Abstract
In this work, the performance of an empirical coagulation-flocculation plant to treat wastewater from a metal-mechanic industry located in an industrial park of Queretaro city, México, is studied. Wastewater samples were obtained from the homogenization tank and treated with the employed industrial reactants through an experimental jar test to obtain statistical data. Then, a response surface methodology with ANOVA analysis was used to model the process, and the ε-constraints methodology was used to optimize the coagulation-flocculation process in terms of economic and environmental impact. The results showed an improvement of phosphates removal, but a minimal increment of 1.01% of operational costs regarding to the current operating conditions. Additionally, the results offered a certain reference value for practical application of the coagulation-flocculation process using calcium hydroxide, aluminum salts, and polyacrylamide/urea for the main removal of phosphates in real effluents.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
The metal-mechanic industry widely uses the phosphate coating process to perform chemical treatment on the surface of its products [1, 2]. Therefore, the generated effluent contains large amounts of phosphates and solids. The inappropriate disposal of these effluents can lead to serious environmental problems, mainly associated to phosphate, because it causes eutrophication. This phenomenon refers to the accelerated growth of algae, causing unsuitable conditions for aquatic life and leading to the death of various aquatic species [3, 4]. In addition, it has been proven that the presence of eutrophication in the bodies of water promotes greenhouse gases [5,6,7]. Therefore, the generated effluents in the metal-mechanical industry must be treated to avoid environmental problems and to promote its reuse in production process contributing to water sustainability.
Several techniques have been employed to remove phosphates from wastewater such as coagulation-flocculation (CF), crystallization, adsorption, or magnetic separation [8, 9]; however, the CF process is the leading technology for removing phosphates from wastewater [10, 11], mainly due to its easy operation, relatively simple design, low cost, and high efficiency [12, 13]. Coagulation consists of adding an agent, commonly trivalent metal ions, or polymerized species to the wastewater to form micro-flocs that are generated by rapid mixing [14,15,16]. After that, flocculation also involves adding a chemical specie, mainly synthetic or organic polymers, so that the primary particles created in the coagulation process, form aggregates that will have a considerable volume and weight that can later be separated from the water through a sedimentation process [17, 18]. The main variables of the coagulation-flocculation process are coagulant/flocculant feed rate, mixing speed, mixing time, pH, type of effluent, and temperature [19]. Traditionally, operational variables of CF process are determined through the well-known jar test [20]. This is an empirical test which depends on the real characteristics of the effluent, so then generating a general mathematical model of the CF process that involves all these variables is a task that seems to be far from being completed [21]. Moreover, operational variables to find the optimal range entail the consumption of time and resources [22].
This last promotes that many industrial plants operate by empirical way and sometimes, environmental regulations are not complied generating environmental pollution. For this reason, it is very important to make an effort for developing or implementing mathematical or statistical models to minimize or even eliminate the empirical operation of wastewater treatment plants. So, one of the main challenges of using the coagulation-flocculation process is to find a mathematical model that can predict how the operational variables (or at least the most important ones) affect the reduction of contaminants. In this context, the response surface methodology (RSM) is an appropriate strategy to model CF process [23, 24]. RSM is a statistical technique that allows representing the influence of a set of variables over the response of interest using a minimum number of experiments [25, 26]. RSM has been proven as a reliable strategy to optimize CF process in several studies, such as effluents from the textile industry to reduce color, turbidity, and chemical oxygen demand (COD) [13, 27]; a tannery industry effluent to reduce COD [28]; a municipal wastewater to reduce turbidity, COD, and biochemical oxygen demand (BOD) [29]; an effluent obtained from a biodiesel production plant to reduce COD, turbidity, and solids [30]; an effluent from the oil industry to reduce COD, turbidity, color, and solids [18]; a surface water to reduce dissolved organic matter (DOM) [23]; and an aqueous solution to reduce hexavalent chromium (Cr(VI)) [31]. Nevertheless, the economic impact is hardly taken into account in the analysis, and real effluents of metal-mechanic industry are hardly studied, because including the economic impact implies a multi-objective optimization problem which involves a more rigorous study.
Therefore, the models generated in these studies could predict optimal solutions to avoid consumption of more resources than necessary and avoiding empirical operation to ensure environmental regulations and promoting water sustainability.
After the actual treatment of the study case, specifications of phosphate concentration are not complied, mainly because it operates by empirical way. Therefore, it is necessary to implement a robust methodology to ensure legal regulations and avoid environmental problems. Thereby, the objective of this research is the statistical modelling and multi-objective optimization in environmental and economic terms of the CF process of a wastewater treatment plant installed in a metal-mechanic industry to treat effluents with high phosphates concentration, focusing on the wastewater quality specifications without compromising operational costs. The methodology developed in this work could be employing for engineering or industrial applications.
2 Description of the Wastewater Treatment Plant
The effluent used on this study was collected directly from a wastewater treatment plant of a metal-mechanic industry located in the city of Queretaro, Mexico. Currently, the wastewater treatment plant operates with a homogenization tank, two coagulation tanks, a flocculation tank in continuous stirred mode of operation, an inclined plate settler to separate sludge from wastewater, and a filter press to compress the sludge. The process ends in a tank to store the generated sludge and a tank to store the treated water, as shown in Fig. 1. In the first coagulation stage, calcium hydroxide (CH) is added as the first coagulating agent. Then, in the second coagulation stage, sulfuric acid (SA) is added to adjust the pH, while aluminum salts (AS) are added as a second coagulant agent. Finally, in the flocculation stage, a mixture of soluble anionic polymers (copolymers of acrylamide and urea) are added as a flocculant agent. In general, the whole process takes 4.5 h to complete, maintaining an operational pH between 6 and 7. It is important to mention that the concentration of AS solution is unknown, since a real reagent employed by the study industry is being used and the information is confidential. However, it is a mixture of typical aluminum salts such as Al2(SO4)3, Al(Cl)3, or NaAlO2.
3 Materials and Methods
The sample used on this study was collected directly from the homogenization tank stage of the wastewater treatment plant. A volume of 50 L was collected in a high-density polyethylene container, and it was stored in absence of light at room temperature. The characterization of the wastewater was performed by determining the pH with a pH meter Pro + Multi2, HACH® company. Turbidity was determined with a U-10 turbidimeter, HORIBA® company. The phosphate concentration (PC) was determined through the vanadomolybdophosphoric acid method and total solids (TS) were determined with the gravimetric technique, both described in the Standard Methods for the Exam of Water and Wastewater made by American Public Health Association (APHA) [32].
An experimental jar test of four stages was carried out to obtain input parameters. In the first stage, CH was added as the first coagulant at a stirring speed of 60 rpm. In the second stage, SA was added to regulate the pH in a range of 6.5 to 7, and AS were added as a second coagulant varying rotational speeds. In the third stage, the flocculant agent was added at a stirring speed of 40 rpm. Finally, in the last stage, the sedimentation of the generated flocs occurs. All stages represent the real CF process, and reagents employed were the same used in the industrial wastewater treatment plant. It is important to mention that after several trials, the agitation speed of the first coagulation stage as well as the agitation speed of the flocculation stage remain constant. Because, in the case of the agitation speed of the flocculation stage, this must be carried out at a slow speed so as not to affect the formation of flocs, so it cannot move significantly. For the cases when coagulation processes are being carried out, it was observed that only the stage of AS has a significant influence on pollutants removal. Finally, generated sludge volume was measured through Imhoff cones.
4 Description of Response Surface Methodology and Optimization Modelling
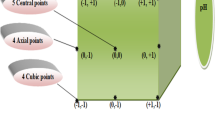
The coagulation-flocculation process was modeled considering the four most influential input variables, that are, CH dosage, AS dosage, flocculant dosage, and stirring speed of the second coagulation stage. The response surface methodology was used to correlate the interactions of the input variables with the output variables (phosphate concentration (PC), total solids (TS) and sludge generation (SG)). The composite central design (CCD) was used since it can be used for two or more variables and five levels can be analyzed for each variable [33, 34]. Table 1 shows all levels and variables analyzed. Although BBD could also be adapted to this case study, CCD was preferred because it has more levels for each variable allowing a better prediction of the behavior of the CF process. This design contains three main points:
-
1.
The factorial portion that includes a complete (or a fraction of) \({2}^{k}\) factorial design whose levels are coded as -1, 1.
-
2.
An axial portion consisting of 2 k points arranged so that two points are chosen on the axis of each control variable at a distance of α from the design center (chosen as the point at the origin of the coordinates system) and the value of α is obtained from Eq. (1). In this study, α takes a value of 2.
-
3.
\({c}_{p}\) center points.
The distance α is given by the following:
The number of experiments (N) to perform in CCD for k variables is calculated with Eq. (2):
where \({N}_{k}\) denotes the number of points in the factorial portion [33, 34]. According to Eq. 2, 36 experiments must be carried out with 4 central points for 4 variables, following the CCD matrix shown in Appendix 1 (Table A.1). STATGRAPHICS® Centurion XVI.I software was used for the design and analysis of the experiments presented in Table A.1. The experimental data were fitted to a second-order model, as shown in Eq. (3), using the least square multiple regression methodology:
where \(Y\) is the model response of interest, \({x}_{i}\) and \({x}_{j}\) are the independent variables, \({\beta }_{0}\) is the constant coefficient, \({\beta }_{i}\) is the linear coefficient, \({\beta }_{ii}\) is the quadratic coefficient, and \({\beta }_{ij}\) is the interaction coefficient. The models were validated in terms of the coefficient of determination (R2), adjusted coefficient of determination (R2-adj.), and the predicted coefficient of determination (R2-pred.) The value of R2 between 0.9 and 1 reveals a best fit of data between experimental and predicted values [29]. Besides, the probability value (P value) at 95% confidence interval was used to evaluate the significance of model terms [19].
Once the response surface models have been obtained, a multi-objective optimization problem is formulated, aiming to minimize operating costs (OC) and environmental impact (EI), measured in terms of the phosphate concentration (PC) and the sludge generation (SG), as shown in Eq. (4):
where \({C}_{PC}\) is the phosphate concentration, \({C}_{TS}\) is the total solids concentration, \({D}_{CH}\) is the dose of CH, \({D}_{AS}\) is the dose of aluminum salts, \({D}_{F}\) is the dose of flocculant, \({E}_{C}\) is the energy consumed, \({D}_{SS}\) the stirring speed, and \({G}_{S}\) the generation of sludge.
The optimization problem has been coded in GAMS® 34.2.0 to perform the multi-objective optimization. This tool is the most used to solve multi-objective optimization problems [35, 36]. The Pareto front was constructed through the ε-constraints method. This technique is based on selecting one of the objective functions to be optimized, while the others are converted into constraints by establishing an upper limit (ε1,….εl) for each of the objective functions [36]. The construction of the Pareto front was performed, with around 100 points, and the Hammersley sampling technique was employed. In this work, the cost function is selected as the main objective. Thus, the optimization problem is reformulated as follows:
In general, the best solution was obtained by normalizing each objective function (OC, PC and SG) with Eq. (6):
where \({z}_{i}\) is the i-th objective function, while the superscripts lo and up refer to the lower and upper bounds of the objective \({z}_{i}\), respectively. To find the solution with the best compromise between the objective functions, the Euclidean distance theorem was applied, as shown in Eq. (7):
This solution will be the one with the smallest distance between all points.
5 Results and discussion
The wastewater was characterized, and pH, turbidity, PC and TS were found to be 6.7 ± 0.01, 25 ± 0.75 NTU, 96.09 ± 0.86 mg L−1 and 690 ± 14 mg L−1, respectively. It is important to emphasize that wastewater samples do not present suspended solids, and TS are associated only with the amount of dissolved solids including phosphates and traces of other metals such as Fe.
The relationship between the four independent variables (CH dosage, AS dosage, flocculant dosage, and stirring speed) and their responses (PC, TS, and SG) was analyzed through the CCD matrix. The results obtained from the three responses were fitted to a second-order mathematical model given by the following equations:
The results of the experiments as well as the results of the predictions of the generated mathematical models are shown in Appendix 1 (Table A.1). According to the experimental results of Table A.1, it is possible to reduce the PC by a maximum of 90.00% and a minimum of 51.10%, while for the concentration of TS, it increased from a minimum of 188.40% to a maximum of 381.15%. The increase in the concentration of TS is because not all of the CH and AS interact in the process, so it remains in the water as dissolved solids. Finally, for the generation of sludge, there is a minimum generation of 16 mL L−1 and a maximum generation of 94 mL L−1. It should be noted that turbidity was not considered in this study, since in all performed experiments, it exceeded 99% removal (not shown in this work) and wastewater composition does not have significant suspended solids concentration.
Results of ANOVA are shown in Appendix 2 (Table A.2). A P value (or Prob. > F) is the probability that the result will equal or exceed the value that was observed if the model produces accurate results. If the model Prob. > F, and no term exceeds the level of significance (e.g., c = 0.05), the model can be considered acceptable within a confidence interval of (1 − c) [19]. Examining the relevance of the obtained parameters (P value < 0.05) in Table A.2, it is observed that \({X}_{1}\), \({X}_{2}\), \({X}_{3}\), \({X}_{4},\) and \({X}_{2}{X}_{2}\) are the most significant elements for PC, being aluminum salts dose the most influential variable, while for TS, \({X}_{1}\), \({X}_{2}\), \({X}_{3}\), \({X}_{4}\), \({X}_{1}{X}_{2}, {X}_{1}{X}_{4},{X}_{2}{X}_{4,}\) and \({X}_{3}{X}_{3}\), all variables are considerably influential. Finally, for SG, \({X}_{1}\), \({X}_{2}\), \({X}_{3}\), \({X}_{4}, {X}_{1}{X}_{1}\), and \({X}_{2}{X}_{2}\) are the most significant elements, being calcium hydroxide dose and aluminum salt dose, the most influential variables. The three-response regression results of ANOVA confirm that all generated models are significant, with high F values (779.41, 634.39, and 472.12 for PC, TS, and SG, respectively) and low P values (< 0.05 for all models). Therefore, the developed models were statistically significant within ± 5% [24, 29]. In order to evaluate the quality of the efficiency of the generated models, the coefficient of determination (R2) was used, finding values very close to 1 (0.976 for FC, 0.970 for TS and 0.997 for SG). On the other hand, the obtained adjusted determination coefficients (R2-adj) were also quite good (0.956 for FC, 0.946 for TS and 0.994 for SG). Thereby, a good correlation between the generated predictive model and the experimental values is ensured.
Figure 2 shows how much of the independent variables affect the different responses. It is observed in Fig. 2a that the dose of aluminum salts presents a significant effect over other variables, this is due to the fact that AS is the coagulant with the most interaction with contaminants, because with AS, a 98% removal of phosphates can be achieved [8]; in addition, CH is used only as a coagulant aid [37, 38]. In the case of total solids, it is observed in Fig. 2b that the main interaction variable is calcium hydroxide, because it promotes an increase or decrease of this variable due to the fact that not of the whole reactant interacts with the contaminants and it remains in the solution in the form of ions. Finally for the generation of sludge, the dose of aluminum salts predominates, since large amounts of sludge could be generated when AS is used as a coagulant [39].
Response surface graphs were made with the variables with highest interaction, and they are shown in Fig. 3. In Fig. 3a, It is observed that the minimum removal of phosphates considering only the effects of CH and AS is 39.64%, and the maximum removal is 86.48%, while by varying CH and AS, a minimum removal of 50.47% and a maximum removal of 93.76% are achieved. In Fig. 3b, it is observed that the minimum and maximum values for the concentration of total solids considering only the effects of CH and stirring speed present a decrease of 27.54% and an increase of 304.34%, respectively. Meanwhile, by varying only the CH and the flocculant, a maximum value of 347.82% and a minimum value of 123.18% were obtained. Figure 3c shows that analyzing only the effects of CH and AS, the minimum and maximum amount of sludge are 5 and 100 mL L−1, respectively. On the other hand, by varying only the AS and the flocculant, a minimum and a maximum of 20 and 110 mL L−1 are achieved. It is confirmed that phosphate removal, sludge generation and total solids removal present a complex interaction, and its behavior is difficult to predict by empirical operation.
A Pareto front was generated for the development of a multi-objective optimization. As a first step, the limits of the objective functions were established through their individual optimization, as shown in Table 2. Once the limits of each objective function were obtained, 100 points of the Pareto front were obtained and represented graphically in Fig. 4. Analyzing the results shown in Fig. 4, the operating costs are strongly related to the PC, since when the operating cost is higher, the PC increases and vice versa, and as the same for total solids. On the other hand, having low values of PC, the concentration of total solids increases. The Euclidean distance theorem was used to obtain the point with the best compromise between objective functions, then an optimal point was found with PC at 23,839 mg L−1, TS at 1611,416 mg L−1, and OC at 7.91 USD/day. The convergence time was 0.594 s for the point found performing a total of 13 iterations. For this optimal point, operational variables CH, AS, flocculant, and stirring speed were obtained at 400 mg L−1, 0.723 mL L−1, 3.32 × 10–3 mg L−1, and 100 rpm, respectively. A phosphate removal efficiency of 75.20% was achieved.
Operational variables currently used in the wastewater treatment plant and optimal values obtained in this work are shown in Table 3. According to these values, an operational cost of 7.80 USD per day of operation is obtained with concentrations of 36.07 mg L−1 of phosphates and 890 mg L−1of total solids. The obtained phosphate concentration after the current process of the wastewater treatment plant is above the limit allowed by the internal regulations of the industrial park (< 30 mg L−1), so its current process conditions are not adequate to comply with the required regulation. According to the results obtained by modelling, it was found that the current process is cheaper than the optimal one found (with a difference of 1.01% per day) and an increase in the concentration of total solids (of 233.53%). Nevertheless, the cost increment is not significant, and the phosphates concentration decreases to comply with the regulation, which is the most important objective for this particular metal-mechanic industry. It is important to remark that the increment of total solids is not environmental harmful according with the local environmental regulations of this particular industrial park, because its mainly composition are non-toxic dissolve salts and non-toxic metal traces, such as Fe. Besides, the concentration of aluminum ions (Al3+) reported in the residual water after the treatment is 0.03 ± 0.004 mg/L, which is a very low value for aluminum concentration. Individual studies must be performed for other study cases, and the methodology presented in this work could be taken as a first keystone.
Table 4 shows other studies of different coagulation processes to remove phosphates, where it is shown that there are processes with more removal efficiency and others with less removal efficiency. However, these do not consider the economic impact, which makes the main difference of this work with the others [11, 40,41,42].
6 Conclusions
In this work, statistical modeling and optimization methodology of a CF process of a metal-mechanic industry have been presented. The generated mathematical model of the CF process was performed with RSM employing a CCD, and the input data was obtained from an experimental jar test method using a real sample from the metal-mechanic industry. Then, a Pareto front was generated using the ε-constraint method to perform a multi-objective optimization. The results of this investigation reveal optimal operating conditions with 400 mg L−1 of CH, 0.723 mL L−1 of AS, 3.32 × 10–3 mg L−1 of flocculant, and 100 rpm agitation speed. The analyzed objectives found their best compromise in 23.839 mg L−1 of PC, 1611.416 mg L−1 of TS, and 7.91 USD/day of operating cost. The results obtained indicate that the CF process is effective to reduce PC and is suitable to treat effluents from the metal-mechanic industry. On the other hand, it was found that the wastewater treatment plant of the study case needs to change its operating conditions to comply with the regulations required by the industrial park where it is located, with a low increment of 1.01% of the cost of operation and an increment of 181% of total solids concentration. The proposed methodology could be applied in effluents with high phosphates content for engineering or industrial applications.
Availability of Data and Material
Not applicable.
Code Availability
Not applicable.
References
Duszczyk, J., Siuzdak, K., Klimczuk, T., Strychalska-Nowak, J., & Zaleska-Medynska, A. (2018). Manganese phosphatizing coatings: The effects of preparation conditions on surface properties. Materials (Basel), 11, 1–22. https://doi.org/10.3390/ma11122585
Statsyuk, V., Fogel, L., Bold, A., Sultanbek, U., Ait, S., & Sassykova, L. (2020). Protective properties of phosphate coatings based on rust converters. Journal of Chemical Technology and Metallurgy, 55, 2151–2157.
Humphrey, C. P. (2022). Phosphate treatment by five onsite wastewater systems in a nutrient sensitive watershed. Earth, 3, 683–698. https://doi.org/10.3390/earth3020039
Schindler, D. W. (2012). The dilemma of controlling cultural eutrophication of lakes. Proceedings of the Royal Society B: Biological Sciences, 279, 4322–4333. https://doi.org/10.1098/rspb.2012.1032
Huttunen, J. T., Alm, J., Liikanen, A., Juutinen, S., Larmola, T., Hammar, T., Silvola, J., & Martikainen, P. J. (2003). Fluxes of methane, carbon dioxide and nitrous oxide in boreal lakes and potential anthropogenic effects on the aquatic greenhouse gas emissions. Chemosphere, 52, 609–621. https://doi.org/10.1016/S0045-6535(03)00243-1
Li, Y., Shang, J., Zhang, C., Zhang, W., Niu, L., Wang, L., & Zhang, H. (2021). The role of freshwater eutrophication in greenhouse gas emissions: A review. Science of the Total Environment, 768, 144582. https://doi.org/10.1016/j.scitotenv.2020.144582
West, W. E., Creamer, K. P., & Jones, S. E. (2016). Productivity and depth regulate lake contributions to atmospheric methane. Limnology and Oceanography, 61, S51–S61. https://doi.org/10.1002/lno.10247
Gong, Y., & Zhao, D. (2014). Physical-chemical processes for phosphorus removal and recovery. Comprehensive Water Quality and Purification, Elsevier Inc. https://doi.org/10.1016/B978-0-12-382182-9.00086-4
Lai, Y. T., Huang, Y. S., Chen, C. H., Lin, Y. C., Jeng, H. T., Chang, M. C., Chen, L. J., Lee, C. Y., Hsu, P. C., & Tai, N. H. (2020). Green treatment of phosphate from wastewater using a porous bio-templated graphene oxide/MgMn-layered double hydroxide composite. iScience, 23, 10106. https://doi.org/10.1016/j.isci.2020.101065
Morse, G. K., Brett, S. W., Guy, J. A., & Lester, J. N. (1998). Review: Phosphorus removal and recovery technologies. Science of the Total Environment, 212, 69–81. https://doi.org/10.1016/S0048-9697(97)00332-X
Sibiya, N. P., Amo-Duodu, G., Tetteh, E. K., & Rathilal, S. (2022). Model prediction of coagulation by magnetised rice starch for wastewater treatment using response surface methodology (RSM) with artificial neural network (ANN). Scientific African, 17, e01282. https://doi.org/10.1016/j.sciaf.2022.e01282
Ozairi, N., Mousavi, S. A., Samadi, M. T., Seidmohammadi, A., & Nayeri, D. (2020). Removal of fluoride from water using coagulation–flocculation procea comparative study. Desalinization and Water Treatment, 180, 265–270. https://doi.org/10.5004/dwt.2020.25064
Sakhi, D., Elmchaouri, A., Rakhila, Y., Abouri, M., Souabi, S., Hamdani, M., & Jada, A. (2020). Optimization of the treatment of a real textile wastewater by coagulation–flocculation processes using central composite design. Desalinization and Water Treatment, 196, 33–40. https://doi.org/10.5004/dwt.2020.25929
Watanabe, Y. (2017). Flocculation and me. Water Research, 114, 88–103. https://doi.org/10.1016/j.watres.2016.12.035
Mahmoudabadi, Z. S., Rashidi, A., & Maklavany, D. M. (2022). Optimizing treatment of alcohol vinasse using a combination of advanced oxidation with porous α-Fe2O3 nanoparticles and coagulation-flocculation. Ecotoxicology and Environmental Safety, 234, 113354. https://doi.org/10.1016/j.ecoenv.2022.113354
Okolo, B. I., Adeyi, O., Oke, E. O., Agu, C. M., Nnaji, P. C., Akatobi, K. N., & Onukwuli, D. O. (2021). Coagulation kinetic study and optimization using response surface methodology for effective removal of turbidity from paint wastewater using natural coagulants. Scientific African, 14, e00959. https://doi.org/10.1016/j.sciaf.2021.e00959
Al-Risheq, D. I. M., Shaikh, S. M. R., Nasser, M. S., Almomani, F., Hussein, I. A., & Hassan, M. K. (2022). Influence of combined natural deep eutectic solvent and polyacrylamide on the flocculation and rheological behaviors of bentonite dispersion. Separation and Purification Technology, 293, 121109. https://doi.org/10.1016/j.seppur.2022.121109
Singh, B., & Kumar, P. (2020). Pre-treatment of petroleum refinery wastewater by coagulation and flocculation using mixed coagulant: Optimization of process parameters using response surface methodology (RSM). Journal of Water Process Engineering, 36, 101317. https://doi.org/10.1016/j.jwpe.2020.101317
Bobadilla, M. C., Lorza, R. L., García, R. E., Gómez, F. S., & González, E. P. V. (2019). Coagulation: Determination of key operating parameters by multi-response surface methodology using desirability functions. Water (Switzerland), 11, 1–21. https://doi.org/10.3390/w11020398
Marín-Galvín, R. (1998). “Jar-test” in water treatment: A valuable tool. Tecnología del agua, 181, 25–34.
Kadooka, H., Miyajima, T., Tanaka, T., Jami, M. S., & Iwata, M. (2017). Simplified flocculation model for inorganic and polymer flocculants. Separation Science and Technology, 52, 2802–2814. https://doi.org/10.1080/01496395.2017.1335322
Zangooei, H., Delnavaz, M., & Asadollahfardi, G. (2016). Prediction of coagulation and flocculation processes using ANN models and fuzzy regression. Water Science and Technology, 74, 1296–1311. https://doi.org/10.2166/wst.2016.315
Khettaf, S., Bouhidel, K. E., Khouni, I., Louhichi, G., Ghrabi, A., Bousselmi, L., & Bouhelassa, M. (2021). Optimization of coagulation-flocculation process in the treatment of surface water for a maximum dissolved organic matter removal using RSM approach. Water Supply, 21, 3042–3056. https://doi.org/10.2166/ws.2021.070
Nayeri, D., & Mousavi, S. A. (2020). Treatment of highly turbid water in disaster conditions using coagulation-flocculation process: Modeling and optimization. Water Quality Research Journal, 55, 358–369. https://doi.org/10.2166/wqrj.2020.120
Iloamaeke, I. M., Nnaji, N. J., Okpala, E. C., Eboatu, A. N., & Onuegbu, T. U. (2021). Mercenaria mercenaria shell: Coagulation-flocculation studies on colour removal by response surface methodology and nephlometric kinetics of an industrial effluent. Journal of Environmental Chemical Engineering, 9, 105715. https://doi.org/10.1016/j.jece.2021.105715
Myers, R. H., & Montgomery, D. C. (1971) Response surface methodology: Process and product optimization using designed experiments, A Wiley-Interscience publication.
Dotto, J., Fagundes-Klen, M. R., Veit, M. T., Palácio, S. M., & Bergamasco, R. (2019). Performance of different coagulants in the coagulation/flocculation process of textile wastewater. Journal of Cleaner Production, 208, 656–665. https://doi.org/10.1016/j.jclepro.2018.10.112
Pinto, M. B., Samanamud, G. R. L., Baston, E. P., França, A. B., Naves, L. L. R., Loures, C. C. A., & Naves, F. L. (2019). Multivariate and multiobjective optimization of tannery industry effluent treatment using Musa sp flower extract in the coagulation and flocculation process. Journal of Cleaner Production, 219, 655–666. https://doi.org/10.1016/j.jclepro.2019.02.060
Joaquin, A. A., & Nirmala, G. (2019). Statistical modeling and process optimization of coagulation–flocculation for treatment of municipal wastewater. Desalinization and Water Treatment, 157, 90–99. https://doi.org/10.5004/dwt.2019.24162
Daud, N. M., Sheikh-Abdullah, S. R., & Abu-Hasan, H. (2018). Response surface methodological analysis for the optimization of acid-catalyzed transesterification biodiesel wastewater pre-treatment using coagulation–flocculation process. Process Safety and Environmental Protection, 113, 184–192. https://doi.org/10.1016/j.psep.2017.10.006
Benmahdi, F., Khettaf, S., & Kolli, M. (2022). Efficient removal of Cr(VI) from aqueous solution using activated carbon synthesized from silver berry seeds: modeling and optimization using central composite design. Biomass Conversion and Biorefinery, 1–15. https://doi.org/10.1007/s13399-022-03041-8
Rice, E. W., Bridgewater, L., Association, A. P. H., Association, A. W. W., & Federation W. E. (2012). Standard methods for the examination of water and wastewater. American Public Health Association.
Bezerra, M. A., Santelli, R. E., Oliveira, E. P., Villar, L. S., & Escaleira, L. A. (2008). Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta, 76, 965–977. https://doi.org/10.1016/j.talanta.2008.05.019
Khuri, A. I., & Mukhopadhyay, S. (2010). Response surface methodology. Wiley Interdisciplinary Reviews: Computational Statistics, 2, 128–149. https://doi.org/10.1002/wics.73
Achenie, L. E. K., Sharifi, Y., & Lee, D. G. (2016). Development of a multiscale strategy and application to chemical vapor deposition. Computer Aided Chemical Engineering, 39, 95–123. https://doi.org/10.1016/B978-0-444-63683-6.00004-6
Arora, J. S. (2017). Multi-objective optimum design concepts and methods, introduction to optimum design. Elsevier Inc. https://doi.org/10.1016/b978-0-12-800806-5.00018-4
Abdul-majeed, B. A., & Oleiwi, H. B. (2015). Evaluation of alum/lime coagulant for the removal of turbidity from Al- Ahdab Iraqi oilfields produced water. Journal of Engineering, 21, 145–153.
Duan, J., Cao, X., Chen, C., Shi, D., Li, G., & Mulcahy, D. (2012). Effects of Ca(OH)2 assisted aluminum sulfate coagulation on the removal of humic acid and the formation potentials of tri-halomethanes and haloacetic acids in chlorination. Journal of Environmental Science (China), 24, 1609–1615. https://doi.org/10.1016/S1001-0742(11)60953-4
Joshi, S., & Shrivastava, K. (2011). Recovery of alum coagulant from water treatment plant sludge: A greener approach for water purification. International Journal of Advanced Computer Research, 1, 101–103.
Smotraiev, R., Nehrii, A., Koltsova, E., Anohina, A., Sorochkina, K., & Ratnaweera, H. (2022). Comparison of wastewater coagulation efficiency of pre-polymerised zirconium and traditional aluminium coagulants. Journal of Water Process Engineering, 47(May), 102827. https://doi.org/10.1016/j.jwpe.2022.102827
Sibiya, N. P., Amo-Duodu, G., Kweinor Tetteh, E., & Rathilal, S. (2022). Response surface optimisation of a magnetic coagulation process for wastewater treatment via Box-Behnken. Materials Today: Proceedings, 62, S122–S126. https://doi.org/10.1016/j.matpr.2022.02.098
Chen, Y., Long, J., Chen, S., Xie, Y., Xu, Z., Ning, Z., Zhang, G., Xiao, T., Yu, M., Ke, Y., Peng, L., & Li, H. (2022). Multi-step purification of electrolytic manganese residue leachate using hydroxide sedimentation, struvite precipitation, chlorination and coagulation: Advanced removal of manganese, ammonium, and phosphate. Science of the Total Environment, 805, 150237. https://doi.org/10.1016/j.scitotenv.2021.150237
Acknowledgements
The authors are grateful to Universidad de Guanajuato for facilities to develop this work and to the company for the information and wastewater samples. Also, Carlos Gómez Rodríguez is grateful to CONACyT for the scholarship granted for his Master’s degree studies.
Funding
This work did not receive any funding source.
Author information
Authors and Affiliations
Contributions
C.G.-R. performed the experiments and wrote the first version of the manuscript. T.P. and F.G.-C. implemented the experiments and the mathematical modelling. Z.G.-A. provided chemicals and laboratory equipment. T.P. wrote the final version of the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
All the material is owned by the authors, and no permissions are required.
Competing Interests
All authors have no competing interests or other interests that might be perceived to influence the results and/or discussion reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gómez-Rodríguez, C., Gómez-Castro, F.I., Gamiño-Arroyo, Z. et al. Modeling and Optimization of Coagulation-Flocculation Process to Remove High Phosphate Concentration in Wastewater from a Metal-Mechanic Industry. Environ Model Assess 29, 845–854 (2024). https://doi.org/10.1007/s10666-024-09967-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10666-024-09967-9