Abstract
The excessive use of pesticides in agriculture and the widespread use of metals in industrial activities and or technological applications has significantly increased the concentrations of these pollutants in both aquatic and terrestrial ecosystems worldwide, making aquatic biota increasingly vulnerable and putting many species at risk of extinction. Most aquatic habitats receive pollutants from various anthropogenic actions, leading to interactions between compounds that make them even more toxic. The aim of this study was to assess the effects of the compounds Chlorpyrifos (insecticide) and Cadmium (metal), both individually and in mixtures, on the cladocerans Ceriodaphnia rigaudi and Ceriodaphnia silvestrii. Acute toxicity tests were conducted for the compounds individually and in mixture, and an ecological risk assessment (ERA) was performed for both compounds. Acute toxicity tests with Cadmium resulted in EC50-48 h of 0.020 mg L−1 for C. rigaudi and 0.026 mg L−1 for C. silvestrii, while tests with Chlorpyrifos resulted in EC50-48 h of 0.047 μg L−1 and 0.062 μg L−1, respectively. The mixture test for C. rigaudi showed the occurrence of additive effects, while for C. silvestrii, antagonistic effects occurred depending on the dose level. The species sensitivity distribution curve for crustaceans, rotifers, amphibians, and fishes resulted in an HC5 of 3.13 and an HC50 of 124.7 mg L−1 for Cadmium; an HC5 of 9.96 and an HC50 of 5.71 μg L−1 for Chlorpyrifos. Regarding the ERA values, Cadmium represented a high risk, while Chlorpyrifos represented an insignificant to a high risk.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Aquatic ecosystems exhibit high vulnerability due to the presence of various compounds. One example are pesticides that enter these environments from multiple sources, such as residues from agricultural activities, caused by pesticide drift, surface runoff (dissolved in water or adsorbed to particles), soil leaching, and groundwater contamination (El-Nahhal and El-Nahhal 2021).
Despite being banned in several European Union countries and the United States (Villar and Schaeffer 2022), Chlorpyrifos is one of the most widely used insecticides globally (Solomon et al. 2014). In the United States, annual usage estimates ranged from 3200 to 4100 tons (Solomon et al. 2014), in Mexico, the usage between 2010 and 2020 was 114,751.77 tons (Ruiz-Arias et al. 2023), in Iran, between 2012 and 2014, the usage was 1134 tons (Morteza et al. 2017), in China, the annual usage is around 18,000 tons (Liu et al. 2014), and in Brazil, in 2022, Chlorpyrifos was the 9th most commercially sold active ingredient, with 17,679.34 tons (IBAMA IB do MA 2022). Measured values in Brazilian rivers have been reported ranging from 0.003 µg L−1 in the Pantanal basin (Laabs et al. 2002) to 0.36 µg L−1 in Ribeirão Preto (CETESB SP 2021).
In Brazil, Chlorpyrifos is used in the cultivation of cotton, peanuts, oats, potatoes, coffee, rye, barley, citrus fruits, peas, beans, chickpeas, lentils, apples, corn, pastures, soybeans, sorghum, wheat, tomatoes, and potatoes, as well as for ant control and as a wood preservative (ANVISA 2021). Its mode of action involves the inhibition of the cholinesterase enzyme in the central and peripheral nervous systems, leading to an accumulation of acetylcholine that promotes neurotoxicity in animals (Zhao et al. 2006).
In addition to pesticides, there are a variety of pollutants present in aquatic ecosystems, such as potentially toxic metals. Originating from mining residues, leaching from landfills, urban runoff, and industrial waste (Gautam et al. 2014), these metals cannot be biodegraded, and bioaccumulation makes them highly hazardous (Baby et al. 2011).
Cadmium is a particularly concerning metal because, unlike others such as copper and zinc, cadmium is not essential for biological systems and can be highly toxic even at low concentrations (Gautam et al. 2014). It is naturally released into the environment through processes like weathering and volcanic eruptions, but there is a rapid and constant increase due to human activities such as mining and industrial processes like electroplating, paint manufacturing, dyes, and batteries (Sobha et al. 1970). According to the National Environment Council Resolution No. 357, dated March 17, 2005 (CONAMA, 2005), the maximum permissible value of cadmium in freshwater is 0.001 mg L−1. However, measured maximum values in Brazilian rivers exceed this limit, such as the São Francisco River with 0.02 mg L−1 (Ribeiro et al. 2012), the Tapajós River with 0.004 mg L−1 (Oliveira et al. 2015), and the Cassiporé River with 0.8 mg L−1 (Lima et al. 2015), all belonging to the Amazon River basin.
In nature, pollutants are unlikely present in isolation, so it is necessary to assess their potential interactions. Despite being common and coexisting in the environment, the combined toxicity of Cadmium and Chlorpyrifos has not been fully elucidated yet (Wang et al. 2017). In order to explain the behavior of compounds in mixture, theoretical models have been developed based on two reference concepts that predict the toxicity of the mixture based on the individual toxicities of the components, assuming no interaction between the compounds (Mansano et al. 2017; Jonker et al. 2005). These are the concentration addition and independent action models (Bliss 1939; Pérez et al. 2011), with the possibility of synergism or antagonism deviations, depending on the dose level (DL) or dose ratio (DR) (Mansano et al. 2017).
Toxicity assessments targeting the effects caused by individual products and their mixtures, coupled with risk assessment, can be suitable for investigating the effects of these stressors on biota. Ecological Risk Assessment (ERA) is a tool that allows the analysis of the probability of adverse effects occurring in the environment due to exposure to certain agents (EPA, 1998). In order to increase representativeness in Brazilian research, the use of native species as test organisms has been recommended (Martins and Bianchini 2011). Furthermore, it promotes a more realistic evaluation of their sensitivity, facilitates logistics, and avoids the introduction of exotic species (Freitas and Rocha 2011; Mansano et al. 2018).
The objectives of this study were (I) to assess the acute effects (immobility) of single and combined concentrations of the compounds Chlorpyrifos and Cadmium Chloride on the native cladocerans C. rigaudi and C. silvestrii; and (II) to perform risk estimates based on risk quotients (RQs) for both compounds.
Methods
Cultivation and maintenance of test organisms
The species Ceriodaphnia rigaudi, Richard, 1894 (Crustacea, Cladocera, Daphnidae), and Ceriodaphnia silvestrii, Daday, 1902 (Crustacea, Cladocera, Daphnidae), were collected from the Broa Reservoir, located between the municipalities of Itirapina – SP and Brotas – SP. After collection, they were acclimatized and maintained according to the procedures described in the protocol 13373 of the Brazilian Association of Technical Standards (ABNT, 2016), in 2 L beakers containing reconstituted water composed of 1.5 g L−1 CaSO4·2H20, 6.1 g L−1 MgSO4·7H2O, 4.8 g L−1 NaHCO3, and 0.2 g L−1 KCl dissolved in distilled water, with total hardness between 40 and 48 mg CaCO3 L−1, pH between 7 and 7.6, and conductivity around 160 μS cm−1. Additionally, 1 drop of Potemin® vitamin was added per liter of reconstituted water.
The culture medium was renewed three times a week, with two complete changes and one partial change. The beakers were covered with plastic film and maintained at a temperature of 25 ± 1 °C and a photoperiod of 12:12 h light/dark.
The diet consisted of algal suspension from the microphyte Raphidocelis subcapitata cultivated in CHU-12 medium at a concentration of 105 cells/liter and 1 mL L−1 of a food additive composed of equal parts of fermented fish feed (TetraMin®) and yeast (Fleischmann® type dry biological yeast and distilled water).
Chemical compounds
The compounds used in this study were pure Cadmium Chloride Monohydrate (CdCl2·H2O, CAS: 7790-78-5) from the Vetec brand (99% purity) and Chlorpyrifos (C9H11Cl3NO3PS, CAS: 2921-88-2) from the Sigma Aldrich brand (98% purity).
For the test with Cadmium, a stock solution of 10 mg L−1 (as CdCl2·H20) was prepared. From this solution, for C. rigaudi, five concentrations were prepared through serial dilutions in reconstituted water (0.02; 0.03; 0.04; 0.05; and 0.06 mg L−1), in addition to the control. For C. silvestrii, concentrations, also prepared from the 10 mg L−1 solution, were 0.04; 0.05; 0.06; 0.07; and 0.08 mg L−1, in addition to the control.
The quantification of Cadmium was carried out following the guidelines of the 23rd edition of Standard Methods (SMWW, 3111 B). The aliquot of the stock solution was withdrawn, acidified with concentrated nitric acid until reaching pH < 2.0, and used for the determination of actual concentrations. These analyses were conducted using an inductively coupled plasma optical emission spectrometer (ICP-OES Optima 8300/Perkin Elmer).
For the test with isolated Chlorpyrifos, a stock solution of 1 mg L−1 was prepared. From this solution, five concentrations were prepared through serial dilutions in reconstituted water (0.01; 0.02; 0.04; 0.08; and 0.16 µg L−1), in addition to the control and the control with the solvent (acetonitrile) at the highest tested concentration (0.0032 ng L−1) of solvent used. For C. silvestrii, concentrations, also prepared from the 1 mg L−1 solution, were 0.01; 0.02; 0.04; 0.08; and 0.16 µg L−1, in addition to the control and the control with the solvent.
The quantification of Chlorpyrifos was carried out via high-performance liquid chromatography coupled with UV detection (HPLC–UV). An aliquot of the stock solution was withdrawn and kept refrigerated until the analysis. The analysis was conducted using the Agilent 1260 Infinity II HPLC equipment, coupled with a UV–Vis detector operating at 210 nm and using a Phenomenex Kinetex column (5 μm, EVO C18, 100 Ä, 150 × 4.6 mm). The mobile phase used was water (A)/acetonitrile (B) in isocratic mode at 30% A/70% B, with a mobile phase flow rate of 0.8 mL min−1, an injection volume of 10 µL, and a column temperature of 40 °C. After quantification, nominal concentrations were adjusted to represent the actual compound values.
For acute toxicity testing of the mixtures, a complete factorial experimental design was used, including 1 test for each compound and 25 combinations of both, which were selected based on the acute toxicity tests conducted individually (Fig. S1). In total, 36 treatments were performed, with 4 replicates for each.
Acute toxicity tests
The acute toxicity tests followed methodologies adapted from the standards of the Brazilian Association of Technical Standards ABNT NBR 12713 (ABNT 2016) and Method 1002.0 (EPA 2002). The tests were static (without medium renewal) and lasted for 48 h. Five independent tests were conducted for each compound and each species. In the case of the mixture, 36 tests were conducted for each cladoceran species.
Organisms aged between 6 and 24 h were placed in plastic containers containing 15 mL of the test solution or reconstituted water (control), with 5 organisms per replicate. Four replicates were conducted for each concentration. The containers were maintained without aeration, feeding, and illumination in an incubator with a controlled temperature of 25 ± 1 °C. pH, electrical conductivity, and dissolved oxygen variables were measured at the beginning and end of each experiment. Hardness was measured only at the beginning. The individuals were observed at the end of 48 h, thus obtaining the number of immobile individuals for calculating the concentration causing an effect on 50% of the organisms compared to the control (CE50-48 h).
Data analysis
The values of CE50-48 h and their respective 95% confidence intervals for acute toxicity tests were calculated through nonlinear regression, fitting a logistic equation to the data using Statistica 12.5 software (STATSOFT 2014).
The data from the mixture tests were analyzed using the concentration addition (CA) and independent action (IA) models. Initially, the observed data were compared with the expected effect for the mixture, calculated based on individual exposures through the MIXTOX tool. Subsequently, extended analyses were performed considering three types of deviations from the reference models: synergistic/antagonistic interactions (S/A), deviation dependent on dose ratio (DR), and deviation dependent on dose level (DL). These deviations were modeled by adding two parameters (“a” and “b”). Parameter “a” takes negative or positive values, indicating synergistic or antagonistic deviation. For dose ratio-dependent deviation (DR), a second parameter “bDR” was included, allowing the identification of the role of each compound in the mixture. For dose level-dependent deviation (DL), another parameter “bDL” was included, indicating at which dose level the deviation occurs. The value of “a” indicates the deviation at low and high doses, while the value of “bDL” indicates the dose level at which the deviation occurs. The data were fitted to conceptual models and deviations, using the maximum likelihood method to select the best fit. Once the most statistically suitable model was identified to describe the deviation, the pattern of effects was deduced directly from the parameter values, and the maximum deviation could be calculated in terms of the effect level (Jonker et al. 2005).
Species sensitivity distribution (SSD)
The values of CE50 obtained in this study were used to compare with LC50 and EC50 values for other test organisms using the species sensitivity distribution (SSD) approach. Data were retrieved from the ECOTOX database of the United States Environmental Protection Agency (US-EPA) (Olker et al. 2022), selecting articles with EC50 or LC50 data for amphibians, fishes, rotifers, and crustaceans exposed to freshwater with measured chemical analyses. When multiple data points were available for the same organism, geometric means were calculated. All references used are provided in the Supplementary Material (Tables S1 and S2).
Log-normal distribution curves were constructed using the R software with the SSDtools package version 1.0.2 (Thorley and Schwarz 2018). The software ETX 2.0 was used to calculate the values of HC5 (hazardous concentration for 5% of species) and HC50 (hazardous concentration for 50% of species) along with their 95% confidence intervals according to Aldenberg and Jaworska (2000). The underlying assumption of the software is the log-normal distribution of the data, and the verification of log-normality was performed using the Anderson–Darling test, available in the ETX software package, with a significance level of 5%.
Ecological risk assessment
Ecological risk assessment was conducted by deriving risk quotients (RQ), calculated as the ratio between measured (MEC) or predicted (PEC) environmental concentrations and the predicted no-effect concentration (PNEC) (Sánchez-Bayo et al. 2002). PNEC values were obtained by dividing the HC5 value resulting from the SSD curve by an assessment factor (AF = 4), selected due to the level of uncertainty associated with the ecotoxicological information available for various taxonomic groups in constructing SSD curves (Moreira et al. 2020; Gomes et al. 2023). RQ values for Cadmium were assessed on the scale: low risk (RQ < 0.1), medium risk (RQ ranging from 0.1 to 1), and high risk (RQ > 1) (Yan et al. 2022; Gomes et al. 2023). For Chlorpyrifos, the scale was as follows: insignificant (RQ < 0.01), low risk (0.01 < RQ < 0.1), medium risk (0.1 < RQ < 1), high risk (RQ = 1), and very high risk (RQ > 1) (Moreira et al. 2020, Sánchez-Bayo et al. 2002).
MEC values were obtained from data available on Web of Science, Scopus, Science Direct, and repositories of theses and dissertations from Brazilian universities. Articles with the described quantification method for Brazilian water bodies were selected and can be consulted in the tables in the Supplementary Material (Tables S3 and S4).
To determine environmental concentrations that would be safe for the analyzed species, the low risk RQ values stipulated by previous studies (Moreira et al. 2020; Sánchez-Bayo et al. 2002; Yan et al. 2022; Gomes et al. 2023) were multiplied by the PNEC values obtained in this study, as follows: Safe MEC = Low risk RQ × PNEC.
Results
Quantification of substances
The concentration of Cadmium quantified in the stock solution can be found in the table in the Supplementary Material (Table S5). The metal concentration remained within a variation of 10% of the desired concentration, as recommended by ISO 10706:2000 (ISO 2000). This consistency reinforces the safety of using nominal concentrations.
The concentration of Chlorpyrifos quantified in the stock solution can be found in the table in the Supplementary Material (Table S6).
Acute toxicity tests
During the tests, physical and chemical parameters were monitored and varied as follows: pH (7.19–7.91), electrical conductivity (139–221 μS cm−1), dissolved oxygen (6.06–8.2 mg L−1), and hardness (40–44 CaCO3 L−1).
For C. rigaudi, tests using the reference substance NaCl ranged from CE50-48 h 0.85–1.08 g L−1, indicating that the sensitivity was within the expected range: 0.84–1.5 g L−1 (Miguel 2020). For C. silvestrii, tests using the reference substance NaCl ranged from CE50-48 h 1.09–1.47 g L−1, indicating that the sensitivity was within the expected range: 0.76–1.46 g L−1 (Takenaka 2007).
The CE50 values found in acute exposures of C. rigaudi to Cadmium and Chlorpyrifos individually are presented in Table 1.
The CE50 values found in acute exposures of C. silvestrii exposed to Cadmium and Chlorpyrifos individually are presented in Table 2.
Species sensitivity distribution curves (SSDs)
Based on the SSD curve for Cadmium (Fig. 1), it is possible to infer that the species tested in this study, C. rigaudi and C. silvestrii, are, respectively, the 11th and 14th most sensitive crustacean species. Additionally, the group most sensitive to Cadmium was the fishes, with Salmo trutta being the most sensitive.
Species sensitivity distribution (SSD) curve of crustaceans, rotifers, amphibians, and fishes to the metal Cadmium, at concentrations ranging from 0.003 to 3.019 mg L−1. The HC5 value is indicated by the dotted line. The species in bold (Ceriodaphnia silvestrii and Ceriodaphnia rigaudi) are the species analyzed in this study
Analyzing the SSD curve related to Chlorpyrifos (Fig. 2), it can be concluded that the species tested in this study (C. silvestrii and C. rigaudi) are, respectively, the second and third most sensitive species. In this case, the most sensitive group was crustaceans, with Daphnia ambigua being the most sensitive species.
Species sensitivity distribution (SSD) curve of crustaceans, rotifers, amphibians, and fish to the pesticide Chlorpyrifos, at concentrations ranging from 0.00004 to 5.926 mg L−1. The HC5 value is indicated by the dotted line. The species in bold (Ceriodaphnia silvestrii and Ceriodaphnia rigaudi) are the species analyzed in this study
The mean values of hazardous concentrations of Cadmium and Chlorpyrifos affecting 5% (HC5) and 50% (HC50) of all species included in the SSDs and their respective 95% confidence intervals (CI) are presented in Table 3.
Ecological risk assessment
The calculated values of PNEC, MEC, and RQ are presented in Table 4. According to the results, for the lowest concentration found, Chlorpyrifos represents an insignificant risk, while for the median concentration it represents a lower risk and for the higher concentrations, it represents a medium risk. On the other hand, Cadmium represents a high risk since the lowest concentration, as the RQ values were higher than 1. With the calculated PNEC value and an RQ value representing low risk, we computed the MEC values which represented low risk for the species included in the SSD. For Cadmium, the MEC value representing low risk would be 0.078 µg L−1, and for Chlorpyrifos, 0.025 µg L−1.
Mixture tests
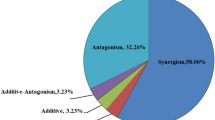
The data obtained from the acute toxicity test with the mixture of Cadmium and Chlorpyrifos for C. rigaudi fit the independent action (IA) reference model, with no occurrence of any of the three possible deviations from the model – synergistic/antagonistic interactions (S/A), dose ratio-dependent deviation (DL), or dose level-dependent deviation (DR) (Fig. 3). The fitting of the mixture data to the IA model resulted in a sum of the squares of residuals (SS) of 50.06 (p < 0.05; r2 = 0.88; n = 36) (Table S7).
For C. silvestrii, the data obtained from the acute toxicity test with the mixture of Cadmium and Chlorpyrifos fit the independent action (IA) reference model, with a deviation dependent on the dose level (DL), showing antagonism at lower concentrations of Cadmium (Fig. 4). The adjustment of the mixture data to the IA model produced a sum of squares of residuals (SS) of 43.05 (p < 0.05; r2 = 0.90, n = 36) (Table S8).
Isobologram of the mixture effects of the metal Cadmium and the pesticide Chlorpyrifos for the cladoceran Ceriodaphnia silvestrii, highlighting the fit to the independent action (IA) reference model, with a deviation dependent on the dose level (DL), showing antagonism at lower concentrations of Cadmium (MIXTOX Tool)
Discussion
In this study, the mean values of CE50-48 h obtained for C. rigaudi (0.02 mg L−1) exposed to Cadmium were about ten times lower than those found by Mohammed (2007) for the same species (CE50-48 h of 0.2 mg L−1), which may be due to differences in methodology such as hardness, in which they used 90–100 mg L−1. In the study of Yoon et al. (2017), there was a decrease in Cadmium toxicity to Daphnia magna with increasing hardness, which was attributed to competition between metals and cations that cause water hardness. Compared to other cladocerans, the CE50 values obtained for C. silvestrii (0.026 mg L−1) were slightly lower than those reported by Biesinger and Christensen (1972) for Daphnia magna (CE50-48 h of 0.065 mg L−1), a standard test species in temperate regions. Both species studied here were also more sensitive compared to Chydorus sphaericus (CE50-48 h of 0.149 mg L−1) (Lalande and Pinel-Alloul 1983). On the other hand, the CE50-48 h of cadmium for Daphnia galeata mendotae reported by Marshall (1979) was 0.04 mg L−1, which denotes that the sensitivity of that species is very close to that of C. silvestrii in the present study.
In addition to cladocerans, the toxicity of cadmium has been documented for various organisms, affecting the growth and replication of aquatic invertebrates and microorganisms, causing hypocalcemia in fish, and affecting the growth, stomatal opening, transpiration, and photosynthesis in plants (World Health Organization 1992). Based on the SSD, among the groups analyzed, fishes were the most sensitive, with Salmo trutta being the most sensitive species, which is supported by the work of Eaton et al. (1978), indicating that among fishes, salmonid species are more sensitive to cadmium. In this study, few data were found for amphibians (two species) and rotifers (seven species) (as seen in Table S1), which were the least sensitive groups. Therefore, a maximum allowed value by law that would protect fish and cladocerans species would also protect amphibians and rotifers.
In the case of Chlorpyrifos, the most sensitive crustaceans were Daphnia ambigua, C. silvestrii, and C. rigaudi, respectively, which is consistent with previous studies (Raymundo et al. 2019), where C. silvestrii was the most sensitive among the tested cladocerans. Additionally, C. rigaudi showed a sensitivity very similar to that found by Harmon et al. (2003) for D. ambigua (CE50-48 h of 0.035 μg L−1) to Chlorpyrifos. Based on the SSD, among the studied groups, crustaceans were the most sensitive, with cladoceran species being the most sensitive, supported by Hanazato (1991) and Havens (1994) who found that cladocerans exhibit higher sensitivity to anthropogenic chemical agents, such as insecticides, compared to other zooplankton groups. Similar to the case of cadmium, for amphibians and rotifers, few data were found, with two species for each compound (as seen in Table S2), which were also the least sensitive.
Regarding the toxicity of Cadmium and Chlorpyrifos mixtures, the results obtained in this study for C. rigaudi differed from those reported by Yu et al. (2019). However, the results for C. silvestrii, where we found dose-dependent antagonistic effects, are supported by those of the same authors. Yu et al. (2019), focusing on the earthworm Eisenia fetida, found antagonism for the Cadmium–Chlorpyrifos binary mixture, the Cadmium–Atrazine–Chlorpyrifos, Cadmium–Chlorpyrifos–Lambda-cyhalothrin, and Cadmium–Chlorpyrifos–Abamectin ternary mixtures, while synergy was observed for the Cadmium–Atrazine–Chlorpyrifos–Abamectin quaternary mixture. They also reported antagonism for the Cadmium–Atrazine–Chlorpyrifos–Lambda-cyhalothrin and Cadmium–Chlorpyrifos–Lambda-cyhalothrin–Abamectin ternary mixtures, as well as the quaternary Cadmium–Atrazine–Chlorpyrifos–Lambda-cyhalothrin–Abamectin mixture. Both studies indicate the occurrence of antagonism in the toxicity of the mixture of cadmium and chlorpyrifos, but there is still little data on the effects of the mixture to different organisms. To understand the ecological relevance of this mixture, future research is necessary, leading to more information and consequently we would perhaps be able to respond to the gaps in relation to this subject.
The study by Zhao et al. (2018) assessed the risk of cadmium in the Longjiang River, located in the Guangxi Zhuang Autonomous Region, China. The HC5 value for species selected by these authors based on NOECs (no observed effect concentration), which included algae, amphibians, aquatic plants, crustaceans, fishes, insects, molluscs, worms, and other invertebrates was 0.714 µg L−1, a value lower than that obtained in the present study for fishes, amphibians, rotifers, and crustaceans based on CL50 or CE50. The ecological risk calculated by Zhao et al. (2018) was high (HQ > 1) during the initial period of pollution caused by an accident and low or no risk (HQ < 1) ~2 years later. In the case of the present study, the risk was high for Brazilian rivers, similar to the onset of pollution in China. These analyses highlight the importance of proper monitoring and management of water resources to protect the health of aquatic ecosystems.
In a study on the toxicity of Chlorpyrifos Rämö et al. (2018) produced a SSD curve for 159 fish species obtaining an HC5 of 6.94 µg L−1 and indicating a low risk for these species. In the present study, the HC5 value obtained (9.96 µg L−1) was similar to that of Rämö et al. (2018), despite our inclusion of more sensitive groups such as crustaceans in the SSD.
According to the Resolution No. 357 of the National Council for the Environment, the maximum allowed value for Cadmium in freshwater quality standards is 0.001 mg L−1 (Brasil 2005). However, based on the data analyzed in this study, this concentration would already pose a medium risk to the considered species. Moreover, concentrations exceeding the maximum permitted values have been reported in waters from the states of São Paulo, Minas Gerais, Amazonas, Amapá, Paraná, and Sergipe (Table S3). Therefore, according to the toxicity results and ecological risk assessment in this study, these values would not be safe for the evaluated species. Based on the data from this study, for the measured environmental concentration to represent a low risk to these species, the value should be below 0.078 µg L−1.
The CONAMA resolution does not cover Chlorpyrifos, and similar to the case of Cadmium the results obtained in this study from measured environmental concentrations (Table S4) indicate that these values are not safe for the species under consideration. Furthermore, based on our data, a value representing negligible risk for these species should be below 0.025 µg L−1.
Therefore, in our view, CONAMA’s values for cadmium should be revised, and Chlorpyrifos should be included in the same. This study can contribute to these decisions, and we recommend that further studies be conducted regarding the compounds individually and in mixture, in order to broaden the range of species tested to include a wider representation of aquatic organisms, including different species of crustaceans, fish, amphibians, mollusks, and plants.
Conclusions
In this study, both compounds individually caused lethal effects on the cladocerans C. rigaudi (EC50-48 h 0.02 mg L−1 for Cadmium and 0.047 µg L−1 for Chlorpyrifos) and C. silvestrii (EC50-48 h 0.026 mg L−1 for Cadmium and 0.062 µg L−1 for Chlorpyrifos). In mixture, the compounds resulted in additive effects for C. rigaudi and dose-dependent antagonism for C. silvestrii, even though they are congeneric species.
The species considered in the SSD showed an HC5 of 3.13 µg L−1 for Cadmium and 9.96 µg L−1 for Chlorpyrifos, with values above these having been measured for both compounds in Brazilian water bodies. The ecological risk assessment conducted in this study indicated that the measured environmental concentrations for both compounds would not be safe for the species considered in the SSD.
Furthermore, both Ceriodaphnia rigaudi and Ceriodaphnia silvestrii seem to be sensitive and suitable as native test organisms for ecotoxicological assays with metals and pesticides, enhancing their applicability in ecological risk assessments in tropical regions. However, further studies are necessary to deepen our understanding, despite the species showing promising results.
Data availability
No datasets were generated or analyzed during the current study.
References
ABNT (2016) NBR 12713:2016 - Ecotoxicologia Aquática – Toxicidade aguda – Método de ensaio comD aphnia spp (Crustacea, Cladocera). Associação Brasileira de Normas Técnicas.
Aldenberg T, Jaworska JS (2000) Uncertainty of the hazardous concentration and fraction affected for normal species sensitivity distributions. Ecotoxicol Environ Saf 46:1–18. https://doi.org/10.1006/eesa.1999.1869
ANVISA (2021) Monografia C-20. Agência Nacional de Vigilância Sanitária. https://www.gov.br/anvisa/pt-br/setorregulado/regularizacao/agrotoxicos/monografias/monografias-autorizadas/c/4226json-file-1/view
Baby J, Raj J, Biby E, et al. (2011) Toxic effect of heavy metals on aquatic environment. Int J Biol Chem Sci 4. https://doi.org/10.4314/ijbcs.v4i4.62976
Biesinger KE, Christensen GM (1972) Effects of various metals on survival, growth, reproduction, and metabolism of Daphnia magna. J Fish Res Board Can 29:1691–1700. https://doi.org/10.1139/f72-269
Bliss CI (1939) The toxicity of poisons applied jointly1. Ann Appl Biol 26:585–615. https://doi.org/10.1111/j.1744-7348.1939.tb06990.x
Brasil (2005) Conselho Nacional de Recursos Hídricos (CNRH) Resolution No. 357, dated March 17, 2005. Classificação de águas, doces, salobras e salinas do Território Nacional. RC n°357, de 17 de março de 2005. Published in the Official Gazette
CETESB (2021) Diagnóstico da contaminação de águas superficiais, subterrâneas e sedimentos por agrotóxicos. São Paulo: Companhia Ambiental do Estado de São Paulo
CONAMA (2005) Resolução no 357, 17 de março de 2005. Conselho Nacional do Meio Ambiente. https://www.icmbio.gov.br/cepsul/images/stories/legislacao/Resolucao/2005/res_conama_357_2005_classificacao_corpos_agua_rtfcda_altrd_res_393_2007_397_2008_410_2009_430_2011.pdf
Eaton JG, McKim JM, Holcombe GW (1978) Metal toxicity to embryos and larvae of seven freshwater fish species—I. Cadmium. Bull Environ Contam Toxicol 19:95–103. https://doi.org/10.1007/BF01685772
El-Nahhal I, El-Nahhal Y (2021) Pesticide residues in drinking water, their potential risk to human health and removal options. J Environ Manag 299:113611. https://doi.org/10.1016/j.jenvman.2021.113611
EPA (1998) Guidelines for ecological risk assessment. U.S. Environmental Protection Agency – EPA/630/R-95/002F
EPA (2002) Ceriodaphnia dubia, survival and reproduction test; chronic toxicity excerpt from: short-term methods for estimating the chronic toxicity of effluents and receiving waters to freshwater organisms - U.S. Environmental Protection Agency – EPA-821-R-02-013.
Freitas EC, Rocha O (2011) Acute toxicity tests with the tropical cladoceran Pseudosida ramosa: the importance of using native species as test organisms. Arch Environ Contam Toxicol 60:241–249. https://doi.org/10.1007/s00244-010-9541-2
Gautam RK, Sharma SK, Mahiya S, Chattopadhyaya MC (2014) Contamination of heavy metals in aquatic media: transport, toxicity and technologies for remediation. In: Heavy metals in water. Royal Society of Chemistry, Cambridge, pp 1–24
Gomes DF, da Silva Pinto TJ, Raymundo LB et al. (2023) Ecological risk assessment for metals in sediment and waters from the Brazilian Amazon region. Chemosphere 345:140413. https://doi.org/10.1016/j.chemosphere.2023.140413
Hanazato T (1991) Effects of repeated application of carbaryl on zooplankton communities in experimental ponds with or without the predator Chaoborus. Environ Pollut 74:309–324. https://doi.org/10.1016/0269-7491(91)90078-B
Harmon SM, Specht WL, Chandler GT (2003) A comparison of the Daphnids Ceriodaphnia dubia and Daphnia ambigua for their utilization in routine toxicity testing in the Southeastern United States. Arch Environ Contam Toxicol 45:79–85. https://doi.org/10.1007/s00244-002-0116-8
Havens KE (1994) An experimental comparison of the effects of two chemical stressors on a freshwater zooplankton assemblage. Environ Pollut 84:245–251. https://doi.org/10.1016/0269-7491(94)90135-X
IBAMA (2022) Relatórios de comercialização de agrotóxicos. Instituto Brasileiro do Meio Ambiente. https://www.gov.br/ibama/pt-br/assuntos/quimicos-e-biologicos/agrotoxicos/relatorios-de-comercializacao-de-agrotoxicos
ISO (2000) Water quality - Determination of long term toxicity of substances to Daphnia magna Straus (Cladocera, Crustacea). ISO 10706:2000, 1–17
Jonker MJ, Svendsen Claus, Bedaux Jacques JM et al. (2005) Significance testing of synergistic/antagonistic, dose level–dependent, or dose ratio–dependent effects in mixture dose–response analysis. Environ Toxicol Chem 24:2701–2713
Laabs V, Amelung W, Pinto AA et al. (2002) Pesticides in surface water, sediment, and rainfall of the northeastern Pantanal Basin, Brazil. J Environ Qual 31:1636–1648. https://doi.org/10.2134/jeq2002.1636
Lalande M, Pinel-Alloul B (1983) Acute toxicity of cadmium, copper, mercury and zinc to Chydorus Sphaericus (Cladocera) from three Quebec Lakes. Water Qual Res J 18:103–114. https://doi.org/10.2166/wqrj.1983.007
Lima DPde, Santos C, Silva RdeS et al. (2015) Contaminação por metais pesados em peixes e água da bacia do rio Cassiporé, Estado do Amapá, Brasil. Acta Amazon 45:405–414. https://doi.org/10.1590/1809-4392201403995
Liu P, Wu C, Chang X et al. (2014) Assessment of chlorpyrifos exposure and absorbed daily doses among infants living in an agricultural area of the Province of Jiangsu, China. Int Arch Occup Environ Health 87:753–762. https://doi.org/10.1007/s00420-013-0918-1
Mansano AS, Moreira RA, Dornfeld HC et al. (2017) Effects of diuron and carbofuran and their mixtures on the microalgae Raphidocelis subcapitata. Ecotoxicol Environ Saf 142:312–321. https://doi.org/10.1016/j.ecoenv.2017.04.024
Mansano AS, Moreira RA, Dornfeld HC et al. (2018) Acute and chronic toxicity of diuron and carbofuran to the neotropical cladoceran Ceriodaphnia silvestrii. Environ Sci Pollut Res 25:13335–13346. https://doi.org/10.1007/s11356-016-8274-9
Marshall JS (1979) Cadmium toxicity to laboratory and field populations of Daphnia galeata mendotae. Bull Environ Contam Toxicol 21:453–457. https://doi.org/10.1007/BF01685452
Martins SE, Bianchini A (2011) Toxicity tests aiming to protect Brazilian aquatic systems: current status and implications for management. J Environ Monit 13:1866. https://doi.org/10.1039/c0em00787k
Miguel M (2020) Efeito do hormônio sintético 17 α-etinilestradiol e do herbicida atrazina em espécies de dois níveis tróficos. Tese (Doutorado em Ciência), Universidade de São Paulo
Mohammed A (2007) Comparative sensitivities of the tropical cladoceran, Ceriodaphnia rigaudii and the temperate species Daphnia magna to seven toxicants. Toxicol Environ Chem 89:347–352. https://doi.org/10.1080/02772240601035888
Moreira RA, Rocha O, Pinto TJ, da S et al. (2020) Life-history traits response to effects of fish predation (kairomones), fipronil and 2,4-D on neotropical cladoceran Ceriodaphnia silvestrii. Arch Environ Contam Toxicol 79:298–309. https://doi.org/10.1007/s00244-020-00754-7
Morteza Z, Mousavi SB, Baghestani MA, Aitio A (2017) An assessment of agricultural pesticide use in Iran, 2012-2014. J Environ Health Sci Eng 15:10. https://doi.org/10.1186/s40201-017-0272-4
Oliveira HSP, França SCA, Rocha EJP (2015) Atividades de mineração e avaliação de metais em água superficial, sedimento de fundo e peixes no Rio Tapajós. In: Amazônia em tempo: estudos climáticos e socioambientais. Embrapa, Belém, pp 195–222
Olker JH, Elonen CM, Pilli A et al. (2022) The ECOTOXicology Knowledgebase: a curated database of ecologically relevant toxicity tests to support environmental research and risk assessment. Environ Toxicol Chem 41:1520–1539. https://doi.org/10.1002/etc.5324
Pérez J, Domingues I, Soares AMVM, Loureiro S (2011) Growth rate of Pseudokirchneriella subcapitata exposed to herbicides found in surface waters in the Alqueva reservoir (Portugal): a bottom-up approach using binary mixtures. Ecotoxicology 20:1167–1175. https://doi.org/10.1007/s10646-011-0661-x
Rämö RA, van den Brink PJ, Ruepert C et al. (2018) Environmental risk assessment of pesticides in the River Madre de Dios, Costa Rica using PERPEST, SSD, and msPAF models. Environ Sci Pollut Res 25:13254–13269. https://doi.org/10.1007/s11356-016-7375-9
Raymundo LB, Rocha O, Moreira RA et al. (2019) Sensitivity of tropical cladocerans to chlorpyrifos and other insecticides as compared to their temperate counterparts. Chemosphere 220:937–942. https://doi.org/10.1016/j.chemosphere.2019.01.005
Ribeiro EV, Magalhães Junior AP, Horn AH, Trindade WM (2012) Metais Pesados e Qualidade da Água do Rio Sao Francisco no Seguimento entre Três Marias e Pirapora – MG: Índice de contaminação. Geonomos. https://doi.org/10.18285/geonomos.v20i1.27
Ruiz-Arias MA, Medina-Díaz IM, Bernal-Hernández YY et al. (2023) The situation of chlorpyrifos in Mexico: a case study in environmental samples and aquatic organisms. Environ Geochem Health 45:6323–6351. https://doi.org/10.1007/s10653-023-01618-4
Sánchez-Bayo F, Baskaran S, Kennedy IR (2002) Ecological relative risk (EcoRR): another approach for risk assessment of pesticides in agriculture. Agric Ecosyst Environ 91:37–57. https://doi.org/10.1016/S0167-8809(01)00258-4
Sobha K, Poornima A, Harini P, Veeraiah K (1970) A study on biochemical changes in the fresh water fish, Catla catla; (Hamilton) exposed to the heavy metal toxicant cadmium chloride. Kathmandu Univ J Sci Eng Technol 3:1–11. https://doi.org/10.3126/kuset.v3i2.2890
Solomon KR, Williams MW, Mackay D, et al. (2014) Properties and uses of chlorpyrifos in the United States. In: Giesy JP, Solomon KR (eds.) Aquatic systems in the United States, 231st edn. Springer Open, Summerfield, pp 13–30
STATSOFT (2014) Statistica: A Comprehensive Software Package for Data Analysis. StatSoft, Inc
Takenaka RA (2007) Avaliação da toxicidade de Microcystis aeruginosa e de florações naturais de cianobactérias de reservatórios do Rio Tietê, SP. Tese (Doutorado), Universidade de São Paulo
Thorley J, Schwarz C (2018) ssdtools: an R package to fit species sensitivity distributions. J Open Source Softw 3:1082. https://doi.org/10.21105/joss.01082
Villar D, Schaeffer DJ (2022) Chlorpyrifos should be banned in agriculture and livestock production in Colombia. Revista Colombiana de Ciencias Pecuarias 35:61–67. https://doi.org/10.17533/udea.rccp.v35n2a7
Wang P, Wang J, Sun Y-J et al. (2017) Cadmium and chlorpyrifos inhibit cellular immune response in spleen of rats. Environ Toxicol 32:1927–1936. https://doi.org/10.1002/tox.22415
World Health Organization (1992) Cadmium: environmental aspects. https://apps.who.int/iris/handle/10665/39366
Yan F, Li N, Yang Z, Qian B (2022) Ecological risk evaluation of Baihetan Dam based on fuzzy hazard quotient model. Water 14:2694. https://doi.org/10.3390/w14172694
Yoon SH, Ha HJ, Lee SJ, Jho EH (2017) Effect of water hardness on toxicity of cadmium and zinc. J Korean Soc Water Environ 33:556–652
Yu Y, Li X, Yang G et al. (2019) Joint toxic effects of cadmium and four pesticides on the earthworm (Eisenia fetida). Chemosphere 227:489–495. https://doi.org/10.1016/j.chemosphere.2019.04.064
Zhao Q, Dourson M, Gadagbui B (2006) A review of the reference dose for chlorpyrifos. Regul Toxicol Pharmacol 44:111–124. https://doi.org/10.1016/j.yrtph.2005.10.003
Zhao X-M, Yao L-A, Ma Q-L et al. (2018) Distribution and ecological risk assessment of cadmium in water and sediment in Longjiang River, China: implication on water quality management after pollution accident. Chemosphere 194:107–116. https://doi.org/10.1016/j.chemosphere.2017.11.127
Acknowledgements
LBR thanks Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
Author information
Authors and Affiliations
Contributions
Larissa Broggio Raymundo: conceptualization, methodology, formal analysis, investigation, writing; Diego Ferreira Gomes: formal analysis, methodology, and review; Mariana Miguel: formal analysis, methodology, and editing; Raquel Aparecida Moreira: formal analysis, methodology, and editing; Odete Rocha: conceptualization, writing, and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Raymundo, L.B., Gomes, D.F., Miguel, M. et al. Effects of acute toxicity of the pesticide Chlorpyrifos and the metal Cadmium, both individually and in mixtures, on two species of native neotropical cladocerans. Ecotoxicology 33, 642–652 (2024). https://doi.org/10.1007/s10646-024-02761-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-024-02761-z