Abstract
Phytosociological study in and around reclaimed coal mine site is an essential requirement for judging restoration impact on a disturbed site. Various studies have been aimed towards assessing the impact of different restoration practices on coal mine wastelands. Plantation scheme in a scientific way is the most suitable approach in this context. During the present investigation, an effort have been made to assess the vegetation dynamics through structure, composition, diversity, and forest floor biomass analysis in and around Bishrampur collieries, Sarguja division, northern Chhattisgarh, India. We have tried to develop strategies for eco-restoration and habitat management of the concerned study sites. Four sites were randomly selected in different directions of the study area. We classified the vegetation community of the study sites into various strata on the basis of height. Two hundred forty quadrats were laid down in various directions of the study area to quantify vegetation under different strata. During our investigation, we found eight different tree species representing four families in the different study sites. The density of the various tree species ranged between 40 and 160 individuals ha−1. The density of sapling, seedling, shrub, and herb ranged between 740 and 1620; 2000 and 6000; 1200 and 2000; and 484,000 and 612,000 individuals ha−1, respectively, in different directions. The diversity indices of the tree reflected highest Shannon index value of 1.91. Simpsons index ranged between 0.28 and 0.50, species richness ranged between 0.27 and 0.61, equitability up to 1.44, and Beta diversity ranged between 2.00 and 4.00. Total forest floor biomass ranged between 4.20 and 5.65 t/ha among the study sites. Highest forest floor biomass occurred in the south direction and lowest at east direction. Total forest floor biomass declined by 6.19% in west, 13.10% in north, and 25.66% in east direction, respectively. The mining activities resulted significant damage to natural vegetation and its dynamics. The study indicated that Acacia mangium, Cassia siamea, and Dalbergia sissoo can be recommended for effective eco-restoration of the concerned sites due to cosmopolitan distribution, high regeneration potential, as well as existence in the form of various girth classes with stable population structure.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Mining has negative impact in its various phases of operations. The diverse nature of deterioration of environment due to mining is a major issue and challenges in modern context. Any alteration in the habitat quality in the form of physical, chemical, and biological processes has significant impact on living entity of the ecosystem. Surface mining imparts significant impact on soil environment that leads to total destruction in soil ecosystem structure and functioning. Several reports on vegetation composition and diversity in mining areas of Chhattisgarh (Singh et al. 2013; Jhariya et al. 2013 and 2016; Kumar et al. 2015 and 2016), Madhya Pradesh (Singh et al. 2004a & b), Jharkhand (Ghose 2004; Guha 2014; Singh et al. 2014), Rajasthan (Kumar et al. 2014), Orissa (Swain et al. 2011; Mohaptra and Goswami 2012), West Bengal (Sadhu et al. 2012), North East India (Rai 2002; Sarma et al. 2010) and other countries (Donggan et al. 2011; Milder et al. 2011; Tom-Dery et al. 2012) are available.
Mining activity imposes its impact on various dimension of environment including biodiversity along with ecological interactions among species. The impact includes reduction in the diversity and abundance of vegetation, plant productivity, and promoting invasive species on the concerned area (Jhariya et al. 2013; Kumar et al. 2015). Such events lead to higher level of deforestation rendering mismanagement and loss of productivity (Jhariya et al. 2016). Coal extraction significantly alters the physico-chemical and biological nature of the excavated site. Surface mining leads to complete denudation of indigenous vegetation due to damage of the soil and its biota (Corbett et al. 1996). Comparative study reflects the nature of intensive pollution of surface mining in comparison to underground mining. Mining activities throughout the world leads to 20% deforestation in developing countries (Bahrami et al. 2010). Scientific effort needs to be developed to quantify the impact of mining activity on biotic systems that would lead to eco-restoration and proper resource management.
A proper understanding of vegetation composition, structure, and community dynamics is vital to address conservation, management, and facilitate restoration of mined out landscape. This study provides an opportunity to understand the changes in vegetation dynamics related with open-cast mining activity in the concerned study site along with proper reclamatory measures.
Material and methods
Study site
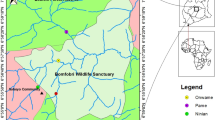
The present study focussed on assessing vegetational structure and dynamics of selected study sites of Bishrampur collieries (23° 21 N and 82.85° E with an average elevation of 527 m) in the district of Surajpur of Northern Hill Zone, Chhattisgarh during the year 2014–2015. Bishrampur is highly concentrated with opencast and underground mining centers (Kumar et al. 2015 and 2016). The climate of this area is sub-tropical in nature with summer from April to June, monsoon season from July to September, post-monsoon from October to December, and winter from January to March. The maximum and minimum temperature generally occurs on May and December, respectively. The Surajpur district receives annual rainfall of 1192.3 mm with 73 normal rainy days.
Sampling design
The directional impact on vegetation of Bishrampur collieries and its surrounding (Fig. 1) was analyzed for its structure, composition, and dynamics in south, north, east, and west wards. Vegetation stands were stratified on the basis of height. Total of 240 quadrats were laid across the sites for quantification of vegetation at different stratum. Random quadrats of 10 × 10 m size were laid for quantification of trees and saplings. In each of this quadrat, a sub-quadrat of 5 × 5 m size was randomly laid for measuring seedling and shrubs. Similarly another quadrat of 50 × 50 cm size within 10 × 10 m quadrat was laid for measuring herbs and forest floor, respectively.
Data collection
The girth at breast height (above 1.37 m from the ground) of all the trees and saplings were measured. For tree species, the individuals >30 cm GBH were categorized as tree, <30 cm but >10 cm as saplings. The seedlings (<10 cm GBH) and shrubs were measured at the collar height. The data were collected up to species level separately in each quadrats in different directions.
Data analysis
Vegetational structure and composition analysis
Vegetational structure and composition were quantitatively analyzed through frequency, density, abundance values (Curtis and McIntosh 1950), and A/F ratio (Whiteford 1949). The relative frequency, relative density, and relative basal area values were calculated following standard methods (Phillips 1959). The sum of relative frequency, relative density, and relative basal area values was used to calculate importance value index (IVI) for the species. The species found in the quadrats were identified with the help of standard text materials (Regional floras of Chhattisgarh & Madhya Pradesh, Handbook of Weeds of Chhattisgarh, flora/encyclopedia; Hooker’s 1875; Pullaiah 2006; Singh et al. 2010).
Raunkiaer (1934) frequency class analysis was used to assess the rarity or commonness of the tree species (Hewit and Kellman 2002). In this classification, the percentage frequency of the species was classified as A, B, C, D, and E, where A represents rare (0–20%), B represents low frequency (20–40%), C represents intermediate frequency (40–60%), D represents moderately high frequency (60–80%), and E represents high frequency or common (80–100%). The population structures were based on different girth classes. The total number of individuals belonging to these girth classes were calculated for each species on each site following Saxena and Singh (1984), Tripathi et al. (1991) and Jhariya et al. (2012).
Diversity indices analysis
The data generated were synthesized to represent the diversity of each direction of mining area for each stratum. Diversity parameters were determined using the Shannon-Weaver information function (Shannon and Weaver 1963). Concentration of dominance was measured following Simpson’s index method (Simpson 1949). Vegetations were also measured for species richness (Marglef 1958), equitability (Pielou 1966) and Beta diversity (Whittaker 1972).
Statistical analysis
To observe the inter-relationship between phytosociological attributes, data were subjected to correlation analysis. Multivariate analysis of the data was done through two-way cluster analysis and principle component analysis through MINITAB presents version 15.0- statistical computer software.
Results and discussion
Impact on overstory
Vegetation structure of tree layer
In four different directions, eight tree species representing four families were recorded during the present investigation (Table 1). The total density of trees ranged between 40 and 160 individuals ha−1 among the various study sites. Highest and lowest total density values were recorded for north and south directions, respectively. The dominant representatives were Dalbergia sissoo, Syzygium cumini, Peltophorum ferrugineum, and Cassia siamea in east, west, north, and south directions, respectively, contributing the higher basal area and IVI in the respective directions. It was apparent from the study that all species were distributed randomly in east and west direction. The A/F ratio for west direction showed random distribution pattern. Results reveal directional influence with respect to tree species distribution.
Development of vegetal cover is a suitable policy towards improving soil health through alteration of various pedogenic factors including increase of soil organic matter, biological nitrogen fixation, nutrient utilization efficiency of herbaceous vegetation, increase water storage and recharge, and building up of soil nutrient pool. Formation of new self-sustaining top soils is mediated by trees. Plant litter and root exudates provide efficient nutrient-cycling to soil (Mertens et al. 2007). During our present investigation, we found that Acacia mangium and Casia siamea can be effectively utilized for eco-restoration of respective study sites. Lower density of tree species in the degraded habitat may be due to intensive logging and felling for mining purpose as well as improper seed dispersal in the degraded habitat from the adjoining forest areas (Robinson and Handel 2000).
Vegetation structure of sapling layer
Sapling layer comprises of ten species representing six families in the different sites (Table 1). The total density ranged between 740 and 1620 individuals ha−1 in different directions. Maximum and minimum total density values were recorded for north and east directions, respectively. Acacia mangium (560 individuals ha−1) recorded higher density in east and south direction, Cassia siamea (400 individuals ha−1) in west direction while Eucalyptus spp. (540 individuals ha−1) in north direction, respectively. The total basal area of sapling among the site ranged between 1.36 and 3.02 m2 ha−1. Highest total basal area was observed in north direction and lowest value was recorded in east direction. Acacia mangium were found to be the most suitable species in terms of establishment and further propagation followed by Cassia siamea.
Phytosociology of vegetation community is a good indicator of ecological condition of land ecosystem. Alteration in the abiotic factors may lead to changes in the community structure in a short span of time (Hassan et al. 2007). Present study reflected density of tree was very low for all the study sites. Majority of species showed random as well as contagious distribution pattern in the study sites. Rahman et al. (2010) reported a total of 715 individuals and 22 invasive species representing 17 families in mining areas. During the investigation, ten species of shrubs, nine species of herbs, and three species of vines were recorded. Sobuj and Rahman (2011) reported a total of 52 (16 tree species, 15 shrubs, 21 herbs) in the natural forest; on the contrary, 31 species of plants (11 trees, 8 shrubs, 12 herb species) were identified in plantation forest. Kumar et al. (2014) found the tree density of 11.67/100 m2, shrub density was 8.33/9 m2, and herb density was 41.11/m2, respectively, in Rajasthan.
The number of tree species was low in all the mining sites due to various mining activities (Sarma et al. 2010). The tree density was more (1040 stems ha−1) in the unmined area in comparison to mined areas (515 and 646 stems ha−1) while density of herbaceous species was higher in mined areas than the unmined area. The trees of medium girth class contributed to the maximum stand density in the mined areas, while in the unmined site, the trees of low girth class contributed to the maximum stand density. In the mined areas it was found that in spite of low stand density, higher basal area were recorded due to existence of trees of high girth class as they were not damaged by the miners during the mining operations (Sarma et al. 2010). From the result, it appears that highest density of tree and sapling layer were recorded in north direction. Therefore, higher regeneration as well as early establishment of species for eco-restoration perspectives can be fruitful in north direction in comparison to other directions.
Impact on understory/ground vegetation
Vegetation structure of seedling layer
In seedling layer, 12 species representing six families were recorded from the different sites (Table 2). The total density of seedling layer in east direction were 2000 individuals ha−1, and the maximum density was recorded by A. mangium (1780 individuals ha−1) and minimum by B. monosperma (80 individuals ha−1). The IVI of individual species ranged between 33.681 and 218.99. In west direction, a sum of 4480 seedlings ha−1 was recorded. The maximum density was recorded by C. siamea (1280 individuals ha−1) and minimum by A. catechu and P. ferrugineum (240 individuals ha−1), respectively. The IVI value ranged between 23.239 and 70.387 in west direction. In north direction, a sum of 5040 individuals ha−1 were observed, and the maximum density was recorded by A. mangium and Eucalyptus spp. (1840 individuals ha−1) and minimum by C. siamea (80 individuals ha−1). The IVI of individual species ranged between 12.803 and 90.704. In south direction, 6000 individuals ha−1 was observed, and the maximum density was recorded by A. mangium (13,440 individuals ha−1), while the minimum by C. fistula and S. cumini (160 individuals ha−1). The IVI of individual species ranged between 21.989 and 146.253. For seedling stage, the highest density of plants was found in south direction representing higher regeneration rate. This therefore may be fruitful for eco-restoration practices for the concerned study sites.
Vegetation structure of herb layer
A total of 48 herb species representing 17 families were recorded in different directions of mining areas (Table 3). East direction comprising 24 species representing 12 families, west direction contains 30 species representing 12 families, and 32 species representing 15 families were recorded towards north direction and 25 species representing 10 families in south direction. The total density of herb species in east direction was 572,000 individuals ha−1. The maximum density was recorded by M. coromandelianum and O. burmannii (96,000 individuals ha−1) and minimum by X. strumarium, V. trilobata, T. quadrivalvis, T. macrodonta, P. vittata, and H. indicus (8000 individuals ha−1). The IVI of individual herb ranged between 6.39 and 37.24; highest value was recorded by O. burmannii and lowest by H. indicus, P. vittata, T. macrodonta, T. quadrivalvis, V. trilobata, and X. strumarium, respectively. A/F ratio of individual species ranged between 0.038 and 0.300. It appears that 29.27% species were distributed randomly and remaining (70.83%) were contagiously distributed. In the west direction, herb density was 612,000 individuals ha−1. The maximum density was recorded by C. rotundus (100,000 individuals ha−1) and minimum by E. hieraciifolium, H. indicus, P. hysterophorus, T. procumbence, V. pilosa, and V. trilobata (4000 individuals ha−1). The IVI of individual herb species ranged between 3.82 and 31.55, highest value were recorded by C. rotundus and lowest by E. hieraciifolium, L. octovalis, P. hysterophorus, T. procumbence, V. pilosa, and V. trilobata, respectively. The north direction reflected 592,000 individuals ha−1. The maximum density was recorded by C. dactylon (68,000 individuals ha−1) and minimum by T. macrodonta and T. procumbence (4000 individuals ha−1). The IVI of individual herb species ranged between 3.71 and 23.25, highest for C. dactylon and O. burmannii and lowest for T. macrodonta and T. procumbence, respectively. The south direction has total herb density of 484,000 individuals ha−1, and the maximum density was recorded by C. tora (64,000 individuals ha−1) and minimum by B. pilosa, E. diarrhena, E. hirta, and T. procumbence (4000 individuals ha−1). The IVI of individual species ranged between 4.62 and 28.40, highest for C. tora and lowest for B. pilosa, E. diarrhena, E. hirta, and T. procumbence, respectively.
From the result, it appears that towards west direction, successful establishment and propagation of herb layer is more prominent due to lower density of tree species providing more light as available resources for the ground vegetation. Findings further supported by the fact that west direction is little bit cooler in comparison to other directions thereby promoting herbaceous growth. The higher number of herbaceous species in the mined areas in comparison to unmined areas could be due to colonizing ability of herbs in the degraded area (Sarma et al. 2010).
Vegetation structure of shrub layer
A sum of nine shrub species representing eight families were found in different directions of study sites (Table 4). The density of shrub species in east direction was 2000 individuals ha−1. The maximum density was recorded by L. camara (1040 individuals ha−1) and minimum by C. gigantea, C. opaca, and L. asiatica (80 individuals ha−1). The total basal area of shrub species in east direction was 0.60 m2 ha−1. The IVI of individual shrub species ranged between 14.24 and 130.96. Highest IVI was recorded for L. camara and lowest for C. gigantea , respectively. In the west direction, total density was 1840 individuals ha−1, and the maximum density was recorded by L. camara (1040 individuals ha−1) and minimum by I. carnea (80 individuals ha−1). The total basal area of shrub species in west direction was 1.060 m2 ha−1. The IVI of individual shrub species ranged between 17.73 and 130.52. Highest IVI value was recorded by L. camara and lowest by I. carnea, respectively. In north direction, a sum of 1600 individuals ha−1 were recorded. The maximum density was recorded for L. camara (720 individuals ha−1) and minimum for Z. rotundifolia (240 individuals ha−1). The total basal area was 0.789 m2 ha−1 towards north direction. The IVI value of shrub species was higher for L. camara and least for Z. rotundifolia. The density of shrub was 1200 individuals ha−1 towards south direction. The maximum density was recorded for L. camara (1040 individuals ha−1) and minimum for V. negundo (160 individuals ha−1). The total basal area for shrub species in south direction was 0.543 m2 ha−1. The IVI of individual species ranged between 44.46 and 255.61, respectively.
Sarma et al. (2010) reported that due to mining, higher number of herbaceous species (31–41) were found in the mining area in comparison to unmined area (23 species). Majority of species showed contagious distribution pattern in the unmined and mined areas. Gupta and Sharma (2015) studied the plant assemblages along an altitudinal gradient in north-west Himalaya and found that the density and basal area of herbage in grassland at different elevations ranged between 649.6 and 1347.9 tillers/m2, 30.0, and 65.7cm2/m2, respectively. Density and basal area of the herbage and plant communities decreased along the elevation.
Lower value of basal area in mining site is a characteristic feature of young stand with low girth class gradation and the density of stand, thereby serves as a reflection of low performance of the trees. Development of extensive root system promotes higher nutrient absorption as well as inhibits growth of ground vegetation due to higher interception of light by the tree species. Absence of tree species may lead to availability of higher solar insolation to the younger plants. There is a gap of knowledge in the context of abundance and distribution of herb, shrub, sapling, and seedling in mining sites both at local and regional level. The reported densities of these vegetation layer of the present investigation are well comparable with some earlier work (Mishra et al. 2008). The fewer number of saplings recorded in relation to seedlings in this study implies that most of the saplings are transiting into young trees. Intense competition leads to mortality of seedling at very early stage (Weidelt 1988; Jhariya et al. 2012, 2014). Among the various girth classes, saplings and seedlings reflect high regeneration potential of the stand (Mishra et al. 2005; Khumbongmayum et al. 2006).
In most of the plant life forms, there were high number of species that occurred singly. The distribution of tree species into Raunkiaer’s frequency classes reflected that most of the species were of rare category and of low frequency class; intermediate class was represented only in the north direction while moderately high and high or common frequency class were totally absent in the study area (Table 5).
Importance value index (IVI) reflects species dominance and ecological succession. High IVI values of a species indicate its high regeneration capacity and greater ecological amplitude. In our study, Acacia mangium and Cassia siamea reflected the highest IVI value. Therefore, these species can be considered as capable of higher regeneration potential in the concerned study sites. The low IVI may be attributed towards rarest occurrence of species (Pascal and Pellissier 1996; Oyun et al. 2009), as confirmed by Raunkiaer’s frequency distribution of the tree species. The rarity of species may be attributed to the occurrence of abundant sporadic species with low frequency in the stands (Oyun et al. 2009). The high percentage of rare species observed in different stratum of vegetation in the collieries confirms that most of the species in an ecological community are rare, rather than common (Magurran and Henderson 2003).
Relationship of density to GBH in collieries
Tree species density-GBH distribution followed an exponential model [Y = exp. (a − bx)] on all the direction of the collieries. The sites thus exhibited a small structure as 69.44–83.33% individuals had ≤10 cm girth, and only 16.67–30.56% were in girth classes exceeding 10 cm GBH (Fig. 2), while 0.0–1.39% individuals were exceeding >50 cm GBH. When data were pooled, the woody species density was related to GBH according to:
-
(1.)
Y = exp. [2289.1–0.784×] for east direction, r 2 = 0.790, p < 5%
-
(2.)
Y = exp. [13799–1.424×] for west direction, r 2 = 0.911, p < 5%
-
(3.)
Y = exp. [18078–1.339×] for north direction, r 2 = 0.966, p < 5%
-
(4.)
Y = exp. [31464–1.794×] for south direction, r 2 = 0.936, p < 5%
Exponential relationship between density and GBH reflected small structure of vegetation where only 0.0–1.39% individuals were in classes >50 cm GBH. This may be related to faster turnover, biotic removal, and low capacity of biomass accumulation in the concerned study sites. Relating woody species density with GBH in dry tropics Singh and Singh (1991) similarly reported same pattern where minor (3–5%) individuals exceeded 50 cm GBH. Similar findings were also reported by Chaturvedi et al. (2011) for tropical dry forest of India. Relating tree density with GBH, in Puerto Rican sub tropical dry forest, Murphy and Lugo (1986) also found a similar pattern where 2–3% individuals exceeded 10 cm GBH. Oraon (2012) reported that nearly 89–94% individuals had ≤10 cm girth and only 1.5–3.7% were in girth classes exceeding 50 cm GBH. Jhariya (2014) found that the forest exhibited a small structure as 86.37–91.71% individuals had ≤10 cm girth, and only 8.29–13.63% were in girth classes exceeding 10 cm GBH, while 1.58–2.18% individuals were exceeding >50 cm GBH. In our study, each tree species is devoid of different girth classes which indicates younger population of tree species. These therefore suggest that it has the potential for frequent reproduction (Knight 1975).
Population structure in mining sites
The population structure revealed the regeneration pattern of a stand or site. The proportion of seedling size class (A) was found to be dominant in east direction (Fig. 3). The older size classes (D) and (E) were totally absent in this direction. The single size class was represented by Saccopetalum tomentosum and Ailanthus excelsa. The sapling size class (B) was nil for Dalbergia sissoo, while the size classes (A), (B), and (C) were represented by Acacia mangium and Butea monosperma.
Similar to east direction, the population structure of tree species in west direction (Fig. 4) revealed that there is no single species which represent the older size classes (D) and (E). Some species reflected only seedling size class (A) exemplified by Acacia catechu, Butea monosperma, and Eucalyptus spp. The young size classes (A) and (B) were represented by Acacia mangium and Tectona grandis while the subsequent class was absent. Sygygium cumini have sapling (B) and young tree class (C) and is devoid of seedling class (A). Casia siamea and Peltophorum ferrugineum were represented by size classes (A), (B), and (C).
In the north direction, seedling class (A) was represented by all the species while the older classes (D) and (E) were totally absent (Fig. 5). The tree class (C) was represented by Acacia mangium, Cassia siamea, Eucalyptus spp., and Peltophorum ferrugineum. The sapling class (B) was represented by each species except Azadirecta indica which were found only in seedling stage.
In the south direction (Fig. 6), most of the species representing single size class only exemplified by Acacia catechu, Butea monosperma, Cassia fistula, and Syzygium cumini. Acacia mangium and Casia siamea represented by size classes (A), (B), and (C), while the older size classes (D) and (E) were found to be totally absent.
Population structures of tree species in south aspect of collieries. a Shannon diversity indices at various directions. b Simpson diversity indices at various directions. c Species richness at various directions. d Equitability indices at various directions. e Variation in beta diversity at various directions
The three life stages (seedlings, saplings, and trees) for different species suggested their possible future status in the given site. The diameter class gradation or distribution of trees has been the significant criteria for assessing the population structure of forests or stand (Saxena and Singh 1984; Khan et al. 1987, Dhaulkhandi et al. 2008; Jhariya et al. 2012). In all the concerned study sites of collieries, it showed that the individuals with small girth class A (<10 cm) were high. A greater population of individuals in lower size classes compared to larger classes as the structure represents frequent reproduction (Knight 1975; West et al. 1981). Due to this heavy stress, the species could not reach in the tree or sapling layer as a dominant species. The hump in the middle size classes may indicate comparatively fast growth or less mortality in individuals. The prevailing habitat conditions in these areas have reduced the chances of regeneration of many species, reducing the number of species in the mined areas (Sarma et al. 2010) once they successfully crosses the sapling layer and attained the first tree size class. According to West et al. (1981), such types of patterns indicate the heavy exploitation of older individuals and greater mortality among young individuals. This species has produced abundant population in the past with better conversion rate from one size class to another but at present, though the seedlings are not comings up frequently, the species might have produced the seeds but, the environment is not supporting their proper establishment.
According to Saxena and Singh (1984), the concentration of individuals in intermediate size classes with general absence or negligible representation of individuals both towards higher and lower size classes reflects the degenerative nature of the concerned population. Khurana (2007) concluded that biotic disturbances impose the site nutrient poor, and top soil erosion makes the inadequate regeneration pattern. Regeneration of a species both depends upon internal forest process and exogenic disturbance (Barker and Kirk Patrick 1994). Local extinction in the forest stands is reflected by the abundance or absence of adult individual or very low population of seedlings and saplings (Dalling et al. 1998). The density values of seedlings and saplings are considered as regeneration potential of the species. Suitability of a species to a given environment is reflected through the presence of good regeneration potential (Dhaulkhandi et al. 2008). Climatic factors and biotic interference influence the regeneration of different species in the vegetation. Higher seedling density values get reduced to sapling due to biotic disturbance and competition for space and nutrients. The greater tree diversity and density towards north direction could be due to the presence of a large number of young individuals in the concerned sites whereas tree diversity declined in other directions due to logging activity during mining and dumping of coal.
Consequences on vegetational diversity
Diversity is considered to be an heterogeneous nature of the gene pool prevailing over a particular area. It is admixture of synthetic measure of the structure, complexity, and stability of a community (Hubble and Foster 1983). It is reflected in two ways, the number of species present, referred to as species richness and the distribution of individuals among species, referred to as species evenness or equability. Species diversity therefore, refers to the variation that exists among the different life forms.
During our present investigation, diversity indices (Fig. 7) of tree species reflected significant level of variation among various sites. Shannon index in different directional sites ranged up to 1.91, Simpsons index from 0.28–0.5, species richness from 0.27–0.61, equitability up to 1.44, and beta diversity from 2.00–4.00, respectively. For sapling layer, the value of Shannon index in different sites varied up to 2.33, Simpsons index 0.22–0.61, species richness up to 0.70, equitability up to 1.36, and beta diversity 1.66–2.50, respectively. In seedling layer, Shannon index varied from 0.64 to 2.53, Simpsons index 0.08–2.20, species richness 0.26–0.71, equitability 0.58–1.30, and beta diversity 1.71–4.00, respectively. Shannon index for herb layer in different directional sites ranged between 4.07 and 4.52, Simpsons index from 0.06 to 0.08, species richness from 1.73 to 2.33, equitability from 1.27 to 1.32, and beta diversity from 1.5 to 2.0, respectively. Shrub layer revealed that the Shannon index values in different directional sites varied from 0.57 to 2.10, Simpsons index from 0.31 to 0.77, species richness from 0.14 to 0.79, equitability from 0.82 to 1.34, and beta diversity from 1.28–4.50, respectively.
During the present investigation, Shannon index value was found to be lower in case of sapling, seedling, and herb towards east direction, and minimum diversity was recorded for tree and shrub species towards south direction. This may be due to lower rate of evolution and diversification of communities (Simpson 1964) and severity in environment (Connel and Oris 1964). The east direction represents less herbaceous vegetation as compared to other directions because of reduction in competition for space and resources.
Sarma et al. (2010) reported that the Shannon diversity index for tree and shrub species was low in mined areas as compared to unmined area. However, diversity index of ground vegetation did not differ among the mined and unmined areas. Simpson’s dominance index shows a reverse trend to that of diversity index. Sobuj and Rahman (2011) compared the plant diversity of natural forest and plantations of Rema-kalenga wildlife sanctuary of Bangladesh and reported that Shannon index were 2.70, 2.72, and 3.12 for trees, shrubs, and herbs, respectively, in the natural forest. However, it was 2.35 for tree species, 2.31 for shrub species, and 2.81 for herb species in the plantation forest.
Study of vegetational structure and composition provides necessary ecological information for various ecosystems along with their ecological health status (Silver et al. 2004; Wang et al. 2004). As large forest have been clear felled in the Bishrampur for opencast coal mining, the major challenges include top soil loss, forest cover reduction, and habitat destruction, essential for maintaining nutrient cycling and ecological balance. Soil stratification, reduced biotic diversity, and alteration of structure and functioning of ecosystems are the major environmental impacts associated with mining (Almas et al. 2004; Ghose 2004). The habitat conditions in these sites have reduced the chances of regeneration of many species, thereby reducing the presence of higher number of species in this region. Sarma et al. (2010) reported that the number of tree species was low in all the mining sites due to various activities during the mining as compared to adjacent unmined area. Majority of species showed contagious distribution pattern in the unmined and mined areas. The tree density was more (1040 stems ha−1) in the unmined area than the mined areas (515 and 646 stems ha−1). The trees of medium girth class contributed maximum stand density in the mined areas, while in the unmined site, the trees of low girth class contributed to the maximum stand density. Hashemi (2010) investigated the plant species diversity in north of Iran and reported a total number of 114 plant species, belonging to 76 genus and 31 families that were gathered in the region of study. Oraon et al. (2015) reported that natural regeneration potential of shrubs was more in disturbed site as compared to undisturbed site.
Suitable environment promotes optimum distribution of species along with its adaptive potential. Distribution pattern of species may be regulated by habitat condition and biological interaction. A/F ratio was used to assess the distribution pattern of the species. In the collieries, it was found that most of the species were distributed contagiously and randomly, whereas, a few species distributed regularly. This can be attributed to the interaction of various factors that are acting together on the population. Inefficient mode of seed dispersal is reflected through clumping nature of distribution (Richards 1996). Comparison of dispersal pattern of trees in tropical and temperate forest of the world, Armesto et al. (1986) recommended prevalent contagious distribution of species in the natural forest, may be attributed by clumping. Odum (1971) emphasized that contagious distribution is the most prevalent pattern observed in nature. Likewise, Kumar et al. (2015), Sinha et al. (2015), Oraon et al. (2014), Kittur et al. (2014), and Jhariya (2014) described that most of the recorded species followed contagious and random distribution pattern, whereas regular distribution was found almost negligible or insignificant.
Disturbance regimes strongly influence biodiversity of an area. Species diversity is the key factor of ecosystems and its functioning (Scherer-Lorenzen et al. 2005). The species diversity reflects trophic relationship and possibilities of invasion (Nicholas and Nicholas 2003). Species diversity in this mining landscape often fluctuates to a considerable amount due to various biotic changes and abiotic factors.
Impact on litter biomass
Forest floor biomass in different site of mining areas (east, west, north, and south direction) was calculated and presented in Table 6. Total forest floor biomass was 4.20 t/ha in east direction. 95.23% of the forest floor biomass comprises of leaf litter and remaining (4.77%) by wood litter. In the west direction, the total forest floor biomass was 5.30 t/ha, of which 90.5% was contributed by leaf litter and remaining (9.5%) by wood litter. In the north direction, the total forest floor was 4.91 t/ha, of which 72.8% was represented by leaf litter and remaining (27.2%) by wood litter. Similarly in the south direction, the total forest floor biomass was 5.65 t/ha, of which 64.51% shared by leaf litter and remaining (35.49%) by wood litter, respectively. The average values of litter biomass across the sites were 4.22 t/ha for leaf, 0.78 t/ha for wood, and 5.02 t/ha total litter mass.
Disturbances have various impact upon vegetation and its dynamics. This includes changes in the structure, composition, their litter production capacity, accumulation, and decomposition rate of tropical forests (Xuluc-Tolosa et al. 2003; Dezzeo et al. 2004; Yadav et al. 2008). Anthropogenic factors significantly influence accumulation of organic matter in the forest floor (Jordan 1985; Aguiar et al. 2010). Quantifying litter fall rate and quality describing any change are due to natural as well as anthropogenic disturbances (Sizer et al. 2000). The value measured for litter biomass was comparable to 3.00–10.00 t ha−1 for tropical forest as previously reported by Murphy and Lugo (1986). The litter biomass for tropical forests ranged between 0.8 and 15.3 t ha−1 (Vitousek 1984) and 4.88 to 6.71 t ha−1 of dry tropical forest (Singh and Singh 1991). Sahu et al. (2013) reported the forest floor biomass in different sites varied from 2.19 to 2.66 t ha−1. Lower levels of fresh leaf litter were found in highly disturbed site. In comparison, fresh leaf litter content appeared to be of same level in the least and moderately disturbed sites. The wood litter ranged from 0.70 to 1.03 t ha−1 in different sites (Pawar et al. 2014). Strategies for eco-restoration may include plantation of well adapted species to the local condition of degraded land. Locally adapted species may promote high litter deposition and decomposition rates and therefore improving the soil health (Kumar et al. 2010).
From our results, it appears that in Bishrampur collieries, soil quality can be effectively restore through addition of organic matter into the soil through addition of litter biomass. Decomposition of litter biomass leads to gradual build-up of nutrient pool in the soil ecosystem (Jhariya 2017). Therefore, litter biomass acts as a effective recycling agent of nutrient under natural condition. More litter is added to the soil, more probability of development of better quality of soil can be achieved. From this perspective, litter biomass assessment is very much significant in restoration studies.
Statistical analysis
From the correlation study, it was observed that tree layer density, frequency is significantly correlated with important value index. Density was positively correlated with frequency. A/F ratio is positively correlated with IVI (Table 7). For sapling layer, density is positively correlated with IVI and basal area. Frequency was found to be positively correlated with IVI and basal area. Basal area was also positively correlated with IVI (Table 8). In shrub layer, almost similar trend was observed for the phytosociological parameters (Table 9). For seedling and herb layer, density and important value index were found to be significantly and positively correlated (Tables 10 and 11).
Two-way cluster analysis was done for the different stratified layer of the vegetation surrounding Bishrampur collieries. In tree layer (Fig. 8), north and west direction framed the smaller cluster, and three major clusters were formed among different species. Cluster 1 comprises of Cassia siamea and Acacia mangium, cluster 2 comprises of Peltophorum ferrugineum and Syzgium cumini, and cluster 3 comprises of Ailanthus excelsa and Butea monosperma, respectively. In sapling layer (Fig. 9), closely spaced clustering of vegetation was observed for north and west direction. Two major clusterings of species were observed in case of sapling layer on the basis of vegetation characteristics. Cluster 1 comprises of Eucalyptus sp., Pongamia pinnata, and Peltophorum ferrugineum, and cluster 2 comprises of Acacia catechu, Tectona grandis, Syzgium cumini, and Saccopetalum tomentosum, respectively. For the seedling layer (Fig. 10), two smaller clusters were framed between north and west direction, east and south direction, respectively. Four major clusterings of species were observed for the seedling layer. Azadirachta indica, Pongamia pinnata, and Peltophorum ferrugineum formed the cluster1, cluster 2 comprises of Cassia fistula and Syzygium cumini, cluster 3 comprises of Tectona grandis and Acacia catechu, cluster 4 comprises of Butea monosperma and Dalbergia sissoo. Cluster analyses of the herb layer (Fig. 11) represent numerous cluster formations due to occurrence of higher number of species. In shrub layer (Fig. 12), east and west direction formed smallest cluster, and the vegetational type reflected distant clustering towards the south direction when compared to other directions. Smaller clusters were formed between Cassia angustifolia and Ipomea carnea, Grewia rothi, Leea asiatica, and Calotropis gigantea, Carissa opaca, and Zizypus rotundifolia. Vegetation stratification studies were similarly reported by Palit and Banerjee (2013) in Senchal wildlife sanctuary, W.B. in India which reflected clustering of plant species in different beats of concerned forest area.
Principle component analysis of the phytosociological attributes of vegetation types of Bishrampur collieries reflected significant level of variation among themselves in various stratified layer of vegetation. Similar studies on PCA analysis of vegetation parameters were reported by Eni et al. (2012) in order to establish the inter-relationship between soil and vegetation of forest of Nigeria. Such studies act as fact finding tool which helps to reduce the complexity in data analysis as well as helps to screen out variables which have made significant contribution in the results. Bi-plot PCA-ordination diagram was plotted for the different phytosociological attributes of plant species belonging to various stratified layer of vegetation. For the tree layer (Table 12 and Fig. 13), the first two principle components account for 91.06% of the total variance in the data set. Therefore, 69 and 21% of variance were accounted for the first and second principle components, respectively. This means that the first principle component is far by most important for representing the variation within the phytosociological attributes in the tree layer for the concerned study sites. In the study area, phytosociological attributes in Ailanthus excelsa and Butea monosperma have similarity in comparison to other tree species. These two species as reflected from the four quarter diagram, both have high correlation with first axis. Therefore, these species reflects strong relation with the variables of the first axis. Acacia mangium and Cassia siamea reflected inverse correlation for the same phytosociological variables of the first axis. Acacia catechu showing positive correlation with A/F ratio.
In sapling layer (Table 13 and Fig. 14), first two principle components account for 99.209% of the total variance in the data set. Therefore, 87.138 and 12.071% of variance were accounted for the first and second principle components, respectively. This means that the first principle component is far by most important for representing the variation within the phytosociological attributes in the sapling layer for the concerned study sites. Due to higher distance between Acacia mangium type from the second axis, this species has a weak relation with factors such as A/F ratio. Butea monosperma, Tectona grandis, Peltophorum ferrugianum, and Eucalyptus spp. showed negative correlation with respect to A/F ratio in axis 2. Syzygium cumini reflected positive correlation with A/F ratio in axis 2.
In seedling layer (Table 14 and Fig. 15), first two principle components account for 99.389% of the total variance in the data set. Therefore, 90.626 and 8.763% of variance were accounted for the first and second principle components, respectively. This means that the first principle component is far by most important for representing the variation within the phytosociological attributes in the sapling layer for the concerned study sites. Due to higher distance between Acacia mangium type from the second axis, this species has a weak relation with factors such as frequency. Butea monosperma, Azadirachta indica, Tectona grandis, and Pongamia pinnata being located on axis 2 thereby reflecting strong negative correlation.
For the herb layer (Table 15 and Fig. 16), components 1 and 2 reflected higher level of variation equally. Therefore, both the components accounted for 75.874% of variance. Most of the herb species were located on or near of axis 1 and 2. This, therefore, reflects the strong positive correlation of the concerned species along with the study variable represented by the respective axis.
Principle component analysis for shrub layer (Table 16 and Fig. 17) reflects variable scenario with respect to herb layer. Zizyphus rotundifolia and Vitex negundo showed higher correlation for axis 2. These species therefore, are strongly correlated with IVI. Component 1 accounted for 99.908% of variance.
Management perspectives
Eco-restoration of degraded habitat is a major challenge towards sustainable development. Plantation is the most suitable strategy among various practices available in eco-restoration program. Plantations promote biological reclamation of mine spoil. For plantation, priority must be given to native species, those having fast growth rate and ability to fix atmospheric nitrogen. Scenario in the mining sector often reflects ill management of the concerned habitat. The features of the site and its subsequent management policies are the most important aspects for eco-restoration and eco-development. As per the rule of ecological interactions, diverse nature of species imposes more stability to the habitat. These should be given due consideration while going for plantation in the coal waste piles and promote mixed plantation (tree, shrub, and grasses) instead of mono species plantation (Li et al. 2006b). Proper management practices should be applied for the betterment of the site. During our present investigation, data revealed natural regenerative potential of Dalbergia sissoo, Syzygium cumini, and Butea monosperma in concerned study sites at various growth stages. Therefore, such indigenous species should be promoted for plantation purpose with the aim of eco-restoration of mined wastelands. Our findings were further supported by Harrington (1999)) who mentioned that indigenous species are much more preferable in comparison to exotics due to their local adaptation of the extreme conditions (Wong 2003; Gonzalez and Gonzalez-Chavez 2006; Mendez and Maier 2008; Ghose 2004; Maiti 2007; Juwarkar and Jumbalkar 2008). Previous study suggests that vegetal cover introduction in the mining areas may promote rhizo-deposition of toxic elements. Future research and development activities are required for application of suitable technology from lab to land scale. Banning of mining in eco-sensitive areas should be discouraged always. In our present study, directional impact on population structure is clearly revealed through statistical interpretation of data. Different girth classes are prevalent in different direction. Selection of suitable species on the basis of high growth rate, regeneration capacity, and stable population structure needs to be emphasized for eco-restoration of mined wastelands.
Conclusion
Surface mining of coal in Bishrampur and adjoining areas has led to significant changes in the land-use pattern. It also promoted alteration of soil quality through higher accumulation of mine spoils. The effect was significant in all the four directions during present investigation. The degradation of habitat has reduced regeneration potential of indigenous plant population. There was predominance of herbaceous species in comparison to other vegetative strata which suggest that mining operations lead to colonization of specific species in newly altered habitat. The study further suggests detrimental impact of mining activities on vegetation on an area. Therefore, such activities need to be regulated properly. Improvement in the site quality is the key prerequisite from management perspective of mined wastelands. Further, regular monitoring and appropriate regulations are indispensable to alleviate the negative effects of the mining upon ecology and environment to make this practice a sustainable one. The major findings of the present investigation include screening of Acacia mangium, Cassia siamea, and Dalbergia sissoo species to be recommended for effective eco-restoration of the concerned sites with due consideration of cosmopolitan distribution in the study sites, high regeneration potential, as well as existence in the form of various girth classes of stable population structure.
References
Aguiar, A. C. F., Bicudo, S. J., Sobrinho, J. R. S. C., Martins, A. L. S., Coelho, K. P., & Moura, E. G. (2010). Nutrient recycling and physical indicators of an alley cropping system in a sandy loam soil in the pre-Amazon region of Brazil. Nutrient Cycling in Agroecosystems, 86, 189–198.
Almas, A. R., Bakken, L. R., & Mulder, J. (2004). Changes in tolerance of soil microbial communities in Zn and Cd contaminated soils. Soil Biol Bioch, 36, 805–813.
Armesto, J. J., Mitchell, J. D., & Villagran, C. (1986). A comparison of spatial pattern of trees in some tropical and temperate forests. Biotropica, 18, 1–11.
Bahrami, A., Emadodin, I., Atashi, M.R., & Bork, H.R. (2010). Land-use change and soil degradation: A case study, North of Iran. Agriculture and Biology Journal of N. America, 605.
Barker, P. C. J., & Kirk Patrick, J. B. (1994). Phyllocladus asplenifolius: variability in the population structure of the regeneration niche and dispersion pattern in Tasmanian forest. Australian Journal of Botany, 42, 163–190.
Chaturvedi, R. K., Raghubanshi, A. S., & Singh, J. S. (2011). Carbon density and accumulation in woody species of tropical dry forest in India. Forest Ecology and Management, 262, 1576–1588.
Connel, J. H., & Oris, E. (1964). The ecological regulation of species diversity. The American Naturalist, 48, 399–414.
Corbett, E. A., Anderson, R. C., & Rodgers, C. S. (1996). Prairie revegetation of a strip mine in Illinois: fifteen years after establishment. Restoration Ecology, 4, 346–354.
Curtis, J. T., & McIntosh, R. P. (1950). The interrelations of certain analytic and synthetic phytosociological characters. Ecology, 31, 434–455.
Dalling, J. W., Hubbel, S. P., & Silvera, K. (1998). Seed dispersal, seedling establishment and gap partitioning among tropical pioneer trees. Journal of Ecology, 86, 674–689.
Dezzeo, N., Chacon, N., Sanoja, E., & Picon, G. (2004). Changes in soil properties and vegetation characteristics along a forest savanna gradient in southern Venezuela. Forest Ecology and Management, 200, 183–193.
Dhaulkhandi, M., Dobhal, A., Bhatt, S., & Kumar, M. (2008). Community structure and regeneration potential of natural forest site in Gangotri, India. Journal of Basic and Applied Sciences, 4(1), 49–52.
Donggan, G., Zhongke, B., Tieliang, S., Hongbo, S., & Wen, Q. (2011). Impacts of coal mining on the aboveground vegetation and soil quality: a case study of Qinxin coal mine in Shanxi province, China. Clean-Soil Air Water, 39(3), 219–225.
Eni, D. D., Iwara, A. I., & Offiong, R. A. (2012). Analysis of soil-vegetation interrelationship in a south-southern secondary forest of Nigeria. International Journal of Forestry Research, 2012, 1–8. doi:10.1155/2012/469326.
Ghose, M. K. (2004). Effect of opencast mining on soil fertility. J Sci Indust Res, 63, 1006–1009.
Gonzalez, R. C., & Gonzalez-Chavez, M. C. A. (2006). Metal accumulation in wild plants surrounding mining wastes: soil and sediment remediation (SSR). Environmental Pollution, 144, 84–92.
Guha, D. (2014). A case study on the effects of coal mining in the environment particularly in relation to soil, water and air causing a socio-economic hazard in Asansol-Raniganj area, India. Int Res J Social Sci., 3(8),39–42.
Gupta, B., & Sharma, N. (2015). Plant asemblages alongs an altitudinal gradient in northwest Himalaya. Journal of Forest and Environmental Science, 31(2), 91–108.
Harrington, C. A. (1999). Forests planted for ecosystem restoration or conservation. New Forest, 17, 175–190.
Hashemi, S. A. (2010). Investigation plant species diversity and physiographical factors in mountain forest in north of Iran. Journal of Forest Science, 26(1), 1–7.
Hassan, A., Wahab, R., Alias, M. A., & Salim, R. M. (2007). Growth performance of 9-year-old selected 5 indigenous wood species planted on degraded forest land. International Journal of Agricultural Research, 2, 302–306.
Hewit, N., & Kellman, M. (2002). True seed dispersal among forest fragments: dispersal ability and biogeographical controls. Journal of Biogeography, 29(3), 351–363.
Hooker, J. D. (1875). Flora of British India Vol. I-VII. England: L. Reeve and Co. Ltd..
Hubble, S.P., & Foster, R.B. (1983). Diversity of canopy trees in a neo-tropical forest and implications to conservation. Tropical Rain Forest: Ecol. Manage., (Sutton L.T.C).
Jhariya, M. (2014). Effect of forest fire on microbial biomass, storage and sequestration of carbon in a tropical deciduous forest of Chhattisgarh. Ph.D. Thesis, I.G.K.V., Raipur (C.G.), pp. 259.
Jhariya, M.K. (2017). Influences of Forest Fire on Forest Floor and Litterfall in Bhoramdeo Wildlife Sanctuary (C.G.), India. Journal of Forest and Environmental Science (In Press).
Jhariya, M. K., Bargali, S. S., Swamy, S. L., & Kittur, B. (2012). Vegetational structure, diversity and fuel loads in fire affected areas of tropical dry deciduous forests in Chhattisgarh. Vegetos, 25(1), 210–224.
Jhariya, M. K., Bargali, S. S., Swamy, S. L., & Oraon, P. R. (2013). Herbaceous diversity in proposed mining area of Rowghat in Narayanpur District of Chhattisgarh, India. Journal of Plant Development Sciences, 5(4), 385–393.
Jhariya, M. K., Bargali, S. S., Swamy, S. L., Kittur, B., Bargali, K., & Pawar, G. V. (2014). Impact of forest fire on biomass and carbon storage pattern of tropical deciduous forests in Bhoramdeo wildlife sanctuary, Chhattisgarh. International Journal of Ecology and Environmental Sciences, 40(1), 57–74.
Jhariya, M. K., Kittur, B. H., & Bargali, S. S. (2016). Assessment of herbaceous biomass: a study in Rowghat mining areas (Chhattisgarh), India. Journal of Applied and Natural Science, 8(2), 645–651.
Jordan, C.F. (1985). Nutrient cycling in tropical forest ecosystems. Wiley. Chichester. UK. p. 190.
Juwarkar, A. A., & Jumbalkar, H. P. (2008). Phytoremedia-tion of coal mine spoil dump through integrated biotechnological approach. Bioresource Technology, 99, 4732–4741.
Khan, M. L., Rai, J. P. N., & Tripathi, R. S. (1987). Population structure of some tree species in disturbed and protected sub-tropical forests of north East India. Acta Oecologica, 8, 247–255.
Khumbongmayum, A. D., Khan, M. L., & Tripathi, R. S. (2006). Biodiversity conservation in sacred groves of Manipur, Northeast India: population structure and regeneration status of woody species. Biodiversity and Conservation, 15, 2439–2456.
Khurana, P. (2007). Tree layer analysis and regeneration in tropical dry deciduous forest of Hastinapur. Indian Forester, 16(1), 43–50.
Kittur, B., Swamy, S. L., Bargali, S. S., & Jhariya, M. K. (2014). Wildland fires and moist deciduous forests of Chhattisgarh, India: divergent component assessment. Journal of Forestry Research, 25(4), 857–866.
Knight, D. H. (1975). A phytosociological analysis of species-rich-tropical forest on Barro Colorado Island, Panama. Ecological Monographs, 45, 259–284.
Kumar, J. I. N., Sajish, P. R., Kumar, R. N., & Bhoi, R. K. (2010). Wood and leaf litter decomposition and nutrient release from Tectona grandis linn. F. in a tropical dry deciduous forest of Rajasthan, western India. Journal of Forest Science, 26(1), 17–23.
Kumar, N., Kumar, A., & Singh, M. (2014). Floristic diversity assessment in ecologically restored limestone (building stone) mine near Chechat village, Kota district, Rajasthan. Ecología, 4(1), 16–25.
Kumar, A., Jhariya, M. K., & Yadav, D. K. (2015). Community characters of herbaceous species in plantation sites of coal mine. Journal of Plant Development Science, 7(11), 809–814.
Kumar, A., Jhariya, M. K., & Yadav, D. K. (2016). Vegetation dynamics in plantation sites of collieries. Nature, Environment and Pollution Technology, 15(4), 1285–1291.
Li, P. B., Hu, Z. Q., Wu, J., Zhao, Y. L., & Zang, Z. R. (2006b). Study on model of vegetation rehabilitation technique on coal waste pile. Journal of Shandong Forestry Science and Technology, 4, 13–15.
Magurran, A. E., & Henderson, P. A. (2003). Explaining the excess of rare species in natural species abundance distributions. Nature, 422, 714–716.
Maiti, S. K. (2007). Bioremediation of coalmine overburden dumps with special emphasis in micronutrients and heavy metals accumulation in tree species. Environmental Monitoring and Assessment, 125, 111–122.
Marglef, D. R. (1958). Information theory in ecology. General Systems Yearbook, 3, 36–71.
Mendez, M. O., & Maier, R. M. (2008). Phytostabilization of mine tailings in arid and semiarid environments—an emerging remediation technology. Environmental Health Perspectives, 116, 278–283.
Mertens, J., Van Nevel, L., De Schrijver, A., Piesschaert, F., Oosterbean, A., Track, F. M. G., & Verheyen, K. (2007). Tree species effect on the redistribution of soil metals. Environmental Pollution, 149(2), 173–181.
Milder, A.I., Fernandez-Santos, B., & Martinez-Ruiz, C. (2011). Colonization patterns of woody species on lands mined for coal in Spain: Preliminary insights for forest expansion. Land Degradation & Development, DOI: 10.1002/Idr.1101.
Mishra, B. P., Tripathi, O. P., & Laloo, R. C. (2005). Community characteristics of a climax subtropical humid forest of Meghalaya and population structure of ten important tree species. Tropical Ecology, 46, 241–251.
Mishra, R. K., Upadhyay, V. P., & Mohanty, R. C. (2008). Vegetation ecology of the Similipal Biosphere reserve, Orissa, India. Journal of Applied Ecology and Environmental Research, 6(2), 89–99.
Mohapatra, H., & Goswami, S. (2012). Impact of coal mining on soil characteristics around Ib river coalfield, Orissa, India. Journal of Environmental Biology, 33, 751–756.
Murphy, P. G., & Lugo, A. E. (1986). Ecology of tropical dry forests. Annual Review of Ecology and Systematics, 17, 67–88.
Nicholas, O. G., & Nicholas, F. M. (2003). Long-term trends in faunal recolonization after bauxite mining in the jarrah forest of south-western Australia. Restoration Ecology, 11, 261–272.
Odum, E. P. (1971). Fundamental of ecology. Philadelphia: Saunders Co..
Oraon, P.R. (2012). Structure and dry matter dynamics along the disturbance gradient of tropical dry deciduous forest in Bhoramdeo Wildlife Sanctuary, Chhattisgarh. Ph.D. Thesis, I.G.K.V., Raipur (C.G.).
Oraon, P. R., Singh, L., & Jhariya, M. K. (2014). Variations in herbaceous composition of dry tropics following anthropogenic disturbed environment. Current World Environment, 9(3), 967–979.
Oraon, P. R., Singh, L., & Jhariya, M. K. (2015). Shrub species divesity in relation to anthropogenic disturbance of Bhoramdeo wildlife sanctuary, Chhattisgarh. Environment and Ecology, 33(2A), 996–1002.
Oyun, M. B., Bada, S. O., & Anjah, G. M. (2009). Comparative analysis of the floral composition at the edge and interior of Agulii Forest reserve, Cameroon. Journal of Biological Sciences, 9(5), 431–437.
Palit, D., & Banerjee, A. (2013). Species diversity and pedological characteristics in selected sites of Senchal wildlife sanctuary, West Bengal, India. Journal of Environment and Ecology, 4(1), 111–137.
Pascal, L. P., & Pellissier, R. (1996). Structure and floristic composition of a tropical Evergreen Forest in south-West India. Journal of Tropical Ecology, 12, 191–210.
Pawar, G. V., Singh, L., Sarvade, S., & Lal, C. (2014). Litter production and soil physico-chemical properties influenced by different degraded sites of tropical deciduous forest, Chhattisgarh, India. The Ecoscan, 8(3&4), 349–352.
Phillips, E.A. (1959). Methods of vegetation study. Holt R and Winston New York USA. pp. 105.
Pielou, E. C. (1966). Species diversity and pattern diversity in the study of ecological succession. Journal of Theoretical Biology, 10, 370–383.
Pullaiah, T. (2006). Encyclopedia of world medicinal plants. New Delhi: Regency publication.
Rahman, H., Khan, M. A. S. A., Fardusi, M. J., & Roy, B. (2010). Status, distribution and diversity of invasive forest undergrowth species in the tropics: a study from northeastern Bangladesh. Journal of Forest Science, 26(3), 149–159.
Rai, R. K. (2002). Implication of coal mining on environmental in Jaintia Hills, Meghalaya. In P. M. Passah & A. K. Sarma (Eds.), Jaintia Hills, a Meghalaya tribe: Its environment (pp. 113–119). New Delhi: Land and People. Reliance Publishing House.
Raunkiaer, C. (1934). The Life Form of Plants and Statistical Plant Geography. Claredon Press ISBN 9978–40–943-2, Oxford.
Richards, P. W. (1996). The tropical Rain Forest: an ecological study (2nd ed.). London: Cambridge University Press.
Robinson, G. R., & Handel, S. N. (2000). Directing spatial patterns of recruitment during an experimental urban woodland reclamation. Ecological Applications, 10, 174–188.
Sadhu, K., Adhikari, K., & Gangopadhyay, A. (2012). Effect of mine spoil on native soil of lower Gondwana coal fields: Raniganj coal mines areas, India. International Journal of Enviromental Sciences, 2(3), 1675–1687.
Sahu, K. P., Singh, L., & Jhariya, M. K. (2013). Fine root biomass, forest floor and nutrient status of soil in an age series of teak plantation in dry tropics. The Bioscan, 8(4), 1149–1152.
Sarma, K., Kushwaha, S. P. S., & Singh, K. J. (2010). Impact of coal mining on plant diversity and tree population structure in Jaintia Hills district of Meghalaya, north East India. New York Science Journal, 3(9), 79–85.
Saxena, A. K., & Singh, J. S. (1984). Tree population structure of central Himalayan forest associations and implications concerning their future composition. Vegetatio, 54, 61–69.
Scherer-Lorenzen, M., Korner, C., & Schulze, E. D. (2005). Forest diversity and function: temperate and boreal systems (p. 399). Berlin: Springer.
Shannon, C. E., & Weaver, W. (1963). The mathematical theory of communication (323). Urbana: University of Illinois Press.
Silver, W. L., Kueppers, L. M., Lugo, A. E., Ostertag, R., & Matzek, V. (2004). Carbon sequestration and plant community dynamics following reforestation of tropical pasture. Ecol. Apli., 14, 1115–1127.
Simpson, E. H. (1949). Measurement of diversity. Nature, 163, 688.
Simpson, G. G. (1964). Species diversity of north American recent mammals. Systematic Zoology, 13, 57–73.
Singh, L., & Singh, J. S. (1991). Species structure, dry matter dynamics and carbon flux of a dry tropical forest in India. Annals of Botany, 68, 263–273.
Singh, A. N., Raghubanshi, A. S., & Singh, J. S. (2004a). Impact of native tree plantations on mine spoil in a dry tropical environment. Forest Ecology and Management, 187, 49–60.
Singh, A. N., Raghubanshi, A. S., & Singh, J. S. (2004b). Comparative performance and restoration potential of two Albizia species planted on mine spoil in a dry tropical region, India. Ecological Engineering, 22, 123–140.
Singh, A.P., Chowdhury, T. and Gupta, S. (2010). Handbook on weeds of Chhattisgarh. IGKV/pub/2010/06, pp 1–146.
Singh, P. K., Imam, A., Singh, R., Singh, D., & Sharma, S. (2013). A study about ecological imbalance in Surguja (India) coalfield area due to mining. Int. Res. J. Environment Sci., 2(4), 10–14.
Singh, R. B. P., Singh, A., & Choudhary, S. K. (2014). Impact of opencast coal mining on the quality of surfacewater, groundwater and vegetation: a case study in Simlong coalfield, Sahibganj, Jharkhand. International Journal on Emergring Technologies, 5(2), 95–105.
Sinha, R., Jhariya, M. K., & Yadav, D. K. (2015). Assessment of Sal seedlings and herbaceous flora in the Khairbar plantation of Sarguja Forest division, Chhattisgarh. Current World Environment, 10(1), 330–337.
Sizer, N. C., Tanner, E. V. J., & Kossmann Ferraz, I. D. (2000). Edge effects on litterfall mass and nutrient concentrations in forest fragments in Central Amazonia. J. Tropical Ecology, 16, 853–863.
Sobuj, N. A., & Rahman, M. (2011). Comparison of plant diversity of natural forest and plantations of Rema-Kalenga wildlife sanctuary of Bangladesh. Journal of Forest Science, 27(3), 127–134.
Swain, B. K., Goswami, S., & Das, M. (2011). Impact of mining on soil quality: a case study from Hingula opencast coal mine, Angul district, Orissa. Vistas in Geolo. Res, 10, 77–81.
Tom-Dery, D., Dagben, Z. J., & Cobbina, S. J. (2012). Effect of illegal small-scale mining operations on vegetation cover of arid northern Ghana. Research Journal of Environmental and Earth Sciences, 4(6), 674–679.
Tripathi, B. C., Rikhari, H. C., Bargali, S. S., & Rawat, Y. S. (1991). Species composition and regeneration in disturbed forest sites in the oak zone in and around Nainital. Proceedings of Indian National Science Academy B, 57, 381–390.
Vitousek, P. M. (1984). Litterfall, nutrient cycling and nutrient limitation in tropical forests. Ecology, 65, 285–298.
Wang, J., Borsboom, A. C., & Smith, G. C. (2004). Flora diversity of farm forestry plantations in southeast Queensland. Ecological Management & Restoration, 5, 43–51.
Weidelt, H. J. (1988). On the diversity of tree species in tropical Rain Forest Eosystems. Plant Research and Development, 28, 110–125.
West, D. C., Shugart Jr., H. H., & Ranney, J. W. (1981). Population structure of forest cover in a large area in the boreal Swedish forest. Forest Science, 27, 701–710.
Whiteford, P. B. (1949). Distribution of woodland plants in relation to succession and colonal growth. Ecology, 30, 199–200.
Whittaker, R. H. (1972). Evolution and measurement of species diversity. Taxon, 21, 213–251.
Wong, M. H. (2003). Ecological restoration of mine degraded soils, with emphasis on metal contaminated soils. Chemosphere, 50, 775–780.
Xuluc-Tolosaa, F. J., Vester, H. F. M., Ramirez-Marcial, N., Castellanos-Albores, J., & Lawrence, D. (2003). Leaf litter decomposition of tree species in three successional phases of tropical dry secondary forest in Campeche, Mexico. Forest Ecology and Management, 174, 401–412.
Yadav, R. S., Yadav, B. L., & Chhipa, B. R. (2008). Litter dynamics and soil properties under different treespecies in a semi-arid region of Rajasthan, India. Agroforestry Systems, 73, 1–12.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, A., Jhariya, M.K., Yadav, D.K. et al. Vegetation dynamics in Bishrampur collieries of northern Chhattisgarh, India: eco-restoration and management perspectives. Environ Monit Assess 189, 371 (2017). https://doi.org/10.1007/s10661-017-6086-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-017-6086-0