Abstract
Environmental pollution has received considerable attention over the last 50 years. Recently, there has been an increasing interest in pollution of the Nile Delta, Egypt, which is one of the longest settled deltaic systems in the world. Pollution in the delta is increasingly recognized as a serious health concern that requires proper management of ecosystems. Therefore, this project aimed to study the distribution and assess the risk associated with selected trace elements (TEs) in different soils (i.e., marine, fluvial, and lacustrine parent materials) in the northern Nile Delta. Mehlich-3 extraction was used to determine the availability of antimony, vanadium, strontium, and molybdenum in agro-ecosystems in this area and their spatial distributions were investigated. Five indices were used to assess ecological risk. Results showed that TEs were higher in the southern part of the study area because it is affected by multiple pollution sources. The available concentrations of TEs were Sr < V < Sb < Mo. The bioavailability of Sr was highest among the studied TEs. The studied indices suggested the study area was moderately polluted by Sr and Sb. Furthermore, the results showed that marine soils had higher TE levels then lacustrine and fluvial soils. The ecological risk assessment indicated that V and Mo were of natural origin, while Sr and Sb were anthropogenically linked. Therefore, the situation calls for planning to reduce pollution sources, especially in the protected north Nile Delta, so these productive soils do not threaten human and ecological health.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The Egyptian Nile Delta supports one of the oldest civilizations based on a deltaic system in the world. This delta was developed by fluvial sedimentary processes beginning in the Miocene and continuing through the current time, built up by the alluvium transported by the seven active Nile branches. It occupies approximately 2% of Egypt’s area; however, it holds around 41% of the population of the country and approximately 63% of its agricultural land. The delta is the major green belt area in the country and is regarded as the basic agricultural wealth of Egypt. However, it is also one of the most densely populated agricultural areas in the world, with 1360 inhabitants per km2. More than 80 million people are mainly dependent on the agricultural and aquacultural products of the Nile Delta (Abu Khatita 2011; El Banna and Frihy 2009; Gu et al. 2013; Elbehiry et al. 2017; Negm et al. 2017; Elbasiouny and Elbehiry 2019).
Trace elements (TEs) are found naturally in soils, but they can also be introduced and accumulate due to their use in industry and agriculture. Various TE indices can be used as indicators to monitor natural variability in soil composition, the probability of anthropogenic contamination, and the toxicological health risks associated with soil pollution (Mehr et al. 2017). For instance, antimony (Sb) released from coal combustion is of environmental concern due to its high solubility and reactivity (Feng et al. 2013). Vanadium (V), which can be introduced through metallurgical enterprises, can have negative effects on human health and crop yields (Vodyanitskii 2016), and excess molybdenum (Mo) in the human diet has been linked to stomach cancer (Xianmao et al. 1990). The effects of stable strontium (Sr) on bone are dose dependent; low doses may induce bone formation, while high doses of this element may stimulate a mineralization defect. The young are more sensitive to excess strontium than older individuals (Kabata-Pendias and Mukherjee 2007). However, the concentrations, availability, and mobility of TEs in soils from many parts of the world, particularly developing countries, are largely unknown, and thus, their impacts on ecosystems can hardly be forecasted.
Human activities may significantly increase concentrations of trace elements in soil and the environment (Steffan et al. 2018). Once contaminated, soils typically remain in this condition for a long time due to the sorption of metals on soil solids, limiting their mobility. Trace elements that are mobilized can be transferred into the food chain, which produces potential human exposure (Telford et al. 2009; Feng et al. 2013; Munthali et al. 2015; Vodyanitskii 2016; Mehr et al. 2017). Hence, TE contamination in agricultural soils is a major concern.
Delineating the spatial and temporal distribution of TEs enables the recognition of sources of such elements in the soil and allows determination of the positions and ranges of over-limit samples in soils for better environmental assessment and management (Khalil and El-Gharabawy 2016; Wang et al. 2016). However, information on the distribution of TEs in the Nile Delta is still quite limited. Trace element data can be used to produce predictive indices, such as the geoaccumulation index (Igeo) and enrichment factor (EF), to evaluate risks posed by TEs based on their total content, bioavailability, and toxicity including the spatial distributions of these risks (Gu et al. 2016). The literature has mainly depended on total (or pseudo-total) analyses of trace elements, whereas environmental monitoring and assessment needs to consider specific elemental mobility and phytoavailability (Giuseppe et al. 2014). The risks associated with TEs are primarily controlled by their mobility and bioavailability, rather than by total amounts (Tiberg et al. 2017). However, the environmental legislation for many countries is based on total soil TEs to define contamination levels and determine any required interventions to mitigate environmental and human health risks (Morgan 2013). Therefore, there is an increasing interest in using the concept of bioavailability in risk assessment and management of contaminated sites because pollutant linkages promoted by TE-contaminated soils do not always correlate with total soil TEs, but rather to TEs’ bioavailability, and it is possible to reduce the exposure of humans and other living organisms with sustainable phytotechnologies (Kumpiene et al. 2017).
This study focused on the availability of selected TEs (Mo, Sb, Sr, and V) because these TEs have been identified as emerging contaminants (ADEQ 2016), but their availability and distribution have not previously been studied in the Nile Delta. In addition, other TEs have been studied (Aitta et al. 2019); therefore, this study adds to that body of knowledge. The primary goals of this study were to:
- 1.
Provide a database for the studied TEs
- 2.
Evaluate the spatial (horizontal and vertical) distribution of these TEs
- 3.
Establish a better understanding of the ecological risk assessment for the TEs that will contribute to sustainable management of this vital area
Material and methods
Study area
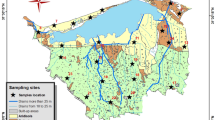
The study area is part of the north Nile Delta, Egypt (i.e., Kafrelsheikh Governorate), which is located south of the Mediterranean Sea (Fig. 1). The area is situated within longitudes 30° 20′ E and 31° 20′ E and latitudes 31° 00′ N and 31° 40′ N, covering 3748 km2. Burullus Lake is an important feature in the study area. Other important natural features and major landforms include flood plain, coastal plain, urban and industrial commercial centers, and some sand dunes in the coastal sections. The main physiographic units include alluvial plain, lacustrine plain, and marine plain (71.08, 19.34, and 9.57% of the total area, respectively). The main soils in this region, classified by US Soil Taxonomy (and World Reference Base), respectively, are as follows: Vertic Torrifluvents (Vertic Eutric Fluvisols), Typic Torrifluvents (Eutric Fluvisols), Typic Natrargids (Solonetz), and Typic Aquisalids (Gleyic Solonchaks) in the alluvial plain; Sodic Aquicambids (Sodic Irragric Cambisols) and Typic Natrargids (Solonetz) in the lacustrine plain; and Typic Torripsamments (Eutric Arenosols) as the most common soils in the marine plain. The main parent materials in the study area are marine (M) (next to the Mediterranean Sea), lacustrine (L) (around Burullus Lake), and fluvial (Fl) (in the southern part of the study area) (Elbasiouny et al. 2014; Elbehiry et al. 2017; Fig. 1). The soil temperature regime is defined as thermic with a torric soil moisture regime (Elbasiouny et al. 2014). The mean temperature of the study area is 19.5 °C, while the mean precipitation is 138 mm/year (Elbasiouny et al. 2014). Most of the area surrounding the Mediterranean Sea is barren with degraded soil; however, there are some cultivated soils in this area that have been reclaimed by local people. The main cropping systems in the reclaimed area are sweet corn, watermelon, and clover in addition to palm and fruits. The area south of Burullus Lake is largely cultivated, with the main crops being rice, cotton, and corn in the summer and wheat, sugar beet, and clover in the winter.
Soil sampling and preparation
Samples were collected from 15 profiles in three different depositional environments, namely M, Fl, and L (Fig. 1). The soil samples were collected in triplicates at three depths (i.e., 0–30, 30–60, and 60–90 cm). The samples were composited, homogenized, air-dried at 25 to 35 °C, crushed, and sieved to 2 mm. Soil properties were determined using the prepared samples as presented in Table 1.
The Mehlich-3 (M3) method was used to extract trace metals in this study, because this method is well correlated with the double acid, 0.1 M HCl or diethylene triamine pentaacetic acid-triethanol amine (DTPA-TEA) (Mehlich 1984). This also avoided the use of certain extraction analyses with harsh reagents (aqua regia) or weak chelating agents (DTPA) that are commonly used to simulate chelating solutions in soils (Giuseppe et al. 2014). The M3 method was developed as a multi-element soil extractant and the M3 extraction was prepared according to Mehlich (1984). The extraction (2 g of soil: 20 ml of Mehlich 3 extractant) was analyzed after 5 min of shaking and filtering with Whatman grade 42 (2.5 μm membranes) filter paper (Mehlich 1984; Yildiz et al. 1998). Elemental analysis was conducted utilizing inductively coupled plasma spectrometry (ICP-OES) with wavelengths of 202.032, 206.834, 421.552, and 292.401 for Mo, Sb, Sr, and V, respectively, and low detection limits (LOD) of 0.026, 0.0126, 0.0198, and 0.056 μg g−1 obtained for Sb, Sr, V, and Mo, respectively.
Pollution indices
Five indices were used for the comprehensive evaluation of soil pollution at the study area. These pollution indices were calculated for all collected samples for the 0–30-cm interval based on the formulas given in Table 2.
Enrichment factor
The EF was used to estimate the status of TEs’ contamination and to distinguish between the possible sources (anthropogenic or natural origin). Aluminum (Al) is most often selected for normalization to minimize particle size effect on the TE values (Roussiez et al. 2005; Xia et al. 2012; Hu et al. 2013). It was utilized as a reference element and the mean EF values for each of the investigated TEs were calculated. Theoretically, EF values of less than 1.5 indicate that the element is completely from crustal sources (e.g., a weathering product), while EF values of more than 1.5 indicate that an important contribution came from noncrustal sources (e.g., biota and/or pollution) (Zhang and Liu 2002; Chen et al. 2007). In this study, the metal pollution level was also determined according to the criteria suggested by Sutherland (2000), where an EF of less than 2 indicates no to minor contamination, 2–5 moderate contamination, 5–20 significant contamination, 20–40 very high contamination, and greater than 40 extremely high contamination.
Geoaccumulation index
This index is a geochemical parameter used to evaluate the level of pollution in soils or sediments by comparing current TE levels to the expected natural values (Table 3) (Müller 1979).
Contamination factor
The contamination factor (CF) is a single-element index that describes the contamination level of a given potentially toxic substance in the environment (Hakanson 1980). The categories used to describe CF are given in Table 3.
Degree of contamination
Degree of contamination (DC) is the sum of CF for all examined elements to assess the extent of multi-element contamination in the surface layers at a certain sampling site. The DC values can be categorized according to the degree of contamination as follows: ≤ 6, 6–12, 12–24, and > 24 as low, moderate, considerable, and very high degree of contamination, respectively (Hakanson 1980).
The pollution load index
The pollution load index (PLI) provides a simple comparative mean for assessing the level of element contamination (Tomlinson et al. 1980). The PLI was obtained as a concentration factor of each trace element with respect to the background value of that element in the soil. The PLI can give an estimate of the element contamination status and the necessary action that should be taken (Angula 1996). Two classes of PLI were proposed by Tomlinson et al. (1980): PLI > 1 and PLI ≤ 1 in the cases of pollution and no element pollution, respectively.
Spatial variation and geostatistical analysis of metals in the studied soils
To interpolate the sampled points for mapping spatial variation of the studied elements, inverse distance weighting (IDW) was applied. The data were processed in ArcGIS 10.1 using a topographic map (1:25,000) of the study area as the base map.
Quality assurance
Glassware and plastic ware for lab work were usually new. They were cleaned using a 10% (v/v) HNO3 soaked for a minimum of 24 h, then they were rinsed well with distilled water. All applied chemicals were analytical reagent grade or equivalent. All equipment was calibrated and uncertainties were calculated. Blanks, measurements, triplicate measurements of measured elements in extracts, and certified reference materials analysis for each metal (Merck) were utilized for quality control. The recovery percentage ranged from 92 to 103% and the mean of relative standard deviation (RSD) was < 3%. The RSD of replicate analysis was < 5%. In very limited cases, the standard deviation (SD) of the measurements was > 5%. These values were excluded from the statistical analyses.
Descriptive statistics for the studied elements were performed using the JMP 11 software (SAS Institute, Cary, NC, USA). All results were analysed using one-way ANOVA to compare the means of differences between soil types. The individual means were compared by Duncan’s test to a level of 5%. A principal component analysis (PCA) was conducted to investigate the relationship between the TEs and the soil chemical and physical characteristics. All tests were performed at p < 0.05.
Results and discussion
Physiochemical characterization of the representative soil samples
The results showed different particle size distribution by depositional environment. The M soils were dominated by a sandy texture, while the Fl and L soils were dominated by silty loam and silty clay, respectively (Table 4). Soil pH was > 7.5 for all samples and there was no significant difference between depositional environments (i.e., M, Fl, and L). The electrical conductivity (EC) values differed significantly between soils, especially in the first layer (0–30 cm) where they ranged from 0.14 to 8.89 dS m−1. The M soils had the highest mean value of EC because of their proximity to the Mediterranean Sea coast.
Soil organic matter (SOM) ranged from 0.2 to 6.7, 5.7 to 30.6, and 7.4 to 21.8 g 100 g−1 in the M, Fl, and L soils, respectively, and decreased with depth. The lowest mean value for SOM was in M soils and was highest in Fl soils, although there was no significant difference in SOM between Fl and L at all studied depths (Table 4). The cation exchange capacity (CEC) ranged from 4.71 to 16.15, 30.37 to 61.64, and 32.94 to 46.92 cmolc kg−1 soil for M, Fl, and L, respectively. The values of CEC for Fl and L soils were significantly higher than the M value for all depths.
The phosphorus (P) concentration was significantly higher in L compared to Fl and M soils with means of 1.48, 2.68, and 6.17 μg g−1 in M, Fl, and L surface soils, respectively. Phosphorus followed a similar trend in the lower layers, where the value was significantly higher in L than in M and Fl (Table 4). Maguire et al. (2009) stated that considerable amounts of sludge cakes are usually utilized in degraded soils in Mediterranean areas. However, the amount of P added with these sludge cakes exceeds crop nutrient requirements and may cause unfavorable changes in soil chemical properties and subsequent environmental contamination. Accumulations of P can impact water bodies through surface run-off or leaching, which may threaten surface and ground waters through eutrophication. Burullus Lake collects agricultural, industrial, and domestic drainage in the delta area, and its water is saturated by elements such as P which in turn affects the P percent in the adjacent soils (i.e., L). Sodium (Na) was the dominant cation in M soils followed by calcium (Ca) and Mg, while Ca was the dominant cation in Fl and L soils. Aluminum, Fe, and Mn were significantly higher in Fl and L soils compared to M soils; they were present in the order Al > Fe > Mn in all studied soils (Table 4).
Availability and spatial variability of studied trace elements
The geospatial distribution of surface soil (0–30 cm) Sb, Sr, V, and Mo is shown in Fig. 2. Due to the different depositional environments and pollution sources, the variability of TEs was different as shown in the geospatial maps for the study area.
Antimony
Antimony is classified as a nonessential element for plants, animals, and humans (Cidu et al. 2013). It is toxic at low concentrations and its excess intake results in many diseases for humans, such as cancers, cardiovascular disease, liver disease, and respiratory disease (Feng et al. 2013). Soil contamination by Sb has caused health problems in regions such as Australia and China (Feng et al. 2013). The soils in this study had low levels of Sb (Table 5). The mean available concentrations in surface soil were 0.44, 0.90, and 0.82 μg g−1 in M, Fl, and L soils, respectively. Antimony levels were higher in Fl and L soils than in M soils; however, no significant difference was found between Fl and L. The available concentrations of Sb in the studied soils were an order of magnitude lower than the lower limit of the maximum allowable concentrations (MAC) in agricultural soil (i.e., 10 μg g−1; Kabata-Pendias 2011). However, the Sb concentrations in the Fl and L soils were higher than the typical background (uncontaminated locations) levels of this element in continental crust and surface soils (0.2–0.61 μg g−1; Kabata-Pendias 2011). Kumpiene et al. (2017) reported that several metalloids such as Sb frequently occur at higher concentrations in TE-contaminated soils than their background values, although they are less studied because they are not subjected to mandatory thresholds in most national policies. The background value (geochemical baseline concentration) is defined as the natural level of an element in soils that have not been affected by human activities. These values can vary widely between regions due to diverse geological properties and dominant soil-forming factors. Because of a lack of true background values for most regions, data such as the average concentration in the continental crust or in shale, preindustrial concentrations, etc. have commonly been used as baseline data when studying metal contamination of soils (Tian et al. 2017). The mobilization of Sb is controlled by soil factors such as metal (hydr)oxides, pH, oxidation–reduction potential (Eh), organic matter (OM), and clay content (Wilson et al. 2010; Huang et al. 2012; Herath et al. 2017). Antimony was once considered to be quite unreactive and immobile in soils, but its behavior in soils has been increasingly highlighted over the last decade (Feng et al. 2013). According to Casado et al. (2007), Sb concentration under oxidizing conditions in soils is not limited by solubility reactions and Sb should be highly mobile under basic pH and oxidizing conditions. Because soils in Fl and L are mostly cultivated by rice (reducing condition), the opposite will be true and Sb is not highly mobile in the studied soils. Antimony was weakly positively correlated with Eh in a study by Frohne et al. (2014). It mainly occurs in the environment as Sb(III) and Sb(V). Compared with Sb(V), Sb(III) is more toxic and less mobile. Antimony in our studied profiles may behave in the same manner.
The geospatial distribution for Sb in the surface layer indicated less spatial variability in the northern part of the Nile Delta (M soils), while the highest available Sb levels were found in the southern part of the study area (Fl soil) (Fig. 2). The higher levels of available Sb in the south may be attributed to the use of agrochemicals and irrigation by wastewater for more than 50 years, both of which are considered pollution sources in this region (Cidu et al. 2013). Other sources of Sb in this region include waste incineration, fossil fuel combustion, ore smelting, and road traffic (Cidu et al. 2013). Antimony and its compounds are hazardous to human health and may be carcinogenic (Cidu et al. 2013; Cheng et al. 2014). The United States Environmental Protection Agency (USEPA), the European Union (EU), the World Health Organization (WHO), and many other countries consider Sb a pollutant of priority interest. For such reasons, this study highlighted the spatial variability of Sb for environmental monitoring purposes in the Nile Delta, a crucial area for food production in Egypt. Although Sb levels do not currently indicate contamination in the study area, values are higher than the expected background values in the surface soils and this work provides a baseline against which future studies of Sb in the northern Nile Delta can be compared.
Strontium
Many anthropogenic activities, including fertilizer application, nuclear power generation, and the burning of coal to generate power, have led to elevated levels of Sr in soil (Burger and Lichtscheidl 2019; George et al. 2015). Strontium is radioactive and the 90Sr isotope has a half-life of around 29 years. Its high potential to accumulate in plants and animals causes serious environmental and human health hazards such as cancers and nervous system disorders (Burger and Lichtscheidl 2019; Sun 2018). The average content of Sr in select materials commonly used in agriculture include 610 mg kg−1 in limestone, 80 mg kg−1 in manure, 75 mg kg−1 in sewage sludge, and 270 mg kg−1 in industrial sludge (Munthali et al. 2015). Phosphate fertilizers can also be a local source of soil Sr contamination (Abdi et al. 2015). There are many soil factors that can lead to releasing immobilized Sr to groundwater such as acidification, dissolution of the solid phase, competitive adsorption with other cations, and complexation by ligands. Therefore, soil contamination by Sr creates concerns for the safety of agricultural soils, irrigation and drinking water, and crops (Munthali et al. 2015).
Strontium concentrations in the surface soils (0–30 cm) ranged from 8.51 to 31.37, 9.05 to 82.62, and 14.27 to 75.49 μg g−1 in the M, Fl, and L soils, respectively (Table 5). The mean worldwide background concentration for Sr in soil is 147 mg kg–1 (147 μg g−1) (Kabata-Pendias 2011). Thus, all Sr concentrations for all soil types in this study were under the background value, indicating there is no pollution by Sr in the study area. Strontium values were highest predominantly in the middle section of the study area (i.e., in Fl and L soils). Because this is an active agricultural area, the higher levels here may be attributed to the use of soil amendments (e.g., phosphors fertilizers, sewage sludge, and lime).
Vanadium
Vanadium is a potentially toxic metal whose soluble salts can adversely affect plant, animal, and human health when present in sufficient amounts (Evans and Barabash 2010; Vodyanitskii 2016). Vanadium primarily creates anionic complexes; nevertheless, it forms neutral and cationic complexes in acidic environments. This variance in charge is caused by the wide variation of chemical properties of this metal. The vanadyl cation (VO2+) is more stable in acidic soils and tightly fixed by humus; however, in neutral environments, the situation becomes more dangerous. In such soils, the vanadate anion (VO−3) increases in importance, and it is more highly mobile and toxic to biota and plants than vanadyl. Subsequently, the hazard of vanadium is higher in cities, where alkalized soils are common (Vodyanitskii 2016). The anions VO3–, VO42–, and H2VO4– are most commonly found in soils. Vanadium mobility in soils appears to be associated mainly with Fe hydrous oxides, clay minerals, and SOM (Kabata-Pendias and Sadurski 2004).
The mean available concentrations of V in the surface soils (0–30 cm) were 1.37, 6.15, and 5.70 μg g−1 in M, Fl, and L soils, respectively, with values that were significantly higher in the Fl and L soils than in the M soils (Table 5). These values are lower than the lower limit of the maximum allowable concentrations in agricultural soil (150 μg g−1; Kabata-Pendias 2011). The global average contents of V in soils vary from 10 μg g−1 in sandy soils to 500 μg g−1 in calcareous soils (Kabata-Pendias 2011). The geospatial distribution map indicated that V levels increased toward the southern portion of the study area, with the lowest V contents mainly observed in soils of light texture (the northern section; Fig. 2). Clay dominates the texture of the southern portion of the study area and likely represents a controlling factor on V mobility and availability in this area.
Molybdenum
Molybdenum is an important micronutrient anion as it is involved in various physiological and enzymatic activities in plants (Mishra et al. 2006) and humans (Schwarz and Belaidi 2013). However, Mo is also a potentially toxic metal in high concentrations and can adversely affect the health of both plants and animals (Evans and Barabash 2010). Available Mo concentrations in the surface soils were 0.09, 0.2, and 0.26 μg g−1 in M, Fl, and L soils, respectively (Table 5). These values were lower than the lower limit of the maximum allowable concentrations in agricultural soils (4–10 μg g−1) and lower than the mean background content of Mo in surface soils as an average of worldwide soils (1.1 μg g−1; Kabata-Pendias 2011). Kumpiene et al. (2017) reported that Mo generally exceeds its background concentration and national guideline values in TE-contaminated soils.
Dehghani et al. (2017) explained the low mobility and bioavailability of Mo in soils by its propensity to form various complex compounds and minerals in soil. Differential adsorption of Mo by Fe, Al, and Mn hydrous oxides contributes to the retention of Mo in surface soils, which may determine its behavior in the environment. Molybdenum from wet alkaline soils is most easily taken up, but the geochemical processes involved in this phenomenon are not completely understood (Kabata-Pendias 2011). The mobile fraction of Mo might be affected by pH and Eh values and is dominated by the MO42− form in moderately alkaline soils. These easily mobile anions are readily co-precipitated by OM, CaCO3, and several cations.
The geospatial distribution of Mo in the surface soils was less variable than Sb and V, but there were high concentrations of Mo in some soils south of Burullus Lake (Fig. 2). The Mo concentrations of our soils were usually similar to those of other studies (e.g., Kubota 1977; Kadunas et al. 1999; Kabata-Pendias 2011).
Vertical distribution of TEs in the soil
To assess the potential transfer or mobility of heavy metals to deeper parts of the soil, samples were collected at three different depths (0–30, 30–60, and 60–90 cm) in all 15 locations. Results of the Sb, Sr, V, and Mo analyses are given in Table 5. In general, the Sb concentrations were highest in the surface interval. In all cases, Sb was lowest in the M soils, with no statistical differences between the Sb values in the Fl and L soils at any depth interval. Antimony has been found to be poorly mobile in soil. In a leaching experiment, Shangguan et al. (2016) found that Sb concentration in the soil solution decreased with depth in each lysimeter they studied, which is similar to the results in our profiles. However, there is a paucity of quantitative data on the distributions of Sb in soil profiles (Shangguan et al. 2016). The results of our study support the idea of restricted mobility of Sb within the soil and lack of movement to deeper parts of the profile.
Strontium levels increased with depth in two M and in one Fl profile. This was due to high concentrations observed in the third layer (60–90 cm depth). Vanadium results indicated mobility to lower depths in the Fl and L soils (Table 5), in contrast with the Sb and most of the Sr results. The distribution pattern of Mo fluctuated sharply with depth (Table 5). These findings are essential for understanding TEs’ mobility and fate in such soils. Kabata-Pendias (2011) explained the mobility of the studied elements (i.e., Mo, Sb, Sr, and V) as follows: Sr is moderately mobile in soils and Sr2+ is the predominant cation form of Sr. Sr2+ is likely to be sorbed in hydrated form by clay minerals and Fe oxides and hydroxides. However, Sb was found to be mostly immobile in the soil near industrial sites and mobility decreases with depth, indicating that it is not likely to contaminate groundwater. Furthermore, Kabata-Pendias (2011) explained that V mobility during weathering depends on the host minerals, with V being adsorbed or incorporated into mineral clay structures or remains in the residual rock-forming minerals or Fe oxides. On the other hand, the mobility of Mo and its availability to plants is highly controlled by soil pH and drainage conditions.
Ecological risk assessment of TEs in the studied soils
Five indices were used to evaluate ecological risk assessment for Sb, Sr, V, and Mo in the agricultural soils in the study area. The indices allowed the assessment of pollution by individual elements (EF, Igeo), evaluation of holistic soil quality (CF, DC), and an estimation of ecological risk (PLI). These indices were calculated for all collected samples and spatial representations of the results are presented in Figs. 3, 4, 5, and 6.
The EF is used to assess the possible impact of anthropogenic activities on the TEs’ concentrations in the soil (Fig. 3). The EF calculated for Sb indicates that some anthropogenic enrichment of this element has occurred, as many of the values were > 1.5 which indicates noncrustal (non-natural) source(s). The EF values for Sr were roughly equally split between not enriched (EF < 1.5) and enriched (EF > 1.5). On the other hand, V and almost all of the Mo values were below 1.5 except for a very small area in the northwestern part of the study area, indicating that the Mo and V came from natural sources. The mean EF values for V and Mo were all lower than 1.5, while the mean EF values for Sb and Sr were higher than 1.5 in all studied soils. This indicated that V and Mo were scarcely influenced by anthropogenic impact and almost entirely came from natural weathering process or crustal materials, while there was an anthropogenic influence on Sb and Sr. Generally, an EF value less than 1.5 suggests that a given element may be entirely from crustal materials or natural weathering processes, and an EF value greater than 1.5 indicates that a significant portion of the trace element was delivered from noncrustal materials or non-natural weathering process (Zhang and Liu 2002; Mazurek et al. 2017). Therefore, contamination by the studied TEs was not of primary concern because almost all the studied soils showed deficiency to minimal enrichment of all elements except Sr.
The Igeo allows the assessment of soil pollution by TEs based on the comparison of TE contents in the surface layer with the geochemical background of the deep soil layers (Kowalska et al. 2016). The TEs were characterized by a narrow range of Igeo values (Fig. 4), with all calculated Igeo values being less than 1. This classifies the soils studied as uncontaminated to moderately contaminated (Zhao et al. 2013).
Spatial depictions of the CF values are shown in Fig. 5. All CF values indicated low to moderate contamination for all studied TEs. Similar to EF, the CF mean values for Sb, Sr, and V were highest in the M soils, while Mo was highest in the L soils.
The calculated PLI values for the four elements in the surface interval are presented in Fig. 6. The values of PLI ranged from 0.03 to 23.36 in all studied soils. The surface soil can be classified as nonpolluted in most of the study area and polluted in some sites in the northeastern part (Tomlinson et al. 1980). The PLI values were higher in Fl and L than M soils.
The DC values ranged from 0.11 to 8.82, indicating low (in most of the study area) to moderate (small area of in the northeastern part of the study area) contamination levels (Fig. 6). The level of pollution differed between TEs and soil types. The DC was greater than 6 in M soil, which was classified as a moderate degree of contamination. The DC evaluation indicated a higher level of pollution than the EF and Igeo indices. This might be explained by the fact that the DC considers multiple TEs that could show higher toxicities and contamination due to the interactions between them (Pekey et al. 2004; Fu et al. 2013).
Based on the different pollution indices (EF, Igeo, CF, PLI, and DC), the M soils are the most polluted followed by the L and Fl soils. The ecological risk assessment indicated that V and Mo were of natural origin, while human activities affected Sr and Sb. Of the studied TEs, Sb and Sr had moderate ecological risk indicators. According to Kabata-Pendias (2011), coal combustion is the main source of Sr pollution. Sr may be concentrated at levels up to 2000 mg kg−1 in phosphorites and is a common source of soil contamination as a result of phosphate fertilization. Because soil alkalinity fixes P, P fertilizers are heavily used in the study area. Antimony contamination can result from construction activities that use concrete and leaded paint, coal combustion products such as ash, and contamination from road traffic (Appleton and Cave 2018). These activities are widespread in the study area, especially construction activities and road traffic which increased dramatically over the last 10 years. Furthermore, Jiménez-Ballesta et al. (2017) studied Sb and Sr, among other TEs, in a similar ecosystem (flood plain soil) in Spain and found that high concentrations of these elements could be attributed to traditional soil fertilization, municipal waste disposal, and urban wastewater treatment discharge into their study area. All these conditions are also present in the Nile Delta.
Source analysis of heavy metals in the surface soils
Correlation matrix
The results of correlation analysis revealed that Sb, Sr, V, and Mo were positively significantly correlated (p < 0.01), suggesting that these elements have similar sources (Lin et al. 2018, Liu et al. 2018; Table 6). Many authors have found strong correlation between some of these elements, such as Dehghani et al. (2017) and Tapia et al. (2018), who found strong correlations between Sb and Mo.
The major factors controlling Sb concentration in soils are P, soil pH, and Ca, which effectively influence mobility and solubility of Sb in the soil solution (Feng et al. 2013). The strongest correlation in this study was observed between Sb and Ca (R2 = 0.72), which is in accordance with Appleton and Cave (2018), whereas Sb was negatively correlated with both pH and P. To form Ca2(SbO3), Ca in soil can co-precipitate with Sb. Ca2(SbO3) is highly insoluble and the dissolution of romeite (Ca[Sbx(OH)y]Z) will govern antimonate leaching at pH values ranging from 8 to 11 (Cornelis et al. 2012; Feng et al. 2013). It is worth noting that the studied soils had pH values between 7.79 and 8.87. Feng et al. (2013) stated that a considerable amount of the Sb in soil is retained by organic matter. They added that Sb solubility has been observed to decline with increasing clay and SOM. However, the results of the current study indicate positive significant correlation between Sb and OM. Furthermore, the concentration of Sb in the soil solution was negatively correlated to the concentration of Na in all studied soils (p < 0.01). Previous studies indicated that Sb and Na2S react to form Na3SbS3. Sb concentration in soil will be affected by the dissolution of Na3SbS3 (Anderson 2012; Shangguan et al. 2016).
The strongest positive correlations with TEs were observed with clay, silt, OM, Na, Mg, and CEC, while the strongest negative correlations were observed between TEs, sand, and pH (Table 6). Other researchers have also documented SOM as an important characteristic affecting TEs’ availability (Khaledian et al. 2017), with SOM having the ability to both increase and decrease the mobility of soil metals (Giuseppe et al. 2014). This is further supported by molybdenum’s significant positive correlation with organic carbon (OC) (r = 0.795**; Mishra et al. 2006). A spatial correlation was also noticed between Ca and Sr at the 0.01 significance level (R2 = 0.91), which indicates the potential for a common origin in the studied soil.
All studied elements were significantly correlated with Mn, while the opposite was true with Fe. Feng et al. (2013) explained, based on several studies (i.e., Leleyter and Probst 1999; Ilgen and Trainor 2011; Guo et al. 2014), that a considerable portion of the Sb in soil was sorbed to iron hydroxides. They also mentioned that powerful evidence indicated that complexes of inner-sphere surfaces favored binding of Sb to iron oxides, as has been shown by measurements of extended X-ray adsorption of fine structures. In addition, there is direct evidence for strong interactions between Sb and Fe–O in red earth soils (Vithanage et al. 2013). This is in contrast to the current study, maybe because of the difference between the study designs. The study of Vithanage et al. (2013) was a spiked experiment performed to study the sorption of Sb(V) in soil on specific mineral surfaces at different pH values, as opposed to this study which investigated Sb in the soil environment at fairly consistent pH values. They also noted that Sb was negatively correlated with Fe and Mn in their four lysimeters and they explained this by the ability of Fe and Mn compounds to strongly adsorb Sb. Therefore, the Vithanage et al. (2013) study partially agrees with ours.
All studied TEs were negatively correlated with EC, Na, and sand except Mo, which was positively correlated with EC. This was attributed to the increase in Mo in sites adjacent to the M soils. Sr had the highest significant negative correlation to EC, Na, and sand. The lowest negative significant correlation with EC, Na, and sand was V with EC and Na and Mo with sand. On the other hand, all studied TEs had significant positive correlations with CEC, silt, clay, K (except Sb and Mo), and Ca. Sr was the highest positively correlated element with the abovementioned soil properties followed by V > Sb > Mo.
Principal component analysis
PCA, which has been shown to be an effective tool for source identification of TEs and to verify the major potential sources of pollution in soils (Anju and Banerjee 2012; Lin et al. 2018), was performed to further identify the source of TEs in the investigated soils. Four principal components were obtained (Table 7 and Fig. 7), and those accounted for 81.38% of the total variation in the data matrix. This indicated that TEs and soil physiochemical characterizations can be described by these four principal components. The TEs and soil properties (CEC, Ca, Mg, K, OM, clay, and silt) were included in the first principal component (PC1), explaining the largest amount (50.37%) of the total variance. The first principal component was mainly characterized by heavy positive loading of Sr and V, indicating that they have similar distribution patterns and sources. Hence, PC1 supported earlier conclusions about the predominant role of geogenic processes from weathering of rocks in determining the concentrations of these elements (Liu et al. 2015). The second principal component (PC2) was composed of Mn, Al, and Fe and explained 15.70% of the total variance. The sources of this second set of elements are considered different than that of the elements in PC1. The third principal component (PC3) included Na and EC and explained 9.24% of the total variance. The fourth principal component (PC4) included only P, explaining 6.07% of the total variance. The results of the PCA were consistent with TEs’ distribution in the studied soils and this further confirmed the source of TEs in the studied area.
Conclusion
This study was developed to provide baseline information on the spatial variability and a risk assessment of selected trace metals in different depositional environments in the northern Nile Delta of Egypt. The results showed variability between available TEs in the different soils in the study area. Sb and V are concentrated in the southern part of the study area, while Sr was dominant in the center of the study area. Mo showed less variability than Sb and V, and high levels of available Mo were recorded mainly in some spots south of Burullus Lake. The risk assessment evaluation of the studied elements indicated V and Mo were of natural origin and not introduced by human activities. The opposite was true for Sr and Sb, where both TEs had moderate ecological risk indicators. Although these TEs do not currently pose a high risk, the continuous monitoring of such elements is necessary because human activities may disturb the dynamics of the ecosystems in the study area. This area is one of the most populated, fertile, and cultivated areas in Egypt, meaning it supports many people. Additionally, this area is subjected to many stresses because it is located close to the sea and hosts many of the industrial and agricultural resources of Egypt as well as many domestic drainages in the Nile Delta. Subsequently, regular TE monitoring and ecological risk assessment in this area is essential to avoid potential environmental and health risks.
References
Abdi, M. R., Saraee, K. R. E., Fard, M. R., & Ghahfarokhi, M. B. (2015). Potential health concerns of trace elements and mineral content in commonly consumed greenhouse vegetables in Isfahan, Iran. Advanced Biomedical Research, 4(1), 214. https://doi.org/10.4103/2277-9175.166152.
Abu Khatita, A. M. (2011). Assessment of soil and sediment contamination in the Middle Nile Delta area (Egypt)—geo-environmental study using combined sedimentological, geophysical and geochemical methods. Doctoral thesis. Friedrich-Alexander-Universität Erlangen-Nürnberg (FAU), Natur wissenschaftliche Fakultät, Germany.
ADEQ. (2016). Emerging contaminants in Arizona water. Phoenix: Arizona Department of Environmental Quality.
Aitta, A., El-Ramady, H., Alshaal, T., El-Henawy, A., Shams, M., Talha, N., Elbehiry, F., & Brevik, E. (2019). Ecological risk assessment and spatial distribution of soil trace elements around Kitchener drain in the Northern Nile Delta, Egypt. Agriculture, 9, 152. https://doi.org/10.3390/agriculture9070152.
Anderson, C. G. (2012). The metallurgy of antimony. Chemie der Erde – Geochemistry, 72, 3–8. https://doi.org/10.1016/j.chemer.2012.04.001.
Angula, E. (1996). The Tomlinson Pollution Index applied to heavy metal, Mussel–Watch data: a useful index to assess coastal pollution. Science of the Total Environment, 187, 19–56. https://doi.org/10.1016/0048-9697(96)05128-5.
Anju, M., & Banerjee, D. K. (2012). Multivariate statistical analysis of heavy metals in soils of a Pb–Zn mining area, India. Environmental Monitoring and Assessment, 184, 4191–4206. https://doi.org/10.1007/s10661-011-2255-8.
Appleton, J. D., & Cave, M. R. (2018). Variation in soil chemistry related to different classes and eras of urbanisation in the London area. Applied Geochemistry, 90, 13–24. https://doi.org/10.1016/j.apgeochem.2017.12.024.
Burger, A., & Lichtscheidl, I. (2019). Strontium in the environment: review about reactions of plants towards stable and radioactive strontium isotopes. Science of the Total Environment, 653, 1458–1512. https://doi.org/10.1016/j.scitotenv.2018.10.312.
Casado, M., Anawar, H. M., Garcia-Sanchez, A., & Regina, I. S. (2007). Antimony and arsenic uptake by plants in an abandoned mining area. Communications in Soil Science and Plant Analysis, 38(9-10), 1255–1275. https://doi.org/10.1080/00103620701328412.
Chen, C. W., Kao, C. M., Chen, C. F., & Dong, C. D. (2007). Distribution and accumulation of heavy metals in the sediments of Kaohsiung Harbor, Taiwan. Chemosphere, 66(8), 1431–1440. https://doi.org/10.1016/j.chemosphere.2006.09.030.
Cheng, H., Li, M., Zhao, C., Li, K., Peng, M., Qin, A., & Cheng, X. (2014). Overview of trace metals in the urban soil of 31 metropolises in China. Journal of Geochemical Exploration, 139, 31–52. https://doi.org/10.1016/j.gexplo.2013.08.012.
Cidu, R., Biddau, R., Dore, E., & Vacca, A. (2013). Antimony dispersion at abandoned mines in Sardinia, Italy. Procedia Earth and Planetary Science, 7, 171–174. https://doi.org/10.1016/j.proeps.2013.03.008.
Cornelis, G., Van Gerven, T., & Vandecasteele, C. (2012). Antimony leaching from MSWI bottom ash: modeling of the effect of pH and carbonation. Waste Management, 32, 278–286. https://doi.org/10.1016/j.wasman.2011.09.018.
Dehghani, S., Moore, F., Keshavarzi, B., & Hale, B. A. (2017). Health risk implications of potentially toxic metals in street dust and surface soil of Tehran, Iran. Ecotoxicology and Environmental Safety, 136, 92–103. https://doi.org/10.1016/j.ecoenv.2016.10.037.
El Banna, M. M., & Frihy, O. E. (2009). Natural and anthropogenic influences in the northeastern coast of the Nile delta, Egypt. Environmental Geology, 57, 1593–1602. https://doi.org/10.1007/s00254-008-1434-6.
Elbasiouny, H., & Elbehiry, F. (2019). Geology. In H. El-Ramady Alshaal, T. N. Bakr, T. Elbana, E. Mohamed, & A.-A. Belal (Eds.), The soils of Egypt (pp. 93–110). Cham: Springer Nature.
Elbasiouny, H., Abowaly, M., Abu Alkheir, A., & Gad, A. (2014). Spatial variation of soil carbon and nitrogen pools by using ordinary kriging method in an area of north Nile Delta, Egypt. Catena, 113, 70–78. https://doi.org/10.1016/j.catena.2013.09.008.
Elbehiry, F., Elbasiouny, H., & El-Henawy, A. (2017). Boron: spatial distribution in an area of North Nile Delta, Egypt. Communications in Soil Science and Plant Analysis, 48(3), 294–306. https://doi.org/10.1080/00103624.2016.1269795.
Evans, L. J., & Barabash, S. J. (2010). Molybdenum, silver, thallium and vanadium. In P. S. Hooda (Ed.), Trace elements in soils (pp. 515–549). Hoboken: Blackwell.
Feng, R., Wei, C., Tu, S., Ding, Y., Wang, R., & Guo, J. (2013). The uptake and detoxification of antimony by plants: a review. Environmental and Experimental Botany, 96, 28–34. https://doi.org/10.1016/j.envexpbot.2013.08.006.
Frohne, T., Rinklebe, J., & Diaz-Bone, R. A. (2014). Contamination of floodplain soils along the Wupper River, Germany, with As, Co, Cu, Ni, Sb, and Zn and the impact of pre-definite redox variations on the mobility of these elements. Soil and Sediment Contamination: An International Journal, 23(7), 779–799. https://doi.org/10.1080/15320383.2014.872597.
Fu, J., Hu, X., Tao, X. C., Yu, H. X., & Zhang, X. W. (2013). Risk and toxicity assessments of heavy metals in sediments and fishes from the Yangtze River and Taihu Lake, China. Chemosphere, 93, 1887–1895. https://doi.org/10.1016/j.chemosphere.2013.06.061.
George, J., Masto, R. E., Ram, L. C., Das, T. B., Rout, T. K., & Mohan, M. (2015). Human exposure risks for metals in soil near a coal-fired power-generating plant. Archives of Environmental Contamination and Toxicology, 68, 451–561. https://doi.org/10.1007/s00244-014-0111-x.
Giuseppe, D. D., Antisari, L. V., Ferronato, C., & Bianchini, G. (2014). New insights on mobility and bioavailability of heavy metals in soils of the Padanian alluvial plain (Ferrara Province, northern Italy). Chemie der Erde, 74, 615–623. https://doi.org/10.1016/j.chemer.2014.02.004.
Gu, J., Salem, A., & Chen, Z. (2013). Lagoons of the Nile delta, Egypt, heavy metal sink: with a special reference to the Yangtze estuary of China. Estuarine, Coastal and Shelf Science, 117, 282–292. https://doi.org/10.1016/j.ecss.2012.06.012.
Gu, Y.-G., Gao, Y.-P., & Lin, Q. (2016). Contamination, bio accessibility and human health risk of heavy metals in exposed-lawn soils from 28 urban parks in southern China’s largest city, Guangzhou. Applied Geochemistry, 67, 52–58. https://doi.org/10.1016/j.apgeochem.2016.02.004.
Guo, X. J., Wu, Z. J., He, M. C., Meng, X. G., Jin, X., Qiu, N., & Zhang, J. (2014). Adsorption of antimony onto iron oxyhydroxides: adsorption behavior and surface structure. Journal of Hazardous Materials, 276, 339–345. https://doi.org/10.1016/j.jhazmat.2014.05.025.
Hakanson, L. (1980). An ecological risk index for aquatic pollution-control—a sedimentological approach. Water Research, 14, 975–1001. https://doi.org/10.1016/0043-1354(80)90143-8.
Herath, I., Vithanag, M., & Bundschuh, J. (2017). Antimony as a global dilemma: geochemistry, mobility, fate and transport. Environmental Pollution, 223, 545–559. https://doi.org/10.1016/j.envpol.2017.01.057.
Hu, B., Li, J., Zhao, J., Yang, J., Bai, F., & Dou, Y. (2013). Heavy metal in surface sediments of the Liaodong Bay, Bohai Sea: distribution, contamination, and sources. Environmental Monitoring and Assessment, 185(6), 5071–5083. https://doi.org/10.1007/s10661-012-2926-0.
Huang, Y., Chen, Z., & Liu, W. (2012). Influence of iron plaque and cultivars on antimony uptake by and translocation in rice (Oryza sativa L.) seedlings exposed to Sb(III) or Sb(V). Plant and Soil, 352, 41–49. https://doi.org/10.1007/s11104-011-0973-x.
Ilgen, A. G., & Trainor, T. P. (2011). Sb(III) and Sb(V) sorption onto Al-rich phases: hydrous Al oxide and the clay minerals kaolinite KGa-1b and oxidized and reduced nontronite NAu-1. Environmental Science & Technology, 46, 843–851. https://doi.org/10.1021/es203027v.
Jiménez-Ballesta, R., García-Navarro, F. J., Bravo, S., Amoros, J. A., Pérez de los Reyes, C., & Mejias, M. (2017). Environmental assessment of potential toxic elements contents in the inundated floodplain área of Tablas de Daimiel wetland (Spain). Environmental Geochemistry and Health, 39, 1159–1177. https://doi.org/10.1007/s10653-016-9884-3.
Kabata-Pendias, A. (2011). Trace elements in soils and plants (4th ed.). Boca Raton: CRC.
Kabata-Pendias, A., & Mukherjee, A. B. (2007). Trace elements from soil to human. Berlin: Springer.
Kabata-Pendias, A., & Sadurski, W. (2004). Trace elements and compounds in soil. In E. Merian, M. Anke, M. Ihnat, & M. Stoeppler (Eds.), Elements and their compounds in the environment (2nd ed., pp. 79–99). Weinheim: Wiley-VCH.
Kadunas, V., Budavicius, R., Gregorauskiene, V., Katinas, V., Kliuugiene, E., Radzevicius, A., & Taraskievicius, R. (1999). Geochemical atlas of Lithuania. Vilnius: Geological Institute.
Khaledian, Y., Pereira, P., Brevik, E. C., Pundyte, N., & Paliulis, D. (2017). The influence of organic carbon and ph on heavy metals, potassium, and magnesium levels in Lithuanian Podzols. Land Degradation and Development, 28, 345–354. https://doi.org/10.1002/ldr.2638.
Khalil, M., & El-Gharabawy, S. (2016). Evaluation of mobile metals in sediments of Burullus Lagoon, Egypt. Marine Pollution Bulletin, 109, 655–660.
Kowalska, J., Mazurek, R., Gąsiorek, M., Setlak, M., Zaleski, T., & Waroszewski, J. (2016). Soil pollution indices conditioned by Medieval metallurgical activity: a case study from Krakow (Poland). Environmental Pollution, 218, 1023–1036. https://doi.org/10.1016/j.marpolbul.2016.04.065.
Kubota, J. (1977). Molybdenum status of United States soils and plants. In W. R. Chappell & K. K. Peterson (Eds.), Molybdenum in the environment (pp. 555–581). New York: Marcel Dekker.
Kumpiene, J., Giagnoni, L., Marschner, B., Denys, S., Mench, M., Adriaensen, K., Vangronsveld, J., Puschenreiter, M., & Renella, M. (2017). Assessment of methods for determining bioavailability of trace elements in soils: a review. Pedosphere, 27(3), 389–406. https://doi.org/10.1016/S1002-0160(17)60337-0.
Leleyter, L., & Probst, J. L. (1999). A new sequential extraction procedure for the speciation of particulate trace elements in river sediments. International Journal of Environmental Analytical Chemistry, 73, 109–128. https://doi.org/10.1080/03067319908032656.
Li, P., Qian, H., Howard, K. W., Wu, J., & Lyu, X. (2014). Anthropogenic pollution and variability of manganese in alluvial sediments of the Yellow River, Ningxia, northwest China. Environmental Monitoring and Assessment, 186(3), 1385–1398. https://doi.org/10.1007/s10661-013-3461-3.
Lin, Y., Ma, J., Zhang, Z., Zhu, Y., Hou, H., Zhao, L., Sun, Z., Xue, W., & Shi, H. (2018). Linkage between human population and trace elements in soils of the Pearl River Delta: implications for source identification and risk assessment. Science of the Total Environment, 610–611, 944–950. https://doi.org/10.1016/j.scitotenv.2017.08.147.
Liu, J., Ma, K., & Qu, L. (2015). Ecological risk assessments and context-dependence analysis of heavy metal contamination in the sediments of mangrove swamp in Leizhou Peninsula, China. Marine Pollution Bulletin, 100, 224–230. https://doi.org/10.1016/j.marpolbul.2015.08.046.
Liu, X., Jiang, J., Yan, Y., Dai, Y., Deng, B., Ding, S., Su, S., Sun, W., Li, Z., & Gan, Z. (2018). Distribution and risk assessment of metals in water, sediments, and wild fish from Jinjiang River in Chengdu, China. Chemosphere, 196, 45–52. https://doi.org/10.1016/j.chemosphere.2017.
Maguire, R. O., Rubæk, G. H., Haggard, B. E., & Foy, B. H. (2009). Critical evaluation of the implementation of mitigation options for phosphorus from field to catchment scales. Journal of Environmental Quality, 38(5), 1989–1997. https://doi.org/10.2134/jeq2007.0659.
Mazurek, R., Kowalska, J., Gasiorek, M., Zadrozny, P., Jozefowska, A., Zaleski, T., Kepka, W., MarylaTymczuk, M., & Orłowska, K. (2017). Assessment of heavy metals contamination in surface layers of Roztocze National Park forest soils (SE Poland) by indices of pollution. Chemosphere, 168, 839–850. https://doi.org/10.1016/j.chemosphere.2016.10.126.
Mehlich, A. (1984). Mehlich 3 soil test extractant: a modification of Mehlich 2 extractant. Communications in Soil Science and Plant Analysis, 15, 1409–1416. https://doi.org/10.1080/00103628409367568.
Mehr, M. R., Keshavarzi, B., Moore, F., Sharifi, R., Lahijanzadeh, A., & Kermani, M. (2017). Distribution, source identification and health risk assessment of soil heavy metals in urban areas of Isfahan province, Iran. Journal of African Earth Sciences, 132, 16–26. https://doi.org/10.1016/j.jafrearsci.2017.04.026.
Mishra, P., Singh, S. K., Srivastava, P. C., & Singh, S. (2006). Distribution of molybdenum and boron in some soils of northern alluvial plain of UP and Uttaranchal in relation to soil characteristics. Agropedology, 16(1), 60–62.
Morgan, R. (2013). Soil, heavy metals, and human health. In E. C. Brevik & L. C. Burgess (Eds.), Soils and human health (pp. 59–82). Boca Raton: CRC.
Müller, G. (1979). Heavy-metals in sediment of the Rhine-changes since 1971. Umschau in Wissenschaft und Technik, 79(24), 778–783.
Munthali, M. W., Johan, E., Aono, H., & Matsue, N. (2015). Cs+ and Sr2+ adsorption selectivity of zeolites in relation to radioactive decontamination. Journal of Asian Ceramic Societies, 3, 245–250. https://doi.org/10.1016/j.jascer.2015.04.002.
Murphy, J., & Riley, J. P. (1962). A modified single solution method for the determination of phosphate in natural waters. Analytica Chimica Acta, 27, 31–36. https://doi.org/10.1016/S0003-2670(00)88444-5.
Negm, A. M., Saavedra, O., & El-Adawy, A. (2017). Nile Delta biography: challenges and opportunities. In A. M. Negm, O. Saavedra, & A. El-Adawy (Eds.), The Nile Delta (pp. 3–18). New York: Springer International Publishing AG.
Nelson, D. W., & Sommers, L. E. (1996). Total carbon, organic carbon and organic matter. In J. M. Bigham (Ed.), Methods of soil analysis: part 3—chemical methods (pp. 961–1010). Madison: Soil Science Society of America.
Pekey, H., Karakas, D., Ayberk, S., Tolun, L., & Bakoglu, M. (2004). Ecological risk assessment using trace elements from surface sediments of Izmit Bay (northeastern Marmara Sea) Turkey. Marine Pollution Bulletin, 48, 946–953. https://doi.org/10.1016/j.marpolbul.2003.11.023.
Roussiez, V., Ludwig, W., Probst, J. L., & Monaco, A. (2005). Background levels of heavy metals in surficial sediments of the Gulf of Lions (NW Mediterranean): an approach based on 133Cs normalization and lead isotope measurements. Environmental Pollution, 138(1), 167–177. https://doi.org/10.1016/j.envpol.2005.02.004.
Rowell, D. L. (1995). Soil Science Methods & Applications. Library of Congress Cataloging – in – Publication data, New York, USA.
Schwarz, G., & Belaidi, A. A. (2013). Molybdenum in human health and disease. Metal Ions in Life Sciences, 13, 415–450. https://doi.org/10.1007/978-94-007-7500-8_13.
Shangguan, Y., Zhao, L., Qin, Y., Hou, H., & Zhang, N. (2016). Antimony release from contaminated mine soils and its migration in four typical soils using lysimeter experiments. Ecotoxicology and Environmental Safety, 133, 1–9. https://doi.org/10.1016/j.ecoenv.2016.06.030.
Sparks, D. L., Page, A. L., Helmke, P. A., Loppert, R. H., Soltanpour, P. N., Tabatabai, M. A., Johnston, C. T., & Summner, M. E. (1996). Methods of soil analysis: chemical methods, Part 3. Madison, WI: Agronomy Society of America and Soil Science Society of America.
Steffan, J. J., Brevik, E. C., Burgess, L. C., & Cerdà, A. (2018). The effect of soil on human health: an overview. European Journal of Soil Science, 69, 159–171. https://doi.org/10.1111/ejss.12451.
Sumner, M.E., Miller, W.P. (1996). Cation exchange capacity and exchange coefficients. In: Sparks, D.L. (Ed.), Methods of Soil Analysis. Part 3. Chemical Methods. Soil Science Society of America, Inc., Madison, Wisconsin, pp. 1201–1229.
Sun, H. (2018). Association of soil selenium, strontium, and magnesium concentrations with Parkinson’s disease mortality rates in the USA. Environmental Geochemistry and Health, 40, 349–357. https://doi.org/10.1007/s10653-017-9915-8.
Sutherland, R. A. (2000). Bed sediment-associated trace metals in an urban stream, Oahu, Hawaii. Environmental Geology, 39(6), 611–627. https://doi.org/10.1007/s002540050473.
Tapia, J., Davenport, J., Townley, B., Dorador, C., Schneider, B., Tolorza, V., & von Tümpling, W. (2018). Sources, enrichment, and redistribution of As, Cd, Cu, Li, Mo, and Sb in the Northern Atacama Region, Chile: implications for arid watersheds affected by mining. Journal of Geochemical Exploration, 185, 33–51. https://doi.org/10.1016/j.gexplo.2017.10.021.
Telford, K., Maher, W., Krikowa, F., Foster, S., Ellwood, M. J., Ashley, P. M., Lockwood, P. V., & Wilson, S. C. (2009). Bioaccumulation of antimony and arsenic in a highly contaminated stream adjacent to the Hillgrove Mine, NSW, Australia. Environmental Chemistry, 6, 133–143. https://doi.org/10.1071/EN08097.
Tian, K., Huang, B., Xing, Z., & Hu, W. (2017). Geochemical baseline establishment and ecological risk evaluation of heavy metals in greenhouse soils from Dongtai, China. Ecological Indicators, 72, 510–520. https://doi.org/10.1016/j.ecolind.2016.08.037.
Tiberg, C., Bendz, D., Theorin, G., & Kleja, D. B. (2017). Evaluating solubility of Zn, Pb, Cu and Cd in pyrite cinder using leaching tests and geochemical modelling. Applied Geochemistry, 85, 106–117. https://doi.org/10.1016/j.apgeochem.2017.09.007.
Tomlinson, D. C., Wilson, J. G., Harris, C. R., & Jeffery, D. W. (1980). Problems in the assessment of heavy metals levels in estuaries and the formation of a pollution index. Helgoländer wissenschaftliche Meeresuntersuchungen, 33, 566–575. https://doi.org/10.1007/BF02414780.
Vithanage, M., Rajapaksha, A. U., Dou, X., Bolan, N. S., Yang, J. E., & Ok, Y. S. (2013). Surface complexation modeling and spectroscopic evidence of antimony adsorption on iron-oxide-rich red earth soils. Journal of Colloid and Interface Science, 406, 217–224. https://doi.org/10.1016/j.jcis.2013.05.053.
Vodyanitskii, Y. N. (2016). Standards for the contents of heavy metals in soils of some states. Annals of Agrarian Science, 14, 257–263. https://doi.org/10.1016/j.aasci.2016.08.011.
Wang, W., Lai, Y., Ma, Y., Liu, Z., Wang, S., & Chenglin, H. (2016). Heavy metal contamination of urban topsoil in a petrochemical industrial city in Xinjiang, China. Journal of Arid Land, 8(6), 871–880. https://doi.org/10.1007/s40333-016-0057-0.
Wilson, S. C., Lockwood, P. V., Ashley, P. M., & Tighe, M. (2010). The chemistry and behavior of antimony in the soil environment with comparisons to arsenic: a critical review. Environmental Pollution, 158, 1169–1181. https://doi.org/10.1016/j.envpol.2009.10.045.
Xia, P., Meng, X., Feng, A., Yin, P., Zhang, J., & Wang, X. (2012). Geochemical characteristics of heavy metals in coastal sediments from the northern Beibu gulf (SW China): the background levels and recent contamination. Environmental Earth Sciences, 66(5), 1337–1344. https://doi.org/10.1007/s12665-011-1343-y.
Xianmao, L., Guogang, H., & Huijan, W. (1990). Effects of molybdenum on etiology, pathogenesis and prevention of esophageal cancer. In T. Jian’an, P. J. Peterson, L. Ribang, & W. Wuyi (Eds.), Environmental life elements and health (pp. 309–317). Beijing: Science Press.
Yildiz, N., Aydemir, O., Aydin, A., & Ulusu, F. (1998). Suitability of Mehlich III method for assessing the plant nutrients in Erzurum plain and acid soils. In D. Anac & P. Martin-PrÉvel (Eds.), Improved crop quality by nutrient management (pp. 281–284). Dordrecht: Springer.
Zhang, J., & Liu, C. L. (2002). Riverine composition and estuarine geochemistry of particulate metals in China—weathering features, anthropogenic impact and chemical fluxes. Estuarine, Coastal and Shelf Science, 54(6), 1051–1070. https://doi.org/10.1006/ecss.2001.0879.
Zhao, Q., Liu, S., Deng, L., Dong, S., & Wang, C. (2013). Longitudinal distribution of heavy metals in sediments of a canyon reservoir in Southwest China due to dam construction. Environmental Monitoring and Assessment, 185, 6101–6110. https://doi.org/10.1007/s10661-012-3010-5.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Elbehiry, F., Elbasiouny, H., El-Ramady, H. et al. Mobility, distribution, and potential risk assessment of selected trace elements in soils of the Nile Delta, Egypt. Environ Monit Assess 191, 713 (2019). https://doi.org/10.1007/s10661-019-7892-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-019-7892-3