Abstract
This investigation was carried out on commercial organic field in the Fayoum Governorate of Egypt under conditions of natural infection, 2020/2021 and 2021/2022 to evaluate the effectiveness of four commercial biocides—Blight Stop (Trichoderma harzianum), Bio Zeid (T. album), Root Guard (Bacillus subtilis), and Bio ARC (B. megaterium)—as well as, four biocontrol agents (T. harzianum, T. album, B. subtilis, and B. megaterium) in controling Cercospora beticola Sacc, the main causal of Cercospra leaf spot (CLS) disease on sugar beet in an organic farming systems. All biocontrol agents and commercial biocides sprayed at the recommended dose in a two spray regime with 15 days between sprays, were significantly reduced Cercospora beticola, total amino acid and juice impurities (K, Na and α-a N %) in comparison to control treatment. All bioagents and biocides put to the test resulted in a significant rise in phenolic compound values, total chlorophyll, sucrose (%), purity (%), root, top and sugar yield quality of sugar beet during both seasons. Spraying Blight Stop was the most effective treatment followed by T. harzianum. B. megaterium was the least effective biocide treatment compared with the control treatment during the two growing seasons.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In Egypt, sugar beet (Beta vulgaris L.), which is ranked as the second-largest sugar crop after sugar cane, is a crop of the temperate regions (Eweis et al., 2006 and Amer et al., 2019).
Many pathogenic fungi affect sugar beet plants leading to severe disease and yield loss. The most devastating foliar disease of sugar beet, Cercospora leaf spot (CLS) is caused by Cercospora beticola Sacc., damages the leaves, negatively affects plant photosynthetic ability, induces biochemical changes in amino acids, phenols, and sugar that decrease sugar yields to 30 and 50%, respectively (Wolf & Verret, 2002; Wolf & Vereet, 2005; Weiland & Koch, 2004; Kaiser et al., 2010 and Skaracis et al., 2010).
CLS reduces root and extractable sucrose yields and raise impurity concentrations, which increases processing losses (Farahat, 2018 and Morsy et al., 2022).
The main methods of disease control include the use of fungicides, the development of resistant cultivars, and crop rotation (Morsy et al., 2022; Tedford et al., 2019). Synthetic fungicides have been applied repeatedly and widely over the past ten years with detrimental effects to the environment and human health.
Biological control as an ecofriendly method providing a logical substitute for synthetic fungicides for the control of various diseases (Bharathi et al., 2004; Galletti et al., 2008 Shahraki et al., 2008; Jacobsen, 2010 and Derbalah et al., 2013).
Many bacterial isolates of Bacillus spp. and fungal isolates of Trichoderma spp. produce antibiotics enzymes, and show mycoparasitism, where a strain of fungus or bacterium preys on other fungi (Harman, 2000). Due to the elicitation of systemic resistance, repeated applications of Bacillus spp. decrease sugar beet CLS symptoms under field conditions (Bargabus et al., 2002).
Different bioagents, including Trichoderma harzianum, T. viride, Gliocladium virens, Bacillus subtilis, and Pseudomonas fluorescens, are successful in reducing disease incidence and increase yield when compared to control treatments (Srivastava, 2004; Patel & Jasrai, 2012; Ray & Swain, 2013 and Sharma, 2015). Trichoderma spp. produce lytic enzymes such as chitinases, peroxidases, polyphenoloxidases, and glucan 1–3 -glucosidases that damaged the pathogen's cell wall. B. subtilis creates a collection of enzymes that decrease the strength of the pathogen's cell wall, and produce volatile chemicals, phytotoxic substances, and antibiotics like bacterocin and subtilisin (Jacobsen et al., 2004 and Muthuvelayudham & Viruthagiri, 2006).
In vitro, Trichoderma koningii (T1), Bacillus subtilis (B1), and Bacillus subtilis (B2) have a strong antagonistic effect on Cercospora beticola. In addition, increased enzyme activities of peroxidase (POX), catalase (CAT), and polyphenol oxidase (PPO) reduce the severity of CLS. As a result of using the product Eminent's superior control, root productivity, total soluble solids (T.S.S.), sucrose percentage, and chlorophyll content improved (Chen et al., 2022 and Hamden et al., 2023).
The purpose of this study was to evaluate the effectiveness of specific recommended biocontrol agents to control Cercospora leaf spot on sugar beet under organic field conditions and to identify which application regimen could be used to manage the disease, produce sugar with high quality and quantity without toxicity at the food chain.
Material and methods
Plant components
Seeds of Sugar beet cv. Gloria were obtained from the Sugar Crops Research Institute (SCRI), Agricultural Research Center, ARC, Giza Governorate, Egypt. Field tests were conducted throughout 2020/2021 and 2021/2022 at Fayoum Governorate, Egypt.
Biological antagonists
Trichoderma harzianum, T. album, Bacillus subtilis and B. megaterium were added at the rate of 1 Lit./50 Lit. water. These biocontrol organisms were kindly provided by the Biological Control Production Unit, Central Laboratory of Organic Agriculture, CLOA, ARC.
Biocide products
The biocide formulations were used as a comparison with other treatments as follow:-
-
Blight Stop is recommended as biocide preparation. It contains Trichoderma harzianum (30 × 106 spore/ml). This product was used at a rate of 1 Lit./50 Lit. water and was kindly provided by the Biological Control Production Unit Central Lab. of Organic Agriculture, CLOA; ARC.
-
Root Guard, a biocide preparation that was kindly provided by the Biological Control Production Unit Central Lab of Organic Agriculture, CLOA; ARC is recommended. It contains Bacillus subtilis (30 × 106 cell/ml) and is used at a rate of 1 Litre per 100 Litres of water.
-
Bio Zeid 25% WP, which was provided by Kafr El Zayat Pesticides and Chemicals (kz), is recommended as a biocide preparation. It contains T. album (2.5% w/w) and is used at a rate of 250 g/100 L water.
-
Bio ARC 6% WP, a biocide preparation that was kindly provided from kz firm is a recommended control product. It contains B. megaterium (6% w/w) and is used at a rate of 250 g/100 L water.
Isolation of Cercospora leaf spot
In a private organic farm in the Fayoum Governorate, during the 2020–2021 season, Cercospora leaf spot disease was discovered in sugar beet cv. Gloria leaves. 20 infected leaves were collected, washed carefully with tap water, cut into small pieces, surface sterilized in 3% sodium hypochlorite solution for three minutes, repeatedly washed in sterile distilled water, dried between two sheets of sterile filter paper (v/v) and transferred onto Petri dishes (9 cm) containing sugar beet leaf extracts dextrose agar medium (SBLEDA). The fresh sugar beet leaves sliced and 200 g was boiled in one liter distilled water for 15 min and strained through double layers of cheese cloth. The SBLEDA medium consists of sugar beet leaf extract (100 ml), dextrose (20 g), and agar (15 g) medium. Plates were incubated at 27 ± 2 °C for 3–7 days and examined daily for occurrence of fungal growth. Fungal growth was examined microscopically and then purified using the hyphal tip method (Dhingra & Sinclair, 1995 and Morsy et al., 2022). Pure cultures of each isolate were maintained on PDA at 4 °C for further examination.
Identification of Cercospora leaf spot
Identification according to the fungal cultural, phytopathological, and microscopic traits (Alexopoulos et al., 1996). Based on identification, one of them was chosen for further examination.
Antagonistic effect of bacterial strains against Cercospora beticola
The antagonists and the commercial preparation suspensions were added to warm sterilized PDA medium at the rate of 10% and poured before solidification into Petri dishes (10 ml/plate). After solidification, a disc (5 mmØ) of Cercospora beticola obtained from the periphery of 7-days old mycelium on the same medium was placed in the center of each plate. Plates containing media without antagonists and inoculated only with abovementioned pathogen served as control treatment. Three plates were used for each treatment. Inoculated plates were incubated at 27 ± 2 °C. The experiment was terminated when mycelial mats covered the surface in the control treatment, all plates were examined and the percentage reduction in mycelial growth of the fungus was calculated using the formula suggested by Ahmed (2005) and Ahmed (2013) as following:
where: G1: growth of the pathogenic fungus in control only, G2: growth of the pathogen against the tested antagonists.
Field trials
In Vivo studies were carried out at a private organic farm, Fayoum Governorate, Egypt, which has a long history of severe infection by Cercospora leaf spot disease (CLS) The experiments assess the effectiveness of different antagonists for the control of Cercospora beticola. Three replicated plots were used in a complete randomized block design for all of the trials. The experimental plot's surface area was 21 m2, made up of 3 rows (6 m × 50 cm) 50 cm apart. Each row was sown with 30 Sugar beet seeds cv. Gloria and were exposed to natural inoculum only. Sugar beet seeds cv. Gloria were planted on 15th October during the two growing seasons, 2020/21 and 2021/22, respectively.
The cultivation, irrigation and compost fertilization, were applied in an equal amount to every plot 15 days before sowing. After 90 days of cultivation, sugar beet plants were sprayed with suspensions of the four bioagent isolates—Trichoderma harzianum, T. album, Bacillus subtilis, and B. megaterium, as well as with the four commercial biocide products, including Blight Stop, Bio Zeid, Root Guard, and Bio ARC at the recommended doses as mentioned above. Super film as a surfactant and sticker material, was mixed with each treatment prior to spraying at a rate of 50 ml/100 L water. All treatments were applied twice with 15 days intervals between each application. Plots that had not been treated (only water sprayed) served as the control.
The following characteristics were evaluated:
Disease severity (DS %) and disease incidence (DI):
14 days after the last treatment disease was scored using the scale developed by Verreet et al. (1996), where 0 = no symptoms; 1 = 1–20%; 2 = 21–40%; 3 = 41–60%; 4 = 61–80%; and 5 = 81–100% infected leaf area, The following formula was used to calculate each foliar disease's severity percentage:
where, D.S.I = disease severity index, n = number of leaves in each category, c = numerical value of each category, C = numerical value of highest category and N = total number of leaves in the sample.
The percentages disease incidence and the efficacy of the tested treatments were estimated using the following equations:
Quality control criteria:-
-
1-Sugar beet total yield
The average root weight per fed. area was evaluated (Mahmoud et al., 2012).
-
2-Sugar, root, and top yield of sugar beet plants (ton/fed.)
The yield tops and roots (ton/fed.) was calculated at harvest. Root yield was multiplied by sucrose % to calculate sugar yield (ton/fed.). According to the method described by McGinnus (1982), quality parameters such as sucrose percentage and impurity content (K, Na, and alpha-amino N %) were measured by the Faiyum Sugar Works Company, Fayoum Governorate. Juice purity was determined using the formula provided by Devillers (1988).
-
3-Total soluble solids (T.S.S %)
Total soluble solids (T.S.S %) in fresh roots were evaluated using a manual refractometer according to McGinnis (1982).
-
4- Chlorophyll content
The total chlorophyll content was determined using method of Moran (1982).
Biochemical changes in sugar beet plants due to application of chemicals
Total phenol
The amount of phenol was calculated using the typical graph made with different catechol concentrations. The catechol equivalents of the phenol content were calculated as mg/g of fresh tissue (Turkmen et al., 2005).
Statistical analysis
All data collected over two successive seasons were statistically analysed and compared using the least significant difference (L.S.D.) at 5% proposed by Snedecor and Cochran (1989).
Results
Effect of different antagonists on the growth of Cercospora beticola
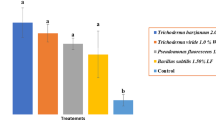
The data in (Table 1) demonstrate that the antagonists’ inhibited the growth of Cercospora beticola in vitro. T. harzianum greatly reduced mycelial growth by 89.60%, followed by T. album (87.50%). The smallest control was found with Bio ARC (68.10%)
The effect of different antagonists on sugar beet Cercospora leaf spot under field conditions during the 2020/21 and 2021/22 growing seasons:
-
1. Diseases parameters
The results in (Table2) indicate that, all tested biological control treatments (Trichoderma harzianum, T. album, Bacillus subtilis, B. megaterium, Blight Stop, Bio Zeid, Root Guard, and Bio ARC) significantly outperformed the control treatment in reducing the incidence and severity of sugar beet Cercospora leaf spot disease in the two growing seasons. Blight Stop biocide showed the best efficacy (78.72 and 89.43%), followed by T. harzianum isolate (77.33 and 86.37%). On the other hand, B. megaterium had the lowest effectiveness (68.62 and 68.98%) in controlling the Cercospora beticola leaf spot disease in both seasons compared with control treatment.
-
2. Total phenols, total chlorophyll and total amino acids content
Presented data in (Table 3) illustrate that all t biological treatments increased total chlorophyll and phenols in comparison with untreated plants during both growing seasons. The biocide Blight Stop provided the highest level of total chlorophyll (79.70 and 80.11) and phenols (10.19 and 10.25) in the majority of cases, followed by T. harzianum. In contrast, B. megaterium demonstrated the smallest effectiveness (72.27 and 73.17 in total chlorophyll and 6.30 and 6.32 in total phenols).
Compared to the control treatment, all biological treatments reduced the total amino acid consentration in sugar beet roots throughout the two growing seasons. Untreated plants had the highest levels of amino acids in both growing seasons. Amino acid levels were found to be lowest in sugar beet plants that had been treated with Blight Stop. B. megaterium produced the highest level of amino acid content.
-
3. Quality of commercial sugar beet production and juice impurities (K, Na and α-amino N %):
The results, which are presented in (Table 4) show that, as compared to the untreated control, all tested treatments considerably reduced juice impurities (K, Na and -amino N%) in comparison with the control treatment. The Blight Stop treatment had the highest reduction in juice impurities followed by Trichoderma harzianum during the two successive growing seasons compared with control treatment.The B. megaterium treatment was the least effective in reducing impurities.
-
4. Total soluble solids (TSS), Sucrose % and Juice purities:
The data (Table 5) demonstrate that all tested biological treatments significantly increased total soluble solids (TSS), sucrose, and purity percentage when compared to the untreated plants. Spraying Blight Stop resulted in the largest increase in TSS, sucrose, and purity during both seasons, followed by T. harzianum. In contrast, B. megaterium was the least effective treatment compared to the untreated control during the two growing seasons 2020/21 and 2021/22.
-
5. Root, top and sugar yields:
Regarding the influence of biological treatments on root and recoverable sugar yields/fed. (Table 6), all tested treatments significantly increased root, top and sugar yields throughout both seasons compared with untreated plants.
The most superior treatments increased the root, top and sugar yields was observed when sugar beet plants were sprayed with Blight Stop twice, followed by T. harzianum compared to the other treatments. The B. megaterium treatment showed the least effect on in root, top and sugar yields in comparison with the control treatment.
Results in Tables 4, 5 and 6 shows that, in comparison to the untreated control all tested treatments resulted in high values for the quality traits (sucrose and purity percentages), root, top and sugar yields of sugar beet, and lowest juice impurity. This discovery may be attributed to a decline in impurities, such as sodium, potassium, and alpha amino-N levels, as well as a decline in disease incidence and severity, which had an impact on root production and sugar content.
Discussion
In recent years, farmers have become aware that using chemical pesticides may be harmful to the environment and to human health and may contribut to the emergence of new pests, reduce the number of natural enemies. In order to produce high-quality food in sufficient quantities and enhance biodiversity, the current work aimed to reduce the use of toxic chemicals in agriculture. In addition, we aimed to find the best non-chemical methods. to protect sugar beet plants from Cercospora beticola leaf spot disease.
Data in Table 1 showe that, T. harzianum greatly reduced mycelial growth, followed by T. album, Blight Stop "T. harzianum", Bio Zeid "T. album", B. subtilis, Root Guard "B. subtilis", and B. megaterium. Bio ARC's "B. megaterium" had the least impact.. Bacillus subtilis held the second position after Trichoderma spp., this may be because it generates a group of enzymes that degrate the pathogen's cell wall (Ahmed, 2005 and Ahmed, 2013), antibiotics such as bacterocin and subtilisin (Hamden et al., 2023), volatile compounds and phytotoxic substances (Stein, 2005). This phenomenon could be explained by the fact that different infections with distinct striations have unique defence mechanisms against the enzymes and toxic materials produced by different antagonists. (Ahmed, 2005, 2013; Muthuvelayudham & Viruthagiri, 2006). Trichoderma species produce lytic enzymes such as chitinases, peroxidases, polyphenoloxidases, and glucan 1–3 B-glucosidases that destroyed the pathogen's cell wall (Jacobsen et al., 2004 and Derbalah et al., 2013). Furthermore, Trichoderma are known to be able to cause systemic acquired resistance (SAR), and this is thought to be one of the most crucial modes of action for this biocontrol agent. This has been described for a range of plant-pathogen systems (Harman et al., 2004).
According to the available data (Table 2), all tested biological control treatments (Trichoderma harzianum, T. album, Bacillus subtilis, B. megaterium, Blight Stop, Bio Zeid, Root Guard, and Bio ARC) significantly outperformed the control treatment in reducing the incidence and severity of sugar beet cercospora leaf spot disease in the two growing seasons of 2020–2021 and 202–2022. The results can be evaluated using both the chemical impact of antioxidants, which clearly improve plant physiology and metabolism and induce systemic resistance (ISR), and the action of biotic factors, which provide growth regulators (Harman et. al., 2004). Due to competition for oxygen, nutrients, and space as well as their capacity for fungal mycoparasitism (El-Sayed et al., 2017) and the secretion of antibiotics, Trichoderma spp. and/or Bacillus spp. have direct effects on pathogenic fungi (Patel & Jasrai, 2012, and Sharma, 2015). Trichoderma spp. can also create antifungal compounds such as trichodermin, alpha-1,3-glucanase, beta-glucosidase, and endo chitinase (Galletti et al., 2008, Stefania et al., 2008 and Hamden et al., 2023).
The biocide Blight Stop provided the highest levels of total chlorophyll and phenols in the majority of cases, followed by T. harzianum as shown in Table 3. In order to reduce the pathogen's harmful effects, bioagents may provide the nutrients and biological elements needed to enhance photosynthesis in the host plants which decrease disease incidence and severity (%), decrease the loss of photosynthetic leaf area, and decrease toxicity from Cercosporin-related toxins, which have a negative impact on plant health and photosynthesis (Scholes & Rolfe, 2009 and El–Mansoub et al., 2017). Cercospora beticola infection of sugar beet impacts chlorophyll and phenol concentration even before causing symptoms, and imaging raw chlorophyll fluorescence allowed for presymptomatic detection of the necrotrophic fungal disease, C. beticola, in sugar beet leaves (Wolf & Vereet, 2005 and Chaerle et al., 2007). The biodegradation of commercial sugar beet production and sugar yield, or the contribution of the pathogen, the hyphae of the fungus absorbing and retaining some of the amino acids for the synthesis of its own proteins, may all contribute to the quantitative increase of specific amino acids in the infected tissues (Rossi et al., 2000; Weiland & Koch, 2004; Kaiser et al., 2010; Skaracis et al., 2010 and El–Mansoub, et al., 2017).
The data in Table 4 suggest that, Blight Stop treatment showed the highest reduction in juice impurities and α-amino N, followed by Trichoderma harzianum during the two successive growing seasons 2020/21 and 2021/22 compared with control treatment. B. megaterium treatment was the least effective. This might be because biological treatments have a role in decreasing disease incidence and severity, which is reflected in a decline in the impurities in juice as argued by Martin et al. (2001) and Hamden et al., 2023.
The data in Table 5 demonstrate that all tested biological treatments significantly increased total soluble solids (TSS), sucrose, and purity percentage when compared to the untreated plants. Spraying Blight Stop resulted in the largest increase in TSS, sucrose and purity percentages during both seasons, followed by T. harzianum. These results are in agreement with those reported by Rossi et al., (2000), Schmittgen, (2015) and Stevens, (2017), who found that BCAs efficiently controlled Cercospora leaf spot, and simultaneously promoted the plants growth (Chen et al., 2022).
The data in Table 6 showe the largest increase the root, top and sugar yield was observed when sugar beet plants were sprayed with Blight Stop twice, followed by T. harzianum compared to the other treatments. The obtained results are in agreement with those reported by Cioni et al. (2004); Stefania et al. (2008), Farahat (2018) and Morsy et al. (2022), who reported that bioagents may increase the nutrients and essential elements required to improve photosynthesis in the host plants in order to reduce the negative effects of the pathogen and increase root and sugar yields. Results in Tables 4,5 and 6 show that, in comparison to the untreated control all tested treatments recorded high values for the quality traits (sucrose and purity percentages), root, top and sugar yields of sugar beet, and lowest juice impurities (K, Na and α-amino N%). This discovery may be attributed to a decline in impurities, such as sodium, potassium, and alpha amino-N levels, as well as a decline in disease incidence and severity, which has an impact on root production and sugar content.
Conclusion
Our finding with the commercial biocides—Blight Stop (Trichoderma harzianum), Bio Zeid (T. album), Root Guard (Bacillus subtilis), and Bio ARC (B. megaterium)—as well as, four biocontrol agents (T. harzianum, T. album, B. subtilis, and B. megaterium) sprayed at the recommended doses with 15 days between sprays show a significant reduction in Cercospora beticola, total amino acid and juice impurities in comparison to control treatment. All bioagents and biocides put to the test showed a significant increase in phenolic compounds, total chlorophyll, sucrose (%), purity (%), root, top and sugar yield quality of sugar beet during both seasons. Spraying Blight Stop was the most effective treatment followed by T. harzianum, however B. megaterium was the least effective biocide treatment compared with control treatment during the two growing seasons 2020/21 and 2021/22.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Ahmed, M. F. A. (2005). Effect of Adding Some Biocontrol Agents on Non-target Microorganisms in Root Diseases Infecting Soybean and Broad bean Plants. M.Sc. Thesis. Fac. Agric., Moshtohor, Benha Univ., 137 p.
Ahmed, M. F. A. (2013). Studies on Non-chemical Methods to Control Some Soil-Borne Fungal Diseases of Bean Plants Phaseolus vulgaris L. Ph.D. Thesis. Fac. Agric., Cairo Univ., pp: 137.
Alexopoulos, C. J., Mims, C. W., & Blackwell, M. (1996). Introductory mycology (4th ed., p. 889p). John willey and Sons.
Amer, M., Aiad, M., Rashed, S., & El-Ramady, H. (2019). Sustainable Irrigation and Fertilization Management of Successive Cultivated Sugar Beet and Cotton under Salt-affected Soil Conditions. Env. Biodiv. Soil Security, 3, 227–239.
Bargabus, R. L., Zidack, N. K., Sherwood, J. E., & Jacobsen, B. J. (2002). Characterization of systemic resistance in sugar beet elicited by a non-pathogenic, phyllosphere-colonizing Bacillus mycoides, biological control agent. Physiological and Molecular Plant Pathology, 61, 289–298.
Bharathi, R., Vivekananthan, R., Harish, S., Ramanathan, A., & Samiyappan, R. (2004). Rhizobacteria-based bio-formulations for the management of fruit rot infection in chilies. Crop Protection, 23(6), 835–843.
Chaerle, L., Hagenbeek, D., De Bruyne, E., & Van Der Straeten, D. (2007). Chlorophyll fluorescence imaging for disease-resistance screening of sugar beet. Plant Cell, Tissue and Organ Culture, 91(2), 97–106.
Chen, Y., Aramrueang, N., Zicari, S., & Zhang, R. (2022). Process development of sugar beet enzymatic hydrolysis with enzyme recycling for soluble sugar production. Bioprocess and Biosystems Engineering, 45(8), 1297–1309.
Cioni, F., Tugnoli, V., & Zavanella, M. (2004). Cercospora leaf spot: the effects on sugar content reduction. In Proc. 67th IIRB Congress, pp. 61–72.
Derbalah, A. S., El-Moghazy, S. M., & Godah, M. I. (2013). Alternative Control Methods of Sugar-beet Leaf Spot Disease Caused by the Fungus Cercospora beticola (Sacc). Egyptian Journal of Biological Pest Control, 23(2).
Devillers, P. (1988). Prevision Du Sucre Melasse. Sucrerie Francaise, 129, 190–200.
Dhingra, O. D., & Sinclair, J. B. (1995). Basic Plant Pathology Methods (2nd ed., p. 434p). CRC Press.
El–Mansoub, M., Fattah, E. A., & Ibrahim, M. (2017). Efficiency of Some Biocides as Safe Alternatives to Fungicides on Cercospora Leaf Spot Control, their Effect on Biochemical Constituent and Yield of Sugar Beet. Egyptian Journal of Phytopathology, 45(2), 145–164.
El-Sayed, A. N., El-Shehawy, A., El-Shabrawy, E. S., & Alkolaly, A. (2017). Safely Application of some Fungicides to Control Cercospora Leaf Spot Disease of Sugar Beet. Egyptian Journal of Phytopathology, 45(2), 219–230.
Eweis, M., Elkholy, S. S., & Elsabee, M. Z. (2006). Antifungal efficacy chitosan and its thiourea derivatives upon the growth of some sugar beet pathogen. International Journal of Biological Macromolecules, 38, 1–8.
Farahat, G. A. (2018). Biosynthesis of Nano Zinc and Using of Some Nanoparticles in Reducing of Cercospora Leaf Spot Disease of Sugar Beet in the Field. Environment, Biodiversity and Soil Security, 2, 103–117.
Galletti, S., Burzi, P. L., Cerato, C., Marinello, S., & Sala, E. (2008). Trichoderma as a potential biocontrol agent for Cercospora leaf spot of sugar beet. BioControl, 53, 917–930.
Hamden, S., Shalaby, M. E., Elkady, E. M., Elfahar, S. A., & El-Emary, S. A. (2023). Potential Impacts of Eminent Fungicide, Certain Bacterial and Fungal Antagonists for Controlling Cercospora Leaf Spot Disease in Sugar Beet. Journal of Sustainable Agricultural Sciences, 49(1), 1–12.
Harman, G. E. (2000). Myths and dogmas of biocontrol: Changes in perception derived from research on Trichoderma harzianum T-22. Plant Disease, 84, 377–393.
Harman, G. E., Howell, C. R., Viterbo, A., Chet, I., & Lorito, M. (2004). Trichoderma species opportunistic, a virulent plant symbionts. Nature Reviews Microbiology, 2(1), 43–56.
Jacobsen, B. J. (2010). Integrated management of Cercospora leaf spot. In R. T. Lartey, J. J. Weiland, L. Panella, P. W. Crous, & C. E. Windels (Eds.), Cercospora leaf spot of sugar beet and related species (pp. 275–284). The American Phytopathological Society.
Jacobsen, B. J., Johnston, M., Zidack, N. K., Eckhoff, J., & Bergman, J. (2004). Effects of high temperatures on Cercospora leaf spot infection and sporulation and effects of variety and number of fungicide sprays on yield. Sugarbeet Research and Extension Reports, 35, 205–208.
Kaiser, U., Kluth, C., & Marlander, B. (2010). Variety specific epidemiology of Cercospora beticola Sacc. and consequences for threshold-based timing of fungicide application in sugar beet. Journal of Phytopathology, 158(4), 296–306.
Mahmoud, E. A., Hassanin, M. A., & Emara, E. I. (2012). Effect of organic and mineral nitrogenous fertilizers and plant density on yield and quality of sugar beet (Beta vulgaris L.). Egyptian Journal of Agronomy, 34(1), 89–103.
Martin, S. S., Narum, J. A., & Chambers, K. H. (2001). Sugar beet biochemical quality changes during factory pile storage. Part II. Non-sugars. Journal of Sugarbeet Research, 38(2), 173–188.
McGinnis, R. A. (1982). Juice purification. In R. A. McGinnis (Ed.), Beet Sugar Technology (pp. 87–103). Beet Sugar Development Foundation: Fort Collins.
Moran, R. (1982). Formulae for determination of chlorophyllous pigments extracted with N N-Dimethylformamide. Plant Physiology, 69(6), 1376–1381.
Morsy, S. Z., Shady, M. F., Gouda, M. I., Kamereldawla, B. A., & Abdelrazek, M. A. S. (2022). A strategy for controlling Cercospora leaf spot, caused by Cercospora beticola, by combining induced host resistance and chemical pathogen control and its implications for sugar beet yield. Canadian Journal of Plant Pathology, 44(4), 518–533.
Muthuvelayudham, R., & Viruthagiri, T. (2006). Fermentative production and kinetics of cellulase protein on Trichoderma reesei using sugarcane bagasse and rice straw. African Journal of Biotechnology, 5(20), 1873–1881.
Patel, R. M., & Jasrai, Y. T. (2012). Bio-control agents (BCAs) as a Potential Tool to Uphold Sustainability of Environment: Types (p. 170). Emerging Trends: Methods and Commercialization. Phytotechnology.
Ray, R.C., & Swain, M. R. (2013). Bio (bacterial) control of pre-and postharvest diseases of root and tuber crops. In D. K. Maheshwari (Ed.), Bacteria in Agrobiology: Disease Management. Chapter: 12 (pp. 321–348). Heidelberg: Springer Berlin.
Rossi, V., Battilani, P., Chiusa, G., Languasco, L., & Racca, P. (2000). Components of rate-reducing resistance to Cercospora leaf spot in sugar beet: Conidiation length, spore yield. Journal of Plant Pathology, 82, 125–132.
Schmittgen, S. (2015). Effects of Cercospora leaf spot disease on sugar beet genotypes with contrasting disease susceptibility. Schriften Des Forschungszentrums Jülich Reihe Energie and Umwelt / Energy and Environment, 244, 1793–1866.
Scholes, J. D., & Rolfe, S. A. (2009). Chlorophyll fluorescence imaging as tool for understanding the impact of fungal diseases on plant performance: A phenomics perspective. Functional Plant Biology, 36(11), 880–892.
Shahraki, M., Heydari, A., Hassanzadeh, N., Rezaei, S., & Naraghi, L. (2008). Investigation on the possibility of biological control of sugar beet seedling damping-off disease. Iranian Journal of Agriculture Science, 13(1), 23–38.
Sharma, P. (2015). Bio fungicides: their role in plant disease management. In New paradigms in diseases management: Conventional and molecular approaches for rapeseed-mustard production, Indian Council of Agricultural Research, Winter School training manual, 113 p.
Skaracis, G. N., Pavli, O. I., & Biancardi, E. (2010). Cercospora leaf spot disease of sugar beet. Sugar Tech, 12, 220–228.
Snedecor, G. W., & Cochran, W. G. (1989). Statistical Methods (8th ed., p. 503). Ames, Iowa USA: Iowa State Univ. Press.
Srivastava, S. N. (2004). Disease management of fruits and vegetables, management of sugar beet diseases, Kluwer Academic Publishers. In K. G. Mukerji (Ed.), Fruit and Vegetable Diseases (Vol. 1, pp. 307–355). Netherlands.
Stefania, G., Pier, L. B., Claudio, C., Simona, M., & Eleonora, S. (2008). Trichoderma as a potential biocontrol agent for Cercospora leaf spot of sugar beet. International Organisation for Biological Control, 53, 917–930.
Stein, T. (2005). Bacillus subtilis antibiotics: Structures, syntheses and specific functions. Molecular Microbiology, 56(4), 845–857.
Stevens, M. (2017). Maximising sugar beet yields via fungicide applications to control foliar diseases. British Sugar Beet Rev., 85(2), 5–8.
Tedford, S. L., Burlakoti, R. R., Schaafsma, A. W., & Trueman, C. L. (2019). Optimizing management of Cercospora leaf spot (Cercospora beticola) of sugar beet in the wake of fungicide resistance. Canadian Journal of Plant Pathology, 41(1), 35–46.
Turkmen, N., Sari, F., & Velioglu, Y. S. (2005). The effect of cooking methods on total phenolic and antioxidant activity of selected green vegetables. Food Chemistry, 93(4), 713–718.
Verreet, J. A., Wolf, P., & Weis, F. J. (1996). Bekämpfungsschwellen als Grundlage für eine integrierte Bekämpfung von Cercospora beticola–Das IPS-Modell Zuckerrübe. Proceedings of the IIRB, 59, 55–69.
Weiland, J., & Koch, G. (2004). Sugar beet leaf spot disease (Cercospora beticola Sacc.). Molecular Plant Pathology, 5(3), 157–166.
Wolf, P. F. J., & Vereet, J. A. (2005). Factors affecting the onset of Cercospora leaf spot epidemics in sugar beet and establishment Disease-Monitoring Thresholds. Phytopathology, 95, 269–274.
Wolf, P. F. J., & Verret, J. A. (2002). An integrated pest management system in Germany for the control of fungal leaf diseases in sugar beet. Plant Disease, 86(4), 336–344.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB). Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
Majority contribution for the whole article belongs to the author. The author read and approved the final manuscript.
Corresponding author
Ethics declarations
Declarations
The authors bear all the ethical responsibilities of this manuscript. They declare that the research was conducted in the absence of any commercial or financial relationship that could be construed as a potential conflict of interest and that it does not include any animal and/or human trials.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The author declares that he has no competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ahmed, M.F.A., Mikhail, S.P.H. & Shaheen, S.I. Performance efficiency of some biocontrol agents on controlling Cercospora leaf spot disease of sugar beet plants under organic agriculture system. Eur J Plant Pathol 167, 145–155 (2023). https://doi.org/10.1007/s10658-023-02729-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-023-02729-5