Abstract
Dissolved organic matter (DOM) plays an important role in promoting or suppressing methylmercury (MeHg) production in wetlands. However, the effects of DOM spectral characteristics on MeHg levels remain poorly understood in boreal peatlands in Northeast China, where is undergoing remarkable climate warming. In the present work, soil samples were collected from 22 peatlands in the Greater Khingan Mountains (GKM) to test the hypothesis that DOM spectral properties control MeHg levels. DOM was characterized by UV–Vis absorption and fluorescence spectroscopy; the three-dimensional fluorescence excitation-emission matrix (EEM) was used to unveil the origin of DOM. The average total mercury (THg) and MeHg contents were 112.76 µg/kg and 12.43 µg/kg across all peatlands, respectively. There was a significantly positive correlation between MeHg and the longitude spanning the range from 120 to 123°E (p < 0.05). Proportions of MeHg to THg (%MeHg), 12.3% on average, were positively correlated with DOM humification degree at p < 0.05 level. Protein-like components of DOM (P-like) were negatively related to %MeHg. DOM had positive effects on THg, and P-like components, HIX and BIX can negatively affect THg as well as MeHg. Our findings demonstrate that the spectral characteristics of DOM in soil are crucial to the content of methyl mercury in the GKM soil.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Mercury (Hg) could be transformed by bacteria into methylmercury, which is with strong lipophilicity, high neurotoxicity and high bioaccumulation (Corrales et al., 2011; Feng et al., 2009; Yang & Wang, 2006). Wetlands could trap Hg for a long time due to the flooded, anaerobic, and carbon-rich soil environments (Bachand et al., 2014), and generally function as Hg sinks (Gong et al., 2014). However, wetlands have high methylmercury (MeHg) and are MeHg sources to the ambient (Jiang et al., 2018; Semeraro et al., 2015). Globally, permafrost regions store 793 Gg Hg in the Northern Hemisphere (Schuster et al., 2018). Previous studies confirmed that climate warming accelerated Hg methylation in soils and sediments from Arctic lake (Hudelson et al., 2020; Yang et al., 2016), permafrost (Mu et al., 2019), tundra (Vasilevich et al., 2014) and wetlands (Lehnherr et al., 2012). MeHg production is usually primarily attributed to biological methylation that is mediated by anaerobic microorganisms, sulfate-reducing bacteria, iron-reducing bacteria, and methanogens. Also, the abiotic methylation process cannot be neglected when electron donors are abundant and present (Celo et al., 2006; Liu & Ding, 2007). The dissolved organic matter (DOM) is one of the most important Hg methylators that could fuel the rapid synthesis of MeHg. Especially, the microbial-derived DOM can stimulate MeHg production in the temperature-sensitive boreal wetlands and permafrost (Lescord et al., 2018), where DOM increasing under climate warming is accompanying more Hg release initially trapped by frozen soil (Barkay et al., 2011; MacMillan et al., 2015; St. Pierre et al., 2014; Yang et al., 2016). Wilds in boreal wetlands may be at risk from elevated MeHg.
Hg methylation is promoted by DOM as electron donors for bacteria (Schaefer & Morel, 2009; Zhang et al., 2011), MeHg variation was not well explained just by DOM contents itself (Schartup et al., 2013), though positive effects of DOM on MeHg were observed in many reports (Graham et al., 2012; Klapstein et al., 2018; Wallschläger et al., 1996). DOM, in addition to concentrations, with high highly aromatic structures were particularly effective at enhancing Hg methylation (Hill et al., 2009; Graham et al., 2012). Fulvic acid is a powerful activator that reactive Hg associated with iron and manganese oxides, thereby stimulating MeHg production (Yao et al., 2004). As a significant fraction of the organic metal-binding ligands, the production of protein-like substances by phytoplankton or microbes could effectively bind with Hg, facilitate Hg utilization by microorganisms, and accelerate MeHg synthesis (He et al., 2010). In lake sediment, organic compounds originating from fresh chlorophyll, proteins and phyto-derived cell wall lipids were demonstrated to fruitfully accelerate MeHg production (Bravo et al., 2017). Although a large body of literature is now available on the interactions of MeHg production and DOM, to the best of our knowledge, how DOM characters affect MeHg production has rarely been examined in boreal peatlands where are susceptible to remarkable climate warming.
The Greater Khingan Mountains (GKM) has the second-largest permafrost area in China and peatlands are widely distributed in the GKM. As the temperature continues to rise in the GKM, permafrost thawing is accelerated and Hg trapped in the frozen soils is now being released (Bracho et al., 2016). DOM contents and spectral characteristics would certainly affect MeHg production in boreal peatlands, but currently, few are available about this topic in the GKM. The objective of this study was to examine relations between MeHg and DOM concentrations and spectral features in peatlands of the GKM, aiming to better our understanding of Hg cycle in boreal peatlands under climate warming.
Materials and methods
Description of the GKM and field sampling
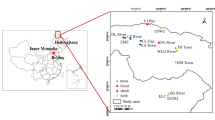
The GKM extends 1200 km from north to south, involving Heilongjiang Province and Inner Mongolia and spanning cold and moderate temperate continental monsoon climate zones (Gao et al., 2016). Widespread wetlands here develop well on the permafrost and seasonally frozen soil. Since 1707, the annual average temperature increased significantly with the tendency rate of 0.06 ℃/10a, and the precipitation increased at the rate of 0.79 mm/10a (Zhao et al., 2016).
A total of 154 surface soil samples were collected from 22 peatlands from 2017 to 2018 (Fig. 1). Sample sites were categorized into four groups, herb (Hc, n = 7), shrub (Sc, n = 8), forest (Fc, n = 5), and moss (Mc, n = 2), according to the predominant vegetation. Soil samples were sealed in polyethylene bags, brought back to the laboratory, air-dried, manually ground with a quartz mortar, hand-sieved through a 100-mesh sieve, and preserved at − 4 ℃ before chemical analysis.
Chemical analysis
Total mercury (THg) was determined according to Zhang et al. (Zheng et al., 2020). In brief, 0.200 g soil samples was digested using 10.0 mL HNO3 (32 N) and 2.5 mL H2SO4 (36 N). All Hg species were determined using an atomic fluorescence spectrometer (AFS-8510, Haiguang Instrument, China).
For MeHg determination, 1.000 g soil sample was soaked in 10 mL 6 N HCl solution overnight; MeHg was then extracted with 5.0 mL dichloromethane (CH2Cl2). The CH2Cl2 was separated and transferred into a heart-shaped bottle. After adding 5.0 mL of deionized water, the heart-shaped bottle was connected with a rotary evaporator at 60℃, and CH2Cl2 was removed while MeHg remained in the deionized water. MeHg was oxidized into Hg2+ by a 1.0 mL brominating agent, and then transformed to Hg0 using hydroxylamine hydrochloride (0.2 g/mL), and finally determined using an atomic fluorescence spectrometer (AFS-8510, Haiguang Instrument, China). Detailed procedures were described in Zhang et al. (Zhang et al., 2009).
The proportions of MeHg to THg (%MeHg) to express the potential of Hg methylation (Weber, 1993) were calculated as the following formula.
Soil organic carbon (SOC), total nitrogen (TN), and total sulfur (Sulfur) contents were analyzed by dry combustion on a Vario EL elemental analyzer (Elementary, Hanau, Germany). DOM was extracted by deionized water at the rate of 1:5 (soil: water) and determined by a total carbon analyzer (TOC-2000, Gangdong, China).
For quality control, standard materials were used to test the precision and accuracy of THg and MeHg determination. The recovery ratios varied in the range of 96 ~ 106%. Each sample was analyzed with three replications. All the chemical reagents were guaranteed reagent, and all glass instruments need to be soaked in 3 N HNO3 for more than 24 h.
The UV–Vis and fluorescence spectrum of DOM
The UV–Vis spectrum and fluorescence spectrum were conducted to characterize DOM source and potential composition. Absorbance measures, analyzed from 190 to 800 nm on a Shimadzu UV–Vis spectrophotometer (UV–2550, Shimadzu, Japan), were used to investigate DOM source, structure, and composition (Table S2). DOM fluorescence was measured on a Hitachi Fluorescence spectrophotometer (F–4700, Hitachi, Japan). Three-dimensional scans were performed at 5 nm excitation steps from 200 to 500 nm, reading emissions at 2 nm steps from 250 to 600 nm, generating excitation-emission matrices (EEMs) for each sample. All spectral corrections were performed using parallel factor analysis (PARAFAC) in the MATLAB software environment.
Three fluorescence indexes were used to judge DOM source and humification degree. The fluorescence index (FI), as the ratio of excitation wavelength (Ex) at 370 nm and emission wavelength (Em) at 470 nm and 520 nm, is used to differentiate DOM sources (Cory et al., 2007). The humification index (HIX) refers to the ratio of integral values of Em at 435 ~ 480 nm and 300 ~ 345 nm when Ex is 254 nm, with higher values indicative of a stronger humification degree (Zsolnay et al., 1999). When HIX < 4, implies that DOM is mainly derived from microbial metabolism. When HIX corresponds 10 ~ 16 for DOM of high levels of humification and DOM is mainly exogenous input (Ohno, 2002). The biological index (BIX) is the ratio of fluorescence intensity of Em at 380 nm and 430 nm when Ex is 310 nm, it can be used to characterize the autogenic property of DOM (Birdwell & Engel, 2010). When BIX < 0.8, the autogenic property of DOM is not obvious. When BIX > 0.8, the autogenic property of DOM is obvious (Huguet et al., 2009).
The UV–Vis absorbance indicated humification degree (represented by SUVA254), the hydrophobic component (represented by SUVA260), and the complexity of soil DOM (represented by A253/A203) varied greatly across different wetlands (Table S3).
Data statistical analysis
All data were standardized to satisfy the normal distribution before analysis. All statistical analysis was carried out using the R software. Principal component analysis was used to test the potential relationship between environmental factors, DOM spectral parameters, THg and MeHg contents. Multiple regression models were implemented to assess the relative importance of each variable for explaining variation in THg, MeHg and %MeHg. All data were standardized before performing the regression analysis. We compared the importance of the different variables using standardized regression coefficients. Statistical significance was tested at 0.05 level.
Results
THg and MeHg contents in wetland soils of the GKM
Overall, THg and MeHg ranged between 27.62 and 347.58 μg/kg, between 2.06 and 31.81 μg/kg, with average values of 112.76 μg/kg and 12.43 μg/kg, respectively. %MeHg varied greatly across sampling sites, ranging from 1.61 to 23.3% with an average of 12.3%.
Forest peatlands had the highest MeHg contents, and moss peatlands had the lowest %MeHg values. THg, MeHg, and %MeHg differed significantly among different groups. THg significantly differed among groups in pairs (p < 0.01), besides forest versus moss categories. MeHg in the forest group was significantly higher than the others. %MeHg in the moss group was significantly lower than the others (Fig. 2).
The variation coefficients of THg and MeHg were 44.5% and 42.8% on average, respectively, and showed considerable variation in spatial (Fig. S1). A longitudinal occurrence of MeHg was observed spanning the range of 120–123°E (p < 0.05, Fig. 3), indicating that MeHg increased with the increasing longitude. However, no similar trend was observed for THg.
Spectral characteristics of DOM
Overall, moss peatlands had the highest SUVA254, SUVA260, A240/A420, A250/A365, A465/A664, and A253/A203 values of DOM. A250/A365 values were greater than 3.5 in all sites, meaning that DOM was dominated by fulvic acid with relatively low molecular weight.
Five components were extracted from the EEMs dataset using the PARAFAC analysis. The C1 (Ex/Em = 240/434) and C2 (Ex/Em = 270/482) belonged to UV fulvic-like fluorescence (peak A) and represented an external source input (He & Hur, 2015). The C3 (Ex/Em = 280/402) belonged to fulvic acid-like in visible light fluorescence (peak C) (He et al., 2015). The C4 (Ex/Em = 220, 280/ 338) and C5 (Ex/Em = 220/314) were fluorescence peak B (tyrosines) and peak T (tryptophans) and represented the metabolites of microorganisms (Chen et al., 2003; Wang et al., 2020). Therefore, there were three fulvic acid-like (F-like) and two protein-like (P-like) substances, which accounted for 35–90% and 10–65% of the total variances (Figs. 4 and S2).
The FI values exceeded 1.9 in peatlands (Table S4), indicating that endogenous products from microbial metabolism contribute most DOM in the GKM. The HIX values in herb and forest peatlands were no more than 4, confirming that DOM mainly was microbial-derived. BIX values in moss peatlands were significantly lower than those in herb and shrub groups (p < 0.05) because low pH and phenolic substance suppress microbial activity in Sphagnum peatlands and yield low biological DOM (Pedersen & Calvert, 1990; Van Breemen, 1995).
Correlation analysis
THg positively related to TN, SOC, DOM, and A240/A420, while negatively related to A2/A1, A3/A2, A3/A1, BIX, HIX, and F-like components at p < 0.05. MeHg positively related to SOC, DOM, and A240/A420, while negatively related to A2/A1 and P-like components at p < 0.05. %MeHg positively related to BIX, HIX, and F-like components while negatively related to TN, SOC, DOM, and P-like components at p < 0.05 (Fig. 5).
Discussion
THg contents in the present work were threefold of the background value, 40 µg/kg, confirming that Hg was accumulating effectively in peatlands of the GKM (Zhang et al., 2015). THg and MeHg in the GKM were much higher than those in peatlands, fens, and permafrost at similar latitudes (Table S5). For example, THg and MeHg in Ontario peatland soils were no more than 99 µg/kg and 3 µg/kg, respectively (Heyes et al., 2000). MeHg contents changed from 1.8 to 10.1 µg/kg in beaver ponds (Herrero Ortega et al., 2018), which was relatively high levels but still lower than those in the GKM peatlands. THg enrichment in the GKM is probably attributed to industrial emission in Heilongjiang and Jilin Province, the heavy industry base of China. In addition, Hg release from coal consumption and deposition might also contribute Hg accumulation in peatlands (Wang et al., 2000). %MeHg values in the GKM region, 12.3% on average, were much higher than those, no more than 3%, in Alaska (Olson et al., 2018), Norwegian Arctic (Halbach et al., 2017; Liu et al., 2015), and Ontario (Heyes et al., 2000). It implied that Hg methylation was effective in peatlands because of the reducing environment that favors Hg accumulation and stimulates Hg methylating microorganisms like sulfate-reducing bacteria and methanogens (Liu et al., 2002).
Vegetation absorption was the prominent Hg source in remote areas (Olson et al., 2018). Hg enters into the soil with litter and could be effectively retained by organic matter. Forest ecosystems represent strong Hg sinks globally and could capture about 1180 Mg/yr of Hg through a huge amount of litterfall (Wang et al., 2016). The present work matched this conclusion well, showing that forest peatlands had high THg and MeHg. Climate warming has introduced shrub or forest inhabitation into peatlands and caused slow expansion northward of forest in the Northern Hemisphere (Liess et al., 2012). The succession of plant under future climate warming scenarios, from herb or moss to shrub or forest, might yield more biomass returned to soils and this brought us particularly noteworthy concern that Hg enrichment and MeHg production were predicted to be accelerated in the GKM regions in the future.
PCA analysis indicated that two principal components were extracted and explained 42.4% of the total variance (Fig. 6). The first principal component (PC1, 23.8% of total variance) represented the humification degree of DOM. Indeed, SUVA254, A3/A1, A3/A2, A2/A1, and SUVA260 were indices of DOM humification and they all had positive loadings on PC1. The second principal component (PC2, 18.6% of total variance) separated plant-derived DOM from endogenous source; this was confirmed by negative loadings of BIX and positive loadings of SUVA254 on PC2. Both THg and MeHg had positive loading on PC2, while %MeHg had positive loadings on PC1. SUVA254, as proxies of terrigenous organic compounds, had positive loading on PC1 and PC2, showing that terrigenous DOM had great impacts on Hg sources (Ouellet et al., 2009), and indicating that the humification degree of DOM could support Hg methylation. Hg(II) is more available for methylation in the presence of high molecular weight DOM (Mitchell & Gilmour, 2008). One potential mechanism is hydrophobic acid fractions of DOM (which includes the humic and fulvic acid fractions) had significantly higher reduced sulfur content (defined as the sum of polysulfide, sulfide, and thiol groups) than the low molecular weight hydrophilic acid fractions (Ravichandran, 2004). It promotes Hg methylation in the presence of stronger complexing thiols (Schaefer et al., 2011). Compared to oxygen ligands, thiols and disulfides are preferred complexation points of Hg to DOM (Mitchell & Gilmour, 2008). The other reason is an additional effect of aromatic carbon, is its potential to enhance the release of Hg from insoluble cinnabar (Waples et al., 2005), which can promote and enhance Hg methylation (Paranjape et al., 2017). The small included angle between %MeHg and A3/A2 in Fig. 6 indicates that the aromatic degree of DOM can promote the ability of mercury methylation potential.
The multiple regression models showed that P-like components (negative), HIX (negative), BIX (negative) and DOM (positive) explained most variations of THg to a significant level. THg was the only factor that has significant positive effects on MeHg, while TN, P-like component, and HIX had significantly negative effects on MeHg (Fig. 7). DOM could improve THg and MeHg effectively due to the strong adsorption of DOM on THg and MeHg in soil (Fig. 5). HIX was negatively correlated with Hg and MeHg. This is likely due to Hg and MeHg preferentially associated with low molecular weight dissolved organic matter in wetland environments (Hill et al., 2009). THg was inversely correlated with BIX and P-like component, and P-like substances significantly reduced MeHg (Fig. 7), suggesting that DOC with microbial signatures could be associated with a decrease in THg and MeHg (Jiang et al., 2018). This might because of the much larger rate of Hg(II) reduction when complexed by the weaker, but more abundant RO/N (e.g., carboxyl and amino) functional groups in DOM in dark Hg(II) reduction (Rocha et al., 2000; Zheng & Hintelmann, 2010). The above results emphasize the important influence of DOM spectral characteristics on mercury methylation.
Conclusions
Unexpected high THg and MeHg contents, 112.76 µg/kg and 12.43 µg/kg, suggested that wetlands served as Hg sinks and MeHg sources in the GKM. Meanwhile, higher THg in forest wetlands implied Hg accumulation would be accelerated by climate change, which favor forest expansion into herb peatlands in these regions. DOM and its spectral characteristics play important roles in controlling Hg methylation. On the whole, DOM positively related to THg while P-like components negatively related to THg and MeHg. Hg and MeHg pollution were speculated to be aggravated by more DOM derived from permafrost thawing with current climate warming in the GKM regions.
Availability of data and materials
The authors confirm that the data supporting the findings of this study are available within the article.
Code availability
Not applicable.
References
Bachand, P. A., Bachand, S. M., Fleck, J. A., Alpers, C. N., Stephenson, M., & Windham-Myers, L. (2014). Methylmercury production in and export from agricultural wetlands in California, USA: The need to account for physical transport processes into and out of the root zone. Science of the Total Environment, 472, 957–970. https://doi.org/10.1016/j.scitotenv.2013.11.086
Barkay, T., Kroer, N., & Poulain, A. J. (2011). Some like it cold: Microbial transformations of mercury in polar regions. Polar Research, 30(1), 15469. https://doi.org/10.3402/polar.v30i0.15469
Birdwell, J. E., & Engel, A. S. (2010). Characterization of dissolved organic matter in cave and spring waters using UV–Vis absorbance and fluorescence spectroscopy. Organic Geochemistry, 41(3), 270–280. https://doi.org/10.1016/j.orggeochem.2009.11.002
Bracho, R., Natali, S., Pegoraro, E., Crummer, K. G., Schädel, C., Celis, G., Hale, L., Wu, L., Yin, H., & Tiedje, J. M. (2016). Temperature sensitivity of organic matter decomposition of permafrost-region soils during laboratory incubations. Soil Biology and Biochemistry, 97, 1–14. https://doi.org/10.1016/j.soilbio.2016.02.008
Bravo, A. G., Bouchet, S., Tolu, J., Björn, E., Mateos-Rivera, A., & Bertilsson, S. (2017). Molecular composition of organic matter controls methylmercury formation in boreal lakes. Nature Communications, 8(1), 1–9. https://doi.org/10.1038/ncomms14255
Celo, V., Lean, D. R., & Scott, S. L. (2006). Abiotic methylation of mercury in the aquatic environment. Science of the Total Environment, 368(1), 126–137. https://doi.org/10.1016/j.scitotenv.2005.09.043
Chen, W., Westerhoff, P., Leenheer, J. A., & Booksh, K. (2003). Fluorescence excitation−emission matrix regional integration to quantify spectra for dissolved organic matter. Environmental Science and Technology, 37(24), 5701–5710. https://doi.org/10.1021/es034354c
Corrales, J., Naja, G. M., Dziuba, C., Rivero, R. G., & Orem, W. (2011). Sulfate threshold target to control methylmercury levels in wetland ecosystems. Science of the Total Environment, 409(11), 2156–2162. https://doi.org/10.1016/j.scitotenv.2011.02.030
Cory, R. M., McKnight, D. M., Chin, Y. P., Miller, P., & Jaros, C. L. (2007). Chemical characteristics of fulvic acids from Arctic surface waters: Microbial contributions and photochemical transformations. Journal of Geophysical Research Biogeosciences. https://doi.org/10.1029/2006JG000343
Feng, X., Qiu, G., Fu, X., He, T., Li, P., & Wang, S. (2009). Mercury pollution in the environment. Chemical Progress, 21(Z1), 436–457.
Gao, Y., & zhao, H., Gao, F., Zhu, H., Qu, H. and Zhao, F. (2016). Climate change trend in future and its influence on wetlands in the greater Khingan Mountains. Journal of Glaciology and Geocryology, 38(01), 47–56.
Gong, P., Wang, X.-P., Xue, Y.-G., Xu, B.-Q., & Yao, T.-D. (2014). Mercury distribution in the foliage and soil profiles of the Tibetan forest: Processes and implications for regional cycling. Environmental Pollution, 188, 94–101. https://doi.org/10.1016/j.envpol.2014.01.020
Graham, A. M., Aiken, G. R., & Gilmour, C. C. (2012). Dissolved organic matter enhances microbial mercury methylation under sulfidic conditions. Environmental Science and Technology, 46(5), 2715–2723. https://doi.org/10.1021/es203658f
Halbach, K., Mikkelsen, Ø., Berg, T., & Steinnes, E. (2017). The presence of mercury and other trace metals in surface soils in the Norwegian Arctic. Chemosphere, 188, 567–574. https://doi.org/10.1016/j.chemosphere.2017.09.012
He, R., Xu, R., & Wei, C. (2015). Spectral characterization of dissolved organic matter in bio-treated effluent of coking wastewater. Environmental Chemistry, 34(1), 129–136.
He, W., & Hur, J. (2015). Conservative behavior of fluorescence EEM-PARAFAC components in resin fractionation processes and its applicability for characterizing dissolved organic matter. Water Research, 83, 217–226. https://doi.org/10.1016/j.watres.2015.06.044
He, X., Xi, B., Wei, Z., Li, M., Geng, C., Yu, H., & Liu, H. (2010). Three-dimensional excitation emission matrix fluorescence spectroscopic characterization of complexation between mercury(II) and dissolved organic matter extracted from landfill leachate. Analytical Chemistry, 38(10), 1417–1422.
Herrero Ortega, S., Catalán, N., Björn, E., Gröntoft, H., Hilmarsson, T. G., Bertilsson, S., Wu, P., Bishop, K., Levanoni, O., & Bravo, A. G. (2018). High methylmercury formation in ponds fueled by fresh humic and algal derived organic matter. Limnology and Oceanography, 63(S1), S44–S53. https://doi.org/10.1002/lno.10722
Heyes, A., Moore, T., Rudd, J. W., & Dugoua, J. (2000). Methyl mercury in pristine and impounded boreal peatlands, experimental Lakes Area, Ontario. Canadian Journal of Fisheries and Aquatic Sciences, 57(11), 2211–2222. https://doi.org/10.1139/f00-197
Hill, J. R., O’Driscoll, N. J., & Lean, D. R. (2009). Size distribution of methylmercury associated with particulate and dissolved organic matter in freshwaters. Science of the Total Environment, 408(2), 408–414. https://doi.org/10.1016/j.scitotenv.2009.09.030
Hudelson, K. E., Drevnick, P. E., Wang, F., Armstrong, D., & Fisk, A. T. (2020). Mercury methylation and demethylation potentials in Arctic lake sediments. Chemosphere, 248, 126001. https://doi.org/10.1016/j.chemosphere.2020.126001
Huguet, A., Vacher, L., Relexans, S., Saubusse, S., Froidefond, J.-M., & Parlanti, E. (2009). Properties of fluorescent dissolved organic matter in the Gironde Estuary. Organic Geochemistry, 40(6), 706–719. https://doi.org/10.1016/j.orggeochem.2009.03.002
Jiang, T., Bravo, A. G., Skyllberg, U., Björn, E., Wang, D., Yan, H., & Green, N. W. (2018). Influence of dissolved organic matter (DOM) characteristics on dissolved mercury (Hg) species composition in sediment porewater of lakes from southwest China. Water Research, 146, 146–158. https://doi.org/10.1016/j.watres.2018.08.054
Klapstein, S. J., Ziegler, S. E., Risk, D. A., & O’Driscoll, N. J. (2018). Assessing the utility of dissolved organic matter photoreactivity as a predictor of in situ methylmercury concentration. Journal of Environmental Sciences, 68, 160–168. https://doi.org/10.1016/j.jes.2018.02.025
Lehnherr, I., & St. Louis, V.L., Emmerton, C.A., Barker, J.D. and Kirk, J.L. (2012). Methylmercury cycling in high Arctic wetland ponds: Sources and sinks. Environmental Science and Technology, 46(19), 10514–10522. https://doi.org/10.1021/es300576
Lescord, G. L., Emilson, E. J., Johnston, T. A., Branfireun, B. A., & Gunn, J. M. (2018). Optical properties of dissolved organic matter and their relation to mercury concentrations in water and biota across a remote freshwater drainage basin. Environmental Science & Technology, 52(6), 3344–3353. https://doi.org/10.1021/acs.est.7b05348
Liess, S., Snyder, P. K., & Harding, K. J. (2012). The effects of boreal forest expansion on the summer Arctic frontal zone. Climate Dynamics, 38(9), 1805–1827. https://doi.org/10.1007/s00382-011-1064-7
Liu, R., Wang, Q., Lu, X., Zhuangwei, M., & Fang, F. (2002). Mercury in the peat bog ecosystem in the Xiaoxing ’an Mountain in China. Environmental Science, 23(4), 102–106.
Liu, J., & Ding, Z. (2007). Progress in research on mercury methylation in environment. Earth and Environment, 03, 215–222.
Liu, Y., Chai, X., Hao, Y., Gao, X., Lu, Z., Zhao, Y., Zhang, J., & Cai, M. (2015). Total mercury and methylmercury distributions in surface sediments from Kongsfjorden, Svalbard, Norwegian Arctic. Environmental Science and Pollution Research, 22(11), 8603–8610. https://doi.org/10.1007/s11356-014-3942-0
MacMillan, G. A., Girard, C., Chételat, J., Laurion, I., & Amyot, M. (2015). High methylmercury in Arctic and subarctic ponds is related to nutrient levels in the warming eastern Canadian Arctic. Environmental Science & Technology, 49(13), 7743–7753. https://doi.org/10.1021/acs.est.5b00763
Mitchell, C. P., & Gilmour, C. C. (2008). Methylmercury production in a chesapeake bay salt marsh. Journal of Geophysical Research: Biogeo Sciences, 113(2), 1.
Mu, C., Zhang, F., Chen, X., Ge, S., Mu, M., Jia, L., Wu, Q., & Zhang, T. (2019). Carbon and mercury export from the Arctic rivers and response to permafrost degradation. Water Research, 161, 54–60. https://doi.org/10.1016/j.watres.2019.05.082
Ohno, T. (2002). Fluorescence inner-filtering correction for determining the humification index of dissolved organic matter. Environmental Science & Technology, 36(4), 742–746. https://doi.org/10.1021/es0155276
Olson, C., Jiskra, M., Biester, H., Chow, J., & Obrist, D. (2018). Mercury in active-layer tundra soils of alaska: concentrations, pools, origins and spatial distribution. Global Biogeochemical Cycles, 32(7), 1058–1073. https://doi.org/10.1029/2017GB005840
Ouellet, J.-F., Lucotte, M., Teisserenc, R., Paquet, S., & Canuel, R. (2009). Lignin biomarkers as tracers of mercury sources in lakes water column. Biogeochemistry, 94(2), 123–140. https://doi.org/10.1007/s10533-009-9314-z
Paranjape, A. R., & Hall, B. D. (2017). Recent advances in the study of mercury methylation in aquatic systems. Facets, 2(1), 85–119. https://doi.org/10.1139/facets-2016-0027
Pedersen, T., & Calvert, S. (1990). Anoxia vs productivity: What controls the formation of organic-carbon-rich sediments and sedimentary rocks? AAPG Bulletin, 74(4), 454–466. https://doi.org/10.1306/0C9B232B-1710-11D7-8645000102C1865D
Ravichandran, M. (2004). Interactions between mercury and dissolved organic matter––a review. Chemosphere, 55(3), 319–331. https://doi.org/10.1016/j.chemosphere.2003.11.011
Rocha, J. C., Junior, É. S., Zara, L. F., Rosa, A. H., dos Santos, A., & Burba, P. (2000). Reduction of mercury (II) by tropical river humic substances (Rio Negro)—a possible process of the mercury cycle in Brazil. Talanta, 53(3), 551–559. https://doi.org/10.1016/S0039-9140(00)00532-4
Schaefer, J. K., & Morel, F. M. (2009). High methylation rates of mercury bound to cysteine by Geobacter sulfurreducens. Nature Geoscience, 2(2), 123–126. https://doi.org/10.1038/ngeo412
Schaefer, J. K., Rocks, S. S., Zheng, W., Liang, L., Gu, B., & Morel, F. M. (2011). Active transport, substrate specificity, and methylation of Hg (II) in anaerobic bacteria. Proceedings of the National Academy of Sciences, 108(21), 8714–8719. https://doi.org/10.1073/pnas.1105781108
Schartup, A. T., Mason, R. P., Balcom, P. H., Hollweg, T. A., & Chen, C. Y. (2013). Methylmercury production in estuarine sediments: Role of organic matter. Environmental Science & Technology, 47(2), 695–700. https://doi.org/10.1021/es302566w
Schuster, P. F., Schaefer, K. M., Aiken, G. R., Antweiler, R. C., Dewild, J. F., Gryziec, J. D., Gusmeroli, A., Hugelius, G., Jafarov, E., Krabbenhoft, D. P., Liu, L., Herman-Mercer, N., Mu, C., Roth, D. A., Schaefer, T., Striegl, R. G., Wickland, K. P., & Zhang, T. (2018). Permafrost stores a globally significant amount of mercury. Geophysical Research Letters, 45(3), 1463–1471. https://doi.org/10.1002/2017GL075571
Semeraro, T., Giannuzzi, C., Beccarisi, L., Aretano, R., De Marco, A., Pasimeni, M. R., Zurlini, G., & Petrosillo, I. (2015). A constructed treatment wetland as an opportunity to enhance biodiversity and ecosystem services. Ecological Engineering, 82, 517–526. https://doi.org/10.1016/j.ecoleng.2015.05.042
St. Pierre, K., Chétélat, J., Yumvihoze, E. and Poulain, A. (2014). Temperature and the sulfur cycle control monomethylmercury cycling in high Arctic coastal marine sediments from Allen Bay, Nunavut Canada. Environmental Science & Technology, 48(5), 2680–2687. https://doi.org/10.1021/es405253g
Van Breemen, N. (1995). How sphagnum bogs down other plants. Trends in Ecology & Evolution, 10(7), 270–275. https://doi.org/10.1016/0169-5347(95)90007-1
Vasilevich, R. S., Beznosikov, V. A., Lodygin, E. D., & Kondratenok, B. M. (2014). Complexation of mercury(II) ions with humic acids in tundra soils. Eurasian Soil Science, 47(3), 162–172. https://doi.org/10.1134/S1064229314030089
Wallschläger, D., Desai, M. V., & Wilken, R.-D. (1996). The role of humic substances in the aqueous mobilization of mercury from contaminated floodplain soils. Water, Air and Soil Pollution, 90(3), 507–520. https://doi.org/10.1007/BF00282665
Wang, S., Qin, J., Xie, B., Liu, C., Chen, Y., Tang, Y., & Sun, H. (2020). Spectroscopic characteristics of dissolved organic matter (DOM) in Zoige alpine peatland soils along a soil moisture gradient. Ecological Environmental Science, 29(04), 676–685. https://doi.org/10.16258/j.cnki.1674-5906.2020.04.005
Wang, Q., Shen, W., & Ma, Z. (2000). Estimation of mercury emission from coal combustion in China. Environmental Science and Technology, 34, 2711–2713. https://doi.org/10.1021/es990774j
Wang, X., Bao, Z., Lin, C.-J., Yuan, W., & Feng, X. (2016). Assessment of global mercury deposition through litterfall. Environmental Science & Technology, 50(16), 8548–8557. https://doi.org/10.1021/acs.est.5b06351
Waples, J. S., Nagy, K. L., Aiken, G. R., & Ryan, J. N. (2005). Dissolution of cinnabar (HgS) in the presence of natural organic matter. Geochimica Et Cosmochimica Acta, 69(6), 1575–1588. https://doi.org/10.1016/j.gca.2004.09.029
Weber, J. H. (1993). Review of possible paths for abiotic methylation of mercury (II) in the aquatic environment. Chemosphere, 26(11), 2063–2077. https://doi.org/10.1016/0045-6535(93)90032-Z
Yang, Y., & Wang, D. (2006). Research progress in temporal and spatial distribution of atmospheric mercury. Sichuan Environment, 25(6), 91–95. https://doi.org/10.14034/j.cnki.schj.2006.06.024
Yang, Z., Fang, W., Lu, X., Sheng, G.-P., Graham, D. E., Liang, L., Wullschleger, S. D., & Gu, B. (2016). Warming increases methylmercury production in an Arctic soil. Environmental Pollution, 214, 504–509. https://doi.org/10.1016/j.envpol.2016.04.069
Yao, A., Qin, C., & Mou, S. (2004). Effect of humus on environmental mobility of mineral bound mercury. Acta Ecologica Sinica, 02, 274–277.
Zhang, M., Dai, J., & Wang, R. (2011). Research progress of the effect of dissolved organic matter(DOM) on adsorption migration and bioavailability of Hg in soil. Environmental Pollution and Control, 33(05), 95–110. https://doi.org/10.15985/j.cnki.1001-3865.2011.05.022
Zhang, J., Lv, P., Wang, L., & Yu, Z. (2015). Spatial variation of heavy metal contents and contamination assessment in forest soils in Daxing ’an Mountains. Chinese Journal of Ecology, 34(03), 810–819. https://doi.org/10.13292/j.1000-4890.2015.0110
Zhang, S., Wang, Q., Cui, J., Sun, X., & Zheng, D. (2009). CV-AFS determination of methyl mercury in sediment and soil samples with ultrasonic extraction. Physical and Chemical Examination (chemistry Section), 45(11), 1273–1275.
Zhang, Z., Li, M., Li, Z., Xue, Z., & Jiang, M. (2020). Unexpected high methylmercury contents related to soil organic carbon and its molecular composition in wetland soils of the Yarlung Tsangbo River. Tibet. Geoderma, 377, 114607. https://doi.org/10.1016/j.geoderma.2020.114607
Zhao, H., Gong, L., Qu, H., Zhu, H., Li, X., & Zhao, F. (2016). The climate change variations in the northern greater Khingan Mountains during the past centuries. Journal of Geographical Sciences, 26(5), 585–602. https://doi.org/10.1007/s11442-016-1287-y
Zheng, D., Zhang, S., Ma, H., & Li, H. (2020). Simulation of methylmercury content and SRB methylation in phragmites australis soil under different salinity conditions. Water, Air, & Soil Pollution, 231(1), 1–8. https://doi.org/10.1007/s11270-019-4382-8
Zheng, W., & Hintelmann, H. (2010). Nuclear field shift effect in isotope fractionation of mercury during abiotic reduction in the absence of light. The Journal of Physical Chemistry A, 114(12), 4238–4245. https://doi.org/10.1021/jp910353y
Zsolnay, A., Baigar, E., Jimenez, M., Steinweg, B., & Saccomandi, F. (1999). Differentiating with fluorescence spectroscopy the sources of dissolved organic matter in soils subjected to drying. Chemosphere, 38(1), 45–50. https://doi.org/10.1016/S0045-6535(98)00166-0
Acknowledgements
The authors appreciate the financial support by the National Natural Science Foundation of China (41771103, U20A2083, and U19A2042) and the Youth Innovation Promotion Association CAS (No. 2018265).
Author information
Authors and Affiliations
Contributions
YX contributed to study design, sample measurement, data curation and analysis, and writing—original draft preparation. XZ contributed to assisting sample collection and sample measurement. DZ contributed to methodology and investigation. ZZ contributed to study design, methodology, and manuscript revision.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval
This work was performed in compliance with ethical standards.
Humans and animals research
Not applicable.
Consent to participate
All authors were informed about and agreed to the protocol.
Consent to publish
All authors were allowed to read and approve the final manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xin, Y., Zhang, X., Zheng, D. et al. Impacts of spectral characteristics of dissolved organic matter on methylmercury contents in peatlands, Northeast China. Environ Geochem Health 45, 913–923 (2023). https://doi.org/10.1007/s10653-022-01257-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10653-022-01257-1