Abstract
The study attempted to evaluate the agricultural soil quality using the Soil Quality Index (SQI) model in two Community Development Blocks, Ausgram-II and Memari-II of Purba Bardhaman District. Total 104 soil samples were collected (0–20 cm depth) from each Block to analyse 13 parameters (bulk density, soil porosity, soil aggregate stability, water holding capacity, infiltration rate, available nitrogen, available phosphorous, available potassium, soil pH, soil organic carbon, electrical conductivity, soil respiration and microbial biomass carbon) in this study. The Integrated Quality Index (IQI) was applied using the weighted additive approach and non-linear scoring technique to retain the Minimum Data Set (MDS). Principal Component Analysis (PCA) identified that SAS, BD, available K, pH, available N, and available P were the key contributing parameters to SQI in Ausgram-II. In contrast, WHC, SR, available N, pH, and SAS contributed the most to SQI in Memari-II. Results revealed that Ausgram-II (0.97) is notably higher SQI than Memari-II (0.69). In Ausgram-II, 99.72% of agricultural lands showed very high SQI (Grade I), whereas, in Memari-II, 49.95% of lands exhibited a moderate SQI (Grade III) and 49.90% showed a high SQI (Grade II). Sustainable Yield Index (SYI), Sensitivity Index (SI) and Efficiency Ratio (ER) were used to validate the SQIs. A positive correlation was observed between SQI and paddy ( R2 = 0.82 & 0.72) and potato yield (R2 = 0.71 & 0.78) in Ausgram-II and Memari-II Block, respectively. This study could evaluate the agricultural soil quality and provide insights for decision-making in fertiliser management practices to promote agricultural sustainability.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Globally, the primary concern is food requirements, leading to land-use changes (Allen et al., 2011). In just 12 years, the population grew from 7 to 8 billion, exerting immense pressure on agriculture and questioning sustainable land management (Rudel et al., 2016; Zeifman et al., 2022; Kaur & Chauhan, 2023). By 2050, it is projected that the world’s food consumption will have increased by around 60%, while one-third of the global soil is undergoing moderate to severe degradation (Bisht & Chauhan, 2020; Tahat et al., 2020). It becomes a huge challenge to meet this massive demand for food while minimising the harmful effect of intensive agricultural systems on soil quality (Tang et al., 2022). Maintaining good soil quality enhances crop production, environmental quality, and overall health for plants, animals, and humans (Sharma et al., 2005). Measurement of soil quality considering soil properties provides an early warning to implement sustainable land management practices (Rahmanipour et al., 2014; Guillot et al., 2021; Choudhury & Mandal, 2021). To evaluate agricultural sustainability, it is crucial to determine the soil properties causing changes in soil quality. These qualities may eventually be identified as soil quality indicators (Masto et al., 2007).
The term “soil quality” was first propounded by Warkentin and Fletcher in 1977 (Rattan, 2015; Wienhold et al., 2004). They emphasised the significance of soil as a resource for purposes beyond agricultural production, waste recycling, and recreational uses (Karlen et al., 2001). The concept of soil quality and its evolution was diffused from North America (Klingebiel & Montgomery, 1961). In the 1980s, North American scientists primarily focused on soil erosion and its impact on production. However, they shifted their attention towards soil quality, sustainable agriculture and environmental health in the following decades (Karlen et al., 2001). Soil Science Society of America (SSSA) defined soil quality as “the capacity (soil) to function.” Later on, Karlen et al., (1997) extended the definition of soil quality as “the capacity of specific kind of soil to function, within natural or managed ecosystem boundaries, to sustain plant and animal productivity, maintain or enhance water and air quality, and support human health and habitation.” Therefore, the main objectives of a proper soil quality assessment are to ensure environmental quality, promote agronomic sustainability, and support socio-economic vitality (Andrews et al., 2002). Soil quality depends on inherent (subsurface) and dynamic (surface) processes. Inherent processes change more naturally, as Jenny (1941) attempted to explain, and these changes tend to happen gradually. In contrast, dynamic changes are influenced more by human decisions, which can be referred to as land management systems (Larson & Pierce, 1994; Karlen et al., 2001). Numerous methods for evaluating soil quality have developed. Larson and Pierce, (1991) were among the first to propose a quantitative formula for measuring soil quality (Karlen et al., 1999; Liebig & Doran, 1999); along with this, spatial variability of soil quality was also introduced (Smith et al., 1993).
For several decades in tropical countries, including India, research has focused on plant biomass, soil productivity, and assessing specific soil-deficient nutrients (Masto et al., 2007). However, in recent times, there has been a shift towards agricultural sustainability, as numerous studies have indicated a decline in productivity resulting from long-term poor land management practices (Bhattacharyya et al., 2015; Srinivasarao et al., 2013; Srivastava et al., 2016). For the first time, the declining productivity of rice was noticed in India with the rice–wheat system (Nambiar & Ghosh, 1984; Nambiar, 1995) and in Philippines with the rice-rice cropping system (Flinn & De Datta, 1984). Farmers have adopted different nutrient management practices in various countries based on their food demands, land size, and available agrochemicals. The application rate (kg/ha) of primary nutrients (N-P2O5-K2O) has been found to be highest in Columbia (615), followed by Egypt (543), China (375), Brazil (369), Chile (359) and Uzbekistan (296) in 2021 (The World Bank, 2023). Whereas India uses 193 kg/ha. In India, the usage of chemical fertilisers (NPK) has risen from 2.65 million tons in 1971–1972 to 41.4 million tons in 2018–2019 (Pahalvi et al., 2021). In the case of fertiliser management practices, NPK + Farm Yard Manure (FYM) has been identified as significantly positive in correlation with the Soil Quality Index (SQI) (Choudhary et al., 2018; Majhi et al., 2019; Manna et al., 2005). While some studies have shown a higher SQI in NPK + Crop residue (Rakshit et al., 2018), the absence of FYM only reduces the SQI by 23.3%. This suggests that applying FYM with NPK positively effects soil health (Saha et al., 2022). However, higher rice production was found under chemical fertiliser alone, followed by Integrated Nutrient Management (INM) in the alkaline soil of India (Ghosh et al., 2020). Although for long-term sustainable rice yield, INM with chemical fertiliser + FYM + crop residues improved the biological properties of the land and increased land productivity (Borase et al., 2021). Poor nutrient management results in a gradual yet significant change in soil properties, making it crucial to assess soil quality (Bhattacharyya et al., 2015; Zahedifar, 2023).
Assessing the soil quality and land productivity through individual soil properties is very complex due to the varied effects of diverse land management practices. (Griffiths et al., 2010; Raiesi & Kabiri, 2016). SQI integrates the physical, chemical, and biological properties of soil into a single quantitative measure (Doran et al., 1996; Karlen et al., 1997), providing a more accurate representation of soil quality than relying solely on individual or paired soil parameters (Masto et al., 2007; Armenise et al., 2013). These soil parameters have been selected based on management goals, soil function and site-specific factors (Andrews et al., 2004). SQI reliably links soil health with management practices (Temjen et al., 2022). Spatial variability of soil quality provides crucial information about fertiliser management practices (Delsouz Khaki et al., 2017; Alabouvette & Cordier, 2018; Shukla et al., 2020; Bel-Lahbib et al., 2023). There are a few approaches; the Integrated Quality Index (IQI) and the Nemoro Quality Index (NQI) are reliable and suitable for evaluating soil quality and linking it with associated land management practices. Qi et al., (2009) addressed the three advantages of these methods: (1) due to their intuitive nature, both indices are very easily understandable (Sun et al., 2003; Wang & Gong, 1998). (2) Both the indices use mathematical equations, which makes them more reliable, practical, and efficient, and (3) the indices create a platform for agricultural planners and soil researchers to evaluate soil quality and other associated characteristics. Rahmanipour et al., (2014) attempted to compare the reliability of these two indices and obtained that compared to NQI, IQI identified the correct estimation of soil quality. Zhou et al., (2022) supported the previous study and showed that IQI and NQI are the most suitable for estimating soil quality up to 0–20 cm and 20–30 cm, respectively. Now, it has become popular in the scientific world, and many studies have applied IQI in their work (Bahena-Osorio et al., 2023; Kafei et al., 2023; Oliveira et al., 2024; Samaei et al., 2022). IQI is further subdivided into simple additive and weighted additive indexes. After analysing the Total Data Set (TDS) using Principal Component Analysis (PCA), the Minimum Data Set (MDS) is selected (Andrews et al., 2002). MDS is an appropriate approach for reducing data redundancy, clarifying data variability, and accelerating the identification of key properties for further analysis (Vasu et al., 2016).
In the recent era, there are numerous researchers of subtropical rice-dominated countries like China (Chen et al., 2021; Ji et al., 2020; Yu et al., 2018; Zhao et al., 2021), Iran (Bakhshandeh et al., 2019; Nehrani et al., 2020; Rezaee et al., 2020; Rezapour et al., 2021; Samaei et al., 2022), Pakistan (Abid et al., 2020; Hussain et al., 2021 Sheikh, 2022; Nasir et al., 2024), Turkey (Doğan and Gülser, 2019; Alaboz et al., 2021), Bangladesh (Chowdhury et al., 2011; Hussain et al., 2013) and Philipines (Posadas et al., 2022) have focused on measurement of agricultural soil quality and nutrient management practices. The precision and reliability of predicting soil quality using geospatial techniques and machine learning approaches surpasses that of conventional soil mapping (Fathizad et al., 2020). The increasing popularity of spatial prediction of soil quality using these techniques is evident in numerous research studies (De et al., 2022; Li et al., 2023; Vaezi et al., 2020; Yadav et al., 2022). Kumar and Jhariya, (2015) utilised organic matter, soil pH, soil P, soil K, slope, and land use-land cover to identify agriculturally suitable areas using multi-criteria decision analysis (MCDA), obtaining a high agricultural suitability of 39.09%. The farmers, while contributing to the degradation of agricultural soil quality through their land management decisions primarily based on experience, also demonstrate the changes in soil quality due to different management systems and crop selection (Jian et al., 2020; Ruf et al., 2018; Yadav et al., 2022).
In India, various studies have been conducted on soil quality assessment, including evaluating soil quality near municipal waste dumping sites (Uma et al., 2016; Deshmukh & Aher, 2017; Choudhury et al., 2022), examining the relationship between soil quality and crop productivity (Ghosh et al., 2014; Singh et al., 2020; Vasu et al., 2016), analysing the effects of land-use changes on soil quality (Abdoalmohamdi et al., 2021; De et al., 2022; Masto et al., 2008), investigating the impact of agroforestry on soil quality (Fahad et al., 2022; Meetei et al., 2020; Prasad et al., 2017) and exploring the connection between cropping systems and soil quality (Sinha et al., 2014; Dhanda et al., 2022) etc. Most of the studies focus on computing the SQI and interpreting it. In contrast, minimal studies, particularly in India, attempt to assess the spatial variability of SQI and identify site-specific fertiliser management systems with crop combination and sustainable yield in a holistic way in a particular area.
The primary objectives of this study are (1) to determine and evaluate 13 soil parameters, including five physical parameters (bulk density, soil porosity, soil aggregate stability, water holding capacity, and infiltration rate), six chemical parameters (available nitrogen, available phosphorous and available potassium, soil pH, soil organic carbon, and electrical conductivity), and two biological parameters (soil respiration and microbial biomass carbon), (2) to compare spatial variability of the agricultural soil quality in two distinct, in terms of soil inherent and dynamics characteristics, C.D. Blocks, Ausgram-II and Memari-II using Inverse Distance Weighting (IDW) technique. (3) to validate the SQI using appropriate methods such as Sensitivity Index (SI), Efficiency Ratio (ER), and Sustainable Yield Index (SYI), and (4) to understand the underlying reason for the spatial variation of soil quality through a view of fertiliser management practices. It is hypothesised that there will be a significant difference in the spatial variation of SQI between the two Blocks. Eventually, crop selection leads to fertiliser management practices effectively impacting spatial variation of soil quality in this study area. The novelty of the work lies in identifying spatial variation of SQIs and understanding the site-specific fertiliser management system with distinct crop combinations. It also focuses on the sustainable fertiliser management practices in these two intensive agro-based C.D. Blocks.
Description of the study area
The eastern part of the erstwhile undivided Bardhaman District was known as the ‘rice bowl of Bengal’ due to the high production of rice. In 2017, the District was divided into Purba Bardhaman (East Bardhaman) and Paschim Bardhaman (West Bardhaman). However, currently, the rice bowl region falls under Purba Bardhaman District. Among the 23 C.D. blocks of Purba Bardhaman, two C.D. Blocks, Memari-II and Ausgram-II, were chosen for the study. Memari-II is located between 23°11′59"N to 23°22′25"N and 88°1′24"E to 88°14′05"E, covering an area of 186 km2. Ausgram-II, on the other hand, is situated between 23°23′55"N to 23°37′38"N and 87°27′44"E to 87°43′51"E, with a total area of 360 km2. The geological formations of Memari-II consist of diara, paskura, and sijua, while Ausgram-II comprises paskura, sijua, and laterite. Ausgram II is higher in elevation (65 m) than Memari-II (25 m). The mean monthly high and low temperatures of the study area are 32 °C (April) and 18 °C (January), respectively, with an annual rainfall of 1350 mm. Most of the soil in Memari-II is entisols, while Ausgram-II primarily comprises alfisols and ultisols. The parent material in Ausgram-II is alluvium-granite gneiss and gneiss-alluvium, while Memari-II has alluvium as its parent material. Based on five-year NPK data (2016–17 to 2020–21) of the 23 Blocks of the District, the authors calculated the imbalance index (FLibI) of the N-P-K level using the following formula (Eq. 1). Previously, numerous researchers have applied the imbalance index (FLibI) in their studies (Rajiv Mehta, 2007; Chand & Pandey, 2009; Bora, 2022). The value of FLibI away from zero measures the magnitude of the imbalance of fertiliser usage. If FLibI equals zero, it indicates a balanced fertiliser use, while a value of one indicates a situation of maximal imbalance. The data revealed that Memari-II is the highest in the imbalance index (FLibI = 0.75). At the same time, Ausgram-II is identified as the lowest (FLibI = 0.003) (Fig. 1). Also, the reason behind the selection of these Blocks is due to the difference in inherent and dynamic characteristics of agricultural soils.
where ‘FLibI’ denotes the imbalance index, ‘a’ refers to the actual dose of NPK and ‘n’ means the norm ratio of NPK (4:2:2). It is generally accepted that an NPK ratio of 4:2:2 (N: P2O5: K2O) is suitable for macro-level monitoring of plant nutrient intake for West Bengal (NAAS, 2009).
Materials and methods
Collection and analysis of the soil samples
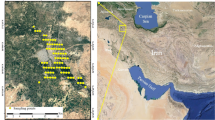
The stratified unaligned grid sampling method was used to select the soil sampling points (Wollenhaupt et al., 1994). Initially, the total area was divided into 1 × 1 km. Then a total of 104 soil samples were randomly collected during the dry season (November–December) from the agricultural lands of each C.D. Block with a confidence level of 95% (Fig. 2). The soil collection was carried out during the dry season due to less compaction of soil, reduction of risk of contamination (Leul et al., 2023) and primarily fields were crop free. Five soil samples (four corners and one centre point) were collected from each grid using an auger at the depth of 0–20 cm as this layer is the most nutrient-enriched, susceptible to land use changes and biologically well-active (Abegaz et al., 2016; Leul et al., 2023). Then, samples were blended, and a considerable amount of soil was sealed in self-locking polythene bags marked with numbers and latitudinal and longitudinal extensions. In the laboratory, the samples were air-dried, ground and passed through a 2 mm size sieve for further analysis. Each sample was tested for thirteen soil properties, viz. five physical (bulk density, soil porosity, soil aggregate stability, water holding capacity, and infiltration rate), six chemical (available nitrogen, available phosphorous and available potassium, soil pH, soil organic carbon and electrical conductivity) and two biological (soil respiration and microbial biomass carbon).
Physical properties
A core cutter technique was applied to assess the dry bulk density (BD) (Blake & Hartge, 1986). A cylinder-shaped iron ring with a radius (r) of 2.25 cm and a height of 7.8 cm was used to measure the volume of the soil core. To determine BD, the weight of the dry soil core was divided by the volume of the soil core. Soil porosity (SP) was derived from the BD value using the formula SP = 1-(BD ÷ 2.65). The default value of 2.65 is used as a rule of thumb based on the average bulk density of rock (USDA-NRCS, 2019). Soil Aggregate Stability (SAS) was determined by the wet sieving or wet aggregates method (Kemper & Rosenau, 1986). Following Kemper et al., (1985) to determine the wetting rates and stability of the samples to standardise the method. Water Holding Capacity (WHC) and Infiltration Rate (IR) were measured by the keen box method (Piper, 1966) and single ring infiltrometer (Tricker, 1978), respectively. Tricker, (1978) determined that a 15 cm diameter was the optimal measurement for infiltration, and a 50 cm ring height was utilised. Plastic was used to cover the open end of the ring and fill it with water to a depth of 5 cm. The ring was then driven 5 cm into the soil, and the plastic was removed from the mouth to measure the infiltration rate at 5-min intervals for 2 h (Tricker, 1978; Johnson, 1963).
Chemical properties
Available nitrogen (available N) was analysed using the Kjeldahl method (Kjeldahl, 1883). Available phosphorus (available P) and available potassium (available K) were determined by Olsen’s method (Olsen et al., 1954) and Flame photometer (Jackson, 1973), respectively. The wet digestion method (Walkley and Black, 1934) and the 1:2 soil/water method (Thomas, 1996) were used to measure Soil Organic Carbon (SOC) and soil pH, respectively. Electrical Conductivity (EC) was measured by EC-meter (Fritz et al., 1999).
Biological properties
Microbial Biomass Carbon (MBC) was measured by chloroform fumigation-extraction (Vance et al., 1987) and Soil Respiration (SR) was analysed by the alkali trap method (Page, 1982).
Modelling of soil quality index
The Soil Quality Index (SQI) is determined through three steps: (1) selecting MDS from TDS by PCA, (2) scoring the selected MDS, for this purpose, linear and non-linear scoring techniques are applied, and (3) integrating the score and selected MDS in a quantitative value (Andrews et al., 2002; Bel-Lahbib et al., 2023; Masto et al., 2008).
Selection of MDS
PCA was used for data redundancy or retention of MDS from TDS (Doran & Parkin, 1994; Qi et al., 2009). PCs with eigenvalue ≥ 1 and cumulatively explained ≥ 70% of the total variance were the criteria for selecting high-loading indicators (Kaiser, 1960; Andrews et al., 2002; Chandel et al., 2018). In each PC, soil parameters are represented with some loading value to show their contribution in a respective PC. The absolute values that fall within 10% of the highest loading factor of the same PC were considered for MDS (Wander and Bollero, 1999; Rahmanipour et al., 2014). Pearson's correlation coefficient was used to eliminate the redundancy of multiple highly weighted indicators. Regarding the correlated values between the two indicators, the soil property with a highly weighted value was considered MDS. However, if the highly loaded indicators were not correlated, both values were retained as MDS. The loading value of MDS is also known as the commonality of the factors, which represents their contribution to explained variance (Johnson & Wichern, 2002). To obtain the weighted value of PCs, the explained variance has been divided by the total cumulative variance (Andrews et al., 2002). From the MDS, the indicators were assigned by non-linear scoring according to their functions (Dadhwal et al., 2011).
Scoring function
The non-linear scoring method was employed to score the MDS. The scoring curve has a scale of 0 to 1 (Karlen & Stott, 1994; Qi et al., 2009). There are three types of curves; firstly, a sigmoid curve with an upper asymptote or ‘more is better’, where the indicators are scored highest (1) in its higher threshold value and other values are determined according to their obtained level. Secondly, the sigmoid curve with lower asymptote or ‘less is better’, where the indicators are scored high in its lowest threshold value, and thirdly, the bell-shaped curve or optimum value is better (Liebig et al., 2001; Andrews & Carroll, 2001 and Karlen et al., 1994). For example, available N is assigned as upper asymptote or ‘more is better’, BD as lower asymptote or ‘less is better’, and soil pH as optimum is better. In this study, IQI has been employed using a weighted additive index. In an unequal number of parameters, the weighted additive index is more suitable (Cherubin et al., 2016). Finally, the SQIs are computed using the following formula (Eq. 2). The entire methodological framework is shown in Fig. 3.
where, \(^{\prime}Wi^{\prime}\) is assigned as the weight of the indicators extracted from PCA, \(^{\prime}Si^{\prime}\) refers to the indicator score and ‘n’ represents the number of indicators.
Methods of evaluating indices for SQI
Sensitivity index (SI)
After calculating the SQI, it is crucial to assess its efficacy (Zhou et al., 2022). The Sensitivity Index (SI) determines the existing quality of the soil. A higher value of SI indicates that the soil is more prone to soil management practices or external disturbances (Masto et al., 2008). It also shows the potential variability of the SQIs in the data set (Sheidai Karkaj et al., 2019). The following formula (Eq. 3) was applied to calculate the sensitivity of the soil.
SI is a ratio of the maximum (SQI(max)) and minimum (SQI(min)) SQIs observed under each indexing procedure.
Efficiency ratio (ER)
ER indicates the efficiency level of SQI, or it determines how reliable the SQI value is (Sheidai Karkaj et al., 2019). ER is obtained by simply dividing the number of highly correlated values by the number of feasible indicators (Eq. 4). Higher ER explains the reliable value of SQI.
‘K’ is the number of correlated indicators in a specific SQI, and ‘N’ is the number of feasible parameters or MDS.
Sustainable yield index (SYI)
A sustainable agricultural system aims to preserve soil quality and land productivity over time (Kanwar, 1994). SYI is an essential quantitative measurement that includes long-term cultivable fields with similar land management practices (Singh et al., 1990). The following formula (Eq. 5) has been used to calculate the SYI.
where ‘\(\overline{{\text{Y}} }\)’ indicates the mean yield of a treatment of a particular crop, ‘\(\sigma\)’ means the standard deviation of that treatment, and ‘Ymax’ is the maximum yield of a particular crop within the treatment tenure.
Statistical technique and software
Maximum, minimum, mean, coefficient of variation (CV), kurtosis, and skewness of the soil parameters were included in the descriptive data set. Principal Component Analysis (PCA) was applied to select the MDS from the TDS. Pearson's correlation coefficient was used to find the correlated indicators for dimension reduction. MS Excel 2016 and IBM SPSS Statistics V25 were used to compute the descriptive statistics.
Spatial variability technique
Inverse Distance Weighting (IDW) assumes the value of unvisited points through the assigned known points (Eq. 6). IDW, a suitable interpolation method, was applied to present the spatial variation of the soil parameters, including SQI. IDW is used to estimate the value of the attribute at unvisited locations based on the data points within a neighbourhood or window surrounding the unvisited point (Bhunia et al., 2018). To run the IDW, ArcMap 10.6 has been used.
\(z\) is the interpolated value at the location \({x}_{0}\); \(n\) indicates the total number of sample points; \(\beta\) denotes the weighting power (β = 2); \({x}_{i}\) is the measured value, and hij is the separation distance between the interpolated value and the measured value.
Result
Statistical description of the soil parameters
The essential descriptive statistics such as minimum, maximum, mean, standard deviation (SD), skewness, kurtosis and coefficient of variance (CV) of the selected 13 soil parameters are properly presented (Table 1). Skewness, kurtosis, and CV are crucial statistical techniques for evaluating data normality. The threshold values for skewness and kurtosis are -2.0 to + 2.0 and -7.0 to + 7.0, respectively (Hair et al., 2010; Byrne, 2010). The analysed data fall within the specified thresholds for skewness and kurtosis. In terms of CV, some researchers classified the ranges into three categories: low (< 10%), moderate (10–100%), and high (> 100%) (Nielsen and Bouma, 1985; Behera and Shukla, 2015). It is widely accepted that CV < 10% indicates very good, 10–20% denotes good, 20–30% is acceptable and CV > 30% means not acceptable (Sedivy et al., 2021; Doan & Hassell, 2023). In Ausgram-II C.D Block, soil respiration (29.03%) and soil pH (5.44%) are the highest and lowest CV, respectively. In the Memari-II Block, soil respiration is the lowest CV (16.64%), and available N is the highest (2.94%) (Table 1). The BD ranges from 1.06 to 1.62 g/cm3 (mean 1.30 g/cm3) and 1.14 to 1.65 g/cm3 (mean 1.41 g/cm3) in Ausgram-II and Memari-II Block, respectively. While SP also varies from 38 at 57% (average 46.92%) in Memari-II compared to ranges of 39 to 60% (average 50.66%) in Ausgram-II Block. BD and SP are recognised as negatively correlated to each other. The infiltration rate of Ausgram-II ranges from 10 to 29 mm/hr (Mean 22.19 mm/hr), whereas Memari-II ranges from 2 to 7 mm/hr (Mean 4.05 mm/hr). The mean SAS in Memari-II Block is 45.14% (34–55%), higher than Ausgram-II with a mean of 40.02% (32–51%). WHC varies from 25 at 49% (Mean 30.51%) in Ausgram-II and ranges from 26 to 52% (mean 36.71%) in Memari-II Block, which is slightly higher than Ausgram-II. The Average value of EC has been observed at 0.43 dS/m (ranges 0.2–0.7 dS/m) in Ausgram-II Block compared to 0.5 dS/m (range 0.1–1.4 dS/m) in Memari-II. The soil pH is neutral for Ausgram-II, having a mean of 6.57 (range of 6.12–7.34), and Memari-II, having a mean of 6.00 (range of 5.12–7.65). Regarding SOC, it is found to be slightly better for Ausgram-II with a mean of 0.72% (range of 52–88%) compared to Memari-II, 0.50% (range of 28–69%). Available N, P and K are relatively high in Memari-II with the mean of 451.19 kg/ha (range of 415–481 kg/ha), 93.04 kg/ha (range of 64–112 kg/ha) and 281.76 kg/ha (range of 234–296 kg/ha), respectively. In contrast, in Ausgram-II, the mean of available N, P and K are found to be 353.87 kg/ha (range of 225–454 kg/ha), 66.58 kg/ha (range of 41–95 kg/ha) and 215.83 kg/ha (range of 148–285 kg/ha), respectively. In Memari-II, SP varies from 14 to 37 CO2-C lbs/acre-3"/day (Mean 22.13 CO2-C lbs/acre-3"/day) compared to a range of 21 to 61 CO2-C lbs/acre-3"/day (mean 38.87 CO2-C lbs/acre-3"/day) for Ausgram-II. Lastly, the MBC shows also better in Ausgram-II with an average of 258.75 µg C/g (range of 284–289 µg C/g) in contrast, Memari-II with 228.15 µg C/g−1 (ranges 214–262 µg C/g). The differences between Ausgram-II and Memari-II Block are evident in various soil properties.
Spatial distribution of the soil parameters
Spatial distribution of the soil data was used to understand the space-specific variability of soil health and estimate associated soil management practices. IDW, a reliable and suitable method, was used to produce the distribution of soil properties by estimating non-sampled points from the known points. The Blocks are distinct textural classes due to their unique geological formation, climate and other physical factors. The soil of Ausgram-II is predominantly sandy loamy and sandy clay loamy (Fig. 4a), whereas Memari-II has a clay loam and silty clay-type texture (Fig. 4b). For Ausgram-II, the Ideal BD (< 1.40 g/cm3) for sandy loamy and sandy clay loamy soil are covered almost 94% of area (Fig. 5a), while predominate BD (< 1.49 g/cm3) shows that 88% of area for Memari-II Block (Fig. 6a), though the ideal BD found < 1.10 for clay loam and silty clay soil. In Ausgram-II, 67.40% (< 50%), 16.68% (50%) 15.91% (> 50%) of the area are covered as low, medium or ideal and high SP (Fig. 5b). Whereas, for Memari-II, it shows 83% of the area is dominated by low porosity, 5% ideal, and 12% high porosity (Fig. 6b). In case of IR, the ideal IR (20–30 mm/hr) is observed 80.56% of northern part and southern part of Ausgram-II Block (Fig. 5c) compared to almost entire of area (96%) is covered with low IR and remaining only 4% of area shows ideal IR for Memari-II Block (Fig. 6c). WHC is observed 62.28% (< 30%), 36.53% (30–40%) and 0.81% (> 40%) for Ausgram-II (Fig. 5d) and 3% (< 30%), 74% (30–40%) and 23% (> 40%) for Memari-II (Fig. 6d). For SAS, low to medium (99.93%) is predominated for Ausgram-II (Fig. 5e) while 95% of the lands of Memari-II is covered by low to medium and 5% shows good (Fig. 6e). In terms of EC, Fig. 5f shows that 99% of the lands of Ausgram-II is covered with non-saline soil (< 0.6 dS/m). For Meamri-II, Fig. 6f shows 87% of the area is dominated by non-saline (< 0.6 dS/m), and the remaining lands, mostly the north-eastern corner part, is covered by slightly saline (0.6–1 dS/m) and moderately saline soil (1–2 dS/m). The spatial variation map (Fig. 5g) of available N shows that 6.90% are low (< 270 kg/ha), observed in the north-eastern portion, 93.03% are medium (270–450 kg/ha), and 0.07% (> 450 kg/ha) are high in Ausgram-II. In comparison, 36.38% are medium (270–450 kg/ha), observed mostly south-eastern part, and 63.62% are high (> 450 kg/ha), the remaining entire area of Memari-II (Fig. 6g). Figure 5h shows 1.46% of low (< 45 kg/ha), mostly observed in the north-eastern part, 97.59% are medium (45–90 kg/ha), and 0.95% are high (> 90 kg/ha), available P of Ausgram-II Block compare to Fig. 6h shows that 26% is identified as medium (45–90 kg/ha) and 74% are high (> 90 kg/ha) in Memari-II Block. In both Blocks, a medium amount (150–340 kg/ha) of available K dominates the entire area (Fig. 5 i and 6i). In terms of soil pH, some scatter portion (57.05%) of the north centre and southern part of Ausgram-II is covered by neutral (pH = 6.6–7.3), and the remaining 42.95% of the area is observed as slightly acidic soil (pH = 6.1–6.5) (Fig. 5j). In Memari-II, Fig. 6j shows 11% strongly acidic (pH = 5.1–5.5), 31% moderately acidic (pH = 5.6–6.0), 41% slightly acidic (pH = 6.1–6.5), 15% neutral and 0.56% (pH = 6.6–7.3), and slightly alkaline soil (pH = 7.4–7.8). For SOC, Fig. 5k shows 68.28% are medium (0.50–0.75%), and 31.72% are high (> 0.75%), while Fig. 6k shows 57% are low (< 0.50) and 43% are medium (0.50–0.75%) in Ausgram-II and Memari-II, respectively. SR is mostly dominated by ideal (32–64 CO2-C lbs/acre-3"/day) with 69% of the area of Ausgram-II and remaining 29% shows medium (16–32 CO2-C lbs/acre-3"/day) in the northern and southern part of the Block (Fig. 5l). In comparison, it is observed in the Fig. 6l that 2% of the area are low (9.5–16 CO2-C lbs/acre-3"/day), 96.04% are medium (16–32 CO2-C lbs/acre-3"/day) and 2% are ideal (32–64 CO2-C lbs/acre-3"/day) in Memari-II. For MBC, about 70% of the area of Ausgram-II is covered with medium (250–275 µg/g) (Fig. 5m) and about 82% of the area of Memari-II is dominated by medium class (220–240 µg/g) (Fig. 6m).
Soil quality index (SQI)
Selection of minimum data set (MDS)
In the Blocks Ausgarm-II and Memari-II, up to five PCs are extended to fulfil the above-mentioned principle. In the case of Ausgram-II, PC1 explains the highest variance (26.71%), followed by PC2 (19.77%), PC3 (10.28%), PC4 (9.39%) and PC5 (8.38%). PC1 is represented by SAS (0.806), SR (0.803) and MBC (0.782). In PC2, BD is the highest negatively correlated value (-0.836), followed by SP (0.835), and available K (0.762). PC3, PC4, and PC5 are represented by highly loaded values of soil pH (-0.754), available N (0.529), and available P (0.715), respectively (Table 2).
For Memari-II, cumulative variance accounts for 80.36% of the five PCs (Table 3). The highest value is explained by PC1 (29.145%), followed by PC2 (16.78%), PC3 (13.940%), PC4 (22.935%) and PC5 (8.565%). In PC1, WHC has the highest positive loading value (0.907), followed by BD (-0.837), SP (0.832), and IR (-0.825). PC2 is represented by SR (0.889) and MBC (0.843). Available N (-0.821) is the highest negative loading in PC3, followed by available P (-0.739). PC4 is represented by SOC (0.663) and Soil pH (0.632). While PC5 is represented only by SAS (0.511).
Data redundancy and retention of MDS
For Ausgram-II Block, from the PC1, only SAS is selected as representative as it is correlated with SR and MBC. In PC2, BD is correlated with SP. Thus, BD and available K are taken as representative as they are both highly loaded but not correlated to each other (Fig. 7). Soil pH, available N and available P are selected for PC3, PC4 and PC5, respectively, as they are only representative of respective PCs. In Memari-II, PC1 is represented by WHC, BD, SP and IR. WHC has been taken for further calculation as WHC is correlated to remaining soil parameters. While SR and MBC represent PC2, and as they are correlated, highly loading SR has been selected as MDS. Similarly, in PC3 and PC4, available N and pH have been taken in MDS, as both parameters correlated to the rest of the highly loading parameters (Fig. 8). SAS is the only representative for PC5, so it has been selected for further computation.
Scoring of MDS
For Ausgram-II Block, SAS, BD, available K, pH and available P have been retained as highly loading components. In contrast, WHC, SR, available N, pH, and SAS have been considered for calculating the SQI for Memari-II. Now, it is necessary to assign a score to the retained components (Tables 4 and 5). SAS, available K and available N have been assigned the score as ‘more is better’, whereas BD and pH have been scored as ‘less is better’ and ‘optimum is better’ (Table 4). In Memari-II, apart from pH, WHC, SR, available N, and SAS have been assigned as ‘more is better’ while pH has been assigned as ‘optimum is better’ (Table 5). To calculate the weight of each parameter, extracted individual variance has been divided by cumulative variance (Andrews et al., 2001; Bhardwaj et al., 2011). For Ausgram-II Block, the SAS (26.71%) shows the highest contribution to SQI followed by BD (19.77%), available K (19.77%), pH (10.28%) available N (9.39%) (Fig. 9a). Weight of the retained components have been given as 0.36 > 0.27 = 0.27 > 0.14 > 0.13 > 0.11.
Among the retained components, WHC (29.15%) contributes the highest contribution to SQI, followed by SR (16.78%), available N (13.94%), pH (11.94%), and SAS (8.57%) in Memari-II Block (Fig. 9b). The weight of the retained components has been given as 0.36 > 0.21 > 0.17 > 0.15 > 0.11.
Compare the SQI between two C.D. Blocks
The ideal indicator of soil quality would be an index that considers multiple highly loaded soil properties, as individual soil attributes do not accurately reflect the overall health of the soil. In this study, the SQI represents the soil health of each Block, and the results show that the SQIs of the Blocks vary significantly. The mean SQI value of Memari-II is 0.69 (ranges 0.57–0.86) compared to the mean value of 0.97 (ranges 0.83–1.08) of Ausgram-II (Fig. 10). Although the SD for both the Blocks is found to same (SD = 0.06) and CV is also very minimal differences (1.37%) (Table 6).
SQI ranges could be divided into five grades (Table 7). The spatial distribution shows that the SQI of Memari-II is observed mostly moderate to high (Grades III and IV), while in Ausgram-II Block, it is very high (Grade I). Understanding the spatial variability of SQI is important because it indicates the extent of land affected by poor soil management systems. In Ausgram-II, 99.72% of the area is classified as very high SQI with grade I, while the remaining 0.28% is classified as high SQI with grade II (Fig. 11a). On the other hand, moderate (Grade III), high (Grade II), and very high (Grade I) SQI values cover the land of 49.95%, 49.90%, and 0.16% of Memari-II, respectively (Fig. 11b). Moderate SQI is dominated on the eastern side and some patches in the western side. In contrast, high grade is dominated mostly on the western side of the Block.
Evaluation of SQI
Sensitivity index (SI) and efficiency ratio (ER)
For SI, the soil of the Memari-II Block (SI = 1.53) is more sensitive than Ausgram-II (SI = 1.30), which indicates higher external disturbances or human interventions to the soil. In the case of ER, out of 12 highly loaded parameters, Ausgram-II has 6, and Memari-II has 5 parameters selected for MDS. Therefore, Ausgram-II (ER = 50%) has a slightly higher ER value than Memari-II Block (ER = 42%).
Validation of SQI by sustainable yield index (SYI)
The study also attempts to understand the relationship between SQI and SYI. Singh et al., 1990 introduced a quantitative formula for SYI, which requires mean, SD, and maximum crop production. SYI ranges from 0 to 1, with high yielding and low SD resulting in higher SYI. A stable management with low SD and high mean provides the best feasible SYI (Wanjari et al., 2004).
The selected Blocks are identified as intensive agro-based. Seven years of annual data (District Statistical Handbook, 2010–11 to 2017–18) have been considered for assessing crop productivity. In Ausgram-II, the highest average yield is found in potatoes, followed by paddy, wheat, mustard, sesame and lentils. A similar sequence of productivity has been seen in Memari-II. However, the cultivated lands are seen differently. In Augram-II, paddy is cultivated in the maximum lands (96%) of total cultivated lands in the respective growing season, followed by potato (65%), mustard (45%), wheat (42%), sesame (32%), and lentils (24%). Whereas, in Meamri-II, paddy (94%) and potato (91%) dominate most of the area in their respective growing season, followed by mustard (20%), wheat (15%), sesame (8%), lentils (6%).
The mean of SYI for Ausgram-II is higher (0.51) than Memari-II (0.43) for the overall period, but Memari-II has a better SYI value for only potato yield (0.65). Ausgram-II has better SYI values for the remaining five crops: paddy (0.88), wheat (0.60), mustard (0.66), lentils (0.22), and sesame (0.43) (Fig. 12). The SQI and SYI have found lower value in Memari-II than Ausgram-II (Table 8). All the crop yields are positively correlated with SQIs in both Blocks. In Ausgram-II, paddy (R2 = 0.82) and Potato (R2 = 0.71) are found to be highly positively correlated, while mustard (R2 = 0.68), wheat (R2 = 0.65), sesame (R2 = 0.65), and lentils (R2 = 0.55) are moderately correlated. In Memai-II, Potato (R2 = 0.78) and paddy (R2 = 0.72) are highly positively correlated, followed by mustard (R2 = 0.67), wheat (R2 = 0.61), sesame (R2 = 0.55), and lentils (R2 = 0.51), respectively (Table 8).
Discussion
Explanation of the classes of soil indicators
In Ausgram-II, BD is lower than the threshold value for sandy loamy and sandy clay loamy soils (1.40 g/cm3) compared to Memari-II; it is slightly above the recommended range (< 1.10 g/cm3) for silty clay and clay loamy soil as per the USDA-NRCS (2019). Numerous studies consider that a porosity of about 50% is appropriate for soil productivity (Klopp & Bly, 2023). So, a soil porosity of 50% is considered as ideal. The infiltration rate is optimum for soil texture for both Blocks, as per the Food and Agricultural Organization (FAO, 1990). For WHC, both the Blocks fall above the threshold value (> 18–20%) (NRCCA, 2010). Soil with less than 2 dS/m is classified as non-saline (USDA-NRCS, 2011). It is also stated that when the EC level exceeds 2.1 dS/m, 12% of the paddy production would decline (USDA-NRCS, 2011). Durand (1983) classified ≤ 0.6 dS/m as non-saline, > 0.6EC ≤ 1 as slightly saline, and > 1EC ≤ 2 as moderately saline soil. More than 50–75% of SAS have been considered as medium to good physical quality (Shadekar, 2018) and > 75% as structural integrity (USDA-NRCS, 2001). As per the standard classification of soil pH (USDA-NRCS, 1998), mostly neutral and slightly acidic soil is found in Ausgram-II and Memari-II C.D.Block, respectively. The low, medium and high SOC are < 0.50%, 0.5–0.75% and > 0.75%, respectively. Optimum ranges of available N, P and K are < 270–450 > kg/ha, < 45–90 > kg/ha and < 150–340 > kg/ha, respectively, in agricultural land (CDAP, Purba Bardhaman 2017–20). Therefore, in Memari-II, available N and available P are slightly excessive than the optimum level. In contrast, the analysed data of N, P and K in Ausgram-II fall below the threshold ranges. USDA-NRCS (2014) defines high SR as 32–64 CO2-C lbs/acre-3"/day and moderate SR as 16–32 CO2-C lbs/acre-3"/day.
Evaluation of retained MDS
Among the total 13 parameters, 5 parameters were retained as MDS using PCA for both the Blocks. MDS is one of the most efficient methods for determining SQI as it reduces the variables, avoids data redundancy and retains the specific and effective variables (Huang et al., 2021; Nabiollahi et al., 2017). This study identified the highest loading factor for SQI in Ausgram-II and Memari-II Block as SAS, contributing 26.71%, and WHC, contributing 29.15 to SQI, respectively. A positive correlation (r = 0.58) between SAS and SOC may be attributed to the high use of organic matter. It was the prime observation of some previous works (Mustafa et al., 2020; Wolschick et al., 2018). Wolschick et al., (2018) showed that the tillage system also influences the SAS. On the other hand. WHC is the highest loading factor and serves as the primary physical parameter. WHC has a positive correlation with SAS (r = 0.75) and a negative correlation with BD (r = -0.62) and IR (r = -0.86). According to Nath, (2014), soil texture significantly impacts WHC in an experimental investigation conducted in Assam, India. The study finds that the WHC positively correlates with clay content (r = 0.80) as opposed to sandy content (r = -0.78), indicating that clay texture enhances WHC. The textural class of Memari-II Block is predominantly clay loam and silty clay-type, which may explain why WHC is higher in this area. However, WHC could be improved using manure, inorganic, and bio-fertiliser (Datt et al., 2013). Shah et al., (2022) showed that both the plots practised with inorganic and organic fertiliser received higher WHC. Available N and soil pH have been retained as MDS for both Blocks, indicating that both indicators influence soil quality. The amount of N-content appears to impact soil pH, with a negative correlation observed for both Blocks. This could be attributed to the acidifying effect of excessive N-fertilisation. In Memari-II, the soil pH is slightly below the neutral level (pH = 7) due to the high N-fertilisation, while the opposite finding is observed in Ausgram-II. Findings that align with the current results suggest that soil pH decreases linearly with increasing N-content in soils. Urea and NH4NO3 were found to be more responsible for acidification than NH4 fertilizer (Tian & Niu, 2015). In this study, urea was one of the primary fertilisers used as a nitrogen source in the soil. Available N contributed 13.94% and 9.31% to SQI of Memari-II and Ausgram-II, respectively. The reason may be the excessive usage of urea (46%N) and Di-ammonium Phosphate (18%N) in the Memari-II Block. Cattle manure contributes very slowly to the nitrogen in the soil. Sharma et al., (2008) found available N as a key indicator of SQI. They reported that nitrogen plays an important role in improving biomass and crop productivity above the soil and below the soil. Available K, the second most important indicator along with BD, contributes 19.77% to the SQI of Ausgram-II Block. Zero tillage sometimes lowers the available K as a minimum, or zero tillage increases bulk density (Sharma et al., 2005; Tisdale et al., 1985). However, BD was also found ideal, contributing 19.77% towards the SQI of Ausgram-II. This means tillage is regular and does not affect the available K. Available K controls the osmotic pull that draws water from the soil into plant roots (Sharma et al., 2005).
Effects of fertiliser management practices on soil properties and SQI
In Ausgram-II, 99.72% of the area is classified as very high SQI with grade I, while the remaining 0.28% is classified as high SQI with grade II. On the other hand, moderate (Grade III), high (Grade II), and very high (Grade I) SQI values cover 49.95%, 49.90%, and 0.16% of Memari-II, respectively. Nguemezi et al. (2020) attempted to investigate land management practices, showing that most lands are identified as having average fertility (46.49%) followed by good fertility (29.04%), poor fertility (21.46%) and very good fertility (3.79%) in sub-Saharan Africa. The major reason they noticed good to very good fertility was high organic matter, and poor fertility was related to severe limitation of physical parameters. A similar study was conducted in China's humid monsoon climatic sub-tropical region (Qi et al., 2009). They found that 90% of the area was covered by moderate soil quality (grade II and III), and the remaining 10% was shared by very high (grade I) and poor soil quality (grade II). Sandy-loamy soil is found to have low soil quality with low SOC and alkaline soil, while clay loamy is quite healthy with high SOC and is nutrient-rich. Although the findings contradict the present study, this may be the reason for differentiated land management systems that make a difference in soil quality. Similarly, Maleki et al., (2022) found that the most dominant soil quality is moderate (grade II and III) due to topographical attributes and irrigation from poor-quality groundwater. They addressed the sustainable and appropriate land management to mitigate soil degradation. A study demonstrates that the highest Soil Quality Index (SQI) is achieved at the optimal NPK fertiliser level (0.90), followed by the intermediate (0.86) and no fertilizer (0.78) levels in the Mediterranean environment (Armenise et al., 2013).
Farmers of Memari-II Block prefer farming with alone inorganic fertiliser, a certain amount of bio-fertiliser, and nearly no organic fertiliser. The common combinations are NPK and NPK + bio-fertiliser. In Ausgram-II Block, major combinations of fertiliser inputs NPK + cattle manure, NPK + cattle manure + bio-fertiliser and NPK + cattle manure + bio-fertiliser + rice husk ash. Studies have shown that high-quality cow dung contains important nutrients such as N (1.44–2.10%), P (48–80%), and K (1.74%) (Ewusi-Mensah et al., 2015). Additionally, cow dung with chicken manure can improve soil health by enhancing pH, SOC, exchangeable Ca, total N, and available P while reducing BD (Van Dang et al., 2021). Rice husk ash also benefits soil health and land productivity, as it contains biomaterials (Kordi et al., 2023). Another study found that combining rice husk biochar and fly ash with inorganic fertilisers can improve soil pH, SOC, CEC, available NPK, and EC, and even using 50% N content with rice ash biochar and fly ash can produce the same yield as 100% of the recommended dose (Munda et al., 2016). Matching with the findings of the present study, Sharma et al., (2010) proved that fertiliser practice with 100% organic (SQI = 2.62) improves soil quality more than 50% organic + 50% inorganic (SQI = 2.35) in rainfed alfisols. Ram et al., (2016) conducted a study on the effect of integrated nutrient management and revealed that continuous use of FYM improved the most to SQI (0.94). Applying the SQI method to 9 different fertiliser treatments, Li et al., (2020) highlighted that vermicompost and pig manure had significantly higher SQI, followed by straw, biochar and single chemical fertiliser. Eventually, to improve the soil health, they suggested applying a combination of chemicals with organic fertiliser. Rezaee et al., (2020) have studied paddy fields in Iran and identified poor soil quality (SQI < 0.58). The primary reason is that rice cultivation does not use optimum phosphorous and potassium-based fertiliser. Dengiz (2020) stated that SQI was dominated by moderate (36.8%) followed by high quality (36.1%), very low (22.5%) and low (4.5%) in Çarsamba Deltaic Plain in Turkey for rice cultivation. Excess slope, low clay content, salinity and water holding capacity have been found as reasons why 25.9% of the study area is unsuitable for rice cultivation.
In recent times, human activities have become more common than natural disturbances when it comes to changes in soil quality. SOC is a crucial indicator of soil quality because it can increase crop productivity and improve yield stability. The present study found that applying chemical fertilisers with manure can yield soil quality that matches previous findings (Han et al., 2016; Zhengchao et al., 2013). Han et al., (2016) found that unbalanced chemical fertilisers had the lowest SOC (0.10) and that chemical fertilisers combined with manure had the highest (0.31). In the present study, SOC was found to be low to moderate due to the excessive use of chemical fertilisers, while it was moderate to high when chemical fertilisers were combined with manure. Additionally, excessive usage of N-fertiliser has been found to reduce soil pH, as demonstrated by extensive research in China over the past 30 years (1980–2010) (Guo et al., 2018). The amount of organic matter has a significant impact on BD. Studies have shown that organic matter generally reduces BD and increases soil aggregate stability (SAS) (Biswas et al., 2023; Ekwue, 1990; Keller & Håkansson, 2010). In the present study, the use of organic fertilisers improved SAS in the study area. While WHC found the highest loading for Memari-II, this may be due to the higher percentages of clay contents in soils (Franzluebbers, 2020) Furthermore, there was a highly positive correlation between SR and MBC for both the Blocks. The presence of organic matter improves SR and, in turn, enhances microbial activities in the soil. MBC was higher in no-tillage systems due to the high percentage of organic matter, while pasture land with no organic matter showed a reduction in MBC (Valpassos et al., 2001).
Association of SQI with crop combination
Armenise et al., (2013) revealed that double cropping of wheat and dry beans has a higher SQI than other crop combinations. In North-West India, the rice–wheat-mungbean crop combination has the highest SQI (0.91) compared to the lowest SQI (0.65) of rice and wheat (Roy et al., 2022). The current study shows that the crop combination of paddy during the kharif season (rainy) and potato, wheat, and mustard during the rabi season (winter) is better than the paddy-potato cropping system. This finding is consistent with Saurabh et al., (2021), who reported that the paddy-wheat and greengram crop combination had the highest SQI and crop yield. In the present study, paddy with potato, wheat, mustard, sesame, and lentils improved SYI and SQI. The diverse crop rotation leads to improving soil health through balancing the nutrient cycle (Amorim et al., 2020; Lal, 2015; McDaniel et al., 2014). In the present study, dominant SQI grades are moderate to very high for Memari-II and high to very high for Ausgram-II Block. Previous studies found the dominant grades to be II and III, covering 58% of corn belt croplands in northern China (Li et al., 2018). In Qazvan Province, Iran, 54.44% of the area was found to have moderate (Grade-III) agricultural soil quality (Rahmanipour et al., 2014). Similar results were reported by Nabiollahi et al., 2018. In Memari-II, Potato is the primary crop, followed by paddy and insignificant cultivated lands of other crops (mustard, wheat, lentils, and sesame). In contrast, in Ausgram-II, farmers cultivated mustard, wheat, lentils, and sesame alongside paddy and potato as main crops. The diverse crop rotation leads to improving soil health through balancing the nutrient cycle (Amorim et al., 2020; Lal, 2015; McDaniel et al., 2014). While continuous paddy cultivation significantly decreased soil pH, SOC, available P and available K (Ren et al., 2020). Singh et al., (2020) identified that potato-green gram-rice would be the best crop combination in crop production. However, the rice-lentils combination mostly improves the soil quality in the Indo-Gangetic Plains of India. This finding matches the present study, which found that intensive paddy and potato combination increases crop yield but not soil quality, which is used to cultivate in Memari-II. Ghorai et al. (2023) proved that rice-mustard-sesame for 22 years fields responded positively towards soil quality with farm yard manure in lower Indo-Gangetic Plains. The present study shows that rice-mustard-sesame, a common crop combination, was used to cultivate in Ausgram-II C.D.Block. Xing et al., (2021) found that very low SQI (ranges from 0.06–0.46) in potato fields in the loess plateau region suggested using organic fertiliser and reasonable use of NPK. As a principal crop of Memari-II is potato, both suggestions are useful for this area. The nutrient (NPK) ratio of Memari-II (5:3:2.5) is found to be comparably higher than Ausgram-II (4:2.1:2). It is generally accepted that an NPK ratio of 4:2:1 (N: P2O5: K2O) is suitable for macro-level monitoring of plant nutrient intake for India as a whole and 4:2:2 for West Bengal in specific (NAAS, 2009). The study reveals a significant variation in the spatiality of SQI between two Blocks, and poor nutrient management is the principal cause of low soil quality.
Explanation of SQI validation
The SI value of the present finding (1.30 & 1.53) is lower than in the previous study (2.268) (Sheidai Karkaj et al., 2019). However, another study showed that the sensitivity of three different SQI approaches was 1.43,1.48 and 1.52 (Masto et al., 2007). Which is higher than the Ausgram-II Block (1.30) but lesser than the Memari-II Block (1.53). In the weighted additive method of MDS, the value of ER was observed to be low at 71.43% (Sheidai Karkaj et al., 2019), which is relatively higher than the present study. Not all TDS parameters are highly loaded in PCA during MDS selection. For SYI, Previous studies showed that the highest SYI was found in NPK + FYM (0.35–0.62), followed by N-fertilizer without manure (0–0.07). SYI can determine the recommended fertiliser dose. Liu et al., (2017) found that the recommended doses of N, P2O5 and K2O fertilisation rates for rice in southeast China were 186, 60, and 96 kg/ha, respectively. The study revealed a positive linear relationship between the recommended dose and SYI. A similar study showed that the highest SYI was achieved with 100% NPK + FYM at 15 t ha−1, which improved soil organic carbon rapidly. However, SYI values were more sustainable for wheat than rice yield (Ram et al., 2016). The present findings show higher SYI in various crop rotation (paddy, paddy, wheat, mustard, sesame and lentils) fields with a fertiliser combination of NPK + Organic manure + Bio-fertiliser + Rice husk ash. In comparison, it is found to be low when NPK is used solely in paddy-potato fields.
Conclusions
The study attempted to compare the two distinct soil inherent and dynamics characterised C.D. Blocks, Ausgram-II and Memari-II, using the SQI model to understand the spatial variation of soil quality and unveil the underlying reasons. Among the thirteen soil indicators, PCA showed that SAS, BD, available K, pH, available N, and available P were the most significant factors contributing to the SQI in Ausgram-II. Similarly, in Memari-II, WHC, SR, available N, pH, and SAS were the highest contributing parameters. The average SQI value is significantly higher in Ausgram-II (0.97), with 99.72% of the area having a very high SQI (Grade-I) compared to Memari-II (0.69) with 49.95% of the area bearing moderate SQI (Grade-II) and 49.90% shown a high SQI (Grade-III). SYI was relatively higher in Ausgram-II Block (0.51) than Memari-II (0.43). The results have shown that the Agricultural lands of Memari-II were identified as more sensitive (SI = 1.53) to external disturbances or land management systems than Asugram-II (SI = 1.30). Differentiated fertiliser management systems, nutrient ratio and crop selection mostly cause the disparity in SQIs. Most importantly, the nutrient ratio is much higher than the recommended ratio in Memari-II C.D.Block compared to Ausgram-II C.D.Block, which is a serious concern. The study found that the combination of fertiliser management practices in Memari-II is predominantly NPK and NPK + bio-fertiliser. In contrast, it is completely different in Ausgam-II, with NPK + organic fertiliser (cattle manure) + bio-fertiliser + rice husk ash. A strong positive correlation was observed between SQI and paddy ( R2 = 0.82 & 0.72) and potato yield (R2 = 0.71 & 0.78). The study focuses on space-specific variations in soil health and identifies faulty fertiliser management practices to enhance sustainable crop productivity. In the future, regions such as the agro-based Indian subcontinent should prioritise similar studies to improve crop yields with appropriate agricultural practices.
Data availability
All data generated during manuscript analysis are included in the article. Further datasets are available from the corresponding author upon request.
References
Abdoalmohamdi, S., Ildoromi, A., & Heshmati, M. (2021). The Effect of land use change on some physical and chemical properties of soil in the Halshi Watershed Kermanshah. Geography and Planning, 25(75), 171–180. https://doi.org/10.22034/gp.2021.10902
Abegaz, A., Winowiecki, L. A., Vågen, T. G., Langan, S., & Smith, J. U. (2016). Spatial and temporal dynamics of soil organic carbon in landscapes of the upper Blue Nile Basin of the Ethiopian Highlands. Agriculture, Ecosystems & Environment, 218, 190–208. https://doi.org/10.1016/j.agee.2015.11.019
Abid, M., Batool, T., Siddique, G., Ali, S., Binyamin, R., Shahid, M. J., Rizwan, M., Alsahli, A. A., & Alyemeni, M. N. (2020). Integrated nutrient management enhances soil quality and crop productivity in maize-based cropping system. Sustainability, 12(23), 10214. https://doi.org/10.3390/su122310214
Alabouvette, C., & Cordier, C. (2018). Fertilité biologique des sols: des microorganismes utiles à la croissance des plantes. Innovations Agronomiques, 69, np. Retrieved from https://hal.science/hal-02058232. Accessed on 08.12.23.
Alaboz, P., Dengiz, O., & Demir, S. (2021). Barley yield estimation performed by ANN integrated with the soil quality index modified by biogas waste application. Zemdirbyste-Agriculture, 108(3). https://doi.org/10.13080/z-a.2021.108.028
Allen, D. E., Singh, B. P., & Dalal, R. C. (2011). Soil health indicators under climate change: a review of current knowledge. Soil Health and Climate Change, 25–45. https://doi.org/10.1007/978-3-642-20256-8_2
Amorim, H. C., Ashworth, A. J., Wienhold, B. J., Savin, M. C., Allen, F. L., Saxton, A. M., Owens, P. R., & Curi, N. (2020). Soil quality indices based on long-term conservation cropping systems management. Agrosystems, Geosciences & Environment, 3(1), e20036. https://doi.org/10.1002/agg2.20036
Andrews, S. S., & Carroll, C. R. (2001). Designing a soil quality assessment tool for sustainable agroecosystem management. Ecological Applications, 11(6), 1573–1585.
Andrews, S. S., Karlen, D. L., & Mitchell, J. P. (2002). A comparison of soil quality indexing methods for vegetable production systems in Northern California. Agriculture, Ecosystems & Environment, 90(1), 25–45. https://doi.org/10.1016/S0167-8809(01)00174-8
Andrews, S. S., Karlen, D. L., & Cambardella, C. A. (2004). The soil management assessment framework: a quantitative soil quality evaluation method. Soil Science Society of America Journal, 68(6), 1945–1962.
Armenise, E., Redmile-Gordon, M. A., Stellacci, A. M., Ciccarese, A., & Rubino, P. (2013). Developing a soil quality index to compare soil fitness for agricultural use under different managements in the Mediterranean environment. Soil and Tillage Research, 130, 91–98. https://doi.org/10.1016/j.still.2013.02.013
Bahena-Osorio, Y., Franco-Hernández, M. O., Pueyo, J. J., & Vásquez-Murrieta, M. S. (2023). Development of a Quality Index to Evaluate the Impact of Abiotic Stress in Saline Soils in the Geothermal Zone of Los Negritos, Michoacán. Mexico. Agronomy, 13(6), 1650. https://doi.org/10.3390/agronomy13061650
Bakhshandeh, E., Hossieni, M., Zeraatpisheh, M., & Francaviglia, R. (2019). Land use change effects on soil quality and biological fertility: A case study in northern Iran. European Journal of Soil Biology, 95, 103119. https://doi.org/10.1016/j.ejsobi.2019.103119
Behera, S. K., & Shukla, A. K. (2015). Spatial distribution of surface soil acidity, electrical conductivity, soil organic carbon content and exchangeable potassium, calcium and magnesium in some cropped acid soils of India. Land Degradation & Development, 26(1), 71–79. https://doi.org/10.1002/ldr.2306
Bel-Lahbib, S., Ibno Namr, K., Rerhou, B., Mosseddaq, F., El Bourhrami, B., & Moughli, L. (2023). Assessment of soil quality by modeling soil quality index and mapping soil parameters using IDW interpolation in moroccan semi-arid. Modeling Earth Systems and Environment, 1–19. https://doi.org/10.1007/s40808-023-01718-1
Bhardwaj, A. K., Jasrotia, P., Hamilton, S. K., & Robertson, G. P. (2011). Ecological management of intensively cropped agro-ecosystems improves soil quality with sustained productivity. Agriculture, Ecosystems & Environment, 140(3–4), 419–429. https://doi.org/10.1016/j.agee.2011.01.005
Bhattacharyya, R., Ghosh, B. N., Mishra, P. K., Mandal, B., Rao, C. S., Sarkar, D., Das, K., Anil, K. S., Lalitha, M., Hati, K. M., & Franzluebbers, A. J. (2015). Soil degradation in India: Challenges and potential solutions. Sustainability, 7(4), 3528–3570. https://doi.org/10.3390/su7043528
Bhunia, G. S., Shit, P. K., & Maiti, R. (2018). Comparison of GIS-based interpolation methods for spatial distribution of soil organic carbon (SOC). Journal of the Saudi Society of Agricultural Sciences, 17(2), 114–126. https://doi.org/10.1016/j.jssas.2016.02.001
Bisht, N., & Chauhan, P. S. (2020). Excessive and disproportionate use of chemicals cause soil contamination and nutritional stress. Soil Contamination-Threats and Sustainable Solutions, 2020, 1–10.
Biswas, S., Singh, P., Rahaman, R., Patil, K. V., & De, N. (2023). Soil quality and crop productivity under 34 years old long-term rainfed rice based cropping system in an Inceptisol of sub-tropical India. Frontiers in Soil Science, 3, 1155712. https://doi.org/10.3389/fsoil.2023.1155712
Blake, G. R., & Hartge, K. H. (1986). Particle density. Methods of soil analysis Part 1 physical and mineralogical methods, 5, 377–382. https://doi.org/10.2136/sssabookser5.1.2ed.c14
Bora, K. (2022). Spatial patterns of fertilizer use and imbalances: Evidence from rice cultivation in India. Environmental Challenges, 7, 100452. https://doi.org/10.1016/j.envc.2022.100452
Borase, D. N., Murugeasn, S., Nath, C. P., Hazra, K. K., Singh, S. S., Kumar, N., Singh, U., & Praharaj, C. S. (2021). Long-term impact of grain legumes and nutrient management practices on soil microbial activity and biochemical properties. Archives of Agronomy and Soil Science, 67(14), 2015–2032.
Byrne, B.M. (2010). Basic Concepts, Applications, and Programming Second Edition Structural.
Byrne, B. M. (2013). Structural equation modeling with Mplus: Basic concepts, applications, and programming. routledge. https://doi.org/10.4324/9780203807644
Chand, R., & Pandey, L. M. (2009). Fertiliser use, nutrient imbalances and subsidies: Trends and implications. Margin: The Journal of Applied Economic Research, 3(4), 409–432. https://doi.org/10.1177/09738010090030040
Chandel, S., Hadda, M. S., & Mahal, A. K. (2018). Soil quality assessment through minimum data set under different land uses of submontane Punjab. Communications in Soil Science and Plant Analysis, 49(6), 658–674. https://doi.org/10.1080/00103624.2018.1425424
Chen, Q., Liu, Z., Zhou, J., Xu, X., & Zhu, Y. (2021). Long-term straw mulching with nitrogen fertilization increases nutrient and microbial determinants of soil quality in a maize–wheat rotation on China’s Loess Plateau. Science of the Total Environment, 775, 145930. https://doi.org/10.1016/j.scitotenv.2021.145930
Cherubin, M. R., Karlen, D. L., Cerri, C. E., Franco, A. L., Tormena, C. A., Davies, C. A., & Cerri, C. C. (2016). Soil quality indexing strategies for evaluating sugarcane expansion in Brazil. PLoS ONE, 11(3), e0150860. https://doi.org/10.1371/journal.pone.0150860
Choudhary, M., Panday, S. C., Meena, V. S., Singh, S., Yadav, R. P., Mahanta, D., Mondal, T., Mishra, P. K., Bisht, J. K., & Pattanayak, A. (2018). Long-term effects of organic manure and inorganic fertilization on sustainability and chemical soil quality indicators of soybean-wheat cropping system in the Indian mid-Himalayas. Agriculture, Ecosystems & Environment, 257, 38–46. https://doi.org/10.1016/j.agee.2018.01.029
Choudhury, B. U., & Mandal, S. (2021). Indexing soil properties through constructing minimum datasets for soil quality assessment of surface and profile soils of intermontane valley (Barak, North East India). Ecological Indicators, 123, 107369. https://doi.org/10.1016/j.ecolind.2021.107369
Choudhury, M., Jyethi, D. S., Dutta, J., Purkayastha, S. P., Deb, D., Das, R., Roy, G., Sen, T., & Bhattacharyya, K. G. (2022). Investigation of groundwater and soil quality near to a municipal waste disposal site in Silchar, Assam, India. International Journal of Energy and Water Resources, 6(1), 37–47. https://doi.org/10.1007/s42108-021-00117-5
Chowdhury, M. A., Khairun, Y., Salequzzaman, M., & Rahman, M. M. (2011). Effect of combined shrimp and rice farming on water and soil quality in Bangladesh. Aquaculture International, 19, 1193–1206. https://doi.org/10.1007/s10499-011-9433-0
Dadhwal, K. S., Mandal, D., & Shrimali, S. S. (2011). Soil quality index for different land use systems in northwestern hilly region of India. Journal of the Indian Society of Soil Science, 59(2), 169–176.
Datt, N., Dubey, Y. P., & Chaudhary, R. (2013). Studies on impact of organic, inorganic and integrated use of nutrients on symbiotic parameters, yield, quality of French-bean (Phaseolus vulgaris L.) vis-à-vis soil properties of an acid Alfisol. African Journal of Agricultural Research, 8(22), 2645–2654. https://doi.org/10.5897/AJAR12.942
De, P., Deb, S., Deb, D., Chakraborty, S., Santra, P., Dutta, P., Hoque, A., & Choudhury, A. (2022). Soil quality under different land uses in eastern India: Evaluation by using soil indicators and quality index. PLoS ONE, 17(9), e0275062. https://doi.org/10.1371/journal.pone.0275062
de Oliveira, E. M., Hermógenes, G. M., da Costa Brito, L., Silva, B. M., Avanzi, J. C., Beniaich, A., & Silva, M. L. N. (2024). Cover crop management systems improves soil quality and mitigate water erosion in tropical olive orchards. Scientia Horticulturae, 330, 113092. https://doi.org/10.1016/j.scienta.2024.113092
Delsouz Khaki, B., Honarjoo, N., Davatgar, N., Jalalian, A., & Torabi Golsefidi, H. (2017). Assessment of two soil fertility indexes to evaluate paddy fields for rice cultivation. Sustainability, 9(8), 1299. https://doi.org/10.3390/su9081299
Dengiz, O. (2020). Soil quality index for paddy fields based on standard scoring functions and weight allocation method. Archives of Agronomy and Soil Science, 66(3), 301–315. https://doi.org/10.1080/03650340.2019.1610880
Deshmukh, K. K., & Aher, S. P. (2017). Assessment of soil fertility around municipal solid waste disposal site near Sangamner City, Maharashtra. India. Current World Environment, 12(2), 401.
Dhanda, S., Yadav, A., Yadav, D. B., & Chauhan, B. S. (2022). Emerging issues and potential opportunities in the rice–wheat cropping system of North-Western India. Frontiers in Plant Science, 13, 832683. https://doi.org/10.3389/fpls.2022.832683
District Statistical Handbook. (2010). Bureau of Applied Economics & Statistics, Department of Statistics & Programme Implementation, Government of West Bengal. Retrieved from https://purbabardhaman.nic.in/district-statistical-handbook/. Accessed on 21.11.2023.
District Statistical Handbook. (2011). Bureau of Applied Economics & Statistics, Department of Statistics & Programme Implementation, Government of West Bengal. Retrieved from https://purbabardhaman.nic.in/district-statistical-handbook/. Accessed on 21.11.2023.
District Statistical Handbook. (2013). Bureau of Applied Economics & Statistics, Department of Statistics & Programme Implementation, Government of West Bengal. Retrieved from https://purbabardhaman.nic.in/district-statistical-handbook/. Accessed on 21.11.2023.
District Statistical Handbook. (2014). Bureau of Applied Economics & Statistics, Department of Statistics & Programme Implementation, Government of West Bengal. Retrieved from https://purbabardhaman.nic.in/district-statisticalhandbook/. Accessed on 21.11.2023.
District Statistical Handbook. (2017). Bureau of Applied Economics & Statistics, Department of Statistics & Programme Implementation, Government of West Bengal. Retrieved from https://purbabardhaman.nic.in/district-statisticalhandbook/. Accessed on 21.11.2023.
District Statistical Handbook. (2018). Bureau of Applied Economics & Statistics, Department of Statistics & Programme Implementation, Government of West Bengal. Retrieved from https://purbabardhaman.nic.in/district-statisticalhandbook/. Accessed on 21.11.2023.
District Statistical Handbook. (2019). Bureau of Applied Economics & Statistics, Department of Statistics & Programme Implementation, Government of West Bengal. Retrieved from https://purbabardhaman.nic.in/district-statisticalhandbook/. Accessed on 21.11.2023.
Doan, D. K., & Hassell, L. A., (2023) Validation of reference intervals and reportable range. Pathologyotlines.com. Retrieved from https://www.pathologyoutlines.com/topic/labadminreferenceintervals.html. Accessed on 16.12.2023.
Doğan, B., & Gülser, C. (2019). Assessment of soil quality for vineyard fields: A case study in Menderes District of Izmir. Turkey. Eurasian journal of soil science, 8(2), 176–183. https://doi.org/10.18393/ejss.551874
Doran, J. W., & Parkin, T. B. (1994). Defining and assessing soil quality. Defining Soil Quality for a Sustainable Environment, 35, 1–21.
Doran, J. W., Sarrantonio, M., & Liebig, M. A. (1996). Soil health and sustainability. Advances in Agronomy, 56, 1–54.
Durand, J. H. (1983). Irrigable soils (p. 338). Pedological study. Presses Universitaires of France.
Ekwue, E. I. (1990). Organic-matter effects on soil strength properties. Soil and Tillage Research, 16(3), 289–297. https://doi.org/10.1016/0167-1987(90)90102-J
Ewusi-Mensah, N., Logah, V., & Akrasi, E. J. (2015). Impact of different systems of manure management on the quality of cow dung. Communications in Soil Science and Plant Analysis, 46(2), 137–147. https://doi.org/10.1080/00103624.2014.967854
Fahad, S., Chavan, S. B., Chichaghare, A. R., Uthappa, A. R., Kumar, M., Kakade, V., ... & Poczai, P. (2022). Agroforestry systems for soil health improvement and maintenance. Sustainability, 14(22), 14877. https://doi.org/10.3390/su142214877
FAO. (1990). FAO Land and Water Development Division, Food and Agriculture Organisation of the United Nation. Retrieved from https://www.fao.org/home/en. Accessed on 05.06.2023.
Fathizad, H., Ardakani, M. A. H., Heung, B., Sodaiezadeh, H., Rahmani, A., Fathabadi, A., Scholten, T., & Taghizadeh-Mehrjardi, R. (2020). Spatio-temporal dynamic of soil quality in the central Iranian desert modeled with machine learning and digital soil assessment techniques. Ecological Indicators, 118, 106736. https://doi.org/10.1016/j.ecolind.2020.106736
Flinn, J. C., & De Datta, S. K. (1984). Trends in irrigated-rice yields under intensive cropping at Philippine research stations. Field Crops Research, 9, 1–15.
Franzluebbers, A. J. (2020). Holding water with capacity to target porosity. Agricultural & Environmental Letters, 5(1), e20029. https://doi.org/10.1002/ael2.20029
Fritz, R. M., Malo, D. D., Schumacher, T. E., Clay, D. E., Carlson, C. G., Ellsbury, M. M., & Dalsted, K. J. (1999). Field comparison of two soil electrical conductivity measurement systems. In Proceedings of the fourth international conference on precision agriculture (pp. 1211–1217). Madison, WI, USA: American Society of Agronomy, Crop Science Society of America, Soil Science Society of America. https://doi.org/10.2134/1999.precisionagproc4.c24b
Ghorai, D., Ziauddin, G., Sarkar, S., Singh, M.S., Chatterjee, J., Ghatak, S., Sinha, G., & Ghosh, P. (2018). Comprehensive district agriculture plan, Purba Bardhaman for 2017-18 to 2019-20. Retrieved from https://www.kvkcrijaf.org.in/Reports/CDAP%20Purba%20Bardhaman. Accessed on 17.06.2023.
Ghorai, P. S., Biswas, S., Purakayastha, T. J., Ahmed, N., Das, T. K., Prasanna, R., Gawade, B. H., Bhattacharyya, K., Sinha, K., Singh, P., & Das, S. (2023). Indicators of soil quality and crop productivity assessment at a long-term experiment site in the lower Indo-Gangetic plains. Soil Use and Management, 39(1), 503–520. https://doi.org/10.1111/sum.12847
Ghosh, B. N., Sharma, N. K., Alam, N. M., Singh, R. J., & Juyal, G. P. (2014). Elevation, slope aspect and integrated nutrient management effects on crop productivity and soil quality in North-west Himalayas, India. Journal of Mountain Science, 11, 1208–1217. https://doi.org/10.1007/s11629-013-2674-9
Ghosh, P. K., Hazra, K. K., Venkatesh, M. S., Praharaj, C. S., Kumar, N., Nath, C. P., Singh, U., & Singh, S. S. (2020). Grain legume inclusion in cereal–cereal rotation increased base crop productivity in the long run. Experimental Agriculture, 56(1), 142–158.
Griffiths, B. S., Ball, B. C., Daniell, T. J., Hallett, P. D., Neilson, R., Wheatley, R. E., Osler, G., & Bohanec, M. (2010). Integrating soil quality changes to arable agricultural systems following organic matter addition, or adoption of a ley-arable rotation. Applied Soil Ecology, 46(1), 43–53. https://doi.org/10.1016/j.apsoil.2010.06.012
Guillot, E., Bertrand, I., Rumpel, C., Gomez, C., Arnal, D., Abadie, J., & Hinsinger, P. (2021). Spatial heterogeneity of soil quality within a Mediterranean alley cropping agroforestry system: Comparison with a monocropping system. European Journal of Soil Biology, 105, 103330. https://doi.org/10.1016/j.ejsobi.2021.103330
Guo, X., Li, H., Yu, H., Li, W., Ye, Y., & Biswas, A. (2018). Drivers of spatio-temporal changes in paddy soil pH in Jiangxi Province, China from 1980 to 2010. Scientific Reports, 8(1), 2702. https://doi.org/10.1038/s41598-018-20873-5
Hair, J.F., Black, W.C., Babin, B.J. & Anderson, R.E. (2010). Multivariate data analysis (7th ed.), Pearson.
Han, P., Zhang, W., Wang, G., Sun, W., & Huang, Y. (2016). Changes in soil organic carbon in croplands subjected to fertilizer management: A global meta-analysis. Scientific Reports, 6(1), 27199. https://doi.org/10.1038/srep27199
Huang, W., Zong, M., Fan, Z., Feng, Y., Li, S., Duan, C., & Li, H. (2021). Determining the impacts of deforestation and corn cultivation on soil quality in tropical acidic red soils using a soil quality index. Ecological Indicators, 125, 107580. https://doi.org/10.1016/j.ecolind.2021.107580
Hussain, M. S., Uddin, M. J., & Mohiuddin, A. S. M. (2013). Assessment of soil quality in Bangladesh with organic carbon as the indicator property. Dhaka University Journal of Biological Sciences, 22(2), 163–174.
Hussain, A., Ali, H., Begum, F., Hussain, A., Khan, M. Z., Guan, Y., Zhou, J., Saif-Ud-Din, & Hussain, K. (2021). Mapping of soil properties under different land uses in Lesser Karakoram range Pakistan. Polish Journal of Environmental Studies, 30, 1181–1189. https://doi.org/10.15244/pjoes/122443
Jackson, M. L. (1973). Soil chemical analysis (Vol. 498, pp. 151–154). Pentice Hall of India Pvt. Ltd.
Ji, L., Ni, K., Wu, Z., Zhang, J., Yi, X., Yang, X., Ling, N., You, Z., Gou, S., & Ruan, J. (2020). Effect of organic substitution rates on soil quality and fungal community composition in a tea plantation with long-term fertilization. Biology and Fertility of Soils, 56, 633–646. https://doi.org/10.1007/s00374-020-01439-y
Jian, J., Du, X., & Stewart, R. D. (2020). A database for global soil health assessment. Scientific Data, 7(1), 16. https://doi.org/10.1038/s41597-020-0356-3
Johnson, A. I. (1963). A field method for measurement of infiltration. In Geological survey water-supply paper, 1544-F. United states government printing office.
Johnson, R. A., & Wichern, D. W. (2002). Applied multivariate statistical analysis. Qatar University.
Kafei, F., Rezapour, S., Dalalian, M. R., Sabbaghtazeh, E., & Rafieyan, O. (2023). Soil quality index as affected by long-time continuous cultivation in a Mediterranean sub-humid region. Rendiconti Lincei. Scienze Fisiche e Naturali, 34(2), 563–575. https://doi.org/10.1007/s12210-023-01148-2
Kaiser, H. F. (1960). The application of electronic computers to factor analysis. Educational and Psychological Measurement, 20(1), 141–151. https://doi.org/10.1177/001316446002000116
Kanwar, J. S. (1994). Relevance of soil management in sustainable agriculture in dryland areas. Bull Ind Soc Soil Sci, 16, 1–11.
Karlen, D. L., & Stott, D. E. (1994). A framework for evaluating physical and chemical indicators of soil quality. Defining Soil Quality for a Sustainable Environment, 35, 53–72. https://doi.org/10.2136/sssaspecpub35.c4
Karlen, D. L., Wollenhaupt, N. C., Erbach, D. C., Berry, E. C., Swan, J. B., Eash, N. S., & Jordahl, J. L. (1994). Crop residue effects on soil quality following 10-years of no-till corn. Soil and Tillage Research, 31(2–3), 149–167. https://doi.org/10.1016/0167-1987(94)90077-9
Karlen, D. L., Mausbach, M. J., Doran, J. W., Cline, R. G., Harris, R. F., & Schuman, G. E. (1997). Soil quality: A concept, definition, and framework for evaluation (a guest editorial). Soil Science Society of America Journal, 61(1), 4–10. https://doi.org/10.2136/sssaj1997.03615995006100010001x
Karlen, D. L., Rosek, M. J., Gardner, J. C., Allan, D. L., Alms, M. J., Bezdicek, D. F., Flock, M., Huggins, D. R., Miller, B. S., & Staben, M. L. (1999). Conservation Reserve Program effects on soil quality indicators. Journal of Soil and Water Conservation, 54(1), 439–444.
Karlen, D. L., Andrews, S. S., & Doran, J. W. (2001). Soil quality: current concepts and applications.
Kaur, S., & Chauhan, B. S. (2023). Challenges and opportunities to sustainable crop production. Plant Small RNA in Food Crops, 25–43. https://doi.org/10.1016/B978-0-323-91722-3.00006-3
Keller, T., & Håkansson, I. (2010). Estimation of reference bulk density from soil particle size distribution and soil organic matter content. Geoderma, 154(3–4), 398–406. https://doi.org/10.1016/j.geoderma.2009.11.013
Kemper, W. D., Rosenau, R., & Nelson, S. (1985). Gas displacement and aggregate stability of soils. Soil Science Society of America Journal, 49(1), 25–28. https://doi.org/10.2136/sssaj1985.03615995004900010004x
Kemper, W. D., & Rosenau, R. C. (1986). Aggregate stability and size distribution. Methods of Soil Analysis: Part 1 Physical and Mineralogical Methods, 5, 425–442. https://doi.org/10.2136/sssabookser5.1.2ed.c17
Kjeldahl, J. G. C. T. (1883). Neue methode zur bestimmung des stickstoffs in organischen körpern. Zeitschrift Für Analytische Chemie, 22(1), 366–382. https://doi.org/10.1007/BF01338151
Klingebiel, A. A., & Montgomery, P. H. (1961). Land-capability classification (No. 210). Soil Conservation Service, US Department of Agriculture.
Klopp H., & Bly A. (2023) Bulk density is an indicator of soil health. SDSU Extension. Retrieved from https://extension.sdstate.edu/bulk-density-indicator-soil-health. Accessed on 18.12.2023.
Kordi, M., Farrokhi, N., Pech-Canul, M. I., & Ahmadikhah, A. (2023). Rice Husk at a Glance: From Agro-Industrial to Modern Applications. Rice Science. https://doi.org/10.1016/j.rsci.2023.08.005
Kumar, T., & Jhariya, D. C. (2015). Land quality index assessment for agricultural purpose using multi-criteria decision analysis (MCDA). Geocarto International, 30(7), 822–841. https://doi.org/10.1080/10106049.2014.997304
Lal, R. (2004). Soil carbon sequestration impacts on global climate change and food security. Science, 304(5677), 1623–1627. https://doi.org/10.1126/science.1097396
Lal, R. (2015). Sequestering carbon and increasing productivity by conservation agriculture. Journal of Soil and Water Conservation, 70, 55A-62A. https://doi.org/10.2489/jswc.70.3.55a
Larson, W. E., & Pierce, F. J. (1991). Conservation and enhancement of soil quality. International Board for Soil Research and Management Inc. (IBSRAM).
Larson, W. E., & Pierce, F. J. (1994). The dynamics of soil quality as a measure of sustainable management. Defining Soil Quality for a Sustainable Environment, 35, 37–51. https://doi.org/10.2136/sssaspecpub35.c3
Leul, Y., Assen, M., Damene, S., & Legass, A. (2023). Effects of land use types on soil quality dynamics in a tropical sub-humid ecosystem, western Ethiopia. Ecological Indicators, 147, 110024. https://doi.org/10.1016/j.ecolind.2023.110024
Li, X., Li, H., Yang, L., & Ren, Y. (2018). Assessment of soil quality of croplands in the Corn Belt of Northeast China. Sustainability, 10(1), 248. https://doi.org/10.3390/su10010248
Li, P., Wu, M., Kang, G., Zhu, B., Li, H., Hu, F., & Jiao, J. (2020). Soil quality response to organic amendments on dryland red soil in subtropical China. Geoderma, 373, 114416. https://doi.org/10.1016/j.geoderma.2020.114416
Li, Y., Ma, J., Li, Y., Jia, Q., Shen, X., & Xia, X. (2023). Spatiotemporal variations in the soil quality of agricultural land and its drivers in China from 1980 to 2018. Science of the Total Environment, 892, 164649. https://doi.org/10.1016/j.scitotenv.2023.164649
Liebig, M. A., & Doran, J. W. (1999). Impact of organic production practices on soil quality indicators (Vol. 28, No. 5, pp. 1601–1609). American Society of Agronomy, Crop Science Society of America, and Soil Science Society of America. https://doi.org/10.2134/jeq1999.00472425002800050026x
Liebig, M. A., Varvel, G., & Doran, J. (2001). A simple performance-based index for assessing multiple agroecosystem functions. Agronomy Journal, 93(2), 313–318. https://doi.org/10.2134/agronj2001.932313x
Liu, C., Liu, Y., Li, Z., Zhang, G., & Chen, F. (2017). A novel way to establish fertilization recommendations based on agronomic efficiency and a sustainable yield index for rice crops. Scientific Reports, 7(1), 1001. https://doi.org/10.1038/s41598-017-01143-2
Majhi, P., Rout, K. K., Nanda, G., & Singh, M. (2019). Soil quality for rice productivity and yield sustainability under long-term fertilizer and manure application. Communications in soil science and plant analysis, 50(11), 1330–1343. https://doi.org/10.1080/00103624.2019.1614607
Maleki, S., Zeraatpisheh, M., Karimi, A., Sareban, G., & Wang, L. (2022). Assessing variation of soil quality in agroecosystem in an arid environment using digital soil mapping. Agronomy, 12(3), 578. https://doi.org/10.3390/agronomy12030578
Manna, M. C., Swarup, A., Wanjari, R. H., Ravankar, H. N., Mishra, B., Saha, M. N., Singh, Y. V., Sahi, D. K., & Sarap, P. A. (2005). Long-term effect of fertilizer and manure application on soil organic carbon storage, soil quality and yield sustainability under sub-humid and semi-arid tropical India. Field Crops Research, 93(2–3), 264–280. https://doi.org/10.1016/j.fcr.2004.10.006
Marzaioli, R., D’Ascoli, R., De Pascale, R. A., & Rutigliano, F. A. (2010). Soil quality in a Mediterranean area of Southern Italy as related to different land use types. Applied Soil Ecology, 44(3), 205–212. https://doi.org/10.1016/j.apsoil.2009.12.007
Masto, R. E., Chhonkar, P. K., Singh, D., & Patra, A. K. (2007). Soil quality response to long-term nutrient and crop management on a semi-arid Inceptisol. Agriculture, Ecosystems & Environment, 118(1–4), 130–142. https://doi.org/10.1016/j.agee.2006.05.008
Masto, R. E., Chhonkar, P. K., Purakayastha, T. J., Patra, A. K., & Singh, D. (2008). Soil quality indices for evaluation of long-term land use and soil management practices in semi-arid sub-tropical India. Land Degradation & Development, 19(5), 516–529. https://doi.org/10.1002/ldr.857
McDaniel, M. D., Tiemann, L. K., & Grandy, A. S. (2014). Does agricultural crop diversity enhance soil microbial biomass and organic matter dynamics? A Meta-Analysis. Ecological Applications, 24(3), 560–570. https://doi.org/10.1890/13-0616.1
Meetei, T. T., Choudhury, B. U., Mohapatra, K. P., Singh, N. U., Das, A., & Devi, Y. B. (2020). Effect of 25 years old agroforestry practices on soil quality attributes in the north eastern Himalayan region of India. IJCS, 8(1), 2371–2379. https://doi.org/10.22271/chemi.2020.v8.i1aj.8623
Mukherjee, A., & Lal, R. (2014). Comparison of soil quality index using three methods. PLoS ONE, 9(8), e105981. https://doi.org/10.1371/journal.pone.0105981
Munda, S., Nayak, A. K., Mishra, P. N., Bhattacharyya, P., Mohanty, S., Kumar, A., Kumar, U., Baig, M. J., Tripathi, R., Shahid, M. A., & Thilagam, V. K. (2016). Combined application of rice husk biochar and fly ash improved the yield of lowland rice. Soil Research, 54(4), 451–459. https://doi.org/10.1071/SR15295
Mustafa, A., Minggang, X., Shah, S. A. A., Abrar, M. M., Nan, S., Baoren, W., Zejiang, Z., Saeed, Q., Naveed, M., & Núñez-Delgado, A. (2020). Soil aggregation and soil aggregate stability regulate organic carbon and nitrogen storage in a red soil of southern China. Journal of Environmental Management, 270, 110894. https://doi.org/10.1016/j.jenvman.2020.110894
NAAS. (2009). Crop response and nutrient ratio, policy paper 42. National Academy of Agricultural Sciences. Retrieved from https://naas.org.in/Policy%20Papers/policy%2042. Accessed on 04.05.2023.
Nabiollahi, K., Taghizadeh-Mehrjardi, R., Kerry, R., & Moradian, S. (2017). Assessment of soil quality indices for salt-affected agricultural land in Kurdistan Province. Iran. Ecological Indicators, 83, 482–494. https://doi.org/10.1016/j.ecolind.2017.08.001
Nabiollahi, K., Golmohamadi, F., Taghizadeh-Mehrjardi, R., Kerry, R., & Davari, M. (2018). Assessing the effects of slope gradient and land use change on soil quality degradation through digital mapping of soil quality indices and soil loss rate. Geoderma, 318, 16–28. https://doi.org/10.1016/j.geoderma.2017.12.024
Nambiar, K. K. M., & Ghosh, A. B. (1984). Highlights of research of a long term fertilizer experiment in India (1971-82). LTFE Research Bulletin No. 1. New Delhi, India: Indian Agricultural Research Institute
Nambiar, K. K. M. (1995). Major Cropping Systems in India. In: Barnett, et al. (Eds.), Agricultural Sustainability: Economics, Environmental and Statistical Considerations, John Wiley & Sons, Chichester, 1995, pp. 135–142.
Nasir, M. J., Haider, M. F., Ali, Z., Akhtar, W., & Alam, S. (2024). Evaluation of soil quality through simple additive soil quality index (SQI) of Tehsil Charsadda, Khyber Pakhtunkhwa, Pakistan. Journal of the Saudi Society of Agricultural Sciences, 23(1), 42–54. https://doi.org/10.1016/j.jssas.2023.09.001
Nath, T. N. (2014). Soil texture and total organic matter content and its influences on soil water holding capacity of some selected tea growing soils in Sivasagar district of Assam India. International Journal of Chemical, 12(4), 1419–1429.
Nehrani, S. H., Askari, M. S., Saadat, S., Delavar, M. A., Taheri, M., & Holden, N. M. (2020). Quantification of soil quality under semi-arid agriculture in the northwest of Iran. Ecological Indicators, 108, 105770. https://doi.org/10.1016/j.ecolind.2019.105770
Nguemezi, C., Tematio, P., Yemefack, M., Tsozue, D., & Silatsa, T. B. F. (2020). Soil quality and soil fertility status in major soil groups at the Tombel area, South-West Cameroon. Heliyon, 6(2). https://doi.org/10.1016/j.heliyon.2020.e03432
Nielsen, D. R., & Bouma, J. (1985). Soil spatial variability. ISSS and the SSSA.
NRCCA. (2010). Soil and water management. Cornel University. Retrieved from https://nrcca.cals.cornell.edu/soil/CA2/index.php. Accessed on 02.11.2023
Olsen, S. R. (1954). Estimation of available phosphorus in soils by extraction with sodium bicarbonate (No. 939). US Department of Agriculture.
Oshi, P. K., Aggarwal, P. K., Tyagi, N. K., & Pandey, D. (2015). Role of development policies in combating climate change issues in Indian Agriculture: A frist order assessment of irrigation and fertilizer policies. https://doi.org/10.22004/ag.econ.211817
Pahalvi, H. N., Rafiya, L., Rashid, S., Nisar, B., & Kamili, A. N. (2021). Chemical fertilizers and their impact on soil health. Microbiota and Biofertilizers, Vol 2: Ecofriendly Tools for Reclamation of Degraded Soil Environs, 1–20. https://doi.org/10.1007/978-3-030-61010-4_1
Page, A. L. (1982). Methods of soil analysis. Part 2. Chemical and microbiological properties (p. 1159). American Society of Agronomy, Soil Science Society of America.
Pan, G., Smith, P., & Pan, W. (2009). The role of soil organic matter in maintaining the productivity and yield stability of cereals in China. Agriculture, Ecosystems & Environment, 129(1–3), 344–348. https://doi.org/10.1016/j.agee.2008.10.008
Patra, A., Sharma, V. K., Nath, D. J., Dutta, A., Purakayastha, T. J., Kumar, S., Barman, M., Chobhe, K. A., & Kumawat, C. (2023). Long-term impact of integrated nutrient management on sustainable yield index of rice and soil quality under acidic inceptisol. Archives of Agronomy and Soil Science, 69(7), 1111–1128. https://doi.org/10.1080/03650340.2022.2056597
Piper, C. S. (1966). Soil and Plant Analysis. Hans Publisher.
Posadas, R. C., Salting, A. A. A., Evangelista, R. D. A., Real, R. M., Jr., Casidsid, L. A., & Sagarino, E. O. (2022). Soil Quality Index for the Two Agricultural Areas along the Naboc River in Davao De Oro, Philippines Contaminated with Mercury. Philippine Journal of Science, 151(5), 2031–2046.
Prasad, R., Newaj, R., Singh, R., Saroj, N. K., Tripathi, V. D., Shukla, A., Singh, P., & Chaturvedi, O. P. (2017). Soil quality index (SQI) for assessing soil health of agroforestry system: effect of Hardwickia binata Roxb. tree density on SQI in Bundelkhand, central India. Indian Journal of Agroforestry, 19(2), 38–45.
Qi, Y., Darilek, J. L., Huang, B., Zhao, Y., Sun, W., & Gu, Z. (2009). Evaluating soil quality indices in an agricultural region of Jiangsu Province China. Geoderma, 149(3–4), 325–334.
Rahmanipour, F., Marzaioli, R., Bahrami, H. A., Fereidouni, Z., & Bandarabadi, S. R. (2014). Assessment of soil quality indices in agricultural lands of Qazvin Province Iran. Ecological Indicators, 40, 19–26. https://doi.org/10.1016/j.ecolind.2013.12.003
Raiesi, F., & Kabiri, V. (2016). Identification of soil quality indicators for assessing the effect of different tillage practices through a soil quality index in a semi-arid environment. Ecological Indicators, 71, 198–207. https://doi.org/10.1016/j.ecolind.2016.06.061
Rajiv Mehta, R. M. (2007). Agricultural growth vision and supply side issues concerning fertilisers.
Rakshit, R., Das, A., Padbhushan, R., Sharma, R. P., & Kumar, S. (2018). Assessment of soil quality and identification of parameters influencing system yield under longterm fertilizer trial. Journal of the Indian Society of Soil Science, 66(2), 166–171. https://doi.org/10.5958/0974-0228.2018.00021.X
Ram, S., Singh, V., & Sirari, P. (2016). Effects of 41 years of application of inorganic fertilizers and farm yard manure on crop yields, soil quality, and sustainable yield index under a rice-wheat cropping system on Mollisols of North India. Communications in Soil Science and Plant Analysis, 47(2), 179–193. https://doi.org/10.1080/00103624.2015.1109653
Rattan, R. K. (2015). Soil quality assessment and management: Critical analysis in the Indian context. Indian Journal of Fertilisers, 11(4), 40–61.
Ren, X., Chen, F., Ma, T., & Hu, Y. (2020). Soil quality characteristics as affected by continuous rice cultivation and changes in cropping systems in South China. Agriculture, 10(10), 443. https://doi.org/10.3390/agriculture10100443
Rezaee, L., Moosavi, A. A., Davatgar, N., & Sepaskhah, A. R. (2020). Soil quality indices of paddy soils in Guilan province of northern Iran: Spatial variability and their influential parameters. Ecological Indicators, 117, 106566. https://doi.org/10.1016/j.ecolind.2020.106566
Rezapour, S., Nouri, A., Jalil, H. M., Hawkins, S. A., & Lukas, S. B. (2021). Influence of treated wastewater irrigation on soil nutritional-chemical attributes using soil quality Index. Sustainability, 13(4), 1952. https://doi.org/10.3390/su13041952
Roy, D., Datta, A., Jat, H. S., Choudhary, M., Sharma, P. C., Singh, P. K., & Jat, M. L. (2022). Impact of long term conservation agriculture on soil quality under cereal based systems of North West India. Geoderma, 405, 115391. https://doi.org/10.1016/j.geoderma.2021.115391
Rudel, T. K., Kwon, O. J., Paul, B. K., Boval, M., Rao, I. M., Burbano, D., McGroddy, M., Lerner, A. M., White, D., Cuchillo, M., Luna, M., & Peters, M. (2016). Do smallholder, mixed crop-livestock livelihoods encourage sustainable agricultural practices? A Meta-Analysis. Land, 5(1), 6. https://doi.org/10.3390/land5010006
Ruf, T., Makselon, J., Udelhoven, T., & Emmerling, C. (2018). Soil quality indicator response to land-use change from annual to perennial bioenergy cropping systems in Germany. Gcb Bioenergy, 10(7), 444–459. https://doi.org/10.1111/gcbb.12513
Saha, S., Saha, B., Mohanty, S., Ray, M., Hazra, G. C., Chatterjee, S., & Mukhopadhyay, S. K. (2022). Identifying suitable soil quality indicators in an Indian rice-wheat system as influenced by long-term (30 years) inorganic-organic fertilization. Archives of Agronomy and Soil Science, 68(5), 704–718. https://doi.org/10.1080/03650340.2020.1851684
Samaei, F., Emami, H., & Lakzian, A. (2022). Assessing soil quality of pasture and agriculture land uses in Shandiz county, northwestern Iran. Ecological Indicators, 139, 108974. https://doi.org/10.1016/j.ecolind.2022.108974
Saurabh, K., Rao, K. K., Mishra, J. S., Kumar, R., Poonia, S. P., Samal, S. K., Roy, H. S., Dubey, A. K., Choubey, A. K., Mondal, S., Bhatt, B. P., Verma, M., & Malik, R. K. (2021). Influence of tillage based crop establishment and residue management practices on soil quality indices and yield sustainability in rice-wheat cropping system of Eastern Indo-Gangetic Plains. Soil and Tillage Research, 206, 104841. https://doi.org/10.1016/j.still.2020.104841
Sedivy, P., Dusilova, T., Hajek, M., Burian, M., Krššák, M., & Dezortova, M. (2021). In vitro 31P MR chemical shifts of in vivo-detectable metabolites at 3T as a basis set for a pilot evaluation of skeletal muscle and liver 31P spectra with LCModel software. Molecules, 26(24), 7571. https://doi.org/10.1017/S001447971300046X
Shadekar, V. (2018). Soil aggregate stability-a soil health physical indicator. Retrieved from https://agcrops.osu.edu/newsletter/corn-newsletter/2018-02/soil-aggregatestability-%E2%80%93-soil-health-physical-indicator. Accessed on 10.08.2023.
Shah, T. I., Shah, A. M., Bangroo, S. A., Sharma, M. P., Aezum, A. M., Kirmani, N. A., ... & Ahmad, L. (2022). Soil quality index as affected by integrated nutrient management in the himalayan foothills. Agronomy, 12(8), 1870. https://doi.org/10.3390/agronomy12081870
Sharma, K. L., Mandal, U. K., Srinivas, K., Vittal, K. P. R., Mandal, B., Grace, J. K., & Ramesh, V. (2005). Long-term soil management effects on crop yields and soil quality in a dryland Alfisol. Soil and Tillage Research, 83(2), 246–259. https://doi.org/10.1016/j.still.2004.08.002
Sharma, K. L., Grace, J. K., Mandal, U. K., Gajbhiye, P. N., Srinivas, K., Korwar, G. R., Bindu, V. H., Ramesh, V., Ramachandran, K., & Yadav, S. K. (2008). Evaluation of long-term soil management practices using key indicators and soil quality indices in a semi-arid tropical Alfisol. Soil Research, 46(4), 368–377. https://doi.org/10.1071/SR07184
Sharma, K. L., Balaguruvaiah, D., Babu, M. V. S., Reddy, B. R., Rao, C. S., Mishra, P. K. G. J., Kusuma, R. G., Madhavi, M., Srinivas, K., Mandal, U. K., Korwar, G. R., Maruthi, S. G., & Chary, G. R. (2010). Long-term impact of soil and nutrient management practices on soil quality in rainfed Alfisols at Anantapur in Andhra Pradesh. Indian Journal of Dryland Agricultural Research and Development, 25(1), 74–85.
Sheidai Karkaj, E., Sepehry, A., Barani, H., Motamedi, J., & Shahbazi, F. (2019). Establishing a suitable soil quality index for semi-arid rangeland ecosystems in northwest of Iran. Journal of Soil Science and Plant Nutrition, 19, 648–658.
Sheikh, A. T. (2022). Essays on the economics of soil quality: Development of soil quality indicators and the adoption of sustainable land management practices and willingness to accept for land restoration and conservation programs among land managers in Punjab, Pakistan. The University of Western Australia.
Shukla, K., Kumar, P., Mann, G. S., & Khare, M. (2020). Mapping spatial distribution of particulate matter using Kriging and Inverse Distance Weighting at supersites of megacity Delhi. Sustainable Cities and Society, 54, 101997.
Singh, R. P., Das, S. K., Bhaskara Rao, U. M., & Narayana Reddy, M. (1990). CRIDA Report (106 pp). CRIDA.
Singh, S. R., Kundu, D. K., Dey, P., Singh, P., & Mahapatra, B. S. (2018). Effect of balanced fertilizers on soil quality and lentil yield in Gangetic alluvial soils of India. The Journal of Agricultural Science, 156(2), 225–240. https://doi.org/10.1017/S0021859618000254
Singh, S. R., Yadav, P., Singh, D., Tripathi, M. K., Bahadur, L., Singh, S. P., Mishra, A., & Kumar, S. (2020). Cropping systems influence microbial diversity, soil quality and crop yields in Indo-Gangetic plains of India. European Journal of Agronomy, 121, 126152. https://doi.org/10.1016/j.eja.2020.126152
Sinha, N. K., Chopra, U. K., & Singh, A. K. (2014). Cropping system effects on soil quality for three agro-ecosystems in India. Experimental Agriculture, 50(3), 321–342. https://doi.org/10.1017/S001447971300046X
Smith, J. L., Halvorson, J. J., & Papendick, R. I. (1993). Using multiple-variable indicator kriging for evaluating soil quality. Soil Science Society of America Journal, 57(3), 743–749.
Srinivasarao, C., Venkateswarlu, B., Lal, R., Singh, A. K., & Kundu, S. (2013). Sustainable management of soils of dryland ecosystems of India for enhancing agronomic productivity and sequestering carbon. Advances in Agronomy, 121, 253–329. https://doi.org/10.1016/B978-0-12-407685-3.00005-0
Srivastava, P., Singh, R., Tripathi, S., & Raghubanshi, A. S. (2016). An urgent need for sustainable thinking in agriculture–An Indian scenario. Ecological Indicators, 67, 611–622. https://doi.org/10.1016/j.ecolind.2016.03.015
Sun, B., Zhou, S., & Zhao, Q. (2003). Evaluation of spatial and temporal changes of soil quality based on geostatistical analysis in the hill region of subtropical China. Geoderma, 115(1–2), 85–99. https://doi.org/10.1016/S0016-7061(03)00078-8
Tahat, M., Alananbeh, K., Othman, Y., & Leskovar, D. (2020). Soil Health and Sustainable Agriculture. Sustainability, 12(12), 4859. https://doi.org/10.3390/su12124859
Tang, Q., Cotton, A., Wei, Z., Xia, Y., Daniell, T., & Yan, X. (2022). How does partial substitution of chemical fertiliser with organic forms increase sustainability of agricultural production? Science of the Total Environment, 803, 149933. https://doi.org/10.1016/j.scitotenv.2021.149933
Temjen, W., Singh, M. R., & Ajungla, T. (2022). Effect of shifting cultivation and fallow on soil quality index in Mokokchung district, Nagaland India. Ecological Processes, 11(1), 42. https://doi.org/10.1186/s13717-022-00386-w
The World Bank. (2023). Fertilizer consumption (kilograms per hectare arable land). Retrieved from https://data.worldbank.org/indicator/AG.CON.FERT.ZS. Accessed on 15.01.2024. Accessed on 24.01.2024
Thomas, G. W. (1996). Soil pH and soil acidity. Methods of Soil Analysis: Part 3 Chemical Methods, 5, 475–490. https://doi.org/10.2136/sssabookser5.3.c16
Tian, D., & Niu, S. (2015). A global analysis of soil acidification caused by nitrogen addition. Environmental Research Letters, 10(2), 024019. https://doi.org/10.1088/1748-9326/10/2/024019
Tisdale, S. L., Nelson, W. L., & Beaton, J. D. (1985). Soil fertility and fertilizers. Collier Macmillan Publishers.
Tricker, A. S. (1978). The infiltration cylinder: Some comments on its use. Journal of Hydrology, 36(3–4), 383–391. https://doi.org/10.1016/0022-1694(78)90156-7
Uma, R. N., Prem Sudha, R., & Murali, K. (2016). Analysis of physico-chemical characteristics of soil and SQI around municipal solid waste dump yard in Vellalore-Coimbatore, Tamilnadu. India. International Journal of Chemical Sciences, 14(4), 3265–3276.
USDA-NRCS. (1998). Soil quality indicators: pH. Retrieved from https://www.fs.usda.gov/Internet/FSE_DOCUMENTS/stelprdb5293776. Accessed on 18.08.2023.
USDA-NRCS. (2001). Rangeland soil quality-aggregate stability. Retrieved from https://www.nrcs.usda.gov/sites/default/files/202212/Rangeland_Soil_Quality_Aggregate_Stability_0. Accessed on 05.07.2023. Accessed on 21.08.2023
USDA-NRCS. (2011). Soil quality indicators. Retrieved from https://www.nrcs.usda.gov/sites/default/files/202210/Soil%20Electrical%20Conductivity. Accessed 07.03.2023
USDA-NRCS. (2014). Soil respiration. Retrieved from https://www.nrcs.usda.gov/sites/default/files/2022-10/Soil%20Respiration. Accessed on 15.10.2023.
USDA-NRCS. (2019). Soil bulk density/moisture/aeration. Retrieved from https://www.nrcs.usda.gov/sites/default/files/202210/Soil%20Bulk%20Density%20Moisture%20Aeration. Accessed on 25.04.2023.
Vaezi, A. R., Mirkhani, R., Rezaei, H., & Esmaeilnejad, L. (2020). Investigating Spatial Variability of Soil Quality Indices in Nazar Abad Fields, West of Alborz Province. Iranian Journal of Soil and Water Research, 51(7), 1755–1768. https://doi.org/10.22059/ijswr.2020.299323.668538
Valpassos, M. A. R., Cavalcante, E. G. S., Cassiolato, A. M. R., & Alves, M. C. (2001). Effects of soil management systems on soil microbial activity, bulk density and chemical properties. Pesquisa Agropecuária Brasileira, 36, 1539–1545. https://doi.org/10.1590/S0100-204X2001001200011
Van Dang, L., Ngoc, N. P., & Hung, N. N. (2021). Soil quality and pomelo productivity as affected by chicken manure and cow dung. The Scientific World Journal, 2021. https://doi.org/10.1155/2021/6289695
Vance, E. D., Brookes, P. C., & Jenkinson, D. S. (1987). An extraction method for measuring soil microbial biomass C. Soil Biology and Biochemistry, 19(6), 703–707. https://doi.org/10.1016/0038-0717(87)90052-6
Vasu, D., Singh, S. K., Ray, S. K., Duraisami, V. P., Tiwary, P., Chandran, P., Nimkar, A. M., & Anantwar, S. G. (2016). Soil quality index (SQI) as a tool to evaluate crop productivity in semi-arid Deccan plateau, India. Geoderma, 282, 70–79. https://doi.org/10.1016/j.geoderma.2016.07
Walkley, A., & Black, I. A. (1934). An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Science, 37(1), 29–38.
Wander, M. M., & Bollero, G. A. (1999). Soil quality assessment of tillage impacts in Illinois. Soil Science Society of America Journal, 63(4), 961–971. https://doi.org/10.2136/sssaj1999.634961x
Wang, X., & Gong, Z. (1998). Assessment and analysis of soil quality changes after eleven years of reclamation in subtropical China. Geoderma, 81(3–4), 339–355. https://doi.org/10.1016/S0016-7061(97)00109-2
Wanjari, R. H., Singh, M. V., & Ghosh, P. K. (2004). Sustainable yield index: An approach to evaluate the sustainability of long-term intensive cropping systems in India. Journal of Sustainable Agriculture, 24(4), 39–56. https://doi.org/10.1300/J064v24n04_05
Warkentin, B. P., & Fletcher, H. F. (1977). Soil quality for intensive agriculture. In Proceedings of the international seminar on soil environment and fertility management in intensive agriculture.
Wienhold, B. J., Andrews, S. S., & Karlen, D. L. (2004). Soil quality: A review of the science and experiences in the USA. Environmental Geochemistry and Health, 26, 89–95.
Wollenhaupt, N. C., Wolkowski, R. P., & Clayton, M. K. (1994). Mapping soil test phosphorus and potassium for variable-rate fertilizer application. Journal of Production Agriculture, 7(4), 441–448. https://doi.org/10.2134/jpa1994.0441
Wolschick, N. H., Barbosa, F. T., Bertol, I., Bagio, B., & Kaufmann, D. S. (2018). Long-term effect of soil use and management on organic carbon and aggregate stability. Revista Brasileira de Ciência do Solo, 42. https://doi.org/10.1590/18069657rbcs20170393
World Bank Open Data (2021) Fertilizer consumption (kilograms per hectare of arable land). Retrieved December 18, 2023, from https://data.worldbank.org/indicator/AG.CON.FERT.ZS. Accessed on 28 September 2023.
Xing, Y., Wang, N., Niu, X., Jiang, W., & Wang, X. (2021). Assessment of potato farmland soil nutrient based on MDS-SQI Model in the Loess Plateau. Sustainability, 13(7), 3957. https://doi.org/10.3390/su13073957
Yadav, K. K., Mali, N. L., Kumar, S., Surya, J. N., Moharana, P. C., Nogiya, M., & Meena, R. L. (2022). Assessment of soil quality and spatial variability of soil properties using geo-spatial techniques in sub-humid Southern Plain of Rajasthan, India. Journal of the Indian Society of Soil Science, 70(1), 69–85.
Yu, P., Liu, S., Zhang, L., Li, Q., & Zhou, D. (2018). Selecting the minimum data set and quantitative soil quality indexing of alkaline soils under different land uses in northeastern China. Science of the Total Environment, 616, 564–571. https://doi.org/10.1016/j.scitotenv.2017.10.301
Zahedifar, M. (2023). Assessing alteration of soil quality, degradation, and resistance indices under different land uses through network and factor analysis. CATENA, 222, 106807. https://doi.org/10.1016/j.catena.2022.106807
Zeifman, L., Hertog, S., Kantorova, V., & Wilmoth, J. (2022). A world of 8 billion. Retrieved from https://www.un.org/development/desa/pd/sites/www.un.org.development.desa.pd/files/undesa_pd_2022_pb_140. Accessed on 22.05.2023.
Zhao, X., Tong, M., He, Y., Han, X., & Wang, L. (2021). A comprehensive, locally adapted soil quality indexing under different land uses in a typical watershed of the eastern Qinghai-Tibet Plateau. Ecological Indicators, 125, 107445. https://doi.org/10.1016/j.ecolind.2021.107445
Zhengchao, Z., Zhuoting, G., Zhouping, S., & Fuping, Z. (2013). Effects of long-term repeated mineral and organic fertilizer applications on soil organic carbon and total nitrogen in a semi-arid cropland. European Journal of Agronomy, 45, 20–26. https://doi.org/10.1016/j.eja.2012.11.002
Zhou, M., Xiao, Y., Li, Y., Zhang, X., Wang, G., Jin, J., ... & Liu, X. (2022). Soil quality index evaluation model in responses to six-year fertilization practices in Mollisols. Archives of Agronomy and Soil Science, 68(2), 180–194. https://doi.org/10.1080/03650340.2020.1827395
Acknowledgements
We are thankful to Kushal Dasgupta and others technical staff of the soil laboratory of Ramkrishna Ashram Krishi Vigyan Kendra, Nimpith, South 24-Parganas, West Bengal for their constant help in soil testing and guide for the critical ranges of the various soil parameters. We are also thankful to Sri Jagannath Chatterjee, the Deputy Director Agriculture for Burdwan (East) for providing relevant data and supportive maps. We also thank to the Joint Director and others staff of Soil Testing and Chemist Department in Purba Bardhaman for their help. Corresponding author also likes to thank the University Grant Commission (UGC) for fellowship assistant.
Funding
The study did not receive any external funding.
Author information
Authors and Affiliations
Contributions
The co-author, Biswaranjan Mistri developed the conceptual framework, verified the analytical results and presentation.
Corresponding author, Piyush Maji took the lead in writing the manuscript, data collection, performed the analytical calculation, interpreted the results and designed the model.
All the authors provided critical feedback and helped to shape the research, analysis and manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Competing interests
The authors declare no competing interests.
Research involving Human Participants and/or Animals
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Informed consent was obtained from all individual participants involved in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Maji, P., Mistri, B. Comparative assessment of soil quality dynamics using SQI modelling approach: a study in rice bowl of West Bengal, India. Environ Monit Assess 196, 567 (2024). https://doi.org/10.1007/s10661-024-12697-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-024-12697-w