Abstract
Purpose
The DTL fibre electrode is commonly used to record the electric potentials elicited by stimulation of the retina. Two positions are commonly used: it is placed either on the cornea along the lower lid or in the conjunctival fornix. The PERG and OPs have previously been examined and compared under both conditions. The aim of this study was to examine the ERG, flicker response and on–off responses with differing electrode positions.
Methods
Before recruitment, all subjects underwent an ophthalmological examination. We enrolled 13 normal control subjects into the study aged 13–64 years, all with a visual acuity of ≥1.0. We recorded scotopic and photopic ERGs, flicker and on–off responses, for both electrode positions. On the first day, one eye had the electrode placed on the cornea along the lower lid and the other eye had it positioned in the conjunctival sac. On a second day, the recordings were repeated with the alternative electrode placements.
Results
ERG, on–off and flicker responses were all smaller by between 20 and 25% when the DTL electrode was positioned in the conjunctival sac, compared to when it was positioned on the cornea, as did the scatter in the data points. This indicates that there is no advantage clinically for one or the other placement.
Conclusions
Our results confirm other reports examining the effect of electrode position on electrophysiological potentials. When recording with the DTL electrode, it is important to ensure that it is placed at the same position in repeat recordings or in multicentre trials and that it is stable and does not move during recording.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In the ISCEV guidelines for performing the electroretinogram (ERG), the position of the active electrode is recommended to have contact with the cornea or the nearby bulbar conjunctiva [1]. It is known, however, that the two positions do not produce identical ERGs: the amplitude decreases with the distance of the electrode from the corneal apex [2].
The DTL electrode, a silver-coated nylon fibre, was developed by Dawson et al. in 1979 [3] and is widely used for recording the ERG. They state “The fibre may be stretched above the cornea of the open eye. The fibre then floats upon the surface film of the eye and is only disturbed a little by blinking”. The fibre is usually simply laid on the cornea along the lower lid, but holders for the electrode to position it correctly are also employed [4, 5]. The problem with this corneal position, apart from blinking, is that eye movements and tearing can dislodge the thread out of the eye or it can be swept to the conjunctival sac, where the potentials are smaller in amplitude. Corneal abrasion can also occur, and an anaesthetic may be required to obtain an acceptable recording. An alternative to placing the electrode on the cornea is to place it in the conjunctival fornix with the advantage that the deeper position is more comfortable for the patient and the use of anaesthetic is not required.
Whereas animal studies have shown that the position of the electrode on the cornea is critical for the amplitude of response [2, 6,7,8,9], there are relatively few studies directly comparing recordings obtained from the two electrode positions in humans: Otto & Bach [10] have examined the differences in the pattern ERG (PERG) and found that when the electrode is placed in the lower fornix, amplitudes are around 80% of those obtained when the electrode is placed across the cornea at the level of the lower lid, and Lachapelle and colleagues [11] have estimated from their experience a 30% decrease in oscillatory potential (OP) amplitudes. It is important therefore to understand the effects of electrode placement on electrophysiological recordings. The rationale here was to examine its effect on the ERG, 9-Hz flicker and on–off responses.
The aim of this study was to examine the recordings obtained from the two electrode placements, to ascertain which is more suitable for clinical use. We recorded scotopic and photopic ERGs as well as flicker and on–off responses. The recordings were repeated on a separate day with the reverse eye settings. Additionally, we examined the interocular differences recorded on different days.
Methods
Subjects
Thirteen subjects aged between 13 and 64 years (mean 42.3 years ± 17.0 SD) were recruited in the study. Nine were female, and three were male. They showed normal results in an ophthalmological examination performed before recruitment. Their corrected visual acuity was ≥1.0.
Methods
The Espion E2 (Diagnosys LLC) system was used to generate and analyse all results. Custom-made DTL electrodes were used to record scotopic (0.001, 0.01. 0.1, 3.0 and 10 cd s m−2) and photopic (3.0 cd s m−2) ERGs according to ISCEV guidelines [1]. Only the b-wave of the 0.001 and 0.01 stimuli were analysed. On–off responses were also recorded with an achromatic stimulus of 80 cd/m2 on a background of 20 cd/m2. The pulse duration was 240 ms with an inter-sweep delay of 500 ms. The recording was bandpass filtered between 10 and 300 Hz. The b- and d-waves were further analysed. Additionally, we recorded the response to a blue 9-Hz flicker stimulus of 3 scot cd/m2 and a pulse duration of 10 ms. The bandpass filter was set to 0.3 and 300 Hz. Three recordings were averaged for the final trace. Here, the amplitude was taken for further analysis.
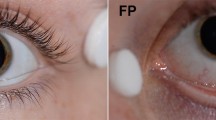
Before recording, pupils were fully dilated to approximately 8 mm with 0.5% tropicamide and the subject dark adapted for 30 min. The signal was recorded from both eyes simultaneously with DTL fibre electrodes attached with its two ends to the lateral and nasal canthus. On the first day, one eye was randomly chosen and the electrode was positioned along the lower lid; in the other eye, it was placed deep in the conjunctival sac, as shown in Fig. 1. Two ground skin electrodes (Ag–AgCl) and one reference electrode were attached to the ipsilateral temples and to the forehead, respectively. The recordings were repeated in a second run of the experiment within 7 days with the electrode placements reversed.
Data analysis
Amplitudes and implicit times for the major inflections in each recording were calculated. The per cent difference between electrode placements for each parameter was calculated for each subject by dividing the amplitude difference by the higher amplitude [12]. The average difference for the two placements was calculated for each subject, and the means and SDs of these averaged data were taken for further analyses. Interocular differences for recordings performed on different days with the same electrode position were calculated in similar manner. Paired comparisons were then performed using the Bland–Altman test [13]. Correction for multiple testing was carried out using the Bonferroni–Holm method [14]. Comparisons were considered significant at p < 0.05 after correction.
Results
In the upper panels of Fig. 2, we show representative dark-adapted ERG recordings for increasing stimulus intensities from one subject with the DTL electrodes placed in one eye on the cornea along the lid (grey) and on the other eye in the conjunctival sac (red). The middle panel shows the 9-Hz flicker results and the bottom panel, the light-adapted recordings (left) and the on–off responses (right). It will be seen that in all cases amplitudes are reduced when the electrode is placed in the conjunctival sac, whereas implicit times remain similar.
The mean differences and SDs between electrode positions are listed for each parameter in Table 1. For the dark-adapted eye, placing the electrodes lower in the conjunctival sac produces smaller amplitudes, on average by 23% for the a-wave and 22% for the b-wave. For the 9-Hz flicker, the mean difference was 20%.
For the light-adapted eye, similar differences to those of the dark-adapted eye are found: The 0.5 photopic step showed a mean amplitude difference of 27% for the a-wave, and 20% for the b-wave. For the on–off responses, the mean difference was 20%. The difference between the two electrode positions is statistically significant for all parameters. Implicit times, on the other hand, show no statistically significant changes.
The mean ERG results and SDs for the a- and b-wave amplitudes for all 26 eyes taken together are shown in Fig. 3. If we look at the SD of average amplitudes, the variance of the data also decreases by about 20% on average with electrode placement in the conjunctival sac.
We additionally calculated the interocular differences for recordings performed on different days with the same electrode position. The average difference for the dark-adapted a-wave amplitudes with the electrode placed along the lower lid was 12%, and that for b-wave amplitudes was 14%. The 9-Hz flicker showed a 16% difference. For the light-adapted retina, the photopic difference was 10 and 14% for a- and b- waves, respectively, and for the on–off responses 27%. These differences were not significant, as were those found if the electrode was placed in the conjunctival sac; 14% for the average of both a- and b- waves of the dark-adapted retina and 22 and 10% for the a and b-waves of the photopic response, respectively. The amplitude differences between eyes are also similar for the 9-Hz flicker (14%) and on–off (23%). In all but one case there were no statistically significant differences between eyes.
Discussion
This study was performed to verify and extend the published literature on the differences between electroretinographic recordings obtained when the DTL fibre electrode is placed on the cornea to those when it is placed in the conjunctival fornix. In the literature both electrode positions are commonly used.
Our results show that ERG, on–off and flicker responses decrease by around 20–25% when the DTL electrode is placed deep in the conjunctival sac, compared to when it is placed on the cornea along the lower lid (see Table 1). These results are in line with the 20% reduction reported by Otto & Bach for the PERG [10] and the 30% reduction estimated by Lachapelle [11] for OPs.
Our data also allow an analysis of the interocular differences, albeit for recordings performed on different days. We find differences ranging from 8 to 27% when the electrode is placed on the cornea and 10–25% when the electrode is placed in the conjunctival fornix. These results are somewhat larger than those of Rotenstreich et al. [12] who found mean interocular percentage differences of between 11 and 14% and Fishman et al. who found a 10% difference. [15]. However, our results were recorded on different days and so will likely reflect a repeatability factor. For recordings of the scotopic ERG, It has been shown that the mean variability between two recording sessions is 3.2% for V max, 1.5% for K [16] and 10.5 and 11.4% for the a- and b- photopic waves, respectively [17]. For OPs variability is generally larger at 25% [18].
In conclusion, our results show that electrode placement plays little role in the diagnostic accuracy of the ERG. Subjects generally found positioning of the electrode in the conjunctival sac more tolerable without anaesthetic, and artefacts caused by blinks tended to be fewer. In young patients, with firmer tissues, however, the electrode may more easily be displaced upwards. Important is that the electrode is in its intended location and does not change its position during recording and that the position used for recording is protocolled in clinical trials and in patient files.
References
McCulloch DL, Marmor MF, Brigell MG, Hamilton R, Holder GE, Tzekov R, Bach M (2015) ISCEV standard for full-field clinical electroretinography (2015 update). Doc Ophthalmol 130(1):1–12. doi:10.1007/s10633-014-9473-7
Mentzer AE, Eifler DM, Montiani-Ferreira F, Tuntivanich N, Forcier JQ, Petersen-Jones SM (2005) Influence of recording electrode type and reference electrode position on the canine electroretinogram. Doc Ophthalmol 111(2):95–106. doi:10.1007/s10633-005-4517-7
Dawson WW, Trick GL, Litzkow CA (1979) Improved electrode for electroretinography. Invest Ophthalmol Vis Sci 18(9):988–991
Thompson DA, Drasdo N (1987) Computation of the luminance and pattern components of the bar pattern electroretinogram. Doc Ophthalmol 66(3):233–244
Mierdel P (1995) An improved holder for the DTL fiber electrode in electroretinography. Doc Ophthalmol 89(3):249–250
Krakau CET (1958) On the potential field of the rabbit electroretinogram. Acta Ophthalmol 36(2):183–207. doi:10.1111/j.1755-3768.1958.tb07707.x
Honda Y (1977) Some characteristics of the c-wave of ERGs recorded by a pair of electrodes on the cornea and sclera. Albrecht von Graefes Archiv für klinische und experimentelle Ophthalmologie 202(1):19–26. doi:10.1007/BF00496765
Doslak MJ, Plonsey R, Thomas CW (1980) The effects of variations of the conducting media inhomogeneities on the electroretinogram. IEEE Trans Biomed Eng 27(2):88–94. doi:10.1109/TBME.1980.326712
Cringle SJ, Alder VA, Brown MJ, Yu DY (1986) Effect of scleral recording location on ERG amplitude. Curr Eye Res 5(12):959–965
Otto T, Bach M (1997) Reproducibility of the pattern electroretinogram. Ophthalmologe 94(3):217–221
Lachapelle P, Benoit J, Little JM, Lachapelle B (1993) Recording the oscillatory potentials of the electroretinogram with the DTL electrode. Doc Ophthalmol 83(2):119–130
Rotenstreich Y, Fishman GA, Anderson RJ, Birch DG (2003) Interocular amplitude differences of the full field electroretinogram in normal subjects. Br J Ophthalmol 87(10):1268–1271
Bland JM, Altman DG (1986) Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1(8476):307–310
Holm S (1979) A simple sequential rejective multiple test procedure. Scand J Stat 6:65–70
Fishman G, Birch D, Holder G (2001) Electrophysiologic testing in disorders of the retina, optic nerve, and visual pathway, 2nd edn. The Foundation of the American Academy of Ophthalmology, San Francisco
Hebert M, Lachapelle P, Dumont M (1996) Reproducibility of electroretinograms recorded with DTL electrodes. Doc Ophthalmol 91(4):333–342
Hebert M, Vaegan Lachapelle P (1999) Reproducibility of ERG responses obtained with the DTL electrode. Vis Res 39(6):1069–1070
Kothe AC, Lovasik JV, Coupland SG (1989) Variability in clinically measured photopic oscillatory potentials. Doc Ophthalmol 71(4):381–395
Funding
The study was supported by Grants from the Erasmus + Programme and the German Ophthalmological Society (DOG) to HL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Statements of human rights
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Statement on the welfare of animals
This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Kurtenbach, A., Kramer, S., Strasser, T. et al. The importance of electrode position in visual electrophysiology. Doc Ophthalmol 134, 129–134 (2017). https://doi.org/10.1007/s10633-017-9579-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10633-017-9579-9