Abstract
This paper focuses on characterizing the current status of physiochemical properties of Mujib Dam sediments. Five types of granulometric textural facies were observed for the bottom sediments of Mujib reservoir bed; these are clayey facies, clayey-silt facies, sand-silt-clay facies, sand facies, and granule facies. This average grain size will likely play a vital role in adsorption-desorption of the majority trace metals to the reservoir lake. Other sediment parameters including the total averages were 5.9% (total organic matter (TOM)), 7.5 (pH), 25.8% (CaCO3), and 88.0 meq/100 g (cation exchange capacity), with dominant mineralogical constituents of quartz, calcite, dolomite, and minor feldspar and with variability in clay mineral types. The vast majority of trace metals in sediment exhibited values in the range or near the upper limit of the normal worldwide soil ranges. TOM and grain size of sediment are major factors governing the trace metal concentrations. The calculated geoaccumulation index (I geo) and enrichment factor (EF) of metals in sediments of Mujib Dam were ranked as follows: cadmium (Cd) > copper (Cu) > zinc (Zn) > lead (Pb) > cobalt (Co) > iron (Fe) > chromium (Cr) > nickel (Ni) > manganese (Mn) > Sr based on the I geo and Cd > Zn > Pb > Co > Cr > Cu > Sr > Ni > Mn according to the EF values. The estimated percentage loss in volumetric capacity of the reservoir due to sedimentation was 1.55% per year, indicating that the sediment currently occupied 18.63% of the original reservoir storage capacity. The maximum life span of reservoir is about 64.46 years.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Mujib Dam was built in 2003 in central western Jordan with a reservoir length of 5 km, a maximum width of 1.1 km, and maximum water levels of 194 and 189 m full supply level (Ijam and Al-Mahamid 2012). With a surface area of about 1.98 km2, the dam’s maximum storage capacity is about 31.2 million cubic meter (MCM) (live storage is about 29.8 MCM, dead storage is 1.4 MCM, and an annual yield of 16.6 MCM). It is founded in the limestone of deeply incised valleys with roller-compacted concrete gravity.
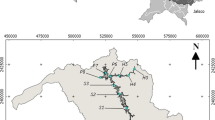
Mujib catchment (of about 4500 km2) is drained by several wadis (Fig. 1) forming the base flow of Mujib Dam and originates primarily from the continuous array of springs within the Dead Sea escarpment and partly from the Ajloun Series and the lower sandy aquifer systems (NWMP 2004). These wadis slope gently along the Jordan Highland and Plateau (of 700–900 m above sea level) before plunging down as they approach the eastern escarpment of the Jordan Rift Valley and flow to the Dead Sea (of 410 m below sea level).
Mujib reservoir, drained by several wadis: Wadi Mujib, Saeda, Balua, and Nukhaila (after JICA Expert/Sabo & Landslide Technical Center 2013)
Total forms of nutrients (including metals) in lakes and water bodies are a function of the factors that influenced their movement across the landscape including stream length, lake order, soil and geologic characteristics of watershed area, and slope and type of drainage system (Fraterrigo and Downing 2008).
Previous studies suggested that reservoir nutrient contents are largely determined by external inputs (Fonseca et al. 2011; Canfield and Bachmann 1981). The relationship between agriculture and micronutrient and macronutrient abundances in water bodies has been widely reported and is presumably a result of the high loadings and low retention of N-P-K fertilizers in cultivated areas (El-Radaideh et al. 2014; Al-Taani et al. 2015a; El-Radaideh 2016). Land development, in the catchment of Mujib Dam, led to an increase in the total area of impervious surfaces, which promotes excessive flood water and reduces the potential of nutrient attenuation in the catchment-watershed area.
The distinct topography of Mujib catchment characterized by a high relief and slope suggests that the dam would receive a significant accumulation of debris and sediment. The majority of soils in the catchment area belong to loamy textural groups ranging from clay loams at the northern part (with moderate infiltration rates) to gravelly and stony sandy loams in the southern area (with higher infiltration rates). Based on Salman et al. (2014), the land use/land cover in the catchment is mainly composed of barren land (53%) that is dominant in the southern area, whereas the northern and western catchments are covered by agricultural farms (14.3%). Coarse-textured soils (of low effective field capacity) are widespread in the catchment and can potentially accelerate contaminant movement to reservoir. Mujib catchment also hosts landfill sites (for liquids and solids).
Heterogeneous soils, cultivation, and land covers of Mujib catchment can strongly influence the chemistry of lake water and its sediments. Among others, cultivated land, livestock farm, gypsum mines, disposal of wastewater in cesspools, and fishing in the lake are the major sources of impacts (Margane et al. 2008).
The sustainability of Mujib Dam is challenged by the high sedimentation and subsequent loss of storage capacity. The frequent and intense storms produce large flood events (the average flood flow is 23 MCM/year) that carry very high sediment loads into the reservoir, and large volumes of dead storage are required. In addition, it also requires check dams in the catchment to control erosion, where the annual sediment yields are estimated to be 60 t/km with an estimated annual sediment load of 17,500 t. On a global basis, about 1% of the total storage capacity of the world’s reservoirs is lost annually due to sedimentation (Mahmood 1987; Yoon 1992), where about 300–400 new dams need to be constructed annually to maintain the current total storage. The estimated cost to replace the existing storage capacity is USD 9 billion (Annandale 2001). In addition to the sedimentation problem, evidence of contamination from human activities has been recently observed (Al-Malahmeh 2006; Hailat and Manasreh 2008; Manasreh et al. 2010), though the Mujib watershed is sparsely inhibited. Monitoring and quality of the reservoir sediments are of significant importance to the region’s sustainable development and prosperity. Dredgability and usability of the accumulated sediments in Mujib Dam reservoir (Fig. 1) have been studied. This study reports, for the first time, the quality properties of the extracting sediments of Mujib Dam through a detailed study of physiochemical, mineralogical, textural distribution patterns, and rate of deposition using representative reservoir sediments. It summarizes the baseline levels of trace metals in the sediments of Mujib and identifies concentration trends with regard to size of particles and concentration heterogeneity. The result of this study will provide reference levels of base metal concentration in sediment of other water bodies in Jordan.
Geology
The Mujib catchment is covered by sedimentary rocks of Mesozoic and Cenozoic with occurrences of basic intrusions. The rock units are exposed in the catchment area of Lower Cretaceous to Recent age. Umm Rijam formation (B4), which is the youngest formation, is exclusively exposed in the eastern watershed with a total thickness of about 90 m. This formation consists primarily of cherts and limestones. The B4 is underlain by Muwaqqar formation (B3) aquiclude of chalky marls with a thickness of 110 m. Amman-Umm Ghudran (B2/B1) formations are cropped out in the central catchment and consist mainly of silicified limestones, cherts, chalky marls, and chalks. Ajlun group (A1–A7), which crops out in the western part, consists largely of a thick sequence of alternating limestone, siltstone, marlstone, nodular limestone, and shales with a thickness of 400–450 m. Kurnub sandstone group exposed in western area is mainly composed of medium- to coarse-grained sandstone with some intercalations of siltstones and clay stones. Amman-Wadi Es Sir is most important aquifer (due to its availability and significant potential). The Jordan-Dead Sea Rift Valley has great influences on the geological structures of the region (Bender 1974; Salman et al. 2014). The stratigraphic sequence of the Mujib Basin is presented in Fig. 2.
Geologic map of Mujib Basin (modified after Salman et al. 2014)
Sediment sampling and analysis
Bottom sediments of Mujib reservoir were sampled at the beginning of summer 2013, corresponding to the end of nutrient input by runoff. Twenty surface sediment (1–15 cm) samples were collected using a Van Veen grab sampler, and sampling points are presented in Fig. 3. Samples were placed into polyethylene bags and transferred to the laboratory for further analyses. Sample preparation and handling were done according to Plumb (1981). Stones and plant fragments were removed by sieving the dried sample through a 2-mm sieve. Sediment samples were initially characterized for grain size distribution, and the results were plotted on a Shepard triangular diagram (Shepard 1954) to produce a facies map (Fig. 3). Following grain size analysis, samples were oven-dried at 55 °C and wet-sieved through a 63-μm sieve to separate the mud from other factions. The fine fraction (63 μm) was further analyzed with a Computerized Sedigraph Analyzer.
Trace metal concentrations in sediments with particle size ≤2 μm were investigated using the pipette analysis method (Williams et al. 2000). The solution of the digested samples was analyzed using the standard methods of atomic absorption spectrophotometry (NOVA A300, Analytik Jena, Leybold, Germany) for iron (Fe), copper (Cu), zinc (Zn), manganese (Mn), cadmium (Cd), cobalt (Co), lead (Pb), chromium (Cr), nickel (Ni), and Sr.
For quality control, triplicates of each sample were analyzed, and average values were calculated. Accuracy of the atomic absorption spectrometer analysis was checked using the National Institute of Standards and Technology standard reference material SRM 1646 (estuarine sediment). Correlation coefficients between sediment variables were calculated with the SPSS software package, Pearson’s correlation coefficient between relevant sediment properties and sediment metal concentration was done to determine the most effective factors influencing the metal concentration in the Mujib sediments. In addition, Person’s correlation coefficient is a simple and effective way in these sorts of studies. Six oriented silty clayey samples were prepared to identify the major and minor mineral composition (Grim 1968) using the scanning electron microscope and XRD.
X-ray diffraction was conducted under the following conditions: 2θ, 0–30°; X-ray radiation, Cu Kα; generator current. 30 mA; generator voltage, 30 kV; and scanning speed. 0.05 cm/min. Non-clay mineral fractions (<63 μm) were also X-rayed for six samples by scanning at 1°/min in the range of 2θ from 5° to 60°. Total organic matter (TOM) in each sample was measured by the titration method (Hesse 1972). The calciometry method (Loring and Rantala 1992) was used to determine the content of carbonate (CO3 −2) in the sediments. In this method, the amount of CO2 released from the reaction is dependent on the amount of CaCO3 in the sample.
Results and discussion
Sedimentation rate and soil in Mujib catchment
In the spring of 2015, we cooperated with Jordan Valley Authority (using their equipments) to conduct a sonic survey to estimate the total sediment volume in Mujib Dam and the amount of sediment loading into the reservoir. The surveys were carried out using digital echo-sounding equipment from a small boat. Post-processing of the data enabled the depth to the top of the deposited sediment layer to be determined. Twelve echo-sounding survey lines were conducted to cover the majority of the Mujib reservoir; distribution and locations of survey lines are presented in Fig. 4.
Schematic diagrams illustrating the survey results are shown in Fig. 5. The cross-sectional profiles in Fig. 5 demonstrated the sediment, water, and base levels in Mujib Dam. It is noteworthy to mention that a detailed map of the pre-dam bottom topography was not available and the calculations were based on a constant base level of about 160 m (Fig. 5).
Based on the survey results, the total volume of sediments deposited behind Mujib Dam reservoir was 5.559084 MCM. This indicates that the estimated long-term sedimentation rate in Mujib reservoir is about 0.4633 MCM/year between dam completion (2003) and the bathymetric survey (2015).
The original volume of Mujib reservoir was 29.846 MCM. The rate at which Mujib reservoir loses its capacity due to sedimentation can be generally expressed as a percentage of original reservoir capacity lost per year. The estimated percentage loss in volumetric capacity of Mujib reservoir is 1.55% per year, indicating that sediments occupied 18.63% of the original reservoir storage capacity (after 12 years of operation). Under the current conditions, the projected life span of Mujib reservoir is 64.46 years (it will be completely infilled with sediment in 2079).
The annual sediment volume, the volumetric sediment accumulation, and the annual flood water entering Mujib Dam obtained in 2015 survey were compared with those of previous years (2005, 2008, and 2009) and are presented in Fig. 6. It appeared that the annual sediment loads are proportional to the annual flood volume. The sediment quantities decreased slightly from 2005 to 2009, but considerable increase in sediment loading occurred between 2009 and 2015. These data also indicate that the annual volume of sediment discharged into the dam is generally fluctuated (unlike that of other dams in Jordan where the quantities remained relatively similar).
According to previous data, the estimated sediment loading into Mujib Dam from 2005 to 2009 was 1.033253 MCM and between 2009 and 2015 was 1.608008 MCM. From 2005 to 2009, the average rate of sediment infilling was about 0.207 MCM/year, whereas it was approximately 0.230 MCM/year during 2009–2015. This indicates that the estimated annual sedimentation rate has increased during the last 7 years. In addition to the increase in flash flood frequency and intensity, land use management and practices in the catchment have probably changed (including agricultural and urban activities), resulting in higher sedimentation rates.
Characteristics of bottom sediments
Granulometric texture
As stated earlier, the sources of sediments in Mujib reservoir are diverse, which determines the rates and types of sedimentation within the reservoir and producing assemblages of weathered products as secondary minerals. Clays, silt, and fine sands (where metals or organic nutrients are preferentially concentrated) are the major types of sediments deposited in the bottom of Mujib reservoir. The grain size distribution is tabulated in Appendix A. Five granulometric textural facies covered Mujib reservoir bed; these are clayey facies, clayey-silt facies, sand-silt-clay facies, and sand facies and granule facies, with average percentages of 23.2% (clay), 45% (silt), 30.7% (sand) and about 1.6% (granule), respectively (Appendix A, Figs. 3 and 7).
Two major modes of transportation processes are responsible for the siltation of the dam; saltation led to deposition of sand fractions, and suspension led to deposition of clay and silt fractions (Fig. 8).
Statistical calculations of particles (population) less than <63 μm in size (analyzed by Sedigraph) showed that the total mean (Mz) was 9.3 μm. These grain sizes with this average play a vital role in carrying and transporting the majority trace metals (including micronutrients) to the reservoir lake.
Bulk mineralogy
Bottom sediments of Mujib reservoir were examined by XRD and SEM. The mineralogy of sediments showed a high similarity in all lithologic facies, in which the textural facies were dominated by calcite, quartz, clay minerals, minor feldspar (anorthoclase), and dolomite.
Clay minerals are composed of palaregoskite, kaolinite, vermiculite, illite, and montmorillonite-smectite. Montmorillonite clay minerals occur as minute particles, which, under SEM, appear as aggregates of irregular, thin laths and flakes. Illite usually occurs as poorly defined flakes and irregular aggregates. Lath- or ribbon-shaped illite particles have also been detected. SEM also showed irregular laths and platy morphologies of kaolinite particles within the detrital quartz matrix (Fig. 9). These mineralogical constituents reflect the geology and soil profiles of catchment, where calcite, dolomite, and anorthoclase (feldspar) were derived from calcareous formations and igneous complex. Quartz was probably derived from the adjacent widely outcropped Kurnub sandstone at the upstream area. Feldspars were probably resulting from chemical weathering of igneous rocks of the NE catchment.
Scanning electron microscopy (SEM) images. a Blocky quartz overgrowth covered adjacent grain (red circle) and illite-smectite clays (red arrow). b Feldspar (anorthoclase) grain and kaolinite ill-stacked booklets of kaolinite. c Spherical aerosol and overgrowth hexagonal quartz. d Hexagonal kaolinite. e Diatom with network ornamentation. f Quartz and overgrowth quartz. g Fibrous wispy illite. h Euhedral rhombohedral calcite/dolomite. i Platelets of kaolinite and tabular illite. j Kaolinite platelets within calcite aggregates. k Partially altered grain of feldspar into kaolinite and euhedral quartz grain. l Well-stacked kaolinite platelets (hexagonal)
The XRD charts indicated that the mineralogical composition of Mujib reservoir sediments is composed of silica (quartz), calcite, dolomite, and anorthoclase (feldspar). (Na,K) (Si3Al)O8 is probably originated from the high-temperature sodium-rich volcanic rocks in the catchment: palygorskite (clay) (Mg, Al)5 (Si,Al)8O20(OH)2, montmorillonite-Na0.3(AL, Mg)2 Si4O10(OH)2·8H2O, vermiculite-Mgx(Mg,Fe)3(Si, Al)4O10(OH)2·4H2O, and kaolinite-Al2Si2O5(OH)4. Fraipontite belonged to kaolinite-serpentine mineral group of detrital origin (Zn, Al, Cu)3 (Si, Al)2O5(OH)4, and metaankoleite (K1.7Ba0.2) (UO2)2(PO4)2·6H2O is a rare secondary mineral in the weathered zone of a complex sericitic matrix of quartz Kurnub sandstone.
The previous major and minor minerals were mainly originated from the heterogeneous lithology of the catchment area, indicating an allochthone origin of the clay minerals as a secondary product in the sediments. This reflects the geological and pedological composition of the reservoir’s watershed. The high variability in mineral composition of Mujib sediment would probably enhance the release of trace metal content to surrounding soils in an oxygenated environment (when disposed of on the surface).
Sediment pH
The pH values (6.9 to 7.8) with an average of 7.5 (Appendix A) are considered convenient for the ready availability of most micronutrients and macronutrients in soils (Donahue et al. 1983). All textural facies are slightly more basic probably due to high concentration of carbonates.
Cation exchange capacity
The cation exchange capacity (CEC) of the bottom samples ranged between 11.0 and 144 meq/100 g with an average value of 88.1 meq/100 g. These values are within the medium interval for soils (100–175 meq/100 g) (Cottenie 1980) (Table 1). CEC increased with decreasing particle size of sediment facies. High levels of CEC were detected in the clay and silty textural facies probably due to high content of different types of available clay minerals and, in particular, palygorskite, montmorillonite, and vermiculite. The high level of CEC values and clay mineralogy play a vital role in the fertility of the bottom sediments, which enhance the availability of both micronutrients and macronutrients.
The CEC values are generally medium to high in Mujib sediment, compared to medium intervals for different granulometric groups of soils. High positive correlations were found between CEC and clay content (Rsq = 0.7), but moderate to high positive correlation was detected between CEC and Co, Fe, Cd, Ni, and Mn (Appendix B).
The high values of total average of trace metals in different granulometric facies (Fig. 10), CEC, TOM, and the clay mineralogy are considered very advantageous with respect to the fertility of the sediments. The presence of palygorskite, montmorillonite, and vermiculite in bottom sediments enhances the availability of micronutrients (Fonseca et al. 1993; El-Radaideh et al. 2014, 2017; Al-Rousan et al. 2016; El-Radaideh 2016). Also, as adsorption and cationic exchange phenomena allow colloidal particles to be a nutrient ion reserve, these sediments probably offer good conditions for agricultural use and natural additives (Sparks, 2002).
Carbonate content (CaCO3)
The CaCO3 of Mujib reservoir sediments varied from 14.8 to 38.4% (averaging 25.8%) (Appendix A). The high content of CaCO3 is mainly derived from the exposed carbonaceous deposits in the catchment. Low positive correlations were observed between CaCO3 content and Cr, K, Mn, and Fe. The positive correlation coefficients with CaCO3 suggest that these elements are partially associated with carbonates (Appendix B).
Macronutrients in sediment
The total average abundances of macronutrients in Mujib sediments are summarized in Appendix A. The average concentrations of PO4, SO4, NO3, F, K, and Ca are 9; 518.2; 5.5; 0.7; 10,515.2; and 103,948.2 mg/kg, respectively (Appendix A). It is believed that these nutrients are likely attributed to the effluent of Al-Lajoun WWTP (Yusuf et al. 2011; Al-Malahmeh 2006). In addition, chemical weathering of rocks in Mujib catchment is a potential source, where the calcareous and igneous rocks bearing Ca, S, and K are widespread in the catchment (Jiries 2001). In addition, intense use of agrochemicals could be another source.
Trace metal content
Most of the trace metals accumulated in bottom reservoir sediments of Mujib Dam are probably transported as suspension with clay-sized fractions (with a high adsorption potential for most trace metals) during the regular quiet water conditions (low discharge), which is necessary for increasing residence time of suspension.
In addition, the relative total content of trace metals in Mujib sediments could also be attributed to high surface areas of organic matter or Fe-oxides. The high percentage of bulk mineral constituents of Mujib sediments are composed of carbonates (with an average value of 25.8%), TOM (with an average value of 6.7%), clay fractions (with an average value of 23.9%), and Fe (with an average value of 3.97%). The surface areas of these particles (with <2 μm in size) as calculated by Forstner and Wittmann (1983) are as follows: calcite (12.5 m2/g), kaolinite (10–50 m2/g), illite (30–80 m2/g), montmorillonite (50–150 m2/g), Fe-hydroxide (300 m2/g), and organic matter particle (1900 m2/g). These constituents with high surface areas can probably accelerate the association of trace element with them.
In this study, there was an association between the physicochemical parameters of Mujib sediments and trace metal contents. Geochemical examinations of Mujib sediments showed that the total forms of the trace metals are in the following decreasing order:
Intertrace metal associations using the Pearson’s correlation coefficients provided also an important view on the possible origin of the trace metals. A significantly positive correlation was established between the trace metal couples Pb with Ni (r = 0.2); Cu with Co (r = 0.3); Cu with Ni (r = 0.6); Cd with Pb, Co, and Ni (r = 0.4); and Sr with Pb (r = 0.4). In addition, moderate to high positive correlation coefficient was also observed between Mn and Cd, Cu, Pb, Co, and Ni, where Pearson’s correlation coefficients ranged from 0.5 to 0.7.
Also, low positive correlation coefficient was found between Fe and Mn, Cd, and Co. High positive correlations were also noticed between clay content and F (r = 0.8), Mn (r = 0.6), Cd (r = .6), Co (r = 0.6), Ni (r = 0.8), Fe (r = 0.4), Cu (r = 0.3), and Pb (r = 0.3) (Appendix B).
The relative high correlation coefficients between trace metals could potentially be an indicator of the same sources (either anthropogenic and geogenic) (Al-Taani et al. 2014; Batayneh et al. 2015). The increases in the concentrations of Zn, Co, Ni, Pb, Co, Cr, Cu, and Mn are probably related to decay of organic materials, sewage, fertilizers, insecticides, agricultural wastes, and wastewater discharges, where the reservoir receives variable amounts of municipal wastewater from many outlets of developed area (for example, the nearby Al-Lajoun wastewater treatment plant). In addition, deposition of total trace metals is accompanied with the deposition of organic matter, carbonates, Fe, and clay minerals in the reservoir bed.
Sediments are particularly important in shallow systems like Mujib reservoir because of the high sediment surface area to water volume ratio. Sediment can serve as sources or sinks for nutrients (Fonseca et al. 2011). Total concentrations of trace metals and physicochemical parameters are summarized in Appendix A. Granulometric textural facies presented in Figs. 3 and 7 are classified as clay, clay silt, sand-silt-clay, sand, and granular facies. These high percentages of clay-sized particle content in the most of granulometric facies which also showed a high percentage of total abundances of most trace metals (Appendix A) are extremely important because they are the highest chemically reactive constituents of any submerged materials. This accelerates the accumulation of the extra-clast organic matter (with subsequent increase in the fertility of poor soils if Mujib sediments are mixed with soils).
Assessment of sediment quality based on metal content
The average concentrations of Fe, Cd, Pb, Zn, Cu, Mn, Co, Ni, Cr, and Sr were 39,620.7; 6.3; 55.5; 278.4; 55.5; 34.7; 24.8; 37.9;114; and 189.5 mg/kg, respectively. The average contents of Fe, Mn, Co, Ni, and Sr were lower than their average concentrations in continental crust (Fortescue 1992). However, the mean levels of Cu, Zn, Cd, Pb, and Cr exceeded their average crustal values (Table 1).
The total concentrations of Fe, Cu, Mn, Co, Pb, and Sr were within the medium values of normal soil; these metals occurred as coating and adsorbed on fine clay-sized fractions of granulometric textural facies, Mn-oxide fractions, and organic grains and as precipitation or co-precipitation of Fe- or Al-oxides and carbonates.
The total abundances of most trace metals (micronutrients) are within or near the normal ranges of worldwide soils (Table 2). Metal micronutrients occur mainly in the clayey and silty facies. These metals are soluble, seldom exceeding the concentrations considered toxic to soils. The high levels of Cd, Cr, Ni, and Zn in Mujib sediments are probably due to their adsorption, precipitation, and co-precipitation avoiding the passage to a soluble phase. The majority of trace metals in bottom sediments of Mujib reservoir showed lower values, indicating that it may potentially be safe to use these sediments in agriculture, particularly after exposure to air. From a pollution potential viewpoint, only Cd and Cr could cause pollution problems. Under aerial conditions, if these sediments are to be used as natural fertilizers, these elements (Cd and Cr) will be oxidized, and their availability to plants will be extremely decreased (According Sparks 2002; Fonseca et al. 2003). According to Fonseca et al. (1993, 2003), the soluble forms of Cd and Zn elements are less toxic and safe when used at agronomic rates. From trace element point of view, Mujib sediments can be a potential fertilizer supplement and soil conditioner that would potentially enhance and restore the eroded and poor soils. Super-accumulator plants can be used to consume the access concentration of these metals (after mixing Mujib sediments with poor soil), which have been used as phytoremediation of contaminated soil (Salt et al. 1995).
Several factors govern the distribution of trace metals in Mujib sediments: among others, the dam’s small size (resulted in a short residence time for settling velocity of variable fractions and higher sequestration of trace metals), total amount of organic matter, pH, and the nature of textural size of sediments (Moura and Sigolo 2002). For evaluating sediment quality, we compared the total average concentrations of trace metals in Mujib sediments with different sets of sediment quality guidelines (Table 2); these are the following:
Typical background levels in US freshwater sediments (NOAA)
Threshold effect level (TEL), NOAA guideline for threshold effect level, concentration at which effects on aquatic organisms might be observed
Probable effect level (PEL), NOAA probable effect level, concentration at which effects on aquatic organisms will likely be observed
Consensus-based estimate of concentration at which adverse effects on aquatic benthic organisms frequently occur and consensus-based probable effect concentrations (PEC) from various agencies (McDonald et al. 2000)
Freshwater sediment benchmark (EPA) and EPA freshwater sediment screening benchmark for use at remediation sites
Screening level guideline of Ontario Ministry of Environment (SLG) (ANZECC and ARMCAZ 2000)
Critical soil concentration (CSC) ranges (Maiti 2003)
NA not applicable
Cadmium
Cd occurs naturally as an impurity in phosphate at concentrations varying from 1 to 200 mg Cd per kilogram P2O5. Cd concentrations in the Mujib reservoir ranged between 1.0 and 12.0 mg/kg, with an average of 6.5 mg/kg. These concentrations exceeded the NOAA, TEL, PEL, PEC, CSC, and EPA benchmark concentrations but less than the SLG (Table 2).
Iron
Fe concentrations in Mujib sediment varied from 33,334.0 to 44,467.0 mg/kg (averaging 39,692.8 mg/kg). No TEL, PEL, or PEC guidelines exist for Fe; however, these values are greater than the benchmark of 20,000 mg/kg for freshwater sediment at contaminated sites (Table 2). The primary source of Fe is probably chemical weathering of basaltic floods which covered the large area of Mujib catchment.
Zinc
Zn contents in sediments ranged between 33.0 and 2151.6 mg/kg, with a mean value of 373.5 mg/kg. Similarly, no TEL, PEL, or PEC guidelines currently exist for Zn. The average Zn in sediments was below the CSC concentration but in excess of the worldwide soil limit (Table 2). The average abundance of Zn in earth’s crust is 76 mg/kg and in soils is 25–68 mg/kg. The permissible limit of Zn in sediment is 121 mg/kg based on the EPA standards.
Copper
Cu concentrations in Mujib bottom sediment ranging between 23.0 and 80.1 mg/kg (average 54.5 mg/kg) exceeded the NOAA. The relative higher values of Cu in some textural facies could be attributed to using Cu-sulfate in nearby water treatment plants. The total average of Cu in sediments was generally less than the PEC, PEL, and SLG limits, but higher than the TEL, CSC, and EPA benchmark concentrations (Table 2). There were positively moderate correlations between Cu and clay content, TOM, Cr, and Sr. The average abundance of Cu is 34 mg/kg, and in soils, it ranges between 14 and 109 mg/kg (Table 2). High level of Cu indicates a higher input of organic matter deposition, which might come from urban and industrial wastes.
Manganese
Mn concentrations varied from 155 to 344 mg/kg, with an average of 268.2 mg/kg. These levels were lower than all sediment quality guidelines and the normal range of soil (Table 2). Mn levels in Mujib sediments can be considered safe based on SLG and lower than CSC ranges. Cu content was positively correlated with Ni and Zn. Mn in earth’s crust is 1060 mg/kg, and in soils, it ranges between 61 and 1060 mg/kg. Mn can be adsorbed onto clay organic fractions, the extent of adsorption depending on the organic content and CEC of sediments.
Cobalt
Co contents in Mujib sediments fluctuated from 10.0 to 42.0 mg/kg (averaging 24.4 mg/kg). These levels were higher than NOAA but less than all sediment quality guidelines (Table 2). The critical and acceptable ranges of Co in soil suggested by Maiti (2003) are 25–50 and 4–20 mg/kg, respectively. Medium to high positive correlations were observed between Co and some physicochemical parameters (clay content r = 0.92, TOM r = 0.63, CEC r = 0.64, K r = 0.61, Pb r = 0.62) and low positive correlations with other parameters (Appendix B). Co is probably derived from biomass of dead cyanobacteria and high input from decaying humic materials from nearby farms.
Lead
Pb concentrations in bottom sediment varied from 21.9 to 101.9 mg/kg (mean value was 55.8 mg/kg). These levels exceeded the NOAA and TEL concentrations, but lower than the PEL, PEC, SLG, CSC, and EPA benchmark limits (Table 2). Pb in natural background soil level ranges between 2.6 and 25 mg/kg (Maiti 2003). Favorable conditions like alkalinity (high available carbonates) enhance Pb abundance (Gowed et al. 2010). Pb content in Mujib sediment was below the CSC range for soils. The Pb-gasoline emissions have been the major sources for the Pb pollution load (Mukai et al. 1994).
Chromium
Cr concentrations in Mujib reservoir sediments ranged between 79.8 and 136.8 mg/kg, with an average of 114.2 mg/kg. Cr contents were higher that the NOAA, TEL, PEL, and the severe limit of SLG. The mean value of Cr was also above the medium value of worldwide soils (11–22 mg/kg) and the CSC ranges (Table 2). Cr is non-essential for plants but is required for animals. The average abundance of Cr in the earth’s crust is 100 mg/kg. Cr has low mobility under medium oxidizing and reducing conditions and neutral pH values. Cr+3 adsorption increases with increasing pH, while Cr+6 adsorption decreases with increasing pH. The relative high concentration of Cr is probably related to bio-accessible processes of Cr enhanced by high content of associated clay. Cr compounds are widely used in pigments, paints, and fungicides (Gowd et al., 2010). Another possible source of Cr is leaching from the surrounding soils. Low to moderate positive correlations with clay content, TOM, CaCO3, pH, CEC, Fe, K, Sr, Cd, Cu, Pb, Co, and Cl were observed (Appendix B).
Nickel
Ni concentrations in Mujib sediments ranged from 17.0 to 78.0 mg/kg, with an average value of 37.6 mg/kg. These values were lower than the severe limit of SLG (75 mg/kg) and the CSC concentration (100 mg/kg) (Maiti 2003) (Table 2). Ni content in sediments showed highly positive correlation with clay content, TOM, and CEC. We observed moderate positive correlation coefficients between Ni and SO4 and NO3 and low positive correlations between Ni and Zn and Ca (Appendix B). Possible origins of Ni contents are probably related to basic igneous rocks cropped out in the surrounding mountain. Other sources may be from fuel combustion at Amman airport.
The degree of contamination by metals was evaluated using the geoaccumulation index (I geo). Muller (1981) suggested the I geo to determine the metal contamination in sediments based on the current concentrations of metal compared with the geochemical background value. We used both the average level in shale (Turekian and Wedepohl 1961) and the average crustal abundance (Fortescue 1992). It can be calculated by the following equation.
where C n is the measured concentration of the examined metal n in the sediment and B n is the geochemical background concentration (or reference value) of the metal n. Factor 1.5 is a background matrix correction factor for lithogenic effects.
The results of I geo calculations of metals are tabulated in Table 2. Based on the Muller scales (1981), Mujib sediments are unpolluted with Fe, Mn, Ni, Cr, and Sr (I geo = −0.07, −0.5, −0.25, −0.23, and −0.58, respectively); unpolluted-moderately polluted with Zn, Co, and Pb (I geo = 0.47, 0.11, and 0.44, respectively); and moderately polluted with Cd and Cu (I geo = 1.32 and 1.17, respectively). Based on the I geo values, the trace metals are ranked in the following order: Cd > Cu > Zn > Pb > Co > Fe > Cr > Ni > Mn > Sr.
For evaluating the geogenic or anthropogenic sources of metal contents in sediments, an enrichment factor was calculated using Fe as a conservative tracer (Ergin et al. 1991; Mucha et al. 2003; Abrahim and Parker 2008; Al-Taani et al. 2015b). According to Ergin et al. (1991), metal enrichment factor (EF) is calculated as follows:
where (M/Fe) sample is the ratio of metal and Fe concentrations in the sample and (M/Fe) crust is the ratio of metal and Fe concentrations in the earth’s crust. In this assessment, the background concentrations of metals were taken from Fortescue (1992) values in crustal abundance and the background value in average shale levels (Turekian and Wedepohl 1961). According to Zhang and Liu (2002) and Zheng et al. (2008), EF values ranging between 0.5 and 1.5 indicate that the metal is entirely from natural processes, whereas EF values greater than 1.5 are more likely to be of anthropogenic origin. Cd, Zn, Pb, and Co have mean EF values of greater than 1.5 (Table 2), with Cd being the most enriched element in sediments. This suggests that these metals were probably originated from anthropogenic sources. Sr, Cr, Cu, Ni, and Mn showed average EF values that are indicative of natural sources, where the mean EF values were less than 1.5. Metals were ranked in the following order: Cd > Zn > Pb > Co > Cr > Cu > Sr > Ni > Mn, according to their EF values.
EF can also assist in determining the degree of trace metal contamination. Five categories are recognized based on EF factors (Sutherland 2000). Based on Sutherland’s classification, the trace metal content in Mujib sediments was deficient to minimal enriched in Fe, Mn, Cr, Cu, Ni, Sr, and Co, whereas they were moderately enriched in Cd, Zn, and Pb.
Conclusions
This paper focuses on characterizing the current status of physiochemical properties of Mujib Dam sediments; potential anthropogenic sources of pollution were also evaluated. The volume of sediment and sedimentation rate in Mujib Dam were also estimated based on sonic surveys. A variety of physical, chemical, biological, and mineralogical properties of bottom sediments collected from Mujib Dam were investigated. These results were employed to investigate the quality of sediments in Mujib reservoir based on several international sediment quality guidelines. This sediment assessment is of significant importance to evaluate the usability of bottom sediments, particularly in agriculture.
Five types of granulometric textural facies were observed for the bottom sediments of Mujib reservoir bed. The fraction of <63 μm showed that the total mean size was 9.3 μm for Mujib. This average grain size will likely play a vital role in adsorption-desorption of the majority trace metals (including micronutrients) to the reservoir lake. For other sediment parameters in Mujib sediments, the total averages were 5.9% (TOM), 7.5 (pH), 25.8% (CaCO3), and 88.0 meq/100 g (CEC), with dominant mineralogical constituents of quartz, calcite, dolomite, and minor feldspar and with variability in clay mineral types. The total concentrations of F, Cl, NO2, NO3, PO4, and SO4 in sediment samples were lower inward for the dam. The vast majority of trace metals in sediment exhibited values in the range or near the upper limit of the normal worldwide soil ranges and that they are within the standard limits recommended for soils in a worldwide basis, except for Cd and Zn. TOM and grain size of sediment are major factors governing the trace metal concentrations. For the calculated I geo and EF of metals in sediments of Mujib Dam, they were ranked as follows: Cd > Cu > Zn > Pb > Co > Fe > Cr > Ni > Mn > Sr based on the I geo and Cd > Zn > Pb > Co > Cr > Cu > Sr > Ni > Mn according to the EF values. Major and trace elements in dam sediments are the result of both anthropogenic and natural sources. The estimated percentage loss in volumetric capacity of Mujib reservoir due to sedimentation was 1.55% per year, indicating that the sediment currently occupied 18.63% of the original reservoir storage capacity. Under the current conditions, the maximum life span of Mujib reservoir is about 64.46 years. Analysis of echo-sounding survey profiles indicated that the annual volume of sediment input into Mujib Dam was generally fluctuated.
References
Abrahim, G. M. S., & Parker, R. J. (2008). Assessment of heavy metal enrichment factors and the degree of contamination in marine sediments from Tamaki Estuary, Auckland, New Zealand. Environmental Monitoring and Assessment, 136, 227–238.
Al-Malahmeh, M. (2006). Quality of treated wastewater produced from Al-Lajoum wastewater treatment plant. M.Sc. thesis, M’tah University, Jordan, 154P.
Al-Rousan, S., Al-Taani, A. A., & Rashdan, M. (2016). Effects of pollution on the geochemical properties of marine sediments across the fringing reef of Aqaba, Red Sea. Marine Pollution Bulletin, 110(1), 546–554.
Al-Taani, A. A., Batayneh, A., Nazzal, Y., Ghrefat, H., Elawadi, E., & Zaman, H. (2014). Status of trace metals in surface seawater of the Gulf of Aqaba, Saudi Arabia. Marine Pollution Bulletin, 86(1–2), 582–590.
Al-Taani, A. A., Batayneh, A., El-Radaideh, N., Al-Rawabdeh, M., Al-Momani, T., & Taani, A. (2015a). Spatial distribution and pollution assessment of heavy metals in surface sediments of Ziqlab Reservoir, Jordan. Environmental Monitoring and Assessment, 187, 1–14. doi:10.1007/s10661-015-4289-9.
Al-Taani, A. A., Rashdan, M., & Khashashneh, S. (2015b). Atmospheric dry deposition of mineral dust to the gulf of Aqaba, red sea: Rate and trace elements. Marine Pollution Bulletin, 92(1–2), 252–258.
Annandale, G.W. (2001). Reservoir conservation and sediment management.
ANZECC & ARMCAZ, (2000). Sediment quality guidelines (in) Australian and New Zealand guidelines for fresh and marine water quality.
Batayneh, A., Ghrefat, H., Zumlot, T., Elawadi, E., Mogren, S., Zaman, Z., Al-Taani, A. A., Nazzal, Y., & Elwaheidi, M. (2015). Assessing of metals and metalloids in surface sediments along the gulf of Aqaba Coast, Northwestern Saudi Arabia. Journal of Coastal Research, 31(1), 163–176.
Bender F., 1974. Geology of Jordan, Borntraeger Berlin 170 pp.
Canfield, D. E., & Bachmann, R. W. (1981). Prediction of total phosphorus concentrations, chlorophyll-alpha, and secchi depths in natural and artificial lakes. Journal of Fish Aquatic Science, 38, 414–423.
Cottenie, A. (1980). Soil and plant testing as a basis of fertilizer recommendations. Food and Agriculture Organization of the United Nations; Soil Resources, Management and Conservation Service Land and Water Development Division. FAO Soils Bulletin, 38(2), 7–93.
Donahue, R. L., Miller, R. W., & Schickluna, J. C. (1983). An introduction to soils and plant growth, In: J. Miller, 5th Edition: Prentice-Hall Inc (p. 667). New Jersey: Englewood Cliffs.
El-Radaideh, N. (2016). Geoenvironmental assessment of Al-Wala Reservoir sediments, Jordan. Geosciences Journal. doi:10.1007/s12303-015-0022-5.
El-Radaideh, N., Al-Taani, A. A., Al-Momani, T., Tarawneh, K., Batayneh, A., & Taani, A. (2014). Evaluating the potential of sediments in Ziqlab Reservoir (northwest Jordan) for soil replacement and amendment. Lake and Reservoir Management, 30, 32–45. doi:10.1080/10402381.2013.870263.
El-Radaideh, N., Al-Taani, A. A., & Al Khateeb, W. (2017). Status of Sedimentation in King Talal Dam, Case study from Jordan. Environmental Earth Sciences, 76(132), 1–14.
Ergin, M., Saydam, C., Basturk, O., Erdem, E., & Yoruk, R. (1991). Heavy metal concentrations in surface sediments from the two coastal inlets (Golden Horn Estuary and Izmit Bay) of the northeastern Sea of Marmara. ChemicalGeology, 91, 269–285.
Fonseca, R., Barriga, F. J. A. S., & Fyfe, W. S. (1993). Suitability for agricultural use of sediments from the Maranhão reservoir, Portugal. In Optimization of plant nutrition (pp. 665–671). Netherlands: Springer. doi:10.1007/978-94-017-2496-8-103.
Fonseca, R., Barriga, S., & Fyfe, W., 2003, Dam reservoir sediments as fertilizers and artificial soils: case studies from Portugal and Brazil. Proceedings of the International Symposium of the Kanazawa University 21st-Century COE Program on Water and Soil Environments, Biological and Geological Perspectives, Kanazawa, March 17–18, p. 55–62.
Fonseca, R., Canario, T., Morais, M., & Barriga, F. (2011). Phosphorus sequestration in Fe-rich sediments from two Brazilian tropical reservoirs. Applied Geochemistry, 26, 16–27. doi:10.1016/j.apgeochem.2011.04.017.
Forstner, U., & Wittmann, G. T. W.(1983). Metal pollution in the aquatic environment: New York, Springer-Verlag, 486 p.
Fortescue, J. (1992). Landscape geochemistry—retrospect and prospect—1990. Applied Geochemistry, 7, 1–54. doi:10.1016/0883-2927 (92)90012-R.
Fraterrigo, J. M., & Downing, J. A. (2008). The influence of land use on lake nutrients varies with watershed transport capacity. Ecosystems, 11(7), 1021–1034. doi:10.1007/s10021-008-9176-6.
Gowd, S. S., Reddy, M. R., & Govil, P. K. (2010). Assessment of heavy metal contamination in soils at Jajmau (Kanpur) and Unnao industrial areas of the Ganga Plain, Uttar Pradesh, India. Journal of Hazardous Materials, 174, 113–121.
Hailat, I. A., & Manasreh, W. A. (2008). The study of the influence of Al-Lajoun basin on water quality of Al-Mujib Dam, Department of chemistry, Mutah University, Al-Karak-Jordan.
Hesse, P. R. (1972). Textbook of soil chemical analysis (p. 520) . New York: Chemical Publications. http://www.mincos.gov.au/publications/australian_and_new_zealand_guidelines_for_fresh_and_marine_water_quality
Ijam, A. Z., & Al-Mahamid, M. (2012). Predicting sedimentation at Mujib Dam Reservoir in Jordan. Jordan Journal of Civil Engineering, 6(4), 448–463.
JICA Expert/Sabo & Landslide Technical Center. (2013). Japan. Akihiko IKEDA: Sediment countermeasures for dams reservoir.
Jiries, A. G. (2001). Chemical evaluation of treated sewage effluent in Karak Province and its suitability for irrigation purposes. Pakistan Journal of Biological Science., 4, 1400–1402.
Kabata-Pendias, A. (2011). Trace elements in soils and plants (4th ed.). Boca-Raton: CRC Press.
Loring, D. H., & Rantala, R. T. (1992). Manual for the geochemical analysis of marine sediments and suspended particulate matter. In Earth science reviews (Vol. 32, pp. 235–263). Amsterdam: Elsevier.
MacDonald, D. D., Ingersoll, C. G., & Berger, T. A. (2000). Development and evaluation of consensus-based sediment quality guidelines for freshwater ecosystems. Archives of Environmental. Contaminant. Toxicology., 39, 20–31.
Mahmood, K. (1987). Reservoir sedimentation: impact, extent, and mitigation. World Bank Technical Paper No. 71, World Bank, Washington, DC.
Maiti, S. K. (2003). Handbook of methods in environmental studies: air, noise, soil and overburden analysis. ABD Publishers.Vol. 2
Manasreh, W., Hailat, I., & El-Hasan, T. (2010). Heavy metal and anionic contamination in the water and sediments in Mujib reservoir. Central Jordan, Environmental Earth Sciences, 60, 613–621.
Margane, A., Subah, A., Hajali, Z., & Almomani, Th. (2008). Delineation of surface water protection zones for the Mujb Dam, Technical Cooperation Project Groundwater Resources Management Technical Report No. 10, Amman, Prepared by BGR and MWI, BGR Archive No. 2005.2110.4.
Moura, C. L., & Sigolo, J. B. (2002). Heavy metals concentration (Cr, Cu, Ni and Zn) in bottom sediments of the Embu-Mirim River, Sp. In Anais da Academia Brasileira de Ciencias, 74 (3). Rio de: Janeiro.
Mucha, A. P., Vasconcelos, M. T. S. D., & Bordalo, A. A. (2003). Macrobenthic community in theDouro Estuary: relations with trace metals and natural sediment characteristics. Environmental Pollution., 121, 169–180.
Mukai, H., Tanaka, A., Fujii, T., & Nakao, M. (1994). Lead isotope ratios of airborne particulate matter as tracers of long-range transport of air pollutants around Japan. Journal of Geophysics Research, 99, 3717–3726.
Muller, G. (1981). Die Schwermetallbelastung der Sedimenten des Neckars und Seiner Nebenflu¨sse. Chemiker-Zeitung, 6, 157–164.
NWMP (National Water Master Plan (NWMP). (2004). Surface water resources: vol(4). Open files: Ministry of Water and Irrigation, Jordan and German Technical Cooperation (GTZ).
Plumb, R. H., Jr. (1981). Procedure for handling and chemical analysis of sediment and water samples. Prepared by Great Lakes Laboratory, State University College at Buffalo, NY, for the US Environmental Protection Agency /US Army Corps of Engineers Technical Committee on Criteria for Dredged and Fill Material. Vicksburg (MS): US Army Engineer Waterways Experiment Station. Tech. Rep. EPA/CE-81–1.
Salman, A., Al-Qinna, M., & Al Kuisi, M. (2014). Spatial analysis of soil and shallow groundwater physicochemical parameters in El-Mujib Basin-central Jordan. Journal of Asian Earth Sciences, 79, 366–381.
Salt, D. E., Blaylock, M., & Kumar, N. P. B. (1995). Phytoremediation: a novel strategy for the removal of toxic metals from the environment using plant. Biotechnology, 13, 468–474.
Shacklette, H. T., & Boerngen, J. G. (1984). Element concentrations in soils and other surficial materials of the conterminous United States. U.S. Geology. Survey Professional Paper 1270.
Shepard, F. P. (1954). Nomenclature based on sand-silt-clay ratios. Journal of Sediment Petrology, 24, 151–158.
Sparks, D. L. (2002). Environmental soil chemistry (p. 345). San Diego: Academic Press.
Sutherland, R. A. (2000). Bed sediment-associated trace metals in an urban stream, Oahu, Hawaii. Environmental Geology, 39, 611–627.
Turekian, K., & Wedepohl, K. H. (1961). Distribution of the elements in some major units of the earth’s crust. Geological Society of America Journal, 72, 175–192.
Williams, T. M., Rees, J. G., & Setiapermana, D. (2000). Metals and trace organic compounds in sediments and waters of Jakarta Bay and the Pulau Seribu Complex, Indonesia. Marine Pollution Bulletin, 40, 277–285.
Yoon, Y. N. (1992). The state and the perspective of the direct sediment removal methods from reservoirs. International Journal of Sediment Research, 7, 99–115.
Yusuf, N., Al-Taani, A. A., Al-Fukaha, F., Al-Shereideh, S., & Mahasneh, M. (2011). Sediments transport across the fringing reef in the gulf of Aqaba, red sea and the implications on live corals. Abhath Al-Yarmouk: Basic Science & Engineering, 20(1), 35–52.
Zhang, J., & Liu, C. L. (2002). Riverine composition and estuarine geochemistry of particulate metals in China—weathering features anthropogenic impact and chemical fluxes. Estuarine, Coastal and Shelf Science, 54, 1051–1070.
Zheng, N., Wang, Q., Liang, Z., & Zheng, D. (2008). Characterization of heavy metal concentrations in the sediments of three freshwater rivers in Huludao City, Northeast China. Environmental Pollution, 154, 135–142.
Acknowledgments
The authors would like to extend their sincere appreciation to the Scientific Research Support Fund at Amman, Jordan, for its funding this research, project number WE/2/05/2012. The authors would like to express their sincere gratitude to the Departments of Earth and Environmental Science, Yarmouk University.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOCX 27 kb)
Rights and permissions
About this article
Cite this article
El-Radaideh, N., Al-Taani, A.A. & Al Khateeb, W.M. Characteristics and quality of reservoir sediments, Mujib Dam, Central Jordan, as a case study. Environ Monit Assess 189, 143 (2017). https://doi.org/10.1007/s10661-017-5836-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-017-5836-3