Abstract
The purpose of this study is to assess metal concentrations and their bioavailability, and relate them to concentrations in fish tissues (gill, muscle and skin) as part of the ecotoxicological and public health assessment of the St Lucia system. Eight metals from six sites (Co, Cr, Cu, Fe, Mn, Ni, Pb and Zn) and five metals (Cu, Fe, Mn, Pb and Zn) from 60 fish, Oreochromis mossambicus, were analysed. Dissolved metal concentrations were highest in the north lake and lowest at the estuary. Sediment metal concentrations were highest in the north lake and lowest in the south lake. Most sediment metals displayed minimal to no anthropogenic enrichment. Normalisation with Fe showed all linear regressions as significant (p < 0.01) and Cr, Cu, Mn, Ni and Zn had R2 values well above 0.9. Only sediment Ni values were above the ERM. Tissue Cu, Fe and Pb concentrations were highest in the gills and Mn and Zn concentrations were highest in the skin. Skin Mn concentrations were highest as compared to the gills and muscle for all sites. The spatial differences in metal concentrations and BCF values for St Lucia suggest distinct populations of fish in the different areas of St Lucia. All concentrations of metals in fish tissues were below the permissible values indicating low human health threat. While higher sediment concentrations justified the economic benefit of mining, evidence of natural enrichment emphasised the need for continual conservation of the greater St Lucia area.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The field of environmental toxicology or ecotoxicology, in its simplest form, seeks to provide an understanding of three basic functions (Landis & Yu, 1995). The first is the interaction of a chemical with the environment. This interaction determines the level of accumulation of the chemical in the medium, either water or sediment, and is referred to as the occurrence of the chemical. The second is the interaction of the chemical with the site of action, from biochemical to physiological levels of interaction. This step determines exposure (Nelke et al., 2020) and includes bioaccumulation (Kuklina et al., 2013). The last function involves the effects of the chemical from the lowest, i.e. individual, level to complex ecosystem level effects (Landis & Yu, 1995). However, very few studies incorporate all these functional steps in the assessment of chemicals in the environment. Most studies combine the first and second, or even the second and third step, with at least one step inferred from the results obtained.
In aquatic environments, metals originate from various natural and anthropogenic sources, including geological weathering, atmospheric deposition and human activities (Du Laing et al., 2008; Izegaegbe et al., 2020). The high metal concentrations associated with very fine sand, silt and mud in estuaries have often been attributed to anthropogenic influences rather than natural enrichment by geological leaching. There has recently been a realisation that metal concentrations differ regionally with some areas having naturally higher concentrations, particularly in sediment (Liu et al., 2013; Newman & Watling, 2007; Chabukdhara & Nema, 2012). Nonetheless, differentiation between naturally and anthropogenically elevated concentrations in estuaries is always difficult because natural leaching, sedimentation and chemical speciation of metals are continuous in estuarine sediments (Cardoso-Silva et al., 2016).
This need to separate metal concentrations and metal bioavailability has necessitated recent studies to emphasise assessment that identifies enrichment due to human activities specifically, in the sediment fraction of the environmental compartment (Newman & Watling, 2007; Chabukdhara & Nema, 2012). The importance of sediment is because of its repository nature and storage potential for contaminants (Riba et al., 2003, Cardoso-Silva et al., 2016; Li et al., 2017). While there may be regional differences in concentrations of metals in sediment, the ratio between metal concentrations in sediment always remains the same (Herut & Sandler, 2006). Therefore, assessment of the relationship of metals with an abundant metal is a reliable tool for all sediment areas. This includes areas such as the east coast of South Africa where naturally high metal concentrations are suggested to occur (Williams & Steenkamp, 2006) but there is a lack of knowledge of what the background concentrations are (Newman & Watling, 2007). Deviations from the expected ratios will therefore indicate enrichment due to human activity. This enrichment usually increases bioavailability by elevating the exchangeable fraction of metals in sediment, which can potentially affect living organisms (Hou et al., 2013). These bioavailability assessments constitute the essence of ecotoxicological exposure studies.
Ecotoxicological studies select mostly the liver, gills and muscle in the assessment of metal concentration in fish tissue. This is because these are target tissues for the accumulation of metals in fish (Rajkowska & Protasowicki, 2013). As this study was directed at danger related to human consumption, only the gills, muscle and skin were selected. In fish, the gills are important as the ion regulation and metal uptake site while the muscle serves as the storage site. And since muscle tissue it is the part that is consumed, the analysis thereof constitutes a public health concern. These tissues are regularly consumed by the local population in the vicinity of the St Lucia area for dietary protein. Fish species, particularly Oreochromis mossambicus, are regularly consumed in a bid to supplement the dire need for protein uptake. It is noteworthy that the locals eat everything (except the digestive tract), including the whole head, and that the gills and skin thus constitute part of the diet. Although the liver also constitutes an important target tissue as it is involved in metal detoxification (Weber et al., 2013), it was excluded from this study as it forms part of the gut which is never consumed.
The St Lucia system is part of the iSimangaliso Wetland Park which is South Africa’s third-largest protected area, spanning 280 km of coastline from the Mozambican border in the north to Maphelane, which is south of the Lake St Lucia estuary, and is made up of approximately 3,280 km2 of natural ecosystems. It is the largest estuarine lake in Africa and was declared a UNESCO World Heritage Site in 1999. Porter et al. (2013) quoted by Humphries et al. (2016) describes the St Lucia system as the largest protected estuarine environment for hippos, crocodiles and aquatic birds on the continent and as the most important nursery for fish on the south-east coast of Africa. The system is shallow, which causes the wind to resuspend bottom sediment (Zikhali et al., 2015), leading to large increases in turbidity, particularly in the northern lake area. Because of its large size, evaporation results in hypersaline conditions (Muir & Perrisinotto, 2011; Taylor et al., 2013) with salinities exceeding three times that of seawater.
The most reported anthropogenic impact to the St Lucia system was the land use change associated with the separation of the inlets of St Lucia and the adjacent Mfolozi estuary to supply water from the Mfolozi river to sugarcane farmers in its catchment (Day, 1981; Lawrie & Stretch, 2011). However, a management plan was in place to reverse the separation and ensure sustainability of the system. The reversal was conducted in 2012, when Mfolozi river water was redirected into the mouth of the estuary. This study was conducted 5 years after the reversal.
The sand dunes of the eastern coast of KwaZulu-Natal (KZN) are rich in heavy metals as evidenced by several mining activities in this area. This situation also exists in the St Lucia dunes and there is currently an application by a local entity, Eyamakhosi Resources, for the right to mine in the south of iSimangaliso Wetland Park of which St Lucia is part (Zululand Observer 17 September 2018). They intend mining for heavy metals which includes iron ore, rutile and zircon. Locals have demonstrated their support for the application through protest action, demanding the right to mine the sand dunes. This strengthens the possibility of an ecological disturbance in the system (Business Day 6 September 2018) and could pose a threat to a very ecologically important and fairly unpolluted system. There is very little to no information on the assessment of chemical pollutants in St Lucia system. It is therefore important to assess levels of metals in media to provide the baseline on occurrence and availability in the estuary before any development is undertaken. Levels in tissues will reveal the threat of metals to local human populations.
Oreochromis mossambicus, commonly known as tilapia, is an important species in the St Lucia estuary. It forms a component of the benthophagous fish community in the estuary, is an important species for anglers (Taylor et al., 2013), and as such, an important source of protein for local communities. It is also one of the few species that are halotolerant having been reported to spawn at a salinity above 100 (Day, 1981). Studies have shown that ecosystem health has been adjudged mostly from the perspective of fish and invertebrate community metrics (O’Brien et al. 2016, Vilmi et al. 2016) and their responses to changes in biological organisation. Although community metrics provide a reliable estimation of the relative vitality of ecosystem functioning, it has been argued that individual or population metrics, including, but not limited to, toxicity testing and bioconcentration may be more useful indicators of ecosystem health as early warning signals (Corsi et al. 2003). This study therefore adopted the use of the bioconcentration factor (BCF) in O. mossambicus as a proxy for estimating the ecosystem health of the St Lucia system.
This study only involved the first two ecotoxicological functions mentioned. Investigating the fate of the chemical was achieved through analysing the metal load in the sediment and as dissolved and suspended fractions. In water, dissolved and particulate metals were determined. Most studies use the enrichment factor (EF) to determine anthropogenic contamination by assessing how far above background sediment metal concentrations are. Though practical, this method is not suitable for the KZN eastern seaboard as background concentrations for the area are not available. High variability in sediment concentrations makes it difficult to employ the background concentrations that are normally used for such studies. In trying to address this void, two methods were used. The first method was through comparative normalisation of sediment samples using Fe. The second method employed compared metals using the effect range low (ERL) and effect range median (ERM) as described by Long and McDonald (2010). The second function, which relates to bioaccumulation, involved the assessment of the different sites of action. These included the gills that serve as the uptake site during breathing, the skin which takes up most metals through both diffusion and active uptake and muscle because accumulation in muscle is a public concern as these fish are consumed by local people.
Due to the large expanse of the St Lucia system, one of the aims was to determine if resident fish species such as O. mossambicus could be differentiated into several populations based on their bioconcentration factor (BCF) levels. The specific objectives of the study were as follows: firstly, to assess spatial variability and enrichment of metals, secondly, to assess public health risk through calculation of the BCF and thirdly, to differentiate distinct fish populations by comparing the BCF between sites.
Materials and methods
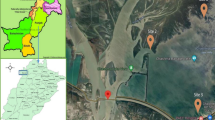
Samples of water and sediment, and 60 fish (O. mossambicus) were collected from six sites in St Lucia estuary. The sampling sites are presented in Fig. 1. The sites were selected such that 2 sites were in the north lake (NL1 and NL2), two sites in the south lake (SL1 and SL2) and two sites in the estuary (E1 and E2). Because of the large size of the system, each of the 6 sites was further divided into three areas such that the water and sediment samples were collected in triplicate from each site. Sediment samples were collected by means of a Zabalocki-type Eckman grab. Sediment samples were collected from all sites except the upper site of the north lake (NL1). The samples for metal analyses were collected from the top 5 cm of the sediment core. All samples were collected during a high flow season in November 2017. All samples were kept in a cooler for analysis in the laboratory. Water quality variables were collected in situ by means of a YSI data logger.
Sample preparation
In the laboratory, all samples were thawed. From the water samples, 250 ml was filtered using a Whatman 45 µm membrane and digested on a hotplate while 0.5 g of sediment was digested using the Ethos Easy Milestone Microwave digester. Fish were weighed and the standard length was measured. Juvenile fish were digested whole on a hot plate while selected tissues (i.e. muscle, skin and gills) were dissected from each adult fish (Dhaneesh et al., 2011) and were dried in the oven at 70 °C for 24 h. From the dried samples, 0.25 g was digested using the Ethos Easy Milestone Microwave digester. All samples were digested using 65% nitric acid and were analysed using the Varian AA50 spectrophotometer. Metals analysed in water and whole fish samples were Co, Cr, Cu, Fe, Mn, Ni and Zn. Only Cu, Fe, Mn, Pb and Zn were analysed in fish tissue and Co, Cr, Cu, Fe, Mn, Ni, Pb and Zn in sediment samples. Water samples were expressed in mg/l while sediment and tissue samples were expressed in µg/g.
Sediment quality guidelines
Sediment samples were compared to ERL and ERM values as described by Long and McDonald (2010).
Statistical analyses
Statistics was performed using IBM SPSS Statistics 25. Comparison of water, sediment and fish was performed using ANOVA and the significance was set at p < 0.05. Regression was used to normalise sediment metal concentrations to sediment Fe concentrations. The BCF was calculated using the equation:
Concentrations of metals in fish were compared to the permissible concentration for tissues to determine the risk for human consumption.
Results
Water quality parameters
Physicochemical parameters measured at St Lucia estuary during the survey are recorded in Table 1. Temperatures ranged from 23.71 °C in the upper south lake to as high as 28.95 °C in the lower north lake. The conductivity and salinity revealed a system with a reverse salinity gradient. High concentrations were found in the upper north lake while very low salinities and conductivity were observed in the estuary, particularly at the mouth (Table 1). Turbidity was very high in the estuary and increased from the estuary to the south lake, and was highest at the north lake. Dissolved oxygen concentrations were generally high in the whole estuary with highest values recorded at the mouth (Table 1).
Water samples
Dissolved metals
Dissolved metal concentrations for all metals except Cr were highest at the north lake. Cr concentrations were highest at E1. Mn and Zn concentrations were highest at the lower site in north lake (NL4) while Co, Cu, Ni and Pb were highest at the upper north lake site (NL1). Dissolved Fe concentrations were highest at SL1. Dissolved concentrations of Cr, Cu and Zn were lowest at south lake while Co, Fe, Mn, Ni and Pb concentrations were lowest at the mouth of the estuary.
Mean concentrations of dissolved metals recorded in the St Lucia system are presented in Table 2. Dissolved concentrations were generally high at NL1, NL2 and SL1. Dissolved Co, Cu, Ni and Pb were highest at NL1, while Mn and Zn were highest at NL2. Except for Cr, which was highest at the estuary, dissolved metals were lowest mostly at the estuary, particularly at E2. Dissolved concentrations of Co, Mn and Zn in the estuary were significantly different (p < 0.05) from the north lake concentrations. The dissolved concentration of Ni at the estuary was significantly different from both the south lake and the north lake concentrations. Dissolved concentrations of Co, Ni and Pb at SL1 were significantly different (p < 0.05) from SL2 concentrations.
Suspended metals
Mean concentrations of suspended metals in the St Lucia system are presented in Table 2. Suspended metals were highest at NL2 and lowest at the estuary (except for Co concentrations which were highest at the mouth, E2). Of the suspended metals that were low in the estuary, only Pb was lowest at E2. Concentrations of suspended Cu, Fe, Mn, Ni and Pb in the estuary were significantly different (p < 0.05) from concentrations at both the south lake and the north lake, while north lake concentrations were also significantly different from south lake concentrations. No significant difference was recorded for suspended Co and Cr concentrations between the different areas of the system. Except for estuarine sites, north lake sites and between SL2 and NL2, all suspended concentrations of Fe from all sites were significantly different from each other.
Sediment samples
Mean concentrations of total sediment metals in the St Lucia system are presented in Table 2 and Fig. 2. Mean concentrations of Co, Fe and Pb were highest at NL2 of the north lake while Cr, Cu, Mn, Ni and Zn were highest at E1, in the upper estuary. All metal concentrations in the sediment were lowest at the south lake. Co, Cr and Fe were lowest at site SL1 while the rest of the metals were lowest at SL2. Sediment concentrations of Fe and Mn were orders of magnitude lower in south lake than in north lake. No large variability was observed in the sediment concentrations of Co and Pb between the various sites in the St Lucia system, particularly between the south lake and the north lake as observed in other metals. Sediment metals from the south lake were significantly different (p < 0.05) from the estuarine and the NL2 metal concentrations, except for concentrations of Pb which were not significantly different between sites.
The order of metal accumulation was different for the different phases and media. Metals in the sediment were ordered as Fe > Mn > Cr > Ni > Zn > Co > Pb > Cu. In the dissolved phase in water, the order of accumulation was Fe > Ni > Co > Pb > Cr > Zn > Mn > Cu. Suspended metals in water, on the other hand, accumulated in the order of Fe > Mn > Cr > Ni > Co > Zn > Cu > Pb. Order of accumulation was the same for the four metals with highest concentrations between sediment and suspended matter.
Comparison with ERL and ERM values
Table 3 presents the comparison of mean sediment metal ranges with ERL and ERM guidelines. Only five of the eight metals analysed in the sediment were compared. Of those, only Ni values (lower and upper value of range) were above the ERL and ERM values. Lower ranges for Cr, Cu and Pb in sediment were lower than the ERL. Sediment Zn’s upper range was similar to the ERL (Table 3).
Normalisation of metals using Fe
The regression of sediment metals against the normaliser element (Fe) and their R2 values are presented in Fig. 3. Four of the six metals in the normalisation had an R2 value well above 0.9. The exception was Co which had an R2 value of 0.74 and Pb with a lower R2 value of 0.4 (Fig. 3). All linear regressions were significant (p ≤ 0.01).
Comparison with other systems
A comparison of metals in St Lucia with other larger systems on the eastern seaboard of KZN is presented in Table 4.
Concentrations of metals in fish tissues
Mean tissue metal concentrations are presented in Fig. 4. Mn, Pb and Zn concentrations in the skin were highest at the estuary. Cu and Fe tissue concentrations were highest in the gills at all sites. Skin Mn concentrations were higher as compared to other tissues for all sites. Metal concentrations were lowest in muscle tissues across all sites.
Correlation of metals in tissues with mass
Metals Fe and Pb were negatively correlated with mass (p = 0.01) while Zn was negatively correlated with Pb (p = 0.05). There were no other strong correlations between metals, with both Cu and Pb correlated with Fe at p = 0.05.
Bioconcentration factor
The bioconcentration factor calculated for fish tissues is presented in Table 5. The highest BCF values in tissues were at E2, at the mouth and at SL2, which is the lower part of the south lake. In the gill tissues, highest BCF values were at E2 for all metals. Skin tissue BCFs for Fe and MN were highest at E2 while for Cu and Zn they were highest at SL2. Skin Mn BCF was very high with the mean BCF above 9. The BCF for Cu in muscle tissues was highest at SL2, while for all other metals it was highest at E2. Mean BCF for muscle tissues was lower than both gill and skin tissue for all metals. Mean Fe BCF for the gills was above 4.
Principal component analysis
Site PCA
In the principal component analysis of metal concentrations in sediment, water and tissues across all sites, the two axes explained 96% of the variation. Estuarine sites were very close together, and NL2 and the south lake sites were also correlated. There was no correlation between the estuarine sites and NL1 (Fig. 5).
Dissolved and suspended metals correlated with NL1. There were no correlations between the BCF values and any sites. Sediment metal concentrations that correlated were grouped together except for Fe which appeared isolated from the group.
Metal PCA
To determine the correlation of metal concentrations in the media with the BCF values, a PCA was performed. The two axes of the PCA explained 92% of the variation. Fe, Mn and Cu were correlated while Cr and Zn formed another group. Mn and Cu were correlated with other sediment metal concentrations from the estuary (E1 and E2) while Fe correlated with suspended metals from NL1 and NL2 (Fig. 6). Tissue BCF for the skin and gill samples correlated with dissolved Pb (at SL2 and E2) and Zn concentrations. Muscle BCFs had no correlations with the suspended metals, but did have a positive correlation with dissolved metal concentrations at E1, SL1 and E2. Suspended metal concentrations from the south lake and the estuary had no correlations with the BCF.
Discussion
Higher concentrations of dissolved metals in the north lake correspond with the higher salinity, as compared to other areas. Salinity increases metal concentrations in the dissolved phase (Hatje et al., 2003). At the same time, the higher turbidity increases the particulate concentrations of metals (Zwolsman & van Eck, 1999). These particulate metals together with the shallow nature of the estuary promote resuspension which will further increase the movement of metals between the dissolved and particulate phases (Zikhali et al., 2015). On the other hand, the low salinity of the estuarine area limits the production of dissolved metals, and the particulate concentrations are also low, particularly at E1. Significant differences in dissolved metal concentrations in the St Lucia system are representative of the influence of different factors within the different areas of the system.
The low concentration of sediment metals in the south lake is related to the nature of the bottom sediment in that part of the lake. Most metals show a reduction in concentration in the south lake: an indication of the inability of the bottom sediment to scavenge metals, as compared to the north lake and the lower estuary.
The significant differences in sediment metal concentrations between the north lake, south lake and the estuary reflect only the nature of sediment and the affinity of the sediment type for metals, which leads to different metal adsorption. The bottom sediment in the north lake is dominated by clay while the upper layer of bottom sediment in the south lake is made of sand that is rich in shell debris (Humphries et al., 2016). A high correlation between the clay and organic content with high metal concentrations is reported by several authors (Du Laing et al., 2008; Hou et al., 2013; Zhang et al., 2014). On the other hand, sandy substrates are rich in carbonates and have less affinity for metals leading to lower concentrations of metals in sandy substrates of aquatic environments (Zhang et al., 2014).
The results of the normalisation indicate that metals from both the north and south lakes, though different at the two localities, are not anthropogenically elevated. The higher concentrations of metals in the sediment at St Lucia as compared to other coastal areas in KZN clearly confirm the continuous pressure of mining in the area. However, the comparison with the ERM guidelines, and the results from the normalisation with Fe, emphasise the pristine nature of the area and the continued need for this (solitary) South African World Heritage marine site to be protected. In unpolluted systems, a large portion of sediment metal is present in crystal lattices (Zhang et al., 2014), but when polluted this proportion is altered. In un-altered or non-polluted sediments, the metals, though elevated, reside in the crystal lattice of minerals and residual fractions which constitute the bottom mud. In polluted areas, metals in sediment mainly exist in soluble, ion-exchangeable forms (Hou et al., 2013), and are metabolically available to living organisms. This alteration is indicated by outliers in the normalisation process and the low R2 value that was not significant. The integrity of the sediment concentrations in the St Lucia system can be explained by the fact that this area is not exposed to industrial development and intense agricultural activities. These activities promote urbanisation which further adds to pollutant discharges into aquatic bodies.
Patterns of sediment concentrations of Pb deviated from the rest of the metals in two main aspects. Pb concentrations were not reduced in south lake and therefore not significantly lower than in north lake. Also, normalisation against Fe returned low R2 values compared to the rest of the metals. Pb contamination is usually caused by accumulation of lead weights from fishing. This does not seem to be the case in this system (St Lucia) as most contamination reported from fishing is localised. There is no immediate explanation for the behaviour of Pb in St Lucia. Although the regression was significant at p < 0.01, the R2 value was low. The uniformity of concentrations in the St Lucia system may indicate the natural deviation which may be localised in St Lucia. It was not reported in Mhlathuze estuary by Mzimela et al. (2014), nor in Richards Bay harbour by Izegaegbe (2020). In both these studies, the R2 values were above 0.7.
The comparison of metals with ERL and ERM guidelines revealed sediment metals that are not enriched. This was supported by the regression of sediment metals against Fe. As the concentration of Fe in the sediment is high, the potential pollution by human activity is limited. In this study, the metals were shown to have a good relationship with Fe (very close to 1), which indicate normal concentrations and sediment that has not been disturbed or contaminated. The ERM for Ni, however, indicated above background concentrations while the relationship with Fe remained good. This may indicate that the enrichment of Ni is natural. This enrichment was also reported by Izegaegbe et al. (2020) in the Richards Bay.
The most important route of uptake of metals in fish is in the dissolved form. Dissolved metal concentrations in the estuarine area of St Lucia were very low but the accumulation of metals by fish was significantly higher as compared to other sites. This supports the biotic ligand model as explained by Di Toro et al. (2001). The model explains that the ‘amount of metal that binds is determined by a competition for metal ions between the biotic ligand and the other aqueous ligands, particularly dissolved organic matter (DOM), and the competition for the biotic ligand between the toxic metal ion and the other metal cations in solution’ (Di Toro et al., 2001). The high salinity levels in the upper areas create this competition with metals for the binding site and for complexation with salts, thereby decreasing the toxicity of the metal (Zhang et al., 2014). Studies have reported on gill complexation in fish that are exposed to metals in the presence of salinity for metals such as silver (Paquin et al., 1999), copper and cadmium (Playle et al., 1992).
All metals that we analysed in fish (except Pb) are essential metals and therefore will accumulate in the body due to storage. Fish are good bioindicators for metal accumulation and are a good food source (Kuklina et al., 2014); the tissues chosen for analysis are target tissues for metals in fish (Rajkowska & Protasowicki, 2013). The gills are the main route of uptake as an organ for respiration, while the skin is also important in uptake. The fish muscle tissues on the other hand are poorly perfused and therefore do not accumulate most metals. The comparison of the muscle concentrations reveals that fish is suitable for consumption. However, local populations in the upper areas of KZN could be under threat because the fish is readily consumed in the northern part of St Lucia and is found sold in markets already fried together with the head and eyes. This may increase the risk from the consumption of fish from the system.
The significant differences between sites may imply that there are distinct populations of O. mossambicus in the St Lucia system. That can be understood from the large size of the system. The whole system spans approximately 3280 km2 (lakes and estuary) and also includes the 280 km2 of coastline. Each of these has unique characteristics that includes a (large) reverse salinity gradient and a large gradient in turbidity. The lower salinity in the lower part is due to the influence of the Mfolozi river which was recently connected to the estuary while the increase in the salinity in the upper areas is due to evaporation occurring in the upper parts of the system. The higher metal concentrations in the lower areas could be as a result of human interventions to limit the hypersaline nature of the system. However, it may increase the risk of the consumption of fish by locals. This risk was further exacerbated by the sampling which occurred during high flow season in summer.
The lack of correlation between metal concentrations in muscle tissue and that in the media was expected. Muscle tissue has been described by Miller et al. (1992) and Dhaneesh et al. (2011) as a poor indicator of metal accumulation as it does not correlate with any medium. The gills, on the other hand, are good indicators and normally correlate with dissolved metal concentrations (Dhaneesh et al., 2011), as was the case in this study.
Apart from the low salinity in the estuary which promotes the availability of metals when compared to salt water, the mouth area of the system has been influenced by human intervention which was to keep the mouth open artificially. The activity of moving sediment changes the form and distribution of metals in the sediment such that they are released from their crystal lattices and therefore interchangeable metals are increased in the sediment (Du Laing et al., 2008). The interchangeable metals constitute the fraction of metals that is bioavailable (Zhang et al., 2014).
Suspended or particulate metals are very important in the St Lucia estuary due to the shallow nature of the estuary and the resultant high turbidity. According to Zhang et al. (2014), fine grained suspended sediment is important in transporting metals in a fluvial system. Resuspension promotes both speciation in the dissolved phase and speciation of metals into labile forms that are highly bioavailable (Zhang et al., 2014).
Metals were shown to be variable in water, with both the dissolved and particulate metals changing with environmental factors, and by resuspension, due to the shallow nature of the system. Variability in sediment metal concentrations was a function of the nature of the sediment. Areas such as the north lake and the estuary with clay bottoms had more metals when compared to the sandy south lake. Enrichment investigations revealed a system that was relatively undisturbed by human activities.
Tissue BCF concentrations in the estuarine area indicated a threat of metals as compared to the upper areas of the St Lucia system. This elevated BCF was due to the lower salinity in the mouth area. The presence of salinity mitigates the uptake of metals by fish and hence the lower BCF in the upper areas. The higher BCF could also be due to anthropogenic leaching of metals as a result of dredging that has been occurring in the mouth area of the estuary.
The different tissue metal concentrations between the different sections of the system suggested distinct populations of O. mossambicus. However, there would be migrations of some individuals into adjacent areas as there are no barriers preventing them. The species was found in areas covering the full range of salinities from 0 to 17 so it is likely that where each population was found within the system reflects its preferred habitat.
Although metal concentrations are high, this does not raise much concern because of their low toxicological risk of bioavailability. This is further mitigated by the fact that locals use mostly the lakes for subsistence fishing and not the estuary. Environmental factors such as pH and redox potential affect the metal availability. With problems such as nutrient enrichment, and urban and rural encroachment observed in areas of iSimangaliso Wetland Park (Adams et al., 2020; an indication that it is not immune), effects that are detrimental to the ecology of the system will soon be observed. Furthermore, Buah-Kwofie and Humphries (2017) reported traces of DDT in the St Lucia system and other parts of iSimangaliso Wetland Park. A coordinated monitoring programme is required that consists of different groups of researchers that will investigate important aspects such as water levels, the chemistry and sediment dynamics of this largest estuarine system in the southern hemisphere. This becomes important as the pressures of mining intensify, and the needs of local communities are beginning to be recognised over environmental concerns.
Data availability
All data generated or analysed during this study are included in this published article.
References
Adams, J. B., Taljard, S., van Niekerk, L., & Lemley, D. A. (2020). Nutrient enrichment as a threat to the ecological resilience and health of South African microtidal estuaries. African Journal of Aquatic Science, 45(1–2), 23–40. https://doi.org/10.2989/16085914.2019.1677212
Buah-Kwofie, A., & Humphries, M. S. (2017). The distribution of organochlorine pesticides in sediments from iSimangaliso Wetland Park: Ecological risks and implications for conservation in a biodiversity hotspot. Environmental Pollution, 229, 715–723.
Cardoso-Silva, S., De Lima Ferreira, P. A., Moschini-Carlos, V., et al. (2016). Temporal and spatial accumulation of heavy metals in the sediments at Paiva Castro Reservoir (Sao Paulo, Brazil). Environmental Earth Sciences, 75, 9.
Chabukdhara, M., & Nema, A. K. (2012). Assessment of heavy metal contamination in Hindon River sediments: a chemometric and geochemical approach. Chemosphere, 87(8), 945–953.
Corsi, I., Mariottini, M., Sensini, C., Lancini, L., & Focardis, S. (2003). Fish as bioindicators of brackish ecosystem health: Integrating biomarker responses and target pollutant concentrations. Oceanologica Acta, 26(1), 129–138.
Day, J.H. (1981). Summaries of current knowledge of 43 estuaries in southern Africa. in J.H. Day (Ed.), Estuarine ecology with particular reference to Southern Africa (pp. 251–330). Rotterdam: A. A. Balkemia.
Dhaneesh, K. V., Gopi, M., Noushad, K. M., Ganeshamurthy, R., Kumar, T. T. A., & Balasubramanian, T. (2011). Determination of metal levels in thirteen fish species from Lakshadweep Sea. Bulletin of Environmental Contamination and Toxicology., 88, 69–73. https://doi.org/10.1007/s00128-011-0459-9
Di Toro, D., Allen, H. E., Bergman, H. L., Meyer, J. S., Paquin, P. R., & Santore, R. V. (2001). Biotic ligand model of the acute toxicity of metals. Environmental Toxicology and Chemistry, 20, 2383–2396.
Du Laing, G., Rinklebe, J., Vandecasteele, B., Meers, E., & Tack, F. M. G. (2008). Trace metal behaviour in estuarine and riverine floodplain soils and sediments: A review. Science of the Total Environment, 407, 3922–3985. https://doi.org/10.1016/j.scitotenv.2008.07.025
Hatje, V., Payne, T. E., Hill, D. M., McOrist, G., Birch, G. F., & Szymczak, R. (2003). Kinetics of trace metals uptake and release by particles in estuarine waters: Effects of pH, salinity and particle loading. Environmental International, 29(5), 619–629.
Herut, B., & Sandler, A. (2006). Normalization method for pollutants in marine sediments: Review and recommendations for the Mediterranean IORL Report H18/2006 Submitted to UNEP/MAP, pp. 23.
Hou, D., He, J., Lu, C., Ren, L., Fan, Q., Wang, J., & Zie, Z. (2013). Distribution characteristics and potential ecological risk assessment of heavy metals (Cu, Pb, Zn, Cd) in water and sediments from Lake Dalinouer. China. Ecotoxicology and Environmental Safety., 93, 135–144.
Humphries, M. S., Green, A. N., & Finch, J. M. (2016). Evidence of EL Niño driven desiccation cycles in a shallow estuarine lake: The evolution and fate of Africa’s largest estuarine system, Lake St Lucia. Global and Planetary Change, 147, 97–105.
Izegaegbe, J. I., Vivier, L., & Mzimela, H. M. M. (2020). Trace metal contamination in sediment in Mhlathuze Estuary, northern KwaZulu-Natal, South Africa. Effects on the macrobenthic community. Environmental Monitoring and Assessment, 192, 401. https://doi.org/10.1007/s10661-020-08352-9
Izegaegbe, J. L. (2020). The macrobenthic community and ecotoxicological status of Mhlathuze Estuary and Richards Bay Harbour, PhD Thesis, University of Zululand. pp 179.
Kuklina, I., Kouba, A., Buřič, M., Horká, I, Ďuriš, Z., & Kozák, P. (2014). Accumulation of heavy metals in crayfish and fish from selected Czech reservoirs. BioMed Research International, 2014, Article ID 306103, pp. 9. https://doi.org/10.1155/2014/306103
Kuklina, I., Kouba, A., & Kozak, P. (2013). Real-time monitoring of water quality using fish and crayfish as bioindicators: a review. Environmental Monitoring Assessment, 185, 5043–5053.
Landis, W. G., & Yu, M. H. (1995). Introduction to environmental toxicology: Impacts upon ecological systems. Florida: Lewis Publishers.
Lawrie, R. A., & Stretch, D. D. (2011). Anthropogenic impacts on the water & salt budgets of st Lucia estuarine lake in South Africa. Estuarine, Coastal and Shelf Science, 93(1), 58–67.
Li, H., Lin, L., Ye, S., Li, H., & Fan, J. (2017). Assessment of nutrient and heavy metal contamination in the sea water and sediment of Yalujiang Estuary. Marine Pollution Bulletin, 117(1–2), 499–506.
Liu, G., Tao, L., Liu, X., Hou, J., Wang, A., & Li, R. (2013). Heavy metal speciation and pollution of agricultural soils along Jishui River in non-ferrous metal area in Jiangxi province, China. Journal of Geochemical Exploration, 132, 156–163.
Long, E. R., & Mc Donald, D. D. (2010). Recommended uses of empirically derived, sediment quality guidelines for marine and estuarine ecosystems. Human and Ecological Risk Assessment: An International Journal, 4, 1019–1039.
Miller, P. A., Munkittrick, K. R., & Dixon, D. G. (1992). Relationship between concentrations of copper and zinc in water, sediment, benthic invertebrates, and tissues of white sucker (Catastomus commersoni) at metal-contaminated sites. Canadian Journal of Fisheries and Aquatic Sciences, 49, 978–985.
Muir, D.G., & Perissinoto, R. (2011). Persistent phytoplankton bloom in Lake St. Lucia (iSimangaliso Wetland Park, South Africa) caused by a cyanobacterium closely associated with the genus Cyanothece (Synechococcaceae, Chroococcales). Applied and Environmental Microbiology, 77(17), 5888–5896.
Mzimela, H. M., Wepener, V., & Cyrus, D. P. (2014). Spatial and temporal variations in selected heavy metals in water and sediment from the Mhlathuze Estuary, Richards Bay. African Journal of Environmental Science and Technology, 8(11), 670–683.
Nelke, E., Mulak, M., Łuczak, K., Pawlak, W., Nienartowitz, J., Szumny, D., Kochman, N., & Gerber, H. (2020). Occurrence and exposure to lead and cadmium and their environmental influence on eyesight. Polish Journal of Environmental Studies, 24(4), 1491–1496. https://doi.org/10.15244/pjoes/39551
Newman, B. K., & Watling, R. J. (2007). Definition of baseline metal concentrations for assessing metal enrichment of sediment from the south-eastern Cape coastline of South Africa. Water Sa, 33(5).
O’Brien, A., Townsend, K., Hale, R., Sharley, D., & Pettigrove, V. (2016). How is ecosystem health defined and measured? A critical review of freshwater and estuarine studies. Ecological Indicators, 69, 722–729.
Paquin, P., Di Toro, D., Santore, R., Trivedi, D., & Wu, K. (1999). A biotic ligand model of the acute toxicity of metals. III. Application to fish and daphnia exposure to silver. In US EPA (Ed.), Integrated approach to assessing the bioavailability and toxicity of metals in surface waters and sediments. EPA-822-E-99–001. Washington, DC, pp 3–59–3–102.
Playle, R., Gensemer, R., & Dixon, D. (1992). Copper accumulation on gills of fathead minnows—Influence of water hardness, complexation, and pH of the gill micro-environment. Environmental Toxicology and Chemistry, 11, 381–391.
Porter, S. N., Branch, G. M., & Sink, K. J. (2013). Biogeographic patterns on shallow subtidal reefs in the western Indian Ocean. Marine Biology, 160, 1271–1283.
Rajkowska, M., & Protasowicki, M. (2013). Distribution of metals (Fe, Mn, Zn, Cu) in fish tissues in two lakes of different trophy in Northwestern Poland. Environmental Monitoring and Assessment, 185, 3493–3502. https://doi.org/10.1007/s10661-012-2805-8
Riba, I., Garcia-Luque, E., Blasco, J., & DelValls, T. A. (2003). Bioavailability of heavy metals bound to estuarine sediments as a function of pH and salinity values. Chemical Speciation and Bioavailability, 15(4), 101–114.
Taylor, R., Fox, C., & Mfeka, S. (2013). Monitoring the St Lucia Estuarine System: A synthesis and interpretation of the monitoring data for March–April 2013. Unpublished Ezemvelo KZN Wildlife report. pp.19.
Vermeulen, L. A., & Wepener, V. (1999). Spatial and temporal variations of metals in Richards Bay Harbour (RBH), South Africa. Marine Pollution bulletin, 39(1–12), 304–307.
Vilmi, A., Karjalainen, S. M., Nokela, T., Tolonen, K., & Hario, J. (2016). Unravelling the drivers of aquatic communities using disparate organismal groups and different taxonomic levels. Ecological Indicators, 60, 108–118.
Weber, P., Everton, R. B., Camila De Lellis, K., Daniel, S. V., Erico, M. F., Valderi, L. D., & Bernardo, B. (2013). Metals in the water, sediment and tissues of two fish species from different trophic levels in a subtropical Brazilian river. Microchemical Journal, 106, 61–66.
Williams, G. E., & Steenkamp, J. D. (2006). Heavy mineral processing at Richards Bay minerals. Southern African Pyrometallurgy, 191–188.
Zhang, C., Yu, Z., Zeng, G., Jiang, M., Yang, Z., Cui, F., Zhu, M., Shen, L., & Wu, L. (2014). Effects of sediment geochemical properties on heavy metal bioavailability. Environmental International, 73, 270–281.
Zikhali, Z., Tirok, K., & Stretch, D. (2015). Sediment resuspension in a shallow lake with muddy substrates: St Lucia, South Africa. Continental Shelf Research, 108, 112–120.
Zwolsman, J. J. G., & van Eck, G. T. M. (1999). Geochemistry of major elements and trace metals in suspended matter of the Scheldt estuary, Southwest Netherland. Marine Chemistry, 66, 91–111.
Acknowledgements
The authors would like to thank Prof L. Vivier and Mr Q. Schutte for help with sampling. Assistance from the staff of the Department of Zoology, University of Zululand, during preparation of samples and analyses is highly appreciated.
Funding
This study was funded by the Department of Zoology, University of Zululand.
Author information
Authors and Affiliations
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mzimela, H.M.M., Izegaegbe, J.I. Metal behaviour in water, sediment and fish from the St Lucia system: implications for public health and ecosystem impact. Environ Monit Assess 193, 807 (2021). https://doi.org/10.1007/s10661-021-09602-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-021-09602-0