Abstract
Jinju Bay is a semi-closed bay located on the mid-southern coast of Korea. The dietary habitats of eight dominant fish species—Liparis tanakae (Tanaka’s snailfish), Zoarces gillii (an eelpout), Pennahia argentata (silver croaker), Conger myriaster (whitespotted conger), Callionymus valenciennei (Valenciennes’ dragonet), Thryssa kammalensis (Kammal thryssa), Sillago japonica (Japanese whiting), and Pholis fangi (a gunnel)—collected from inside and outside of Jinju Bay were analyzed using stomach content analyses. As a result of multivariate analysis for each season, there were significant differences in dietary composition between species and sampling sites in all seasons, except between study sites during summer. The eight common species were divided into four groups based on the cluster analysis. The first group contained C. valenciennei and Z. gillii, which showed preferences for amphipods, while the second group, comprising T. kammalensis, fed mainly on crabs and mantis shrimps. The two species (P. fangi and S. japonica) in the third group tended to specialize in feeding on polychaetes, whereas the three species (L. tanakae, C. myriaster, and P. argentata) in the fourth group preyed on carid shrimp and teleosts. Although the eight species occurred in the same habitat and ingested a similar range of food resources, the contributions of different prey items were different in each species’ diet—differences in food resource use between species were evident. In conclusion, the results of stomach content analyses of the eight species showed evidence of ecological separation between species occurring inside and outside Jinju Bay.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The size of coastal fishing grounds in Korean seas is gradually decreasing due to coastal development. Consequently, the biological resources in coastal waters are being depleted due to habitat loss, overfishing, and environmental pollution (Yoo et al. 1999; Zhang et al. 2003; Yoon et al. 2014). Coastal areas often act as spawning and nursery grounds for numerous marine organisms, and are thus an important habitat during their early life history (Hajisamae and Chou 2003; Song et al. 2012). These areas also have high economic value owing to their high fishing productivity (Houde and Rutherford 1993). Among the 10 sea zones designated as fisheries resource-protected areas in Korea, eight are located on the southern coast; thus, the southern coastal regions are highly regarded for their importance in terms of fishery resources (Yoo et al. 1999; Lee et al. 2009). The study area, Jinju Bay, is located in the central part of the southern coast and is one of the previously mentioned fisheries resource-protected areas, connecting the Namgang River in the northern part to the open sea on the east side (Song et al. 2012). Approximately 25% of the bay area consists of a shallow intertidal zone (Choo and Yoon 2015).
Generally, bay environments are rich in planktonic organisms because of the introduction of a large amount of nutrients from the land through river discharge (Selleslagh et al. 2009; Newton et al. 2014); these areas provide abundant food resources during the early growth of young marine organisms (Swearer et al. 1999). As such, bay environmental conditions have partially attracted attention as spawning and nursery grounds to facilitate early growth and high survival rates during the early life history of marine organisms (Vasconcelos et al. 2010). Studies on fish communities in the southern coastal areas of Korea have been conducted in Gwangyang Bay and Yeoja Bay (Cha and Park 1997; Lee et al. 2011), and have reported that small-sized fish species such as Nuchequula nuchalis and Thryssa adelae, and/or younger specimens of Konosirus punctatus and Pennahia argentata, were dominant in these areas. In addition, a number of studies on the feeding relationships among co-occurring species in shallow coastal habitats have been conducted to explain their ecological function and biological relationships (e.g., Motta et al. 1995; Kwak et al. 2015). Recently, Park et al. (2018) reported on the feeding relationship among six major fish species in Gwangyang Bay in southern Korea and concluded that each species showed different feeding characteristics according to their preferences for different food resources. These results indicate that various fishes occurring in the same area show different life-history strategies to minimize interspecific competition by sharing or separating available food resources (Platell and Potter 2001).
The eight dominant species in this study (Myoung et al. 2020)—Liparis tanakae (Tanaka’s snailfish), Zoarces gillii (an eelpout), Pennahia argentata (silver croaker), Conger myriaster (whitespotted conger), Callionymus valenciennei (Valenciennes’ dragonet), Thryssa kammalensis (Kammal thryssa), Sillago japonica (Japanese whiting), and Pholis fangi (a gunnel)—are mainly distributed in the shallow coastal area of southern Korea, including Jinju Bay, occasionally forming dominant fish groups (Kwak and Huh 2003; Lee et al. 2011; Kwak et al. 2012; Jeong et al. 2013; Park et al. 2020a, b). Jinju Bay may be used as a spawning and nursery ground for several fish species because their eggs and larvae have been collected from Jinju Bay (Myoung et al. 2021). Among the above species, L. tanakae, Z. gillii, P. argentata, and C. myriaster are important commercial fish species in Korea (Kim et al. 2005). For example, L. tanakae is a representative winter commercial species in Korea; not only the fish body but also the eggs attached to fishing gear (e.g., traps and gill nets) are used as food (Kosaka 1971; Kim et al. 1986; Myoung et al. 2016). In addition, C. myriaster is an economically important fish species that is exported to Japan (Ryu et al. 2009).
The purposes of this study were (1) to identify the feeding characteristics of eight abundant fish species inhabiting Jinju Bay via stomach content analyses and (2) to examine any differences in the utilization of food resources among the species. The results of this study can identify the feeding relationship between major fish species occurring in the bay and will serve as basic ecological data for the resource management of dominant species inhabiting the areas inside and outside of Jinju Bay. In addition, it is important to understand the characteristics of the bay ecosystem by identifying the structure of the food network between these co-occurring species.
Materials and methods
Survey area and sampling method
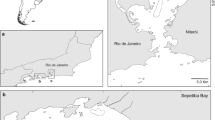
In this study, fish sampling was conducted at stations inside and outside Jinju Bay, located in the middle-southern waters off Korea (Fig. 1). The distance between the two stations was approximately 15 km. The sampling stations were set based on the difference in the intensity of salinity stratification according to the strength of freshwater flows from Namgang Dam, located in the northern part of Jinju Bay (Kim et al. 2010; Kang et al. 2011). Fish samples were collected monthly from March 2015 to November 2015 using a shrimp beam trawl net with 6-m width, 1-m height, and 20-mm mesh. The shrimp beam trawl was towed at approximately 1.6 knots for 1 h, with an estimated 17,780 m2 coverage per sampling event.
The collected fishes were identified according to the guideline of taxonomic classification for Korean fishes (Kim et al. 2005; Nakabo 2013), and the scientific name and taxonomic classification followed Kim et al. (2019) and Nelson et al. (2016). This study analyzed the stomach contents of the eight abundant fish species among a total of 81 species collected by shrimp beam trawl (Myoung et al. 2020). For each specimen, the TL and weight were measured to the nearest 0.1 cm and 0.1 g, respectively. The stomach of each specimen was removed, and the contents were preserved in 99% ethanol.
Stomach contents analysis
The stomachs were cut open and incised using tweezers and scissors under a dissecting microscope. All prey items were identified to the lowest possible taxonomic level, and individuals that proved difficult to identify were classified to the family or order level. The number and weight of all food organisms in the stomach contents were measured. Because gravimetric dietary data are considered to best represent the relative importance of each prey taxon, especially when different-sized prey are ingested (Hyslop 1980), the dietary data were expressed as a percentage weight (%W = 100 × Wi × WT−1) of all prey organisms for each specimen’s stomach, where Wi is the weight of prey individual i and WT is the total weight of prey items. The %W of each prey item in each stomach is expressed as the average value for all individuals.
Data analysis
Cumulative prey curves were calculated for each species to assess whether a sufficient number of stomachs were analyzed to describe the diet (Ferry and Cailliet 1996). The dietary data were randomized 100 times, and the cumulative number of new prey taxa was recounted for each randomization. Because visual examination of prey curves for an asymptote is unreliable, the slope of the linear regression (b) through the last five subsamples validated the sample size, where b ≤ 0.05 signified acceptable leveling off of the prey curve for diet analyses (Bizzarro et al. 2009). The analysis was performed with R version 4.2.1 (R Core Team 2022) using the ‘vegan’ packages (Oksanen et al. 2022).
Multivariate analyses based on weight dietary data were used to analyze inter- and intraspecific feeding relationships. In this study, the dietary data of eight species were analyzed from two perspectives. First, since the eight dominant species did not occur together in each season, analyses of the intra- and interspecific feeding characteristics against seasonal available food resources were conducted separately for each season. Consequently, the feeding relationship (variations between species) in two habitats (inside and outside Jinju Bay) was analyzed according to each season (spring, summer, and autumn). Second, although the occurrence trends of the dominant species did not overlap seasonally, the sampling method and time (seasonal sampling) had limitations in including the true occurrence of dominant fish species in the study area. In addition, the overall feeding characteristics of fish species can be reflected by their average food consumption (aggregating feeding over potential habitats and seasons) inherent in each species (Gerking 1994). Therefore, the population level feeding relationships were analyzed by considering only the “species.”
To analyze the feeding relationship, three to five individuals collected at the same station and season were randomly sorted by species into subgroups, and the average weight percentage of the prey taxa was calculated for each subgroup to make a sample for multivariate analysis (Park et al. 2017b). Such subgrouping can reduce the number of prey taxa that show a weight contribution of “0” in the stomach contents, thus increasing the effectiveness of multivariate analysis (Platell and Potter 2001; White et al. 2004; Marshall et al. 2008). The dietary data were square root-transformed to avoid any tendency of the main dietary components being excessively dominant, and a similarity matrix was constructed using the Bray–Curtis similarity (Platell and Potter 2001). The Bray–Curtis similarity matrix was visualized using non-metric multidimensional scaling (nMDS) and cluster analysis. Significant differences in dietary composition between species and/or habitats were tested using permutational multivariate analysis of variance (PERMANOVA). PERMANOVA is a non-parametric distance-based analysis of variance that uses the permutation method to test a hypothesis. If there was a significant difference in the PERMANOVA analysis, posteriori pairwise PERMANOVA comparisons were performed to determine which interaction terms differed significantly among variables within each factor level. Canonical analysis of principal coordinates (CAP) was used to determine which prey taxon showed correlation coefficients contributing to the difference in the feeding relationships among species. The prey taxa with correlation coefficients higher than 0.4 were plotted on CAP component axes 1 and 2. For this analysis, the PRIMER v7 multivariate statistics package with the PERMANOVA + add-on module was used (Anderson et al. 2008; Clarke and Gorley 2015).
Diet overlap among eight species was calculated using Schoener’s index (Cxy; Schoener 1970), as this method best estimates dietary similarities over the potential range of overlap (Wallace 1981). Cxy was calculated as follows: Cxy = 1 − 0·5 (Σ|Pxi − Pyi|), where Pxi and Pyi are the proportions of prey taxa i (in terms of relative weight contribution) in the diet of species x and y. Cxy ranged from 0 (diets containing no items in common) to 1 (complete overlap). An index value > 0.6 indicated high diet similarity and was considered biologically significant (Langton 1982).
Results
Composition of stomach contents
In this study, the diets of 976 individuals of the eight species were examined. The proportion of empty stomachs ranged from 1.3% (L. tanakae) to 42.5% (C. valenciennei). Nineteen identifiable prey taxa were identified in the remaining 820 stomachs (after the removal of empty ones) (Table 1). Cumulative prey curves for all species aggregating all seasons and sites reached stable asymptotes, indicating sufficient sample sizes for an accurate description of the diets of the species included in the study (Fig. S1). While the proportion of individuals sampled for stomach contents was not reached for some seasonal and spatial subgroups of P. argentata, C. valenciennei, C. myriaster, and S. japonica, all cumulative prey curves calculated for both sites according to three seasons for the diets of L. tanakae, Z. gillii, T. kammalensis, and P. fangi attained asymptoticity (Fig. S2).
In the diets of L. tanakae, carid shrimps were the most dominant prey taxon, with a %W of 85.4%. In the diets of Z. gillii, gammarid amphipods were the most abundant (63.1% in %W), followed by bivalves (10.9%). Both teleosts and carid shrimps were important prey items in the diets of P. argentata and C. myriaster, constituting 34.4% and 31.3% of the %W (teleosts) and 27.3% and 32.0% of the %W (carid shrimps), respectively. C. valenciennei mainly consumed bivalves (37.1%) and gammarid amphipods (24.8%), whereas T. kammalensis mostly ingested crabs (52.9%), followed by stomatopods (16.6%). Polychaetes only contributed to the diets of S. japonica and P. fangi, with %W values of 54.0% and 47.9%, respectively. Among the minor prey taxa, ostracods occurred only in the diets of C. valenciennei (9.4%), cumaceans in Z. gillii (8.0%), isopods in P. argentata (0.4%), and mysids in T. kammalensis (0.6%), whereas seaweed was found only in the diets of L. tanakae in small amounts (< 0.1%).
Variations in stomach contents according to season and station
The eight dominant fish species showed different trends in food consumption according to season and habitat (Fig. 2). During the spring, L. tanakae and Z. gillii indicated similar diets inside and outside the bay in that they exclusively fed on carid shrimps and amphipods, respectively. C. myriaster and P. fangi showed a gradually increased consumption of carid shrimps and polychaetes, but decreased consumption of teleosts and carid shrimps at the outside site, respectively. However, S. japonica showed a more diverse dietary composition inside than outside the bay. Among the fish species that occurred in summer, C. myriaster, P. argentata, T. kammalensis, and Z. gillii showed significant differences in their diets between the inside and outside sites. These differences were mainly due to the increasing or decreasing consumption of specific food items at the outside site. For example, carid shrimps were more abundant in the diets of C. myriaster and P. argentata, and the consumption of crabs increased in the diets of T. kammalensis at the outside site. P. fangi and S. japonica collectively consumed polychaetes at both sites, although the consumption of other food items differed slightly between the two sites. Z. gillii consumed mostly gammarid amphipods at the inside site, but both gammarid amphipods and bivalves contributed to the diet. Fish species collected during autumn also showed differences in their dietary compositions, as indicated by the spring and summer trends. P. argentata and C. myriaster collected at the inside site fed evenly on various food items, whereas both species mainly ingested teleosts at the outside site. In the case of C. valenciennei, gammarid amphipods mainly contributed to the diets at the inside site, whereas bivalves showed the highest contribution to the diet at the outside site. S. japonica fed mainly on polychaetes at both the inside and outside sites, and T. kammalensis showed a surprisingly high consumption of stomatopods at the outside site.
Mean percentage weight contributions (%W) of the different dietary component of eight fish species in Jinju Bay, Korea. Species codes were shown in Table 1 legend
The two-way PERMANOVA for each season showed significant differences in the diets between species and between stations in all seasons except for between sites during summer (Table 2). There also were significant interactions between species and sites in all seasons. The PERMANOVA pairwise comparison of species–site interactions showed that the diets between the inside and outside sites were significantly different for S. japonica and Z. gillii during spring; for S. japonica, T. kammalensis, and Z. gillii during summer; and for P. argentata, C. valenciennei, and S. japonica during autumn (Table 3). In addition, there were significant differences in most comparisons between species in each season within each site. Between-species comparisons of L. tanakae and P. fangi, L. tanakae and Z. gillii, and P. fangi and Z. gillii collected during spring showed significant differences at both the inside and outside sites. During summer, 19 pairwise species comparisons were significantly different at the outside site, while only five comparisons were significantly different at the inside site. The pairs P. argentata and C. valenciennei and P. argentata and S. japonica collected during autumn also showed significant differences at both sites (Table 4).
Population level feeding relationships among eight species
PERMANOVA with unrestricted permutations of raw data showed a significant difference in dietary composition among fish species (PERMANOVA, pseudo-F = 51.898, p = 0.001). The differences in the dietary compositions among the fish species were visually divided into four groups based on both cluster analysis and nMDS ordination plot at a similarity level of 55% (Fig. 3). The first group included Z. gillii and C. valenciennei, which mainly consumed gammarid amphipods and bivalves. Only one species (T. kammalensis) belonged to the second group. The third group (P. fangi and S. japonica) preferred polychaetes as their primary food source. The fourth group included three species (L. tanakae, C. myriaster, and P. argentata), which ingested both carid shrimps and teleosts as their main prey items.
Cluster analysis (A) and non-metric multidimensional scaling (nMDS) ordination (B) of dietary compositions constructed from Bray–Curtis similarity matrix for eight fish species inhabiting Jinju Bay, Korea. Species codes are shown in the Table 1 legend
Canonical analyses of principal coordinates (CAP) further demonstrated that each feeding group could be classified according to the different contributions of each food item (Fig. 4). Gammarid amphipods distinguished Z. gillii and C. valenciennei from other groups, and polychaetes were key in separating P. fangi and S. japonica from other species. Three prey taxa (carid shrimps, teleosts, and stomatopods) were characteristic of the diets of L. tanakae, P. argentata, and C. myriaster, and crabs showed a strong contribution to the diets of T. kammalensis.
Canonical analysis of principal coordinates (CAP) ordination plot to discriminate among the diets of species. Species correlations with each canonical axis are represented as vectors for species with correlations greater than 0.4. Vectors represent Pearson correlations, and the circle indicates a correlation of 1. Species codes are shown in the Table 1 legend
Dietary overlaps between species comparisons were mostly lower than the biological significance of 0.6; only the comparison between C. myriaster and P. argentata, and between P. fangi and S. japonica, revealed an overlap of > 0.6 (Table 5), whereas the overlap values were relatively low between species in feeding group A or B and group D (see Fig. 3), indicating that these groups of species had dissimilar diets.
Discussion
Individuals of the eight species collected from Jinju Bay were mostly juveniles comparable in size to the adults or small-sized specimens, but only some species included their adult conspecifics (Kim et al. 2005; Froese and Pauly 2022). For example, the length of maturity of P. argentata is 19.3-cm TL (Jeon et al. 2020) and the standard length of L. tanakae is 38.2 cm (Park 2010). In this study, only 16 mature individuals (> 21.7 cm TL) of the former species and one specimen (40.8 cm TL) of the latter species were collected; the proportions of immature individuals were 89.3% and 98.8%, respectively. In addition, the sizes of all C. myriaster specimens collected were less than the maturity size of 50.8 cm TL (Kim et al. 2011). Callionymus valenciennei, T. kammalensis, and P. fangi are relatively small-sized species, and their sizes at maturity do not reach 20-cm TL (Froese and Pauly 2022). Only Z. gillii and S. japonica included both their juvenile and adult specimens. Shallow coastal regions, including estuaries and seagrass habitats, are known as good places for nursery grounds for the early stages of marine organisms (Vasconcelos et al. 2010; Park and Kwak 2018); they provide a variety of food organisms compared to offshore coastal areas and are a safe habitat for the early growth and survival of various marine organisms (Lefcheck et al. 2019; Youn et al. 2019). Among the 81 fish species collected in the study area, 57 species from both the inside and outside sites were all immature specimens (Myoung et al. 2020). Song et al. (2019) also showed that more smaller and juvenile specimens were collected in the inner bay than in the offshore coastal waters of Namhae Island, Korea.
Most of the species analyzed in this study mainly consumed benthic prey organisms, but teleosts also showed moderate contributions to the diets of P. argentata and C. myriaster (Table 1). Several previous studies have reported similar dietary compositions to the current study. In previous studies, L. tanakae, Z. gillii, and C. valenciennei inhabiting the southern sea of Korea were found to mainly consume benthic carid shrimps, gammarid amphipods, and polychaetes, respectively, which were also dominant in the diets of the same species in this study (Huh 1997; Huh and Kwak 1998; Huh and Baeck 2000, 2003). In addition, higher contributions of teleosts to the diets of P. argentata and C. myriaster have been reported previously (Huh and Kwak 1998; Koh et al. 2014). Although T. kammalensis is classified as a pelagic fish, it mainly preys on crabs. However, all crabs found in the diets of T. kammalensis were in the zoea stage. In previous studies, T. kammalensis was also found to feed on arrow worms and megalopa-stage crabs (Baeck et al. 2014). Therefore, T. kammalensis likely prefers large pelagic prey organisms with weak mobility.
In this study, C. myriaster and P. argentata consumed more teleosts than did other fish species. When comparing similar-sized specimens from previous studies, similar results to this study were also observed in the diets of C. myriaster inhabiting an eelgrass bed in Gwangyang Bay and P. argentata occurring around Sejon Island on the southern coast of Korea (Huh and Kwak 1998; Koh et al. 2014). However, P. argentata inhabiting the Nakdong River estuary showed results different from those in the present study (Huh et al. 2018). In the Nakdong River estuary, a similar total length of P. argentata (9.0–23.9 cm TL) consumed only a small amount of teleosts, but carid shrimps constituted almost half of the diets (Huh et al. 2018). This is thought to be due to the high abundance of carid shrimps as food resources for predators in estuarine environments (Upchurch and Wenner 2008). Therefore, the regional differences in preferred food organisms are the result of the different types of available food resources harboring different habitats (Kim et al. 2013).
Carid shrimps accounted for most of the dietary content of L. tanakae, with crangonid shrimp being the most common carid species in the diets (Table S1). A high ratio of crangonid shrimps has also been found in the diets of L. tanakae occurring elsewhere, including the southern coast of Korea (Huh 1997), the southern part of the East Sea (Park et al. 2017b), the Seto Inland Sea of Japan (Kobayashi and Hiyama 1991), and the Yellow Sea off China (Jin et al. 2010). Crangonid shrimps mainly inhabit shallow coastal waters, including estuaries and inside the bay (Hanamura and Matsuoka 2003; Maher et al. 2013), and constitute a major group in the benthic community of Jinju Bay (Song et al. 2012). Abundant prey organisms in the marine ecosystem have a high probability of encountering predators and consequently provide an increase in catch success for predators (Persson and Diehl 1990). Similarly, the highly diverse polychaetes in Jinju Bay (accounting for 80% of all benthic infauna; Kang et al. 2002) likely enhanced the foraging ratio of polychaetes for P. fangi and S. japonica.
Although stomatopods were not a common food item for the fish inhabiting Jinju Bay, it showed a tendency to increase in the diets of fishes collected outside of the bay during autumn. Stomatopods were ingested by C. valenciennei, S. japonica, P. argentata, and T. kammalensis during autumn and constituted 56.6% and 23.8% of the diets of T. kammalensis and P. argentata, respectively. Most stomatopods ingested by the fishes are young juveniles of Oratosquilla oratoria. The spawning season of stomatopods occurs between May and September in the innermost coastal habitats of the Northwest Pacific regions, including Korean waters, and then they gradually move to deeper waters toward offshore as they grow (Hamano et al. 1987; Kodama et al. 2004). Because our autumn sampling was conducted between September and November, some fish species in this study likely had a high chance of capturing young stomatopods outside of Jinju Bay. In addition, because the stomatopods consumed were mostly pre-settled larvae and/or juveniles on the benthic layer (Hamano et al. 1987), they were more abundant in the diets of benthopelagic fishes (i.e., P. argentata and T. kammalensis), but were less important for other benthic fishes.
Although the eight species in this study ingested a similar range of food resources, the contribution of each prey item was different in each species’ diet, and they were consequently divided into four feeding groups according to the type of food they consumed. Such differentiation of prey items among the eight species may reflect their different foraging behaviors and/or mouth morphologies (Hyndes et al. 1997; Park et al. 2017a). The two species (Z. gillii and C. valenciennei) in the first group are typical benthic fishes and show the development of a bottom-faced mouth morphology that facilitates feeding more on epifaunal prey items, such as gammarid amphipods, inhabiting the surface of the benthic layer (Fugi et al. 2001; Bonato et al. 2017). The body of T. kammalensis is suitable for feeding on small-sized pelagic prey items because the species is a small-sized pelagic fish and shows pelagic feeding behavior, unlike other fish species (Baeck et al. 2014). The two species (S. japonica and P. fangi) in the third group have relatively small heads and protruding mouth structures that are suitable for feeding on infaunal prey items such as polychaetes (Huh et al. 2018; Park et al. 2020a, b). In contrast, the fourth group of fishes, comprising L. tanakae, P. argentata, and C. myriaster, have relatively large mouth sizes and high mobility, which are advantageous for preying on large and mobile prey items, such as teleosts and carid shrimps (Huh 1997; Park et al. 2017b; Huh et al. 2018). Diet overlaps also indicated relatively higher values between species within the same feeding group, but lower between different feeding groups, especially between groups A (epifaunal predators) and D (benthopelagic predators). Therefore, each of the major fish species in this study feeds on different prey items based on differences in their mouth morphology, behavior (pelagic vs. benthic), feeding strategy, and foraging ability; consequently, they can coexist by utilizing different types of food resources in the same area. Such differences in resource utilization are common among different species inhabiting the same habitat (Kwak et al. 2004; Nanjo et al. 2008), even within the same family (e.g., Park et al. 2017a), and/or the same genus (e.g.,Hajisamae et al. 2006; Huh et al. 2016; Amariles et al. 2017), and can be important for maintaining ecosystem functions by minimizing competition for limited food resources and preventing the depletion of food resources (Krajewski et al. 2006; Adam et al. 2015; Park et al. 2018).
This study analyzed the dietary contents of eight abundant fish species inhabiting sites inside and outside of Jinju Bay. Except for T. kammalensis, all of the other studied species were benthic or semi-benthic predators, which commonly consumed carid shrimps, gammarid amphipods, polychaetes, and bentho-pelagic teleosts. However, the ratio of prey items was different in the diets of each species, and consequently, they could coexist in the same area by reducing competition for food resources. The feeding relationships among the eight dominant species in Jinju Bay were also evident according to the habitat and/or season. However, a lack of samples for identifying populations of prey organisms imposed limitations on our ability to describe direct prey–predator relationships in benthic ecosystems. The results from this study are important not only as a scientific basis for studying the trophic flow associated with the demersal food web in Jinju Bay, but also as basic data for further comparisons of the diets of the same species inhabiting different environments.
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author upon request.
References
Adam TC, Kelley M, Ruttenberg BI, Burkepile DE (2015) Resource partitioning along multiple niche axes drives functional diversity in parrotfishes on Caribbean coral reefs. Oecologia 179(4):1173–1185. https://doi.org/10.1007/s00442-015-3406-3
Amariles DF, Navia AF, Giraldo A (2017) Food resource partitioning of the Mustelus lunulatus and Mustelus henlei (Elasmobranchii: Carcharhiniformes). Environ Biol Fishes 100(6):717–732. https://doi.org/10.1007/s10641-017-0598-x
Anderson MJ, Gorley RN, Clarke KR (2008) PERMANOVA+ for PRIMER: guide to software and statistical methods. PRIMER-E Ltd., Plymouth, p 214
Baeck GW, Park JM, Huh SH, Kim HJ, Jeong JM (2014) Feeding habits of Kammal thryssa Thryssa kammalensis (Bleeker, 1849) in the coastal waters of Gadeok-do, Korea. Anim Cells Syst 18(2):154–159. https://doi.org/10.1080/19768354.2014.907206
Bizzarro JJ, Smith WD, Márquez-Farías JF, Tyminski J, Hueter RE (2009) Temporal variation in the artisanal elasmobranch fishery of Sonora, Mexico. Fish Res 97:103–117. https://doi.org/10.1016/j.fishres.2009.01.009
Bonato KO, Burress ED, Fialho CB (2017) Dietary differentiation in relation to mouth and tooth morphology of a neotropical characid fish community. Zool Anz 267:31–40. https://doi.org/10.1016/j.jcz.2017.01.003
Cha SS, Park KJ (1997) Seasonal changes in species composition of fishes collected with a bottom trawl in Kwangyang Bay, Korea. Korean J Ichthyol 9(2):235–243
Choo HS, Yoon EC (2015) Temporal and spatial variations of sea surface temperature in Jinju Bay in the South Coast of Korea. J Kor Soc Mar Environ Safe 21(4):315–326. https://doi.org/10.7837/kosomes.2015.21.4.315
Clarke KR, Gorley RN (2015) Getting started with PRIMER v7. PRIMER-E: Plymouth, Plymouth Marine Laboratory
Ferry LA, Cailliet GM (1996) Sample size and data analysis: are we characterizing and comparing diet properly? In: MacKinlay D, Shearer K (eds) Gutshop ’96: feeding ecology and nutrition in fish symposium proceedings. American Fisheries Society, Physiology Section, San Francisco, pp 71–80
Froese R, Pauly D (2022) FishBase. World Wide Web electronic publication. www.fishbase.org, version (02/2022)
Fugi R, Agostinho A, Hahn N (2001) Trophic morphology of five benthic-feeding fish species of a tropical floodplain. Rev Bras Biol 61(1):27–33
Gerking S (1994) Feeding ecology of fish. Academic Press Inc., San Diego, p 416
Hajisamae S, Chou LM (2003) Do shallow water habitats of an impacted coastal strait serve as nursery grounds for fish? Estuar Coast Shelf Sci 56(2):281–290. https://doi.org/10.1016/S0272-7714(02)00162-2
Hajisamae S, Yeesin P, Ibrahim S (2006) Feeding ecology of two sillaginid fishes and trophic interrelations with other co-existing species in the southern part of South China Sea. Environ Biol Fishes 76(2):167–176. https://doi.org/10.1007/s10641-006-9018-3
Hamano T, Morrissy NM, Matsuura S (1987) Ecological information on Oratosquilla oratoria (Stomatopoda, Crustacea) with an attempt to estimate the annual settlement date from growth parameters. J Shimonoseki Univ Fish 36(9):27
Hanamura Y, Matsuoka M (2003) Feeding habits of the Sand Shrimp, Crangon urital Hayashi & Kim, 1999, in the Central Seto Inland Sea, Japan. Crustaceana 76:1017–1024. https://doi.org/10.1007/s00435-015-0270-y
Houde ED, Rutherford ES (1993) Recent trends in estuarine fisheries: predictions of fish production and yield. Estuaries 16(2):161–176. https://doi.org/10.2307/1352488
Huh SH (1997) Feeding habits of snailfish, Liparis Tanakai. Korean J Ichthyol 9(1):71–78
Huh SH, Baeck GW (2000) Feeding habits of blotched eelpout, Zoarces gilli collected in the coastal water off Gadeok-do, Korea. Korean J Ichthyol 12(1):54–61
Huh SH, Choi HC, Park JM (2018) Feeding relationship between co-occurring silver croaker (Pennahia argentata) and Japanese sillago (Sillago japonica) in the Nakdong River estuary, Korea. Korean J Ichthyol 30(4):224–231
Huh SH, Kwak SN (1998) Feeding habits of Conger myriaster in the eelgrass (Zostera marina) bed in Kwangyang Bay. Korean J Fish Aquat Sci 31(5):665–672
Huh SH, Park JM, Baeck GW (2016) Diet partitioning between co-occurring Amblychaeturichthys hexanema and Amblychaeturichthys sciistius in the southeastern Korean waters. Korean J Ichthyol 28(2):79–86
Huh SH, Baeck GW (2003) Feeding habits of Repomucenus valencienei collected in the coastal waters of Gadeok-do of Korea. Korean J Ichthyol 15:289–294
Hyndes GA, Platell ME, Potter IC (1997) Relationships between diet and body size, mouth morphology, habitat and movements of six sillaginid species in coastal waters: implications for resource partitioning. Mar Biol 128(4):585–598. https://doi.org/10.1007/s002270050125
Hyslop E (1980) Stomach contents analysis-a review of methods and their application. J Fish Biol 17(4):411–429. https://doi.org/10.1111/j.1095-8649.1980.tb02775.x
Jeong JM, Park JM, Huh SH, Ye SJ, Kim HJ, Baeck GW (2013) Seasonal variation in the species composition of fish assemblages in the coastal waters off Gadeok-do, South sea, Korea. Korean J Fish Aquat Sci 46(6):948–956. https://doi.org/10.5657/KFAS.2013.0948
Jeon BS, Choi JH, Im YJ, Lee HW, Kim JW (2020) Maturity and spawning of white croaker Pennahia argentatus in the southern Sea of Korea. Korean J Fish Aquat Sci 53(1):36–42. https://doi.org/10.5657/KFAS.2020.0036
Jin X, Zhang B, Xue Y (2010) The response of the diets of four carnivorous fishes to variations in the Yellow Sea ecosystem. Deep Sea Res 2 Top Stud Oceanogr 57(11–12):996–1000
Kang CK, Baik MS, Kim JB, Lee PY (2002) Seasonal and spatial distribution of soft-bottom Polychaetesin Jinju Bay of the Southern Coast of Korea. Korean J Fish Aquat Sci 35(1):35–45. https://doi.org/10.5657/kfas.2002.35.1.035
Kang YS, Chae YK, Lee HR (2011) Variation of density stratification due to fresh water discharge in the Kwangyang Bay and Jinju Bay. J Korean Soc Coast Ocean Eng 23(1):126–137. https://doi.org/10.9765/KSCOE.2011.23.1.126
Kim YU, Park YS, Myoung JG (1986) Egg development and larvae of the snailfish, Liparis tanakai (Gilbert et Burke). Korean J Fish Aquat Sci 19(4):380–386
Kim CK, Lee JT, Jang HS (2010) Water circulation structure in the Chinju Bay of Korea. J Korean Soc Coast Ocean Eng 22(4):215–223
Kim IS, Choi Y, Lee CL, Lee YJ, Kim BJ, Kim JH (2005) Illustrated book of Korean fishes. Kyo-Hak Publishing Co., Seoul, p 615
Kim YH, Lee EH, Kim JN, Choi JH, Oh TY, Lee DW (2011) Age and growth of whitespotted conger Conger myriaster in the southern coastal waters of Korea. Korean J Fish Aquat Sci 44(6):689–694. https://doi.org/10.5657/KFAS.2011.0689
Kim MJ, Youn SH, Kim JY, Oh CW (2013) Feeding characteristics of the Japanese anchovy, Engraulis japonicus according to the distribution of zooplankton in the coastal waters of southern Korea. Korean J Environ Biol 31(4):275–287. https://doi.org/10.11626/KJEB.2013.31.4.275
Kim JK, Ryu JH, Kwun HJ, Ji HS, Park JH, Myoung SH, Song YS, Lee SJ, Yu HJ, Bae SE, Jang SH, Lee WJ (2019) Distribution map of sea fishes in Korean peninsula. Mapledesign, Busan, p 541
Kobayashi T, Hiyama S (1991) Distribution, abundance, and food habits of the snailfish Liparis tanakai in the Suo Sea, Seto Inland Sea. Japan J Ichthyol 38:207–210
Kodama K, Shimizu T, Yamakawa T, Aoki I (2004) Reproductive biology of the female Japanese mantis shrimp Oratosquilla oratoria (Stomatopoda) in relation to changes in the seasonal pattern of larval occurrence in Tokyo Bay. Japan Fish Sci 70(5):734–745. https://doi.org/10.1111/j.1444-2906.2004.00866.x
Koh EH, An YS, Baeck GW, Jang CS (2014) Feeding habits of white croaker, Pennahia argentata in the coastal waters off Sejon island, Korea. J Kor Soc Fish Tech 50(2):139–146. https://doi.org/10.3796/KSFT.2014.50.2.139
Kosaka M (1971) On the ecological niche of the seasnails, Liparis tanakai in Sendai Bay. J Coll Mar Sci Tech Tokai Univ 5:27–41
Krajewski JP, Bonaldo RM, Sazima C, Sazima I (2006) Foraging activity and behaviour of two goatfish species (Perciformes: Mullidae) at Fernando de Noronha Archipelago, tropical West Atlantic. Environ Biol Fishes 77(1):1–8. https://doi.org/10.1007/s10641-006-9046-z
Kwak SN, Huh SH, Kim HW (2012) Change in fish assemblage inhabiting around Dae Island in Gwangyang Bay, Korea. J Kor Soc Mar Environ Safe 18(3):175–184. https://doi.org/10.7837/kosomes.2012.18.3.175
Kwak SN, Klumpp DW, Park JM (2015) Feeding relationships among juveniles of abundant fish species inhabiting tropical seagrass beds in Cockle Bay, North Queensland, Australia. N Z J Mar Freshwater Res 49(2):205–223. https://doi.org/10.1080/00288330.2014.990467
Kwak SN, Huh SH (2003) Changes in species composition of fishes in the Nakdong River Estuary. Korean J Fish Aquat Sci 36(2):129–135. https://doi.org/10.5657/kfas.2003.36.2.129
Kwak SN, Huh SH, Klumpp DW (2004) Partitioning of food resources among Sillago japonica, Ditremma temmincki, Tridentiger trigonocephalus, Hippocampus japonicus and Petroscirtes breviceps in an eelgrass, Zostera marina, bed. Environ Biol Fishes 71(4):353–364. https://doi.org/10.1007/s10641-004-1259-4
Langton RW (1982) Diet overlap between Atlantic cod, Gadus morhua, silver hake Merluccius bilinearis and fifteen other northwest Atlantic finfish. Fish B-NOAA 80:745–759
Lee CR, Kang HK, Noh JH (2009) Temporal and spatial variation of zooplankton community structure post construction of Saemangeum dyke. Ocean Polar Res 31(4):327–338. https://doi.org/10.4217/OPR.2009.31.4.327
Lee SK, Seo YI, Kim JI, Kim HY, Choi MS (2011) Seasonal species composition and fluctuation of fishes by beam trawl in Yeoja Bay. Korean J Ichthyol 23(3):206–216
Lefcheck JS, Hughes BB, Johnson AJ, Pfirrmann BW, Rasher DB, Smyth AR, Williams BL, Beck MW, Orth RJ (2019) Are coastal habitats important nurseries? A meta-analysis. Conserv Lett 12(4):e12645. https://doi.org/10.1111/conl.12645
Maher I, Song KJ, Park HM, Oh CW (2013) Feeding ecology of the sand shrimp Crangon hakodatei Rathbun, 1902 (Decapoda: Crangonidae) in the East Sea of Korea. Anim Cells Syst 17(1):44–52. https://doi.org/10.1080/19768354.2013.769897
Marshall AD, Kyne PM, Bennett MB (2008) Comparing the diet of two sympatric urolophid elasmobranchs (Trygonoptera testacea Müller & Henle and Urolophus kapalensis Yearsley & Last): evidence of ontogenetic shifts and possible resource partitioning. J Fish Biol 72(4):883–898. https://doi.org/10.1111/j.1095-8649.2007.01762.x
Motta PJ, Clifton KB, Hernandez P, Eggold BT, Giordano SD, Wilcox R (1995) Feeding relationships among nine species of seagrass fishes of Tampa Bay, Florida. Bull Mar Sci 56(1):185–200
Myoung SH, Ban TW, Kim JK (2016) Population structure of Liparis tanakae (PISCES, Liparidae) from Korea based on morphological and molecular traits. Korean J Fish Aquat Sci 49(2):198–207. https://doi.org/10.5657/KFAS.2016.0198
Myoung SH, Kwak SN, Kim JK, Lee WC, Kim JB, Kim HC, Williamson JE (2020) Juvenile fish assemblages in the Jinju Bay region, Korea. Fish Aquatic Sci 23(1):1–16. https://doi.org/10.1186/s41240-020-00175-6
Myoung SH, Kwak SN, Kim JK, Williamson JE (2021) Effect of freshwater discharge from Namgang Dam on ichthyoplankton assemblage structure in Jinju Bay, Korea. Aquat Living Resour 34:18. https://doi.org/10.1051/alr/2021017
Nakabo T (2013) Fishes of Japan with pictorial keys to the species. Hadano, Tokai University Press, I: xlix+ 1–864 p; II: xxxii+ 865–1747 p; III: xvi+ 1751–2428 p
Nanjo K, Kohno H, Sano M (2008) Food habits of fishes in the mangrove estuary of Urauchi River, Iriomote Island, southern Japan. Fish Sci 74(5):1024–1033. https://doi.org/10.1111/j.1444-2906.2008.01620.x
Nelson JS, Grande TC, Wilson MV (2016) Fishes of the World, 5th edn. John Wiley and Sons Inc., New Jersey, p 386
Newton A, Icely J, Cristina S, Brito A, Cardoso AC, Colijn F, Dalla- Riva S, Gertz F, Hansen J, Holmer M, Ivanova K, Leppäkoski E, Mocenni C, Mudge S, Murray N, Pejrup M, Razinkovas A, Reizopoulou S, Pérez-Ruzafa A, Schernewski G, Schubert H, Seeram L, Solidoro C, Viaroli P, Zaldívar JM (2014) An overview of ecological status, vulnerability and future perspectives of European large shallow, semi-enclosed coastal systems, lagoons and transitional waters. Estuar Coast Shelf Sci 140:95–122. https://doi.org/10.1016/j.ecss.2013.05.023
Oksanen J, Simpson GL, Blanchet FG, Kindt R, Legendre P, Minchin PR, O’Hara RB, Solymos P, Stevens MHH, Szoecs E, Wagner H, Barbour M, Bedward M, Bolker B, Borcard D, Carvalho G, Chirico M, De Caceres M, Durand S, Evangelista HBA, FitzJohn R, Friendly M, Furneaux B, Hannigan G, Hill MO, Lahti L, McGlinn D, Ouellette M-H, Cunha ER, Smith T, Stier A, Braak CT, Weedon J (2022) Vegan: community ecology package. r package version 2.6–2. Available at: https://CRAN.R-project.org/package=vegan
Park JM (2010) Species composition and reproductive ecology of fishes in the coastal waters off Gori, Korea (Doctoral dissertation, Ph. D Thesis, Pukyoung Natl Univ, Busan, Korea, 236)
Park JM, Coburn E, Platell ME, Gaston TF, Taylor MD, Williamson JE (2017a) Diets and resource partitioning among three sympatric gurnards in northeastern Tasmanian waters, Australia. Mar Coast Fish 9(1):305–319. https://doi.org/10.1080/19425120.2017.1320342
Park JM, Kwak SN (2018) Seagrass fish assemblages in the Namhae Island, Korea: the influences of seagrass vegetation and biomass. J Sea Res 139:41–49. https://doi.org/10.1016/j.seares.2018.06.007
Park JM, Kwak SN, Han IS (2018) Feeding relationships among six seagrass-associated fishes in the Northeastern Gwangyang Bay, Southern Korea. Ocean Sci J 53(1):63–72. https://doi.org/10.1007/s12601-017-0060-1
Park JM, Kwak SN, Huh SH, Han IS (2017) Diets and niche overlap among nine co-occurring demersal fishes in the southern continental shelf of East/Japan Sea, Korea. Deep Sea Res 2 Top Stud Oceanogr 143:100–109. https://doi.org/10.1016/j.dsr2.2017.06.002
Park JM, Kwak SN, Lee WC (2020a) Dietary study using set-nets produces bias in prey choice of fish: a case of three coastal fishes inhabiting southern Korean waters. J Sea Res 157:101846. https://doi.org/10.1016/j.seares.2020.101846
Park JM, Riedel R, Ju HH, Choi HC (2020b) Fish assemblage structure comparison between freshwater and estuarine habitats in the lower Nakdong River, South Korea. J Mar Sci Eng 8(7):496. https://doi.org/10.3390/jmse8070496
Persson L, Diehl S (1990) Mechanistic individual-based approaches in the population/community ecology of fish. Ann Zool Fenn 27:165–182
Platell ME, Potter IC (2001) Partitioning of food resources amongst 18 abundant benthic carnivorous fish species in marine waters on the lower west coast of Australia. J Exp Mar Biol Ecol 261(1):31–54. https://doi.org/10.1016/S0022-0981(01)00257-X
R Core Team (2022) R: A language and environment for statistical computing (Vienna, Austria: R Foundation for Statistical Computing). Available at: https://www.R-project.org/
Ryu KY, Shim SL, Kim W, Jung MS, Hwang IM, Kim JH, Hong CH, Jung CH, Kim KS (2009) Analysis of the seasonal change of the proximate composition and taste components in the conger eels (Conger myriaster). J Korean Soc Food Sci Nutr 38(8):1069–1075. https://doi.org/10.3746/jkfn.2009.38.8.1069
Schoener TW (1970) Nonsynchronous spatial overlap of lizards in patchy habitats. Ecology 51(3):408–418. https://doi.org/10.2307/1935376
Selleslagh J, Amara R, Laffargue P, Lesourd S, Lepage M, Girardin M (2009) Fish composition and assemblage structure in three Eastern English Channel macrotidal estuaries: a comparison with other French estuaries. Estuar Coast Shelf Sci 81(2):149–159. https://doi.org/10.1016/j.ecss.2008.10.008
Song MY, Kim JI, Kim ST, Lee JH, Lee JB (2012) Seasonal variation in species composition of catch by a coastal beam trawl in Jinhae Bay and Jinju Bay, Korea. J Kor Soc Fish Tech 48(4):428–444. https://doi.org/10.3796/KSFT.2012.48.4.428
Song SH, Jeong JM, Lee SH, Kim DH (2019) Species composition and community structure of fish by shrimp beam trawl between Sacheon Bay and coastal waters off Namhae, Korea. J Kor Soc Fish Tech 55(3):217–232. https://doi.org/10.3796/KSFOT.2019.55.3.217
Swearer SE, Caselle JE, Lea DW, Warner RR (1999) Larval retention and recruitment in an island population of a coral-reef fish. Nature 402(6763):799–802. https://doi.org/10.1038/45533
Upchurch S, Wenner E (2008) Fish and decapod crustacean assemblages from the Ashepoo-Combahee-Edisto Basin, South Carolina (1993–1999). J Coast Res 10055:200–213. https://doi.org/10.2112/SI55-017.1
Vasconcelos RP, Reis-Santos P, Maia A, Fonseca V, França S, Wouters N, Costa MJ, Cabral HN (2010) Nursery use patterns of commercially important marine fish species in estuarine systems along the Portuguese coast. Estuar Coast Shelf Sci 86(4):613–624. https://doi.org/10.1016/j.ecss.2009.11.029
Wallace RK (1981) An assessment of diet-overlap indexes. T Am Fish Soc 110:72–76
White W, Platell M, Potter I (2004) Comparisons between the diets of four abundant species of elasmobranchs in a subtropical embayment: implications for resource partitioning. Mar Biol 144(3):439–448. https://doi.org/10.1007/s00227-003-1218-1
Yoo JM, Lee EK, Kim S (1999) Distribution of ichthyoplankton in the adjacent waters of Yousu. Korean J Fish Aquat Sci 32(3):295–302
Yoon SC, Jeong YK, Zhang CI, Yang JH, Choi KH, Lee DW (2014) Characteristics of Korean coastal fisheries. Korean J Fish Aquat Sci 47(6):1037–1054. https://doi.org/10.5657/KFAS.2014.1037
Youn BI, Lee SJ, Sohn MH, Han Sh, Lee HB, Mj K, Han KH (2019) Distribution of Ichthyoplankton in the West coastal waters of Korea. J Kor Soc Fish Tech 55(3):243–251. https://doi.org/10.3796/KSFOT.2019.55.3.243
Zhang CI, Lee SI, Kim JM (2003) Ecosystem-based management of fisheries resources in marine ranching areas. J Korean Soc Fish Res 6:71–83
Acknowledgements
We are grateful to Mr. Ho Ik Choi (local fisher) for his assistance with the field sampling. We would also like to thank all members of the Ichthyological Laboratory at Pukyong National University for their assistance with sampling and laboratory processes.
Funding
This research was funded by the Korea Institute of Ocean Science and Technology (grant numbers PEA0013 and PEA0016).
Author information
Authors and Affiliations
Contributions
SHM, JKK, SNK, and JMP conceived the study. SHM and SNK contributed to sample preparation for stomach content analysis. JMP reviewed and revised the manuscript. JKK supervised the study.
Corresponding author
Ethics declarations
Ethic approval
Not applicable for that section.
Consent to participate
All authors have agreed to participate.
Consent for publication
All authors have read and agreed to the published version of the manuscript.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Myoung, S.H., Kim, JK., Kwak, S.N. et al. Variations in food resource usage among eight fish species inhabiting inside and outside Jinju Bay, Korea. Environ Biol Fish 105, 1219–1235 (2022). https://doi.org/10.1007/s10641-022-01341-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-022-01341-6