Abstract
Although plant breeding programs can vary in operations and objectives, all must contain high performing germplasm and diverse testing environments. Additionally, these elements must be well understood in order for the program to be successful. The purpose of this research was to investigate Texas A&M (TAM) hard red winter wheat variety trial locations and germplasm. The objective of this study was to gain a better understanding of the environments and germplasm by utilizing yield data from 2008 to 2012 advanced variety trials and biplot analysis. Results revealed high significant differences (P < 0.0001) amongst environments, varieties, and variety-by-environment interaction. Three mega-environments within Texas were identified as the High Plains, Rolling Plains, and Blacklands/South Texas and several environments were found to produce high yields each year. ‘Duster’ (PI 639233) was found to be the highest yielding and most stable variety across environments, in this limited set of varieties; while ‘TAMW-101’ (CItr 15324) was the lowest yielding and unstable.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Texas is a very large and diverse state with wheat producing regions ranging from temperate zones in the panhandle to the sub-tropics in the south. Abiotic and biotic stresses vary not only amongst locations, but also from 1 year to the next at the same location. The goal of the Texas A&M AgriLife Wheat Breeding Program is to develop higher yielding wheat varieties that are adapted to Texas and other states in the Southern Great Plains. In order to capture the level of variability that exists, and develop adapted varieties, over 25 locations are utilized for testing across the state annually. However, due to limited resources and increasing labor and travel costs, it is imperative that redundancy is minimized and that each testing location contributes meaningful information that will be used in the variety selection process.
Statistical software such as Microsoft Excel (Microsoft Corp. Redmond, WA 2013) and SAS (SAS Institute Cary, North Carolina 2013) are commonly used to identify superior testing locations and varieties. However, genotype-by-environment (G × E) interactions can complicate this task. G × E has been defined as genotypes failing to perform consistent relative to each other across various environments (Ghaderi et al. 1980). One method implemented by plant breeders in order to overcome this problem is combining similar varieties or environments into groups and then breeding for each independently (Malla et al. 2010; Abdalla et al. 1997). Although several methods such as variety-by-location interaction mean squares (Horner and Frey 1957), correlations of yield among locations (Guitard 1960), and two-way pattern analysis based on yield performance (Byth et al. 1976) have been used for grouping homogeneous groups, cluster analysis, which was first used by Abou-El-Fittough et al. (1969), has been relied on the most.
One tool used for identifying clusters is biplot analysis. The biplot, which is a graphical display used for evaluating multi-environment data, was first applied in agriculture by Gabriel (1971). A popular biplot among plant breeders, developed by Yan (2001), is the “GGE [Genotype (G) Genotype-by-environment (GE)] biplot”. This biplot has been used to determine the discriminating ability and representativeness of environments, identify the best performing variety in an environment, identify the most suitable environment for a given variety, and determine the average yield and stability of each of the varieties (Malla et al. 2010; Yan and Tinker 2006). Environments that provide diverse stresses and are highly discriminating will produce the most useful information to a breeding program. The “Average Tester Coordination for Tester Evaluation” biplot, which summarizes the interrelationships between test locations (Karimizadeh et al. 2013), is used to compare one environment to another and group homogenous locations together. Grouping of environments into mega-environments that perform similarly is done by breeders (Gauch and Zobel 1997; Mohammadi et al. 2011) in order to appropriately target certain environments for particular regions. The “Which-Won-Where” biplot is typically used to compare and identify varieties that performed best in each environment as well as their stability (Yan and Hunt 2002). The most highly regarded varieties are those that consistently produce high yields across environments.
The main objective of this study was to evaluate our state-wide wheat variety program in regards to the importance and contribution of each variety trial location and germplasm performance. Using biplot analysis, the most discriminating and/or representative testing environment as well as the best performing varieties and their stability in each location was determined.
Materials and methods
Yield data from the hard red winter wheat uniform variety trial (UVT) was used for this study. Collaboration between faculty of Texas A&M AgriLife Research and Texas A&M AgriLife Extension Service has been ongoing since 2004 in conducting a UVT across Texas. The UVT is comprised of a uniform list of 30–35 entries that are planted annually at over 25 locations across the state. This trial has been divided into four major geographic regions which include the High Plains, Rolling Plains, Blacklands, and South/Central (Fig. 1). The High Plains region has the greatest wheat production while south Texas has the lowest production. The same seed source was used to plant all locations which usually consisted of three replications laid out in a randomized complete block design (RCBD). Cultural practices varied by location but were representative for each region. Plots were planted with a small plot planter under no-till or conventional till practices and the total plot size was 1/1000th of an acre or larger. Seed treatments and additional insecticide and herbicide applications were performed with labeled pesticides as needed but fungicides were not applied so that disease resistance could be measured. A small plot combine (Wintersteiger, Ried im Innkreis, Austria 2018) was used to harvest the plots and yield and test weights were determined using a scale and USDA test weight apparatus. Abiotic stress such as drought or hail damage resulted in some locations not being harvested each year. If multiple years of data were present, the mean value was used for each location. All yield data was standardized to report grain yield in kilograms per hectare.
In this study, yield data of 16 varieties (Table 1) planted at 19 locations (95 total site-years) (Fig. 1) in the UVT from 2008 to 2012 was used to evaluate environment and germplasm performance. These 16 varieties were selected for analysis as they were present at all locations while the remaining entries within the UVT varied across locations. Environmental performance across years and amongst locations was examined and a combined environment analysis of variation (ANOVA) was conducted using SAS v9.3. Biplot analysis was conducted using the GGE biplot software (Yan 2001). GGE biplots were used to identify mega-environments, the most and least discriminating environments, the highest and lowest yielding varieties, and the most stable varieties across locations. Environments were evaluated based on their ability to discriminate between varieties and the mean performance of varieties planted at each location. Locations that produced similar results were grouped into mega-environments. Furthermore, the best and worst performing varieties were identified for each mega-environment and the stability of these varieties was also determined.
Results and discussion
A combined environment ANOVA for grain yield (Table 2) showed highly significant differences (P < 0.0001) in each source. The term “yxl” (year-by-location) was used to represent environments (example: College Station 2011 and College Station 2012 were considered two different environments). Highly significant differences amongst environments indicates (1) at least one location did not perform similarly from 1 year to another, (2) at least two locations did not perform similarly, or (3) a combination of these two, although it does not provide information on which case is present. Differences amongst environments can be better seen in Table 3 which shows the mean grain yield for each location across years. The ANOVA table also showed a high significant difference (P < 0.0001) between varieties indicating significant variation in yield between at least two of these varieties. Finally, the variety-by-environment interaction was found to be highly significant (P < 0.0001) indicating that there was variation in the yield of varieties amongst environments.
Locations that consistently produce high yields are an important aspect of a breeding program. These types of locations can assist breeders in assessing new varieties for their full yield potential as well as be a suitable place for growing seed increase plots in preparation for the release of a variety. The mean grain yield data (Table 3) revealed several locations that would be suitable for this purpose. Comparing this data with national average yields of hard red winter wheat, which ranged from 2266 to 2730 kg per hectare from 2012 to 2015 (USDA-ERS 2015), several locations were identified that reliably produce yields that are well above the national average. Clovis and Bushland under irrigated conditions as well as Castroville and Prosper were found to be the highest yielding locations across Texas.
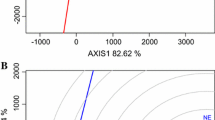
Biplot analysis is commonly used to determine similarities amongst testing locations and identify mega-environments (Malla et al. 2010; Munaro et al. 2014; Yan et al. 2000). This was done using the ‘Average Tester Coordination for Tester Evaluation’ biplot (Fig. 2). This biplot showed three clusters of locations: Etter Dry, Etter Irrigated, Canadian, Hereford, Clovis Dry, Clovis Irrigated, Bushland Dry, Bushland Irrigated, and Dalhart (Cluster 1); Abilene, Brady, Castro County, Chillicothe, and Hardeman (Cluster 2); College Station, Ellis County, Prosper, Castroville, and McGregor (Cluster 3). These clusters were determined to be separate mega-environments identified as the High Plains (cluster 1), Rolling Plains (cluster 2), and Blacklands/South Texas (cluster 3). The red circle lying on the red line that runs through the biplot indicates the average for all locations in the analysis. A location that appears close to this point would be considered representative for the entire state of Texas. Therefore, Chillicothe and Castro County were found to be the most representative locations and would be the best for testing and selecting varieties that are generally adapted for Texas. A similar analysis previously conducted using 2004–2008 UVT data had revealed four clusters (Blacklands and South Texas locations were grouped separately) and found Brady to be the best representative of all testing locations (Dr. Amir M.H. Ibrahim- Personal Communication). These changes are most likely due to the drought conditions that were seen throughout Texas especially in 2010. Another feature of this biplot is the vectors that connect each location to the center of the concentric circles which are used to approximate discriminating ability. Those with a long vector are considered very discriminating meaning that they greatly show the differences between superior and non-superior varieties. Those with very short vectors show very little variation between varieties and, therefore, contribute the least amount of useful information for selecting varieties. In this analysis, Abilene had the shortest vector indicating it was the least discriminating environment. Many of the locations located in the South Texas/Blacklands and High Plains regions had long vectors and contributed the most useful information in selecting varieties. Yan (2001) describes an “ideal” location as the one that best combines discriminating ability and representativeness. The best location was Chillicothe as it was the most discriminating testing site closest to the ideal location (Fig. 2). The information gathered from this biplot can greatly assist in using resources efficiently. Variety performance within a mega-environment of tightly clustered locations should be similar and, therefore, some can be eliminated without much loss of information. Additionally, locations that are very discriminating or representative will be retained while those that are not can be discarded.
GGE biplot showing environment discrimination grouping of environments and average wheat testing location for Texas. Location Abbreviations: ABI = Abilene, BD = Bushland Dry, BI = Bushland Irrigated, BRD = Brady, CAN = Canadian, CAS = Castroville, CH = Chillicothe, CS = College Station, CTRO = Castro County, CVD = Clovis Dry, CVI = Clovis Irrigated, DAL = Dalhart, ED = Etter Dry, EI = Etter Irrigated, ELS = Ellis County, HFD = Hereford, HG = Hardeman, MCG = McGregor, PRO = Prosper
The ‘which wins where and which is best for what’ biplot (Fig. 3) shows which varieties performed best in which environments. In this graph, the varieties most distant from the biplot origin are connected to create a polygon so that all other varieties are contained within it. Perpendicular lines are then drawn from the origin to make a right angle with each side of the polygon. Varieties that are located at the vertices of the polygon are either the best or poorest performing varieties in one or more environments. From the biplot, ‘TAM 112’ (PI 643143; Rudd et al. 2014) was found to be the best performing variety in the cluster of locations identified as the High Plains region. ‘TAM 111’ (PI 631352; Lazar et al. 2004), ‘TAM 113’ (PI 666125; Rudd et al. 2011a, b), and ‘Endurance’ (PI 639233; Carver et al. 2006) were also adapted to this region but were not as high yielding. In the cluster comprised mostly of Rolling Plains locations, Duster (Edwards et al. 2012) was found to be the top performing variety. ‘TAM 304’ (PI 655234; Rudd et al. 2015) was the best performing variety in the cluster of South Texas/Blacklands locations. ‘TAM 203’ (PI 655234), ‘Greer’, ‘Jackpot’ (PI 658007), ‘Santa Fe’, and ‘Fuller’ (PI 653521) were also adapted to these regions but were not as high yielding as TAM 304. The varieties TAMW-101 (Porter 1974), ‘Jagger’ (PI 593688; Sears et al. 1997), ‘Fannin’ (PI 639231), and ‘TAM 401’ (PI 658500; Rudd et al. 2011a, b) did not perform well across a broad mega-environment. ‘Fannin’ (PI 639231), which has a tendency to lodge under optimum fertility and moisture conditions due to below average straw strength (Watson and Steve 2009), and ‘TAM 401’, which performs well in South Texas locations but does not fare as well in the northern part of the state, were identified as the worst performers overall.
GGE biplot showing best and poorest performing cultivars for each test environment. Cultivars that appear at the vertex of the polygon close to a location is best suited for that location. Cultivars that do not appear close to any location are not well suited for any of the locations. Testing locations in red, cultivars in blue. Location Abbreviations: ABI = Abilene, BD = Bushland Dry, BI = Bushland Irrigated, BRD = Brady, CAN = Canadian, CAS = Castroville, CH = Chillicothe, CS = College Station, CTRO = Castro County, CVD = Clovis Dry, CVI = Clovis Irrigated, DAL = Dalhart, ED = Etter Dry, EI = Etter Irrigated, ELS = Ellis County, HFD = Hereford, HG = Hardeman, MCG = McGregor, PRO = Prosper
Figure 4 demonstrates the stability of individual varieties across environments. This figure is comprised of a red line that passes through the biplot origin and is referred to as the average-tester axis. The small red circle that lies on this line indicates the position of the average tester which is defined by the average of PC1 and PC2 scores across all testers. This figure also contains a blue line that passes through the biplot origin and is perpendicular to the average-tester axis which separates entries with below-average means from those with above-average means with average yield increasing from left to right. Finally, a vector is used to connect each variety to the average-tester axis which represents the stability of each entry. Varieties with a short vector were stable whereas those with long vectors were not stable across environments. Duster was found to be the highest yielding and most stable variety, which could be due to its resistance to leaf rust (Puccinia triticina) and soil-borne wheat mosaic virus (Virgaviridae Furovirus) and moderate resistance to stripe rust (Puccinia striiformis) and powdery mildew (Blumeria graminis f. sp. tritici) (Oklahoma Foundation Seed Stocks 2010). ‘TAM 112’ was also high yielding but was very unstable across environments. This is most likely due to leaf and stripe rust susceptibility in humid conditions such as those typically observed in South Texas locations. As seen in the Fig. 3, ‘Fannin’ and ‘TAM 401’ were two of the lowest yielding varieties and also unstable across environments. This study is beneficial as varieties found to be both high yielding and stable, and their progenies, will be used as parental lines for new varieties.
GGE biplot showing each cultivar’s mean performance and stability. Location Abbreviations: ABI = Abilene, BD = Bush Dry, BI = Bush Irrigated, BRD = Brady, CAN = Canadian, CAS = Castroville, CH = Chillicothe, CS = College Station, CTRO = Castro County, CVD = Clovis Dry, CVI = Clovis Irrigated, DAL = Dalhart, ED = Etter Dry, EI = Etter Irrigated, ELS = Ellis County, HFD = Hereford, HG = Hardeman, MCG = McGregor, PRO = Prosper
Conclusion
Biplot analysis is a powerful tool for plant breeders in evaluating multi-environment data as it allows for effective evaluation of the varieties and testing environments that are used in a breeding program. In this study, 5 years of UVT data was analyzed using biplots in order to better understand the various winter wheat growing environments in Texas. In contrast to a previous report which clustered locations into four mega-environments, only three were found in this study. As new analyses are conducted in the future, it may be possible to better understand and therefore predict how the best regionally adapted varieties change due to various environmental factors such as severe drought. Researchers can use this information, and the varieties identified as being stable across environments, to develop the next generation of adapted cultivars as well as make recommendations to producers.
References
Abdalla OS, Crossa J, Cornelius PL (1997) Results and biological interpretation of shifted multiplicative model clustering of durum wheat cultivars and test site. Crop Sci 37:88–97
Abou-El-Fittough HA, Rawlings JO, Miller PA (1969) Classification of environments to control genotype by environment interactions with an application to cotton. Crop Sci 9:135–140
Byth DE, Eisemann RL, De Lacy IH (1976) Two-way pattern analysis of a large data set to evaluate genotypic adaptation. Heredity 37:215–230
Carver BF, Smith EL, Hunger RM, Klatt AR, Edwards JT, Porter DR, Verchot-Lubicz J, Rayas-Duarte P, Bai G-H, Martin BC, Krenzer EG, Seabourn BW (2006) Registration of ‘Endurance’ wheat. Crop Sci 46:1816–1817
Edwards JT, Hunger RM, Smith EL, Horn GW, Chen MS, Yan L, Bai G, Bowden RL, Klatt AR, Rayas-Duarte P, Osburn RD, Giles KL, Kolmer JA, Jin Y, Porter DR, Seabourn BW, Bayles MB, Carver BF (2012) ‘Duster’ wheat: a durable, dual-purpose cultivar adapted to the southern great plains of the USA. J Plant Reg 6:37–48
Gabriel KR (1971) The biplot graphic display of matrices with application to principle component analysis. Biometrika 58:453–467
Gauch HG, Zobel RW (1997) Identifying mega-environments and targeting genotypes. Crop Sci 37:311–326
Ghaderi A, Everson EH, Cress CE (1980) Classification of environments and genotypes in wheat. Crop Sci 20:707–710
Guitard AA (1960) The use of diallel correlations for determining the relative locational performance of varieties of barley. Can J Plant Sci 40:645–651
Horner TW, Frey KJ (1957) Methods for determining natural areas for oat varietal recommendations. Agron J 49:313–315
Karimizadeh R, Mohammadi M, Sabaghni N, Mahmoodi A, Roustami B, Seyyedi F, Akbari F (2013) GGE biplot analysis of yield stability in multi-environment trials of lentil genotypes under rainfed condition. Not Sci Biol 5(2):256–262
Lazar MD, Worrall WD, Peterson GL, Fritz AK, Marshall D, Nelson LR, Rooney LW (2004) Registration of ‘TAM 111’ wheat. Crop Sci 44:355–356
Malla S, Ibrahim A, Little R, Kalsbeck S, Glover K, Ren C (2010) Comparison of shifted multiplicative model, rank correlation, and biplot analysis for clustering winter wheat production environments. Euphytica 174:357–370
Microsoft Corporation (2013) Microsoft Office 2013. Redmond, Washington. Available at: https://www.microsoft.com/en-us/. Accessed 4 Apr 2018
Mohammadi R, Armion M, Sadeghzadeh D, Amri A, Nachit M (2011) Analysis of genotype-by-environment interaction for agronomic traits of durum wheat in Iran. Plant Prod Sci 14:15–21
Munaro L, Genin G, Marchioro V, Franco F, Silva R, Silva C, Beche E (2014) Brazilian spring wheat homogeneous adaption regions can be dissected in major megaenvironments. Crop Sci 54:1374–1383
Oklahoma Foundation Seed Stocks (2010) Duster hard red winter wheat. Available at: http://www.okgenetics.com/files/Duster%20Brochurefeb%202014.pdf. Accessed 6 Oct 2015
Porter KB (1974) Registration of TAM W-101 wheat. Crop Sci 14:608
Rudd JC, Devkota RN, Fritz AK, Baker JA, Obert DE, Worrall D, Sutton R, Rooney LW, Nelson LR, Weng Y, Morgan GD, Bean B, Ibrahim AM, Klatt AR, Bowden RL, Graybosch RA, Jin Y, Seabourn BW (2011a) Registration of ‘TAM 401’ wheat. J Plant Reg 6:60–65
Rudd JC, Devkota RN, Baker JA, Ibrahim AM, Worrall D, Lazar MD, Sutton R, Rooney LW, Nelson LR, Bean B, Duncan R, Seabourn BW, Bowden RL, Jin Y, Graybosch RA (2011b) Registration of ‘TAM 113’ wheat. J Plant Reg 7:63–68
Rudd JC, Devkota RN, Baker JA, Peterson GL, Lazar MD, Bean B, Worrall D, Baughman T, Marshall D, Sutton R, Rooney LW, Nelson LR, Fritz AK, Weng Y, Morgan GD, Seabourn BW (2014) ‘TAM 112’ wheat, resistant to greenbug and Wheat Curl Mite and adapted to the dryland production system in the Southern High Plains. J Plant Reg 8:291–297
Rudd JC, Devkota RN, Ibrahim AM, Marshall D, Sutton R, Baker JA, Peterson GL, Herrington R, Rooney LW, Nelson LR, Morgan GD, Fritz AK, Erickson CA, Seabourn BW (2015) ‘TAM 304’ wheat, adapted to the adequate rainfall or high-input irrigated production system in the Southern Great Plains. J Plant Reg 9:331–337
SAS Institue Inc. (2013) SAS Software Version 9.4. Cary, North Carolina. Available at: https://www.sas.com/en_us/home.html. Accessed 4 Apr 2018
Sears RG, Moffatt JM, Martin TJ, Cox TS, Bequette RK, Curran SP, Chung OK, Heer WF, Long JH, Witt MD (1997) registration of ‘Jagger’ wheat. Crop Sci 37:1010
United States Department of Agriculture- Economic Research Service (2015) Wheat data overview. Available at: http://www.ers.usda.gov/data-products/wheat-data.aspx. Accessed 21 Sept 2015
Watson S (2009) Wheat varieties for Kansas and the Great Plains, 2010: your best choices. Lone Tree Publishing, Topeka
Wintersteiger AG (2018) Ried im Innkreis, Austria. Available at: https://www.wintersteiger.com/us/Home. Accessed 24 May 2018
Yan W (2001) GGE biplot-a windows application for graphical analysis of multi-environment trial data and other types of two way data. Agron J 93:1111–1118
Yan W, Hunt LA (2002) Biplot analysis of multi-environment trial data. In: Kang MS (ed) Quantitative genetics, genomics, and plant breeding. CABI Pub, Wallingford, New York
Yan W, Tinker N (2006) Biplot analysis of multi-environment trial data: principles and applications. Can J Plant Sci 86:623–645
Yan W, Hunt LA, Sheng Q, Szlavnics Z (2000) Cultivar evaluation and mega-environment investigation based on the GGE biplot. Crop Sci 40:597–605
Acknowledgements
We thank all of the members of the Texas A&M AgriLife Research and Extension Small Grains program who provided assistance in planting, growing, and harvesting these research plots across the state. We would also like to thank the Texas Wheat Producers Board for providing the funding to conduct the UVT trials.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gerrish, B.J., Ibrahim, A.M.H., Rudd, J.C. et al. Identifying mega-environments for hard red winter wheat (Triticum aestivum L.) production in Texas. Euphytica 215, 129 (2019). https://doi.org/10.1007/s10681-019-2448-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10681-019-2448-8