Abstract
Global warming has profound effects on the living conditions and metabolism of organisms, including fish. The metabolic rate of fish increases as the temperature increases within its thermal tolerance range. Temperature changes can trigger a range of physiological reactions, including the activation of the stress axis and the production of HSPs. Under stress conditions, HSPs play a crucial role in antioxidant systems, immune responses, and enzyme activation. This study examined the effects of heat shock products (HSPs) on fish under temperature stress. Various HSP inducers (HSPis), including Pro-Tex®, amygdalin, and novel synthetic compounds derived from pirano piranazole (SZ, MZ, HN-P1, and HN-P2), were evaluated in isolated cells of sterlet sturgeon (Acipenser ruthenus) treated with temperature changes (18, 22, and 26 °C). Cells from the liver, kidney, and gills were cultured in vitro in the presence and absence of temperature stress and treated with HSPi compounds. To assess HSP27, HSP70, and HSP90 expression patterns, Western blotting was used. The HSPis and HSPi + temperature stress treatments affected the antioxidant capacity and immune parameters, among other enzyme activities. The results showed that HSPi compounds increase cell survival in vitro, positively modulate HSP expression and antioxidant levels, and decrease immune parameters. HSPi can increase A. ruthenus tolerance to temperature stress. In addition, the results indicate that these compounds can reverse adverse temperature effects. Further research is needed to determine how these ecological factors affect fish species' health in vivo and in combination with other stressors.
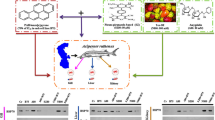
Graphical abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Climate change and global warming are serious environmental problems. Environmental security is more prominent in the Caspian Sea. Due to its geopolitical and geoeconomic dimensions as well as its unique characteristics, this large lake is highly vulnerable to climate change. In fact, the natural isolation of the Caspian Sea has made its global warming conditions unique (Yazdanpanah Dero et al. 2020). From 1982 to 2019, the water surface temperature of the Caspian Sea in Iran increased between 5 and 8°C (Leroy et al. 2020; Zakerinejad et al. 2022).
In the aquatic environment, temperature is a crucial variable that directly affects marine organism survival. Temperature also influences the physiological stress response of fish (Cho et al. 2015). The stress response of fish depends on the duration of exposure and the temperature approaching the upper and lower limits (Islam et al. 2022). The adverse effects of temperature changes in fish have been fully proven to be related to oxidative stress (Birnie-Gauvin et al. 2019; Klein et al. 2017). Reactive oxygen species (ROS) may be produced by climate change. High levels of ROS can damage DNA, proteins, and lipids in cells as well as lysozymes, peroxidases, and complement factors in humoral immunity (Filho 2007; Kim et al. 2021; Mozanzadeh et al. 2021). Increased ROS production can lead to the production of proinflammatory cytokines, which further link inflammation to inflammatory diseases (Dominguez et al. 2005). A number of immune system processes are regulated by ROS, such as proliferation, differentiation, intracellular signaling, chemoattraction, and antigen cross-presentation (Birnie-Gauvin et al. 2019). Among the enzymes that protect against oxidative stress are glutathione S-transferases (GST), glutathione peroxidases (GPx), and total antioxidant capacity (TAC) (Ghafoori et al. 2017). Fish can maintain homeostasis under various stressors, including temperature and salinity changes, by scavenging ROS (Neamatallah et al. 2022). Cortisol, a corticosteroid, regulates water balance and is produced during stress (Lin et al. 2000). Stress triggers physiological mechanisms that maintain body homeostasis, affecting fish immunity (Lu et al. 2022). The fish innate immune system includes complement component 3 (C3), lysozyme (LYZ), and immunoglobulin M (IgM), which respond to pathogens and environmental stressors (Biller-Takahashi and Urbinati 2014). Successful aquaculture depends on the organism's environment. During the culture period, therefore, it is essential to maintain an optimum environment in order maximize productivity (Sharma et al. 2017).
Several factors contribute to the plight of Caspian sturgeon populations, including overfishing, increased water pollution, changes in water chemistry, and blocked migration routes. These factors put sturgeons at critical risk in approximately 85% of cases (Moghim et al. 2006; Prokopchuk et al. 2016). Acipenser ruthenus is one of the smallest sturgeon species (Stanic et al. 2006), while the sterlet sturgeon is a critically endangered species in the Caspian Sea (Lenhardt et al. 2006). Iran's Fisheries Organization maintains sturgeon stocks in the Caspian Sea by releasing up to three million fingerling fish into rivers every year (Baharloei et al. 2020). However, after release, fish fingerlings often suffer extensive economic losses and mass mortality due to environmental stresses (Khodabandeh et al. 2009). Stressors can cause fish to experience different stress reactions at the cellular level. In 1998, 24.5 million sturgeon fingerlings were released into the Caspian Sea, but in 2008, this number decreased to 10 million. In recent years, this number has reached one million (Afraei Bandpei et al. 2010; Hajirezaee et al. 2017). It is challenging for the Fisheries Organization to maintain sturgeon stocks.
As living organisms are subjected to constantly changing conditions, maintaining cellular protein homeostasis is critical for cell survival and integrity because protein misfolding and aggregation can cause protein malfunctions and a variety of diseases (Boshoff et al. 2004; Hu et al. 2022). Heat shock proteins (HSPs) are ubiquitous and conserved proteins that function to maintain proteostasis in both prokaryotes and eukaryotes (Pirali et al. 2020; Taheri et al. 2022). The stress-coping abilities of HSPs were long known to function. Environmental factors and oxidative stress increase HSP27, HSP70, and HSP90 protein levels (Mahanty et al. 2017; Yu et al. 2022). Aquaculture could benefit from natural compounds that induce HSP in animals without causing traumatic injury. A variety of natural and manufactured compounds can stimulate HSP expression, resulting in the activation of HSP and the induction of HSP. Pro-Tex® induces HSPs in Persian sturgeons and other organisms as a resistant precursor. Pro-Tex® is a soluble form of TEX-OE®, a plant-derived substance made from Opuntia ficus indica or Nopal cactus (NOP) (Otaka et al. 2007; Salmani et al. 2024; Vahdatiraad et al. 2023b). Another commonly used HSPi is amygdalin (AMG) (Ahmed et al. 2012). Numerous studies have shown that pyrazole-based compounds activate the HSF1 gene to upregulate HSP70 expression (Brough et al. 2005; Küçükgüzel and ŞenkardeŞ 2015).
Fish cell lines have been developed from a broad range of tissues, such as the ovary, fin, swim bladder, heart, spleen, liver, kidney, gill, eye muscle, vertebrae, brain, and skin (Lakra et al. 2011). A gill cell regulates acid‒base balance, whereas a liver cell regulates metabolism and detoxification (Stapp et al. 2015; Tresguerres et al. 2020). This study investigated whether HSPi can mitigate the effects of temperature changes on Acipenser ruthenus liver, gill, and kidney cells. Specifically, we will assess the effects of HSPis, including Tex-OE and amygdalin, together with newly synthesized pirano-piranazole-based inducers of HSPs called SZ, MZ, HN-P1, and HN-P2, on HSP27, HSP70, and HSP90 protein expression and antioxidant and immunological responses. Our hope is that this research will provide insights into the mechanisms underlying the protective effects of HSP induction and contribute to the development of effective strategies for mitigating the side effects of environmental changes on sturgeons.
Materials and methods
Synthesis of novel compounds based on pirano-piranazole
The synthesis of pyrazolone or 5-methyl-2,4-dihydro-3H-pyrazole-3-one is carried out via a condensation reaction between ethyl acetoacetate and hydrazine. Acetic acid is used as a catalyst for this reaction.
Synthesis of 4,4- (1,4-phenylene) bis(5-amino-3-methyl-1,4-dihydropyrano [2,3-c]pyrazole-6-carbonitrile: SZ)
This compound was synthesized from the reaction of 2 mmol of pyrazolone, 1.2 mmol of malonitrile, and 1 mmol of terephthalaldehyde in the presence of 5% sodium hydroxide solution (NaOH) as a catalyst in ethanol under reflux conditions. H-NMR, C-NMR, and FT-IR were used to confirm the structure of the synthesized compound (Supplementary information (SI): Fig. 1a, b, and c, respectively) (Shahriyari-Nejad et al. 2020; Zarei et al. 2024a).
Synthesis of 4,4- (1,3-phenylene) bis(5-amino-3-methyl-1,4-dihydropyrano [2,3-c]pyrazole-6-carbonitrile: MZ)
Under reflux conditions, 2 mmol of pyrazolone was reacted with 1.2 mmol of malonitrile and 1 mmol of isophthalaldehyde in ethanol with 5% sodium hydroxide (NaOH) as a catalyst. By using FT-IR measurements, the structure of the synthesized compound was confirmed (see SI Fig. 2) (Shahriyari-Nejad et al. 2020).
Synthesis of 6-amino-4-(2-methoxyphenyl)-3-methyl-1,4-dihydropyrano [2,3-c]pyrazole-6-carbonitrile (HN-P1)
Synthesis was carried out using 1 mmol of a pyrazolone base compound, 1.1 mmol of malonitrile, and 1 mmol of 2-methoxybenzaldehyde (a methoxyarylaldehyde). The structure of the synthesized compound was confirmed by FT-IR (SI Fig. 3) (Pourmousavi et al. 2022).
Synthesis of 6-amino-4-(3-methoxyphenyl)-3-methyl-1,4-dihydropyrano [2,3-c]pyrazole-5-carbonitrile (HN-P2)
1 mmol of pyrazolone base compound was reacted with 1.1 mmol of malonitrile and 1 mmol of 3-methoxybenzoaldehyde to yield this compound. FT-IR confirmed the structure of the synthesized compound (SI Fig. 4) (Pourmousavi et al. 2022).
Isolation of cells from liver, gill, and kidney tissue
In this study, a fingerling species, Acipenser ruthenus, which measured 18 cm in length and weighed 15.28 g, was used. The specimens were obtained from the International Sturgeon Research Institute. The liver, gill, and kidney tissues were dissected under completely sterile conditions and transferred to separate T-25 cm2 flasks. The culture was performed in a T-25 cm2 flask at 22°C and 95% humidity with essential medium supplemented with culture medium [80% Leibovitz's L-15 medium (L15), 20% fetal bovine serum (FBS), 100 U/ml streptomycin/penicillin (1%) and 100 U/ml amphotericin B (1%) prepared by Sigma‒Aldrich]. The complete medium was replaced every 24 h. Under a microscope, the cells were examined daily for contamination and growth. Monolayers of liver, gill, and kidney cells formed after 10–15 days. Two washes with phosphate-buffered saline (PBS, pH: 7.4) were conducted to remove any remaining culture medium from the cells adhering to the bottom of the flask. Trypsin–EDTA (0.25%; Sigma‒Aldrich) was then used to detach the cells from the flask. The monolayer cells were dispersed throughout the flask by using trypsin–EDTA. After 3 min, the cells were centrifuged for 5 min at 3000 rpm. For treatment, the supernatant was discarded, and the cell sediment was transferred to a flask (Butler 2004; Zarei et al. 2024a).
Determination of the optimal HSPi dose
Treatment with HSPis and MTT assay
Liver, kidney, and gill cells (5 × 105 cells/ml) were counted and plated in 96-well plates. At 22°C, 5% CO2, and 95% humidity, the cells were incubated in DMEM with 10% FBS, 100 U/ml streptomycin/penicillin (1%) and 100 U/ml amphotericin B (1%). After 24 h, the medium was replaced with DMEM containing 1% FBS. The HSPi concentrations used were as follows: NOP (Source Naturals, INC, Box 2118, Santa Cruz, CA 95062): 0, 50, 100, 200, 400, 800, and 1600 mM; AMG (Sigma‒Aldrich): 0, 1.25, 2.5, 5, 10, 20, 40, and 80 mM; and synthetic compounds: 0, 5, 10, 20, 40, 80, and 160 µM for 24 h. A group without treatment was considered the control group. To determine the toxicity of the HSPi compounds and cell viability, a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) test was conducted, with a final volume of 200 µl for each well. Each concentration was tested three times.
The MTT assay measures the production of purple‒blue color when tetrazolium salts are reduced to formazan forms, which can be measured spectrophotometrically. At the end of the treatment period, the medium without FBS was replaced with 10 µl (10% of final volume) of MTT reagent (5 mg/ml, Sigma‒Aldrich), and the plates were incubated at 22°C, 5% CO2, and 95% humidity for 3‒4 h. The final volume of each well was 100 µl. The supernatant was removed, and the formazan crystals formed were solubilized in 100 μl of dimethyl sulfoxide (DMSO; Sigma‒Aldrich) for 30 min. The absorbance was measured at 570 nm and is expressed as a percentage of the control (which is considered 100%) (Ahmadi et al. 2014). The viability was calculated using the following formula:
The optimal doses of HSPi were determined based on results from the MTT assay (Fig. 1): 800 mM NOP (N800), 80 mM AMG (A80) and 80 µM SZ (SZ80) (Zarei et al. 2024a; Zarei et al. 2024b).
The optimal dose of each compound (HSPi) was determined (means ± SEM). Livers, gills, and kidneys of sterlet cells were treated with a) (0–160) µM SZ, b) (0–160) µM MZ, c) (0–160) µM HN-P1, d) (0–160) µM HN-P2, e) (0–1600) mM Nopal Endurance (NOP), and f) (0–80) mM Amygdalin (AMG). Each treatment group was repeated three times. Within each column, different letters indicate significantly different groups according to Duncan's test (a, b, c, and…; P < 0.0001)
Grouping of treatments
A temperature of 22°C (T22) was considered the control temperature, and temperatures of 18 (T18) and 26°C (T26) were considered stress temperatures (Ndong et al. 2007; Wiles et al. 2020). The 5 × 105 cells/ml were counted and transferred to a 24-well plate with culture medium [90% DMEM, 10% FBS, 100 U/ml streptomycin/penicillin (1%) and 100 U/ml amphotericin B (1%)]. The plates were incubated at 22°C, 5% CO2, and 95% humidity. After 24 h, the culture medium was replaced with medium containing 1% FBS, and the cells were divided into four groups (Fig. 2): 1) the control group (without HSPi; T22, T18, and T26), 2) the group in which the cells received different concentrations of HSPi (N800, A80, and SZ80; at 22°C: HSPi + T22), 3) the group in which the cells were treated with HSPi for 24 h at 22°C and then placed at 18°C (HSPi + T18), and 4) the group in which the cells were treated with HSPi for 24 h at 22°C and then incubated at 26°C (HSPi + T26). Afterward, the cells were treated for 24 h and then centrifuged for 5 min at 4°C at 5000 rpm. Each group was repeated 3 times. The final volume of each well was 500 µl. Aseptic conditions were followed in all patients.
Grouping of treatments Twelve groups were considered: 1) Control cells without treatment at 22°C (T22), HSPi groups containing 22°C, 2) 80 mM amygdalin (A80 + T22), 3) 80 µM SZ (SZ80 + T22), 4) 800 mM Nopal Endurance (N800 + T22), 5) cells without treatment at 18 °C (T18), and HSPi groups incubated at 22°C. After 24 h, the cells were transferred to a temperature of 18°C, 6) 80 mM amygdalin (A80 + T18), 7) 80 µM SZ (SZ80 + T18), 8) 800 mM Nopal Endurance (N800 + T18), 9) cells without treatment at 26°C (T26), and HSPi groups incubated at 22°C. After 24 h, the cells were transferred to a temperature of 26°C: 10) 80 mM amygdalin (A80 + T26), 11) 80 µM SZ (SZ80 + T26), and 12) 800 mM Nopal Endurance (N800 + T26). A total of 5 × 105 cells/ml from each cell line (kidney, liver and gill) were counted and transferred to 24-well plates. Each group was repeated 3 times, and after 24 h, the cells were collected from the wells and used for subsequent tests
The protein concentration in each treatment group (5 × 105 cells/ml) was determined using the Bradford assay (Bradford 1976), with bovine plasma albumin (Sigma‒Aldrich) used as a standard (SI section 1-3).
Western blotting analysis
A western blotting technique described by Werner et al. was used to determine the expression patterns of HSP27, HSP70, and HSP90 (Werner et al. 2007). Briefly, 25 µg of protein from each sample was electrophoresed on a 12% polyacrylamide gel and 5% stacking gel using sodium dodecyl sulfate‒polyacrylamide gel electrophoresis (SDS‒PAGE). The proteins were then transferred to a polyvinylidene fluoride membrane using an electroblotter (Millipore, Bedford, Massachusetts). Primary antibodies [sc-13132 (Santa Cruz Biotechnology, INC), H5147, and H1775 from Sigma‒Aldrich] were used for immunoblotting of HSP27, HSP70, and HSP90. For ß-actin, which is a housekeeping protein (control), monoclonal anti-ß-actin (A3854) and anti-mouse IgG-peroxidase conjugate (A2304, Sigma‒Aldrich) were utilized as primary and secondary antibodies, respectively. The primary antibody binds specifically to the protein of interest, while the secondary antibody recognizes and binds to the primary antibody, allowing for detection of the protein. Finally, 3,3′-diaminobenzidine (DAB; Sigma‒Aldrich, D-7304) and H2O2 were used as substrates to generate a visible signal indicating the presence of the protein. Note that gill and kidney cells were analyzed using western blots, and the enzyme assay was not performed on these cells.
Antioxidant assay in liver cells
GST activity was assessed by measuring the increase in absorption at 340 nm in 100 mM Na-phosphate buffer (pH 6.5) containing 1 mM 1-chloro-2,4-dinitrobenzene (CDNB, Sigma-Aldrich) and 1 mM GSH (Pacini et al. 2013).
GPx activity was measured at 340 nm using 100 mM Na-phosphate buffer (pH 7.5), 1 mM EDTA, 0.12 mM NADPH, 2 mM GSH, 1 mM NaN3, 1 U glutathione reductase (GR), and 0.6 mM H2O2 (ZellBio GmbH, Germany).
TAC levels were determined using an ELISA kit (ZellBio GmbH, Germany) at 490 nm, similar to GPx (Li et al. 2017).
Immune responses in liver cells
IgM was measured using an ELISA quantification kit (Hangzhou East Biopharm Co. Ltd.) (Valipour et al. 2018). IgM standards and supernatant samples were analyzed manually. The wavelength at 450 nm was determined within 15 min after all the necessary steps were completed. Three measurements were performed.
Quantitative C3 levels were determined using an ELISA sandwich (enzyme-linked immunosorbent assay) with a fish ELISA kit (Hangzhou East Biopharm Co. Ltd.) (Baharloei et al. 2020). The C3 fish monoclonal antibody was previously coated with an antibody-enzyme monoclonal well. The biotin-labeled C3 antibody was mixed with streptavidin–horseradish peroxidase and incubated at 37°C to form an immune complex. To remove the unmixed enzyme, the plate was washed. The liquid color changed to yellow when sulfuric acid was added. The OD at 450 nm was measured with a microplate reader, and the C3 concentration was determined.
LYZ activity was assessed using a turbidimetric method (Kim et al. 2019). Micrococcus lysodeikticus (Sigma‒Aldrich) was used as a substrate (0.2 mg/ml 0.05 M phosphate buffer, pH 6.6). In this study, lyophilized chicken egg white LYZ was used to construct a standard curve, and changes in turbidity at 530 nm were measured. Chicken egg white LYZ activity was determined from the results.
Cortisol assay
An enzyme-linked immunosorbent assay (ELISA) kit (Nanjing Jiancheng Institute, Nanjing, China) was used to measure cortisol levels in liver cells. Color changes were detected using a spectrophotometric spectrophotometer (450 nm) (Long et al. 2019) (SI section 1-3).
Statistical analysis
For the statistical analysis, SPSS/PC + 23 (SPSS Inc.) and GraphPad Prism 8 were used. All experiments were repeated three times, and the numerical data are presented as the mean ± SEM. The first step was to check and confirm the normality of the data by performing a one-sample Kolmogorov‒Smirnov test. Significant differences between groups were analyzed using one-way analysis of variance (ANOVA) and Duncan's test for multiple comparisons. To evaluate the differences in the main components between the studied parameters and different temperature stress treatments, principal component analysis (PCA) was performed. Pearson correlation analysis was conducted to examine the possibility of a relationship between the biomarkers and fish cells. The significance level was set at P < 0.0001.
Results
Cell viability
Evaluation of the viability (%) of cells treated with HSPi
The liver, gill, and kidney cells were exposed individually to various concentrations of HSPi, including SZ, MZ, HN-P1, HN-P2, AMG, and NOP (Fig. 1; P < 0.0001). The concentrations of the compounds MZ, HN-P1, and HN-P2 (Fig. 1 b, c and d, respectively) strongly fluctuated in all three cell lines and did not follow a continuous increasing or decreasing trend. A sporadic change pattern was observed. These compounds were ignored in subsequent tests. SZ (at a concentration of 160 µM: 138.36% in the liver, 143.40% in the gill, and 141.88% in the kidney; Fig. 1 a) and AMG (at a concentration of 80 mM: 148% in the liver, 139.21% in the gill, and 143.66% in the kidney; Fig. 1 f) had the best survival rates in all three cell lines compared to the control group (100%). In contrast, individuals with higher NOP concentrations (at 1600 mM: 80.33% in the liver, 81.66% in the gill, and 78.29% in the kidney; Fig. 1 e) had lower survival rates (P < 0.0001). Furthermore, the optimal dose of HSPi was determined based on the cell survival results for the following concentrations of NOP: 800 mM NOP (N800), AMG: 80 mM (A80), and SZ: 80 µM (SZ80).
Overview of cell viability at different treatment doses (%) under temperature stress
We grouped the treatments as shown in Fig. 2 and assessed their survival rates (Fig. 3). The levels of cell survival differed among the treatments chosen for liver, gill, and kidney cells. The T26 treatment had a reduced effect on cell viability (%) in all three treated cell tissues (liver: 90%, gill: 89.66%, and kidney: 88.67%) compared to the control (100%; P < 0.0001). The T18 group showed a similar effect to the control (T22) group in all three cell lines. A temperature of 26°C had a decreasing effect on cell viability. Among all HSPi compounds, N800 had the lowest cell viability (liver: 86.10%, gill: 86.61%, and kidney: 82.88%). In all three cell lines, cell viability increased when the cells were first treated with AMG (A80) and then exposed to stress temperatures (T18 and T26).
The cell survival rate (%) of cells isolated from Acipenser ruthenus tissues (means ± SEM) treated with various doses and under various temperatures was investigated. A dose of 800 mM NOP (N800), 80 mM AMG (A80), and 80 µM SZ (SZ80) was selected as the optimal dose of HSPi. Additionally, the temperature stress groups (18, 22, and 26°C) were selected for the test. In the 12 treatment groups, each parameter was measured three times. Within each column, different letters indicate significantly different groups according to Duncan's test (a, b, c, and..; P < 0.0001)
HSP response to temperature stress
HSP27
The expression of the HSP27 protein in liver, gill, and kidney cells varied significantly (Fig. 4; P < 0.0001). Compared to the control group (T22), the T26 group had increased HSP27 expression in liver cells (Fig. 4 a and d; P < 0.0001). Among all the HSPi compounds, SZ80 expressed the least amount of protein (SZ80 + T22: 1.153-fold, SZ80 + T26: 1.453-fold). A80 + T26 (2.546-fold) and A80 + T22 (2.448-fold) showed the highest HSP27 expression (P < 0.0001). In the gill, the T26 group (3.036-fold) showed maximal HSP27 expression compared with the control group (Fig. 4 b and d). Treatment with N800 + T26 also had a greater effect on HSP27 expression (2.377-fold; P < 0.0001). Similar to those in liver cells, the protein expression levels in gill cells in the SZ80 treatment groups were the lowest among all the HSPi compounds (P < 0.0001). In kidney cells (Fig. 4c and d), SZ80 + T22 (0.088-fold) and N800 + T26 (2.926-fold) cells expressed less and more HSP27, respectively, than did the controls. The HSP27 protein expression in the T26 group (1.631-fold) was slightly greater than that in the control group. In cells treated with HSPi compounds, especially N800 and A80, followed by temperature stress (T26), protein expression increased (HSPi + T26; P < 0.0001).
The relative protein expression of HSP27 was analyzed by western blotting. a) Liver, b) gill, and c) kidney cells. d) Eight groups were considered: 1) control cells without treatment at 22°C (T22) and HSPi groups containing 22°C, 2) 80 mM amygdalin (A80 + T22), 3) 80 µM SZ (SZ80 + T22), 4) 800 mM Nopal Endurance (N800 + T22), and 5) cells without treatment at 26°C (T26). The HSPi groups were incubated at 22°C for 24 h, after which they were transferred to a temperature of 26°C: 6) 80 mM amygdalin (A80 + T26), 7) 80 µM SZ (SZ80 + T26), and 8) 800 mM Nopal Endurance (N800 + T26). There were 8 treatment groups in which each parameter was measured three times. The HSP27 protein levels are shown as the fold change relative to the control values (means ± SEM), and within each column, different letters indicate significantly different groups according to Duncan's test (a, b, c, and…; P < 0.0001)
HSP70
Under temperature stress conditions (T26), HSP70 expression was evaluated in sterlet sturgeon liver cells (Fig. 5a and d), gills (Fig. 5b and d), and kidneys (Fig. 5c and d). The changes in the expression of the HSP70 protein in all three cell lines were almost the same. The N800 + T26 group showed the greatest changes in HSP70 protein expression in liver, gill, and kidney cells. Compared with that of the other inducing compounds, the protein expression of the synthetic compounds (SZ80 + T22 and SZ80 + T26) was the lowest. HSP70 protein expression was slightly greater under stress conditions (group T26) than under the control conditions (P < 0.0001). In general, all three cell lines showed an increase in HSP70 expression when first treated with inducing compounds (especially N800) and then exposed to temperature stress (P < 0.0001).
The relative protein expression of HSP70 was analyzed by western blotting. a) Liver, b) gill, and c) kidney cells. d) Eight groups were considered: 1) control cells without treatment at 22°C (T22) and HSPi groups containing 22°C, 2) 80 mM amygdalin (A80 + T22), 3) 80 µM SZ (SZ80 + T22), 4) 800 mM Nopal Endurance (N800 + T22), and 5) cells without treatment at 26°C (T26). The HSPi groups were incubated at 22°C for 24 h, after which they were transferred to a temperature of 26°C: 6) 80 mM amygdalin (A80 + T26), 7) 80 µM SZ (SZ80 + T26), and 8) 800 mM Nopal Endurance (N800 + T26). There were 8 treatment groups in which each parameter was measured three times. The HSP70 protein levels are shown as the fold change relative to the control values (means ± SEM), and within each column, different letters indicate significantly different groups using Duncan's test (a, b, c, and…; P < 0.0001)
HSP90
HSP90 protein expression was altered by HSPi and temperature treatments in liver, gill, and kidney cells (Fig. 6; P < 0.0001). In the liver, the highest HSP90 expression was observed in the A80 + T26 treatment group (2.553-fold), while the control group had the lowest HSP90 expression (Fig. 6a and d). Compared to those in the T26 treatment group, the HSP90 expression in the A80 + T26 and N800 + T26 treatment groups significantly increased (P < 0.0001). The liver cells expressed the highest concentration of the HSP90 protein as a result of AMG (A80). In the gills, the N800 + T26 treatment group (5.536-fold) had higher levels of HSP90 expression than did the control group, while SZ80 (SZ80 + T22 and SZ80 + T26) had the lowest protein expression (Fig. 6b and d). Compared with those in the T26 group, the groups that received the inducing compounds first and then underwent temperature stress showed significantly greater HSP90 expression (P < 0.0001). In the kidney, the N800 + T26 treatment group (2.870-fold) had higher levels of HSP90 expression, while the SZ80 + T22 treatment group (0.861-fold) had lower levels (Fig. 6c and d). The T26 group had increased HSP90 expression in kidney cells compared to that in the control group (T22). HSP90 expression was significantly greater in N800 + T26 (2.870-fold) than in A80 + T26 (2.156-fold) and SZ80 + T26 (1.742; P < 0.0001). Note that original photos of the western blot gel are available in SI Figs. 5, 6, 7 and 8.
The relative expression of HSP90 was analyzed via western blotting. a) Liver, b) gill, and c) kidney of sterlet cells. d) Eight groups were considered: 1) control cells without treatment at 22°C (T22) and HSPi groups containing 22°C, 2) 80 mM amygdalin (A80 + T22), 3) 80 µM SZ (SZ80 + T22), 4) 800 mM Nopal Endurance (N800 + T22), and 5) cells without treatment at 26°C (T26). The HSPi groups were incubated at 22°C for 24 h, after which they were transferred to a temperature of 26°C: 6) 80 mM amygdalin (A80 + T26), 7) 80 µM SZ (SZ80 + T26), and 8) 800 mM Nopal Endurance (N800 + T26). In eight treatment groups, each parameter was measured three times. The HSP90 protein levels are shown as the fold change relative to the control values (means ± SEM), and within each column, different letters indicate significantly different groups according to Duncan's test (a, b, c, and…; P < 0.0001)
Antioxidant activity
Liver cell antioxidant activity was assessed using GST, GPx, and TAC measurements (Table 1; P < 0.0001). The changes in all the aforementioned enzymes were similar. Enzyme activity significantly increased at stress temperatures (T18 and T26) compared to that at the control temperature (T22). The N800 + T26 group exhibited the highest activity of all three enzymes. Among all the HSPi compounds, SZ80 had the lowest activity (SZ80 + T22 < SZ80 + T18 < SZ80 + T26). Overall, the enzyme activity was greater in the treatment groups at 26°C than at 18°C (HSPi + T26 ˃ HSPi + T18 ˃ HSPi + T22).
Immune responses
Table 2 shows the measured activities of C3, IgM, and LYZ in liver cells exposed to different treatments (P < 0.0001). The control group had an apparent increase in C3 activity (1.58 mg/g), while the other treatment groups showed a clear decrease in activity. C3 levels were greater at 18°C than at 26°C, and treatment groups at 22°C had higher C3 levels than those at 18°C (the order of changes in the groups was treatments + T22 > treatments + T18 > treatments + T26; P < 0.0001). Similarly, the highest level of IgM was observed in the control group (2.7 mg/g), and the other treatment groups had a decreasing effect. Changes in groups T18 and T26 were similar. Among the HSPi compounds, SZ80 had the highest level (P < 0.0001). LYZ activity in groups T18 and T22 was the same (0.196 mg/g), while it decreased in the other groups. The LYZ levels in the treatment groups were greater at 18°C than at 26°C, and the LYZ levels were greater at 22°C than at 18°C (the order of differences between the groups was groups + T22 > groups + T18 > groups + T26; P < 0.0001).
Assessing PCA
PCA was conducted on 20 variables and 36 treatments, revealing that the first, second, and third principal components accounted for 59.20%, 20.26%, and 7.09% of the changes, respectively (Table 3). Variables with a score higher than 0.6 were used to interpret the components (SI Fig. 10, Tables 1 and 2). The variables measured during temperature stress played a significant role in determining the relationships between the treatments and the tested variables in the first and second components (Fig. 7). Finally, Pearson’s correlation analysis was performed on all measured parameters (SI Table 3).
Principal component analysis (PCA) for temperature stress. The three components accounted for 86.56% of the total variation. The variables measured during temperature stress played a significant role in determining the relationships between the treatments and the tested variables in the first and second components. The abbreviations are L: liver, G: gill, K: kidney, LV: liver viability, GV: gill viability, and KV: kidney viability
Discussion
Sturgeon in the Caspian Sea has been cultivated artificially in recent years due to extinction conditions (Baharloei et al. 2021). The Sefidroud River, which flows into the Caspian Sea, is one of the fish release stations in Guilan Province (Hajirezaee et al. 2017). The water temperature of this river has fluctuated in different seasons in recent years due to the increase in the Earth's temperature and decrease in rainfall (Abdullah et al. 2022; Khoshakhlagh et al. 2016). On the other hand, due to its ecological conditions, the temperature of the Caspian Sea has increased by approximately 8°C in the last 40 years (Leroy et al. 2020; Zakerinejad et al. 2022). Sturgeons are anadromous fish that migrate to rivers to spawn. During this migration between the river and the sea, the fish encounter water temperature changes, which are considered stressors (Afraei Bandpei et al. 2010; Khodabandeh et al. 2009; Prokopchuk et al. 2016). Additionally, when sturgeons migrate from fresh to saltwater, their blood plasma ion levels increase, causing severe stress (Khodabandeh et al. 2009; Kim et al. 2021). Different fish species react differently to climate change and environmental changes. Some species may adapt to changing conditions, while others struggle to survive. It is imperative to understand these differences to develop effective conservation strategies (Carosi et al. 2019).
HSP27, HSP70, and HSP90 are proteins that act as cellular chaperones and may be involved in environmental factors (Deane and Woo 2011). Temperature stimulates the upregulation of HSP proteins, particularly HSP27, HSP70, and HSP90. As a result, this upregulation is caused by cellular stress induced by temperature fluctuations (Ji et al. 2016; Mohanty et al. 2018; Wang et al. 2007). In fish and shellfish, HSP inducers play a significant role in stress responses (Baharloei et al. 2020; Baruah et al. 2014). Environmental factors can negatively affect fish health by generating reactive oxygen species (ROS), which disrupt the immune system (Eissa et al. 2017). Fish health can be determined by studying safety parameters (Banaee et al. 2013). Moreover, antioxidant parameters can indicate how fish respond to environmental changes (Hattori et al. 2009). By investigating the effects of stress factors on fish environments, these changes provide an effective tool for managing fish health (Werner et al. 2007).
An ecological model of Acipenser ruthenus was used to examine liver, kidney, and gill cell survival. It was hypothesized that inducing HSPs in sterlet cells could increase protection and readiness to cope with temperature stress in vitro. First, MTT assays were used to investigate cytotoxicity (Fig. 1). Temperature negatively affected sterlet cell viability (Fig. 3). Additionally, temperature treatment altered HSP27, HSP70, and HSP90 expression in all three cell lines (Figs. 4, 5 and 6). As a consequence, temperature stress increased the oxidative activity of GST, TAC, and GPx enzymes (Table 1). The immune parameters IgM, LYZ, and C3 were also reduced (Table 2).
Despite our initial findings about temperature stress and its effects on cells, we encountered significant challenges in the continuation of our study. We discovered that HSPs can cause similar alterations without reducing cell viability or increasing cell death. Furthermore, normal conditions can be restored by combining the HSP and temperature. To investigate whether HSPi could counteract the temperature effects on A. ruthenus cells, we tested NOP-, AMG-, and pirano piranazole-derived compounds (SZ, MZ, HN-P1, and HN-P2; SI Figs. 1 to 4). We examined the viability of cells after applying the compounds mentioned above to better understand this challenge. MTT assays revealed that AMG and SZ increased cell viability, while NOP decreased cell viability (Fig. 1). Furthermore, cells treated with the inducing compounds and then exposed to temperature showed increased viability (Fig. 3).
Fish farming is particularly important for the preservation of endangered fish populations. This can be achieved by bringing them to special farms and releasing them into freshwater once they reach maturity. However, after being raised on farms under optimal conditions, the fish are released into water that differs from the water they were raised in, which can be a disadvantage. By using HSPis for the first aspect, this problem may be alleviated. For the second aspect of the challenge, reducing or eliminating temperature stress, combining temperature with HSPis is essential (Afraei Bandpei et al. 2010; Salmani et al. 2024; Vahdatiraad et al. 2023b; Vahdatiraad et al. 2023a). In all three cell lines (Figs. 4, 5 and 6), AMG and NOP regulate all HSPs (HSP27, HSP70, and HSP90) and increase their expression (HSPi + stress).
In aquatic organisms, temperature increases affect parameters such as metabolic rate and oxygen consumption, causing oxidative stress (Madeira et al. 2013). Thus, the induction of antioxidant defenses is an important component of the stress response to oxidative stress in biological systems (Han et al. 2018; Lushchak and Bagnyukova 2006). Our results are generally in accordance with previous findings, where oxidative stress biomarkers (both degradation products and antioxidants) are highly sensitive to temperature due to temperature-induced ROS production. Salmani et al. showed that the use of an HSP inducer in combination with temperature exposure resulted in increased activity of superoxide dismutase, catalase, and TAC enzymes in sturgeon fingerlings. The increase in the activity of these enzymes, particularly on the first day, indicated the activation of antioxidant defense mechanisms in response to changes in temperature-induced oxidative stress (Salmani et al. 2024). HSPs, including HSP70, have been shown to have antioxidant properties and can help protect cells from oxidative damage (Eissa et al. 2017; Lu et al. 2022). Therefore, it is reasonable to speculate that HSPs may serve as a protective mechanism against the oxidative stress induced by changes in temperature, particularly during the initial days. Overall, our findings suggest that the use of HSP-inducing compounds in combination with a temperature of 26°C (HSPi + 26°C ˃ HSPi + 18°C) improves antioxidant enzyme activity (Table 1). In the cells of sterlet sturgeon subjected to environmental stress, ROS can be controlled, and antioxidant defense mechanisms can be enhanced, potentially reducing the effects of temperature stress and environmental stressors on aquatic organisms. Salmonids and tilapia exhibit species-specific immune responses to temperature changes (Bailey et al. 2017; Ndong et al. 2007). In another study, the presence of HSPi was found to moderate immune activity in sturgeon fish (Vahdatiraad et al. 2023a). Overall, there was a decreasing trend between the treatment and control groups with regard to the immune response (C3, IgM, and LYZ) according to the present study. Cells treated with NOP and AMG first and then subjected to temperature stress produced the best results (Table 2). Based on PCA and correlation analysis, HSP expression in different tissues was related to the most studied parameters (Table 3, SI Tables 1 to 3). As a result, this family plays a significant role in a variety of biological states. Using PCA, we identified the appropriate treatment for temperature stress. This study indicated that HSP expression is directly related to antioxidant parameters (GST and GPx) and the immune response (IgM; Fig. 7). Thus, NOP and AMG could improve temperature stress and restore normal cell function.
Conclusions
Using cells isolated from Acipenser ruthenus sturgeons, we investigated the modulatory effects of HSP inducers on temperature stress. In vitro, HSP inducers are capable of modifying HSP expression, cortisol levels, and antioxidant activity. As a result, the use of HSP inducers may be a powerful and reliable method for increasing A. ruthenus resistance to temperature stress. However, our results also suggest that these compounds can reverse the harmful effects of temperature stress. Further research is necessary to investigate the effects of these ecological factors, especially in vivo and in combination with other stresses, on the health status of species in real-life situations to effectively implement this approach.
Data availability
On request, the data will be provided by the corresponding authors.
References
Abdullah AHJ, Abdullah SA, Ziyadi MSF, Al-Faiz NA (2022) Investigation of changes in the fish assemblage building and abundance in the Garmat Ali River, Southern Iraq. J Fish Taxon 25:31–40
Ahmadi K, Mirvaghefei AR, Banaee M, Vosoghei AR (2014) Effects of long-term diazinon exposure on some immunological and haematological parameters in rainbow Trout Oncorhynchus mykiss (Walbaum, 1792). Toxicol Environ Heal Sci 6(1):1–7
Ahmed K et al (2012) Chemical inducers of heat shock proteins derived from medicinal plants and cytoprotective genes response. Int J Hyperth 28(1):1–8
Baharloei M, Heidari B, Zamani H, Hadavi M (2020) Effects of Pro-Tex® on the expression of Hsp70 gene and immune response parameters in the Persian sturgeon fingerlings, Acipenser persicus, infected with Aeromonas hydrophila. J Appl Ichthyol 36(4):393–401
Baharloei M et al (2021) Effects of heat shock protein inducer on Hsp70 gene expression and immune parameters during Streptococcus iniae infection in a Persian sturgeon fry. Vet Res Forum 12(4):473–479
Bailey C, Segner H, Casanova-Nakayama A, Wahli T (2017) Who needs the hotspot? The effect of temperature on the fish host immune response to Tetracapsuloides bryosalmonae the causative agent of proliferative kidney disease. Fish Shellfish Immunol 63:424–437
Banaee M, Sureda A, Mirvaghefi AR, Ahmadi K (2013) Biochemical and histological changes in the liver tissue of rainbow trout (Oncorhynchus mykiss) exposed to sub-lethal concentrations of diazinon. Fish Physiol Biochem 39(3):489–501
Bandpei A, Ali M, Mashhor M, Abdolmaleki S, El-Sayed AFM (2010) Population dynamics of Caspian Kutum, Rutilus frisii kutum (Cyprinidae) in Southern Caspian Sea, Iran. Cybium 34(3):285–292
Baruah K et al (2014) Reactive oxygen species generated by a heat shock protein (Hsp) inducing product contributes to Hsp70 production and Hsp70-mediated protective immunity in Artemia franciscana against pathogenic vibrios. Dev Comp Immunol 46(2):470–479
Biller-Takahashi JD, Urbinati EC (2014) Fish Immunology. The modification and manipulation of the innate immune system: Brazilian studies. An Acad Bras Ciencias 86(3):1483–1495
Birnie-Gauvin K et al (2019) Cortisol predicts migration timing and success in both Atlantic salmon and sea trout kelts. Sci Rep 9(1):2422
Boshoff A et al (2004) Molecular chaperones in biology, medicine and protein biotechnology. S Afr J Sci 100(11–12):665–677
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254
Brough PA et al (2005) 3-(5-Chloro-2,4-Dihydroxyphenyl)-Pyrazole-4-carboxamides as inhibitors of the Hsp90 molecular chaperone. Bioorg Med Chem Lett 15(23):5197–5201
Butler M (2004) Animal Cell Culture and Technology Animal Cell Culture and Technology. Taylor & Francis
Carosi A, Padula R, Ghetti L, Lorenzoni M (2019) Endemic freshwater fish range shifts related to global climate changes: A long-term study provides some observational evidence for the Mediterranean area. Water (Switzerland) 11(11):2349
Cho HC, Kim JE, Kim HB, Baek HJ (2015) Effects of water temperature change on the hematological responses and plasma cortisol levels in growing of red spotted grouper, Epinephelus skaara. Dev Reproduciton 19(1):19–24
Deane EE, Woo NYS (2011) Advances and perspectives on the regulation and expression of piscine heat shock proteins. Rev Fish Biol Fisheries 21(2):153–185
Dominguez M, Takemura A, Tsuchiya M (2005) Effects of changes in environmental factors on the non-specific immune response of Nile tilapia, Oreochromis niloticus L. Aquac Res 36(4):391–397
Eissa N et al (2017) Expression of Hsp 70, Igf1, and three oxidative stress biomarkers in response to handling and salt treatment at different water temperatures in yellow perch, Perca flavescens. Front Physiol 8(SEP):683
Filho DW (2007) Reactive oxygen species, antioxidants and fish mitochondria. Front Biosci 12(4):1229–1237
Ghafoori H, Rezaei M, Mohammadi A (2017) Anti-inflammatory effects of novel thiazolidinone derivatives as bioactive heterocycles on Raw264.7 cells. Iran J Allergy Asthma Immunol 16(1):28–38
Hajirezaee S, Mirvaghefi A, Farahmand H, Agh N (2017) NMR-based metabolomic study on the toxicological effects of pesticide, diazinon on adaptation to sea water by endangered Persian sturgeon, Acipenser persicus fingerlings. Chemosphere 185:213–226
Han SY et al (2018) A comparative study on oxidative stress response in the hepatopancreas and midgut of the white shrimp litopenaeus vannamei under gradual changes to low or high pH environment. Fish Shellfish Immunol 76:27–34
Hattori RS et al (2009) Cortisol-induced masculinization: does thermal stress affect gonadal fate in pejerrey, a teleost fish with temperature-dependent sex determination? PLoS ONE 4(8):e6548
Hu C et al (2022) Heat shock proteins: Biological functions, pathological roles, and therapeutic opportunities. MedComm 3(3):e161
Islam MJ, Kunzmann A, Slater MJ (2022) Responses of aquaculture fish to climate change-induced extreme temperatures: A review. J World Aquacult Soc 53(2):314–366
Ji L et al (2016) Low temperature stress on the hematological parameters and HSP gene expression in the Turbot Scophthalmus maximus. Chin J Oceanol Limnol 34(3):430–440
Khodabandeh S, Mosafer S, Khoshnood Z (2009) Efectos de la aclimatización Al cortisol y a la Salinidad En El Na+/K+/2Cl?- cotransportador de expresión génica y en la actividad Na+, K+-ATPasa en las Branquias de juveniles del esturión de Persia. Sci Mar 73(SUPPL. 1):111–116
Khoshakhlagh F et al (2016) Trend of the Caspian Sea surface temperature changes. Nat Environ Change 2(1):57–66
Kim JH, Kim SK, Hur YB (2019) Temperature-mediated changes in stress responses, acetylcholinesterase, and immune responses of juvenile olive flounder Paralichthys olivaceus in a bio-floc environment. Aquaculture 506(March):453–458
Kim JH et al (2021) Salinity-mediated changes in hematological parameters, stress, antioxidant responses, and acetylcholinesterase of juvenile olive flounders (Paralichthys olivaceus). Environ Toxicol Pharmacol 83:103597
Klein RD et al (2017) Effects of increasing temperature on antioxidant defense system and oxidative stress parameters in the Antarctic fish Notothenia coriiceps and Notothenia rossii. J Therm Biol 68:110–118
Küçükgüzel G, ŞenkardeŞ S (2015) Recent advances in bioactive pyrazoles. Eur J Med Chem 97(1):786–815
Lakra WS, Raja Swaminathan T, Joy KP (2011) Development, characterization, conservation and storage of fish cell lines: A review. Fish Physiol Biochem 37(1):1–20
Lenhardt M, Jaric I, Kalauzi A, Cvijanovic G (2006) Assessment of extinction risk and reasons for decline in sturgeon. Biodivers Conserv 15(6):1967–1976
Leroy SAG et al (2020) Past and current changes in the largest lake of the world: the caspian sea. Springer Water, pp 65–107
Li Q et al (2017) Effects of dietary lipid sources on growth performance, lipid metabolism and antioxidant status of juvenile Russian sturgeon Acipenser gueldenstaedtii. Aquac Nutr 23(3):500–510
Lin LY, Weng CF, Hwang PP (2000) Effects of cortisol and salinity challenge on water balance in developing larvae of tilapia (Oreochromis mossambicus). Physiol Biochem Zool 73(3):283–289
Long L et al (2019) Effects of stocking density on growth, stress, and immune responses of juvenile chinese sturgeon (Acipenser sinensis) in a recirculating aquaculture system. Comp Biochem Physiol - C: Toxicol Pharmacol 219:25–34
Lu M, Maoliang Su, Liu N, Zhang J (2022) Effects of environmental salinity on the immune response of the coastal fish Scatophagus argus during bacterial infection. Fish Shellfish Immunol 124:401–410
Lushchak VI, Bagnyukova TV (2006) Temperature increase results in oxidative stress in goldfish tissues. 2. Antioxidant and associated enzymes. Comp Biochem Physiol - C Toxicol Pharmacol 143(1):36–41
Madeira D et al (2013) Influence of temperature in thermal and oxidative stress responses in estuarine fish. Comp Biochem Physiol - Mol Integr Physiol 166(2):237–243
Mahanty A, Mohanty S, Mohanty BP (2017) Dietary supplementation of curcumin augments heat stress tolerance through upregulation of nrf-2-mediated antioxidative enzymes and hsps in puntius sophore. Fish Physiol Biochem 43(4):1131–1141
Moghim M, Kor D, Tavakolieshkalak M, Khoshghalb MB (2006) Stock status of Persian Sturgeon (Acipenser Persicus Borodin, 1897) along the Iranian Coast of the Caspian Sea. J Appl Ichthyol 22(SUPPL. 1):99–107
Mohanty BP et al (2018) Heat shock proteins in stress in teleosts. Regulation of heat shock protein responses, pp 71–94
Mozanzadeh MT et al (2021) The effect of salinity on growth performance, digestive and antioxidant enzymes, humoral immunity and stress indices in two euryhaline fish species: Yellowfin seabream (Acanthopagrus latus) and Asian seabass (Lates calcarifer). Aquaculture 534:736329
Ndong D et al (2007) The immune response of tilapia Oreochromis mossambicus and its susceptibility to Streptococcus iniae under stress in low and high temperatures. Fish Shellfish Immunol 22(6):686–694
Neamatallah WA et al (2022) 2, 3-Dimethylsuccinic acid and fulvic acid attenuate lead-induced oxidative misbalance in brain tissues of Nile tilapia Oreochromis niloticus. Environ Sci Pollut Res 29(15):21998–22011
Otaka M et al (2007) The induction mechanism of the molecular chaperone HSP70 in the gastric mucosa by Geranylgeranylacetone (HSP-inducer). Biochem Biophys Res Commun 353(2):399–404
Pacini N et al (2013) Antioxidant response versus selenium accumulation in the liver and kidney of the Siberian sturgeon (Acipenser baeri). Chemosphere 93(10):2405–2412
Pirali M et al (2020) Artesunate, as a HSP70 ATPase activity inhibitor, induces apoptosis in breast cancer cells. Int J Biol Macromol 164:3369–3375
Pourmousavi S, Heidari G, Manoochehri H, Sharifi E (2022) Poly (aniline-co-melamine)@ MnFe2O4 nanocatalyst for the synthesis of 4, 4′-(arylmethylene) bis (1H-pyrazole-5-Ol) derivatives, and 1, 4-dihydropyrano [2, 3-c] pyrazoles and evaluation of their antioxidant, and anticancer activities. Front Chem 10
Prokopchuk G, Dzyuba B, Rodina M, Cosson J (2016) Control of sturgeon sperm motility: Antagonism between K+ ions concentration and osmolality. Anim Reprod Sci 164:82–89
Salmani E, Heidari B, Vahdatiraad L, Zarei S (2024) Protection against thermal stress: the role of tex-OE® in inducing HSP70 and boosting defense mechanisms in acipenser stellatus fry. Aquacult Int 1–19
Shahriyari-Nejad H et al (2020) Synthesis of pirano-based piranazole-based compounds to induce apoptosis by reducing the expression of anti-apoptotic protein B2 cell lymphoma protein in the MCF-7 human breast cancer cell category. Armaghane Danesh 25(1):40–54
Sharma JG, Singh SP, Chakrabarti R (2017) Effect of temperature on digestive physiology, immune-modulatory parameters, and expression level of Hsp and LDH genes in Catla catla (Hamilton, 1822). Aquaculture 479:134–141
Stanic B et al (2006) Assessing pollution in the Danube River near Novi Sad (Serbia) using several biomarkers in sterlet (Acipenser Ruthenus L.). Ecotoxicol Environ Saf 65(3):395–402
Stapp LS, Kreiss CM, Pörtner HO, Lannig G (2015) Differential impacts of elevated CO2 and acidosis on the energy budget of gill and liver cells from Atlantic cod, Gadus morhua. Comp Biochem Physiol - A : Mol Integr Physiol 187:160–167
Taheri M et al (2022) Synthesis, in vitro biological evaluation and molecular modelling of new 2-chloro-3-hydrazinopyrazine derivatives as potent acetylcholinesterase inhibitors on PC12 cells. BMC Chem 16(1):1–12
Tresguerres M et al (2020) Evolutionary links between intra- and extracellular acid-base regulation in fish and other aquatic animals. J Exp Zool A: Ecol Integr Physiol 333(6):449–465
Vahdatiraad L et al (2023a) Biological responses of stellate sturgeon fingerlings (Acipenser stellatus) immersed in HSP inducer to salinity changes. Mar Environ Res 191:106145
Vahdatiraad L et al (2023b) Protective effects of HSP Inducer on diazinon-exposed stellate sturgeon (Acipenser stellatus) fry: Insights on HSP70 gene expression, immune response, and enzyme indices. PLoS ONE 18(11):e0294188
Valipour A, Heidari B, Hadavi M, Yousefi A (2018) Changes in immune parameters (Lysozyme, IgM, C3) in early life stages and broodstock of Siberian sturgeon, Acipenser baerii. Fish Aquat Life 26(1):21–30
Wang J et al (2007) The identification of heat shock protein genes in goldfish (Carassius auratus) and their expression in a complex environment in Gaobeidian Lake, Beijing, China. Comp Biochem Physiol - C Toxicol Pharmacol 145(3):350–362
Werner I, Linares-Casenave J, Van Eenennaam JP, Doroshov SI (2007) The effect of temperature stress on development and heat-shock protein expression in larval green sturgeon (Acipenser mirostris). Environ Biol Fishes 79(3–4):191–200
Wiles SC et al (2020) Long-term pharmaceutical contamination and temperature stress disrupt fish behavior. Environ Sci Technol 54(13):8072–8082
Yazdanpanah Dero Q, Yari E, Charrahy Z (2020) Global warming, environmental security and its geo-economic dimensions case study: Caspian Sea level changes on the balance of transit channels. J Environ Health Sci Eng 18(2):541–57
Yu M et al (2022) HSP27–HSP40–HSP70–HSP90 pathway participated in molecular mechanism of selenium alleviating lead-caused oxidative damage and proteotoxicity in chicken bursa of fabricius. Anim Biotechnol 1–12
Zakerinejad R, Movahedi S, Jahanian E (2022) Investigation of Sea Surface temperature changes in the Oman Sea and the Persian gulf using satellite images and its comparison with the Caspian Sea Trend. Spatial Planning 12(4):65–80
Zarei S, Ghafouri H, Vahdatiraad L, Heidari B et al (2024a) Enhancing resistance and cell survival in Acipenser ruthenus liver, gill, and kidney cells: The potential of heat shock protein inducers against PAH-benzo[a]pyrene stress. Environ Sci Pollut Res 31(6):9445–9460
Zarei S, Ghafouri H, Vahdatiraad L, Moghaddam VA et al (2024b) Using heat shock protein (HSP) inducers as an approach to increase the viability of sterlet (Pisces; Acipenseridae; Acipenser ruthenus) cells against environmental diazinon toxicity. J Hazard Mater 465:133194
Acknowledgements
We would like to thank the Shahid Beheshti Sturgeon Breeding and Rearing Center (Rasht, Iran) for providing the fish for our project.
Funding
This work was supported by the Iranian Biological Resource Center (IBRC, Grant No. 961113100001) and the Student Affairs Organization of the Ministry of Science, Research and Technology (Grant No. 3913929).
Author information
Authors and Affiliations
Contributions
S.Z: Performing the experiments, Writing –original draft, Statistical Analysis. H.Gh: Conceptualization, Supervision, Methodology, Writing – review &; editing. L.V: Performing the experiments. T.S: Providing the sample and advice in the fish physiology. B.H: Methodology, Advisor, Writing – review &; editing.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The authors declare that they have no known competing financial interests or personal relationships that seem to affect the work reported in this article. We declare that we have no human participants, human data, or human tissues.
Consent for publication
All of the authors have read and approved the paper for publication.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zarei, S., Ghafoori, H., Vahdatiraad, L. et al. Effects of HSP inducers on the gene expression of Heat Shock Proteins (HSPs) in cells extracted from sterlet sturgeon under temperature stress with antioxidant and immunity responses. Fish Physiol Biochem 50, 1409–1428 (2024). https://doi.org/10.1007/s10695-024-01347-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-024-01347-0