Abstract
Background
The long-term outcomes of endoscopic resection of superficial submucosal colorectal cancer (CRC) had not been adequately compared with those of surgical resection.
Aims
We aimed to compare the long-term clinical outcomes of endoscopic resection of superficial submucosal CRC to those of surgical resection.
Methods
Submucosal CRC patients with a tumor depth of sm1 or less than 1 mm from the muscularis mucosa were enrolled. Patients with unfavorable histology, such as poorly differentiated cancer or lymphovascular invasion, were excluded. Recurrence-free survival and overall survival were investigated in 87 patients who underwent endoscopic resection and in 171 patients who underwent surgical resection.
Results
The mean ages of the endoscopic and surgical resection groups were 59.7 and 59.8 years, respectively. Hospital stay was shorter in the endoscopic resection group (1.7 ± 1.6 vs. 8.6 ± 3.8 days; p < 0.001). The 3- and 5-year recurrence-free survival rates were 98.7 and 96.7 % in the endoscopic resection group and 98.7 and 97.5 % in the surgical resection group, respectively (p = 0.837). The 3- and 5-year overall survival rates were 100.0 and 95.2 % in the endoscopic resection group and 98.7 and 92.8 % in the surgical resection group, respectively (p = 0.928). Recurred cases showed an unfavorable histology that was overlooked at the time of initial resection.
Conclusions
Long-term outcomes after endoscopic resection of superficial submucosal CRC are comparable to those after surgical resection. Thorough initial histopathological evaluations are needed to guarantee the correct indication for endoscopic resection of submucosal CRC.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Early colorectal cancer (CRC) is defined as a carcinoma whose invasion depth is confined to the mucosa or submucosa, regardless of the presence of lymph node metastasis [1]. While metastasis does not occur in mucosal CRC, the lymph node metastasis rate is reported as 6.6–14.4 % in submucosal CRC [2–6]. The high risk of lymph node metastasis in submucosal CRC is significantly associated with deep submucosal invasion, lymphovascular invasion, tumor budding, and poor differentiation [2, 7, 8]. A recent consensus thus recommends additional surgical treatment after endoscopic resection of submucosal CRC when any of the following unfavorable histological features are present: (1) depth of submucosal invasion ≥1,000 μm; (2) positive lymphovascular invasion; (3) poorly differentiated adenocarcinoma, signet ring cell carcinoma, or mucinous carcinoma; or (4) grade 2/3 budding at the site of deepest invasion [9, 10]. These features are collectively classified as an unfavorable histology that is considered to carry a high risk of lymph node metastasis.
Previous studies have reported a good prognosis after endoscopic resection of submucosal CRC with favorable histology [11]. However, these studies are limited in that relatively small polyps may have been included, which could be resected by a simple snare resection technique; new resection techniques, such as endoscopic submucosal dissection, were not used; there was no appropriate comparison group, such as patients managed with surgical colectomy. Therefore, a re-evaluation of the role of endoscopic resection of submucosal CRC with favorable histology is necessary by comparing its long-term prognosis with that after surgical resection. We thus aimed in our current study to evaluate clinical outcomes, including long-term prognosis, of patients with superficial submucosal CRC with favorable histology who were treated with an endoscopic resection and to directly compare these outcomes with a superficial submucosal CRC population who had received a surgical resection.
Patients and Methods
Patients
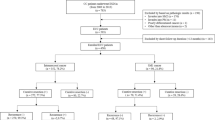
From August 1991 to January 2013, 1,673 submucosal CRC patients were treated in our institution. Of these, we included 275 patients in our current analyses whose invasion depth of primary CRC was the superficial submucosal layer, which was defined as a tumor depth of sm1 or less than 1,000 μm from the muscularis mucosa. Patients with high risk of lymph node metastasis were excluded, specifically those with any of the following unfavorable histological features: (1) poorly differentiated adenocarcinoma; (2) lymphovascular invasion; or (3) tumor budding in the endoscopically or surgically resected specimen. We also excluded those who were treated initially by endoscopic resection and further managed by surgery or radiation therapy due to a positive resection margin at the endoscopically resected specimen or patients’ or physicians’ decision. Negative endoscopic resection margin was defined when the resection margin showed definite absence of tumor regardless of the distance between the deepest tumor front and the resection margin. Those which did not meet these criteria were considered a positive resection margin. Surgical resection meant only radical resection with lymph node dissection, and we also excluded those who were treated using local surgical resection techniques without lymph node dissection, such as transanal excision. A final total of 258 patients with superficial submucosal CRC were recruited (Fig. 1): 87 had been treated by endoscopic resection and 171 using surgical resection.
Inclusion of patients in this study. *High risk means high risk of lymph node metastasis, namely with any of the following unfavorable histological features: (1) poorly differentiated adenocarcinoma; (2) lymphovascular invasion; or (3) tumor budding in the endoscopically or surgically resected specimen
Outcome Analysis
Medical records of the enrolled patients were reviewed for outcome analysis. We investigated the baseline characteristics of these cases such as tumor size and histology. Types of endoscopic resection techniques were reviewed in the endoscopic resection group. Lymph node involvement was assessed in the surgery group. Short-term outcomes including adverse events and hospital stay were investigated in both groups. In the endoscopic resection group, clinical follow-up visits were usually recommended to take place at 1 month, 1 year, and annually thereafter. Colonoscopy follow-up was generally performed at 1 and 3 years in case of en bloc resection. Colonoscopy was recommended at around 6 months and individualized thereafter in the case of piecemeal resection. Imaging studies with abdominopelvic CT scan and chest X-ray with or without chest CT scan were usually recommended annually after endoscopic resection. The follow-up strategy was similar in the surgical resection group. Long-term outcomes such as recurrence-free survival and overall survival were analyzed in both groups. Recurrences were categorized as local and metastatic. Local recurrence was defined as tumors diagnosed histologically at the initial resection margins. Metastatic recurrence was defined as lymph node involvement of cancer or metastasis to extracolonic organs such as liver and lung. We reviewed the pathology slides of initial resection specimens in recurrence cases.
Statistics
Continuous data were compared using a Student’s t test, and categorical data were analyzed using a Pearson Chi-square test or Fisher’s exact test. The Kaplan–Meier method was used to calculate the recurrence-free survival and overall survival rates. A p value less than 0.05 was considered statistically significant. Statistical analysis was performed using SPSS (SPSS 18.0 for Windows; SPSS Inc, Chicago, IL). The protocol of this study was approved by the institutional review board of our institution (IRB No. S2014-0166).
Results
Baseline Characteristics
The male to female ratios were 62:25 and 101:70 in the endoscopic and surgical resection groups, respectively. The mean ages were 59.7 ± 10.5 years (range 38–85 years) and 59.8 ± 10.4 years (range 26–83 years) in the endoscopic and surgical resection groups, respectively. Endoscopic resection was performed as en bloc in 78 patients (89.7 %) and as piecemeal in 9 patients (10.3 %). The types of endoscopic en bloc resection techniques were submucosal saline injection with snaring (n = 63, 72.5 %) and endoscopic submucosal dissection (n = 15, 17.2 %). No visible tumor was left after piecemeal resection. However, argon plasma coagulation was added in four cases based on the endoscopists’ discretion. Tumor location and shape were not significantly different between the two groups (Table 1). However, tumor size was greater in the surgical resection group than the endoscopic resection group (p < 0.01).
Short-Term Outcomes
Hospital stay was longer in the surgical resection group than the endoscopic resection group (8.6 ± 3.8 vs. 1.7 ± 1.6 days, p < 0.01). There were several types of adverse events after the initial treatment in both groups. The frequency of overall adverse events was not different between the two groups (p = 0.47; Table 2).
Long-Term Outcomes
The mean follow-up durations of the endoscopic and surgical resection groups were 45.7 ± 25.3 and 43.9 ± 25.6 months, respectively (p = 0.59). During this period, there were two cases of recurrence (2/87, 2.3 %) in the endoscopic resection group and three cases of recurrence (3/171, 1.8 %) in the surgical resection group. Regarding recurrence cases in the endoscopic resection group, one was local recurrence of adenoma and the other was metastatic recurrence. Regarding recurrence cases in the surgical resection group, one was a local recurrence at the anastomosis site and the other two were metastatic recurrences. The 3- and 5-year recurrence-free survival rates were 98.7 and 96.7 % in the endoscopic resection group and 98.7 and 97.5 % in the surgical resection group, respectively (p = 0.84) (Table 3; Fig. 2).
Six patients died in the endoscopic resection group during the mean follow-up period of 68.1 ± 38.0 months. There was no case of disease-specific mortality. Twelve patients died in the surgical resection group during the mean follow-up of 67.1 ± 41.7 months. There were two cases of disease-specific mortality. The 3- and 5-year overall survival rates were 100.0 and 95.2 % in the endoscopic resection group and 98.7 and 92.8 % in the surgery group, respectively (p = 0.93) (Table 3; Fig. 3).
When we retrospectively reviewed the pathology slides of initial endoscopic and surgical resection specimens of recurrence cases in both groups, we found one or more unfavorable histological features in all cases. For example, lymphovascular invasion of cancer cells was detected in an endoscopically resected specimen in a patient in whom the tumor had been reported to have no lymphovascular invasion at the time of endoscopic resection. A poorly differentiated cancer cluster was detected in the other recurrence patient of the endoscopic resection group in whom the tumor had initially been reported as a well-differentiated adenocarcinoma. Similarly, there were also unfavorable histological features such as tumor budding, a poorly differentiated cancer cluster, and/or lymphovascular invasion in the initial surgical resection specimen of all three recurrence cases after surgical resection. The findings of the five recurrence cases are detailed in Table 4.
Discussion
This comparative study has revealed that there are no significant differences in the long-term prognosis of superficial submucosal CRC cases with favorable histology treated by endoscopic or surgical resection. Hospital stay was shorter, however, in the endoscopic resection group. These results support the current strategy of endoscopic treatment for submucosal CRC [12]. Recurrence rates after endoscopic resection of submucosal CRC with favorable histology have been reported as 0.0–0.8 % in previous studies [11, 13–16]. In our present study, the recurrence rate in the endoscopic resection group was 2.3 % (2/87), which appears to be higher than that previously reported. Although the reason for this difference is not clear, the definition of recurrence may be one explanation. In our present study, we considered a case in which adenoma was detected at the previous endoscopic resection site as a local recurrence, whereas only cancer recurrence was considered a recurrence in most previous studies. If we were to exclude the adenoma recurrence case in our current series, the recurrence rate would be 1.1 % (1/87), which is more similar to the 0.0–0.8 % rates reported in previous studies. Another reason may be the difference in the follow-up period between studies. The cumulative recurrence-free survival rate is the indicator that incorporates the concept of time, which may compensate for differences in the follow-up duration. The 5-year recurrence-free survival was 96.7 % in the endoscopic resection group of our study, which is similar to the 95.9–98 % range of previous reports [13–15].
The most important finding in our current study may be that the long-term prognosis of superficial submucosal CRC with favorable histology was found to be similar between endoscopic resection and surgical resection in terms of recurrence-free survival and overall survival. In addition, the frequency of adverse events was not different between our study groups, while the hospital stay was shorter in the endoscopic resection group. This indicates that endoscopic resection may be recommended as the first choice of treatment for superficial submucosal CRC with favorable histology based on similar long-term oncologic outcomes, similar safety profiles, and shorter hospital stay compared with surgical resection. A recent study comparing endoscopic submucosal dissection with laparoscopic-assisted colorectal surgery for the treatment of low-risk early CRC showed similar findings [17]. In addition, endoscopic resection has another advantage in that it can preserve the colorectum, which may allow functional preservation after treatment. In comparison, surgical resection may be complicated by functional adverse outcomes such as frequent bowel movement, sexual dysfunction, and adhesion [18–20].
We reviewed the pathology slides of initial resected specimens of recurrence cases and found one or more unfavorable histological features in the specimens that had not been considered to have this profile at the time of initial endoscopic and surgical resection. Based on these results, we suggest that a thorough histopathological evaluation should be mandatory if endoscopic resection is to be adopted as the treatment of choice. An incorrect histopathological assessment of an endoscopically resected specimen could result in clinicians neglecting to perform additional surgery that would reduce the risk of lymph node metastasis in the case of unfavorable histology. A lack of surgery after endoscopic resection of superficial submucosal CRC with unfavorable histological features could result in such fatal outcomes as metastatic recurrence, as shown here. For optimal histopathological assessment, pathologists should focus on the detailed examination of unfavorable histological features such as differentiation and tumor budding [21, 22]. In addition, not only routine hematoxylin and eosin staining, but also extra effort, such as special staining for D2-40 and CD31, could be performed to clearly visualize the lymphatics and vessels, thereby more easily detecting lymphovascular invasion of cancer [23–29].
Although the accurate assessment of unfavorable histological features is crucial, the positive predictive value of these features is not satisfactory. In a previous study of 390 patients with submucosal CRC, positive predictive value of lymphatic vessel infiltration for poor clinical outcome such as recurrence was 44.4 %. Those for poor grading of tumor stage and incomplete endoscopic resection were 42.9 and 19.6 %, respectively [30]. Another study showed the incidence of lymph node metastasis was only 1.2 % even in case of submucosal CRC with invasion depth of 1,000 μm or deeper if they did not show poor differentiation, vascular invasion, and high grade tumor budding [31]. Therefore, although precise evaluation of unfavorable histological features is important to identify patients with risk of recurrence, we need further useful predictors for poor clinical outcome to minimize unnecessary additional surgery after endoscopic resection of submucosal CRC.
While the shape and location of tumors were not different between the two groups, the tumor size was larger in the surgical resection group than the endoscopic resection group. Endoscopic resection of large colorectal tumors is technically demanding, especially in the case of en bloc resection, which may have skewed the tumor size toward larger tumors in the surgery group. Recently, endoscopic submucosal dissection has been successfully performed to resect large colorectal tumors in an en bloc fashion [14, 32, 33]. Because accurate histopathological evaluation of resected specimen is difficult and local recurrence is common after piecemeal resection, piecemeal resection may not be a good option for management of superficial submucosal CRC. In our study, 89.7 % of cases were resected as en bloc and only 10.3 % were resected as piecemeal. Thus, we suggest that en bloc resection should be achieved even in large submucosal CRC cases and endoscopic submucosal dissection may be useful in this regard. More widespread use of endoscopic submucosal dissection in the colorectum in the near future may enable the inclusion of larger submucosal CRCs in endoscopic resection group.
There are some limitations to this study. First, the definition of superficial submucosal CRC was not uniform. We adopted two definitions—sm1 or less than 1,000 μm from the muscularis mucosa—because the concept of absolute depth of submucosal invasion of 1,000 μm was introduced around 2004 [4]. Second, although the follow-up strategy was similar in most patients, there were some cases in which surveillance examinations were irregularly performed. Third, the small number of patients and recurred cases limits the power to generalize the results of this study. Finally, the histological review of resected specimen was performed only for recurrence cases, which may have resulted in incorrect inclusion of some cases in our study. Despite these limitations, we believe that our findings are meaningful because they provide evidence for the use of endoscopic resection as the first therapeutic option for superficial submucosal CRC with favorable histology, with long-term outcomes similar to those of surgical resection.
In conclusion, the long-term prognosis after endoscopic resection of superficial submucosal CRC with favorable histology may be comparable to that after surgical resection. Intimate collaboration with the pathology department is required for more accurate initial histopathological evaluation of endoscopically resected specimens to correctly assess the presence of unfavorable histological features and to determine the necessity of additional surgical resection.
References
Kudo S. Endoscopic mucosal resection of flat and depressed types of early colorectal cancer. Endoscopy. 1993;25:455–461.
Ueno H, Mochizuki H, Hashiguchi Y, et al. Risk factors for an adverse outcome in early invasive colorectal carcinoma. Gastroenterology. 2004;127:385–394.
Tominaga K, Nakanishi Y, Nimura S, Yoshimura K, Sakai Y, Shimoda T. Predictive histopathologic factors for lymph node metastasis in patients with nonpedunculated submucosal invasive colorectal carcinoma. Dis Colon Rectum. 2005;48:92–100.
Kitajima K, Fujimori T, Fujii S, et al. Correlations between lymph node metastasis and depth of submucosal invasion in submucosal invasive colorectal carcinoma: a Japanese collaborative study. J Gastroenterol. 2004;39:534–543.
Kikuchi R, Takano M, Takagi K, et al. Management of early invasive colorectal cancer. Risk of recurrence and clinical guidelines. Dis Colon Rectum. 1995;38:1286–1295.
Cooper HS. Surgical pathology of endoscopically removed malignant polyps of the colon and rectum. Am J Surg Pathol. 1983;7:613–623.
Nascimbeni R, Burgart LJ, Nivatvongs S, Larson DR. Risk of lymph node metastasis in T1 carcinoma of the colon and rectum. Dis Colon Rectum. 2002;45:200–206.
Muller S, Chesner IM, Egan MJ, et al. Significance of venous and lymphatic invasion in malignant polyps of the colon and rectum. Gut. 1989;30:1385–1391.
Choi DH, Sohn DK, Chang HJ, Lim SB, Choi HS, Jeong SY. Indications for subsequent surgery after endoscopic resection of submucosally invasive colorectal carcinomas: a prospective cohort study. Dis Colon Rectum. 2009;52:438–445.
Watanabe T, Itabashi M, Shimada Y, et al. Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines 2010 for the treatment of colorectal cancer. Int J Clin Oncol. 2012;17:1–29.
Cooper HS, Deppisch LM, Gourley WK, et al. Endoscopically removed malignant colorectal polyps: clinicopathologic correlations. Gastroenterology. 1995;108:1657–1665.
Participants in the Paris Workshop. The Paris endoscopic classification of superficial neoplastic lesions: esophagus, stomach, and colon: November 30 to December 1, 2002. Gastrointest Endosc. 2003;58:S3–S43.
Yoda Y, Ikematsu H, Matsuda T, et al. A large-scale multicenter study of long-term outcomes after endoscopic resection for submucosal invasive colorectal cancer. Endoscopy. 2013;45:718–724.
Toyonaga T, Man-i M, East JE, et al. 1,635 Endoscopic submucosal dissection cases in the esophagus, stomach, and colorectum: complication rates and long-term outcomes. Surg Endosc. 2013;27:1000–1008.
Ikematsu H, Yoda Y, Matsuda T, et al. Long-term outcomes after resection for submucosal invasive colorectal cancers. Gastroenterology. 2013;144:551–559.
Volk EE, Goldblum JR, Petras RE, Carey WD, Fazio VW. Management and outcome of patients with invasive carcinoma arising in colorectal polyps. Gastroenterology. 1995;109:1801–1807.
Kiriyama S, Saito Y, Yamamoto S, et al. Comparison of endoscopic submucosal dissection with laparoscopic-assisted colorectal surgery for early-stage colorectal cancer: a retrospective analysis. Endoscopy. 2012;44:1024–1030.
Mrak K, Eberl T, Laske A, Jagoditsch M, Fritz J, Tschmelitsch J. Impact of postoperative complications on long-term survival after resection for rectal cancer. Dis Colon Rectum. 2013;56:20–28.
Henneman D, Ten Berge MG, Snijders HS, et al. Safety of elective colorectal cancer surgery: non-surgical complications and colectomies are targets for quality improvement. J Surg Oncol. 2014;109:567–573.
Grosso G, Biondi A, Marventano S, Mistretta A, Calabrese G, Basile F. Major postoperative complications and survival for colon cancer elderly patients. BMC Surg. 2012;12:S20.
Masaki T, Matsuoka H, Sugiyama M, Abe N, Sakamoto A, Atomi Y. Actual number of tumor budding as a new tool for the individualization of treatment of T1 colorectal carcinomas. J Gastroenterol Hepatol. 2006;21:1115–1121.
Smith KJ, Jones PF, Burke DA, Treanor D, Finan PJ, Quirke P. Lymphatic vessel distribution in the mucosa and submucosa and potential implications for T1 colorectal tumors. Dis Colon Rectum. 2011;54:35–40.
Moreira LR, Schenka AA, Latuf-Filho P, et al. Immunohistochemical analysis of vascular density and area in colorectal carcinoma using different markers and comparison with clinicopathologic prognostic factors. Tumour Biol. 2011;32:527–534.
Harris EI, Lewin DN, Wang HL, et al. Lymphovascular invasion in colorectal cancer: an interobserver variability study. Am J Surg Pathol. 2008;32:1816–1821.
Walgenbach-Bruenagel G, Tolba RH, Varnai AD, Bollmann M, Hirner A, Walgenbach KJ. Detection of lymphatic invasion in early stage primary colorectal cancer with the monoclonal antibody D2-40. Eur Surg Res. 2006;38:438–444.
Wada H, Shiozawa M, Sugano N, et al. Lymphatic invasion identified with D2-40 immunostaining as a risk factor of nodal metastasis in T1 colorectal cancer. Int J Clin Oncol. 2013;18:1025–1031.
Barresi V. Reggiani Bonetti L, Vitarelli E, Di Gregorio C, Ponz de Leon M, Barresi G. Immunohistochemical assessment of lymphovascular invasion in stage I colorectal carcinoma: prognostic relevance and correlation with nodal micrometastases. Am J Surg Pathol. 2012;36:66–72.
Suzuki A, Togashi K, Nokubi M, et al. Evaluation of venous invasion by Elastica van Gieson stain and tumor budding predicts local and distant metastases in patients with T1 stage colorectal cancer. Am J Surg Pathol. 2009;33:1601–1607.
Rose AE, Christos PJ, Lackaye D, et al. Clinical relevance of detection of lymphovascular invasion in primary melanoma using endothelial markers D2-40 and CD34. Am J Surg Pathol. 2011;35:1441–1449.
Meining A, von Delius S, Eames TM, Popp B, Seib HJ, Schmitt W. Risk factors for unfavorable outcomes after endoscopic removal of submucosal invasive colorectal tumors. Clin Gastroenterol Hepatol. 2011;9:590–594.
Nakadoi K, Tanaka S, Kanao H. Management of T1 colorectal carcinoma with special reference to criteria for curative endoscopic resection. J Gastroenterol Hepatol. 2012;27:1057–1062.
Byeon JS, Yang DH, Kim KJ, et al. Endoscopic submucosal dissection with or without snaring for colorectal neoplasms. Gastrointest Endosc. 2011;74:1075–1083.
Saito Y, Uraoka T, Yamaguchi Y, et al. A prospective, multicenter study of 1111 colorectal endoscopic submucosal dissections (with video). Gastrointest Endosc. 2010;72:1217–1225.
Conflict of interest
The authors declare no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, JB., Lee, H.S., Lee, H.J. et al. Long-Term Outcomes of Endoscopic Versus Surgical Resection of Superficial Submucosal Colorectal Cancer. Dig Dis Sci 60, 2785–2792 (2015). https://doi.org/10.1007/s10620-015-3530-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-015-3530-2