Abstract
Background
Gastric signet ring cell carcinoma (GSRC) is a rare but increasingly prevalent tumor histotype whose clinical features and natural history are poorly understood, particularly in the USA and minorities.
Aims
To examine the occurrence, clinico-demographic characteristics, oncologic features, treatment, and outcomes of GSRC in a predominantly minority county hospital setting and benchmark them against data from the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) program.
Methods
We queried biopsy-proven GSRC cases at a Los Angeles County hospital, from 2004 to 2017. Clinical characteristics, treatment, and survival data were collected and compared to SEER data.
Results
We identified 63 patients with GSRC. Compared to SEER, our cohort was significantly younger (52.6 vs. 63.5 years, p < 0.01), Hispanic/Latino predominant (81% vs. 20%, p < 0.01), had higher overall stage (86% vs. 69% with stage III/IV, p < 0.01), and more frequent node involvement (89% vs. 49%, p < 0.01). Lower tumor stage, Helicobacter pylori positivity, and surgical intervention were associated with significantly longer median survival (all p < 0.05), which was similar in our study compared to SEER (median 12.6 vs. 9.0 months, p = 0.26).
Conclusions
Patients with GSRC within the Los Angeles County population have different clinical characteristics compared to what has been reported in SEER. Our cohort was younger, and despite having more advanced disease, did not have shorter survival. Further study is needed to better identify protective and risk factors in this population and improve understanding of the etiopathogenesis and natural history of this malignancy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gastric cancer is the sixth most common malignancy worldwide, with approximately 1.2 million new cases diagnosed in 2016 [1]. Despite its overall declining incidence in recent years, gastric cancer is still the second leading cause of cancer-related death worldwide [1, 2]. Furthermore, the incidence of a specific histotype of gastric cancer, signet ring cell carcinoma (SRC), has been increasing in recent years [3]. Indeed, data from the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) program demonstrate a tenfold increase in the incidence of SRC between 1973 and 2000 [3, 4]. Newer SEER data have shown that even in light of national cancer detection and prevention efforts, the incidence of SRC has not decreased [5].
Despite these troubling statistics, data from the USA regarding GSRC are particularly scarce, in part due to SRC being an uncommon malignancy. According to the World Health Organization (WHO) classification, GSRC is a histological type of epithelial carcinoma based on more than 50% of the tumor containing intracytoplasmic mucin. This globoid, optically clear glycoprotein pushes the cells’ nuclei against the cell membrane, creating what has been classically described as a “signet ring” appearance [6]. Compared to other gastric cancer types, it has been reported to have a female predominance, younger age at diagnosis, and late presentation, making treatment particularly challenging [7, 8]. Although there have been studies of the clinicopathological features and prognosis of GSRC, results have been inconsistent, with some reports showing SRC to have a better prognosis than other gastric carcinomas [9], some showing no significant difference [10, 11], and others reporting a poorer prognosis [8].
At our institution, a predominantly minority county hospital in Los Angeles County, it was independently observed by both the Department of Pathology and Department of Medicine that the number of GSRC cases seemed surprisingly large and the presentation commonly in young patients. We therefore aimed to examine the occurrence, features, and outcomes of GSRC in this unique population and benchmarked these characteristics against SEER data.
Methods
This study was approved by the Institutional Review Board of Olive View-UCLA Medical Center.
Patients and Setting
We retrospectively identified all patients greater than 18 years of age with a biopsy-proven diagnosis of GSRC at Olive View-UCLA Medical Center between the years of 2004 and 2017. Olive View-UCLA Medical Center is a 377-bed academic hospital which serves a population of over 2 million people within a catchment area of nearly 1000 square miles and is operated by the Los Angeles County Department of Health Services, the second largest municipal health care system in the USA [12]. Approximately 65% of the population served by the institution identify as Hispanic/Latino (hereinafter “Latino”), and over 50% speak a primary language other than English [13]. Sixty percent of patients are foreign born, of which 20% were born in Mexico, 7% in the Philippines, 6% in El Salvador, 4% in Guatemala, and another 5% from other Central and South American countries.
In order to compare the characteristics and outcomes of patients in this study with that of other cohorts, we abstracted data from the National Cancer Institute’s SEER program. The SEER database represents the most comprehensive cancer registry in the USA, representing approximately 27.8% of the US population [14]. We selected patients with GSRC using the International Classification of Diseases for Oncology, 3rd edition (ICD-O-3) code 8490 and filtering only for cases of gastric primary. SEER data that were abstracted included gender, race, tumor location, T stage, N stage, overall cancer stage, tumor differentiation, and survival time.
Study Variables and Outcomes
Comprehensive demographic, clinical, endoscopic, histopathologic, imaging, and oncologic data were abstracted from the medical record using a standardized data collection form, including: sex, age at diagnosis, race, tumor size, tumor location, TMN stage, differentiation, lymph node involvement, type of surgery, neoadjuvant and adjuvant chemotherapy, Helicobacter pylori (H. pylori) infection status, and time to death or hospice.
The primary outcome was survival, defined as time to either death or hospice. Covariates included patient characteristics (age, gender, race), presenting tumor features (stage, size, tumor location, tumor differentiation), and H. pylori status. We compared these features to SEER data when available, both overall as well as within the subgroup of Latino patients. Age at diagnosis was determined by subtracting the year of birth from the year of diagnosis. Similarly, time to death or hospice was determined by subtracting the date of death or date of hospice initiation from the date of diagnosis. Pathological stage was classified according to the American Joint Committee on Cancer (AJCC) staging manual, 7th edition [15]. Helicobacter pylori status was determined by reviewing histopathology reports (most often confirmed by Warthin-Starry silver stain), serum H. pylori IgG antibody, or stool H. pylori antigen testing results. Patients were categorized as H. pylori positive if any of the three testing methods were positive and H. pylori negative if all H. pylori tests that were performed were negative. Patients without documented H. pylori testing were categorized as unknown status.
Statistical Analysis
Demographic and clinical characteristics were examined using descriptive statistics. Means were compared using independent t-tests. Variables which were not normally distributed (or that otherwise did not satisfy assumptions for parametric tests) were summarized and compared using nonparametric tests. Overall and subgroup (sex, age, tumor size, tumor stage, H. pylori status) survival curves were generated using the Kaplan–Meier method and then compared with the log-rank test. Time to hospice or death (by any cause) was used as a proxy for approximating survival. All study data were analyzed using IBM SPSS Statistics 23 (SPSS, Inc., Chicago, IL). SEER data were analyzed using the SEER*Stat software (version 8.3.5, National Cancer Institute). Patient characteristics were benchmarked against SEER data.
Results
Clinical and Demographic Characteristics
A total of 63 patients with a diagnosis of GSRC during the study period were identified, yielding an overall estimated annual incidence of approximately 6.22 per 100,000 persons. Of these 63 patients, 81.0% were Latino, 7.9% White, 6.3% Other, and 4.8% Asian/Pacific Islander (Table 1). Cases of GSRC had a female predominance (57.1%). The mean age at diagnosis was 52.6 (standard deviation [SD] 11.9) years. Of the 41 patients (65.1%) in our sample who had H. pylori testing data, recent or ongoing H. pylori infection was present in 75.6% of patients, of which 38.7% were diagnosed by serum antibody, 48.4% by histopathology, 6.5% by stool antigen, and 6.5% by a combination of serum antibody and histopathology. Median follow-up was 8.3 months (interquartile range 2.2–18.1 months). The most common presenting symptoms were abdominal pain, nausea, vomiting, and/or dyspepsia. Four patients were asymptomatic at initial presentation; of these four, two underwent endoscopy for evaluation of iron deficiency anemia, one for esophageal varix screening, and one for an incidental finding on radiographic imaging.
Tumor Presentation
Mean tumor size was 6.8 (SD 5.1) cm (Table 2). Approximately 8.9% of cases were located at the gastroesophageal junction, 2.2% in the gastric fundus, 37.8% in the body, 24.4% in the antrum, and 36.7% in multiple locations or diffusely. Over 75% of patients presented with tumor stages T3 or T4, and nearly 90% presented with lymph node metastases. Eighty-six percent of patients presented with cancer stage III or IV, and approximately 87% had poorly differentiated (G3) or undifferentiated (G4) histology.
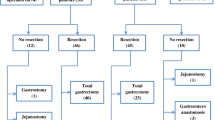
Oncologic Treatment
Sixty-two percent of patients underwent either subtotal or total gastrectomy (Table 3). Approximately 10% had neoadjuvant chemotherapy consisting of capecitabine and an alkylating antineoplastic agent (either cisplatin or oxaliplatin), ± epirubicin. Seventy-six percent of patients received adjuvant chemotherapy (most frequently with 5-fluorouracil and radiotherapy via the Macdonald protocol) with either palliative or curative intent.
Survival Outcomes
The median time between date of diagnosis and date of death or hospice initiation was 12.6 months, and 5-year survival was 14.3% (Table 3 and Figs. 1 and 2). Within 2 years of diagnosis, 88% of patients were either deceased or in hospice care. Median survival in males and females did not differ (12.9 vs. 12.6 months, p = 0.406), and there was no significant difference in survival between patients who were younger than 52 (the median age of patients in this study) and older than 52 years of age at diagnosis (15.8 vs. 11.4 months, p = 0.291). Similarly, there was no significant difference in median survival between patients with tumors smaller than 5.5 cm (the median tumor size in this study) and larger than 5.5 cm (20.3 vs. 12.9 months, p = 0.171), though there appeared to be a clinically significant trend toward shorter survival with larger tumors. Higher overall cancer stage (stage IV vs. stage I–III) was associated with shorter survival (4.2 months vs. 20.4 months, p = 0.001). Patients with recent or active H. pylori infection were found to have longer survival than patients who tested negative for H. pylori (17.9 vs. 5.6 months, p = 0.017). Patients who underwent surgical intervention had longer survival than patients who did not undergo surgery (20.4 vs. 3.3 months, p < 0.001). Survival did not appear to be associated with whether or not patients received chemotherapy (13.0 vs. 17.9 months, p = 0.618). These predictors of survival are shown in Fig. 2.
Kaplan–Meier survival curves of patients with gastric signet ring cell carcinoma. Kaplan–Meier survival curves comparing months of survival in gastric signet ring cell carcinoma (GSRC) are shown for a all patients, b by gender (p = 0.406), c by age at diagnosis (p = 0.291), d by tumor size (p = 0.171), e by cancer stage (p = 0.004), f by H. pylori infection status (p = 0.017), g by surgical intervention (p < 0.001), and h by chemotherapy (p = 0.618). All comparisons performed by log-rank tests
Demographic and clinical predictors of survival. Shown in the plotted data is median survival as a function of various dichotomized demographic and clinical variables. The following variables were associated with shortened survival (i.e. shorter time to death or hospice), as indicated by an asterisk: advanced TNM stage (p = 0.001), H. pylori negativity (p = 0.017), and no surgery (p < 0.001). Whiskers indicate 95% confidence intervals
Comparisons to SEER Data
Compared to SEER data, the mean age at GSRC diagnosis was younger in our study cohort, with the average age of diagnosis being 10 years earlier (52.6 vs. 63.5 years, p < 0.01). Our study cohort also had a higher percentage of females, with 57.1% of patients with a new diagnosis of GSRC being female compared to 45% in SEER (p = 0.15). A higher percentage of patients presented with advanced cancer staging; 86% of patients presented with stage III or IV disease compared with 68.8% in SEER (p < 0.01). Lymph node involvement was also higher in this population (89.2% vs. 48.5%, p < 0.01). However, a lower percentage of patients had poorly differentiated or undifferentiated tumors (87.5% vs. 95.6%, p < 0.01). Despite more frequent advanced staging and lymph node involvement, patients in our study appeared to have similar survival to those in SEER (12.6 vs. 9.0 months, p = 0.26).
Further analysis comparing only Latino patients in our cohort to Latino patients in SEER revealed similar findings: average age of diagnosis was still younger in our cohort (though the age gap was smaller, 52.6 vs. 57.0 years, p = 0.01), the proportion of females with GSRC was higher (62.7% vs. 51.3%, p = 0.10), the percentage of patients with advanced cancer staging (stage III or IV) and lymph node involvement was higher (80% vs. 64.1%, p = 0.03 and 73.9% vs. 23.0%, p < 0.01, respectively), the percentage of patients with poorly differentiated of undifferentiated tumors was lower (84.3% vs. 97.6%), and survival was similar (12.2 vs. 10.0 months, p = 0.49).
Discussion
GSRC is a rare but highly lethal malignancy whose incidence, in contrast to gastric cancer overall, does not appear to be decreasing in the USA. In this study, we aimed to examine and better understand the clinical epidemiology of GSRC in the USA and specifically the predominantly minority patient population of Los Angeles County. Our main findings are that patients with GSRC in our cohort, compared to SEER, had: (1) younger age at diagnosis, (2) more advanced staging and lymph node involvement, and (3) similar survival times. Furthermore, we estimated the incidence of GSRC in our patient population to be 6.22 per 100,000 persons, higher than what has been reported in SEER, 1.27 per 100,000 persons [16]. However, this is likely an overestimate due to the fact that this study was performed at a tertiary referral center.
We believe that ethnoracial (and possibly geographical and environmental) factors may play a role in the differences in mean age at GSRC diagnosis observed between our Los Angeles County-based cohort and SEER. Indeed, when including only Latino patients from the SEER database, we found the average age of diagnosis to be 57 years, which is more similar to our cohort (52.6 years) and younger than SEER patients overall (63.5 years). This suggests that Latino patients may represent a high-risk group for developing GSRC (i.e., at a younger age) [17]. In addition, the younger age of diagnosis in Latino patients appears to be similar to and in some cases even earlier than other populations traditionally considered to be high-risk [18,19,20]. Cancer registries from Latin American countries are not available at this time to determine if the natural history of GSRC in Latino patients is similar internationally compared to the USA.
Our finding that 57.1% of patients with a new diagnoses of GSRC were female is higher than both SEER data as well as prior studies of patients with GSRC, though was not significant at the 0.05 level [10, 11, 18,19,20]. While it is unclear why the proportion of females was higher in this study, several other studies have found that a higher percentage of GSRC patients are female when compared with non-GSRC patients. It has been hypothesized that SRC histology may be influenced by female sex hormones, as estrogen receptors have frequently been found to be expressed in poorly differentiated gastric cancer, thus suggesting a relationship between hormone environment and SRC [21].
A higher percentage of patients presented with advanced cancer staging and lymph node involvement. Interestingly, the presentation of GSRC has been observed to differ between studies from the West compared to the East; for example, US studies have reported GSRC to be associated with advanced staging upon presentation, while many Asian studies suggest that GSRC is diagnosed at earlier stages [11, 18, 19, 22]. One potential explanation for this difference is variation in preventative cancer screening practices throughout the world. Asian countries such as Korea and Japan have programs for and high participation rates in gastric cancer screening [23, 24], which would likely contribute to cancers being detected in earlier stages. Conversely, in Western countries, diagnostic testing is often performed only after symptom onset. In the present study, patients presented with even more advanced presentations than has been previously reported in Western studies, potentially in part due to delays in and difficulties with access to health care in this largely uninsured population. Routine screening for gastric cancer in asymptomatic patients is not currently performed at our hospital.
Even with more advanced staging and lymph node involvement, survival appeared to be similar in this population compared to SEER data. In SEER, the median survival was 9.0 months. Although we were not able to calculate true median survival due to limited survival data, by including time until hospice, we found the mean time to death or initiation of hospice to be 12.6 months, indicating the possible existence of protective factors in this population. From their analysis of SEER data from 2004 to 2007, Taghavi et al. previously indicated that age of diagnosis, Hispanic ethnicity, size and extent of the tumor (T), nodal involvement (N), histologic grade, and overall stage are associated with increased mortality [11]. In our study, though the majority of patients were Latino and had relatively high overall stage and lymph node involvement, the average age of diagnosis was younger, and the percentage of high grade tumors (poorly differentiated and undifferentiated combined) was lower (86.8% vs. 96.5%), thus ostensibly contributing to better survival outcomes [17].
In survival analyses, we found no significant difference in survival based on gender, age, tumor size, and treatment with chemotherapy. However, as expected, longer survival times were observed for patients with lower tumor stages. We also found improved survival with surgery (gastrectomy) versus no surgery, but the no surgery group was expectedly biased toward more advanced tumor staging (all had T4 disease), as it was the patients with metastatic disease who were usually not surgical candidates. Lastly, we found improved survival with H. pylori positivity. Previous studies of H. pylori and gastric cancer have found that most patients with gastric cancer are H. pylori-positive, with reported seropositivity rates varying from 80 to 100% [25, 26]. For the small percentage of patients with H. pylori-negative gastric cancer, the most common subtype is SRC [27]. However, the role of H. pylori in GSRC is unclear, as there have been very few studies on this subject. Horiuchi et al. found that the proliferative ability of cancer was higher in GSRC patients who were H. pylori-positive than those who were H. pylori negative, suggesting that H. pylori-positive gastric cancers of the SRC subtype progress more rapidly [27]. Although our results do not seem consistent with this previous finding, a direct comparison cannot be made, as survival was not analyzed in the aforementioned study. One possible explanation for our findings is that H. pylori-positive patients, by having earlier symptoms, may undergo earlier testing, resulting in earlier diagnosis and treatment. We believe more studies are needed to better evaluate and understand the impact of H. pylori on GSRC outcomes.
Our study has several limitations. Because the incidence of GSRC is low and our sample size is small, we have limited ability to generalize our findings to other settings. Conclusions regarding the role of risks factors such as H. pylori positivity are also limited by the sample size and the availability of H. pylori infection data in only approximately two-thirds of patients. A related concern is the lack of mortality data for some patients; to approximate survival, we created a composite of time to death and time to hospice initiation, which may systematically underestimate mean survival. In addition, this study could not assess the impact of several key demographic and socioeconomic variables, including family income level, education level, geographic location, duration of residence in the USA, and country of origin, on the development, features, and outcomes of GSRC.
Despite the aforementioned limitations, our study has multiple strengths. Although the sample size is small, it represents one of the largest well-characterized GSRC cohorts in the USA and the only cohort based in a county hospital setting. Our study also highlights important differences in the presentation and prognosis of this malignancy in minority populations and suggests areas in need of future research.
Conclusions
In this first USA-based study to examine the occurrence, features, and outcomes of GSRC at a predominately minority Los Angeles County medical center, we found the mean age at GSRC diagnosis was younger and overall stage was more advanced than that which has been reported in SEER data. Despite presenting with higher overall stage and more lymph node involvement, median survival was similar, suggesting possible protective factors in this population. Additional studies are needed and planned to better understand the epidemiology, pathogenesis, natural history, and treatment outcomes of GSRC and evaluate whether there may be a need for tailored screening programs in this unique and traditionally understudied population.
References
Fitzmaurice C, Allen C, Barber RM, et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 32 cancer groups, 1990 to 2015: a systematic analysis for the global burden of disease study. JAMA Oncol. 2017;3:524–548.
Bertuccio P, Chatenoud L, Levi F, et al. Recent patterns in gastric cancer: a global overview. Int J Cancer. 2009;125:666–673.
Henson DE, Dittus C, Younes M, Nguyen H, Albores-Saavedra J. Differential trends in the intestinal and diffuse types of gastric carcinoma in the United States, 1973–2000: increase in the signet ring cell type. Arch Pathol Lab Med. 2004;128:765–770.
Pernot S, Voron T, Perkins G, Lagorce-Pages C, Berger A, Taieb J. Signet-ring cell carcinoma of the stomach: impact on prognosis and specific therapeutic challenge. World J Gastroenterol. 2015;21:11428–11438.
Pokala SK, Zhang C, Chen Z, et al. Incidence, survival, and predictors of lymph node involvement in early-stage gastric signet ring cell carcinoma in the US. J Gastrointest Surg. 2018;22:569–577.
Aaltonen LA, Hamilton SR, World Health Organization, International Agency for Research on Cancer, eds. Pathology and Genetics of Tumours of the Digestive System. Lyon: IARC Press; 2000.
Antonioli DA, Goldman H. Changes in the location and type of gastric adenocarcinoma. Cancer. 1982;50:775–781.
Lu M, Yang Z, Feng Q, et al. The characteristics and prognostic value of signet ring cell histology in gastric cancer: a retrospective cohort study of 2199 consecutive patients. Medicine (Baltimore). 2016;95:e4052.
Kim DY, Park YK, Joo JK, et al. Clinicopathological characteristics of signet ring cell carcinoma of the stomach. ANZ J Surg. 2004;74:1060–1064.
Zhang M, Zhu G, Zhang H, Gao H, Xue Y. Clinicopathologic features of gastric carcinoma with signet ring cell histology. J Gastrointest Surg. 2010;14:601–606.
Taghavi S, Jayarajan SN, Davey A, Willis AI. Prognostic significance of signet ring gastric cancer. J Clin Oncol. 2012;30:3493–3498.
Los Angeles County Department of Public Health, Office of Health Assessment and Epidemiology. Key Indicators of Health by Service Planning Area. 2017.
Valley Care Community Consortium. Assessing the Community’s Health Needs: A Triennial Report on San Fernando & Santa Clarita Valleys. 2013.
Surveillance, Epidemiology, and End Results (SEER) Program. SEER*Stat Database Details. November 2017 Submission [Internet]. [cited 2018 Jul 21].
Washington K. 7th Edition of the AJCC cancer staging manual: stomach. Ann Surg Oncol. 2010;17:3077–3079.
Li H, Zong Z, Zhou T, et al. Trends of incidence and survival in patients with gastroenteropancreatic signet ring cell carcinoma: an analysis from the surveillance, epidemiology, and end results program. J Gastrointest Oncol. 2019;10:979–988.
Surveillance, Epidemiology, and End Results (SEER) Program. SEER*Stat Database: Incidence—SEER 18 Regs Research Data + Hurricane Katrina Impacted Louisiana Cases, Nov 2017 Sub (1973–2015 varying)—Linked To County Attributes—Total U.S., 1969–2016 Counties, National Cancer Institute, DCCPS, Surveillance Research Program, released April 2018, based on the November 2017 submission.
Jiang H, Zhang H, Tian L, Zhang X, Xue Y. The difference in clinic-pathological features between signet ring cell carcinoma and gastric mucinous adenocarcinoma. Tumor Biol. 2013;34:2625–2631.
Chon HJ, Hyung WJ, Kim C, et al. Differential prognostic implications of gastric signet ring cell carcinoma. Ann Surg. 2017;265:946–953.
Kwon K-J, Shim K-N, Song E-M, et al. Clinicopathological characteristics and prognosis of signet ring cell carcinoma of the stomach. Gastric Cancer. 2014;17:43–53.
Matsui M, Kojima O, Kawakami S, Uehara Y, Takahashi T. The prognosis of patients with gastric cancer possessing sex hormone receptors. Surg Today. 1992;22:421–425.
Postlewait LM, Squires MH 3rd, Kooby DA, et al. The prognostic value of signet-ring cell histology in resected gastric adenocarcinoma. Ann Surg Oncol. 2015;22:S832–S839.
Suh M, Song S, Cho HN, et al. Trends in participation rates for the national cancer screening program in Korea, 2002–2012. Cancer Res Treat.. 2017;49:798–806.
Hamashima C. Cancer screening guidelines and policy making: 15 years of experience in cancer screening guideline development in Japan. Jpn J Clin Oncol. 2018;48:278–286.
Huang J-Q, Sridhar S, Chen Y, Hunt RH. Meta-analysis of the relationship between Helicobacter pylori seropositivity and gastric cancer. Gastroenterology. 1998;114:1169–1179.
Uemura N, Yamaguchi S, Schlemper RJ. Helicobacter pylori infection and the development of gastric cancer. N Engl J Med. 2001;345:784–789.
Horiuchi Y, Fujisaki J, Yamamoto N, et al. Biological behavior of the intramucosal Helicobacter pylori-negative undifferentiated-type early gastric cancer: comparison with Helicobacter pylori-positive early gastric cancer. Gastric Cancer. 2016;19:160–165.
Acknowledgments
The authors would like to thank the late Dr. Jeffrey M. Miller for his assistance with data interpretation and the UCLA Clinical and Translational Science Institute (NIH National Center for Advancing Translational Science Grant UL1TR001881) for providing statistical and analytical support.
Author information
Authors and Affiliations
Contributions
BMF, MP, NP, and JHT designed the study; BMF, MP, NP, and NLO collected data; BMF, MP, NP, and JHT analyzed results; BMF drafted the manuscript and tables; JHT and AFB provided revisions of the manuscript; all approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fung, B.M., Patel, M., Patel, N. et al. Signet Ring Cell Gastric Carcinoma: Clinical Epidemiology and Outcomes in a Predominantly Latino County Hospital Population. Dig Dis Sci 66, 1240–1248 (2021). https://doi.org/10.1007/s10620-020-06341-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-020-06341-z