Abstract
Surface oil sands mining and extraction in northern Alberta’s Athabasca oil sands region produce large volumes of oil sands process–affected water (OSPW). OSPW is a complex mixture containing major contaminant classes including trace metals, polycyclic aromatic hydrocarbons, and naphthenic acid fraction compounds (NAFCs). Naphthenic acids (NAs) are the primary organic toxicants in OSPW, and reducing their concentrations is a priority for oil sands companies. Previous evidence has shown that constructed wetland treatment systems (CWTSs) are capable of reducing the concentration of NAs and the toxicity of OSPW through bioremediation. In this study, we constructed greenhouse mesocosms with OSPW or lab process water (LPW) (i.e., water designed to mimic OSPW minus the NAFC content) with three treatments: (1) OSPW planted with Carex aquatilis; (2) OSPW, no plants; and (3) LPW, no plants. The OSPW–C. aquatilis treatment saw a significant reduction in NAFC concentrations in comparison to OSPW, no plant treatments, but both changed the distribution of the NAFCs in similar ways. Upon completion of the study, treatments with OSPW saw fewer high-molecular-weight NAs and an increase in the abundance of O3- and O4-containing formulae. Results from this study provide invaluable information on how constructed wetlands can be used in future remediation of OSPW in a way that previous studies were unable to achieve due to uncontrollable environmental factors in field experiments and the active, high-energy processes used in CWTSs pilot studies.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The Athabasca oil sands region in northern Alberta contains the third largest oil reserve in the world, accounting for an estimated 165 billion barrels of extractable oil reserves with approximately 2.9 million barrels produced daily (Canadian Association of Petroleum Producers, 2022; Government of Canada, n.d.). Surface mining of bituminous sand produces large volumes of oil sands process–affected water (OSPW) and tailings as a by-product, wherein the OSPW in tailings ponds is recycled throughout the extraction process (AER (Alberta Energy Regulator), 2017). A “zero discharge” policy has been in place since 1993, resulting in the accumulation of over 1.3 billion m3 of tailings being stored in tailings ponds across the mineable oil sands region (AER, 2017; Canadian Association of Petroleum Producers, 2022; Government of Canada, n.d.). The free water in tailings ponds, or OSPW, are comprised of < 5% solids and contain varying amounts of salt, organic compounds, and trace metals (AER, 2017; Canadian Association of Petroleum Producers, 2022; Government of Canada, n.d.). OSPW presents considerable environmental and economic challenges due to the volume, complexity, and toxicity of the mixture (Allen, 2008). Within OSPW, there are several major classes of contaminants including naphthenic acids (NAs), polycyclic aromatic hydrocarbons (PAHs), BTEX (benzene, toluene, ethylbenzene, and xylenes), phenols, heavy metals, and ions (Alharbi et al., 2021; Allen, 2008; Cancelli & Gobas, 2020; McQueen et al., 2017a, b; Puttaswamy & Liber, 2012).
Of the many components in OSPW, naphthenic acid fractional compounds (NAFCs) are some of the primary organic toxicants of concern. This complex class of organic acids includes naphthenic acids (NAs), which have been consistently implicated as being particularly toxic (Hughes et al., 2017; Morandi et al., 2015). The general formula for NAs follows the format CnH2n+ZO2, where n is the number of carbon atoms and z is a negative even integer that indicates hydrogen deficiencies caused by the presence of a ring structure (Brown & Ulrich, 2015; Grewer et al., 2010; Headley et al., 2013; Wang et al., 2016; Xue et al., 2018). The broader compound class encompassed by NAFCs includes diverse organic acids that can be aromatic, contain sulfur or nitrogen heteroatoms, and may be polyoxygenated (e.g., x ≥ 2) (Brown & Ulrich, 2015; Grewer et al., 2010; Headley et al., 2013; Wang et al., 2016; Xue et al., 2018). NAs can be particularly resistant to biodegradation when functionalized with an ante-iso-(β)-alkyl substituted moiety, a ternary substitution other than β to the carboxylic group, or a methyl substitution on a ring structure is present (Quagraine et al., 2007). Additionally, NAFCs with more rings, higher molecular weights, and increased branching tend to be more resistant to biodegradation (Biryukova et al., 2007; Del Rio et al., 2006; Han et al., 2008). Molecular features and level of unsaturation can therefore be important determinants of environmental persistence of NAFCs.
As the most toxic component of NAFCs, NAs pose considerable toxicological risks for aquatic and terrestrial biota with acute and chronic adverse responses (Anderson et al., 2012; Bartlett et al., 2017; Hagen et al., 2014; Kavanagh et al., 2013). As an example, results of studies 24 and 25 showed that the lethal concentration that causes 50% mortality (LC50) estimated for wood frog (Xenopus tropicalis) tadpoles was 4.76 mg/L of technical grade NAs (C10H18O2) over a 24-hexposure period (Melvin & Trudeau, 2012), while fathead minnows (Pimephales promelas) exposed to 10 mg/L of NAs for 21 days spawned fewer eggs with an LC50 of 32.8 mg/L of NAs for minnow embryos (Kavanagh et al., 2012). Cattails (Typha latifolia) were also susceptible to acute toxic effects when exposed to OSPW-derived NAs at 60 mg/L (Armstrong et al., 2009). In OSPW, NA concentrations and compositions can vary depending on the extraction processes, type of ore, and age of OSPW (Allen, 2008; Holowenko et al., 2002), as NAs have an estimated half-life of approximately 13 years within tailings ponds (Han et al., 2009). Quantitation across sources and methods must be done sparingly; although NAs in OSPW can range between 20 and 120 mg/L, this depends on the source of OSPW (Ajaero et al., 2018; Cancelli & Gobas, 2022; Vander Meulen et al., 2021). Measurement methods must be kept consistent within study designs, as amounts of NAs and NAFCs will also vary between different quantification methods, whether using FTIR (Grewer et al., 2010; Ripmeester & Duford, 2019; Rogers et al., 2002), gas chromatography–mass spectrometry (Grewer et al., 2010), or various forms of liquid chromatography–mass spectrometry (Ajaero et al., 2018; Duncan et al., 2016; Headley et al., 2011a, b; Janfada et al., 2006).
Due to the persistence of NAFCs and its toxicity in OSPW, NAFCs and OSPW must be adequately degraded and toxicity reduced prior to the potential discharge back into the environment. The development of effective and efficient approaches for treating OSPW is required. Many active treatment techniques are being investigated (e.g., electrical or chemical oxidation, sequestration, and/or filtration techniques) for treating NAFCs in OSPW (Abdalrhman & El-din, 2020; Allen, 2008; Alpatova et al., 2014; Kim et al., 2012; Wang et al., 2016), but there have been few treatment strategies that have shown promise for large-scale and cost-effective implementation (Quinlan & Tam, 2015; Wang et al., 2016).
Constructed wetland treatment systems (CWTS) can facilitate the remediation of OSPW by degrading NAFCs through the microbial communities and wetland plants (Cancelli & Gobas, 2020; Hendrikse et al., 2018; McQueen et al., 2017a, b). Plants are incorporated into CWTS to directly or indirectly promote microbial activity by increasing aeration and accumulating organic matter (Allen, 2008; Phillips et al., 2010; Truu et al., 2015). Biodegradation of NAFCs by leveraging natural processes could be an effective method to reduce NAFC concentrations and related toxicity compared to resource- and energy-intensive chemical and physical approaches. It is imperative to understand the various parameters that affect the success of CWTS in reducing OSPW toxicity, particularly in northern areas like the Athabasca oil sands region with short growing seasons and cold winter temperatures (Allen, 2008; Truu et al., 2009).
Previous successful petroleum bioremediation projects have focused on the potential of wetland graminoids to degrade OSPW in a CWTS (Hendrikse et al., 2018; McQueen et al., 2017a, b; Simair et al., 2021). A variety of wetland graminoids (Typha latifolia, Phragmites australis, and Scirpus acutus) were able to selectively enhance the dissipation of nonionized NA compounds, effectively reducing the toxicity of NAs in the system over 30 days (Armstrong et al., 2009). Slender wheatgrass (Elymus trachycaulus) seedlings grown in substrate saturated with OSPW were found to directly uptake five types of NAs that represent aliphatic, single-ring, and three-ring diamondoid NA classes (Alberts et al., 2021). Sandbar willow (Salix exigua) plants decreased concentrations of single-ring and triple-ring diamondoid NAs in a hydroponic system within 6 h by 95% and 84%, respectively; for the single-ring NAs, the concentration decreased by < 20% within the first 30 min (Alberts et al., 2021). Various studies focused on the bioremediation of NAs in OSPW with CWTS using C. aquatilis, a locally common species to the oil sandss region, as it is a highly competitive species capable of tolerating tailings pond water, becoming the dominant species in these systems (Caners & Lieffers, 2014; Crowe et al., 2002). C. aquatilis has outperformed other tested species in reducing the toxicity of NAFCs in OSPW (Cancelli & Gobas, 2022; Crowe et al., 2002; Simair et al., 2021).
While field- and pilot-scale CWTS projects have shown they are a viable option for NAFC remediation (Ajaero et al., 2018; Simair et al., 2021), it is essential to gain a better understanding of how these passive, low-energy systems degrade OSPW (Cancelli & Gobas, 2020). Field studies are influenced by a large variety of uncontrollable environmental variables (e.g., rainfall events, seasonality, mechanical failure, and leakages) which lead to reduced experimental replicability, thus making conclusive observations difficult (Cappello & Yakimov, 2010; McQueen et al., 2017a, b; Simair et al., 2021). Mesocosm systems are built-in environments where factors that would be integral components in a CWTS can be controlled and evaluated (Cappello & Yakimov, 2010). Mesocosm systems create a representative exposure for test organisms while maintaining enough control to examine the effects of individual experimental parameters (e.g., OSPW typology, substrate, plant species, temperature), track biotic changes, and examine chemical dissipation pathways (Boyle & Fairchild, 1997; Schindler, 1998).
This study aims to evaluate the impacts of C. aquatilis on the attenuation of NAFCs in OSPW. Our study is conducted in a continuous surface flow CWTS constructed using sand and peat. The parameters examined within this mesocosm study will help to understand the different attenuation processes occurring in a CWTS.
Materials and methods
Experimental design
The experiment was set up in the Northern Forestry Centre’s greenhouse in Edmonton, AB. It consisted of three treatments with 4 replicates each: unplanted mesocosms with lab process water (LPW) not containing NAFCs, unplanted mesocosms with OSPW, and established C. aquatilis in mesocosms with OSPW. A total of 12 individual C. aquatilis plants was transplanted into each mesocosm prior to initiating the experiment. The mesocosms were randomly placed in the greenhouse bay.
The mesocosms were constructed out of polypropylene (50.8 cm height × 33.0 cm width × 129.5 cm length; 248.1 L) and were filled evenly with 10 cm (42.8 L) coarse sand tailings (CST) topped with 10 cm (42.8 L) peat–mineral mix (PMM). The CST and PMM represent locally abundant CWTS substrates and were collected from a mine located in the Alberta oil sandss. Either LPW or OSPW from the same mine was used to fill each mesocosm to 25 cm (139 L) above the substrate surface. The LPW was created to represent a similar chemical composition to OSPW excluding NAs to isolate the potential leaching of NAFCs from the substrate. The LPW was created using reverse osmosis water (RO) water containing 100 mg/L of calcium carbonate, 350 mg/L of sodium sulfate, and 100 mg/L of sodium chloride. The mesocosms were operated as surface flow closed loop systems, where the OSPW or LPW was pumped from a 159 L reservoir tank into the mesocosm. The pumps were set to cycle 28.8 L/day by pumping intermittently every 10 min, with a flow rate of approximately 20 mL/min. This flow rate sets each cycle to be 4 days (i.e., the amount of time required to exchange the entire volume of free water in the mesocosm with the reservoir) or a hydraulic retention time of 4.8 days; this experiment ran for 84 days or 21 cycles.
The C. aquatilis used in this study were grown from seeds collected from natural stands in seed zone DM1.3 (Dry Mixedwood). C. aquatilis seedlings were grown in a peat plug for 3 months in standard styroblock containers (size 512A). Seedlings were fertilized three times a week with Miracle Grow All Purpose water-soluble plant food (N-P-K ratio of 24–8–16). At the time of planting in the mesocosms, C. aquatilis plants were between 63 to 110 cm tall (mean = 83 cm). To acclimate C. aquatilis in the mesocosms prior to adding OSPW, RO water was used during the acclimation period (32 days) where the water level was slowly raised to achieve the final depth of 25 cm. The mesocosms were then drained and filled with 50% OSPW or LPW and 50% RO water for 7 days to acclimate the plants to OSPW, after which the mesocosms were drained and 100% OSPW or LPW was added to the desired depth of 25 cm. Throughout the experiment, the greenhouse temperature was maintained at approximately 20ºC with a 16-h daily photoperiod supplemented using LED grow lights. To account for evapotranspiration from the C. aquatilis, RO water was added to the reservoir tanks to maintain a consistent volume of water throughout the experiment.
General chemistry parameters for the substrate, OSPW, and LPW were analyzed at the beginning and the conclusion of the experiment. Substrate and OSPW samples were sent to Element Materials Technology (Edmonton, AB, Canada). The initial LPW samples were sent to the analytical lab at the Northern Forestry Centre (Edmonton, AB, Canada). Analysis for the substrates included pH, electrical conductivity (EC), sodium adsorption ratio (SAR), major nutrients, total petroleum hydrocarbons, and metal concentrations. Characterization of pH, EC, SAR, and major nutrients was determined using the saturated paste method, and metal concentration was completed using an ICP method with boron measured using hot water extraction. The analysis of OSPW and LPW included pH, EC, hardness, total dissolved solids, metals, major anions, and total alkalinity.
Various water, substrate, and plant parameters were measured at the end of every 4-day cycle. A YSI® Professional Plus Multiparameter instrument was used to measure pH, dissolved oxygen, salinity, oxidation reduction potential (ORP), and conductivity of the water. Vegetation assessments included assessments of plant survival, vegetation cover, and height.
To determine the major ion concentrations for plant tissue, above-ground biomass for each mesocosm was collected and sent to Element Materials Technology for analysis (Edmonton, AB, Canada).
Sample preparation and high-resolution MS analysis of NAFCs
Water samples were extracted using an ENV + solid-phase extraction (SPE) method, as has been previously described by Headley et al. (2002). SPE cartridges were rinsed with 6 mL of Milli-Q water, 6 mL of LC–MS grade methanol (Fisher Scientific, Hampton NH, USA), and conditioned with a further 6 mL of Milli-Q water prior to sampling. Sample aliquots were measured to 100 mL and acidified to pH < 2 with formic acid. Acidified samples were drawn through prepared cartridges at 1–2 mL/min under vacuum conditions, rinsed with 6 mL of Milli-Q water to desalt, and then dried under gentle vacuum. Sample extracts were eluted with 6 mL of LC–MS grade methanol then evaporated at 40 °C under a gentle flow of 5.0-grade ultrahigh purity N2 (Linde Canada, Saskatoon, SK). Dried sample eluents were reconstituted into 1 mL of 50:50 ACN:H2O with 0.1% NH4OH, then transferred to clean and labeled 2.0 mL LC–MS vials. Sample extracts were analyzed via loop injection to an LTQ Orbitrap Velos Elite™ mass spectrometer (Thermo Fisher Scientific, Waltham, MA) operating at 240,000 resolution (measured at 400 m/z) in negative ion electrospray ionization mode, as was previously described in the literature by Headley et al. (2011a). Concentrations of NAFCs were determined using a 5-point external standard calibration of Athabasca oil sands OSPW-derived NAFCs at known concentrations as described elsewhere (Ahad et al., 2020).
Carboxyl group targeted carbon isotope analysis
Non-bitumen-derived organic matter such as fatty acids and humic material extracted with NAFCs can confound interpretations of naphthenic acid distributions in environmental samples (Ahad et al., 2013; Ajaero et al., 2017). Carbon isotope ratios of the CO2 generated by the pyrolytic decarboxylation (δ13Cpyr) of NAFCs (also referred to as acid-extractable organics) were thus carried out to quantify the proportion of OSPW contributing to NAFC concentrations at three sampling points over the course of the experiment in the planted and unplanted OSPW mesocosms. The δ13Cpyr values were determined by thermal conversion/elemental analysis–isotope ratio mass spectrometry (TC/EA-IRMS) at the Delta-Lab of the Geological Survey of Canada (Québec, QC, Canada) using a Delta Plus XL isotope ratio mass spectrometer (Thermo Fisher Scientific, Waltham, MA) following a protocol adapted from Ahad et al. (2012). Around 1 L of water was collected on days 28, 41, and 84, acidified to pH 4.5, and extracted using loose Strata-X-A solid-phase extraction (SPE) sorbent (Phenomenex, Torrance, CA). The sorbent was then filtered from the aqueous phase under vacuum and NAFCs eluted with methanol containing 10% formic acid and pure methanol. The extracts were evaporated to dryness under ultrahigh purity N2 and re-dissolved in methanol. Small aliquots were transferred by syringe into a 40-μL rigid silver capsule, dried, and sealed with pliers prior to isotopic analysis. The δ13Cpyr values were determined using isotopically calibrated CO2 gas purchased from Oztech Trading Corp. (Safford, AZ, USA). Based on replicate standard and sample analyses, the uncertainty for δ13Cpyr values was ± 0.5‰.
Data analysis and visualization
High-resolution mass spectrometry data were processed and background-subtracted in XCalibur software (version 2.2) (Thermofisher, 2011) and then imported into Composer64 software (version 1.5.6) (Paulssen & Gieg, 2019) to assign formulae from exact mass using a 3-ppm mass tolerance restricted to those containing H, C, N, O, and S. This non-targeted analysis workflow generates data with a level 4 identification confidence level (Schymanski et al., 2014). All formula data were imported into R Software (version 4.1.0) (Team, 2021) for wrangling and visualization. Plots were generated using ggplot2 (Wickham, 2016) or Microsoft Excel 2016 if the desired plot could not be achieved in R (Microsoft Corporation, 2018).
Prior to principal component analysis (PCA), base–peak normalized spectral abundance data were arranged, centered and Pareto-scaled (Ivosev et al., 2008; van den Berg et al., 2006), and then analyzed using the built-in R function prcomp() (Team, 2021). Where possible, figures were color-coded using colorblind-friendly viridis color palettes (Garnier, 2022).
To complete the analysis on the total NAFC concentration data, the glmmTMB package and the tidyverse packages were installed (Brooks et al., 2017; Wickham et al., 2019) and generalized linear mixed-effects models used to model the effect of treatments over time (intervals at Day 1, Day 40, and Day 83) on total NAFC concentrations (Bolker et al., 2009; Zuur et al., 2010). A generalized linear model was used to model the effect of the various soil and water parameters. Initial LPW parameters were not included in analysis computed in R since the parameters were collected from a different lab. The random effect that was used in the model was “mesocosm” to account for non-independent measures within each mesocosm. The DHARMa package was installed and used to complete model diagnostics (Harting, 2021), and the model with the best fit was used for visualization. A Gaussian identity link function was used for each of the following: total NAFC concentrations, CST (pH, arsenic, boron, chloride cobalt, lead, potassium, and nickel), and PMM (cadmium, chromium, cobalt, lead, nickel, vanadium, and zinc). Gamma log link function was used for each of the following: on all water parameters, CST (SAR, EC, calcium, sulfur, sodium, magnesium, vanadium, and zinc), and PMM (EC, pH, arsenic, barium, beryllium, boron, calcium, chloride, copper, potassium, magnesium, sodium, and sulfur). To test the fixed effects in models for their significance via the Anova() function of the car package (Fox & Weisberg, 2018), a Wald chi-square test was conducted on each model. Based on the fitted model, the estimated marginal means were calculated using the emmeans package (Lenth, 2018). The P-values for multiple-mean comparisons were adjusted using the Tukey method. Plant growth and plant health over time are presented descriptively to provide a general idea but were not analyzed statistically due to the low number of replicated samples.
Results and discussion
Substrate quality and characteristics
CST is a unique substrate that varies between operators due to the variability in ore type (Sarkar & Sadrekarimi, 2022). The CST used in this study was dominated by coarse sand (98% sand ± 0 (2 mm to 50 µm), 1.5% silts ± 0.6 (2–50 µm), 0.5% clay ± 0.6 (< 2 µm)). Trace metals, petroleum hydrocarbons (C10–C50), EC, and SAR in CST did not exceed any of the provincial soil guidelines (Table 1) (Government of Alberta, 2022).
The PMM used in this study had a lower SAR, EC, and cation exchange capacity than the general range of PMM used for reclamation in this region (MacKenzie & Quideau, 2012; Pinno et al., 2012; Schott et al., 2016), and the pH and trace metal concentrations were within the general acceptable range, meeting local guidelines (Table 2) (Government of Alberta, 2022; MacKenzie & Quideau, 2012; Pinno et al., 2012). However, B increased substantially from the initial baseline concentration to the final concentration in the OSPW treatments, and the final concentration of B in PMM exceeded the provincial limit (Table 2) (Government of Alberta, 2022). It is hypothesized that OSPW is the likely source of boron in this experiment (Table 3). Boron adsorption in organic humic substrates is four times higher than clays or other substrates with little organic matter (Lehto, 1995; Parks & White, 1952). Therefore, it is reasonable to expect more B accumulated in the PMM compared to CST (Tables 1 and 2).
Water quality and characteristics
Dissolved oxygen concentrations remained above 5 mg/L throughout the experiment (Table 3) and generally varied with water temperature. During the experiment, ORP were measured between 10–20 cm depths, the values fluctuated between 60 to 200 mV across all treatments and remained positive during the entire study period (Table 3). The pH in the OSPW–C. aquatilis and LPW–unplanted treatments decreased over time compared to OSPW–unplanted treatment (Table 3), where it increased slightly. The LPW, which started with an alkaline pH due to the presence of calcium carbonate, reached equilibrium with atmospheric CO2 over time, leading to the stabilization of pH to around 7.2 (Table 3). The EC decreased across all treatment types (Table 3). The final general characteristics of the OSPW are similar to the findings of other studies (Cancelli & Gobas, 2020; McQueen et al., 2017a, b; Simair et al., 2021).
The presence of boron in the untreated OSPW (1.2 mg/L; Table 3), though only marginally below the provincial limit (1.5 mg/L)(Government of Alberta, 2018), showed a decrease after 84 days to 0.5 and 0.4 mg/L in the unplanted and the planted treatments, respectively.
Naphthenic acid fraction compound dynamics
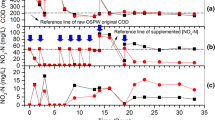
Both quantitative and qualitative data describing NAFCs in the aqueous phase were collected throughout the experiment. Concentrations of NAFCs were dynamic, especially in OSPW-containing mesocosms. This is evident in the mesocosms with OSPW gradually reaching a maximum concentration after approximately 12 days, with gradually decreasing NAFC concentrations thereafter (Fig. 1). The concentrations of NAFCs were likely impacted by diffusion and adsorption during the first 12 days, dissipating afterwards in the remaining 72 days. The LPW–unplanted treatment had relatively little to no NAFCs with no significant change over time (P > 0.857) (0.6 mg/L initial to 0.6 mg/L final), suggesting minimal NAFC desorption from CST or PMM. After 84 days, concentrations of NAFCs in the planted mesocosms significantly decreased (P < 0.015) over time (72.1 mg/L initial to 17.1 mg/L final), whereas the unplanted treatments saw no significant decrease (P > 0.851) in total NAFCs over time (64.5 mg/L initial to 59.0 mg/L final) (Fig. 1). Between the OSPW–unplanted and OSPW–C. aquatilis treatment, there are large variations from the initial 4 mesocosm measurements. The initial (day 1) variation is likely because the OSPW has not had time to equilibrate within each mesocosm. There was a significant difference between treatments (P < 0.001) and a significant interaction effect from treatment and time (P < 0.001); however, timing of sample collection was not significant (P = 0.828) (Fig. 1). Further research into the influence of plants in conjunction with microbial community in the mesocosm will help identify the mechanism for the decrease of NAFCs.
The δ13Cpyr values in the unplanted mesocosms ranged from − 22.5 to − 19.5‰ and from − 21.8 to − 20.9‰ in the planted mesocosms (Table 4). Previous work has demonstrated a significantly more positive δ13Cpyr value for OSPW (− 20.7 ± 0.6‰) compared to non-bitumen, plant-derived NAFCs (− 27.9‰) (Ahad et al., 2013, 2020). The relatively narrow range of values calculated in both the unplanted and planted mesocosms thus indicates a predominantly OSPW-derived origin for NAFCs, with little contribution from non-bitumen-derived organics. Using a two end-member isotopic mass balance incorporating δ13Cpyr values for OSPW and non-bitumen contaminated background sources, the calculated proportions of OSPW in NAFCs were between 74–100% and 85–97% in the unplanted and planted mesocosms, respectively (Table 4). The results from δ13Cpyr measurements thus verify that the trends in NAFC concentrations shown in Fig. 1 and in the Orbitrap MS data, presented in subsequent figures, can be almost exclusively attributed to changes in OSPW-derived rather than plant-derived NAFCs, particularly during the latter stages of the experiment (i.e., days 41 and 84).
To examine the changes in molecular formulae of NAFCs over the course of the study, data were organized according to unique heteroatoms (i.e., anything other than carbon or hydrogen) and plotted according to percent spectral abundance (Fig. 2). Sample data from the mesocosms with LPW were excluded from Fig. 2 owing to trace concentrations of polar organic compounds.
As compared to initial background conditions (i.e., day -8), the spectral signature of OSPW (i.e., high relative percent abundance of O2–NAFCs) remained strong throughout the 84-day treatment period in treatments with OSPW. Nonetheless, in both treatments, formula characteristics gradually shifted, where O2–NAFCs gradually decreased in relative abundance while O3– and O4–NAFCs gradually increased in relative abundance. The rate of decrease of O2–NAFCs (i.e., classical NAs) is almost negligible, decreasing by ~ 10% spectral intensity over the span of 84 days (Fig. 2). In a comparable study, the half-lives of selected NAs were estimated at 12–23 days (Ajaero et al., 2018). However, there are important differences between these two studies; Ajaero et al. (2018) used wetland mesocosms with upward vertical flow of OSPW through the entire growth medium, whereas the mesocosms in the present study operated with horizontal surface flow. Observable changes in surface water may be obfuscated by gradual intermixing and/or equilibrium with the OSPW in the pore water and the OSPW saturated porous media. The study by Ajaero et al. (2018) also used inorganic growth media (e.g., sand or gravel), making OSPW-derived organics the only available source of carbon, whereas the substrate used here included substantial organic-rich PMM, which may provide a favorable source of carbon for microbes. Further, Ajaero et al. (2018) used OSPW supplied by a different operator; accordingly, the typology of OSPW from their study and ours is not chemically identical (Frank et al., 2016), so it should not be expected to biodegrade identically.
Although the bulk percent abundance of heteroatom classes remained relatively static, the spectra of certain mixture components changed over time. A gradual shift in mixture composition is supported by a principal component analysis (PCA) of all OSPW-containing mesocosm samples, shown in Fig. 3. Although sample data resolves incompletely during PCA, groupings of sample data from this experiment are best explained by sampling date. In contrast, there were no compelling differences between planted and unplanted systems when examined with PCA. The similarity between planted and unplanted systems likely could be th effect of adsorption to PMM. Further research into this hypothesis is currently underway.
To examine the connection between sampling dates with the presence, emergence, or absence of particular formulae over time, the Spearman rank correlation between sampling dates and base–peak normalized relative formula abundances were calculated. The Spearman rank correlation was used to color-code a Kendrick mass plot and carbon number (#C) versus double bonding equivalents (DBE) plots in Fig. 4. High carbon number O2–NAFCs tend to have a low-to-negative correlation with sampling date, suggesting that many of these formulae, especially those with high molecular weight (i.e., > 225 m/z) and saturated formulae (i.e., KMD = 0.05–0.10), will diminish in relative spectral abundance over time. The two formulae with the most negative correlations from Fig. 4a are found in the O3S panel of Fig. 4b, corresponding to C14H24O3S and C17H28O3S. On the other hand, many O3–NAFCs have a considerable positive correlation with sampling date, suggesting that most of these formulae consistently increased in spectral relative abundance over time. The correlations of O4–NAFCs are moderately positive-to-neutral, suggesting that some of these formulae increased, while others persisted with unchanged abundance over the treatment period.
Plots summarizing spearman rank correlations of individual formula abundances using a Kendrick mass plot of all formulae detected across samples from OSPW-containing mesocosms, where this figure has been decomposed into major oxygen-containing formula heteroatom classes (b) that constitute approximately 95% of spectral intensity by carbon number and double bonding equivalents for clearer interpretation of trends. In both cases, scatterplot point transparency has been scaled by a factor of rho2 to highlight the strongest correlations
Plant response
Throughout the experiment, OSPW–C. aquatilis had a 98% survival rate. Furthermore, all C. aquatilis individuals, apart from one, remained at 90–99% alive tissue through the experiment. However, all individuals in the OSPW–C. aquatilis presented some symptoms of chlorosis, necrosis, and/or mottling by the end of the experiment. Some individuals also presented signs of deformed and crinkled leaves. By day 40, C. aquatilis likely reached maturity with a growth threshold of around 150–154 cm in height (Fig. 5). Depending on the site conditions, the average height for C. aquatilis in Alberta is between 20 and 155 cm (Hauser, 2006; Johnson et al., 1995; Tannas, 2003; Vitt et al., 2020).
A majority of the ion concentrations were not notably elevated compared to other studies (Table 5). Although the mean manganese concentration in Table 5 is several orders of magnitude higher than that of other ions, the manganese concentrations in tissue generally vary between plant species (30–500 µg/g), making the results reasonable (Li et al., 2019; Millaleo et al., 2005). However, studies have shown toxicity effects in concentrations as low as 176 µg/g in Juncus effusus L. (soft rush) (Najeeb et al., 2009) and 150 µg/g dry weight in some crop species (Li et al., 2019; Millaleo et al., 2005). C. aquatilis in the mesocosm study started to show signs of toxicity expressed through chlorosis, necrosis, crinkling of the leaves, and mottling, which indicate reduced overall health over this 84-day study. It is unclear whether the decline in plant health was attributed to NAs in the OSPW or other ions in OSPW. Although commercial NAs are found to be more toxic than NAs derived from OSPW, Armstrong et al. (2009) found that systems with 30–60 mg/L of commercial NAs significantly reduced wetland plant growth. In the present study, the OSPW–C. aquatilis treatment had concentrations of 10.2–72 mg/L of NAFCs, of which 85–97% originated from OSPW. Further research needs to look at the concentration threshold for NAFCs in OSPW where toxicity effects are evident and the process of translocating NAFCS from the roots to the shoot tissues through the vascular system and how that relates to the concentration of NAFCs in plant tissues (young leaves, lateral shoots and roots, and root tips). Alberts et al. (2021) found that various NA groups are likely being partially bio-transformed when entering the central metabolism where the carbon from these compounds is assimilated into other macromolecules (e.g., lipids, starch, and cellulose). Although plant mortality and reduced growth in C. aquatilis were not observed here, further research is needed for improved analysis of NAs within plant tissue to more effectively evaluate potential toxicity effects from NAFCs (Alberts et al., 2021; Armstrong et al., 2009).
Conclusions
The present study reinforces the weight of evidence that CWTSs are a potential treatment method for OSPW as it is capable of attenuating NAFCs. Treatments with or without plants attenuated NAFCs in the water column according to similar distributions of molecular characteristics, though planted treatments seemed to outperform the unplanted treatments. Although results observed in this study showed NAFC attenuation in the water column to a lesser degree than previous work (Ajaero et al., 2018; Simair et al., 2021), important differences between the experiments likely contribute to the divergence in outcomes. First, previous studies used wetland mesocosms grown in mineral substrates (e.g., sand and/or gravel), with systems configured for vertical flow, and some systems included active aeration as well. These differences in study design likely impacted the distribution and/or degradation of NAFCs in the water column, namely, the adsorption and, by association, the temporary removal of NAFCs from the water phase to the organic substrate phase. This temporary removal conversely limited the availability of labile carbon in systems (e.g., system with no organic substrates) which could facilitate the degradation and assimilation of NAFCs for continued microbial growth. In addition, the vertical flow of water may enhance mixing across the water column, and increased aeration can maximize the potential for aerobic metabolism of NAFCs. In contrast, the mesocosm systems in the present study had surface flow and recirculation of OSPW over a porous mineral and organic medium, which in comparison could potentially adsorb considerable amounts of NAFCs temporarily. Slower diffusion and partitioning effects can obfuscate the rate at which transformation of NAFC species might occur, as noted in this study, where the period in which NAFCs changed the most rapidly occurred in the last 28 days of this study. This experiment also used OSPW from a single operator; it thus remains to be seen whether other OSPW types would undergo similar patterns of attenuation, further investigation into this is recommended. Past work has shown that gradual maturing and ecological succession in reclaimed wetlands can be associated with amelioration of toxicity therein (Armstrong, 2009, Cancelli & Gobas, 2020, Hendrikse et al., 2018; McQueen et al., 2017a, b. The effect of wetland age and related community maturity is therefore likely an important parameter that must be further evaluated to better understand relationships between ecosystem succession and weather of NAFCs.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- OSPW:
-
oil sandss process-affected water
- LPW:
-
Lab process water
- CST:
-
Coarse sand tailings
- PMM:
-
Peat mineral mix
- NAs:
-
Naphthenic acids
- NAFCs:
-
Naphthenic acid fractional compounds
- CWTS:
-
Constructed wetland treatment system
References
Abdalrhman, A. S., & El-din, M. G. (2020). Degradation of organics in real oil sandss process water by electro-oxidation using graphite and dimensionally stable anodes. Chemical Engineering Journal, 38, 124406. https://doi.org/10.1016/j.cej.2020.124406
AER (Alberta Energy Reguator). (2017). Directive 085. November 1–13.
Ahad, J. M. E., Pakdel, H., Gammon, P. R., Mayer, B., Savard, M. M., Peru, K. M., & Headley, J. V. (2020). Distinguishing natural from anthropogenic sources of acid extractable organics in groundwater near oil sandss tailings ponds. Environmental Science and Technology, 54(5), 2790–2799. https://doi.org/10.1021/acs.est.9b06875
Ahad, J. M. E., Pakdel, H., Savard, M. M., Calderhead, A. I., Gammon, P. R., Rivera, A., Peru, K. M., & Headley, J. V. (2013). Characterization and quantification of mining-related “naphthenic acids” in groundwater near a major oil sandss tailings pond. Environmental Science and Technology, 47(10), 5023–5030. https://doi.org/10.1021/es3051313
Ahad, J. M. E., Pakdel, H., Savard, M. M., Simard, M. C., & Smirnoff, A. (2012). Extraction, separation, and intramolecular carbon isotope characterization of Athabasca oil sandss acids in environmental samples. Analytical Chemistry, 84(23), 10419–10425. https://doi.org/10.1021/ac302680y
Ajaero, C., McMartin, D. W., Peru, K. M., Bailey, J., Haakensen, M., Friesen, V., Martz, R., Hughes, S. A., Brown, C., Chen, H., McKenna, A. M., Corilo, Y. E., & Headley, J. V. (2017). Fourier transform ion cyclotron resonance mass spectrometry characterization of Athabasca oil sands process-affected waters incubated in the presence of wetland plants. Energy and Fuels, 31(2), 1731–1740. https://doi.org/10.1021/acs.energyfuels.6b02643
Ajaero, C., Peru, K. M., Simair, M., Friesen, V., O’Sullivan, G., Hughes, S. A., McMartin, D. W., & Headley, J. V. (2018). Fate and behavior of oil sandss naphthenic acids in a pilot-scale treatment wetland as characterized by negative-ion electrospray ionization Orbitrap mass spectrometry. Science of the Total Environment, 631–632, 829–839. https://doi.org/10.1016/j.scitotenv.2018.03.079
Alberts, M. E., Wong, J., Hindle, R., Degenhardt, D., Krygier, R., Turner, R. J., & Muench, D. G. (2021). Detection of naphthenic acid uptake into root and shoot tissues indicates a direct role for plants in the remediation of oil sandss process-affected water. Science of the Total Environment, 795, 148857. https://doi.org/10.1016/j.scitotenv.2021.148857
Alharbi, H. A., Wiseman, S. B., & Giesy, J. P. (2021). Effects of oil sandss process-affected water on measured concentrations of polycyclic aromatic hydrocarbons. ACS ES&T Water, 1(3), 698–706. https://doi.org/10.1021/acsestwater.0c00183
Allen, E. W. (2008). Process water treatment in Canada’s oil sandss industry: II. A review of emerging technologies. Journal of Environmental Engineering and Science, 7(5), 499–524. https://doi.org/10.1139/S08-020
Alpatova, A., Kim, E. S., Dong, S., Sun, N., Chelme-Ayala, P., & Gamal El-Din, M. (2014). Treatment of oil sandss process-affected water with ceramic ultrafiltration membrane: Effects of operating conditions on membrane performance. Separation and Purification Technology, 122, 170–182. https://doi.org/10.1016/j.seppur.2013.11.005
Anderson, J., Wiseman, S. B., Moustafa, A., Gamal El-Din, M., Liber, K., & Giesy, J. P. (2012). Effects of exposure to oil sandss process-affected water from experimental reclamation ponds on Chironomus dilutus. Water Research, 46(6), 1662–1672. https://doi.org/10.1016/j.watres.2011.12.007
Armstrong, S. A., Headley, J. V., Peru, K. M., & Germida, J. J. (2009). Differences in phytotoxicity and dissipation between ionized and nonionized oil sandss naphthenic acids in wetland plants. Environmental Toxicology and Chemistry, 28(10), 2167–2174. https://doi.org/10.1897/09-059.1
Bartlett, A. J., Frank, R. A., Gillis, P. L., Parrott, J. L., Marentette, J. R., Brown, L. R., Hooey, T., Vanderveen, R., McInnis, R., Brunswick, P., Shang, D., Headley, J. V., Peru, K. M., & Hewitt, L. M. (2017). Toxicity of naphthenic acids to invertebrates: Extracts from oil sandss process-affected water versus commercial mixtures. Environmental Pollution, 227, 271–279.
Biryukova, O., Fedorak, P., & Quideau, S. (2007). Biodegradation of naphthenic acids by rhizosphere microorganisms. Chemosphere, 67(10), 2058–2064. https://doi.org/10.1016/j.chemosphere.2006.11.063
Bolker, B. M., Brooks, M. E., Clark, C. J., Geange, S. W., Poulsen, J. R., Stevens, M. H. H., & White, J. S. S. (2009). Generalized linear mixed models: A practical guide for ecology and evolution. Trends in Ecology and Evolution, 24(3), 127–135. https://doi.org/10.1016/j.tree.2008.10.008
Boyle, T. P., & Fairchild, J. F. (1997). The role of mesocosm studies in ecological risk analysis. Ecological Applications, 7(4), 1099–1102. https://doi.org/10.1890/1051-0761(1997)007[1099:TROMSI]2.0.CO;2
Brooks, M. E., Kristensen, K., van Benthem, K. J., Magnusson, A., Berg, C. W., Nielsen, A., Skaug, H. J., Mächler, M., & Bolker, B. M. (2017). GlmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R Journal, 9(2), 378–400. https://doi.org/10.32614/rj-2017-066
Brown, L. D., & Ulrich, A. C. (2015). oil sandss naphthenic acids: A review of properties, measurement, and treatment. Chemosphere, 127, 276–290. https://doi.org/10.1016/j.chemosphere.2015.02.003
Canadian Association of Petroleum Producers. (2022). What are the oil sandss?. Retrieved June 20, 2021, https://www.capp.ca/oil/what-are-the-oil-sands/
Cancelli, A. M., & Gobas, F. A. P. C. (2020). Treatment of polycyclic aromatic hydrocarbons in oil sandss process-affected water with a surface flow treatment wetland. Environments - MDPI, 7(9), 1–16. https://doi.org/10.3390/environments7090064
Cancelli, A. M., & Gobas, F. A. P. C. (2022). Treatment of naphthenic acids in oil sandss process-affected waters with a surface flow treatment wetland: Mass removal, half-life, and toxicity-reduction. Environmental Research, 213, 113755. https://doi.org/10.1016/j.envres.2022.113755
Caners, R., & Lieffers, V. (2014). Divergent pathways of successional recovery for in situ oil sandss exploration drilling pads on wooded moderate-rich fens in Alberta, Canada. Restoration Ecology, 22(5), 657–667. https://doi.org/10.1111/rec.12123
Cappello, S., & Yakimov, M. M. (2010). Handbook of hydrocarbon and lipid microbiology. Handbook of Hydrocarbon and Lipid Microbiology, January. https://doi.org/10.1007/978-3-540-77587-4
Crowe, A. U., Plant, A. L., & Kermode, A. R. (2002). Effects of an industrial effluent on plant colonization and on the germination and post-germinative growth of seeds of terrestrial and aquatic plant species. Environmental Pollution, 117(1), 179–189. https://doi.org/10.1016/S0269-7491(01)00131-2
Del Rio, L. F., Hadwin, A. K. M., Pinto, L. J., MacKinnon, M. D., & Moore, M. M. (2006). Degradation of naphthenic acids by sediment micro-organisms. Journal of Applied Microbiology, 101(5), 1049–1061. https://doi.org/10.1111/j.1365-2672.2006.03005.x
Duncan, K. D., Letourneau, D. R., Vandergrift, G. W., Jobst, K., Reiner, E., Gill, C. G., & Krogh, E. T. (2016). A semi-quantitative approach for the rapid screening and mass profiling of naphthenic acids directly in contaminated aqueous samples: Rapid screening of naphthenic acids. Journal of Mass Spectrometry, 51(1), 44–52. https://doi.org/10.1002/jms.3721
Fox, J., & Weisberg, S. (2018). Nonlinear regression, nonlinear least squares, and nonlinear mixed models in R. An R Companion to Applied Regression, 31.
Frank, R. A., Milestone, C. B., Rowland, S. J., Headley, J. V., Kavanagh, R. J., Lengger, S. K., Scarlett, A. G., West, C. E., Peru, K. M., & Hewitt, L. M. (2016). Assessing spatial and temporal variability of acid-extractable organics in oil sandss process-affected waters. Chemosphere, 160, 303–313. https://doi.org/10.1016/j.chemosphere.2016.06.093
Garnier, S. (2022). Package ‘ viridis ’ (pp. 1–7). Retrieved Sep 20, 2022, https://cran.r-project.org/package=viridisLite
Government of Alberta. (2018). Environmental quality guidelines for Alberta surface waters (pp. 1–53). Water Policy Branch, Alberta Environment and Parks.
Government of Alberta. (2022). Alberta Tier 1 soil and groundwater remediation guidelines. In 2021 Cropping Alternatives (Issue February).
Government of Canada. (n.d.). What are the oil sandss? Retrieved June 11, 2021, from https://www.nrcan.gc.ca/our-natural-resources/energy-sources-distribution/fossil-fuels/crude-oil/what-are-oil-sands/18089
Grewer, D. M., Young, R. F., Whittal, R. M., & Fedorak, P. M. (2010). Science of the total environment naphthenic acids and other acid-extractables in water samples from Alberta: What is being measured ? The Science of the Total Environment, 408(23), 5997–6010. https://doi.org/10.1016/j.scitotenv.2010.08.013
Hagen, M. O., Katzenback, B. A., Shahinoor Islam, M. D., El-Din, M. G., & Belosevic, M. (2014). The analysis of goldfish (Carassius auratus L.) innate immune responses after acute and subchronic exposures to oil sandss process-affected water. Toxicological Sciences, 138(1), 59–68. https://doi.org/10.1093/toxsci/kft272
Han, X., MacKinnon, M. D., & Martin, J. W. (2009). Estimating the in situ biodegradation of naphthenic acids in oil sandss process waters by HPLC/HRMS. Chemosphere, 76(1), 63–70. https://doi.org/10.1016/j.chemosphere.2009.02.026
Han, X., Scott, A. C., Fedorak, P. M., & Bataineh, M. (2008). Influence of molecular structure on the biodegradability of naphthenic acids. Environmental Science & Technology, 42(4), 1290–1295.
Harting, F. (2021). DHARMa: Residual diagnostics for hierarchical (multi-level/mixed) regression models. Retrieved Feb 2, 2021, https://cran.r-project.org/web/packages/DHARMa/index.html
Hauser, S. (2006). Carex aquatilis. In: Fire Effects Information System, [Online]. Retrieved Feb 2, 2021, https://www.fs.usda.gov/database/feis/plants/graminoid/caraqu/all.html#96
Headley, J. V., Barrow, M. P., Peru, K., & Derrick, P. J. (2011a). Salting-out effects on the characterization of naphthenic acids from Athabasca oil sandss using electrospray ionization. Journal of Environmental Science and Health - Part A Toxic/hazardous Substances and Environmental Engineering, 46(8), 844–854. https://doi.org/10.1080/10934529.2011.579857
Headley, J. V., Peru, K. M., Janfada, A., Fahlman, B., Gu, C., & Hassan, S. (2011b). Characterization of oil sandss acids in plant tissue using Orbitrap ultra-high resolution mass spectrometry with electrospray ionization: Letter to the editor. Rapid Communications in Mass Spectrometry, 25(3), 459–462. https://doi.org/10.1002/rcm.4877
Headley, J. V., Peru, K. M., McMartin, D. W., & Winkler, M. (2002). Determination of dissolved naphthenic acids in natural waters by using negative-ion electrospray mass spectrometry. Journal of AOAC International, 85(1), 182–187. https://doi.org/10.1093/jaoac/85.1.182
Headley, J. V., Peru, K. M., Mohamed, M. H., Frank, R. A., Martin, J. W., Hazewinkel, R. R. O., Humphries, D., Gurprasad, N. P., Hewitt, L. M., Muir, D. C. G., Lindeman, D., Strub, R., Young, R. F., Grewer, D. M., Whittal, R. M., Fedorak, P. M., Birkholz, D. A., Hindle, R., Reisdorph, R., & Wrona, F. J. (2013). Chemical fingerprinting of naphthenic acids and oil sandss process waters-A review of analytical methods for environmental samples. Journal of Environmental Science and Health - Part A Toxic/hazardous Substances and Environmental Engineering, 48(10), 1145–1163. https://doi.org/10.1080/10934529.2013.776332
Hendrikse, M., Gaspari, D. P., McQueen, A. D., Kinley, C. M., Calomeni, A. J., Geer, T. D., Simair, M. C., Peru, K. M., Headley, J. V., Rodgers, J. H., & Castle, J. W. (2018). Treatment of oil sandss process-affected waters using a pilot-scale hybrid constructed wetland. Ecological Engineering, 115, 45–57. https://doi.org/10.1016/j.ecoleng.2018.02.009
Hersikorn, B. D., & Smits, J. E. G. (2011). Compromised metamorphosis and thyroid hormone changes in wood frogs (Lithobates sylvaticus) raised on reclaimed wetlands on the Athabasca oil sandss. Environmental Pollution, 159(2), 596–601. https://doi.org/10.1016/j.envpol.2010.10.005
Holowenko, F. M., MacKinnon, M. D., & Fedorak, P. M. (2002). Characterization of naphthenic acids in oil sandss wastewaters by gas chromatography-mass spectrometry. Water Research, 36(11), 2843–2855. https://doi.org/10.1016/S0043-1354(01)00492-4
Hughes, S. A., Mahaffey, A., Shore, B., Baker, J., Kilgour, B., Brown, C., Peru, K. M., Headley, J. V., & Bailey, H. C. (2017). Using ultrahigh-resolution mass spectrometry and toxicity identification techniques to characterize the toxicity of oil sandss process-affected water: The case for classical naphthenic acids. Environmental Toxicology and Chemistry, 36(11), 3148–3157. https://doi.org/10.1002/etc.3892
Ivosev, G., Burton, L., & Bonner, R. (2008). Dimensionality reduction and visualization in principal component analysis. Analytical Chemistry, 80(13), 4933–4944. https://doi.org/10.1021/ac800110w
Janfada, A., Headley, J. V., Peru, K. M., & Barbour, S. L. (2006). A laboratory evaluation of the sorption of oil sandss naphthenic acids on organic rich soils. Journal of Environmental Science and Health, Part A, 41(6), 985–997. https://doi.org/10.1080/10934520600620105
Johnson, D., Kershaw, L., MacKinnon, A., & Pojar, J. (1995). Plants of the western forest boreal and aspen parkland. Lone Pine Publishing.
Kavanagh, R. J., Frank, R. A., Burnison, B. K., Young, R. F., Fedorak, P. M., Solomon, K. R., & Van Der Kraak, G. (2012). Fathead minnow (Pimephales promelas) reproduction is impaired when exposed to a naphthenic acid extract. Aquatic Toxicology, 116–117, 34–42. https://doi.org/10.1016/j.aquatox.2012.03.002
Kavanagh, R. J., Frank, R. A., Solomon, K. R., & Van Der Kraak, G. (2013). Reproductive and health assessment of fathead minnows (Pimephales promelas) inhabiting a pond containing oil sandss process-affected water. Aquatic Toxicology, 130–131, 201–209. https://doi.org/10.1016/j.aquatox.2013.01.007
Kim, E. S., Liu, Y., & Gamal El-Din, M. (2012). Evaluation of membrane fouling for in-line filtration of oil sandss process-affected water: The effects of pretreatment conditions. Environmental Science and Technology, 46(5), 2877–2884. https://doi.org/10.1021/es203813s
Lehto, T. (1995). Boron retention in limed forest mor. Forest Ecology and Management, 78(1–3), 11–20. https://doi.org/10.1016/0378-1127(95)03599-7
Lenth, R. (2018). Emmeans: Estimated marginal means, aka least-squares means. R Package Version, 1, 1.
Li, J., Jia, Y., Dong, R., Huang, R., Liu, P., Li, X., Wang, Z., Liu, G., & Chen, Z. (2019). Advances in the mechanisms of plant tolerance to manganese toxicity. International Journal of Molecular Sciences, 20(20). https://doi.org/10.3390/ijms20205096
MacKenzie, M. D., & Quideau, S. A. (2012). Laboratory-based nitrogen mineralization and biogeochemistry of two soils used in oil sandss reclamation. Canadian Journal of Soil Science, 92(1), 131–142. https://doi.org/10.4141/CJSS2010-070
McQueen, A. D., Hendrikse, M., Gaspari, D. P., Kinley, C. M., Rodgers, J. H., & Castle, J. W. (2017a). Performance of a hybrid pilot-scale constructed wetland system for treating oil sandss process-affected water from the Athabasca oil sandss. Ecological Engineering, 102, 152–165. https://doi.org/10.1016/j.ecoleng.2017.01.024
McQueen, A. D., Kinley, C. M., Hendrikse, M., Gaspari, D. P., Calomeni, A. J., Iwinski, K. J., Castle, J. W., Haakensen, M. C., Peru, K. M., Headley, J. V., & Rodgers, J. H. (2017b). A risk-based approach for identifying constituents of concern in oil sandss process-affected water from the Athabasca oil sandss region. Chemosphere, 173, 340–350. https://doi.org/10.1016/j.chemosphere.2017.01.072
Melvin, S. D., & Trudeau, V. L. (2012). Toxicity of naphthenic acids to wood frog tadpoles (Lithobates sylvaticus). Journal of Toxicology and Environmental Health - Part A: Current Issues, 75(3), 170–173. https://doi.org/10.1080/15287394.2012.640092
Microsoft Corporation. (2018). Microsoft Excel. Retrieved June 11, 2021, https://office.microsoft.com/excel
Millaleo, R., Ivanov, A. G., Mora, M. L., & Alberdi, M. (2005). Manganese as essential and toxic element for plants: Transport, accumulation and resistance mechanisms.
Morandi, G. D., Wiseman, S. B., Pereira, A., Mankidy, R., Gault, I. G. M., Martin, J. W., & Giesy, J. P. (2015). Effects-directed analysis of dissolved organic compounds in oil sandss process-affected water. Environmental Science and Technology, 49(20), 12395–12404. https://doi.org/10.1021/acs.est.5b02586
Najeeb, U., Xu, L., Ali, S., Jilani, G., Gong, H. J., Shen, W. Q., & Zhou, W. J. (2009). Citric acid enhances the phytoextraction of manganese and plant growth by alleviating the ultrastructural damages in Juncus effusus L. Journal of Hazardous Materials, 170(2–3), 1156–1163. https://doi.org/10.1016/j.jhazmat.2009.05.084
Parks, W. L., & White, J. L. (1952). Boron retention by clay and humus systems saturated with various cations. In Soil Science Society of America journal: Vol. v. 16. Soil Science Society of America.
Paulssen, J. M., & Gieg, L. M. (2019). Biodegradation of 1-adamantanecarboxylic acid by algal-bacterial microbial communities derived from oil sandss tailings ponds. Algal Research, 41, 101528. https://doi.org/10.1016/j.algal.2019.101528
Phillips, L. A., Armstrong, S. A., Headley, J. V., Greer, C. W., & Germida, J. J. (2010). Shifts in root-associated microbial communities of Typha latifolia growing in naphthenic acids and relationship to plant health. International Journal of Phytoremediation, 12(8), 745–760. https://doi.org/10.1080/15226510903535106
Pinno, B. D., Landhäusser, S. M., Derek MacKenzie, M., Quideau, S. A., & Chow, P. S. (2012). Trembling aspen seedling establishment, growth and response to fertilization on contrasting soils used in oil sandss reclamation. Canadian Journal of Soil Science, 92(1), 143–151. https://doi.org/10.4141/CJSS2011-004
Puttaswamy, N., & Liber, K. (2012). Influence of inorganic anions on metals release from oil sandss coke and on toxicity of nickel and vanadium to Ceriodaphnia dubia. Chemosphere, 86(5), 521–529. https://doi.org/10.1016/j.chemosphere.2011.10.018
Quagraine, E. K., Peterson, H. G., & Headley, J. V. (2007). In situ bioremediation of naphthenic acids contaminated tailing pond waters in the Athabasca oil sandss region—Demonstrated field studies and plausible options: A review in situ bioremediation of naphthenic acids contaminated tailing pond waters in the 4529. https://doi.org/10.1081/ESE-200046649
Quinlan, P. J., & Tam, K. C. (2015). Water treatment technologies for the remediation of naphthenic acids in oil sandss process-affected water. Chemical Engineering Journal, 279, 696–714. https://doi.org/10.1016/j.cej.2015.05.062
Ripmeester, M. J., & Duford, D. A. (2019). Method for routine “naphthenic acids fraction compounds” determination in oil sandss process-affected water by liquid-liquid extraction in dichloromethane and Fourier-transform infrared spectroscopy. Chemosphere, 233, 687–696. https://doi.org/10.1016/j.chemosphere.2019.05.222
Rogers, V. V., Liber, K., & MacKinnon, M. D. (2002). Isolation and characterization of naphthenic acids from Athabasca oil sandss tailings pond water. Chemosphere, 48(5), 519–527. https://doi.org/10.1016/S0045-6535(02)00133-9
Sarkar, G., & Sadrekarimi, A. (2022). Undrained shearing behaviour of oil sandss tailings. Soil Dynamics and Earthquake Engineering, 161, 107410. https://doi.org/10.1016/j.soildyn.2022.107410
Schindler, D. W. (1998). Replication versus realism: The need for ecosystem-scale experiments. Ecosystems, 1(4), 323–334. https://doi.org/10.1007/s100219900026
Schott, K. M., Snively, A. E. K., Landhäusser, S. M., & Pinno, B. D. (2016). Nutrient loaded seedlings reduce the need for field fertilization and vegetation management on boreal forest reclamation sites. New Forests, 47(3), 393–410. https://doi.org/10.1007/s11056-015-9522-4
Schymanski, E. L., Jeon, J., Gulde, R., Fenner, K., Ruff, M., Singer, H. P., & Hollender, J. (2014). Identifying small molecules via high resolution mass spectrometry: Communicating confidence. Environmental Science and Technology, 48(4), 2097–2098. https://doi.org/10.1021/es5002105
Simair, M. C., Parrott, J. L., le Roux, M., Gupta, V., Frank, R. A., Peru, K. M., Ajaero, C., McMartin, D. W., & Headley, J. V. (2021). Treatment of oil sandss process affected waters by constructed wetlands: Evaluation of designs and plant types. Science of the Total Environment, 772, 145508. https://doi.org/10.1016/j.scitotenv.2021.145508
Tannas, K. (2003). Common plants of the western rangelands volume 1 grasses and grass-like species. Alberta Agriculture and Rual Development.
Team, R. C. (2021). The R Project for Statistical Computing. Retrieved June 11, 2021, https://www.r-project.org/
Thermofisher. (2011). Xcalibur version 2.2 software (Issue January). Retrieved March 14, 2021, https://thermo-xcalibur.software.informer.com/2.2/
Truu, J., Truu, M., Espenberg, M., Nõlvak, H., & Juhanson, J. (2015). Send Orders for Reprints to reprints@benthamscience.ae Phytoremediation and plant-assisted bioremediation in soil and treatment wetlands: A review. The Open Biotechnology Journal, 9, 85–92.
Truu, M., Juhanson, J., & Truu, J. (2009). Microbial biomass, activity and community composition in constructed wetlands. Science of the Total Environment, 407(13), 3958–3971. https://doi.org/10.1016/j.scitotenv.2008.11.036
van den Berg, R. A., Hoefsloot, H. C. J., Westerhuis, J. A., Smilde, A. K., & van der Werf, M. J. (2006). Centering, scaling, and transformations: Improving the biological information content of metabolomics data. BMC Genomics, 7, 1–15. https://doi.org/10.1186/1471-2164-7-142
Vander Meulen, I. J., Klemish, J. L., Peru, K. M., Da Yong Chen, D., Pyle, G. G., & Headley, J. V. (2021). Molecular profiles of naphthenic acid fraction compounds from mine lease wetlands in the Athabasca oil sandss region. Chemosphere, 129892. https://doi.org/10.1016/j.chemosphere.2021.129892
Vitt, D. H., Glaeser, L. C., House, M., & Kitchen, S. P. (2020). Structural and functional responses of Carex aquatilis to increasing sodium concentrations. Wetlands Ecology and Management, 28(5), 753–763. https://doi.org/10.1007/s11273-020-09746-9
Wang, C., Klamerth, N., Messele, S. A., Singh, A., Belosevic, M., & Gamal El-Din, M. (2016). Comparison of UV/hydrogen peroxide, potassium ferrate(VI), and ozone in oxidizing the organic fraction of oil sandss process-affected water (OSPW). Water Research, 100, 476–485. https://doi.org/10.1016/j.watres.2016.05.037
Wickham, H. (2016). Ggplot2. In Data analysis (pp. 189–201). Springer, Cham. https://doi.org/10.1007/978-3-319-24277-4_9
Wickham, H., Averick, M., Bryan, J., Chang, W., McGowan, L., François, R., Grolemund, G., Hayes, A., Henry, L., Hester, J., Kuhn, M., Pedersen, T., Miller, E., Bache, S., Müller, K., Ooms, J., Robinson, D., Seidel, D., Spinu, V., … & Yutani, H. (2019). Welcome to the Tidyverse. Journal of Open Source Software, 4(43), 1686. https://doi.org/10.21105/joss.01686
Xue, J., Huang, C., Zhang, Y., Liu, Y., & Gamal El-Din, M. (2018). Bioreactors for oil sandss process-affected water (OSPW) treatment: A critical review. Science of the Total Environment, 627, 916–933. https://doi.org/10.1016/j.scitotenv.2018.01.292
Zuur, A. F., Ieno, E. N., & Elphick, C. S. (2010). A protocol for data exploration to avoid common statistical problems. Methods in Ecology and Evolution, 1(1), 3–14. https://doi.org/10.1111/j.2041-210x.2009.00001.x
Acknowledgements
We wish to thank Delta-Lab staff (GSC-Québec) for carrying out isotopic analyses. We would like to thank Imperial Oil for supplying us with the materials and Asfaw Bekele and Carolina Berdugo-Clavijo for providing their comments on the manuscript. We would also like to thank Christine Martineau and Douglas Muench for assisting with experimental design and designing the mesocosms and Angeline Van Dongen and Caren Jones for helping to set up and complete sample collection. This research is part of a Genome Canada Large Scale Applied Research Project (LSARP, grant #18207). We acknowledge that experiments and analyses were carried out on Treaty 6 territory using materials sourced from Treaty 8 territory, and we honor and acknowledge all of the First Nations, Métis, and Inuit peoples who have lived, traveled, and gathered on these lands for thousands of years.
Funding
Open Access funding provided by Natural Resources Canada. This work was funded by the Canadian Forest Service Cumulative Effects funding program. John Headley’s laboratory team acknowledges funding from the Natural Resources Canada through the Energy Innovation Program led by the Office of Energy Research and Development.
Author information
Authors and Affiliations
Contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript. Kaitlyn Trepanier: experiment design, greenhouse experiment, analysis, writing, and editing. Ian J. Vander Meulen: sample processing, data analysis, writing, and editing. Jason M. E. Ahad: data analysis, writing, and editing. Dani Degenhardt: project management, experiment design, funding acquisition, writing, and editing. John V. Headley: experiment design, funding acquisition, and editing.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
No ethics approvals or consent to participate was needed for this study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Trepanier, K.E., Vander Meulen, I.J., Ahad, J.M.E. et al. Evaluating the attenuation of naphthenic acids in constructed wetland mesocosms planted with Carex aquatilis. Environ Monit Assess 195, 1228 (2023). https://doi.org/10.1007/s10661-023-11776-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-023-11776-8