Abstract
A detailed field study was carried out to monitor (i) the arsenic contents in irrigation groundwater and paddy soil and (ii) the accumulation of arsenic in the roots and grains of different paddy varieties grown in the arsenic-contaminated middle Indo-Gangetic Plains of Northern India. Results showed the highest arsenic contamination in the irrigation groundwater (312 μg l−1) and in paddy soil (35 mg kg−1) values that were significantly exceeded the recommended threshold values of 100 μg l−1 (EU) and 20 mg kg−1 (FAO), respectively. The paddy soil arsenic content ranged from 3 to 35 mg kg−1 with a mean value of 15 mg kg−1. The soil arsenic content was found to be influenced by the soil texture, carbon, macronutrients, phosphorus, sulfur, hydrolases, and oxidoreductases properties of the paddy soils as revealed in the principal component analyses. Higher root accumulation (>10 mg kg−1) of arsenic was observed in 6 of the 17 paddy varieties grown in the study area. The range of arsenic content accumulated in the paddy roots was 4.1 to 16.2 mg kg−1 dry weight (dw) and in the grains 0.179 to 0.932 mg kg−1 dw. Out of 17 paddy varieties, eight had 0 > .55 mg kg−1 grain arsenic content and were found unsafe for subsistence maximum daily tolerable dietary intake (MTDI) by human beings according to the regulatory standards.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Arsenic (As) is a toxic metalloid, ubiquitous in the environment and affecting over 150 million people worldwide through consumption of arsenic-contaminated potable water (Ahamed et al. 2006; Rahman et al. 2009). The arsenic contamination in groundwater through geogenic origin has been reported in the USA, Chile, Mexico, Japan, Argentina, etc. (Das et al. 2004; Patel et al. 2005; Bhattacharya et al. 2007; Casentini et al. 2011; Bundschuh et al. 2012). There has been a global concern about the toxicity originated from the use of arsenic-contaminated groundwater for drinking and irrigation purposes, especially in Asia and Southeast Asia (Bhattacharya et al. 2007; Brammer and Ravenscroft 2009). The groundwater from tubewells used for irrigating agricultural fields is adding large quantities of arsenic every year, which has resulted into elevated levels of arsenic in the agricultural soils as well as in the crops grown on these soils (Stroud et al. 2011; Norton et al. 2012). Compared to other cereals, arsenic accumulation in paddy crop, a major staple food, is more efficient owing to its flooded agronomic practice (Su et al. 2010; Zhao et al. 2010; Hua et al. 2011). Arsenic accumulation in paddy crop can cause many human health risks (Hite 2013). Consumption of arsenic-laden rice grains is causing arsenic-induced toxic health symptoms and diseases among the human population living in the affected areas (Islam et al. 2000; Patel et al. 2005; Bhattacharya et al. 2007; Bundschuh et al. 2012). In addition to human health risks, high arsenic levels in soil may reduce root growth, plant height, and grain yields in paddy crop along with the occurrence of other physiological disorder like straight-head disease in rice (Yan et al. 2008; Khan et al. 2009; Hua et al. 2011). Arsenic toxicity in paddy leads to a reduction in crop yield as well as contamination of the food chain (Panaullah et al. 2009). The temporal and spatial distribution of arsenic in paddy soils and paddy crop may be affected by the level of arsenic in irrigation groundwater, soil properties, and different rice varieties/genotypes (Alam and Rahman 2003; Meharg and Rahman 2003; Zheng et al. 2011). Rice is a major staple food of the world, especially the South and Southeast Asia. India is a major rice-producing nation, where paddy cultivation occupies 23.3 % of gross cropped area in India. An average intake of an adult human is about 300–600 g cooked rice per day, especially in the Asian countries (Banerjee et al. 2013). Rice cultivation occupies 13.5 million ha area in the Indo-Gangetic Plains (IGP) of South Asian region.
It is important to find out arsenic contamination levels in the irrigation groundwater sources and related agricultural fields under paddy cultivation in the unexplored geographical areas, as the middle IGP region (Rahman et al. 2009; Sidhu et al. 2012). It may help not only in developing remediation strategies to reduce arsenic uptake in crops like rice but also in evaluating the potential health risks involved. In the IGP region of the Northern India under Ganga-Meghna-Brahmaputra basin, high groundwater arsenic levels (up to 500 μg l−1) in drinking water sources have been reported by many researchers (Chakraborti et al. 2004; Mukherjee et al. 2006; Bhattacharya et al. 2007). The groundwater, which has been found unsafe for drinking purposes in a UNICEF survey during 2005, is being used continuously for irrigating agricultural fields in the IGP region. For evaluating arsenic accumulation in paddy crop, several studies have been carried out as hydroponics and greenhouse pot experiments using arsenic spiked soils and/or irrigating rice plants with arsenic-contaminated waters (Abedin et al. 2002; Rahman et al. 2008; Srivastava et al. 2013). Norton et al. (2009b) have suggested developing knowledge of paddy grain arsenic content across multiple environments under field conditions. The understanding of an interaction between different soil properties and soil arsenic content is also necessary to identify key soil properties influencing soil arsenic content under the field conditions. However, data on arsenic accumulation in the soil and paddy crop with special reference to actual field conditions are urgently required to reveal the spread of arsenic problem in the middle IGP region (Srivastava et al. 2013). A review of the literature suggests that none of the studies have included dedicated field study on the accumulation of arsenic in paddy soil and its uptake by different paddy varieties in the middle IGP region.
It was postulated that the use of arsenic-laden groundwater for irrigation was contributing to a significant buildup of arsenic in the paddy fields and contaminating different paddy varieties growing in the middle IGP region. It was also speculated that some key soil properties may regulate soil arsenic content under the field condition in the targeted region. Hence, the aims of the present study were (1) to find out arsenic contents in the paddy soils and different paddy varieties growing in the middle IGP region; (2) to monitor arsenic contents in the irrigation groundwater sources, which were main sources of arsenic input into the paddy soils; and (3) to assess the role of main soil properties in influencing arsenic content in the paddy soils.
Materials and methods
Study area
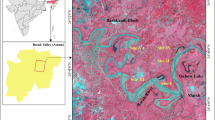
The study was conducted in five different administrative districts (dist.) of the state of Uttar Pradesh. These dist. are Ballia (25° 45′ to 25° 74′ N and 84° 10′ to 84 °55′ E), Ghazipur (25° 29′ to 25° 55′ N and 83° 18′ to 83° 55′ E), Gorakhpur (26° 46′ N 83° 22′ E), Bahraich (27° 17′ to 27° 58′ N and 81° 31′ to 81° 59′ E) and Lakhimpur-Kheri (27° 56′ to 28° 27′ N and 80° 35′ to 80° 46′ E) located in the middle IGP of the Northern India, which represents a major arsenic-affected region in the country (Fig. 1). The climate is mainly subtropical humid to sub-humid. The temperature ranges 32–45 °C in the summers and 2–16 °C in the winters. These sites in the alluvial plain are under paddy cultivation over several decades. The arsenic content in groundwater sources, which is also used for drinking purposes at these sites has been found above the threshold level of 10 μg l−1 (WHO 2005) as revealed by the UNICEF and the Uttar Pradesh Water Corporation, India, in a joint survey during 2005. Out of the total 58 villages in 18 different administrative blocks selected for the study in these 5 districts, 6 villages of 2 blocks (Palia and Issanagar) are located in the Lakhimpur-Kheri; 17 villages in 4 blocks (Jarwal, Phakharpur, Tejvapur, Huzoorpur) of Bahraich; 5 villages in 1 block (Campiereganj) of Gorakhpur; 21 villages in 7 blocks (Maniyar, Bansdeeh, Revati, Bairiya, Muralichhapara, Belhari, Dubhad) of Ballia; and 9 villages in 4 blocks (Karanda, Reotipur, Jamania, Saidpur) of Ghazipur. The detailed site description of all these 58 villages is given in Table S1.
Sampling of water, soil, and paddy crop
In the year 2011 and 2012, the sampling of groundwater was done from the tubewell sources, which were being used for irrigation of paddy fields. The irrigation groundwater sampling was done at the time of planting of rice in the fields, followed by two subsequent samplings per month during paddy cultivation. Water samples (250 ml) (n = 10) were collected in the sterilized polyethylene bottles fitted with liquid-tight stopper after running the tubewell for 10 min. Then the water samples were immediately acidified with 1 % HNO3 and analyzed for arsenic content within 7 days of sampling.
In October–November during the year 2011 and 2012 just prior to harvesting of paddy crop, 10 soil cores (0–45 cm) were taken from each field (10 m2 area) (randomly across the field excluding border area) as replicates and mixed together to have a single representative composite sample of the field. The field was irrigated by the same groundwater source (already sampled for water samples at the time of rice planting) in each village. The air-dried, homogenized, and sieved soil samples were used for physicochemical analysis, and fresh (moist) samples (grounded earlier) were kept at 4 °C for soil enzyme analysis. At the time of soil sampling, 50 paddy plants with ripe panicles were carefully harvested from the same field to represent crop sample for each field. The paddy plants were segregated into roots and seeds. The segregated plant roots were washed thoroughly using deionized water thrice to remove adhering soil particles. The husks were removed from the rice seeds, and grains were oven-dried (70 °C) and then finely grounded in a ball mill.
The physicochemical properties of the soil viz bulk density (BD), particle density (PD), water holding capacity (WHC), clay content, pH, electrical conductivity (EC), total organic carbon (TOC), microbial biomass carbon (MBC), and available forms of phosphorus (AP = available phosphorus), sulfur as available sulfate (AS), nitrogen (AN), potassium (AK), calcium (A-Ca), and sodium (A-Na), and the enzyme activities viz, dehydrogenase (DHA), fluorescein diacetate hydrolysis assay (FDA), protease (ProA), alkaline phosphatase (APA), cellulase (CelA), β-glucosidase (β-GA), arylsulfatase (ArylS) were done using the standard methods of Black (1965) and Dick (2011) as described earlier by (Srivastava et al. 2011a, 2012).
Arsenic estimation
All water samples were filtered through Whatman filter paper no. 40 prior to arsenic estimation. The digestion of air-dried and finely grounded soil (0.5 g) was made by using 5 ml of HNO3:30%H2O2:HF (5:1:1) following the microwave digest procedure (BERGHOF-Speedwave-MWS-3+). The digestion of oven-dried plant part (initially air-dried <40 °C, then kept in the oven at 60 °C for 48 h) was done after chopping and sieving them to the <2 mm size. The digestion of plant part samples (0.1 g) was carried out using 5 ml of HNO3:30%H2O2 (5:1) by the microwave digest procedure. The digested samples were then cooled, diluted with some deionized water and filtered using Whatman filter paper no. 42 and 41 for soil and plant samples, respectively, followed by volume makeup to 15 ml with deionized water. Total arsenic content in water, soil and paddy plant part samples was estimated using inductively coupled plasma-mass spectrometry (ICP-MS) (Agilent, 7500ce).
Quality control and quality assurance
Quality control and quality assurance of total arsenic estimation were done according to the method as described earlier (Srivastava et al. 2011b). Mean rice flour certified reference material (CRM NIST 1568a) and spike recoveries of total As were 90 % (±2.5; n = 5) and 85 % (±3.3; n = 5), respectively.
Statistical analysis
The data were subjected to analysis of variance-general linear model (ANOVA-GLM) and correlation analysis to study significant differences at p < 0.05 using SPSS 11.0 statistical package. Comparison of means and level of significance were carried out and established according to post hoc Tukey’s test. One-way ANOVA was used to test for statistically significant differences between arsenic contents for the paddy varieties at individual field sites. For the paddy varieties commonly grown across the two districts, GLM was used with paddy variety and field site as the main effects. Pearson’s correlation coefficient analysis was performed between rice arsenic concentrations and soil properties. The principal component analysis (PCA) was performed to find out the principal soil factors influencing arsenic accumulation in soil including physical, chemical properties, and enzyme activities of soil. The absolute value of the loadings more than 0.1 of the total variance and an eigenvalue >1 was set as the threshold for component extraction after stepwise regression to remove unreasonable factors.
Results
Arsenic in irrigation groundwater
The field study revealed large variations in arsenic contents in water samples of five districts (Fig. 2). The results showed that the irrigation groundwater samples from the middle IGP had arsenic contents ranging from zero (not detected) to as high as 312 μg l−1. The arsenic content in irrigation groundwater was much higher than the recommended threshold limit for irrigation water (100 μg l−1, EU). The highest arsenic content was recorded in the Belhari Block of Dist. Ballia (312 μg l−1). The arsenic contents (>100 μg l−1) in waters were observed in decreasing order at Phakharpur (Bahraich) > Maniyar (Ballia) > Revati (Ballia) > Bansdeeh (Ballia) > Reotipur (Ghazipur) > Bairiya (Ballia) > Jamania (Ghazipur) > Saidpur (Ghazipur) > Belhari (Ballia) > Palia (Lakhimpur-Kheri) > Tejvapur (Bahraich) > Campieerganj (Gorakhpur) > Murlichhapara (Ballia) > Dubhad (Ballia) > Huzoorpur (Bahraich) > Jarwal (Bahraich) > Karanda (Ghazipur) > Issanagar (Lakhimpur-Kheri). Of the total water samples, 67 % ranged between 100 and 200 μg arsenic l−1 and 15 % >200 μg arsenic l−1. Previous studies have revealed that 10 to 44 % of water samples collected from the studied areas had above 300 μg l−1 of arsenic (Ahamed et al. 2006; Srivastava and Sharma 2013).
Soil arsenic contents
In paddy soil, the arsenic content ranged from 3 to 35 mg kg−1 (Fig. 2). The arsenic contents in the soil of the studied region showed that the values of arsenic exceeded the normal world content, i.e., 5 mg kg−1 at most of the study sites. Arsenic levels in 17 villages were found above threshold limit of 20 mg kg−1 as per the FAO standard for agricultural soils. These sites were covering 26 % of total soil samples under the study. Among these 17 villages, 2 were from Dist. Ghazipur (blocks: Jamania and Reotipur), 6 from Dist. Bahraich (blocks: Phakharpur and Tejvapur), and 9 from Dist. Ballia (blocks: Maniyar, Bairiya, Revati, and Belhari). Srivastava and Sharma (2013) have also reported 5–15 mg kg−1 of arsenic content in soil samples of the studied region and found arsenic contamination in locally grown vegetables such as beans, tomato, spinach, etc. A significant (p < 0.05) correlation was observed between arsenic contents in irrigation groundwater and paddy soils.
Rice arsenic contents
The uptake of arsenic in different paddy plant parts is shown in Fig. 3. Results indicated a considerable accumulation of arsenic (mg kg−1) in the roots (4.1 to 16.2) and the grains (0.179 to 0.932). Seventeen paddy varieties have been found to be commonly grown in the studied region. Out of these 17 paddy varieties, higher arsenic contents in grains (>0.5 mg kg−1) were found in 8 paddy varieties, namely Swarana sub-1, Kasturi, Sarjoo-52, Arize-6444, BPT-3291, Varadhan, IPB-1, and Sugandha-4/Pusa-1121. The study was revealed that these varieties are more susceptible to high arsenic content in the soil and had accumulated comparatively higher arsenic content in the grains. None of the samples of paddy grains exceeded the recommended threshold limit of 1.0 mg kg−1. But, these eight paddy varieties contained grain arsenic content unsafe for subsistence maximum daily tolerable dietary intake by humans (Williams et al. 2005). The arsenic content in roots showed significant differences among paddy varieties. The arsenic content in order of highest to lowest was Bengal Juhi > Kalanamak > IPB-1 > BPT-5204 > Arize-6444 > NDR-359, while grain arsenic content of Bengal Juhi, Kalanamak, NDR-359, and BPT-5204 was found in comparatively low arsenic category. Such paddy varieties can be considered as low grain arsenic accumulating safe paddy varieties according to Williams et al. (2005). There were significant varietal differences (p < 0.05) in the arsenic contents of the paddy plant parts observed among all 17 paddy varieties grown in the studied region.
Correlation analysis with reference to arsenic mapping
The Pearson correlation coefficients among the arsenic contents in the irrigation water, soil, and paddy grains were computed. A significant positive correlation (p < 0.05) was observed between the arsenic content in water and soil samples (Fig. 4). Likewise, arsenic contents in grains of different paddy varieties were influenced by the root arsenic and the soil arsenic contents (Figures S1, S2, and S3). The arsenic contents in paddy soil, roots, and grains were significantly different (p < 0.05) and positively correlated with each other, conforming to some of the earlier reports (Hossain et al. 2008; Sidhu et al. 2012). The results showed a significant positive influence of the total arsenic content in the soil on the arsenic accumulation in the paddy crop in the studied region. In the case of traditional paddy varieties like Bengal Juhi, Kalanamak, and Indrasaan, the correlation coefficients (r 2) were 0.259 and 0.586 between grain arsenic versus root arsenic, and soil arsenic versus root arsenic, respectively, confirming the low grain arsenic accumulation in these varieties.
Paddy soil characteristics and principal component analysis (PCA)
The study of physicochemical properties of paddy soils revealed that the soil is slightly alkaline, with a mean value of pH 7.42 (ranging from 6.82 to 8.0) and clay loam to clay in texture (clay content mean value 31.6 %, ranging between 20 to 42 %) (Tables S2 and S3). The average mean values of total organic carbon and nitrogen contents were 2.52 % and 73.4 mg kg−1, respectively, ranging between 1.85 to 2.91 % and 48.2 to 126.09 mg kg−1, respectively. Among other soil properties, the mean values of available contents (mg kg−1) of phosphorus, sulfur, sodium, potassium, and microbial biomass carbon were 256.6, 6.84, 1.98, 171.03, and 163.01, respectively. The average DHA and FDA contents of paddy soils had mean values of 3.43 μg TPF (g soil)−1 h−1 and 7.08 fluorescein formed nanomol (g soil)−1 h−1, respectively (Table S4).
The paddy soils with higher arsenic contents showed comparatively lower enzyme activities among the various samples. All the enzyme activities were significantly (p < 0.05) and negatively correlated with the soil arsenic content. The r 2 values between the soil clay content, carbon, macronutrients, phosphorus, sulfur, hydrolases, and oxidoreductases versus the soil arsenic content showed a linear relationship.
A long-term accumulation of arsenic in soil adversely affects microbial biomass carbon, respiration (Van Zwieten et al. 2003; Ghosh et al. 2004), and dehydrogenase activity (Fernandez et al. 2005). The soil arsenic may be involved in reducing the soil enzyme activities by masking catalytically active groups, denaturing protein conformation, or competing with metals required to form active enzyme-substrate complexes (Karaca et al. 2010; Koo et al. 2012).
The PCA was carried out between soil arsenic content and different soil physical, chemical properties, enzyme activities to find out the principal components influencing the soil arsenic content. The PCA with soil physical properties produced two principal components, which accounted together for 74.2 % of the total variance (43.6 % for the PC1 and 30.5 % for the PC2) (Fig. 5). The PCA indicated that soil texture (importantly the combination of clay and water holding capacity; and the combination of bulk density and particle density) was the key factor determining the soil arsenic content in the case of soil physical properties. The PCA with soil chemical properties yielded 78.1 % of the total variance with 46.3 % variance for the first factor (PC1), which appeared to be associated with the soil phosphorus and soil sulfur content and 31.8 % variance for the second factor (PC2), which appeared to be associated with the soil pH and soil carbon (Fig. 6). The PCA with soil enzyme activities showed 74.7 % of the total variance with 41.3 % variance for the PC1 representing soil DHA, arylsulfatase, and (-) FDA and 33.4 % variance for the PC2 representing soil β-glucosidase and cellulase activities (Fig. 7).
PCA biplot of soil arsenic and soil chemical properties of paddy soils in the middle IGP region. MBC microbial biomass carbon, A-Na available sodium, AN available nitrogen, A-Ca available calcium, AK available potassium, TOC total organic carbon, EC electrical conductivity, AP available phosphorus, AS available sulfate-sulfur
Discussion
Eight of the total 18 administrative blocks located in five different districts in the middle IGP exhibited higher arsenic contamination in the irrigation groundwater sources and the paddy soils above the threshold limits of 100 μg l−1 (EU) and 20 mg kg−1 (FAO), respectively.
The arsenic content of groundwater showed significant spatial variations throughout the studied region with tremendous substantially high contents (as mean value) at the Phakharpur (230.1 μg l−1) block of dist. Bahraich, Maniyar (228.4 μg l−1), and Revati (213.3 μg l−1) block of dist. Ballia. The districts of Ballia and Bahraich had the most affected sites among total five districts studied. Variation of arsenic contents in groundwater may be affected by the bedrock, its arsenic content, and chemical dissolution processes that usually occurred during heavy pulling out of groundwater (Islam et al. 2000). Notably, groundwater sources of 35 villages in the studied region contained arsenic above 100 μg l−1, indicating the magnitude of arsenic contamination problem and the associated human health hazards. Earlier, the health-related arsenic toxicity in the population of the region has been reported due to the consumption of arsenic-contaminated groundwater for drinking purpose (Ahamed et al. 2006). Studies have revealed that reducing conditions coupled with microbial organic matter decomposition may enrich the arsenic mobilization in soils (Singh et al. 2010). Further, the arsenic-tainted groundwater may result in the arsenic buildup in the crop root zone of soils (Khan et al. 2009).
In the present study, arsenic contents in soils ranged from 3 to 35 mg kg−1. The highest level of arsenic in soil was found in the samples collected from dist. Ballia among all the study sites. The upper layer (0–45 cm) of soil is mainly contaminated by the arsenic inputs from contaminated irrigation groundwater (Panaullah et al. 2009). Differential arsenic contamination pattern in paddy soils was observed due to varying agronomic practices in terms of the volume of irrigation water used and due to the difference of arsenic levels in the groundwater at different sites (Meharg and Rahman 2003). Meharg and Rahman (2003) have observed that the soil arsenic level can reach up to 57–83 mg kg−1 in areas due to continuous use of high arsenic-tainted groundwater. Earlier, a survey of four villages in districts Ballia and Ghazipur had revealed the presence of arsenic in soil samples ranging from 5.4 to 15.4 mg kg−1 (Srivastava and Sharma 2013). Panaullah et al. (2009) have suggested that arsenic added through irrigation water can be quantitatively retained in the soil year by year over long-term use of contaminated water. This accumulation of soil arsenic may lead to soil arsenic mobilization through lateral redistribution of arsenic over adjacent areas depending upon irrigation water flow pattern and surface water dynamics (Khan et al. 2009).
Zhao et al. (2010) have explained that the bioavailability of arsenic is more enhanced to paddy crop under the flooded conditions. Furthermore, the uptake of arsenite (reduced inorganic species) through the aquaglyceroporin channels is highly efficient in paddy roots. Additionally, the arsenate (oxidized inorganic species) may enter paddy roots via phosphate/arsenate cotransporters. Consequently, arsenic content in paddy grains may be derived from either xylem transport from paddy roots or remobilization of shoot arsenic pools through phloem loading during the grain filling (Norton et al. 2010). In the present study, the paddy roots showed significantly higher arsenic contents compared to other plant parts. Rahman et al. (2007) have also revealed that the uptake of arsenic was 75-folds higher in paddy roots compared to grains. Significant differences (p < 0.05) in the arsenic contents were observed in the paddy grains of 17 paddy varieties grown in the studied region (Table 1). The earlier studies have also demonstrated differences in arsenic accumulation among various paddy genotypes (Norton et al. 2009a; Wu et al. 2011; Lei et al. 2012). The arsenic uptake by paddy varieties ranked as hybrids (like Arize-6444, PHB-71) > traditional (like Indrasan, Kalanamak, Bengal Juhi) > > released varieties (based on R&D for the traits like submergence tolerance, yield, aroma/quality, short duration late varieties, like Swarana sub-1, Sugandha-4/Pusa1121, Sarjoo-52, BPT-3291) (Figures S1, S2, and S3). The uptake of arsenic in rice grains (mg kg−1) varied spatially among cultivated paddy varieties in the studied region (Zavala and Duxbury 2008). Total dietary arsenic intake ranges from 10 to 200 μg per person per day in different countries (Zhao et al. 2010). All the paddy grain samples had arsenic content less than the recommended threshold limit of grain arsenic (1.0 mg kg−1, WHO), while some of the paddy varieties were found to be unsafe for the subsistence diet according to Williams et al. (2005) (Table 1). A health risk for the population living in the middle IGP is quite possible through ingestion of arsenic-laden rice grains of eight paddy varieties (Table 1). The arsenic level buildup in the paddy soil may lead to high arsenic in rice grain. Thus, the amount of arsenic ingested by the human and the cattle populations of the studied region may increase as argued by Meharg and Rahman (2003) in an earlier study. Among the different approaches to minimize arsenic uptake in paddy, a field-based selection of paddy varieties with respect to low arsenic content in their grains is considered to be a feasible strategy. All the paddy varieties grown within the middle IGP region revealed significant differences in the paddy root arsenic to paddy grain arsenic ratio along with the spatial effect. Based on the grain arsenic content of paddy varieties, a number of potentially promising varieties can be identified for their use as breeding material to develop new breeding cultivars with low grain arsenic (Norton et al. 2012). In the present study, the Bengal Juhi, Kalanamak, NDR-359, and BPT-5204 paddy varieties were ranked as low grain arsenic varieties across the entire studied region. The findings of the present study can be useful in breeding new low grain arsenic rice variety using existing low grain arsenic paddy varieties, which are adapted to local climate, edaphic factors, paddy agronomy practice, etc. (Norton et al. 2009a). The main factors contributing to the variation of grain arsenic content are the field site, microclimate, paddy genotypes, and the field site-genotype interaction (different genotypes behave differently at different sites) (Norton et al. 2009b).
The variations in total grain arsenic at different sites for the same cultivars might be due to the difference in soil arsenic content, different soil properties, and the field management practices. Different soil properties, like pH, organic matter, clay, etc., influence the arsenic accumulation and its mobility in soil (Koo et al. 2012). Bhattacharyya et al. (2008) and Koo et al. (2012) have revealed significant and negative correlations between soil metalloid contents and soil enzyme activities. The soil arsenic contents differed largely depending on the underlying site-specific soil properties as revealed through the PCA in the study. The PCA showed that soil texture, carbon, phosphorus, sulfur, hydrolases, and oxidoreductases are the most important soil properties influencing the soil arsenic content. Zhao et al. (2010) have explained that soil characteristics (like composition of microbial community, redox state, etc.) may affect the soil arsenic content and subsequent arsenic accumulation in paddy crop. Das et al. (2013) have also found that microbial biomass C and enzyme activities were significantly inhibited by the arsenic accumulation in soils. They explained that arsenic would inactivate enzymes through reacting with –SH groups and affect the enzyme conformation. Thus, the arsenic stress may result into a decrease in the biochemical activities of soils, which are essential for normal soil functions, like fertility, etc. Sulfate, molybdate, and nitrate have a greater tendency compared to other anions to displace arsenic from the mineral complexes (Sinha and Bhattacharyya 2011). The arsenate entry into the plant roots is also affected by the soil phosphorus content (Zhao et al. 2010).
Conclusion
The arsenic content in paddy grains depends on the arsenic contents in irrigation groundwater, paddy soils, and paddy varieties. The soil arsenic content is influenced by different soil properties, especially soil texture, carbon, macronutrients, phosphorus, sulfur, hydrolases, and oxidoreductases. Among paddy varieties grown in the studied region, the Swarana sub-1, Sarjoo-52, BPT-3291, IPB-1, and Arize-6444 accumulated high arsenic in their roots and grains, while the Bengal Juhi, Kalanamak, NDR-359, and BPT-5204 were less responsive. These arsenic accumulating paddy varieties are popularly grown in the middle Indo-Gangetic Plains and the consumption of produce of these varieties may be unsuitable for the people’s health. The study provided information about the magnitude of the arsenic contamination in the water-soil-paddy continuum in the middle IGP region. This study reveals that arsenic contamination to paddy crop in the IGP is affected not only by paddy genotypes but also by the field site and the site-specific environmental factors, like physicochemical properties of paddy soil. It is suggested that regular arsenic monitoring studies should be conducted by the regulatory agencies to determine the increasing magnitude of arsenic accumulation in the agricultural soils and the crop produces.
References
Abedin, M. J., Feldmann, J., & Meharg, A. A. (2002). Uptake kinetics of arsenic species in rice plants. Plant Physiology, 128, 1120–1128.
Ahamed, S., et al. (2006). Arsenic groundwater contamination and its health effects in the state of Uttar Pradesh (UP) in upper and middle Ganga plain. India: a severe danger. Science of the Total Environment, 370, 310–322. doi:10.1016/j.scitotenv.2006.06.015.
Alam, M., Rahman, M. (2003). Accumulation of arsenic in rice plant from arsenic contaminated irrigation water and effect on nutrient content. Paper presented at the BUET-UNU International symposium of fate of arsenic in the environment, Dhaka, Bangladesh.
Banerjee, M., et al. (2013). High arsenic in rice is associated with elevated genotoxic effects in humans. Science Reports, 3, 2195.
Bhattacharya, P., Welch, A. H., Stollenwerk, K. G., McLaughlin, M. J., Bundschuh, J., & Panaullah, G. (2007). Arsenic in the environment: Biology and Chemistry. Science of the Total Environment, 379, 109–120. doi:10.1016/j.scitotenv.2007.02.037.
Bhattacharyya, P., Tripathy, S., Kim, K., & Kim, S. H. (2008). Arsenic fractions and enzyme activities in arsenic-contaminated soils by groundwater irrigation in West Bengal. Ecotoxicology and Environment Safety, 71, 149–156. doi:10.1016/j.ecoenv.2007.08.015.
Black, C. A. (ed) (1965) Method of Soil Analysis. Part 2, Chemical and Microbiological Properties. American Society of Agronomy, Inc, Publisher, Madison, Wisconsin USA.
Brammer, H., & Ravenscroft, P. (2009). Arsenic in groundwater: a threat to sustainable agriculture in South and South-east Asia. Environmental International, 35, 647–654. doi:10.1016/j.envint.2008.10.004.
Bundschuh, J., et al. (2012). Arsenic in the human food chain: the Latin American perspective. Science of the Total Environment, 429, 92–106. doi:10.1016/j.scitotenv.2011.09.069.
Casentini, B., Hug, S. J., & Nikolaidis, N. P. (2011). Arsenic accumulation in irrigated agricultural soils in Northern Greece. Science of the Total Environment, 409, 4802–4810. doi:10.1016/j.scitotenv.2011.07.064.
Chakraborti, D., et al. (2004). Groundwater arsenic contamination and its health effects in the Ganga-Meghna-Brahmaputra plain. Journal of Environmental Monitoring, 6, 74–83.
Das, H. K., Mitra, A. K., Sengupta, P. K., Hossain, A., Islam, F., & Rabbani, G. H. (2004). Arsenic concentrations in rice, vegetables, and fish in Bangladesh: a preliminary study. Environmental International, 30, 383–387. doi:10.1016/j.envint.2003.09.005.
Das, S., Jean, S. H., Kar, S., & Chakraborty, S. (2013). Effect of arsenic contamination on bacterial and fungal biomass and enzyme activities in tropical arsenic-contaminated soils. Biology and Fertility of Soils, 49, 757–765.
Dick, R. P. (ed) (2011) Methods of Soil Enzymology. Issue 9 of Soil Science Society of America book series. Soil Science Society of America, Madison, WI, USA.
Fernandez, P., Sommer, I., Cram, S., Rosas, I., & Gutierrez, M. (2005). The influence of water-soluble As(III) and As(V) on dehydrogenase activity in soils affected by mine tailings. Science of the Total Environment, 348, 231–243. doi:10.1016/j.scitotenv.2004.12.065.
Ghosh, A. K., Bhattacharyya, P., & Pal, R. (2004). Effect of arsenic contamination on microbial biomass and its activities in arsenic contaminated soils of Gangetic West Bengal. India Environmental International, 30, 491–499. doi:10.1016/j.envint.2003.10.002.
Hite, A. H. (2013). Arsenic and rice: a call for regulation. Nutrition, 29, 353–354. doi:10.1016/j.nut.2012.10.001.
Hossain, M. B., Jahiruddin, M., Panaullah, G. M., Loeppert, R. H., Islam, M. R., & Duxbury, J. M. (2008). Spatial variability of arsenic concentration in soils and plants, and its relationship with iron, manganese and phosphorus. Environmental Pollution, 156, 739–744. doi:10.1016/j.envpol.2008.06.015.
Hua, B., Yan, W., Wang, J., Deng, B., & Yang, J. (2011). Arsenic accumulation in rice grains: effects of cultivars and water management practices. Environmental Engineering Science, 28, 591–596.
Islam, M. R., Salminen, R., & Lahermo, P. W. (2000). Arsenic and other toxic elemental contamination of groundwater, surface water and soil in Bangladesh and its possible effects on human health. Environmental Geochemistry and Health, 22, 33–53.
Karaca, A., Cetin, S. C., Turgay, O. C., & Kizilkaya, R. (2010). Effects of heavy metals on soil enzyme activities. In I. Sherameti & A. Varma (Eds.), Soil heavy metals (pp. 237–262). Berlin, Germany: Springer.
Khan, M., Islam, M., Panaullah, G., Duxbury, J., Jahiruddin, M., & Loeppert, R. (2009). Fate of irrigation-water arsenic in rice soils of Bangladesh. Plant and Soil, 322, 263–277.
Koo, N., Lee, S., & Kim, J. (2012). Arsenic mobility in the amended mine tailings and its impact on soil enzyme activity. Environmental Geochemistry and Health, 34, 337–348.
Lei, M., Tie, B., Zeng, M., Qing, P., Song, Z., Williams, P. N., & Huang, Y. (2012). An arsenic-contaminated field trial to assess the uptake and translocation of arsenic by genotypes of rice. Environmental Geochemistry and Health, 35, 379–390.
Meharg, A. A., & Rahman, M. M. (2003). Arsenic contamination of Bangladesh paddy field soils: implications for rice contribution to arsenic consumption. Environmental Science and Technology, 37, 229–234.
Mukherjee, A. B., Bhattacharya, P., Jacks,G., Banerjee, D. M., Ramanathan, A. L., Mahanta, C. (2006). Groundwater arsenic contamination in India: Extent and severity. In: R. Naidu ES, G. Owens, P. Bhattacharya, & P. Nadebaum (ed) Managing arsenic in the environment: From soil to human health CSRIO Publishing, Melbourne, pp 553–594.
Norton, G. J., et al. (2009a). Environmental and genetic control of arsenic accumulation and speciation in rice grain: comparing a range of common cultivars grown in contaminated sites across Bangladesh, China, and India. Environmental Science and Technology, 43, 8381–8386. doi:10.1021/es901844q.
Norton, G. J., et al. (2009b). Identification of low inorganic and total grain arsenic rice cultivars from Bangladesh. Environmental Science and Technology, 43, 6070–6075.
Norton, G. J., et al. (2010). Arsenic shoot-grain relationships in field grown rice cultivars. Environmental Science and Technology, 44, 1471–1477. doi:10.1021/es902992d.
Norton, G. J., et al. (2012). Variation in grain arsenic assessed in a diverse panel of rice (Oryza sativa) grown in multiple sites. New Phytologist, 193, 650–664. doi:10.1111/j.1469-8137.2011.03983.x.
Panaullah, G., et al. (2009). Arsenic toxicity to rice (Oryza sativa L.) in Bangladesh. Plant and Soil, 317, 31–39.
Patel, K. S., Shrivas, K., Brandt, R., Jakubowski, N., Corns, W., & Hoffmann, P. (2005). Arsenic contamination in water, soil, sediment and rice of central India. Environmental Geochemistry and Health, 27, 131–145. doi:10.1007/s10653-005-0120-9.
Rahman, M. A., Hasegawa, H., Rahman, M. M., & Miah, M. A. (2007). Accumulation of arsenic in tissues of rice plant (Oryza sativa L.) and its distribution in fractions of rice grain. Chemosphere, 69, 942–948. doi:10.1016/j.chemosphere.2007.05.044.
Rahman, M. A., Hasegawa, H., Rahman, M. M., Miah, M. A. M., & Tasmin, A. (2008). Arsenic accumulation in rice (Oryza sativa L.): human exposure through food chain. Ecotoxicology and Environment Safety, 69, 317–324. doi:10.1016/j.ecoenv.2007.01.005.
Rahman, M. M., Naidu, R., & Bhattacharya, P. (2009). Arsenic contamination in groundwater in the Southeast Asia region. Environmental Geochemistry and Health, 31, 9–21. doi:10.1007/s10653-008-9233-2.
Sidhu, S. S., Brar, J. S., Biswas, A., Banger, K., & Saroa, G. S. (2012). Arsenic contamination in soil-water-plant (rice, Oryza sativa L.) continuum in central and sub-mountainous Punjab, India. Bull Environ Contam Toxicol, 89, 1046–1050. doi:10.1007/s00128-012-0799-0.
Singh, M., Singh, A. K., Srivastava, S. N., Singh, S., & Chowdhary, A. K. (2010). Arsenic mobility in fluvial environment of the Ganga Plain, Northern India. Environmental Earth Sciences, 59, 1703–1715.
Sinha, B., & Bhattacharyya, K. (2011). Retention and release isotherm of arsenic in arsenic-humic/fulvic equilibrium study. Biology and Fertility of Soils, 47, 815–822.
Srivastava, P. K., Gupta, M., Upadhyay, R. K., Sharma, S., Shikha, S. N., Tewari, S. K., & Singh, B. (2012). Effects of combined application of vermicompost and mineral fertilizer on the growth of Allium cepa L. and soil fertility. Journal of Plant Nutrition and Soil Science, 175, 101–107.
Srivastava, P. K., Singh, M., Singh, N., & Tripathi, R. D. (2013). Soil arsenic pollution: a threat to crops. Journal of Bioremediation & Biodegradation, 4, 7.
Srivastava, P. K., Singh, P. C., Gupta, M., Sinha, A., Vaish, A., Shukla, A., Singh, N., & Tewari, S. K. (2011a). Influence of earthworm culture on fertilization potential and biological activities of vermicomposts prepared from different plant wastes. Journal of Plant Nutrition and Soil Science, 174, 420–429.
Srivastava, P. K., Vaish, A., Dwivedi, S., Chakrabarty, D., Singh, N., & Tripathi, R. D. (2011b). Biological removal of arsenic pollution by soil fungi. Science of the Total Environment, 409, 2430–2442. doi:10.1016/j.scitotenv.2011.03.002.
Srivastava, S., & Sharma, Y. K. (2013). Arsenic occurrence and accumulation in soil and water of eastern districts of Uttar Pradesh, India. Environmental Monitoring and Assessment, 185, 4995–5002.
Stroud, J. L., et al. (2011). The dynamics of arsenic in four paddy fields in the Bengal delta. Environmental Pollution, 159, 947–953. doi:10.1016/j.envpol.2010.12.016.
Su, Y. H., McGrath, S. P., & Zhao, F. J. (2010). Rice is more efficient in arsenite uptake and translocation than wheat and barley. Plant and Soil, 328, 27–34.
Van Zwieten, L., Ayres, M. R., & Morris, S. G. (2003). Influence of arsenic co-contamination on DDT breakdown and microbial activity. Environmental Pollution, 124, 331–339.
WHO (2005). Water sanitation and health. Guidelines for drinking water quality. 3rd edn. World Health Organization.
Williams, P. N., Prince, A. H., Raab, A., Hossain, S. A., Feldmann, J., & Meharg, A. A. (2005). Variation in arsenic speciation and concentration in paddy rice related to dietary exposure. Environmental Science and Technology, 39, 5531–5540.
Wu, C., Ye, Z. H., Shu, W. S., Zhu, Y. G., & Wong, M. H. (2011). Arsenic accumulation and speciation in rice are affected by root aeration and variation of genotypes. Journal of Experimental Botany, 62, 2889–2898. doi:10.1093/jxb/erq462.
Yan, W., Agrama, A. H., Slaton, A. N., & Gibbons, W. J. (2008). Soil and plant minerals associated with rice straighthead disorder induced by arsenic. Agronomy Journal, 100, 1655.
Zavala, Y. J., & Duxbury, J. M. (2008). Arsenic in rice: I. Estimating normal levels of total arsenic in rice grain. Environmental Science and Technology, 42, 3856–3860.
Zhao, F. J., McGrath, S. P., & Meharg, A. A. (2010). Arsenic as a food chain contaminant: mechanisms of plant uptake and metabolism and mitigation strategies. Annual Review of Plant Biology, 61, 535–559.
Zheng, M. Z., et al. (2011). Spatial distribution of arsenic and temporal variation of its concentration in rice. New Phytologist, 189, 200–209. doi:10.1111/j.1469-8137.2010.03456.x.
Acknowledgments
The authors gratefully acknowledge financial support for this work from the Department of Biotechnology, Govt. of India. Authors are also thankful to the Director, CSIR–National Botanical Research Institute, Lucknow, India, for providing the institutional support. Ms. Manvi Singh, UGC-SRF (ref. no. 3185/NET-DEC-2011) is thankful to UGC for financial support to her as SRF. The contributions of Prof. R. S. Tripathi and Prof. S. A Ranade for required linguistic correction of the paper are also gratefully acknowledged.
Compliance with ethical standards
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Figure S1
Correlations among soil, root and grain arsenic (As) contents of R&D based released paddy varieties grown in the middle IGP region. (DOC 54 kb)
Figure S2
Correlations among soil, root and grain arsenic (As) contents of traditional paddy varieties grown in the middle IGP region. (DOC 40 kb)
Figure S3
Correlations among soil, root and grain arsenic (As) contents of company hybrids paddy varieties grown in the middle IGP region. (DOC 44 kb)
Table S1
GPS details of all 18 blocks in different districts as project sites in the middle IGP region of the state of Uttar Pradesh, India. (DOC 41 kb)
Table S2
Different soil physical properties (Mean ± SE) of paddy soils in the middle IGP region. (DOC 54 kb)
Table S3
Different soil chemical properties (Mean ± SE) of paddy soils in the middle IGP region. (DOC 87 kb)
Table S4
Different soil enzyme activities (Mean ± SE) of paddy soils in the middle IGP region. (DOC 57 kb)
Rights and permissions
About this article
Cite this article
Srivastava, P.K., Singh, M., Gupta, M. et al. Mapping of arsenic pollution with reference to paddy cultivation in the middle Indo-Gangetic Plains. Environ Monit Assess 187, 198 (2015). https://doi.org/10.1007/s10661-015-4418-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-015-4418-5