Abstract
We examined 10 subsurface water, 5 benthic water and 19 sediment (02 cm) samples along a 518 km of the middle segment of the Ganga River to assess the possible improvements that resulted from the industrial shutdown during the COVID-19 pandemic. The sites included the main stem river, tributary confluences, and two point sources, one of which releases metal-rich effluents and the other flushes municipal sewage. We found significant declines in the carbon, nutrient and metal concentrations in both the water and sediment. Even the most polluted zones did not show hypoxia (dissolve oxygen; DO < 2.0 mg L−1) that had been observed in the previous year. Despite a significant decline in carbon and nitrogen as substrates, the activities of extracellular enzymes (EEs), such as β-D-glucosidase, FDAase and protease in sediment (0–2 cm depth), increased significantly (p < 0.05) in response to the declining metal concentrations resulting from the industrial shutdown. We found strong negative correlations between EE activity and the concentrations of metal pollutants measured in 2019, but the correlations between these variables appeared poor in 2020 (lockdown period). Also, we found large variances (low stability coefficients) during the period of strong anthropogenic effects (2019). The study indicates that industrial sources are important contributors of metal pollution in the Ganga River and has relevance exploring river ecosystem recovery windows for management decisions.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The novel coronavirus (COVID-19) pandemic emerged as the largest threat to human health during 2020–2021. This virus created a pandemic situation starting on 31st December 2019, wherein the first case was confirmed by the WHO in Wuhan, China, with symptoms of an unexplained low respiratory infection. The director of the WHO declared a pandemic situation on 11th March 2020 because the transmission rate of positive cases increased 13-fold, and approximately 114 countries had shown several thousand positive cases and deaths (World Health Organization, 2020). The transmission of COVID-19 increased very rapidly, and the whole world adopted curfew/lockdown policies by implementing human mobility restrictions. In India, the first case of COVID-19 was reported on 30th January 2020. The lockdown in India occurred from March 23 to April 14, 2020 (Lockdown 1). It was further extended for 19 days, until May 3, 2020 (Lockdown 2), and again until May 17, 2020 (Lockdown 3). During the lockdown period, social, economic, industrial and urban activities were all completely prohibited. As a result, the water of the Ganga River appeared clear, and its turbidity declined relative to its previous status (Gard et al., 2020).
The Ganga River has unique hydrological and morphological features but is under strong human pressure. Approximately 6614 MLD (million litres per day) of wastewater with an organic pollution load of 426 TPD (tonnes per day) is discharged annually into the Ganga River (Central Pollution Control Board, 2016). Total industrial input in the Ganga River contributes 8% flow-wise; however, in terms of the percentage of the organic pollution load, industrial input contributes up to 31% (Central Pollution Control Board, 2016). During the COVID-19 pandemic, industrial activity nearly ceased completely, but sewage release continued, and because there was no agricultural runoff (the lockdown period coincided with the dry season), the industrial shutdown could be considered to be accountable for the improvement in the water quality of the Ganga River.
Three groups of pollutants, carbon, nutrients and heavy metals, have received great attention as the cause of the Ganga water quality (CPCB, 2013). Carbon and nutrients are largely added through sewage and agricultural runoff, whereas metals come primarily from industrial effluents (CPCB, 2013; Yadav & Pandey, 2017). Carbon enhances the turbidity and biological oxygen demand (BOD), and nutrients enhance eutrophy, leading to the progressive accumulation of biomass (Pandey et al., 2014), whereas an excess of heavy metals causes detrimental effects on microbial activity (Jaiswal & Pandey, 2018) and aquatic consumers (Siddiqui & Pandey, 2019). Because rivers impacted by humans simultaneously receive multiple pollutants from a variety of sources, collateral and sequential effects or stressor synergies may constrain the prediction of specific responses resulting from individual stressors (Jaiswal et al., 2021; Tonkin et al., 2019). Thus, parallel enrichment and the collateral effects from these pollutants may hinder predicting the rate of carbon degradation and the magnitude of metal toxicity (Jaiswal & Pandey, 2018, 2019a). Furthermore, carbon-demanding substances and other oxygen-demanding substances (ODSs) lead to a dissolved oxygen deficit (DOD) that causes a series of sequential effects, including positive feedback and the loss of resilience and overall ecosystem performance (Jaiswal et al., 2021). Therefore, lockdown period–driven reductions in one or more pollutants can help to understand and determine how the mechanisms/key processes or amounts of specific pollutants respond to changes to a degree beyond which the key process or ecosystem performance is constrained.
Microbial activities and extracellular enzymes (EEs) are the key regulators of detritus food webs and carbon and nutrient cycling (Sinsabaugh et al., 2009). EE activity is favoured by carbon and nutrient substrates, regulated by the sources of these substrates and environmental conditions (Hill et al., 2010), and constrained by toxicants such as heavy metals (Jaiswal & Pandey, 2018). Thus, EE activity can be used to demarcate the boundaries of microbial responses to source distribution, substrate availability and changes in environmental conditions, including the presence and concentrations of toxicants. Here, we attempted to analyse the impact of the restrictions imposed on human mobility during the lockdown period on the water and sediment quality of the Ganga River. We attempted to explore whether a reduction in the level of metal pollution, as was expected during a lockdown period, could improve microbial/EE activities at the sediment–water interface. Because industrial sources are generally considered major contributors to heavy metal pollution, this study will be highly useful in clarifying the role of industrial activity on the heavy metal pollution and associated shifts in sediment microbial activity and other functional attributes in the Ganga River.
Materials and methods
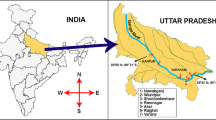
Here, we included a 518-km channel of the Ganga River to collect data on the improvements in water and sediment quality as influenced by the COVID-19 lockdown (2020). To quantify the magnitude of the changes, data comparisons were made with those generated in 2019. The seven study sites selected here were located between Kanpur (26°50′ N; 80°31′ E) and Varanasi (25°32′ N; 83°04′ E), constituting the middle segment of the Ganga River (Fig. 1).
The upstream site, Nawabganj (Site 1; Nwbj), is located upstream of Kanpur city. Jajmau (Site 2; Jjmu), located downstream of Kanpur city, is under strong urban influence. Wazidpur (Site 3; Wpdr), also located downstream of Kanpur city, represents the direct influence of the leather industry. Sampling Site 4, Adalpura (Adpr), situated upstream of Varanasi city, represents rural influence and agricultural land use. Sites Assi (Asdr), Rajghat (Rjhtup) and Varuna (Vrun), Sites 5, 6 and 7, respectively, are under the influence of point sources (Asdr), urban release (Rjhtup) and Varuna tributaries (Vrun). Differences in the sites in terms of urban-industrial flushing were considered to mechanistically link the causal connections with improvements in water/sediment quality resulting from the COVID-19 restrictions.
Experimental protocol
To evaluate the effects on the Ganga River that resulted from the restrictions imposed during the COVID-19 lockdown period, we examined 10 subsurface water quality variables, 5 benthic water quality parameters and 19 sediment (0–2 cm) quality parameters, subject to feasibility of measurement under the imposed restrictions. This study is a continuation of our previous year data collection (2019) and is useful to compare the changes resulting from the imposed restrictions. The river tract considered here represents the most polluted stretch of the Ganga River. The overall observations are the result of three sets of studies conducted at different spatial-scales: the first set represents a main stem investigation considering 7 sampling sites along a 518-km middle part of the main river from Kanpur to Varanasi. In total, 132 sediment samples and 132 water samples were collected from three locations of each site for detailed analysis. The subsurface water samples were collected in triplicate in the third week of June from ~ 25 m of the traverse reach 15 to 25 cm below the water surface. Water samples were also collected from the sediment–water interface. The sediment samples were collected from two spatial locations. At sediment–water interface (SWI), samples were collected from 0 to 2 cm depth, and from riverbed (2–10 cm depth), samples were collected at 1.5–6 m water column depth from 25 m reaches in the main stem of the river. To increase comparability, we also collected freshly deposited sediment (0–2 cm deep) in triplicate from each sampling site. In all cases, samples were collected from three sub sites and mixed to form composite samples. Samples were kept in acid-rinsed polyethylene bags and plastic bottles and brought to the laboratory for the analysis following standard protocols. After collection of the fresh samples, a portion was acidified by adding 1 mL of concentrated nitric acid per 100 mL of sample to avoid microbial consumption of metals. For analysis, the sediment samples were sub-divided into three parts. One part was used for the analysis of carbon, nutrients and heavy metals after air drying for 3–4 days at room temperature. The dried samples were grinded using mortar and pestle and then sieved using 2 mm mesh. The second part was kept at 4 °C for the incubation experiments. The third part of the samples was incubated in desiccators to obtain a moisture level of 80%. These samples were used for measuring microbial biomass/activity and extracellular enzymes. All samples were kept in dark and stored at 4 °C in an ice-box for maintaining the original status.
Methods
Biological oxygen demand (BOD) was quantified as BOD5 (after 5 days of incubation; Eaton et al., 1995). Dissolved oxygen (DO) was fixed at the respective site and analysed volumetrically following azide modification according to Winkler’s method (APHA, 1998). The chemical oxygen demand (COD) was estimated using the potassium dichromate method (APHA, 1998) and the amount of dissolved organic carbon (DOC) was evaluated following the KMnO4 digestion procedure (Michel, 1984). The dissolved oxygen deficit (DOD) was calculated using the formula given by Sánchez et al. (2007). We measured the content of NO3- as per Voghel (1971) and that of NH4+ as per Park et al. (2009). For reactive phosphorus, we used the ammonium molybdate method (Murphy & Riley, 1962). For heavy metals (Cr, Cu, Cd, Zn, Ni and Pb), water samples were digested in a tri-acid mixture (Allen et al., 1986) and quantified by atomic absorption spectroscopy (AAS; manufactured by PerkinElmer, USA, model Analyst 800).
Total organic carbon (TOC) in the sediment was measured following the protocol of Michael (1984). For the measurement of NO3- and NH4+, the phenol disulfonic acid method (Chapman, 1996) and phenate method (Park et al., 2009) were used, respectively. Soluble reactive phosphorus (SRP) was quantified using the ammonium molybdate stannous chloride method (Murphy & Riley, 1962). P release was measured as described in Hu et al. (2001). For quantitative estimation of heavy metals, the dried sediment samples were digested in a mixture of HNO3, H2SO4 and HClO4 (5:1:1 ratio, v/v, Allen et al., 1986) and quantified by AAS. For quality control, we followed Jaiswal and Pandey (2018).
The microbial biomass-carbon (Cmic) was estimated by chloroform fumigation-extraction (Jenkinson & Powlson, 1976). The microbial metabolic quotient (qCO2) was quantified according to Wardle (1993). The activity of β-D-glucosidase was estimated spectrophotometrically (Eivazi & Tabatabai, 1988). Alkaline phosphatase (AP) content was determined using buffered p-nitrophenyl phosphate (Tabatabai & Bremner, 1969). Fluorescein diacetate hydrolysis (FDAase) was assayed as per Schnürer and Rosswall (1982). Protease (L-tyrosine equivalent) activity was determined as described by Ladd and Buttler (1972). Nitrate reductase (NR) activity was assessed following Nowak et al. (2002). Sediment oxygen demand (SOD) was quantified as per Ling et al. (2009). For calculation purposes, we used a plot of DO in the SOD chamber with respect to time and computed SOD as described by Doyle and Rounds (2003).
The results were statistically analysed by analysis of variance (ANOVA). Data are presented as the mean supported by the standard error (SE). Correlation analyses were used to test the level of significance between variables. The stability coefficient for the different variables was calculated using the coefficient of variation (cv) following Kardol et al. (2018).
Results and discussion
The worldwide spread of COVID-19 during the pandemic and the enforcement of lockdown policies caused a dramatic decrease in industrial activities, road traffic and tourism; consequently, air and water quality was expected to show improvement. Some quickly published studies support these expectations (Yunus et al., 2020; Häder et al., 2020; Global Carbon Project, 2020). A similar scenario could also be expected for the Ganga River because a sizable amount of the water quality degradation in this river is caused by industrial activities (CPCB, 2013). Here, we observed two different but interlinked forms of changes measured in context of lockdown-driven alterations in the river water and sediment attributes. The first form of change includes “quality determinants” measured in terms of decrease in the concentrations of carbon, nutrient and heavy metal pollutants during lockdown period. The second form of change represents “response determinants” measured in terms of increases in extracellular enzyme (EE) activity as influence by declining concentration of metal pollutants in the river. The study explores causal linkage between these variables and lockdown period restrictions and industrial shut-down.
Nutrient enrichment enhances eutrophy, leading to a progressive accumulation of biomass and the associated changes (Pandey et al., 2014). Excessive loading of carbon enhances the BOD, whereas an excess of metals causes detrimental effects on microbial activity (Jaiswal & Pandey, 2018) and to aquatic consumers (Siddiqui & Pandey, 2019). We found a significant (p < 0.05) decline in heavy metal concentrations in the water and sediment samples in 2020 compared to those from 2019 at all sites (Figs. 2 and 3). The microbial metabolic quotient (a measure of the stress conditions) showed a synchronous trend. In contrast, microbial biomass and the activities of the extracellular enzymes FDAase, β-D-glucosidase and protease all increased during the lockdown period (2020) compared to the previous year (Fig. 4). The effects of both the year and site were significant (p < 0.01; ANOVA). We found a strong negative correlation between EE activity and the concentrations of metal pollutants measured in 2019. The correlation between these variables appeared poor during the lockdown period (Tables 1 and 2). Furthermore, we found greater stability coefficient values (low variance) for the different variables measured during 2020 (lockdown period) compared to those measured in 2019 (Table 3).
Because industrial flushing is the main source of heavy metal input to the Ganga River, a reduced concentration of metals in the water and in the upper layer of sediment is indicative of reduced industrial activity during the lockdown, and an increase in enzymatic activity provides a second indicator of industrial shutdown–associated improvement as the lockdown progressed. The extracellular enzymes, especially FDAase, β-D-glucosidase and protease considered here, are used to relate and monitor the shifts in river ecosystem functioning. FDAase is commonly used as a proximate to total microbial activity (Aftabuddin et al., 2017; Jaiswal & Pandey, 2019b), β-D-glucosidase as a marker enzyme for C- acquisition (Sinsabaugh et al., 2009) and protease is considered as a proxy of N acquisition (Rejsek et al., 2008). A decline in the denitrification enzyme nitrate reductase indicated an improvement in the benthic environment with respect to DO (Pascual et al., 2000), and the decline in alkaline phosphatase (AP) indicated a decreased benthic P supply. Furthermore, because industrial flushing adds organic C to the river and because microbial extracellular enzymes drive organic C decomposition, a reduction in total organic C compared to the previous year (2019) is a third indicator of industrial lockdown. An increase in extracellular enzyme (EE) activity indicates a temporary recovery in river health. This is because EEs are highly sensitive to metal stressors and as the concentration of metal pollutants increases, the enzyme activity declines (Jaiswal & Pandey, 2018). Metal pollutants, at high concentration, are capable of inhibiting microbial communities that produce extracellular enzymes (Jaiswal & Pandey, 2019a) and or directly inhibiting the EE activity by altering the enzyme–substrate binding sites (Jaiswal & Pandey, 2019b). For this reason, the increases in EE activities during industrial shutdown, a signature of decreased concentration of metal pollutants, could be considered as a “recovery window” in river ecosystem functioning. Collateral effects often change the overall impacts that constrain the prediction of specific responses to individual stressors. Such effects could be seen with the parallel entry of carbon, nutrients and heavy metal pollutants. As observed here, the EE activity, which is stimulated by carbon and nutrient substrates, increased with the decline in the concentrations of metal pollutants. Heavy metals, which were present in higher amounts in 2019, outweighed the stimulatory effects of carbon and nutrients; thus, inhibitory effects on EE activity were observed, which is consistent with previous results (Jaiswal & Pandey, 2018). This has also been impressed upon by correlative evidences. We found a strong negative correlation between EE activity and the concentrations of metal pollutants measured in 2019. However, the correlations between these variables were found to be poor during the lockdown period indicating further a decline in the concentration of metal pollutants. Furthermore, we found greater stability coefficient values for the different variables measured during the lockdown period compared to those measured in 2019, suggesting that increases in multiple anthropogenic perturbations can be judged by increases in variance. Furthermore, because the fluxes in carbon and metal vary in response to the strengths of different sources, we found wide variations in the collateral effects along the river tract. The COVID-19 lockdown period–driven reduction in these pollutants, especially heavy metals, may help understand the changes in these collateral effects and is relevant to determining the specific attributes of river ecosystems, which need prompt attention for management decisions.
The dissolved oxygen in both the subsurface water (DOW) and at the sediment–water interface (DODSW) showed improvement, increasing with respect to the values from the previous year by 11 and 22% and − 67 and − 64% at Adpr (the least polluted site) and − 63 and − 65% and 367 and 216% at Wpdr (the most polluted site), respectively (Figs. 5 and 6). At all sites, the DOSW remained > 2.0 mg L−1. Biological and chemical oxygen demand (BOD and COD) showed the opposite trend and declined at all sites, but more sharply at those sites close to industrial flushing. Compared to Adpr, at Wpdr, the BODW was higher by 129% and 258%, respectively, in 2019 and 2020, respectively (Figs. 5 and 6). The sediment oxygen demand (SOD) followed a trend similar to that of BOD. The concentrations of total organic carbon (TOC), the main causal factor of BOD, and nutrients declined significantly (p < 0.05) in 2020. Associated with these determinants, the dissolved oxygen deficit at the sediment–water interface (DODSW) was relatively less marked in 2020 than that in 2019. As has been previously observed (Pandey et al., 2019), during extremely low flow, many places along the Ganga River show a mosaic of hypoxic zones. Here, hypoxia at the sediment–water interface (DOSW < 2.0 mg L−1) was also observed at the Wpdr, Asdr and Vrun sites during the 2019 low flow. These sites are characterised by an industrial point source (Wpdr) and the highly polluted urban tributaries Assi (Asdr) and Varuna (Vrun). Interestingly, however, none of the sites showed hypoxic conditions (DOSW < 2.0 mg L−1) at the sediment–water interface during the 2020 lockdown. This could be linked to the reduced concentration of oxygen-demanding substances, as reflected by the increased DOW and DOSW during the lockdown low-flow period in 2020. Over 760 grossly polluting industries discharge their effluent directly into the Ganga River (CPCB, 2016), but there was almost complete industrial shutdown during the COVID-19 lockdown. Improvements in DOW and DOSW were coupled with an asynchronous trend in BOD and COD in the river. This was also reflected in terms of the decreased SOD during 2020, indicating a reduced sedimentation load of oxygen-demanding substances (ODSs) during this period. This is relevant because SOD is an important contributor to DODSW in the Ganga River (Jaiswal & Pandey, 2019a). This effect was more pronounced at Wpdr, which is under the strong influence of an industrial point source that flushes a sizable amount of other ODSs.
Overall, this study showed that the industrial shutdown during the COVID-19 lockdown caused a considerable positive impact on Ganga River water quality measured in terms of significant improvements in carbon, nutrient and metal pollution. Even the highly polluted zones did not show the signs of hypoxia (DO < 2.0 mg L−1) that had been observed in the previous year. Despite a significant decrease in carbon concentration (an important substrate for enzymatic activities), the apparent decline in heavy metal concentrations created a window for the enzymatic activity to intensify. Because heavy metal decline can be directly linked to industrial shutdown, our study identified industrial release as an important source of metal pollution in the Ganga River. The study emphasises the need to complement change variables (such as shifts in water and sediment chemistry) with more comprehensive response determinants (such as enzyme activity and dissolved oxygen deficit) to more appropriately address changes in river ecosystem health. We suggest that diversion of industrial release and moving beyond monitoring are needed to incorporate ecosystem response attributes during river management decisions.
Conclusions
We argue that the COVID-19 lockdown period dramatically improved the water and sediment quality of the Ganga River. Industrial shutdown during this period led to a significant decrease in the concentrations of heavy metals and consequently the simultaneous and collateral impacts of carbon and heavy metal pollutants, suggesting that industrial release is an important source of metal pollution in the Ganga River. These changes were markedly reflected in river responses measured in terms of microbial extracellular enzyme (EE) activity. Mechanistic links between EE activity and regional-scale multiple anthropogenic stressors established for the Ganga River suggest that microbial EE activity can be used as a sensitive indicator of river responses to changing human perturbations. The study also revealed that declining variance can be used as a potential indicator of diminishing anthropogenic perturbations. The study has potential relevance in evaluating shifts in river responses to anthropogenic changes and in deciding specific attributes of river ecosystems that need prompt attention during management decisions.
Data availability
Data will be made available upon request.
References
Aftabuddin, M., Hassan, M. A., Das, A. K., Jha, B. C., & Sharma, A. P. (2017). Effect of river connectivity on hydrochemistry, sediment enzyme activity and biotic communities of wetlands. Aquatic Ecosystem Health and Management, 20, 40–150.
Allen, S. E., Grimshaw, H. M., & Rowland, A. P. (1986). Chemical analysis. In P. D. Moore & S. B. Chapman (Eds.), Methods in plant ecology (pp. 285–344). Blackwell Scientific Publication, Oxford.
APHA. (1998). Standard methods for the examination of water and wastewater. American Public Health Association.
Central Pollution Control Board. (2013). Pollution assessment: River Ganga. Ministry of Environment and Forests, Govt. of India, Parivesh Bhawan, Delhi.
Central Pollution Control Board. (2016). Bulletin Vol-I. Ministry of Environment and Forests, Govt. of India, Parivesh Bhawan, Delhi.
Chapman DV (Ed.). (1996). Water quality assessments: A guide to the use of biota, sediments and water in environmental monitoring. CRC Press.
Doyle MC, Rounds SA (2003). The effect of chamber mixing velocity on bias in measurement of sediment oxygen demand rates in the Tualatin River basin, Oregon (Vol. 3, No. 4097). US Department of the Interior, US Geological Survey.
Eaton, A. D., Clesceri, L. S., & Greenberg, A. E. (1995). Standard methods for the examination of water and wastewater (19th ed.). American Public Health Association.
Eivazi, F., & Tabatabai, M. A. (1988). Glucosidase and galactosidases in soils. Soil Biology & Biochemistry, 20, 601–606.
Gard, V., Aggrawal, S. P., & Chauhan, P. (2020). Changes in turbidity along Ganga River using Sentinel-2 satellite data during lockdown associated with COVID-19. Geomat Nat Haz Hisk, 11, 1175–1195.
Global Carbon Project: Carbon brief. (2020). https://www.globalcarbonproject.org/carbonbudget/archive.htm
Häder, D. P., Banaszak, A. T., Villafañe, V. E., Narvarte, M. A., González, R. A., & Helbling, E. W. (2020). Anthropogenic pollution of aquatic ecosystems: Emerging problems with global implications. Science of the Total Environment, 713, 136586.
Hill, B. H., Elonen, C. M., Jicha, T. M., Bolgrien, D. W., & Moffett, M. F. (2010). Sediment microbial enzyme activity as an indicator of nutrient limitation in the great rivers of the Upper Mississippi River basin. Biogeochemistry, 97, 195–209.
Hu, W. F., Lo, W., Chua, H., Sin, S. N., & Yu, P. H. F. (2001). Nutrient release and sediment oxygen demand in a eutrophic land-locked embayment in Hong Kong. Environment International, 26, 369–375.
Jaiswal, D., & Pandey, J. (2018). Impact of heavy metal on activity of some microbial enzymes in the riverbed sediments: Ecotoxicological implications in the Ganga River (India). Ecotoxicology and Environmental Safety, 150, 104–115.
Jaiswal, D., & Pandey, J. (2019a). Carbon dioxide emission coupled extracellular enzyme activity at land-water interface predict C-eutrophication and heavy metal contamination in Ganga River, India. Ecological Indicators, 99, 349–364.
Jaiswal, D., & Pandey, J. (2019b). An ecological response index for simultaneous prediction of eutrophication and metal pollution in large rivers. Wat Res, 161, 423–438.
Jaiswal, D., Pandey, U., Mishra, V., & Pandey, J. (2021). Integrating resilience with functional ecosystem measures: A novel paradigm for management decisions under multiple-stressor interplay in freshwater ecosystems. Global Change Biology. https://doi.org/10.1111/gcb.15662
Jenkinson, D. S., & Powlson, D. S. (1976). The effects of biocidal treatments on metabolism in soil – V: A method for measuring soil biomass. Soil Biology & Biochemistry, 8, 209–213.
Kardol, P., Fanin, N., & Wardle, D. A. (2018). Long-term effects of species loss on community properties across contrasting ecosystems. Nature, 557, 710–713.
Ladd, J. N., & Buttler, H. A. (1972). Short-term assays of soil proteolytic enzyme activities using proteins and dipeptide derivates as substrates. Soil Biology & Biochemistry, 4, 19–30.
Ling, T. Y., Ng, C. S., Lee, N., & Buda, D. (2009). Oxygen demand of the sediment from the Semariang Batu River, Malaysia. World Applied Sciences Journal, 7, 440–447.
Michel P. (1984). Ecological methods for field and laboratory investigation. Tata McGraw–Hill Publication Company, New Delhi, India.
Murphy, J., & Riley, J. (1962). A modified single solution method for the determination of phosphate in natural waters. Analytica Chimica Acta, 27, 31–36.
Nowak, J., Kaklewski, K., & Klodka, D. (2002). Influence of various concentrations of selenic acid (IV) on the activity of soil enzymes. Science of the Total Environment, 291, 105–110.
Pandey, J., Jaiswal, D., & Pandey, U. (2019). Point source-driven seasonal hypoxia signals habitat fragmentation and ecosystem change in River Ganga. Current Science, 117, 1947–1949.
Pandey, J., Pandey, U., & Singh, A. V. (2014). Impact of changing atmospheric deposition chemistry on carbon and nutrient loading to Ganga River: Integrating land–atmosphere–water components to uncover cross-domain carbon linkages. Biogeochemistry, 119, 179–198.
Park, G., Oh, H., & Ahn, S. (2009). Improvement of the ammonia analysis by the phenate method in water and wastewater. Bulletin of the Korean Chemical Society, 30, 2032–2038.
Pascual, J. A., Garcia, C., Hernandez, T., Moreno, J. L., & Ros, M. (2000). Soil microbial activity as a biomarker of degradation and remediation processes. Soil Biology & Biochemistry, 32, 1877–1883.
Rejsek, K., Formanek, P., & Pavelka, M. (2008). Estimation of protease activity in soils at low temperatures by case in amendment and with substitution of buffer by demineralized water. Amino Acids, 35, 411–417.
Sánchez, E., Colmenarejo, M. F., Vicente, J., Rubio, A., García, M. G., Travieso, L., & Borja, R. (2007). Use of the water quality index and dissolved oxygen deficit as simple indicators of watersheds pollution. Ecological Indicators, 7, 315–328.
Schnürer, J., & Rosswall, T. (1982). Fluorescein diacetate hydrolysis as a measure of total microbial activity in soil and litter. Applied and Environment Microbiology, 43, 1256–1261.
Siddiqui, E., & Pandey, J. (2019). Assessment of heavy metal pollution in water and surface sediment and evaluation of ecological risks associated with sediment contamination in the Ganga River: A basin-scale study. Environmental Science and Pollution Research, 26, 10926–10940.
Sinsabaugh, R. L., Hill, B. R., & Shah, J. J. F. (2009). Ectoenzymatic stoichiometry of microbial nutrient acquisition in soil and sediment. Nature, 462, 795–798.
Tabatabai, M. A., & Bremner, J. M. (1969). Use of p-nitrophenyl phosphate for assay of soil phosphatase activity. Soil Biology & Biochemistry, 1, 301–307.
Tonkin, J. D., Poff, N. L., Bond, N. R., Horne, A., Merritt, D. M., Reynolds, L. V., Julian, D. O., Albert, R., & Lytle, D. A. (2019). Prepare river ecosystems for an uncertain future. Nature, 570, 301–303.
Voghel, A. I. (1971). A text book of quantitative inorganic analysis, 4th edn. The English Language Book Society.
Wardle, D. A. (1993). Changes in the microbial biomass and metabolic quotient during leaf litter succession in some New Zealand forest and scrubland ecosystems. Functional Ecology, 7, 346–355.
World Health Organization. (2020). WHO-Director-general-s-opening-remarks-at-themedia-briefing-on-covid-19. Retrieved from: www.who.int/dg/speeches/detail/ who-director-general-s-opening-remarks-at-the-media-briefing-on-COVID-19.
Yadav, A., & Pandey, J. (2017). Water quality interaction with alkaline phosphatase in the Ganga River: Implications for river health. Bullet Environ Cont Toxicol, 99, 75–82.
Yunus, E. A. P., Masago, Y., & Hijioka, Y. (2020). COVID-19 and surface water quality: Improved lake water quality during the lockdown. Science of the Total Environment, 731, 139012.
Acknowledgements
The authors thank the Head of the Department of Botany, Banaras Hindu University, for the facilities. We acknowledge the University Grant Commission (grant no. DSKPDF-BL/18-19/0173) for funding support.
Funding
The University Grants Commission, New Delhi, provided financial support to Dr. Madhulika Singh (as UGC Dr. DSKF; grant no. DSKPDF-BL/18–19/0173).
Author information
Authors and Affiliations
Contributions
Madhulika Singh: sample collection and formal analysis, methodology, data validation, writing: original draft, visualisation and funding acquisition. Jitendra Pandey: conceptualization, hypothesis, writing: review and editing, visualisation, project administration and supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• The COVID-19 lockdown dramatically improved the water and sediment quality of the Ganga River.
• Coupling change variables with response determinants more comprehensively addresses changes in river ecosystem health.
• Extracellular enzymes served as a proxy of P, C and N starvation and benthic anoxia/hypoxia-driven feedback.
• Increases in extracellular enzymes indicated a temporary recovery in river health during lockdown.
• Declining variances can be used as a potential indicator of the recovery phase.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Singh, M., Pandey, J. COVID-19 lockdown–driven changes in the Ganga River ecosystem in response to human perturbations. Environ Monit Assess 194, 858 (2022). https://doi.org/10.1007/s10661-022-10507-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-022-10507-9