °C in 2012 at the MWP site, and 19.3 °C in 2010 and 18.6 °C in 2011 at the BS site. During the same period, cumulated rainfall were 359 mm in 2011 and 188 mm in 2012 at MWP, and 282 mm in 2010 and 390 mm in 2011 at BS. The corn heat units (CHU) cumulated from seeding to harvest, a climatic index calculated as per Brown and Bootsma (1993), were 2207 CHU in 2011 and 2250 CHU in 2012 at MWP, and 2095 CHU in 2010 and 1940 CHU in 2011 at BS. Soil types were a St. Bernard sandy loam (coarse-loamy, mixed, nonacid, calcareous, frigid Eutrochrept) at the MWP site, and a St. Antoine sandy loam (fine-loamy, mixed, acid, frigid Haplorthod) at the BS site. Additional characteristics of the experimental sites are presented in Thivierge et al. (2015). At both sites, sweet pearl millet hybrid CSSPM7 and sweet sorghum hybrid CSSH45 (AERC Inc., Delhi, Ontario, Canada) were compared.
Experimental set-up and crop management
A split-plot factorial design with four replicates was used at each site, with species as the main factor (main plots) and N fertilization treatments as the sub-factor (subplots). The complete description of the experimental design can be found in Thivierge et al. (2015).
Briefly, each subplot was 6.3 m2 in size at the MWP site, and 9.7 m2 at the BS site. Row spacing was 0.18 m in all cases. Seedbed preparation consisted of mouldboard ploughing in the fall and harrowing twice in the spring to prepare seedbed. The seeding rate was 10 kg ha−1 of pure live seeds. Seeding was performed at a depth of 2.5 cm using a Wintersteiger plot seeder (Wintersteiger, Salt Lake City, UT). The growing seasons were from June 10 (seeding) to Sept. 9 (harvesting) at the BS site in 2010 (91 days), from June 6 to Aug. 31 at the BS site in 2011 (86 days), from June 16 to Sept. 7 at the MWP site in 2011 (84 days), and from June 7 to Aug. 30 at the MWP site in 2012 (85 days).
Bentazone (3-isopropyl-1H-2.1.3-benzothiadiazin-4(3H)-one 2.2-dioxide) was applied at a rate of 1.08 kg active ingredient ha−1 between the three- and six-leaf stage for both species to suppress dicotyledonous weeds. At the BS site, glyphosate (N-(phosphonomethyl-glycine) was applied in spring 2010 (0.89 kg active ingredient ha−1) to control weeds. Hand weeding of the sweet sorghum plots was done at the 10-leaf stage at both sites, while it was not necessary in sweet pearl millet plots.
Fertilization treatments
The fertilization treatments included calcium ammonium nitrate (27-0-0) at rates of 40, 80, 120 and 160 kg N ha−1, with 40 kg N ha−1 broadcast at seeding and the remaining of N sidedressed at the four-leaf stage for treatments with 80, 120 and 160 kg N ha−1. A 0 N control treatment was included. The mineral N treatments will be hereafter referred to as 0, 40, 80-S, 120-S, and 160-S, where S stands for split application. Two organic N sources, liquid swine manure (LSM) and liquid dairy manure (LDM), were broadcasted at seeding, at a target rate of 80 kg ha−1 total N, based on preliminary analysis (treatments hereafter referred to as LSM80 and LDM80). The actual amounts of N applied with LSM were 87.6 kg N ha−1 in 2011 and 75.0 kg N ha−1 in 2012 at the MWP site, and 86.4 kg N ha−1 in 2010 and 86.7 kg N ha−1 in 2011 at BS. The actual amounts of N applied with LDM were 79.7 kg N ha−1 in 2011 and 80.8 kg N ha−1 in 2012 at MWP, and 89.8 kg N ha−1 in 2010 and 85.5 kg N ha−1 in 2011 at BS. The main characteristics of the slurries can be found in Thivierge et al. (2015). The last N fertilization treatment was calcium ammonium nitrate broadcast at 80 kg N ha−1 at seeding (single-application treatment, referred to as 80) for comparisons with the organic N sources (80 vs. LSM80 and LDM80) and with the split application (80 vs. 80-S). Phosphorus (triple superphosphate; 40 and 20 kg P2O5 ha−1 at MWP and BS sites, respectively) and potassium chloride (60 and 40 kg K2O ha−1 at MWP and BS sites, respectively) were applied annually to the plots receiving mineral N, based on soil analyses and local recommendations (CRAAQ 2010). Phosphorus and K were not added to the plots fertilized with organic N sources since the manures provided these nutrients.
In the center of each subplot, a 1-m2 microplot was delineated and was fertilized with the same N source and rate than the rest of the subplot, but using a 15N-enriched fertilizer. The subplots fertilized with 160 kg ha−1 of mineral N did not receive 15N-enriched fertilizer because of scientific evidence that such a high N rate is not suitable for sweet pearl millet and sweet sorghum (Geng et al. 1989; Barbanti et al. 2006; Leblanc et al. 2012). The microplots receiving mineral N treatments (40, 80, 80-S, and 120-S) were fertilized with double-labelled ammonium nitrate (15NH 154 NO3 at 5 at.% 15N). A concentrated 15N solution was prepared in the laboratory. For each microplot, the appropriate amount of that solution was completed to 2 L with distilled water, which was applied evenly to the microplot using a watering can. To ensure a rapid infiltration of the solution into the soil, an additional 2 L of distilled water was immediately applied to the microplot. The actual atomic percentage excesses (APE) of the concentrated 15N solutions were 4.85 and 5.19 % at seeding and 4-leaf stage, respectively, at the BS site in 2010, 5.18 and 5.23 % at BS in 2011, 5.10 and 5.26 % at MWP in 2011, and 5.20 and 5.17 % at MWP in 2012.
Microplots receiving organic N treatments were fertilized with slurries to which ammonium sulfate ((15NH4)2SO4 at 99 at.% 15N) was added to raise the APE of the NH4-N fraction of the slurries to approximately 5 % without unbalancing its NH4-N/total N ratio. This allowed tracing the fate of slurry-derived NH4-N. The detailed analyses of the 15N-labelled slurries, including APE, are given in Table 1. The actual excess 15N added to the system with each fertilization treatment is detailed in Table 2.
All organic amendments applied at seeding were manually broadcast on the soil surface and incorporated into the top 5 cm of soil with hand tools within two hours of application to minimize ammonia volatilization (Rochette et al. 2001).
Samplings and analyses
The LSM and LDM were analyzed for N forms and concentrations on a composite subsample that was collected during manure application. Total N concentration of manures was determined by acid digestion whereas the concentrations of NH4-N and NO3-N were determined by KCl extraction, as described in Chantigny et al. (2007). The NH4-N concentration in the acid digestates and NH4-N and NO3-N concentrations in KCl extracts were measured with an automated continuous-flow injection analyzer (Model QuickChem 8000 FIA+, Lachat Instruments, Loveland, CO).
Both crop species were harvested at the maximum growth stage that it was possible to reach at each site, but before the risk of lodging was too high, in order to use their sweet juice for ethanol production. At MWP, the site with the warmest temperature, the harvesting stage corresponded to 3/4 of the inflorescence emerged on at least 75 % of the stalks. At BS, the site with the coldest temperature, the harvesting stage corresponded to 3/4 of the inflorescence emerged on at least 50 % of the stalks. Determination of DM yield is detailed in Thivierge et al. (2015). The central section (36 cm × 36 cm) of each microplot was harvested to avoid edge effects, and stalks were cut 5 cm above the soil surface. The harvested biomass was cut into 10-cm pieces and mixed. A 250-g subsample was taken and dried at 55 °C until constant weight to determine DM concentration, and ground to 1 mm (Whiley mill, Standard model 3, Arthur H. Thomas Co., Philadelphia, PA) to determine total N concentration by dry combustion (LECO CNS-1000, Leco Corp., St. Joseph, MI).
Three 30-cm soil cores were collected with a stainless steel auger (2 cm diam.) in the center of each microplot. Each core was divided in 10-cm increments and mixed to make one composite sample per depth for each microplot. Soil samples were air dried and ground to 0.15 mm (ball mixer-mill MM400, Retsch, Germany) to determine total N concentration.
Stable isotope ratio (15N/14N) was determined in aboveground crop biomass, soil, and manure samples with a continuous flow isotope ratio mass spectrometer (Model Optima, VG Isothech, Middlewich, Cheshire, UK) after sample combustion to N2 at 1020 °C with a combustion analyzer (Model NA1500, Carlo Erba Strumentazione, Rodano, Milan, Italy). Soils from the 0 N control plots were analyzed to determine natural 15N abundance at each site.
Calculations and statistical analyses
The crop N uptake (kg ha−1) was calculated as the product of crop DM yield and its N concentration. The 15N fertilizer N uptake efficiency (NupE) in the aboveground biomass (Eq. 1) and 15N recovery in the soil (0–30 cm) (Eq. 2) were calculated as per Muñoz et al. (2004) and Nyiraneza et al. (2010), respectively:
where a is the 15N abundance in aboveground biomass harvested in the microplot, b is the 15N abundance in the fertilizer, c is the 15N natural abundance measured in the soil of the control plot, and s is the 15N abundance in soil harvested in the microplot. The amounts of 15N recovered in plant aboveground biomass and soil were summed to calculate N budgets. Similar calculations were made for manure treatments (LSM80 and LDM80), and results were expressed on the basis of NH4-N applied with the manures, as the organic fraction of manure N was not labelled with 15N. The 15N unaccounted for in the soil–plant system was considered to represent environment loss, assuming that 15N remaining in soil below 30 cm was negligible.
The proportion of crop N uptake that derived from the fertilizer was calculated as per Fillery and Recous (2001; Eq. 3):
The contribution of soil to crop N uptake was taken as the difference between total N uptake and the fertilizer-derived N uptake (Fillery and Recous 2001).
The analysis of variance was carried out separately for each site, using the MIXED procedure in SAS (SAS Institute 2003) for the dependent variables: N concentration, N uptake, fertilizer-derived N uptake, soil-derived N uptake, and NupE. Treatment replication was considered random effect, whereas years, species, and N treatments were considered fixed effects. Data normality was verified using the UNIVARIATE procedure, using the Shapiro–Wilk test (Shapiro and Wilk 1965) to determine whether the residuals were normally distributed. The homogeneity of variance was verified visually with graphics of residuals. Standard error of means (SEM) is reported in Tables.
Quantitative contrasts were used to test the effect of increasing mineral N rate on plant N concentration, N uptake, fertilizer-derived N uptake, soil-derived N uptake, and NupE. Contrast analysis was also used to test the effect of N source (80 vs. LSM80 and LDM80), to compare the two organic N sources (LSM80 vs. LDM80), and to compare the effect of split versus single application of mineral N (80-S vs. 80). Statistical significance was postulated at P ≤ 0.05.
Results and discussion
Differences between species and response to increasing rates of mineral N
The average N concentration in aboveground biomass was similar for both species at the MWP site, whereas sweet pearl millet had a greater N concentration than sweet sorghum at the BS site (Table 3). The range of N concentrations reported here (5.3–12.6 g N kg−1 at MWP and 8.5–19.4 g N kg−1 at BS) echoed those reported for sweet sorghum by Han et al. (2011) in Northern China (7.5–10.2 g N kg−1 DM at 40 days after anthesis) and by Ceotto et al. (2014) in Italy (4.7 to 15.9 g N kg−1 DM). Crop N concentration responded similarly to N fertilization rate in both species, as indicated by the absence of interaction between species and N treatments, or species and years (Table 3). However, there was an interaction between N treatments and years at both sites. As a general trend, there was a linear increase in plant N concentration with increasing mineral N rate. Increase in plant N concentration with N rate was previously observed in sweet pearl millet (Leblanc et al. 2012) and in fiber sorghum (Barbanti et al. 2006), but it is observed for the first time in sweet sorghum. At the MWP site, this increase in N concentration was more acute in 2011 (5.3–12.6 g N kg−1 DM) than in 2012 (5.9–11.3 g N kg−1 DM). Also, the lower N concentration of the LSM80 treatment in 2011 than in 2012 (5.7 and 6.9 g N kg−1 DM, respectively) might have contributed to the interaction between N treatments and years. At the BS site, N concentration levelled off at 120 kg N ha−1 in 2010 and reached a maximum with 160 kg N ha−1 in 2011.
In regards to crop N uptake, there was an interaction between species and years at the MWP site (Table 4): the difference in crop N uptake between years was larger for sweet pearl millet (110 vs. 159 kg N ha−1) than for sweet sorghum (113 vs. 133 kg N ha−1). At the BS site, there were significant differences between species (153 kg N ha−1 for sweet pearl millet vs. 130 kg N ha−1 for sweet sorghum) and between years (167 kg N ha−1 in 2010 vs. 117 kg N ha−1 in 2011), but without interaction. There were no interaction between N treatments and years, or N treatments and species, at any site. However, at both sites, N uptake increased linearly with mineral N rates. An increase in N uptake with N rate was previously observed in sweet sorghum (Cosentino et al. 2012; Ceotto et al. 2014) and in corn (Tran et al. 1997), but was not observed before for sweet pearl millet. Crop N uptake ranged from 74 to 217 kg N ha−1 at MWP and from 76 to 202 kg N ha−1 at BS (Table 4). These values are in the range (100–270 kg N ha−1) reported for different varieties of sweet sorghum (Barbanti et al. 2006; Propheter and Staggenborg 2010; Han et al. 2011). Nitrogen uptake of 216 and 290 kg N ha−1 were also reported for grain sorghum (Lemaire et al. 1996; Ra et al. 2012). Propheter and Staggenborg (2010) compared many annual and perennial grasses used for energy production and found greater nutrient removal rates with annual grasses, particularly sweet sorghum, than with perennial grasses. This difference could be attributed to the capacity of perennial grasses to store minerals into the crown and rhizomes for overwintering.
For all experimental treatments, the crop N uptake was superior to the quantity of N applied, resulting in a negative N balance. The capacity for sweet pearl millet and sweet sorghum to efficiently capture soil N could be attributable to morphological characteristics of their root system. For instance, Wang et al. (2005) demonstrated in corn that root morphology influences the crop’s capacity to compete for soil N. Moreover, mycorrhizal associations have been observed in grain pearl millet and grain sorghum roots (Bagayoko et al. 2000), and grain sorghum was found to have a greater ability than corn to directly absorb organic N (Okamoto and Okada 2004). In the last decade, some authors discovered that grain sorghum and grain pearl millet roots have the capacity to exudate secondary metabolites that act as nitrification inhibitors in soil. This phenomenon, known as biological nitrification inhibition, keeps fertilizer N in the form of NH4-N in soils for a longer period, which improves crop N uptake and NupE, while reducing N losses through leaching and denitrification (Zakir et al. 2008; Subbarao et al. 2013).
In the present study, the fact that N uptake continued to increase linearly up to the highest N rate (160 kg ha−1) at both sites (Table 4) was unexpected, as Thivierge et al. (2015) reported that, for the same sites, the maximum DM yield was reached with 107–121 kg N ha−1. This finding suggests that these species expressed a phenomenon called N luxury consumption. This phenomenon occurs when N application to meet physiological needs (i.e. to achieve maximum growth) is exceeded: N accumulation in the plant therefore increases without affecting yield (Isfan et al. 1995). Nitrogen luxury consumption was described as a competitive strategy, as it deprives neighboring plants of a limiting nutrient (De Mazancourt and Schwartz 2012). It was previously observed in sweet sorghum by Cosentino et al. (2012).
The proportion of crop N uptake derived from the fertilizers (fertilizer-derived N, Table 5) amounted to 36 % on average at the MWP site, and 33 % at the BS site. When considering only the mineral N treatments, an average of 39 and 36 % of crop N uptake was derived from the fertilizer at MWP and BS, respectively. Under similar climatic conditions, Tran et al. (1997) measured that on average 46 % of N uptake in grain and silage corn was derived from mineral fertilizers at harvest, while Nyiraneza et al. (2010) reported values between 24 and 66 % for silage corn fertilized with 160 kg mineral N ha−1. These previous studies and the present one indicate that the main source of N to the crops generally comes from the soil reserve, in agreement with several previous studies with various crops, reviewed by Stevens et al. (2005). There were not any differences in fertilizer-derived N between species or between years. At both sites, there was a significant effect of N treatments, with a linear increase in fertilizer-derived N along with mineral N rate. Indeed, the amount of fertilizer N recovered by the crops increased substantially (+76 kg N ha−1 at MWP and +69 kg N ha−1 at BS) with mineral N rate from 40 to 120 kg ha−1 (Table 5), and the contribution of fertilizer N to crop N uptake increased from 22 to 52 % at the MWP site and from 22 to 46 % at BS, confirming the positive response of crops to fertilizer N rate.
Interestingly, the amount of soil-derived N also increased linearly with applied mineral N rate, but to a much smaller extent (+13 kg N ha−1 at MWP and +26 kg N ha−1 at BS). The increase in amounts of soil-derived N with increasing N rates was also observed in corn by Reddy and Reddy (1993), who suspected either an increased mineralization of soil organic matter, or a pool substitution between 14N from the soil and 15N from the fertilizer. The amount of soil-derived N was affected by years at both sites: 62 and 97 kg N ha−1 in 2011and 2012, respectively, at MWP; 122 and 69 kg N ha−1 in 2010 and 2011, respectively, at BS. This was expected since soil N supply depends on inter-annual variations in climatic factors affecting soil N mineralization.
The NupE (proportion of applied fertilizer N recovered in crop biomass), was similar for sweet pearl millet and sweet sorghum at both sites (Table 5). There was no significant difference between years at any site, although the difference was almost significant at the BS site (P = 0.052) with 54 % in 2010 and 65 % in 2011. At both sites, the fertilizer N rate had a linear effect on NupE in both species, which ranged from 54 to 82 % (Table 5; Fig. 1). These NupE are relatively high since many studies on corn reported NupE ranging from 28 to 60 % (Reddy and Reddy 1993; Liang and MacKenzie 1994; Tran et al. 1997; Stevens et al. 2005; Nyiraneza et al. 2010). Moreover, in corn, it has been demonstrated that NupE stays relatively constant (Stevens et al. 2005) or decreases as mineral N rate increases (Liang and MacKenzie 1994; Tran et al. 1997). In our case the linear increase in NupE with fertilizer N rate could relate to N luxury consumption. Indeed, N luxury consumption has been positively related to soil N availability (Tripler et al. 2002). In agreement, our results suggest that the phenomenon was amplified with higher N fertilization rates. The continued increase in crop N uptake to the highest N rate (160 kg ha−1) and the strong linear relationship (P < 0.001) between fertilizer-derived N uptake and N rates at both sites (Tables 4, 5) corroborate this finding. A concern stemming from possible N luxury consumption is the management of N-rich residues. After extraction of juice from the stalks, the residues can be either returned to the field or fed to cattle. In either case, care must be taken to avoid environmental loss of N from the field or animal health problems.
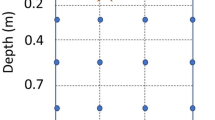
Partitioning of the applied 15N in the soil–plant system for sweet pearl millet and sweet sorghum at the Mixedwood Plains (a) and Boreal Shield (b) sites, in response to six fertilization treatments. Vertical bars represent standard deviation of the proportion of the 15N recovered in the soil–plant system. S, N rate is split-applied with 40 kg N ha−1 at seeding and the rest side-dressed at the 4-leaf stage. NupE, fertilizer N uptake efficiency; LSM80, liquid swine manure applied at a target rate of 80 kg total N ha−1; LDM80, liquid dairy manure applied at a target rate 80 kg total N ha−1. For LSM80 and LDM80, results are expressed based on applied NH4-N, as manure organic N was not labelled with 15N
As a result of the high NupE, the amounts of residual fertilizer found in soil (0–30 cm) at harvest were generally low with the mineral fertilizer (Fig. 1). For all treatments, 72–90 % of the residual N was recovered in the top 10 cm of soil. This is in agreement with previous studies demonstrating that applied N does not significantly migrate at depth during the growing season wherever it is applied as LSM (Morvan et al. 1997; Chantigny et al. 2014), LDM (Muñoz et al. 2004), or mineral fertilizers (Reddy and Reddy 1993; Nyiraneza et al. 2010).
In the present study, the small proportion of fertilizer N recovered in the 20–30 cm soil layer (2.1–3.5 % at MWP, and 1.5–2.3 % at BS; Fig. 1) suggested that little applied N was present at greater depths, and that N not recovered in the top 30 cm of soil and in crop aboveground biomass was actually lost. In regards to the amount of unrecovered N, there was an interaction between N treatment and species at the MWP site (P = 0.018). However, the differences among N treatments and between species were very small, as unrecovered N ranged from 0 to 12 kg N ha−1. At the BS site, there was an interaction between N treatments and years (P = 0.029). The main difference between years was for the treatment 120-S, with higher amount of unrecovered N in 2010 than in 2011 (24 vs. 10 kg N ha−1, respectively). Overall, the unrecovered N at BS was low and ranged from 6 to 24 kg N ha−1 for the mineral N treatments. This further confirms the high efficiency of sweet pearl millet and sweet sorghum to capture applied N. These results are in accordance with previous studies on sweet sorghum, where the sum of nitrate leaching and ammonia volatilization with mineral fertilization rates of 60 and 120 kg N ha−1 were 7.1 and 7.7 kg N ha−1, respectively, in Cosentino et al. (2012), and 18.2 and 19.1 kg N ha−1, respectively, in Barbanti et al. (2006). In corn (Reddy and Reddy 1993; Tran et al. 1997), unrecovered fertilizer N increased from 14 to 103 kg ha−1 with N rates from 60 to 200 kg N ha−1. Therefore, the direct environmental impact from N fertilization of sweet pearl millet and sweet sorghum may be lower than that of corn, especially at higher N rates. Lemaire et al. (1996) reached similar conclusions when comparing the risk of nitrate leaching under grain sorghum and corn.
Response to mineral and organic N sources
At both sites, plant N concentration and N uptake were lower with organic N sources than with the mineral N source for both species (Tables 3, 4). This was expected since the fertilizer value of manures reported by Thivierge et al. (2015) varied from 15 to 52 %, indicating that manure N was less available to the crops than N from the mineral fertilizer. As a result, NupE was smaller with manures (34 % of manure NH4-N at MWP and 43 % at BS) than with the equivalent mineral N treatment (67 % of applied N at MWP and 75 % at BS; Table 5). The lower uptake of manure-derived N was expected because only 31 and 72 % of manure total N was readily available as ammonia-N (Table 1). This is in agreement with Carter et al. (2010) who observed that the apparent NupE in orchardgrass (Dactylis glomerata L.) and reed canarygrass (Phalaris arundinacea L.) was greater with mineral fertilizer N than with LDM.
Different factors could explain why crop N uptake was lower with manures than with mineral N. Ammonia loss may have occurred through volatilization following manure application. However, the short 2-h delay elapsed between application and incorporation likely limited those losses (Rochette et al. 2001). It is also possible that N immobilization occurred as highly decomposable carbon was applied with the manures (Kirchmann and Lundvall 1993). This hypothesis is supported by the fact that most of the manures (LDM80 and LSM80 at MWP, and LDM80 at BS) resulted in a larger proportion of N recovered in the top soil layers than mineral N treatments (Fig. 1). Morvan et al. (1997) reported that 15–35 % of the NH4-N from LSM could be immobilized. Moreover, they argued that immobilization of manure N is favored when crops are not actively growing at the time of manure application, as in our experiment, because ammonia-N remains available to microorganisms for a longer period of time. The immobilized N could eventually be lost and have possible negative impact on the environment, if not taken up by subsequent crops (Chantigny et al. 2014; Sieling et al. 2014).
At the BS site, the proportion of applied N presumably lost to the environment (unrecovered N) was larger for manures than for the mineral fertilizer (18 % of NH4-N for LDM and 33 % for LSM versus 10 % of applied N for treatment 80, P = 0.006), and was larger for LSM than for LDM (P = 0.012, Fig. 1). There were no significant differences in unrecovered N between manures and mineral treatment at the MWP site (13 % of NH4-N for LDM, 19 % for LSM, and 10 % for mineral treatment 80, P = 0.172). The amount of unrecovered N was lower than 20 kg N ha−1 for all organic treatments for all sites and years, except for LSM80 in 2010 at the BS site where it reached 32 kg N ha−1.
At the MWP site, LSM80 resulted in slightly greater plant N concentration, N uptake, and fertilizer-derived N than LDM80 (Tables 3, 4, 5). However, NupE were similar between manures at both sites. At the BS site, the proportion of N recovered in the 0-10 cm soil layer was greater for LDM than for LSM (P < 0.001). Immobilization of soil N following manure application could also explain these differences between manures, as LDM may induce greater N immobilization, and therefore be less available to crop, than LSM (Kirchmann and Lundvall 1993).
Response to single and split application of mineral N
Splitting N application between seeding and the 4-leaf stage, rather than applying all N at seeding, did not affect plant N concentration and N uptake at any site (Tables 3, 4). In comparison with corn, Abassi et al. (2013) found an increase in N concentration and N uptake with split N application, whereas Dordas et al. (2008) did not observe any difference. The split N application had no effect on NupE and on the proportion of crop N uptake derived from the soil at any site, and had only a small effect (P = 0.059) on fertilizer-derived N at MWP, where the split application increased N uptake by 7 kg when compared to the single application (Table 5). Finally, the split N application slightly lowered the proportion of unrecovered N at MWP (10 % for single application versus 3 % for split, P = 0.037, Fig. 1), and the proportion of N recovered in the soil at BS (24 % for single application vs. 19 % for split, P < 0.001, Fig. 1). Along with the finding by Thivierge et al. (2015) that splitting N did not significantly increase crop yields in sweet pearl millet and sweet sorghum, the small differences that were observed in the present study do not justify recommending split N application for these crop species in eastern Canada, at least with N rate of 80 kg ha−1.
Conclusion
This investigation confirmed the high NupE of sweet pearl millet and sweet sorghum under wet and cool conditions in eastern Canada. The NupE ranged from 54 to 82 % of applied mineral N, values much higher than those reported for other crops such as corn. Moreover, N uptake was large for both species at both sites and at all N rates, indicating that these crops have the ability to efficiently recover N from fertilizers and from the soil reserve. Luxury N consumption likely occurred in both species and, consequently, N-rich residues remaining after extraction of juice from the stalks will need to be managed with care to prevent further N losses. More research is required to identify the factors explaining the peculiar ability of sweet pearl millet and sweet sorghum to recover N from soil and fertilizer, such as morphological characteristics of their root system, possible mycorrhizal associations, and biological nitrification inhibition. Crop N uptake and NupE were lower with animal manures than mineral fertilizer, presumably because of soil N immobilization in the days following manure application. Because of the very high NupE, the presumed environmental losses (unrecovered N at harvest) were low for all treatments including manures. Overall, these findings together with the low N requirements (e.g. Thivierge et al. 2015) indicate that sweet sorghum and sweet pearl millet could be successfully grown in eastern Canada with little risk to the environment provided that their N-rich residues are properly managed.
References
Abassi MK, Tahir MM, Rahim N (2013) Effect of N fertilizer source and timing on yield and N use efficiency of rainfed maize (Zea mays L.) in Kashmir–Pakistan. Geoderma 195–196:87–93
Bagayoko M, Buerkert A, Lung G, Bationo A, Römheld V (2000) Cereal/legume rotation effects on cereal growth in Sudano-Sahelian West Africa: soil mineral nitrogen, mycorrhizae and nematodes. Plant Soil 218:103–116
Barbanti L, Grandi S, Vecchi A, Venturi G (2006) Sweet and fibre sorghum (Sorghum bicolor (L.) Moench), energy species in the frame of environmental protection from excessive nitrogen loads. Eur J Agron 25:30–39
Brown DM, Bootsma A (1993) Crop heat units for corn and other warm season species in Ontario. Factsheet, Agdex 111/31. Ontario Ministry of Agric., Food, and Rural Affairs, Queen’s Park
Carter JE, Jokela WE, Bosworth SC (2010) Grass forage response to broadcast or surface-banded liquid dairy manure and nitrogen fertilizer. Agron J 102:1123–1131
Centre de référence en agriculture et agroalimentaire du Québec, CRAAQ (2010) Guide de référence en fertilisation, 2nd edn. CRAAQ, QC, Canada
Ceotto E, Castelli F, Moschella A, Diozzi M, Di Candilo M (2014) It is not worthwhile to fertilize sweet sorghum (Sorghum bicolor L. Moench) with cattle slurry: productivity and nitrogen-use efficiency. Ind Crop Prod 62:380–386
Chantigny MH, Angers DA, Rochette P, Bélanger G, Massé D, Côté D (2007) Gaseous nitrogen emissions and forage nitrogen uptake on soils fertilized with raw and treated swine manure. J Environ Qual 36:1864–1872
Chantigny MH, Angers DA, Rochette P, Pomar C, Pelster DE (2014) Evidencing overwinter loss of residual organic and clay-fixed nitrogen from spring-applied, 15N-labelled pig slurry. Can J Soil Sci 94:1–8
Cosentino SL, Mantineo M, Testa G (2012) Water and nitrogen balance of sweet sorghum (Sorghum bicolor moench (L.)) cv. Keller under semi-arid conditions. Ind. Crop. Prod. 36:329–342
De Mazancourt C, Schwartz MW (2012) Starve a competitor: evolution of luxury consumption as a competitive strategy. Theor Ecol 5:37–49
Dordas CA, Lithourgidis AS, Matsi T, Barbayiannis N (2008) Application of liquid cattle manure and inorganic fertilizers affect dry matter, nitrogen accumulation, and partitioning in maize. Nutr Cycl Agroecosyst 80:283–296
Dos Passos Bernardes A, Tremblay GF, Bélanger G, Brégard A, Seguin P, Vanasse A (2015) Sugar yield of sweet pearl millet and sweet sorghum as influenced by harvest dates and delays between biomass chopping and pressing. Bioenerg Res 8:100–108. doi:10.1007/s12155-014-9504-y
Ecological Stratification Working Group, ESWG (1995) A National Ecological Framework for Canada. Agriculture and Agri-Food Canada, Research Branch, Centre for Land and Biological Resources Research and Environment Canada, State of the Environment Directorate, Ecozone Analysis Branch, Ottawa/Hull. http://sis.agr.gc.ca/cansis/publications/manuals/1996/A42-65-1996-national-ecological-framework.pdf. Accessed 3 November 2014
Fillery IRP, Recous S (2001) Use of enriched 15N sources to study soil N transformations. In: Unkovich M et al (eds) Stable isotope techniques in the study of biological processes and functioning of ecosystems. Kluwer Academic Publishers, Boston, pp 167–194
Financière agricole du Québec, FADQ (2015) Statistiques. Assurance-récolte. Rendements réels pour les années 1995 à 2012. www.fadq.qc.ca/statistiques_et_taux/statistiques/assurance_recolte/rendements_reels.html. Accessed 9 February 2015
Gagnon B, Ziadi N (2010) Grain corn and soil nitrogen responses to sidedress nitrogen sources and applications. Agron J 102:1014–1022
Geng S, Hills FJ, Johnson SS, Sah RN (1989) Potential yields and on-farm ethanol production cost of corn, sweet sorghum, fodderbeet, and sugarbeet. J Agron Crop Sci 162:21–29
Han LP, Steinberger Y, Zhao YL, Xie GH (2011) Accumulation and partitioning of nitrogen, phosphorus and potassium in different varieties of sweet sorghum. Field Crop Res 120:230–240
Isfan D, Zizka J, D’Avignon A, Deschênes M (1995) Relationships between nitrogen rate, plant nitrogen concentration, yield and residual soil nitrate-nitrogen in silage corn. Commun Soil Sci Plant Anal 26:2531–2557
Kirchmann H, Lundvall A (1993) Relationship between N immobilization and volatile fatty acids in soil after application of pig slurry and cattle slurry. Biol Fertil Soils 15:161–164
Leblanc V, Vanasse A, Bélanger G, Seguin P (2012) Sweet pearl millet yields and nutritive value as influenced by fertilization and harvest dates. Agron J 104:542–549
Lemaire G, Charrier X, Hébert Y (1996) Nitrogen uptake capacities of maize and sorghum crops in different nitrogen and water supply conditions. Agronomie 16:231–246
Liang BC, MacKenzie AF (1994) Corn yield, nitrogen uptake and nitrogen use efficiency as influenced by nitrogen fertilization. Can J Soil Sci 74:235–240
Liang BC, MacKenzie AF, Zhang TQ (1996) Grain yields and grain nitrogen concentration of corn as influenced by fertilizer nitrogen rate. J Agron Crop Sci 177:217–223
Morvan T, Leterme P, Arsène GG, Mary B (1997) Nitrogen transformations after the spreading of pig slurry on bare soil and ryegrass using 15 N-labelled ammonium. Eur J Agron 7:181–188
Muñoz GR, Kelling KA, Powell JM, Speth PE (2004) Comparison of estimates of first-year dairy manure nitrogen availability or recovery using nitrogen-15 and other techniques. J Environ Qual 33:719–727
Nyiraneza J, Chantigny MH, N’Dayegamiye A, Laverdière MR (2010) Long-term manure application and forages reduce nitrogen requirements of silage corn-cereal cropping systems. Agron J 102:1244–1251
Okamoto M, Okada K (2004) Differential responses of growth and nitrogen uptake to organic nitrogen in four gramineous crops. J Exp Bot 55:1577–1585
Propheter JL, Staggenborg S (2010) Performance of annual and perennial biofuel crops: nutrient removal during the first two years. Agron J 102:798–805
Ra K, Shiotsu F, Abe J, Morita S (2012) Biomass yield and nitrogen use efficiency of cellulosic energy crops for ethanol production. Biomass Bioenerg 37:330–334
Reddy GB, Reddy KR (1993) Fate of nitrogen-15 enriched ammonium nitrate applied to corn. Soil Sci Soc Am J 57:111–115
Rochette P, Chantigny MH, Angers DA, Bertrand N, Côté D (2001) Ammonia volatilization and soil nitrogen dynamics following fall application of pig slurry on canola crop residues. Can J Soil Sci 81:515–523
SAS Institute (2003) User’s guide V.9.1.3. SAS Inst., Cary
Shapiro SS, Wilk MB (1965) An analysis of variance test for normality (complete samples). Biometrika 52:591–611
Sieling K, Ni K, Kage H (2014) Application of pig slurry—first year and residual effects on yield and N balance. Eur J Agron 59:13–21
Stevens WB, Hoeft RG, Mulvaney RL (2005) Fate of nitrogen-15 in a long-term nitrogen rate study: II. Nitrogen uptake efficiency. Agron J 97:1046–1053
Subbarao GV, Nakahara K, Ishikawa T, Ono H, Yoshida M, Yoshihashi T, Zhu Y, Zakir HAKM, Deshpande SP, Hash CT, Sahrawat KL (2013) Biological nitrification inhibition (BNI) activity in sorghum and its characterization. Plant Soil 366:243–259
Thivierge M-N, Chantigny MH, Bélanger G, Seguin P, Bertrand A, Vanasse A (2015) Response to nitrogen of sweet pearl millet and sweet sorghum grown for ethanol in eastern Canada. Bioenerg Res. doi:10.1007/s12155-014-9558-x
Tran TS, Giroux M, Cescas MP (1997) Effect of N rate and application methods on 15 N-labelled fertilizer use by corn. Can J Soil Sci 77:9–19
Tripler CE, Canham CD, Inouye RS, Schnurr JL (2002) Soil nitrogen availability, plant luxury consumption, and herbivory by white-tailed deer. Oecologia 133:517–524
Wang Y, Mi G, Chen F, Zhang J, Zhang F (2005) Response of root morphology to nitrate supply and its contribution to nitrogen accumulation in maize. J Plant Nutr 27:2189–2202
Yu J, Zhang T, Zhong J, Zhang X, Tan T (2012) Biorefinery of sweet sorghum stem. Biotechnol Adv 20:811–816
Zakir HAKM, Subbarao GV, Pearse SJ, Gopalakrishnan S, Ito O, Ishikawa T, Kawano N, Nakahara K, Yoshihashi T, Ono H, Yoshida M (2008) Detection, isolation and characterization of a root-exuded compound, methyl 3-(4-hydroxyphenyl) propionate, responsible for biological nitrification inhibition by sorghum (Sorghum bicolor). New Phytol 180:442–451
Acknowledgments
The senior author gratefully acknowledges the support of the Natural Sciences and Engineering Research Council of Canada (NSERC) for a fellowship. The work was financially supported by the Programme de soutien à l’innovation en agroalimentaire (PSIA) of the Ministère de l’agriculture des pêches et de l’alimentation du Québec (MAPAQ). We thank Annie Brégard for statistical advice, and Valérie Bélanger, Marie-Eve Bernard, Amélie Desilets Roy, Johanne Tremblay, and Gabriel Lévesque for assistance during field and laboratory work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thivierge, MN., Chantigny, M.H., Seguin, P. et al. Sweet pearl millet and sweet sorghum have high nitrogen uptake efficiency under cool and wet climate. Nutr Cycl Agroecosyst 102, 195–208 (2015). https://doi.org/10.1007/s10705-015-9689-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10705-015-9689-2