Abstract
For an introduced species to expand its range, individuals must be able to adapt to novel stressors (biotic and abiotic) that they may encounter. Florida has a distinct temperature gradient and rapid climate zone transitions from warmer subtropical regions to cooler temperate regions. Mean minimum temperatures are thought to prohibit northward movement of introduced fish species in south Florida. As the introduced Belonesox belizanus (pike killifish) expands its introduced range northward, it may be gaining cold tolerance. This study sought to compare thermal tolerances and cessation of feeding between adult B. belizanus from southern (initial, introduced population) and northern (spreading, fringe edge population) populations from its introduced range. To compare thermal tolerances between locations, southern individuals and northern individuals were acclimated to three temperatures (20, 25, and 30 °C) and their upper and lower lethal temperatures and cessation of feeding temperatures were measured. Two-way ANOVAs revealed that lower lethal temperatures and cessation of feeding temperatures were significantly different between northern and southern populations, with northern individuals having a lower lethal temperature approximately 0.5 °C lower and a cessation of feeding temperature approximately 2 °C lower compared with southern individuals. Directional selection may be influencing this non-native species’ thermal tolerance and feeding ability benefitting their continued effort to disperse from subtropical to temperate environments within Florida. This study indicates that B. belizanus may be gaining cold tolerance as it continues to spread northward in its introduced range.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Through the process of invasion, non-native fish populations are subjected to various ecological constraints in the novel environment, and their ability to survive depends on their ability to overcome these constraints (Stauffer 1984, Taylor et al. 1984, Sakai et al. 2001, Lockwood et al. 2007). For an introduced species to successfully spread and maintain physiological homeostasis, it must have adaptations for biotic and abiotic stressors to cope in its immediate heterogeneous environment. Biotic (e.g., inter- and intraspecific competition, parasitism, and predation) and abiotic (e.g., temperature and habitat connectivity) factors influencing the number of propagules, mode of dispersal, and mortality are critical factors regulating the subsequent spread and dispersal of non-native species from the original point of introduction (Sakai et al. 2001).
The establishment of an non-native species population involves a three-stage process, with each stage requiring the introduced species to overcome several ecological barriers (Lockwood et al. 2007). The first stage occurs when the introduced species survives the initial transport and adjusts to potentially novel environmental regimes, such as variable dissolved oxygen, salinity, and temperature combinations and potential novel biotic pressures, including predation and competition for limited resources. The second stage involves establishment where the introduced colony survives long enough to create a persistent, self-sustaining population in their novel range (Marchetti et al. 2004, Lockwood et al. 2007). The third stage occurs when the introduced population increases in abundance and expands its range. To expand its range, the introduced species must have the capability to endure novel heterogenous habitats surrounding its present range, otherwise populations are limited and localized (Marchetti et al. 2004, Carveth et al. 2006, Capps et al. 2011).
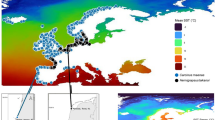
Temperature tolerance studies seek to investigate the thermal limits within which a species can survive and potentially expand their range in thermally variable environments. Florida has a distinct temperature gradient and rapid climate zone transitions (Beaver et al. 1981, Sanford 2003), from warmer subtropical regions in the south to cooler temperate regions of the north. Mean minimum temperatures are thought to prohibit south-north movement of non-native fish species in south Florida (Shafland and Pestrak 1982, Trexler et al. 2005) (Fig. 1). Studies have suggested that physiological and behavioral adaptations to variable thermal regimes (e.g., thermal plasticity or the use of thermal refugia) can occur on localized scales in some fishes that may aid in subtle range expansion over time (i.e., cichlid species (Cichlasoma urophthalmus and Hemichromis letourneuxi) (Schofield et al. 2010), sockeye salmon (Oncorhynchus nerka) (Eliason et al. 2011), and common killifish (Fundulus heteroclitus) (Healy and Schulte 2012)).
Isotherms (following Sanford 2003) depicting estimated mean minimum January temperatures (°C) in Florida (created by R. Maliao). The inset represents the Belonesox belizanus collection sites in south Florida. Individuals representing the southern location were collected along Highway 9336 that runs through Everglades National Park and are labeled by open circles. Individuals representing the northern location were collected from canals along Interstate 75 and SR 839 that run through the northern portion of Big Cypress National Preserve labeled by open triangles
In 1957, the non-indigenous pike killifish, Belonesox belizanus Kner, was released into a Miami-Dade County, Florida canal (Belshe 1961). Belonesox belizanus persisted as a small population in several canals east of the Everglades for more than 20 years after it was introduced and its geographical range expanded in the 1980 and 1990 s to the Everglades National Park (Courtenay 1997, Trexler et al. 2000). From its original introduction, B. belizanus has spread and established populations throughout Miami-Dade, Collier, Broward, and Monroe counties including Big Cypress and Everglades National Parks, an area approximately 10,000 km2 (Belshe 1961, Dunker 2003, Loftus et al. 2004). A previous study indicated that the magnitude of change in density of introduced B. belizanus varies inversely with the magnitude of change in temperature in south Florida populations (Kerfoot et al. 2011). Another study highlighted the variability in temperature tolerance throughout ontogeny for this species, with the juvenile size class having the largest thermal tolerance polygonal area and a 2–4 °C lower minimum lethal thermal limit compared with adults or neonates (Kerfoot 2012). This disparate influence of temperature on B. belizanus life history tolerances underscores the need to determine if there exists differences in thermal tolerances in populations of adult B. belizanus from the northern and southern reaches of their introduced range. As B. belizanus has continued to spread to northern locations in Florida (and to lower minimum temperatures, Fig. 1), has it acclimated to a lower thermal environment? The specific goal of this study was to determine whether the temperature tolerance limits and cessation of feeding temperatures of adult B. belizanus differ between the northern and southern locations of its south Florida distribution. The specific hypotheses tested in this study is that there are significant differences in thermal tolerance ranges and temperatures at which individuals cease to feed between northern and southern populations of introduced B. belizanus.
Materials and methods
Specimen collection
Belonesox belizanus adults (standard length [SL] > 55 mm) (Turner & Snelson 1984) were collected using a 9.5 mm mesh seine, dip nets and cast nets from the Everglades National Park (southern location) along Highway 9336 that runs through the park. Fish were transported live to the Florida Institute of Technology’s Fish Ecophysiology lab. In separate collections approximately 100 km from the southern location, adults were collected live from northern locations of Big Cypress National Preserve (northern location) along canals lining Interstate 75 and SR 839. Collections of B. belizanus in the northern and southern locations occurred during the dry season (January – May) in 2008 and 2009 (Fig. 1, inset). The estimates of minimum January temperature in Fig. 1 are based on mean air temperatures but can be used as approximations of surface water temperatures. Northern locations fall between the 12.2–13.3 °C isotherms, whereas southern locations are bound by 13.3–15.6 °C isotherms (Fig. 1).
All specimens were housed in the laboratory in 90.9 L aquaria at 25 °C for two weeks prior to the start of the experiments to acclimate the fish to laboratory conditions. Individuals were fed ad libitum western mosquitofish, Gambusia affinis. Mosquitofish were collected from retention ponds on Florida Institute of Technology property. Tanks were maintained and cleaned daily to remove uneaten prey and waste material.
Experimental design
Acclimation temperatures
To calculate upper and lower thermal tolerance ranges for adults of this species, individuals from each location were randomly assigned to one of three different acclimation temperatures: 20, 25 or 30 °C. These acclimation temperatures were chosen based on data of the average seasonal temperature fluctuations in south Florida (Kerfoot 2012, South Florida Water Management District database). Fish were acclimated to these temperatures for two weeks. During the acclimation period, fish were fed daily ad libitum and their tanks cleaned daily to remove waste material.
Lethal maximum and minimum temperatures
Lethal maximum (Lmax – upper tolerance, higher temperature threshold) and lethal minimum (Lmin – lower tolerance, lower temperature threshold) were used to assess the upper and lower limits of B. belizanus thermal tolerance between locations. Lmax and Lmin are the two dynamic approaches commonly used to evaluate tolerance limits and are defined as the thermal endpoints where death occurs (Hickman and Dewey 1973, Shafland and Pestrak 1982, Beitinger et al. 2000). For trials, three 145 cm x 29 cm x 29 cm acrylic tanks were divided into five equal, smaller compartments, yielding fifteen 24.4 L tanks. These tanks were set up in a re-circulating flow-through system where water was pumped into each tank from a common sump. To control thermal levels in the experimental tanks, two 500 W titanium heaters were housed in the sump and an external Delta Star Chiller (1/4 hp, 115 V, 5.4 amp, 3080 BTU, 8/15gpm) was plumbed into the re-circulating system. A programmable thermal regime was designed to regulate the chiller and heater systems. The Automatically Regulated Thermal Unit Regime (ARThUR) was developed through a collaboration between A. Kunkle (Florida Institute of Technology’s Department of Computer Science) and J. Kerfoot that allowed maintenance of precise thermal gradients throughout the experiment (modified from Schaefer 2006, precise components used are outlined in Kerfoot 2012). ARThUR was programmed to change the temperature in the experimental tanks 1.0 °C day− 1 starting at 0900 h each day, and to monitor and log temperature in the tanks every 30 min (Fig. 2).
Graph depicting temperature change in experimental tanks over time, controlled by ARThUR during a preliminary Lmin treatment. The experimental design required temperature to change at a rate of 1.0 °C day− 1. Regression analysis indicated that ARThUR maintained 99.5% accuracy in regulating a 1.0 °C day− 1 temperature change in the experimental system
Individual fish were randomly assigned to an experimental tank at their appropriate acclimation temperature. To ensure that deaths of individuals were attributed only to temperature and not handling stress due to movement to experimental tanks, individuals remained in the experimental tank for a 24-hour period and were monitored. After the 24-hour period, temperature was increased (for Lmax trials) or decreased (for Lmin trials) at a rate of 1.0 °C day− 1 until death occurred (following Shafland and Pestrak 1982). The response variables measured were temperatures at which death and cessation of feeding occurred. The cessation of feeding temperature was defined as the first temperature observed where the individual did not feed over a 48-hour period. Once the lethal limit was reached, fish were removed from the experimental tank. To avoid any confounding effects of a pseudo-replicated design, Lmin and Lmax trials were staggered, and acclimation temperatures were randomly assigned.
To investigate thermal tolerance and cessation of feeding temperatures between locations, 30 individuals from the southern location and 27 adult individuals from the northern location were used to calculate tolerance ranges (Table 1). To control for sexual dimorphism in adult B. belizanus, Lmax and Lmin values were analyzed using separate Mann-Whitney non-parametric tests with sex as the independent variable and Lmin and Lmax as the dependent variables. Results indicated that there were no significant differences in Lmin or Lmax measurements between sexes which allowed sexes to be grouped together and used in subsequent analyses (Southern adults: Lmin – U = 18.5, N = 15, P = 0.681, Lmax – U = 11.5, N = 15, P = 0.067; Northern adults: Lmin – U = 14.0, N = 13, P = 0.278, Lmax – U = 12.0, N = 13, P = 0.135).
Statistical analyses
To test the hypothesis that there are significant differences in thermal tolerance ranges and cessation of feeding temperatures between locations of adult B. belizanus, separate two-way factorial Analyses of Variance (ANOVAs) were used. Location and acclimation temperature were used as independent factors and Lmin and Lmax were used as dependent variables. Neither of the dependent variables met the assumptions of parametric tests and were rank-transformed following Quinn and Keough (2002). The rank-transformation method is an acceptable technique and can aid in increasing the robustness of factorial ANOVA designs involving non-parametric data (Quinn and Keough 2002). A separate two-way ANOVA was performed on rank-transformed cessation of feeding temperature measurements using location and acclimation temperature as independent factors and the temperature at which individuals ceased to feed as the dependent variable. If acclimation temperature effect was significant in any two-way ANOVA, a univariate Tukey’s multiple comparisons test was run to investigate which acclimation temperature was significantly different for each dependent variable. To control for possible differences in adult size between locations, an independent t-test was performed on standard lengths of adult individuals from southern and northern locations to test for significant differences in size between locations. No significant difference in average standard length between locations was observed (t = 0.129, df = 55, P = 0.898).
Finally, to represent the thermal tolerance range of adults at each location, thermal tolerance polygons were developed and measured using average lethal temperatures (Lmin and Lmax) across acclimation temperatures. These average lethal temperatures formed lower and upper bounds of the thermal tolerance polygons. Estimates of thermal tolerance polygons are formed by the area which the polygon covers and are limited by the lowest and highest acclimation temperatures. Thermal tolerance polygonal areas are given in units of °C2.
All statistical analyses were performed in SPSS 17.0 and Sigma Plot 2001 at an α value of 0.05.
Results
There were similarities in the upper thermal tolerances of northern and southern populations of adult B. belizanus (Fig. 3; Table 1). Lmax limits were not different between locations at any acclimation temperature (Table 2). However, analyses indicated that lower lethal tolerances (Lmin) were significantly different between locations and between acclimation temperatures (location: F = 5.137, df = 1, P = 0.034; acclimation temperature: F = 11.346, df = 2, P < 0.001; Fig. 3; Table 2). Individuals from northern locations had lower thermal limits across acclimation temperatures compared to individuals from southern locations, especially at an acclimation temperature of 25 °C (Table 1). At an acclimation temperature of 25 °C, individuals from northern locations had Lmin values of 9.6 ± 0.54 °C and were significantly different from the Lmin values for adults from southern locations, which had slightly higher limits of 10.0 ± 0.00 °C (Table 1). Results of a Tukey’s multiple comparison test investigating the differences in Lmin limits between acclimation temperatures, indicated that Lmin measurements were significantly higher for the 30 °C acclimation temperature compared with the other two acclimation temperatures (20 vs. 25 °C: P = 0.329, 25 vs. 30 °C: P = 0.007, 30 vs. 20 °C: P < 0.001). Neither of the interaction terms between location and acclimation temperature were significant for Lmax or Lmin measures, indicating that the significant differences in Lmin between locations can be explained without evoking acclimation temperature and vice versa (Table 1).
Mean upper and lower lethal temperature tolerances (Lmax and Lmin) of B. belizanus between locations and across acclimation temperatures. Adults from the northern location are represented by closed circles (●) and those from southern locations are represented by open circles (○). Data are given in Mean ± S.d. Solid lines represent the thermal tolerance polygon for southern individuals and dashed lines represent the thermal tolerance polygons for northern individuals
The thermal tolerance polygonal areas were similar between locations with adults from southern locations having a polygonal area of 266.08°C2 and those from northern locations having an area of 268.86°C2 (Fig. 3). The similarities in thermal tolerance polygonal areas show no difference between overall tolerance ranges between individuals from different locations; however, these tolerance ranges may have shifted to include lower temperatures for northern adults (Fig. 3).
Individuals from northern locations ceased to feed at significantly lower temperatures compared to individuals from southern locations (F = 6.624, df = 1, P = 0.017; Fig. 4; Table 3). Across acclimation temperatures, mean cessation of feeding temperatures ranged from 13.0 to 14.0 °C for northern adults and 14.4–16.0 °C for southern adults. This indicated that northern individuals ceased feeding approximately 2 °C lower compared to southern individuals. Analyses also indicated that there was no significant effect of acclimation temperature on temperature at cessation of feeding, and there was no significant interaction between location and acclimation temperature (Table 3).
Mean cessation of feeding temperatures for adults from northern and southern locations across acclimation temperatures during Lmin trials. Individuals from southern locations are represented by open circles (○) and individuals from northern locations are represented by closed circles (●). Data are given in Mean ± S.d
Discussion
Comparison of thermal tolerances between adult B. belizanus from northern and southern fringe populations indicated no difference in lethal maximum (Lmax) temperatures. This may be a result of the species’ ability to gain heat tolerance more readily than cold tolerance and a very distinct upper temperature limit needed for basic physiological functioning. There was a significant difference in lower lethal temperatures between locations, with individuals from the northern location having lower Lmin limits compared to individuals from the southern location. This may indicate that B. belizanus at the northern edge of the distribution are becoming more cold-tolerant as they move northward from south Florida. The significant differences between locations in their thermal tolerances are on a scale of 0.4–0.6 °C, which might not be biologically meaningful. However, results here correspond to values presented in other poecilid thermal tolerance studies on Gambusia affinis from the Quashnet River population (Cape Cod, MA) showing lower thermal tolerance differences between males (Lmin = 0.6 °C) and females (Lmin = 0.2 °C) (Al-Habbib and Yacoob 1993, Wood et al. 2020). Interestingly, in the case of the slight difference in tolerance between male and female mosquitofish, this shorter-lived species may escape the cold by employing seed banks that persist through winter, especially in Poeciliids (Gambusia spp. and B. belizanus) where females are known to store sperm after mating (Turner and Snelson 1984, Robbins et al. 1987) allowing for regeneration of the population once favorable environmental conditions return (Podrabsky et al. 2015, Wood et al. 2020). Directional selection can work on small differences in thermal tolerances if they are heritable and increase reproductive success of individuals possessing those beneficial variations (lower Lmin and cessation of feeding temperatures) (Feminella and Matthews 1984). Studies have indicated that the ecology of offspring may be influenced by environmental conditions experienced by the parents, including their thermal adaptive ability to changing environments (Jonsson and Jonsson 2014, Lighten et al. 2016, Jonsson and Jonsson 2019). Genetic evolution may be the explanation for the extreme cold tolerance of the Quashnet mosquitofish due to being isolated for more than two decades (Wood et al. 2020) and may be applicable to this current study. The influence of an environmental stress on an organism is typically similar for both the individuals experiencing the stress and their offspring (Bonduriansky et al. 2012, Jonsson and Jonsson 2014). As B. belizanus continues to establish northward it is conceivable that offspring will continue to have lower thermal tolerances until temperature’s effect on physiology becomes limiting. A previous study hypothesizes that juvenile B. belizanus might be the dispersal stage due to their lower thermal tolerance compared with other life-history stages from the southern population (Kerfoot 2012). Other studies have shown that small changes (i.e., seasonal or diel shifts) in water temperature can have marked effects on fish kinematic performance (Özbilgin and Wardle 2002 [escape behavior], Allan et al. 2015 [predator-prey interactions]). Even though thermal tolerance ranges were similar between locations, there was a slight shift in these to lower thermal limits for northern adults, which may add credence to this species’ ability to gain cold tolerance as it moves northward. Interestingly, a population of B. belizanus was found in 1997 near Tampa Bay, Florida, approximately 181 km north of the northern fringe edge population in south Florida. The Tampa Bay population was not used in this study because individuals are thought to be escapees from a nearby ornamental fish farm and not from the original source population of those released in 1957 (Greenwood 2012).
In addition, the significant differences in cessation of feeding temperatures between locations across acclimation temperatures may demonstrate a behavioral/physiological adaptation to encountering lower thermal environments. Adult B. belizanus from northern fringe-edge populations ceased to feed an average of 2 °C lower across acclimation temperatures compared with their southern counterparts. This 2 °C shift in cessation of feeding follows the approximate 2 °C difference between locations based on estimated mean minimum January temperatures shown in Fig. 1. The ability of northern populations to gain lower thermal tolerance and feed at lower temperatures may provide a physiological mechanism allowing northern adults to feed and remain in lower temperature habitats, continuing to disperse northward. Over longer time scales, seasonal changes in temperature can modify thermal preferenda and elicit a variety of physiological performance responses at low temperatures (Johnson and Bennett 1995, Johnston and Temple 2002). It is believed that genetic evolution in acclimation responses (whether they be thermal plasticity, directional selection, or selection for plasticity) are adaptive and enhance the fitness of an organism in its new environment (Johnston and Temple 2002), which would be beneficial for the continued dispersal of a non-native species in Florida. While this study did not demonstrate a direct link between temperature and thermal plasticity of this species, results provide evidence of thermal plasticity as a potential mechanism aiding in continual dispersal of this species. Based on these results, I fail to reject the hypothesis that there are significant differences in thermal tolerance ranges and cessation of feeding temperatures of adult B. belizanus between northern and southern locations.
Implications for continued range expansion of Belonesox belizanus in south Florida
This study has demonstrated, indirectly, that physiological adaptation to lower thermal environments may be occurring in the introduced B. belizanus. Adults from the northern fringe-edge had lower thermal tolerances and ceased to feed at lower temperatures, which would bestow an advantage to those individuals in adapting to a lower thermal regime. Similarly, other studies have shown intraspecific differences in thermal tolerance measures such as variability in heat shock protein expression, critical thermal maxima, and short-term growth at relatively localized scales within small watersheds (Strange et al. 2002, Stitt et al. 2014). The ability to successfully feed at certain temperatures usurps any results of thermal tolerance limits when investigating the potential spread of a species into surrounding environments, making cessation of feeding temperatures important for predicting potential spread of a species. If the species cannot successfully utilize the available prey resources, then it cannot successfully invade new territories. Populations where directional selection for lower Lmin and cessation of feeding temperatures are occurring, or in those that have the ability for thermal plasticity, would benefit most in their effort to disperse from subtropical to temperate environments within Florida. With the challenge of global climate change, increasing temperature will promote the increased establishment and subsequent spread of tropical ornamental fish from the fish trade and aquaculture (Wolter and Röhr 2010). For non-native species there are likely many physiological and behavioral trade-offs that have allowed them to maintain function over a wide range of situations, allowing them to continue to add to their range, while not performing any of the functions optimally (Brett 1956, Randall and Brauner 1991). It is uncertain how temperature directly influences feeding in the introduced B. belizanus. Its ability to efficiently feed across temperatures may primarily dictate the final distribution of this species within its novel range in Florida.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Allan BJM, Domenici P, Munday PL, McCormick MI (2015) Feeling the heat: the effect of acute temperature changes on predator-prey interactions in coral reef fish. Conserv Physiol 3:cov011–cov011. https://doi.org/10.1093/conphys/cov011
Al-Habbib OAM, Yacoob MP (1993) Effects of acclimation and experience to changing heat and cold shock temperature on lethal temperature and thermal tolerance of Gambusia affinis (Baird & Girard) (Poeciliidae). Cybium 17:265–272
Beaver JR, Crisman TL, Bays JS (1981) Thermal regimes of Florida Lakes. Hydrobiologia 83:267–273. https://doi.org/10.1007/BF00008277
Beitinger TL, Bennett WA, McCauley RW (2000) Temperature tolerances of North American freshwater fishes exposed to dynamic changes in temperature. Environ Biol Fish 58:237–275. https://doi.org/10.1023/A:1007676325825
Belshe JF (1961) Observations on an introduced tropical fish, Belonesox belizanus, in southern Florida. Master’s Thesis, University of Miami
Bonduriansky R, Crean AJ, Day T (2012) The implications of nongenetic inheritance for evolution in changing environments. Evolutionary Applications 5:192–201. https://doi.org/10.1111/j.1752-4571.2011.00213.x
Brett JR (1956) Some principles in the thermal requirements of fishes. Q Rev Biol 31:75–87. https://doi.org/10.1086/401257
Capps KA, Nico LG, Mendoza-Carranza M, Arévalo-Frías W, Ropicki AJ, Heilpern SA, Rodiles-Hernández R (2011) Salinity tolerance of non-native suckermouth armoured catfish (Loricariidae: Pterygoplichthys) in south-eastern Mexico: implications for invasion and dispersal. Aquat Conserv 21:528–540. https://doi.org/10.1002/aqc.1210
Carveth CJ, Widmer AM, Bonar SA (2006) Comparison of upper thermal tolerances of native and nonnative fish species in Arizona. Trans Am Fish Soc 135:1433–1440. https://doi.org/10.1577/T05-025.1
Courtenay WR Jr (1997) Nonindigenous fishes. In: Simberloff DS, Schmitz DC, Brown TC (eds) Strangers in Paradise. Island Press, Washington DC, pp 109–122
Dunker KJ (2003) Non-indigenous fishes in restored and natural wetlands on the Big Cypress Seminole Indian Reservation. Master’s Thesis, Florida Atlantic University
Eliason EJ, Clark TD, Hague MJ, Hanson LM, Gallagher ZS, Jeffries KM, Gale MK, Patterson DA, Hinch SG, Farrell AP (2011) Differences in Thermal Tolerance among sockeye salmon populations. Science 332:109–112. https://doi.org/10.1126/science.1199158
Feminella JW, Matthews WJ (1984) Intraspecific differences in thermal tolerance of Etheostoma spectabile (Agassiz) in constant versus fluctuating environments. J Fish Biol 25:455–461. https://doi.org/10.1111/j.1095-8649.1984.tb04892.x
Greenwood MFD (2012) Assessing effects of the nonindigenous Pike Killifish on indigenous fishes in Tampa Bay, Florida, using a weighted-evidence approach. T Am Fish Soc 141:84–99. https://doi.org/10.1080/00028487.2011.651552
Healy T, Schulte PM (2012) Factors affecting plasticity in whole-organism thermal tolerance in common killifish (Fundulus heteroclitus). J Comp Physiol 182:49–62. https://doi.org/10.1007/s00360-011-0595-x
Hickman GD, Dewey MR (1973) Notes on the upper lethal temperature of the duskystripe shiner, Notropis pilsbryi, and the bluegill, Lepomis macrochirus. Ttans Am Fish Soc 102:838–840. https://doi.org/10.1577/1548-8659(1973)102<838:NOTULT>2.0.CO;2
Kerfoot JR Jr (2012) Thermal tolerance of the invasive Belonesox belizanus, pike killifish, throughout ontogeny. J Exp Zool Part A 317:266–274. https://doi.org/10.1002/jez.1720
Kerfoot JR Jr, Lorenz JJ, Turingan RG (2011) Environmental correlates of the abundance and distribution of Belonesox belizanus in a novel environment. Environ Biol Fish 92:125–139. https://doi.org/10.1007/s10641-011-9822-2
Johnson T, Bennett A (1995) The thermal acclimation of burst escape performance in fish: an integrated study of molecular and cellular physiology and organismal performance. J Exp Biol 198:2165–2175. https://doi.org/10.1242/jeb.198.10.2165
Johnston IA, Temple GK (2002) Thermal plasticity of skeletal muscle phenotype in ectothermic vertebrates and its significance for locomotory behavior. J Exp Biol 205:2305–2322. https://doi.org/10.1242/jeb.205.15.2305
Jonsson B, Jonsson N (2014) Early environment influences later performance in fishes. J Fish Biol 85:151–188. https://doi.org/10.1111/jfb.12432
Jonsson B, Jonsson N (2019) Phenotypic plasticity and epigenetics of fish: embro temperature affects later-developing life-history traits. Aquat Biol 28:21–32. https://doi.org/10.3354/ab00707
Lighten J, Incarnato D, Ward BJ, van Oosterhout C, Bradbury I, Hanson M, Bentzen P (2016) Adaptive phenotypic response to climate enabled by epigenetics in a K-strategy species, the fish Leucoraja ocellate (Rajidae). Roy Soc Open Sci 3:160299. https://doi.org/10.1098/rsos.160299
Lockwood JL, Hoopes MF, Marchetti MP (2007) Chap. 1: An Introduction to Invasion Ecology. In: Lockwood JL, Hoopes MF, Marchetti MP (eds) Invasion Ecology. Blackwell Publishing, Malden, pp 1–18
Loftus WF, Ellis G, Zokan M, Lorenz J (2004) Inventory of freshwater fish species within Big Cypress National Preserve: The basis for a long-term sampling program (USGS Fact Sheet 2004–3131). USGS, U.S. Department of the Interior (US)
Marchetti MP, Moyle PB, Levine R (2004) Invasive species profiling? Exploring the characteristics of non-native fishes across invasion stages in California. Freshw Biol 49:646–661. https://doi.org/10.1111/j.1365-2427.2004.01202.x
Özbilgin H, Wardle CS (2002) Effect of seasonal temperature changes on the escape behavior of haddock, Melanogrammus aeglefinus, from the cod end. Fish Res 58:323–331. https://doi.org/10.1016/S0165-7836(01)00394-0
Podrabsky J, Riggs C, Wagner J (2015) Tolerance of environmental stress. In: Garcia G, Berois B, de Sa R (eds) Annual fishes: life history strategy, diversity, and evolution. CRC Taylor & Francis, Boca Raton, pp 159–184
Quinn GP, Keough MJ (2002) Chap. 9: Multifactor analysis of variance. In: Quinn GP, Keough MJ (eds) Experimental design and data analysis for biologists. Cambridge University Press, Cambridge, pp 208–261
Randall D, Brauner C (1991) Effects of environmental factors on exercise in fish. J Exp Biol 160:113–126. https://doi.org/10.1242/jeb.160.1.113
Robbins LW, Hartman GD, Smith MH (1987) Dispersal, reproductive strategies, and the maintenance of genetic variability in mosquitofish (Gambusia affinis). Copeia 1:156–164. https://doi.org/10.2307/1446047
Sakai AK, Allendorf FW, Holt JS, Lodge DM, Molofsky J, With KA, Baughman S, Cabin RJ, Cohen JE, Ellstrand NC, McCauley DE, O’neil P, Parker IM, Thompson JN, Weller SG (2001) The Population Biology of Invasive Species. Annu Rev Ecol Syst 32:305–332. https://doi.org/10.1146/annurev.ecolsys.32.081501.114037
Sanford MT (2003) Florida’s climate and its beekeeping (University of Florida Fact Sheet ENY-134). University of Florida, Gainesville, FL
Schaefer JF (2006) A simple and economical method for precise, programmable thermal regimes in recirculating aquatic mesocosms. N Am J Aquacult 68:240–244. https://doi.org/10.1577/A05-069.1
Schofield PJ, Loftus WF, Kobza RM, Cook MI, Slone DH (2010) Tolerance of nonindigenous cichlid fishes (Cichlasoma urophthalmus, Hemichromis letoureuxi) to low temperature: laboratory and field experiments in south Florida. Biol Invasions 12:2441–2457. https://doi.org/10.1007/s10530-009-9654-6
Shafland PL, Pestrak JM (1982) Lower lethal temperatures for fourteen non-native fishes in Florida. Environ Biol Fish 7:149–156. https://doi.org/10.1007/BF00001785
Stauffer JR Jr (1984) Chap. 2: Colonization theory relative to introduced populations. In: Courtenay WR Jr, Stauffer JR Jr (eds) Distribution, biology and management of exotic fishes. Johns Hopkins University Press, Baltimore, pp 8–21
Stitt BC, Burness G, Burgomaster KA, Currie S, McDermid JL, Wilson CC (2014) Intraspecific variation in thermal tolerance and acclimation capacity n brook trout (Salvelinus fontinalis): Physiological implication for climate change. Physiol Biochem Zool 87:15–29. https://doi.org/10.1086/675259
Strange KT, Vokoun JC, Noltie DB (2002) Thermal tolerance and growth differences in Orangethroat darter (Etheostoma spectabile) from thermally contrasting adjoining streams. Am Midl Nat 148:120–128. https://doi.org/10.1674/0003-0031(2002)148[0120:TTAGDI]2.0.CO;2
Taylor JN, Courtenay WR Jr, McCann JA (1984) Chap. 16: Known impacts of exotic fishes in the continental United States. In: Courtenay WR Jr, Stauffer JR Jr (eds) Distribution, biology and management of exotic fishes. Johns Hopkins University Press, Baltimore, pp 322–373
Trexler JC, Loftus WF, Jordan F, Lorenz JJ, Chick JH, Kobza RM (2000) Empirical assessment of fish introductions in a subtropical wetland: an evaluation of contrasting views. Biol Invasions 2:265–277. https://doi.org/10.1023/A:1011488118444
Trexler JC, Loftus WF, Perry S (2005) Disturbance frequency and community structure in a twenty-five-year intervention study. Oecologia 145:140–152. https://doi.org/10.1007/s00442-005-0094-4
Turner JS, Snelson FF (1984) Population structure, reproduction, and laboratory behavior of the introduced Belonesox belizanus (Poeciliidae) in Florida. Environ Biol Fish 10:89–100. https://doi.org/10.1007/BF00001665
Wolter C, Röhr F (2010) Distribution history of non-native freshwater fish species in Germany: how invasive are they? J Appl Ichthyol 26:19–27. https://doi.org/10.1111/j.1439-0426.2010.01505.x
Wood ZT, Shepard ID, Hurley ST, Paisker MR, Hensley VR, Kinnison MT (2020) Sex-dependent cold tolerance at the northern invasive range limit of Gambusia affinis on Cape Cod, Massachusetts. Copeia 108:670-678. https://doi.org/10.1643/CE-19-332
Acknowledgements
I would like to thank Matt Wittenrich, Ronald Maliao, and Paul Sexton for their assistance in collecting B. belizanus from the Everglades and Big Cypress National Parks. I would also like to recognize Arthur Kunkle for his assistance in designing the computer-controlled temperature environment used for the experiments. I appreciate the valuable input that Ralph Turingan offered in implementing the experiments and the use of his laboratory space. Finally, I would like to thank Charley Kate Barcroft, Mark Bolyard, Gabi Mirabella, Darius Mullin, Dylan Parmely, Sam Ross, Tayler Schudel, and anonymous reviewers for their valuable comments on earlier drafts of this manuscript. This study was completed under Florida Institute of Technology’s Animal Care and Use Committee guidelines (IACUC Permit No. 99 − 01).
Funding
Funding in part from the Florida Institute of Technology, Graduate Research Grant.
Author information
Authors and Affiliations
Contributions
Experimentation, data collection, analysis, and manuscript writing were done by J. Kerfoot.
Corresponding author
Ethics declarations
Ethics approval
This study was completed under Florida Institute of Technology’s Animal Care and Use Committee guidelines (IACUC Permit No. 99 − 01).
Consent to participate
Not applicable.
Consent for publication
J. Kerfoot consents for publication of this manuscript.
Conflicts of interest/Competing interests
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kerfoot, J.R. Northward expansion leads to cold tolerance? Investigating thermal adaptation of the non-native pike killifish (Belonesox belizanus) in Florida. Environ Biol Fish 105, 487–497 (2022). https://doi.org/10.1007/s10641-022-01248-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-022-01248-2