Abstract
Reproductive patterns observed in organisms are direct outcomes of the interaction among parameters such as phylogeny, body size, and environmental characteristics. However, the action of these parameters is rarely observed in an integrated perspective in the literature. Here, we collected 109 specimens of two congeneric species of treefrogs living in the same microhabitat (bromeligenous species of genus Ololygon) to compare the reproductive patterns of the two species, evaluating the effect of different parameters on the reproductive ecology of the organisms. Our results show that morphometric measurements between females of the two species were not significantly different, indicating similar body sizes. The species exhibited different degrees of sexual dimorphism, and interspecifically, females showed significant differences in breeding traits, with the species O. perpusilla demonstrating higher reproductive effort, characterized by increased ovarian mass and increased average egg size compared to O. littorea. We observed that differences in reproductive effort were strongly associated with egg size, which drove higher reproductive investment in O. perpusilla females. We conclude that although the species share many traits commonly related to the degree of reproductive investment, other drivers, not yet completely understood, may influence the reproductive aspects of organisms, generating unexpected patterns.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In anurans, typically, females tend to be larger than males. This pattern of sexual size dimorphism (SSD) is proposed as a consequence of the increase in fecundity that females undergo with increasing body size, promoting larger body sizes in females, when compared to males (Darwin 1874; Shine 1989). The amount of energy invested in reproduction is directly related to offspring survival capacity (see Dziminski and Roberts 2006 for details) and females may invest in reproduction through different modes of partitioning the energy allocated for reproduction, which can vary in quantity (i.e., fecundity) and size (i.e., quality) of the produced eggs (Dziminski et al. 2009; Pupin et al. 2018; Dugas et al. 2023; Justicia et al. 2023). Generally, the reproductive effort (RE) in females is expected to be positively related to female body size, and this relationship has been seen in all anurans reproducing in aquatic habitats (Nali et al. 2014; Silva et al. 2020). However, females of different species may present differences in patterns of RE as consequence of changes in body size-RE relationships (i.e., reproductive allometry) as well as in magnitude of RE, when variation is not explained by body size (Reiss 1989; Camargo et al. 2008).
Anurans exhibit diverse reproductive strategies (Nunes-de-Almeida et al. 2021). These different reproductive modes are intricately related to specific behaviors and evolutionary trade-offs. Among neotropical amphibians, one reproductive mode stands out: the intimate association between tadpoles and plants belonging to the Bromeliaceae family. Anurans that depend on bromeliads to reproduce are termed bromeligenous (Peixoto 1995). Many bromeliads present water-retaining structures, creating a favorable environment for tadpole development even in minimal water volumes, and the reproduction can occur throughout the year (e.g., Lantyer-Silva et al. 2014; Ferreira et al. 2019). Both our study species are prolonged breeders, and reproductive activity is observed during all year after rain events (Alves-Silva and Silva 2009). Remarkably, this bromeligenous behavior appears to have evolved independently across various families of anurans (Tonini et al. 2020). However, due to the restrictions that the bromeliad microhabitat imposes (e.g. water volume, high temperatures), it is plausible that reproductive traits exhibit similarities among different species of bromeligenous frogs. On the other hand, there are unresolved questions regarding breeding traits of anurans. For instance, while small eggs are expected in warm habitats, frogs that lay their eggs in small bodies of water, such as bromeliads, produce larger eggs compared to closely related species that breed in larger bodies of water, because these sites typically offer limited food for developing larvae (Wells 2007). An alternative to overcome spatial and nutritional limitations in the bromelias microhabitat may be the behavior of spawning in a partitioned manner between different tanks of the same bromeliad or even neighboring bromeliads (Alves-Silva and Silva 2009). Despite spatial limitations, bromeliads are capable of retaining water for longer periods than other aquatic environments with similar volume, being a more temporally stable reproductive site (Cogliatti-Carvalho et al. 2010; Barberis et al. 2023).
In the present work we investigated how reproductive traits are conserved or differentiated between congeneric species that inhabit bromeliads in two sites. We also investigated potential differences in these traits between seasons as well as the sexual size dimorphism. Specifically, we hypothesized there is sexual dimorphism seen on body size between females and males in an intraspecific perspective (H1), and we expected females would be greater than males. We supposed that females of Ololygon perpusilla and Ololygon littorea do not have difference in body size and breeding traits (H2), in view of its that share the same microhabitats conditions (bromeliad) and are very closely phylogenetic relationship (same genus). Then, we also supposed that its traits would not undergo seasonal variations (H3) due stable abiotic conditions in bromeliads. Finally, we hypothesized that females show the same reproductive allometry (i.e., allocation pattern) and magnitude in reproductive effort (H4).
Materials and methods
Study area and species models
We conducted this research at two sites in Rio de Janeiro State - Brazil, that contain the bromeliads Alcantarea glaziouana. The first area encompasses the Natural Monument of Morro da Urca and Sugar Loaf (22°57’S, 43°09’W) in the municipality of Rio de Janeiro. The second area is located at Costão de Itacoatiara (22°58’S, 43°01’W) within the State Park of Serra da Tiririca, spanning the municipalities of Niterói and Maricá (Fig. 1). These two areas are coastal Inselbergs with similar structural composition, origin, and age of formation (Meirelles et al. 1999). The vegetation in these both areas is primarily rupicolous dominated by bromeliads.
Both species of treefrog are endemic to the Atlantic Rain Forest biome and live on coastal inselbergs. The life cycles of Ololygon species are completed in association with plants of the family Bromeliaceae (known as bromeligenous behaviour). The larvae of these species develop in the water stored in the bromeliads, and all Ololygon “perpusilla group” species have similar morphology (i.e., body size and shape) and behaviour (Peixoto 1987; Alves-Silva and Silva 2009), and they are, in fact, probably monophyletic (Alves-Silva and Silva 2009). The tadpoles of both bromeligenous Ololygon species are found during the rainy and dry seasons. They are exotrophic and known to feed on detritus and aquatic organisms in proportion to their abundances (Sabagh et al. 2012).
Field sampling and laboratory procedures
We sampled anurans between the years 2008 to 2010 in two dry seasons (July-August) and two rainy seasons (January-February). Each Ololygon species was found in distinct Inselberg environments: O. perpusilla was identified at the Natural Monument of Morro da Urca and Sugar Loaf, while O. littorea was observed at Costão de Itacoatiara. When checking the bromeliads Alcantarea glaziouana for the presence of treefrogs we did not follow vocalizations to avoid bias in sex ratio. The Ololygon species were collected manually, anesthetized, fixed in 10% formalin, and preserved in 70% ethyl alcohol. This study was approved by environment agencies and is in accordance with Brazilian laws. All specimens were carefully euthanized avoiding stress, pain, or suffering. The specimens were also being used for other studies (e.g. Sabagh and Rocha 2014; Sabagh et al. 2022).
We measured the snout–vent length (SVL) of treefrogs with digital vernier calipers (nearest 0.1 mm) and weighed them on an analytical balance (nearest 0.001 g). Frogs were dissected and the ovaries were weighed and the total number of eggs found was counted and measured a under millimeter ocular (five from each ovary, totaling 10 per female). Measurements of egg diameters were based on mature ovarian eggs (pigmented) and do not include jelly capsules surrounding the eggs. The reproductive effort was calculated as the ratio of total ovarian mass divided by total body mass of each female (Camargo et al. 2008).
Data analysis
We employed a sexual dimorphism index (SDI) to quantify the extent of sex-biased body size within species and to contrast SSD levels between the two species (Gibbons and Lovich 1990). When females exhibit greater size, the ratio value is arbitrarily positive; conversely, when males are larger, it is negative. The SDI equaling 1 denotes the absence of body size difference, whereas deviations from 1 indicate significant variations in body size.
We verified the normality (Shapiro–Wilk test) of all data sets used (Shapiro and Wilk 1965). Then, for comparisons of variables between species or seasons, we conducted Mann–Whitney U-tests when data were non-normally distributed and T-tests when data were normally distributed. In the light of conducting multiple comparisons (i.e., within each species during different seasons and between species), we performed the false discovery rate (FDR) procedure at a significance level of 5% to adjust the p-values when analyzing the results (Benjamini and Hochberg 1995). We conducted, separately, linear regressions with log-transformed variables for both species to evaluate body size-RE (i.e., reproductive allometry) patterns in each species. Then, to compare whether body size-RE relationships vary significantly in slope and intercept between species we used ANCOVA analysis with body mass as a covariate. To test how egg size and the number of eggs explain reproductive investment level in species we used multiple linear regression models grouping species. All statistical analyses were performed using R 4.3.2 (R Development Core Team 2023), and all graphics were made within the ggplot2 package (Wickham 2016).
Results
We collected 109 individuals of two species of tree frogs living inside Alcantarea glaziouana bromeliads. Ololygon perpusilla individuals (n = 56, Female = 24, Male = 32) were documented at Natural Monument of Morro da Urca and Sugar Loaf sites, while Ololygon littorea individuals (n = 53; Female = 22; Male = 31) were documented founded at Costão de Itacoatiara sites. The basic statistics are summarized in Table 1.We found significant differences in SVL and body mass between females and males of the two species indicating there is intraspecific sexual size dimorphism (Fig. 2). On average, females of Ololygon perpusilla were 29.3% larger in SVL (T-test, p < 0.001, T = 13.594, d.f. = 43.71) and 100% larger in body mass (U-test, p < 0.001, U = 654) compared to males. Likewise, females of Ololygon littorea were, on average, 16.5% larger in SVL (U-test, p < 0.001, U = 520.5) and 53.65% larger in body mass (T-test, p = 0.003504, T = -3.2406, d.f. = 23.81) compared to males. The SDI exhibits positive values in both species (O. perpusilla = 2.00; O. littorea = 1.53) and it was more accentuated in Ololygon perpusilla, which showed about 30.7% greater than SDI seen in Ololygon littorea. These results support our predictions for the H1 hypothesis.
Comparing the females of the two species by morphometric traits, we expected that females should not diverge in these traits, and this was supported. In both SVL (U-test, p = 0.232, U = 172.5) and body mass (U-test, p = 0.153, U = 163) comparisons, there were no significant differences among females.”
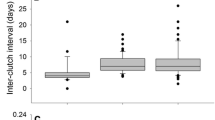
We found differences in breeding traits and reproductive effort between females of the two species, so our H2 hypothesis was partially supported. Females presented a significant divergence in reproductive effort and Ololygon perpusilla invests on average 135.9% more in reproduction than Ololygon littorea (U-test, p < 0.001, U = 80) (Fig. 3A). For breeding traits, we found that ovarian mass in Ololygon perpusilla was on average 136.0% greater than Ololygon littorea and this difference was statistically significant (U-test, p = 0.0013, U = 92.5) (Fig. 3B). We found a significant difference in mean egg diameter (mm), that was on average 11.1% in Ololygon perpusilla females (U-test, p = 0.0121, U = 41) (Fig. 3C). The difference in number of eggs between species was not statistically significant (T-test, p = 0.7704, T = -0.29554, d.f. = 21.306) (Fig. 3D). We found no significant differences in morphometrics (p > 0.05) or breeding traits (p > 0.05) arising within each species under seasons, then supporting our H3 hypothesis (Table S2). Nonetheless, we also consider the possibility that these results may be a consequence of a low sampling, in view the dispersion of data indicates changes between seasons in breeding traits for both species (Figure S1). Finally, we suppose that more conclusive assertions about seasonality could be made with a bigger sample.
We found positively significant relationships in body mass-RE in O. littorea (β = 1,1416, p = 0.00804) and O. perpsuilla (β = 1.22112, p = 0.042), indicating that females of both species tend to invest more in reproduction as long as greater (Fig. 4). Both regressions had a low adjustment (< 30%) and it can be explained by the oversimplification of the model when evaluating only body mass-RE relationship. This oversimplification occurs by excluding the influence of ovarian mass, which is regulated by breeding parameters such as egg size and egg number, on reproductive effort. On the other hand, the data dispersion also showed some large females with a very low reproductive effort. We consider these females may have reproduced shortly before being collected, which could have directly influenced the observed patterns. However, it is challenging to access this information accurately for more conclusive assertions. When comparing reproductive allometry with body mass as a covariate, we found no significant difference in body mass-RE relationships slopes between species (ANCOVA, df = 1, F = 0.01, p = 0.92186). On the other hand, we found significant differences between intercepts (ANCOVA, df = 1, F = 23.68, p < 0.001). These results support partially our H4 hypothesis, in view we noted a clear different magnitude of reproductive efforts between females of the two species.
Finally, we performed a multiple linear regression to evaluate how mean egg diameter and number of eggs are related with reproductive effort of the two species grouped together. Our MLR (adjR2 = 0.6053) showed that reproductive effort was significantly affected by mean diameter of eggs (p < 0.001) and total number of eggs (p = 0.00673). We found the mean diameter of eggs a positive slope (β = 1.6412) greater than total number of eggs (β = 0.5949). The MLR results indicates how these mechanisms are regulating reproductive effort.
Discussion
We confirmed that females are larger than males for both Ololygon species while their females did not differ in size. However, in contrast to our expectations, we found differences in breeding traits between Ololygon females with the exception of the number of eggs. Therefore, the greater reproductive of O. perpusilla effort must be associated with the larger diameter of the egg for this species.
Our results are congruent with the female-biased sexual size dimorphism seen in frogs, in which females are larger than males, which is ubiquitous in most (∼ 90%) of species (Shine 1979; Han and Fu 2013; Nali et al. 2014). Females of both species were larger than males in SVL as well as body mass and this is also in accordance with SSD literature for the Ololygon genus (Pombal et al. 2010; Lourenço et al. 2013; Pereira-Ribeiro et al. 2020). The main mechanism we propose to explain this pattern of SSD observed in our results is the fecundity selection, whereby there exists a direct relationship between female body size and fecundity (increasing fitness), and then, in an intraspecific view, intersexual decoupling of body size towards larger females than males (Darwin 1874; Shine 1989). The occurrence of this pattern is reinforced in both species studied, considering that female size is directly related with fecundity in almost all frogs exhibiting aquatic reproduction, and besides also is positively correlated with both explosive and intermittent breeding species (Nali et al. 2014). Finally, when interpreting sexual size dimorphism between the two species we consider the possibility of more accentuated value of SDI in Ololygon perpusilla arising from an increase on reproductive investment when compared with Ololygon littorea, which we expected to intensify the degree of SDI.
As we expected, morphometric traits remain similar across the two studied species reinforcing the notion of a phylogenetically-biased distribution in female size for both species, in view that phylogenetic proximity is an evident proxy for elucidating body size distributions among anurans (Phung et al. 2020). Anurans have been related as being dependent on different drivers for body size, which impacts directly in the diverse body size distribution seen in this group, varying from small to large body sizes (Amado et al. 2019, 2021; Acevedo et al. 2022). Additionally, there is clear evidence that anuran body size is also regulated by microhabitat conditions, with overlapping size distributions among species sharing similar abiotic conditions (Womack and Bell 2020). The bromeliad environment, characterized by more stable microhabitats, offers favorable and similar conditions such as water availability, humidity, and reproductive sites for the organisms that inhabit them (Zotz and Thomas 1999; de Oliveira and Navas 2004). Therefore, we interpret these results as a consequence of both phylogenetic proximity and shared abiotic conditions within bromeliad microhabitats, which collectively intensify similar pressures on morphometric tendencies. However, considering breeding traits, contrary to our expectations, we found significant differences in parameters such as ovarian mass, mean egg size and reproductive effort, while number of eggs did not vary between species. Significant differences in reproductive effort between congeneric species has been noted for females with the same size (i.e., SVL) but that diverge in body mass (Camargo et al. 2008). In contrast, our results expose new evidence that differences in reproductive investment can arise even if morphometric traits do not vary, despite being highly correlated. It suggests that differently from what is expected in literature, morphometric traits can explain reproductive patterns, but only partly, and other drivers can emerge to additionally elucidate the reproductive effort patterns seen in females.
Our findings indicated no significant difference influenced by seasonality on the body size and reproductive patterns of both species, supporting our hypothesis. However, these findings contrast with the literature, where effects of environmental changes on body size patterns observed in anurans are described (Olalla-Tárraga et al. 2009; Oyamaguchi et al. 2017; Goldberg et al. 2018; Pupin et al. 2020). We attribute these results to the relatively stable environmental conditions within the bromeliad microhabitat, which mitigate the impact of seasonal variations and favor phylogenetic-biased phenotypic convergence in body size between the two species.
Finally, our results demonstrate there is a positive relationship between body size (i.e., body mass) and reproductive investment for both species and it is a general trend seen in reproductive studies in frogs (Prado and Haddad 2005; Camargo et al. 2008; Pupin et al. 2010; Pereira and Maneyro 2012), which has been used to explain the female-biased sexual size dimorphism in most of anuran species (Shine 1979; Woolbright 1983; Han and Fu 2013; Nali et al. 2014). When compared to each other, females showed no significant differences in the slopes of the regressions between body mass-RE, indicating similar patterns of reproductive allocation. However, species differed significantly in intercept comparisons, suggesting that the magnitudes of reproductive effort vary between these species regardless of body mass. In this context, in which body size did not change between species, it suggests that the females invest different energy quantities for reproduction, which can be generated by resource limitation or increase of survival costs (Castellano et al. 2004; Camargo et al. 2008). Maintaining equivalent patterns of energy allocation, we suppose that the discrepancy in reproductive investment magnitude may be driven by lower energy availability for reproduction, thus reducing reproductive magnitude without necessarily influencing the allocation pattern (Stearns 1998; Roff et al. 2006; Cox et al. 2010). However, we also support the possibility that the reduction in reproductive effort among species may result from unmeasured environmental variations in the microhabitat. According to personal observations by the authors (i.e., L.T. Sabagh, unpublished data), it was noted that bromeliads located on the Costão de Itacoatiara, where O. littorea specimens were found, were more exposed to sunlight and had lower water accumulated, suggesting a higher risk of desiccation compared to bromeliads in the Natural Monument of Morro da Urca and Sugar Loaf where lives O. perpusilla. Empirical studies suggest a reduction in egg size in desiccation environments, as part of a strategy to shorten egg development time and increase offspring survival chances by taking advantage of the shorter temporal water availability range (McLaren and Cooley 1972; Kaplan 1985; Richter-Boix et al. 2011). Furthermore, it is acknowledged that environmental variation directly impacts the quality pattern of eggs, leading to an optimal egg size that enhances fitness according to environmental changes, which results in different optimal egg sizes depending on the environment (Kaplan and Cooper 1984; McGinley et al. 1987; Morrongiello et al. 2012; Marshall et al. 2018). Therefore, the observed differences between species may arise as a response to the lower water availability in bromeliads more exposed to sunlight. In this scenario, reducing total energy investment in reproduction, leading to a subsequent decrease in egg size (i.e., quality), could be the mechanism responsible for regulating this response to environmental variation. Despite the size and quantity of eggs being positively related to reproductive effort, the slope of the regressions demonstrates that egg size is more strongly associated with reproductive effort. Associating these results with comparison tests for breeding traits, in which the egg size of Ololygon perpusilla was on average 11% larger than that of Ololygon littorea, it was evident that the differences in egg size drove the variation in reproductive effort observed between the species.
Given the assumptions made in this study, for a better understanding of the factors influencing reproductive aspects and organism’s sensitivity to these factors, we suggest studies that assess intrapopulation variations, even within the same microhabitat. These studies should quantify fluctuations in biotic and abiotic parameters to better comprehend the nature and mechanisms of exogenous agents influencing reproductive patterns in organisms. Ideally, these studies should be designed based on future climate change scenarios.
In conclusion, this paper presents new perspectives the reproductive studies using treefrogs. Here, we highlight that reproduction can vary between species even if they are closely phylogenetically, share the same microhabitat and do not differ in morphometric traits. We suggest that regular drivers (e.g., body size) used to explain reproductive patterns described in literature may be partially sensitive to other parameters that impact the reproductive investment of females. We demonstrate that egg size is the main mechanism explaining the differences in reproductive effort between females. Our findings indicate that females differ in reproductive effort due to investments in egg quality even though the investments in the number of offspring are the same.
References
Acevedo AA, Palma RE, Olalla-Tárraga MÁ (2022) Ecological and evolutionary trends of body size in Pristimantis frogs, the world’s most diverse vertebrate genus. Sci Rep 12:18106. https://doi.org/10.1038/s41598-022-22181-5
Alves-Silva R, da Silva HR (2009) Life in bromeliads: reproductive behaviour and the monophyly of the Scinax perpusillus species group (Anura: Hylidae). J Nat Hist 43:205–217. https://doi.org/10.1080/00222930802568808
Amado TF, Bidau CJ, Olalla-Tárraga MÁ (2019) Geographic variation of body size in New World anurans: energy and water in a balance. Ecography 42:456–466. https://doi.org/10.1111/ecog.03889
Amado TF, Martinez PA, Pincheira-Donoso D, Olalla‐Tárraga MÁ (2021) Body size distributions of anurans are explained by diversification rates and the environment. Glob Ecol Biogeogr 30:154–164. https://doi.org/10.1111/geb.13206
Barberis IM, Freire RM, Montero GA (2023) Tank Bromeliads as a Water Reservoir used by humans: an Important Overlooked Ecosystem Service in Xerophytic forests. J Ethnobiol 43:373–385. https://doi.org/10.1177/02780771231209131
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc B 57:289–300. https://doi.org/10.1111/j.2517-6161.1995.tb02031.x
Camargo A, Sarroca M, Maneyro R (2008) Reproductive effort and the egg number vs. size trade-off in Physalaemus frogs (Anura: Leiuperidae). Acta Oecol 34:163–171. https://doi.org/10.1016/j.actao.2008.05.003
Castellano S, Cucco M, Giacoma C (2004) Reproductive investment of female green toads (Bufo viridis). Copeia 2004:659–664. https://doi.org/10.1643/CE-03-182R2
Cogliatti-Carvalho L, Rocha-Pessôa TC, Nunes-Freitas AF, Rocha CFD (2010) Volume de água armazenado no tanque de bromélias, em restingas da costa brasileira. Acta Bot Bras 24:84–95. https://doi.org/10.1590/S0102-33062010000100009
Cox RM, Parker EU, Cheney DM, Liebl AL, Martin LB, Calsbeek R (2010) Experimental evidence for physiological costs underlying the trade-off between reproduction and survival. Funct Ecol 24:1262–1269. https://doi.org/10.1111/j.1365-2435.2010.01756.x
Darwin CR (1874) The descent of man, and selection in relation to sex, 2nd edn. John Murray
de Oliveira FB, Navas CA (2004) Plant selection and seasonal patterns of vocal activity in two populations of the bromeligen treefrog Scinax perpusillus (Anura, Hylidae). J Herpetol 38:331–339. https://doi.org/10.1670/205-03A
Dugas MB, Stevens TC, Cosman ME, Van Dyke EE, Hernandez MD (2023) Proximate determinants of family size in an obligate egg-feeding frog. Evol Ecol 1–12. https://doi.org/10.1007/s10682-023-10255-3
Dziminski MA, Roberts JD (2006) Fitness consequences of variable maternal provisioning in quacking frogs (Crinia georgiana). J Evol Biol 19:144–155. https://doi.org/10.1111/j.1420-9101.2005.00978.x
Dziminski MA, Vercoe PE, Roberts JD (2009) Variable offspring provisioning and fitness: a direct test in the field. Funct Ecol 23:164–171
Ferreira RB, Mônico AT, Zocca CZ, Santos MTT, Lírio FC, Tonini JF, Duca C (2019) Uncovering the natural history of the bromeligenous frog Crossodactylodes izecksohni (Leptodactylidae, Paratelmatobiinae). South Am J Herpetol 14:136–145. https://doi.org/10.2994/SAJH-D-17-00092.1
Gibbons JW, Lovich JE (1990) Sexual dimorphism in turtles with emphasis on the slider turtle (Trachemys scripta). Herpetol Monogr 4:1–29. https://doi.org/10.2307/1466966
Goldberg J, Cardozo D, Brusquetti F, Bueno Villafañe D, Caballero Gini A, Bianchi C (2018) Body size variation and sexual size dimorphism across climatic gradients in the widespread treefrog Scinax fuscovarius (Anura, Hylidae). Austral Ecol 43:35–45. https://doi.org/10.1111/aec.12532
Han X, Fu J (2013) Does life history shape sexual size dimorphism in anurans? A comparative analysis. BMC Evol Biol 13:1–11. https://doi.org/10.1186/1471-2148-13-27
Justicia L, Mayer M, Lorioux-Chevalier U, Dittrich C, Rojas B, Chouteau M (2023) Intraspecific divergence of sexual size dimorphism and reproductive strategies in a polytypic poison frog. Evol Ecol 1–19. https://doi.org/10.1007/s10682-023-10280-2
Kaplan RH (1985) Maternal influences on offspring development in the California newt, Taricha torosa. Copeia 1985:1028–1035. https://doi.org/10.2307/1445258
Kaplan RH, Cooper WS (1984) The evolution of developmental plasticity in reproductive characteristics: an application of the adaptive coin-flipping principle. Am Nat 123:393–410. https://doi.org/10.1086/284211
Lantyer-Silva AS, Sole M, Zina J (2014) Reproductive biology of a bromeligenous frog endemic to the Atlantic Forest: Aparasphenodon arapapa Pimenta, Napoli and Haddad, 2009 (Anura: Hylidae). Acad Bras Cienc 86:867–880. https://doi.org/10.1590/0001-3765201420130521
Lourenço ACC, Carvalho ALG, Baeta D, Pezzuti TL, Leite FSF (2013) A new species of the Scinax catharinae group (Anura, Hylidae) from Serra Da Canastra, southwestern state of Minas Gerais, Brazil. Zootaxa 3613:573–588. https://doi.org/10.11646/zootaxa.3613.6.4
Marshall DJ, Pettersen AK, Cameron H (2018) A global synthesis of offspring size variation, its eco-evolutionary causes and consequences. Funct Ecol 32:1436–1446. https://doi.org/10.1111/1365-2435.13099
McGinley MA, Temme DH, Geber MA (1987) Parental investment in offspring in variable environments: theoretical and empirical considerations. Am Nat 130:370–398. https://doi.org/10.1086/284716
McLaren I, Cooley JM (1972) Temperature adaptation of embryonic development rate among frogs. Physiol Zool 45:223–228. https://doi.org/10.1086/physzool.45.3.30152501
Meirelles ST, Pivello VR, Joly CA (1999) The vegetation of granite rock outcrops in Rio De Janeiro, Brazil and the need of this protection. Environ Conserv 26:10–20. https://doi.org/10.1017/S0376892999000041
Morrongiello JR, Bond NR, Crook DA, Wong BB (2012) Spatial variation in egg size and egg number reflects trade-offs and bet‐hedging in a freshwater fish. J Anim Ecol 81:806–817. https://doi.org/10.1111/j.1365-2656.2012.01961.x
Nali RC, Zamudio KR, Haddad CF, Prado CP (2014) Size-dependent selective mechanisms on males and females and the evolution of sexual size dimorphism in frogs. Am Nat 184:727–740. https://doi.org/10.1086/678455
Nunes-de-Almeida CHL, Haddad CFB, Toledo LF (2021) A revised classification of the amphibian reproductive modes. Salamandra 57:413–427
Olalla-Tárraga MÁ, Diniz‐Filho JAF, Bastos RP, Rodríguez MÁ (2009) Geographic body size gradients in tropical regions: water deficit and anuran body size in the Brazilian Cerrado. Ecography 32:581–590. https://doi.org/10.1111/j.1600-0587.2008.05632.x
Oyamaguchi HM, Oliveira E, Smith TB (2017) Environmental drivers of body size variation in the lesser treefrog (Dendropsophus minutus) across the Amazon-Cerrado gradient. Bio J Linn Soc 120:363–370. https://doi.org/10.1111/bij.12879
Peixoto OL (1987) Caracterização do grupo perpusilla e revalidação da posição taxonômica de Ololygon perpusilla perpusilla e Ololygon perpusilla v-signata (Amphibia, Anura, Hylidae). Arq Univ Fed Rural Rio J 10:37–49
Peixoto OL (1995) Associação De anuros a bromeliáceas na Mata Atlântica. Rev Ciênc Vida 17:75–83
Pereira G, Maneyro R (2012) Size-fecundity relationships and reproductive investment in females of Physalaemus riograndensis Milstead, 1960 (Anura, Leiuperidae) in Uruguay. Herpetol J 22:145–150
Pereira-Ribeiro J, Linause TM, Ferreguetti AC, Cozer JS, Bergallo HG, Rocha CFD (2020) Ecological aspects of the endemic tree frog Ololygon kautskyi (Anura: Hylidae) in an Atlantic Forest area of Southeastern Brazil. J Nat Hist 54:1499–1511. https://doi.org/10.1080/00222933.2020.1810799
Phung TX, Nascimento JC, Novarro AJ, Wiens JJ (2020) Correlated and decoupled evolution of adult and larval body size in frogs. Proc R Soc B 287:20201474. https://doi.org/10.1098/rspb.2020.1474
Pombal JP, Carvalho RR, Canelas MAS, Bastos RP (2010) A new Scinax of the S. catharinae species group from Central Brazil (Amphibia: Anura: Hylidae). Zoologia (Curitiba) 27:795–802. https://doi.org/10.1590/S1984-46702010000500016
Prado C, Haddad CF (2005) Size-fecundity relationships and reproductive investment in female frogs in the Pantanal, south-western Brazil. Herpetol J 15:181–189
Pupin NC, Gasparini JL, Bastos RP, Haddad CF, Prado C (2010) Reproductive biology of an endemic Physalaemus of the Brazilian Atlantic forest, and the trade-off between clutch and egg size in terrestrial breeders of the P. signifer group. Herpetol J 20:147–156
Pupin NC, Haddad CF, Prado CP (2018) Maternal provisioning by foam-nesting frogs of the genus Physalaemus (Anura, Leptodactylidae) in contrasting environments. Amphi Reptil 39:120–125. https://doi.org/10.1163/15685381-00003146
Pupin NC, Brusquetti F, Haddad CFB (2020) Seasonality drives body size variation in a widely distributed neotropical treefrog. J Zool 312:85–93. https://doi.org/10.1111/jzo.12787
R Core Team (2023) R: a language and environment for statistical computing. R foundation for statistical computing
Reiss MJ (1989) The allometry of growth and reproduction. Cambridge University Press. https://doi.org/10.1017/CBO9780511608483
Richter-Boix A, Tejedo M, Rezende EL (2011) Evolution and plasticity of anuran larval development in response to desiccation. A comparative analysis. Ecol Evol 1:15–25. https://doi.org/10.1002/ece3.2
Roff DA, Heibo E, Vøllestad LA (2006) The importance of growth and mortality costs in the evolution of the optimal life history. J Evol Biol 19:1920–1930. https://doi.org/10.1111/j.1420-9101.2006.01155.x
Sabagh LT, Rocha CF (2014) Bromeliad treefrogs as phoretic hosts of ostracods. Naturwissenschaften 101:493–497. https://doi.org/10.1007/s00114-014-1178-y
Sabagh LT, Ferreira GL, Branco CW, Rocha CFD, Dias NY (2012) Larval diet in bromeliad pools: a case study of tadpoles of two species in the genus Scinax (Hylidae). Copeia 2012:683–689. https://doi.org/10.1643/CE-12-012
Sabagh LT, Neutzling AS, Rocha CFD (2022) Phytophagous consumption by frogs inhabiting bromeliads from Atlantic Forest. Ethol Ecol Evol 34:165–179. https://doi.org/10.1080/03949370.2021.1936652
Shapiro SS, Wilk MB (1965) An analysis of variance test for normality (complete samples). Biometrika 52(3/4):591–611. https://doi.org/10.2307/2333709
Shine R (1979) Sexual selection and sexual dimorphism in the Amphibia. Copeia 1979:297–306. https://doi.org/10.2307/1443418
Shine R (1989) Ecological causes for the evolution of sexual dimorphism: a review of the evidence. Q Rev Biol 64:19–461. https://doi.org/10.1086/416458
Silva NR, Berneck BV, da Silva HR, Haddad CF, Zamudio KR, Mott T, Prado CP (2020) Egg-laying site, fecundity and degree of sexual size dimorphism in frogs. Biol J Linn Soc 131:600–610. https://doi.org/10.1093/biolinnean/blaa126
Stearns SC (1998) The evolution of life histories. Oxford University Press
Tonini JFR, Ferreira RB, Pyron RA (2020) Specialized breeding in plants affects diversification trajectories in neotropical frogs. Evolution 74:1815–1825. https://doi.org/10.1111/evo.14037
Wells KD (2007) Ecology and behavior of amphibians. University of Chicago Press. https://doi.org/10.7208/chicago/9780226893334.001.0001
Wickham H (2016) Ggplot2: elegant graphics for data analysis. Springer Int. https://doi.org/10.1007/978-3-319-24277-4
Womack MC, Bell RC (2020) Two-hundred million years of anuran body‐size evolution in relation to geography, ecology and life history. J Evol Biol 33:1417–1432. https://doi.org/10.1111/jeb.13679
Woolbright LL (1983) Sexual selection and size dimorphism in anuran amphibia. Am Nat 121:110–119. https://doi.org/10.1098/rspb.2002.2170
Zotz G, Thomas V (1999) How much water is in the tank? Model calculations for two epiphytic bromeliads. Ann Bot 83:183–192. https://doi.org/10.1006/anbo.1998.0809
Acknowledgements
We thank the SMAC/Rio de Janeiro (Process no. 14/000.631/2008) and INEA (Authorization #021/2008) for permits to research at conservation areas. CFDR received grants from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (Processes No. 302974/2015-6, 424473/2016-0 and 304375/2020-9), and of the Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) through the Cientistas do Nosso Estado Program (processes No E-26/202.803/2018 and E-26/201.083/2022) and Prociência scholarship from Universidade do Estado do Rio de Janeiro – UERJ. MNH was supported by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP 2023/09413-8). J C Proença-Gomes kindly revised the text offering helpful suggestions.
Author information
Authors and Affiliations
Contributions
Conceptualization: LTS; Investigation: LTS, MAS; Data curation: MNH, LTS; Formal analysis: MNH, LTS; Funding acquisition: CFDR; Methodology: MNH, CFDR and LTS; Visualization: MNH; Writing—original draft: MNH; Writing—review and editing: MNH, LTS and CFDR.
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Horato, M.N., Almeida-Santos, M., Duarte Rocha, C.F. et al. Reproductive ecology of treefrogs: egg size promotes reproductive effort differences between females. Evol Ecol 38, 481–493 (2024). https://doi.org/10.1007/s10682-024-10306-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-024-10306-3