Abstract
Aegilops tauschii, as one of wheat wild relatives, possesses a wide range of resistance to biotic/abiotic factors. To better utilize and provide the genetic basis of breeding for improving phosphorus deficiency tolerance, large-scale screening for tolerant A. tauschii to phosphorus deficiency was conducted using hydroponic experiment. A total of 380 A. tauschii genotypes were used to evaluate their phosphorus deficiency tolerance using root system at seedling stage. The results indicate that compared to applied phosphorus condition, genetic variation of the tested traits among 380 A. tauschii genotypes was significant under non-applied phosphorus condition. Heritability estimates showed that root dry weight, shoot dry weight, and total dry weight is highly inheritable. Principal component (PC) analysis showed that first four PCs explained 81.387 % of total variation. S value and phosphorus deficiency tolerance index (PDTI) showed that all A. tauschii genotypes can be divided into three groups, high (17 %), moderate (58 %) and low (25 %) tolerance groups. Compared with other cultivated wheat using S value and PDTI, the top five tolerant A. tauschii genotypes have stronger ability of phosphorus deficiency tolerance than other cultivated wheat. In conclusion, we systematically evaluated large-scale A. tauschii genotypes, and 63 tolerant lines and some root traits used as selection criteria were selected. The top five tolerant A. tauschii genotypes have relative higher tolerance than other wheat, and can be further used for wheat genetic improvement and molecular breeding for abiotic stress tolerance.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Phosphorus (P) is of definite importance for the production of food crops and the demand for P fertilizer is increasing worldwide. Terrestrial plants generally take up soil P in its inorganic form (Pi) (Marschner 1995). However, 50–80 % of the total P in agricultural soils exists as organic phosphate, in which, up to 60–80 % is myoinositol hexakisphosphate (phytate) (Iyamuremye et al. 1996; Turner et al. 2002; George and Richardson 2008). Because phytate-P is not directly available to plants, low P availability becomes one of the limiting factors to plant growth and development. Not surprisingly, application of P fertilizer is the main solution. However, high inputs and low recovery rates of P fertilizers not only increase the cost of crop production, but also accelerate the exhaustion of non-renewable phosphate resources (CAY 1991, 2006). It has been estimated that the P will be exhausted by the end of this century both in China (Ma et al. 2005) and worldwide (Vance et al. 2003).
In response to available P deficiency condition, plant have developed numerous adaptive mechanisms for better growth, including modification of root morphology and architecture, activation of high-affinity Pi transporters, improvement of internal phosphatase activity, and secretion of organic acids and phosphatases (Raghothama and Karthikeyan 2005; Vance et al. 2003). Plants can increase acquisition and utilization efficiency of P through these adaptive mechanisms, which are referred to as P-efficiency (Wang et al. 2010). But there are many differences in P-efficiency among different plant species. Meanwhile, mechanisms of P-efficiency can be different from one genotype to other within a given plant species, such as wheat (Levent et al. 2005), sweet maize (Lu et al. 2010) and rice (Li et al. 2005). In wheat, the differences of P-efficiency have the following characters: (i) modern cultivars exceed traditional cultivars (Horst et al. 1993), (ii) semi-dwarf plant type exceeds common plant type (Wu 1990), and (iii) cultivars that developed from distant hybridization exceed common cultivars (Zhong et al. 2003). In other words, different genotypes of plant species exist obvious genetic diversity on P-efficiency, high P-efficiency genotypes of plant species often demonstrate high tolerance to P deficiency, thereby, screening and utilizing high tolerance genotypes to P deficiency of a given plant species could be a good solution to face available P deficiency.
In the long process of biological evolution, wild species survive in various stress conditions and conserve some favorable traits, such as, drought resistance, saline-alkaline resistance, barren soil resistance, disease resistance and P deficiency resistance (Cui et al. 2004). Thus, taking advantages of these favorable traits in wild relatives or ancestors of wheat can improve wheat breeding efficiency. Aegilops tauschii is one of the ancestral species of wheat, which is the donor species of D genome of wheat. It possesses with a wide range of resistance/tolerances to biotic/abiotic factors (Valkoun et al. 1990; Cox et al. 1992) that can contribute to wheat improvement, (Yen and Yang 1984). In addition, screening and evaluation of high P-efficiency genotypes more concentrated in common wheat in China and abroad at present (Yang et al. 2012; Xiong et al. 2008), excellent genetic resources of wild species also remains to excavate, so breeders increasingly focused on A. tauschii.
In the study, 380 A. tauschii genotypes from all over the world were evaluated using root system through hydroponic experiments. The digital image analysis was introduced to collect root characters, because it provides more accurate and less subjective measures than the characteristics that the human eye can detect (Bouma et al. 2000). Thus, we use digital image analysis to investigate root characters of A. tauschii under applied phosphorus (AP) and non-applied phosphorus (NAP) conditions at seedling stage. The objectives of this study were to establish the evaluation system of wild relatives using multiple root related traits, and to screen excellent gene resources with high tolerance to P deficiency from 380 A. tauschii, laying the foundation for wheat breeding for improving abiotic stresses.
Materials and methods
Plant materials
A total of 380 Aegilops tauschii accessions (2n = 14, DD),two hexaploid wheat accessions, CS and CN16, (Triticum aestivum L, 2n = 42, AABBDD), two tetraploid wheat accessions, AS2255 and AS313, (Triticum turgidum L., 2n = 28, AABB) and two synthetic hexaploid wheat lines, LAN-2404 and LAN-2399 (2n = 42, AABBDD) were used in the study. These accessions were maintained by Triticeae Research Institute of Sichuan Agricultural University (SAU). CS accession is susceptible to P deficiency (Su et al. 2006; Ren 2012). LAN-2404 and LAN-2399 were screened as tolerant genotypes under drought and nutrients deficiency stresses in our previous studies. CN16 is a native wheat cultivar possessing good comprehensive characters and is an ideal recurrent parent for back-cross breeding. These accessions were maintained by Triticeae Research Institute of Sichuan Agricultural University (SAU).
Growth conditions
All the A. tauschii were grown in phytotron in Wenjiang, Sichuan Province, China, during September 2012–March 2013. The materials were evaluated by hydroponic experiments under two phosphorus conditions, applied phosphorus (AP) and non-applied phosphorus (NAP) in a completely randomized design, each with four replications. The AP and NAP treatments contained normal nutrient solution with and without NH4H2PO4 (1 mmol/L), respectively. The normal nutrient solution was modified from Hoagland’s nutrient solution (Hoagland and Arnon 1950) in order to optimize growth condition of A. tauschii. The modified Hoagland’s nutrient solution is consisted of Ca(NO3)2·4H2O (4 mmol/L), KNO3 (6 mmol/L); MgSO4·7H2O (2 mmol/L), H3BO3 (46 µmol/L), Na·Fe·EDTA (100 µmol/L); MnCl2·4H2O (9.146 µmol/L), ZnSO4·7H2O (0.76 µmol/L), CuSO4·5H2O (0.32 µmol/L), (NH4)6Mo7O24·4H2O (0.0161 µmol/L).
Uniform seedlings grown from eight-day germinated seeds were transplanted into a hole of foamboard, a sponge stripe was needed to twine around the seedlings in case of their falling down from foamboard, and then put the foamboard with seedlings into the box which filled with nutrient solution. Make sure all the little root of seedlings must be fully exposed to nutrient solution so that they can absorb nutrients. In order to ensure the regular growth of all the seedlings after transplanting, they should be grown in normal solution with phosphorus for four days. Subsequently, normal solution with NH4H2PO4, and without NH4H2PO4 were used to water A. tauschii genotypes under AP and NAP conditions, respectively, and the solutions were replaced every 4 days. The growth environment of all seedlings was 25/22 (±1) °C day/night temperature and 65/85 % day/night relative humidity, and a 16-hour photoperiod with 500 μmol m−2 s−1 photon flux density at the level of the plant canopy.
Data collection and analysis
Shoot characteristics and root morphology were evaluated after 18 days of transplantation. Seedlings was washed with clean water carefully and then root morphology was scanned with the scanner Epson XL 10,000×, using WinRHizo Pro 2008a image analysis system (Régent Instr. Inc., Quebec, Canada) to obtain root characteristics including root length (RL), root diameter (RDM), root surface area (RSA), root volume (RV), root tips (RT) and root forks (RF). Then the plants were separated into shoot and root, stored in paper bags, and heated at 105 °C for 30 min to kill the living cells, and then dried at 75 °C until obtaining constant mass, root dry weight (RDW) and shoot dry weight (SDW) (Table 1).
In order to eliminate inherent biology and genetics difference of different A. tauschii genotypes, the phosphorus deficiency tolerance index (PDTI) was used as an indicator to measure diversity of different genotypes with PDTI = TNAP/TAP (Ren 2012; Zhang et al. 2004a, b), where TNAP and TAP are the traits measured under NAP and AP conditions respectively. The estimate of heritability is defined by the formula H = VG/(VG + VE), where VG and VE represent estimates of genetic and environmental variances, respectively (Smith et al. 1998). Stepwise multiple linear regression was used according to Draper and Smith (1966) to determine the variable accounting for the majority of total TDW variability. Descriptive analysis, analysis of variance, correlation analysis, principal component analysis (PCA) and multiple linear stepwise regression were conducted for the tested traits using IBM SPSS Statistics for windows, Version 20.0 (IBM Corp. Released 2011). Weighting method was utilized to acquire S value of each A. tauschii genotype, which calculated as \({{S = \sum\nolimits_{i = 1}^{k} {r_{i} Y_{i} } } \mathord{\left/ {\vphantom {{S = \sum\nolimits_{i = 1}^{k} {r_{i} Y_{i} } } {\sum\nolimits_{i = 1}^{k} {r_{i} } }}} \right. \kern-0pt} {\sum\nolimits_{i = 1}^{k} {r_{i} } }}\), where r i is the first i eigenvalues of principal component (PC), Y i is the first i score of PC that calculated by SPSS factor analysis using PDTI, k is the number of PC.
Results
Variance analysis and the basic parameter estimation
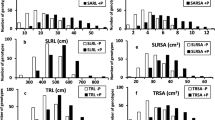
Analysis of variance revealed obvious variation among genotypes for all tested traits (Table 2). The level of variation is reflected by distribution of the traits representing different P deficiency tolerance criteria (Fig. 1). Ten traits except RL in 380 A. tauschii genotypes have significant variation. Significant difference was also observed under the NAP and AP conditions for the tested traits except RDM. The results indicate that the tested traits were significantly influenced by phosphorus supply among 380 A. tauschii genotypes and they could be used for further genetic analyses. The basic parameter estimation including mean, standard deviation and coefficient of variation also showed substantial phenotypic variation among genotypes for each trait (Table 3). Coefficient of variation under NAP condition ranged from 13.113 to 41.436 %, while under AP condition it ranged from 5.043 to 36.288 %. Compared with AP condition, the mean value of RL, RDW, SDW, RDM, RSA, RV and TDW decreased under NAP condition, on the contrary, the rest traits including RT, RF and R/S increased under NAP condition.
Heritability estimated
Heritability estimates were obtained for all the traits (Table 3), and it was generally higher under NAP condition than that under AP condition. Among them, TDW had the highest heritability (0.905 for NAP and 0.818 for AP) and RDM had the lowest heritability (0.685 for NAP and 0.550 for AP). For the other traits, heritability estimates were relatively high, ranging from 0.618 (RL under AP condition) to 0.900 (RDW under NAP condition). It is noteworthy that biomass-relative traits like RDW, SDW and TDW showed high heritability estimate (0.900 for NAP and 0.801 for AP; 0.868 for NAP condition and 0.796 for AP condition; 0.905 for NAP condition and 0.818 for AP condition; respectively). RSA (0.838 for NAP and 0.753 for AP), RV (0.823 for NAP and 0.759 for AP), RT (0.756 for NAP and 0.659 for AP) and RF (0.797 for NAP and 0.733 for AP) had an intermediate level of heritability. These results indicate that RDW, SDW and TDW are highly inheritable and can be used as a good indicator for screening A. tauschii genotypes for phosphorus deficiency tolerance.
Genetic correlations among tested traits
Pearson correlations among 10 traits were calculated (Table 4), and there are 28 and 27 correlation pairs reached the significant levels under NAP and AP conditions, respectively. Root-related traits, such as RT and RF (r = 0.888 and r = 0.745 for NAP and AP, respectively), RDW and SDW (r = 0.837 and r = 0.821 for NAP and AP, respectively), RL and RDW (r = 0.655 and r = 0.643 for NAP and AP, respectively) showed relatively high correlations under two P treatments. TDW was significantly correlated with SDW (0.978 for NAP and 0.985 for AP) and RL (0.655 for NAP and 0.735 for AP).
Principal component analysis (PCA) and multiple linear regression analysis
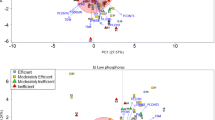
Bartlett test of sphericity Chi square value was 2293.627, which reached significant level at P < 0.001 and indicated there is high correlation among various traits. Therefore, PCA were performed for all the tested traits (Table 5) and plot for the first principal component (PC1) and the second principal component (PC2) are shown in Fig. 2. The first four principal components (PCs) were selected and the cumulative contribution of them was 81.387 %. The eigenvalue of first PC was 3.046, explained 35.463 % of phenotype variation. RDW, SDW and TDW were the three major factors among corresponding characteristic vector. They represent the plant biomass and can be defined as biomass factor. The eigenvalue of second PC was 2.204, explained 22.036 % of phenotype variation. RSA, RV, RT and RF were the four major factors among corresponding characteristic vector, and they influence the ability of obtaining phosphorus nutrition and can be defined as phosphorus obtaining factor. The eigenvalue of third principal component was 1.253, contributed to 12.534 % of phenotype variation. R/S was the only one major factor and it suggests the environment is conducive to root growth or shoot growth. The eigenvalue of fourth PC was 1.136, with 11.355 % of explained phenotype variation, and RDM was the only one major factor. RL, RT, RF and R/S were negative values and it shows that excessive RDM increasing will reduce the value of RL, RT, RF and R/S.
To establish selection indices involving multiple P deficiency tolerant traits, multiple linear regression was performed for the tested traits. Here, we built the regression to explain total dry weight through stepwise regression that contains regression models in which the choice of predictive variables was carried out (Table 6). The final stepwise model built based on P deficiency criteria explained 88.2 % and 86.6 % of phenotypic variation for dry weight under NAP and AP conditions. The built model included six important traits for NAP condition, their relative contribution are: RL (40.9 %), RDW (39.1 %), RDM (7.2 %), RSA (0.6 %), RT (0.3 %) and RF (0.1 %). There are seven important traits for AP condition, their relative contribution are: RL (42.3 %), RDW (38.6 %), RDM (4.0 %), RSA (0.7 %), RV (0.5 %), RT (0.3 %) and RF (0.2 %).
Screening for A. tauschii tolerant genotypes using root system
We used weighting method to calculate S value for each A. tauschii genotype, and the phosphorus deficiency tolerance index (PDTI) for each trait was calculated as well. The genotypes with relatively higher and lower S value and PDTI are shown in the Table 7. According to the principle of high S value with high tolerance ability, among 380 A. tauschii, AS623282 had the highest S value of 3.853 and AS623395 had lowest S value of −1.257, which was selected as extremely tolerant and sensitive genotypes, respectively. Overall, there are 63, 221 and 96 genotypes with high (S ≥ 0.5), moderate (−0.5 ≤ S < 0.5) and low tolerant ability (S < −0.5), accounting for 17, 58 and 25 %, respectively. PDTI calculated for A. tauschii genotypes also reflect the capability of P deficiency tolerance, and plants often enhance P deficiency tolerant ability with increasing PDTI. It is observed that A. tauschii with a higher S value also has a higher PDTI (Table 7), which illustrates the two selection indicators are effective for screening A. tauschii under P deficiency condition. Among 63 A. tauschii genotypes that possess high tolerance under phosphorus deficiency, we found most of them come from the west of Asia and distribute nearby Caspian Sea (Table 8; Fig. 3) through analysis on their geographic origin. It can be seen that 63 A. tauschii genotypes were found in 12 countries, of which 13 from Iran, nine from Turkey and eight from Afghanistan. They distributed mainly in a zone of Turkey-Iran-Afghanistan, which is in keeping with the road of A. tauschii spreading.
Comparison of tolerance ability among different origin of A. tauschii
PDTI for each traits and average S value across all the test traits were used to compare different P deficiency tolerance among different origin of A. tauschii. Table 8 listed their average S values for A. tauschii genotypes from each origin. China, Turkey, Uzbekistan and Azerbaijan had higher S value, which were 0.212, 0.210, 0.049 and 0.011, respectively. The percentage of the selected tolerant genotypes was 13.333, 22.500, 23.529 and 17.647 % of the total accessions from China, Turkey, Uzbekistan and Azerbaijan, respectively. For PDTI, we compared their values for 10 traits in nine countries, each with more than 10 A. tauschii genotypes (Fig. 4). The nine countries included Afghanistan, Armenia, Azerbaijan, China, Iran, Pakistan, Tajikistan, Turkey and Uzbekistan, and there are 51, 14, 34, 15, 81, 42, 12, 40 and 17 genotypes respectively (Table 8). The four countries China, Turkey, Uzbekistan and Azerbaijan with higher S value also have larger PDTI value for traits, RDW, SDW, RSA, RV, RT, RF and TDW than others (Fig. 4). The results suggest the A. tauschii from China, Turkey, Uzbekistan and Azerbaijan have relatively higher P deficiency tolerance ability.
Comparison of tolerance ability between A. tauschii and other wheat
According to the S value, AS623282, AS623011, AS623285-1, AS623305 and AS623020 are listed as the top five genotypes with high tolerance ability. To compare the tolerance ability to phosphorus deficiency between A. tauschii that have been screened out and other cultivated wheat, tetraploid wheat (AS2255 and AS313), hexaploid wheat (CS and CN16) and synthetic hexaploid wheat (LAN-2404 and LAN-2399) were used in the study. The selected A. tauschii genotypes have higher PDTI and S values than other cultivated wheat (Table 9). CS shows susceptible character under P deficiency in our study and previous reports (Su et al. 2006; Ren 2012). Synthetic hexaploid wheat, LAN-2404 and LAN-2399, were hybrids crossing by tetraploid wheat and A. tauschii..They were selected as tolerant genotypes under drought and nutrient deficiency environments in our previous study. Here, we found it showed moderate tolerance to P deficiency. In addition, the synthetic hexaploid wheat (LAN-2404 and LAN-2399) had higher S value than tetraploid wheat (AS2255 and AS313) and hexaploid wheat (CS and CN16). The results showed that the top five A. tauschii genotypes selected based on S value and PDTI have higher tolerance in response to phosphorus deficiency than other cultivated wheat tested in the study.
Discussion
The importance of root morphological traits for germplasm evaluation
Plants with an extensive root system can absorb more nutrients under conditions of nutrient stress than can plants with a less extensive system. To survive during low phosphorus stress, plants modify their root system to increase the acquisition of phosphorus by increasing the root/shoot ratio, altering root morphology and architecture, stimulating lateral root initiation, and increasing the density and elongation of root hairs (Hammond and White 2008; Hermans et al. 2006; Jain et al. 2007; Lynch 2007). In the present study, compared with AP condition, root tips (RT), root forks (RF), root/shoot ratio (R/S) increased under NAP condition, which is consistent with previous studies. Therefore, root morphology should be considered as an important factor to evaluate germplasms. Nevertheless, very little research has been done on root morphology for evaluation of phosphorus deficiency tolerance for a long time, due to difficulty in root sampling and detection methods. With the aid of the WinRHizo digital images analysis system, this work become feasible, especially for early assessment of root system (Magalhães et al. 2011). The data measured by this analytical system is fast and accurate, and displays great efficiency in early discrimination of phosphorus deficiency tolerance genotypes.
Evaluation of phosphorus deficiency tolerance at seedling stage
A large number of studies in many plant species have shown that phosphorus supply is critical for optimum crop yield in early season, withholding phosphorus during early plant growth will limit crop growth and production from which the plant may not recover. Phosphorus limitation later in the season has a much smaller impact on crop production than do limitations early in growth (Grant et al. 2001). In other words, plants of seedling stage are sensitive to phosphorus stress and most researchers choose seedling stage as period to evaluate tolerance of nutrient deficiency (Kong et al. 2010; Zhao et al. 2011; Guo et al. 2012; Li et al. 2012; Ren 2012;). Besides, the traits related to P deficiency tolerance at seedling stage are associated with crop productivity and yield, and plants with high tolerance to P deficiency at seedling stage tend to get higher yield (Liu et al. 2005; Zhang et al. 2008; Tahir et al. 2011; Teng et al. 2013). There are several advantages for evaluation of A. tauschii genotypes at seedling stage in our study. First of all, the experiment environments can be well controlled so that uniform stress can be applied. Second, compared with the whole stage of plants, phenotypic variation caused by experimental errors can be controlled better because the plants are much more uniform at an early seedling stage. Thirdly, large-scale and high-throughput screening can be carried out in a phytotron using digital images analysis system. It greatly shorten screening period and would be labor-saving and time-saving.
Traits and their relative importance in P deficiency tolerance evaluation
There is a spate of reports about evaluation traits which associated with P tolerant genotypes in China and abroad, but, they can’t reach an agreement. In wheat, evaluation traits mainly includes the following three aspects (Sun and Zhang 2002; Bai et al. 2007): (i) root-related traits, for example, root configuration, root length, root weight, lateral root length, root tips, root forks, root diameter, root surface area, root volume and root shoot ratio; (ii) yield-related traits, such as plant height, tiller number, thousand seed weight, biomass and grain yield; (iii) physiology and biochemistry-related traits, for example, acid phosphatase activity, the total organic acid, PH of rhizosphere, chlorophyll content. At present, root or shoot dry weight (biomass traits) and grain yield become the basic and most reliable evaluation traits, root-related traits are frequently and widely used in evaluation of phosphorus deficiency tolerance in numerous crop species (Hammond et al. 2009). Application of evaluation traits are the key points of precise screening germplasm for P deficiency tolerance and statistical model is help for trait selection. Thus, PCA and multiple linear regression analysis were used in the present study. The central idea of PCA is to reduce the dimensionality of a data set consisting of a large number of interrelated variables, while retaining as much as possible of the variation present in the data set (Jolliffe 2002). In this study, the first four PCs contributed more than 80 % of total variations, and biomass factor and P obtaining factor become the main indicators, which are useful for germplasm screening under P deficiency stress. Multiple linear regression analysis shows that RL, RDW and RDM have larger contribution to TDW under both NAP and AP condition. Token together, the results suggested biomass related traits such as RDW, SDW and TDW become preferred indicators in the evaluation, which is consistent with previous conclusions (Yang et al. 2010; Hammond and White 2008; Hermans et al. 2006). In addition, root-related traits such as RSA, RV, RT and RF make a major contribution to our evaluation and they can also be used as important selection criteria in other researches.
Utilizing the resources of wheat wild relatives, A. tauschii
Wild A. tauschii related to cultivated wheat possess numerous genes of agronomic interest and can be valuable sources of resistance to diseases, pests and extreme environmental factors. These genes can be incorporated into the wheat genome via intergenetic crossing, following, where necessary, the development of chromosome addition and substitution lines from the resulting hybrids. The A. tauschii are the most closely related to wheat, exhibit great genetic diversity, the exploitation of which has been the subject of experimentation for more than a century (Annamária et al. 2008). Many of the species in the Aegilops genus have good resistance to disease and insect pests (Gill et al. 1983, 1985, 1987; Raupp et al. 1993, 1995, 1997; Zaharieva et al. 2001). Among other things, some Aegilops species are excellent sources of resistance to various rusts and powdery mildew (Damania and Pecetti 1990; Dimov et al. 1993). Many agronomically useful traits (disease and insect resistance, stress tolerance) have been incorporated into the wheat (Cox and Hatchett 1994; Raupp et al. 1993; Friebe et al. 1996).
Since 1990s, a large amount of synthetic wheat were introduced in China from International Maize and Wheat Improvement Center (CIMMYT), opened a new insight for mining good genes, breeding innovation, creating novel breeding materials and nurturing new wheat cultivars. Representatively, a new commercial wheat cultivar, Chuanmai 42 (CM42), was derived from synthetics (Triticum durum × Aegilops tauschii) by Sichuan Academy of Agricultural Sciences of China. It was resistant or immune to Chinese new stripe rust races, and was a heavy and large spike variety with high quality. The average yield of CM42 exceeded 6t/ha in regional testing of two years and it was the first officially released variety from synthetics in the world (Zhang et al. 2004a; Li et al. 2006). Recently, another new wheat cultivar, Shumai969 (unpublished, derived from synthetic wheat SHW-L1 and CM42), was authorized by Sichuan province of China, which represents big breakthrough of wheat yield in Sichuan. These successful commercial wheat cultivars suggest A. tauschii can enhance efficiency for wheat improvement. Additionally, two synthetic hexaploid wheat (Lan-2399 and Lan-2404) used in our study showed better P deficiency tolerance than other wheat, because the synthetic wheat were derived from A. tauschii crossing with tetraploid wheat. We found the tetraploid wheat had low tolerance to P deficiency, so tolerant genes for P deficiency may come from A. tauschii. Large-scale screening of A. tauschii help us to discover more tolerant gemplasms for P deficiency. CN16 is a native commercial wheat cultivar with good comprehensive agronomic characters, and it can be use as a recurrent parent for transferring excellent tolerant genes and for developing new wheat cultivars.
Judging from what we have discussed above, P deficiency tolerance ability should be one of the essential of A. tauschii, and the good genes also could be utilizing in wheat breeding. However, there is few people did screen job for phosphorus deficiency tolerance, let alone breed high P-efficient common wheat. Here, a natural A. tauschii population was used for genetic evaluation for phosphorus deficiency resistance. The results indicate the A. tauschii from China, Turkey, Uzbekistan and Azerbaijan have relatively higher P deficiency tolerance ability than that from others. Using PDTI for each traits and average S value across all the test traits, 63 A. tauschii genotypes with high tolerance under phosphorus deficiency was identified. Most of them were from Western Asia and distributed around the zone of Turkey-Iran-Afghanistan, which is in keeping with the road of A. tauschii spreading. Aegilops species are found in Mediterranean climates, being indigenous from the Canary Islands to the western part of Asia, in Afghanistan and West China (van Slageren 1994). Their ability to spread so widely was probably due to their great adaptability, which also explains why they carry so many agronomically valuable traits such as drought tolerance (Molnár et al. 2004), salt tolerance (Colmer et al. 2006) and phosphorus deficiency tolerance. These genotypes with high tolerance such as AS623282, AS623011, AS623285-1, AS623305 and AS623020 can be used as germplasm resources that could widen the genetic diversity of cultivated wheat, as a result, to shorten the procedure of wheat breeding for tolerance to phosphorus deficiency.
In conclusion, we systematically evaluated large-scale A. tauschii genotypes, and several tolerant and sensitive genotypes were selected. Root related traits used in the study such as RDW, SDW, TDW RSA, RV, RT and RF could be used as selection criteria to evaluate and screen other wheat related germplasm resources. The top five tolerant A. tauschii genotypes, AS623282, AS623011, AS623285-1, AS623305 and AS623020 that have relative higher tolerance than other wheat can be further used for wheat genetic improvement and molecular breeding for abiotic stress tolerance.
References
Annamária S, Istvá M, Márta ML (2008) Utilisation of Aegilops (goatgrass) species to widen the genetic diversity of cultivated wheat. Euphytica 163:1–19
Bai DY, Feng GH, Zhang HY, Chen RZ, Wang XJ (2007) Screening of wheat genotypes with high phosphorus efficiency under low phosphorus stress. J Triticeae Crops 27:407–410
Bouma TJ, Nielsen KL, Koutstaal B (2000) Sample preparation and scanning protocol for computerized analysis of root length and diameter. Plant Soil 218:185–196
CAY (1991) China Agriculture Yearbook. Chinese Agricultural Press, Beijing
CAY (2006) China Agriculture Yearbook. Chinese Agricultural Press, Beijing
Colmer TD, Flowers TJ, Munns R (2006) Use of wild relatives to improve salt tolerance in wheat. J Exp Bot 57:1059–1078
Cox TS, Hatchett JH (1994) Hessian fly resistance gene H26 transferred from Triticum tauschii to common wheat. Crop Sci 34:958–960
Cox TS, Raupp WJ, Wilson DL, Gill BS, Leath S, Bockus WW et al (1992) Resistance to foliar diseases in a collection of Triticum tauschii germplasm. Plant Dis 76:1061–1064
Cui DQ, Hao X, Li Y, Liu YY (2004) Progress of genetic study on phosphorus efficiency of wheat. J Triticeae Crops 24:110–113
Damania AB, Pecetti L (1990) Varialility in a collection of Aegilops species and evaluation for yellow rust resistance at two location in northern Syria. J Genet Breed 44:97–102
Dimov A, Zaharieva M, Mihova S (1993) Rust and powdery mildew resistance in Aegilops accessions from Bulgaria. In: Damania AB (ed) Biodiversity and wheat improvement. Wiley, New York, pp 165–169
Draper NR, Smith H (1966) Applied regression analysis. Wiley, New York, p 7407
Friebe B, Jiang J, Raupp WJ, McIntosh RA, Gill BS (1996) Characterization of wheat alien translocations conferring resistance to diseases and pests: current status. Euphytica 71:59–83
George TS, Richardson AE (2008) Potential and limitations to improving crops for enhanced phosphorus utilization. In: White PJ, Hammond JP (eds) Ecophysiology of plant-phosphorus interactions. Springer, Dordrecht, pp 247–270
Gill BS, Browder LE, Hatchett JH, Harvey TL, Martin TJ, Raupp WJ, Sharma HC, Waines JG (1983) Disease and insect resistance in wild wheats. In: Sakamoto S (ed) Proc 6th Int Wheat Genet Symp, Faculty of Agriculture, Kyoto University, Japan, pp 783–792
Gill BS, Sharma HC, Raupp WJ, Browder LE, Hatchett JH, Harvey TL (1985) Evaluation of Aegilops species for resistance to wheat powdery mildew, wheat leaf rust, Hessian fly and greenbuy. Plant Breed 69:314–316
Gill BS, Hatchett JH, Raupp WJ (1987) Chromosomal mapping of Hessian fly resistance gene H13 in the D genome of wheat. J Hered 78:97–100
Grant CA, Flaten DN, Tomasiewicz DJ, Sheppard SC (2001) The importance of early season phosphorus nutrition. Can J Plant Sci 81:211–224
Guo Y, Kong FM, Xu YF, Zhao Y, Liang X, Wang YY et al (2012) QTL mapping for seedling traits in wheat grown under varying concentrations of N, P and K nutrients. Theor Appl Genet 124:851–865
Hammond JP, White PJ (2008) Sucrose transport in the phloem: integrating root responses to phosphorus starvation. J Exp Bot 59:93–109
Hammond JP, Broadley MR, White PJ, King GJ, Bowen HC, Hayden R, Meacham MC, Mead A, Overs T, Spracklen WP, Greenwood DJ (2009) Shoot yield drives phosphorus use efficiency in Brassica oleracea and correlates with root architecture traits. J Exp Bot 60:1953–1968
Hermans C, Hammond JP, White PJ, Verbruggen N (2006) How do plants respond to nutrient shortage by biomass alocaion? Trends Plant Sci 11:610–617
Hoagland DR, Arnon IR (1950) The water culture method for growing plants without soils. Circ Calif Agric Exp Stn 347:4–32
Horst WJ, Abdou M, Wiesler F (1993) Genotypic differences in phophorus efficiency of wheat. Plant Soil 155:293–296
Iyamuremye F, Dick RP, Baham J (1996) Organic amendment and phosphorus dynamics: II. Distribution of soil phosphorus fractions. Soil Sci 161:436–443
Jain A, Vasconcelos MJ, Raghothama KG, Sahi SV (2007) Molecular mechanisms of plant adaptation to phosphate deficiency. In: Janick J (ed) Plant breeding reviews. Wiley, NJ, pp 359–419
Jolliffe IT (2002) Principal component analysis, 2nd edn. Springer, New York, pp 1–10
Kong ZX, Yang LL, Zhang ZZ, Ma ZQ (2010) Screening of wheat germplasms tolerant to low phosphorus. J Triticeae Crops 30:591–595
Levent O, Selim E, Bulent T, Ismail C (2005) Variation in phosphorus efficiency among 73 bread and durum wheat genotypes grown in a phosphorus-deficient calcareous soil. Plant Soil 269:69–80
Li YF, Luo AC, Wang WM, Cai BX, Hu XY, Yang X (2005) Genotypic variation of rice in yield, phosphorus uptake and utilization at different phosphorus supply. Chin J Soil Sci 36:365–368
Li GQ, Li ZF, Yang WY, Zhang Y, He ZH, Xu SC, Singh RP, Qu YY, Xia XC (2006) Molecular mapping of stripe rust resistance gene YrCH42 in Chinese wheat cultivar Chuanmai 42 and its allelism with Yr 24 and Yr26. Theor Appl Genet 112:1434–1440
Li CY, Zhang H, Ma L, Li C (2012) Comprehensive evaluation on physiological indices of nitrogen absorbion and utilization in winter wheat at the seedling stage. Plant Nutr Fertil Sci 18:523–530
Liu Y, Li ZC, Mi GH, Zhang HL, Mu P, Wang XK (2005) Screening and identification for tolerance to low-phosphorus stress of rice germplasm (Oryza sativa L.). Acta Agron Sin 31:238–242
Lu DL, Yan FB, Lu WP (2010) Genotypic difference of phosphorus uptake and utilization in sweet maize varieties. J Yangzhou Univ 31:60–64
Lynch JP (2007) Roots of the second green revolution. Aust J Bot 55:493–512
Ma WQ, Zhang FS, Zhang WF (2005) Fertilizer production and consumption and the resources, environment, food security and sustainable development in China. Resour Sci 27:33–40
Magalhães PC, De Souza TC, Cantão FRO (2011) Early evaluation of root morphology of maize genotypes under phosphorus deficiency. Plant Soil Environ 57:135–138
Marschner H (1995) Functions of mineral nutrients: macronutrients. Miner Nutr High Plants 2:379–396
Molnár I, Gáspár L, Sárvári É, Dulai S, Hoffmann B, Molnár-Láng M, Galiba G (2004) Physiological and morphological responses to water stress in Aegilops biuncialis and Triticum aestivum genotypes with differing tolerance to drought. Funct Plant Biol 31:1149–1159
Raghothama KG, Karthikeyan AS (2005) Phosphate acquisition. Plant Soil 274:37–49
Raupp WJ, Amri A, Hatchett JH, Gill BS, Wilson DL, Cox TS (1993) Chromosomal location of Hessian fly-resistance genes H22, H23 and H24 derived from Triticum tauschii in the D genome of wheat. J Hered 84:142–145
Raupp WJ, Gill BS, Friebe B, Wilson DL, Cox TS, Sears RG (1995) The Wheat Genetic Resource Center: Germplasm conservation, evaluation and utilization. Proc 8th Int Wheat Genet Symp, China Agricultural Scientech Press, Beijing, China pp 469–475
Raupp WJ, Friebe B, Wilson DL, Cox TS, Gill BS (1997) Kansas State’s Wheat Genetics Resource Center provides unique oasis for germplasm research. Diversity 13:21–23
Ren YZ (2012) Effects of low phosphorus stress on different wheat varieties at seedling stage. Chin Agric Sci Bull 28:40–44
Smith SE, Kuehl RO, Ray IM, Hui R, Soleri D (1998) Evaluation of simple methods for estimating broad-sense heritability in stands of randonly planted genotypes. Crop Sci 38:1125–1129
Su JY, Xiao YM, Li M, Liu QY, Li B, Tong YP, Jia JZ, Li ZS (2006) Mapping QTLs for phosphorus-deficiency tolerance at wheat seedling stage. Plant Soil 281:25–36
Sun HG, Zhang FS (2002) Morphology of wheat roots under low-phosphorus stress. Chin J Appl Ecol 13:295–299
Tahir S, Alam SM, Ahmad Z (2011) Wheat yield and phosphorus fertilizer efficiency as influenced by pre-incubated use of single superphosphate and poultry litter and its time of application. J Plant Nutr 34:1034–1040
Teng W, Deng Y, Chen XP, Xu XF, Chen RY, Lv Y et al (2013) Characterization of root response to phosphorus supply from morphology to gene analysis in field-grown wheat. Exp Bot 64:1403–1411
Turner BL, Paphazy MJ, Haygarth PM, Mckelvie ID (2002) Inositol phosphates in the environment. Philos Trans R Soc Lond B Biol Sci 357:449–469
Valkoun J, Dostal J, Kucerová D (1990) Triticum × Aegilops hybrids through embryo culture. Biotechnol Agric For 13:152–166
Van Slageren MW (1994) Wild wheats: a monograph of Aegilops L. and Amblyopyrum (Jaub & Spach) Eig (Poaceae). Wageningen Agricultural Univerisity Papers, Wageningen, The Netherlands, pp 7–94
Vance CP (2001) Plant nutrition in a world of declining renewable resources. Plant Physiol 127:390–397
Vance CP, Uhde-Stone C, Allan DL (2003) Phosphorus acquisition and use: critical adaptations by plants for securing a nonrenewable resource. New Phytol 157:423–447
Wang XR, Shen JB, Liao H (2010) Acquisition or utilization, which is more critical for enhancing phosphorus efficiency in modern crops. Plant Sci 179:302–305
Wu ZS (1990) Wheat breeding. Chinese Agricultural Press, Beijing
Xiong YS, Yuan JF, Hao FX, Zhao SJ, Peng CL, Xu XY (2008) Advance on wheat genotype diversity with high nutrient efficiency. Hubei Agric Sci 47:1084–1087
Yang M, Ding GD, Shi L, Feng J, Xu FS, Meng JL (2010) Quantitative trait loci for root morphology in response to low phosphorus stress in Brassica napus. Theor Appl Genet 121:181–193
Yang XB, Zhang XZ, Li TX, Wu DY (2012) Differences in phosphorus utilization efficiency among wheat cultivars. Chin J Appl Ecol 23:60–66
Yen J, Yang JL (1984) Aegilops tauschii in Yili Xinjiang region. Crops J 3:1–8
Zaharieva M, Monneveux P, Henry M, Rivoal R, Valkoun J, Nachit MM (2001) Evaluation of a collection of wild wheat relative Aegilops geniculata Roth and identification of potential sources for useful traits. Euphytica 119:33–38
Zhang LM, He LY, Li JS, Xu SZ (2004a) Investigation of Maize Inbred Lines on Tolerance to Low-Phosphorus Stress at Seedling Stage. Sci Agric Sin 37:1955–1959
Zhang Y, Yang WY, Hu XR, Yu Y, Zou YC, Li QM (2004b) Anaylsis of agronomic characters of new wheat variety Chuanmai 42 derived from synthetics (Triticum duru Aegilops tauschii). Southwest China J Agric Sci 17:141–145
Zhang JH, Gao SB, Yang KC, Zhang ZM, Lin HJ, Huang N et al (2008) Screening and identification for tolerance to low phosphorus stress of Maize germplasm resources. J Plant Genet Resour 9:335–339
Zhao HT, Wang RF, Xu YF, An DG (2011) Screening and evaluating low nitrogen tolerant wheat genotype at seedling stage. Chin J Eco-Agric 19:1199–1204
Zhong GC, Mu SM, Zhang ZB (2003) Distant hybridization of triticeae. Science Press, Beijing
Acknowledgments
We thank Assad Siham (ICARDA, Syria), Jon W. Raupp (Kansas State University, USA), Shuhei Nasuda (Komugi, Japan) and Harold Bockelman (USDA, USA) for plant materials. This work was supported by the National Natural Science Foundation of China (31301317) and the National Basic Research Program of China (2014CB147200).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, L., Liu, K., Mao, S. et al. Large-scale screening for Aegilops tauschii tolerant genotypes to phosphorus deficiency at seedling stage. Euphytica 204, 571–586 (2015). https://doi.org/10.1007/s10681-014-1327-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10681-014-1327-6