Abstract
Cancer cells can leave their primary sites and travel through the circulation to distant sites, where they lodge as disseminated cancer cells (DCCs), even during the early and asymptomatic stages of tumor progression. In experimental models and clinical samples, DCCs can be detected in a non-proliferative state, defined as cellular dormancy. This state can persist for extended periods until DCCs reawaken, usually in response to niche-derived reactivation signals. Therefore, their clinical detection in sites like lymph nodes and bone marrow is linked to poor survival. Current cancer therapy designs are based on the biology of the primary tumor and do not target the biology of the dormant DCC population and thus fail to eradicate the initial or subsequent waves of metastasis. In this brief review, we discuss the current methods for detecting DCCs and highlight new strategies that aim to target DCCs that constitute minimal residual disease to reduce or prevent metastasis formation. Furthermore, we present current evidence on the relevance of DCCs derived from early stages of tumor progression in metastatic disease and describe the animal models available for their study. We also discuss our current understanding of the dissemination mechanisms utilized by genetically less- and more-advanced cancer cells, which include the functional analysis of intermediate or hybrid states of epithelial–mesenchymal transition (EMT). Finally, we raise some intriguing questions regarding the clinical impact of studying the crosstalk between evolutionary waves of DCCs and the initiation of metastatic disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
For decades, the hallmarks of cancer scheme [1] has helped the cancer community to describe the multistep development of human tumors including metastatic dissemination. A variable difficult to represent in the hallmarks of cancer’s scheme [1] is time, i.e., the duration of each event (and what controls this duration, particularly after cancer cells have reached distant sites), and when metastatic dissemination occurs. For example, while the development of a primary tumor can take a few years to decades, intravasation and dissemination through the blood system can occur within minutes or hours. Moreover, after disseminated cancer cells (DCCs) lodge at distant sites, they can remain undetected for years or decades by entering a non-proliferative state defined as cellular dormancy (BOX 1 [2,3,4,5,6]). A recent review addressed the definition and mechanisms of cellular dormancy [7]. Eventually, DCCs that exit this dormancy state will form life-threatening metastases. For many years, we have accepted the notion that cancer progression follows a linear model whereby the cumulative acquisition of genetic and epigenetic alterations allows fully malignant cells from advanced stages to spread and metastasize. Over the last 20 years, the concept of parallel progression of primary tumors and metastases has gained traction throughout the scientific community. The parallel progression model posits that cancer cells, presumably with fewer genetic alterations, can disseminate to distant sites before developing a fully malignant phenotype at the primary site. With time, these cells can accumulate independent genetic and epigenetic alterations at distant sites [8], ultimately resulting in metastasis development [9]. In this review, we discuss several lines of evidence that support the early timing of metastatic dissemination. For instance, an experimental breast cancer mouse model revealed that early DCCs (eDCCs) originating from genetically less-advanced cancer cells (e.g., cancer cells found in hyperplastic lesions that resemble human ductal carcinoma in situ (DCIS)) also undergo dormancy and might, under the right conditions, establish metastases with time [10,11,12] (Fig. 1). In clinical settings, 14–31% of patients with DCIS lesions, classified as non-invasive early lesions, have detectable DCCs in the bone marrow compartment [13,14,15,16,17,18], arguing that these pre-malignant cells have the capacity to disseminate. Notably, a small percentage of patients with DCIS die of breast metastases 15–20 years later without experiencing the formation of an invasive carcinoma [19, 20], suggesting that eDCCs derived from the DCIS lesion can lead to metastasis formation after an extended dormancy phase.
Dissemination of cancer cells. Cancer cells disseminate in waves throughout primary tumor evolution, from early stages (early dissemination, green cells) to late stages (late dissemination, red cells), through blood or lymphatic vessels. While in circulation, circulating cancer cells (CCCs) display a spectrum of epithelial (Ep)/mesenchymal (M) phenotypes that are associated with aggressiveness, chemotherapy resistance, and survival, which appears to be maintained by cancer cells after arrival at secondary organs (see section entitled “Partial EMT is linked to dissemination and metastatic colonization”). Generally, DCCs remain as single cells after activating a cellular dormancy program. As shown in breast cancer mouse models, during the early stages of tumor evolution, the lungs of animals harbor a considerable number of early DCCs (eDCCs, green cells in the lungs) characterized by mesenchymal and hybrid phenotypes [12]. Although eDCCs are capable of reactivation, they remain as dormant single cells for prolonged periods [10,11,12], suggesting that the required signals to escape dormancy are not present or not sufficiently abundant at this stage of the disease. Gradually, the primary tumor evolves into a genetically advanced lesion (red primary tumor), from which late DCCs or lDCCs disseminate (red cells in the lungs). Through the circulation, lDCCs reach secondary organs that have already been colonized by eDCCs, where they can potentially interact. Subsequently, the reactivation signals reach a sufficient level to stimulate DCCs to exit from dormancy and development of metastatic tumors. Clinical [19,20,21,22,23] and experimental evidence [10,11,12] suggest that in certain types of tumors, metastases arise from eDCCs (parallel dissemination [9], metastasis with a majority of cells in green). (Color figure online)
An extensive body of literature has dissected (and continues to do so) the mechanisms and signaling pathways relevant to disease progression in cancers of multiple origins, although it mainly focuses on primary sites. Although these investigations have described the milestones needed for the discovery and design of therapies that have significantly improved the life expectancy and quality of life of patients for decades, such as paclitaxel and trastuzumab [24], it is undeniable that current therapies frequently fail to treat metastatic disease. One reason for this is that the design of current treatments does not take into consideration the biology of dormant DCC subpopulations and thus fail to eradicate metastases, which are responsible for most deaths in patients diagnosed with solid tumors [25]. Therefore, it is reasonable to investigate whether targeting DCC subpopulations would yield any advantage in treating metastasis. To accomplish this goal, we need to overcome the limitations from our poor understanding of DCC biology compared to tumor mass biology.
Incorporating these emerging crucial concepts (cancer cell dormancy and the parallel progression model) in the study of metastatic disease gives rise to key questions: Do eDCCs (derived from the early stages of tumor progression) survive long enough at distant sites to be solely responsible for relapses in secondary organs? In patients with invasive stages, do eDCCs interact with more genetically evolved DCCs derived from fully developed primary tumors? Which DCC subpopulation (derived from early lesions or genetically evolved primary tumors) is lethal? These questions are critical, as they could possibly explain and more accurately predict the timing of metastasis. Likewise, the time DCCs spend in a non-proliferative dormant state and/or, for any reason, fail to develop into metastatic lesions provides a significant temporal window for therapeutic intervention that the oncology community has largely underutilized. Here we provide a perspective on how DCC biology, including eDCCs spreading during early stages of tumor progression, may lead to a new understanding of the metastatic process.
Models of cancer dissemination
Early metastatic dissemination
The parallel model of tumor progression was proposed based on previous efforts to measure human cancer growth rates and suggested that metastasis must be initiated before a primary tumor is detectable [26, 27]. The reason behind this idea was that considering similar growth rates for primary tumors and metastases then the size of the metastases at diagnosis were too large if they originated from the late stages of the primary tumor. This model challenges the widely accepted linear model of progression [9, 28] and has brought into question when metastatic dissemination occurs. Historically, it has been inferred from histopathological observations that cancer cells acquire invasive properties at the late stages of tumor development [29]; therefore, dissemination must be restricted to this stage onward. However, compelling evidence favoring early dissemination in humans and mouse models has been presented for breast [10, 11, 13, 17, 19, 30,31,32,33,34], melanoma [8, 35,36,37], colorectal [21, 22], pancreatic [38], lung [39, 40], ovarian [41], and esophageal cancers [42].
For instance, 3.3% of women diagnosed with DCIS, a predominantly non-invasive breast tumor, carry a risk of dying from breast cancer even 15 years after diagnosis, which is greater than that of the general US population [19, 20]. The risk is at least twice as high for women diagnosed before 35 years of age, as well as for black women compared to non-Hispanic white women [19]. Considering that half of the women diagnosed with DCIS who eventually died never developed signs of local recurrence (ipsilateral invasive or contralateral invasive breast cancer), it is probable that their metastatic disease was derived from cancer cells that disseminated from DCIS. Alternatively, it can be argued that clinicians might have missed the invasive front in the DCIS biopsies, given the limited sampling in biopsy procedures. Therefore, improved methods for detecting early spread, including the use of large format sections in histopathology practice, are necessary. As such, new classifications in the DCIS field have been proposed based on an imaging biomarker system (BOX 2) that may help to predict early spread. Several pre-clinical models of breast cancer have supported the idea that cancer cells derived from stages resembling human DCIS could form metastasis [11, 13], and see Table 1 in “Experimental animal models to study early metastatic dissemination” section for more details of the models).
Importantly, the rates of distant metastasis and survival of women diagnosed with early-stage (I or II) breast cancer were similar to those who underwent radical mastectomy (removal of the entire breast) or lumpectomy (removal of the tumor mass with a narrow margin of normal tissue). This observation opens the possibility that early cancer clones contribute to distant metastasis and, therefore, survival. However, the incidence of local tumor recurrence in the ipsilateral breast was greater in the lumpectomy group than in the mastectomy group [64, 65], arguing that different biology governs local and distant recurrence.
Although it may seem counterintuitive that small in situ-staged tumors disseminate, this is strongly supported by extensive literature [8, 10, 11, 13, 19, 21, 22, 32,34,35,35, 38]. For instance, metastatic cancers of unknown primary tumors (or carcinoma of unknown primary (CUP)) may be examples of the early acquisition of a migratory phenotype. Melanoma is the most frequent CUP, and several studies have demonstrated early spread from indolent primary lesions in human samples and in a melanoma mouse model ([8, 35] and see Table 1 in “Experimental animal models to study early metastatic dissemination” section for more details of the model). A recent study described the proposed role of the sodium leak channel non-selective protein (NALCN) as a regulator of cancer cell shedding in different adenocarcinomas as well as normal epithelial cells lacking oncogenic alleles [66]. This suggests the possibility of decoupling dissemination from different stages of tumorigenesis. Whether early cancer lesions could exploit ion channel-dependent mechanisms to spread remains to be evaluated.
Interestingly, patients with DCIS and stage T1 breast cancer have the same number of DCCs in their bone marrow aspirates, suggesting that the DCIS stages are capable of dissemination to the same extent as invasive carcinomas [13]. Moreover, an intriguing study involving patients with invasive breast cancer showed that 10 mm tumors that grow to 90 mm in size exhibit an increase in the rate of distant metastasis ranging from approximately 0.5–26%. In contrast, the contribution of larger tumors (> 60 mm) to distant metastasis plateaued (at approximately 26%), suggesting that the propensity to metastasize and perhaps the capacity of cancer cells to disseminate are more evident at early stages [67]. In support of this evidence, a breast cancer mouse model revealed that small lesions present with a higher dissemination rate to the bone marrow than large primary tumors [10]. This observation was attributed to the gradual increase in the expression of the oncogene HER2, which decreases the progesterone receptor (PR)-dependent early spread signature, favoring tumor growth over dissemination.
Genetic profile comparisons between primary tumors, metastases, and DCCs also support early dissemination. A study involving 743 patients showed that the number of cytokeratin positive (CK+) cells in the bone marrow of patients with early-stage breast cancer was higher than that of control patients with benign breast lesions. In these patients, the presence of CK + cells in the bone marrow was associated with overt metastatic disease and death but not locoregional relapse [32, 68], suggesting different mechanisms for controlling distant and locoregional recurrence. Notably, chromosomal abnormalities were profiled in DCCs from patients at M0 stage, primary tumors, and metastasis, and DCCs exhibited fewer aberrations than primary tumors and metastasis [13]. Similar results were obtained by Schmidt-Kittler et al., who showed that DCCs found in the bone marrow of patients with breast cancer (M0 stage) had fewer genetic alterations than primary cancer cells [34], demonstrating that DCCs seed the bone marrow early in disease progression. Phylogenetic analysis of genomic data comparing normal tissue to primary tumors and metastases for 40 patients with 13 types of cancer, including lung, pancreas, breast, head and neck, and colon cancers, was able to define a tumor evolutionary clock that revealed that metastatic lineages arose early in tumor development [23]. In agreement, dissemination of pancreatic cancer cells from early lesions has been described in a Pdx1-Cre-dependent knock-in mouse model for KrasG12D with a conditional null allele of P53, in presence of a YFP as reporter (also known as KPYC model for KrasG12D; p53fl/+; RosaYFP; Pdx1-Cre) ([38] and see Table 1 in “Experimental animal models to study early metastatic dissemination” section for more details of the model). In here, eDCCs and early circulating clones have been identified in liver and in blood, respectively.
Similar conclusions were drawn by Hu et al. after profiling the evolutionary dynamics of 118 biopsies from 23 patients diagnosed with colorectal cancer with metastasis to the liver or brain. Using a spatial computational model of tumor growth and metastasis that could determine the age of the primary tumor at the time of metastatic dissemination, they showed driver mutations were acquired early, and in 81% of the cases, early DCCs seeded metastases while the primary tumor was still clinically undetected [21]. Although the phylogenetic tree analyses in this study were not constructed using genomic data from single DCCs but rather overt metastasis, the evolutionary pattern and genetic divergence suggest that the identified clones with proven metastatic capacity arose early in the progression of tumor disease. A follow-up study that included patients with colorectal, breast, and lung cancers estimated that metastatic dissemination occurs 2–4 years before the diagnosis of the primary tumors [22]. Lastly, clonal phylogenetic analysis using breast primary tumors and matched lung metastases from MMTV-HER2 females showed that ~ 80% of metastases derived from eDCCs ([10] and see Table 1 in “Experimental animal models to study early metastatic dissemination” section for more details of the model).
Overall, the above-mentioned clinical and experimental literature supports early dissemination events in a variety of cancers and the potential contribution of eDCCs to the metastatic process.
Other models of dissemination in advanced tumors
Other models of dissemination have been proposed using phylogenic analyses. Phylogenetic analysis of paired primary tumors and metastases in patients with established esophageal adenocarcinomas revealed that the spread of malignant clones occurs rapidly after emergence in an evolved primary tumor (diaspora theory) [69], suggesting that certain genetic alterations confer these malignant clones with the ability to rapidly disseminate from the primary site. It would be interesting to determine whether specific genetic alterations occurring during the clinically undetectable early stages of esophageal adenocarcinoma and other diseases facilitate early spread. Along these lines, Hafner et al., were able to show that benign human epidermal tumors acquired the same oncogenic mutations seen in malignant melanomas and independent benign lesions from the same patient seemed to share a clonal origin [70]. Similarly, approximately 70% of analyzed DCIS-invasive breast carcinoma pairs shared the same genetic alterations, with trp53 mutation being one of the most common [71]. Interestingly, a specific somatic trp53 mutation (R245W) facilitated breast cancer formation and early metastatic dissemination in a mouse model, arguing that specific mutations can drive a parallel evolution model of metastasis formation [72].
The Big Bang model of tumor evolution proposed in a colorectal cancer model suggests that intratumor heterogeneity arises at the early stages in the absence of clonal selection by a single expansion of fit subclones which may carry metastatic potential [21, 73]. Interestingly, in the above-mentioned Big Bang model, the early subclones were not dominant at the primary tumor [73]. Likewise, in a renal carcinoma model, subpopulations of cells that were not dominant at the primary tumor spread and subsequently became the main contributors to later relapses [74]. However, in this scenario, epigenetic changes were responsible for cellular competition between subpopulations of cells in the primary sites, which led to the displacement of less-fit clones into the circulation which eventually became responsible for subsequent distant relapses. Further studies may shed light on whether the above-mentioned models of dissemination seen in advanced tumors are possibly even at early stages of tumor progression.
Defining the metastatic potential of disseminated cancer cells
One of the unanswered questions regarding the parallel progression model of tumors and metastasis formulated by Christoph Klein in 2009 [9] pertains to why the size of the primary tumor informs on survival prognosis (e.g. patients with smaller tumors have better survival), considering that the dissemination of future metastatic seeds happens prior to the clinically detectable tumor mass. Perhaps, as proposed by Klein, cumulative signals released from the primary tumor are necessary to favor secondary tumor growth. We would like to propose a complementary hypothesis: in cancers where eDCCs and lDCCs co-exist at distant organs and metastases are founded by eDCCs, we propose that lDCCs activate the latent potential of eDCCs to initiate metastasis. In this scenario, a large primary tumor will spend more time releasing waves of late DCCs than a small tumor, increasing the opportunities for eDCCs reawakening. Thus, the size of the primary tumor could affect relapses, even if eDCCs are the founders of metastasis.
In cancers where eDCCs found metastases, we propose a new perspective on the definition of the metastatic potential of cancer cells as a temporal crosstalk between distinct evolutionary waves of dormant DCCs and their interactions with the niche (Fig. 2). In this context, similar to the pieces of a puzzle, only the precise combination of genetically less-advanced cancer cells (eDCCs) with genetically advanced tumor cells (lDCCs) and the remodeled microenvironment at the right time will induce the exit from dormancy and metastatic outgrowth of eDCCs (Fig. 2). Therefore, studies investigating the mechanisms of interaction/cooperation between eDCCs and lDCCs may shed light on the biology underlying metastasis formation. Further, research efforts to profile eDCCs and late DCCs, and identify patients carrying these distinct subpopulations of DCCs are of utmost importance for effective therapeutic designs.
Metastatic potential of dormant DCCs. DCCs that enter cellular dormancy at secondary sites activate different cellular sub-programs, including quiescence, survival, immune evasion, and reprograming at the metabolic and epigenetic levels (see BOX 1 for more detail). These sub-programs have been predominantly studied in late DCCs (due to the accessibility of late models) and are expected to be similarly necessary in eDCCs released from early stages of the disease. These sub-programs create multiple bottlenecks that aid in the selection of cancer cells with potential metastatic capacity. In some types of cancer, the reactivation of eDCCs will be favored. In this context, the presence of eDCCs (green) and, later on, the arrival of late DCCs (lDCCs) might play critical roles in reprogramming their specific tumor microenvironments (TME; eTME for early DCCs and lTME for late DCCs), including immune and stromal cells and non-cellular components such as the ECM. Eventually, the right combination of eDCCs, lDCCs and TME changes will mediate eDCCs reactivation and formation of clinically detectable metastatic disease. (Color figure online)
Clinical relevance of minimal residual disease
Detection of DCCs and current treatment paradigms for patients carrying DCCs
Detecting single DCC or small clusters in patients with cancer is challenging. Standard pathological techniques for the identification of invasion signs and/or the presence of metastasis in draining lymph nodes often fail to detect single DCCs or small clusters of cancer cells. Serial sectioning in combination with immunohistochemistry and immunofluorescence has improved the detection of these events in different types of cancer including melanoma and head and neck squamous cell carcinoma (HNSCC) [8, 37, 75]. Detection of eDCCs in lymph nodes has been shown in patients with early stage lesions. For instance, immunostaining using antibodies recognizing pan-cytokeratin (including cytokeratin 8, 18, and/or 19 by monoclonal antibodies A45-B/B3 or AE1/AE3) [18, 32, 76] has revealed the presence of single or small clusters of DCCs in sentinel lymph nodes in 1–13% of patients diagnosed with DCIS [14, 15, 77,78,79,80], supporting early dissemination events. Similarly, Werner-Klein et al., showed detection of eDCCs in sentinel lymph nodes of melanoma patients using anti-gp100 staining [8]. The presence of lymph nodes DCCs has been correlated with increased risk for death [37]. Excision of lymph nodes is not always recommended if draining lymph nodes present a normal appearance under noninvasive imaging techniques in early-stage disease [81]. While it is understandable that no clinician wants to subject patients to procedures that do not offer a high benefit/risk ratio [81], the fact that detection of single cancer cells in metastasis-free organs is linked to poor prognosis in cancers such as melanoma [37] raises the question of whether excision of lymph nodes with a normal appearance should be recommended to better assess long-term disease progression.
Similarly, multiple clinical studies have shown that detection of DCCs in the bone marrow has been associated with metastatic relapse in patients diagnosed with cancers of the lungs [39], prostate [82], colon [83], pancreatic [84], esophageal [85], and breast [86]. When performed, and as in the lymph nodes, detection of DCCs in the bone marrow relies on immunostaining using antibodies against markers of epithelial lineage such as cytokeratins (including cytokeratin 7, 8, 18, and/or 19 by antibodies A45-B/B3, AE1/AE3 or 2E11) or sialomucin (E29). Importantly, bone marrow DCCs have been detected in 14–31% of DCIS patients supporting events of early dissemination from DCIS stages [13,14,15,16,17,18]. A large clinical study including 10,307 patients with breast cancer (ranging from T1-4, N1-3, M0) recruited from 10 different centers across Europe and the United States was able to confirm what previous and smaller studies had suggested [87,88,89,90,91,92,93,94,95,96,97,98]: DCC detection in the bone marrow is an independent prognostic marker for overall survival (OS), disease-free survival (DFS), and distal disease-free survival (DDFS) [86]. Despite methodological differences across participating centers (antibody choice and sample preparation) that might have affected detection rates (range from 13 to 48%, with 27% overall detection rate), a total of 2814 patients were DCC positive. With a median follow-up of almost 8 years, the authors concluded that patients with DCC detection in the bone marrow had lower OS and DDFS even at early stages of disease (T1 and N0) and there was no association with locoregional relapse-free survival. Interestingly, the DFS value of DCCs in the bone marrow was lost when patients were treated with neoadjuvant therapy targeting HER2, suggesting HER2 targeting might be controlling DCC capacity to form macrometastasis. Overall, this report suggests that bone marrow aspiration could be collected at time of primary tumor resection, with minimal discomfort to patients while under anesthesia, and be informative to determine which patients need a closer follow-up. In addition, and since a significant fraction of patients with overt tumors and DCC detection did not progress to metastatic disease during the follow up period, it can be speculated that the nature of dormant DCCs at secondary organs is indeed heterogenous. Hence, identifying which cancer cell population is relevant for disease progression will offer a clearer vision for the design of targeted therapies.
Simultaneously with the emergency of new mechanisms describing dissemination, dormancy, and reactivation of DCCs in pre-clinical models, new clinical strategies are being developed to target DCCs, increase survival, and reduce the risk of death in patients with cancer. Bisphosphonates have been extensively studied and were originally reported to prevent bone damage by inhibiting osteoclast-mediated resorption. Zoledronic acid (ZOL), one of the most potent bisphosphonates, has been shown to improve DFS in postmenopausal patients with breast cancer when administered immediately after or following a delay after letrozole administration (ZO-FAST phase III study involving 1065 metastasis-free patients at stages I–III with ER+/PR+breast cancer) [99]. ZOL-treated patients also developed fewer local and distal recurrences, suggesting that DCCs or their microenvironments may be targeted by ZOL. Following this, Arf et al. showed that the proportion of patients with DCC detection after combined chemotherapy and ZOL administration was less than that in patients treated with chemotherapy alone (clinical breast cancer stages II–III; phase 2 randomized study; N = 120). However, this difference was not statistically significant [100]. In another study, Vidula et al. [101] showed that when patients with breast cancer at stages I–III were treated with ZOL after adjuvant therapy (chemotherapy and tamoxifen or an aromatase inhibitor when classified as hormone receptor-positive), a significant reduction in DCCs in the bone marrow at 12 and 24 months post-treatment was observed. Although this study included only 45 patients and did not have a control arm, the authors also reported a DCC clearance rate of approximately 30%. A different study showed that ZOL increased DCC clearance in patients with early breast cancer (pT1-4, N1-2, M0) after 12 months of treatment with a combination of adjuvant therapy and ZOL when compared to the control arm receiving adjuvant therapy alone (N = 96) [102]. Lastly, Banys et al. demonstrated that a significantly smaller number of patients that received ZOL treatment and adjuvant therapy compared to adjuvant therapy alone developed metastatic or recurrent disease during follow-up [103] (NCT00172068). Whether these strategies can target the bulk or specific subsets of DCCs in bone marrow or other secondary organs still needs to be investigated. Moreover, their mechanisms of action remain to be studied; however, some evidence proposes that they could inhibit osteoclastic bone resorption, which reactivates dormant DCCs [104].
Similar to ZOL, denosumab, a monoclonal antibody targeting RANKL, a protein that mediates bone resorption, was evaluated in combination with neoadjuvant or adjuvant chemotherapy to determine whether disease outcomes in women with high-risk early breast cancer improved (Phase 3 D-CARE trial, stages II–III, no DCC status evaluated). No improvement was observed [105]. Researchers have also proposed evaluating whether denosumab has an effect on DFS through DCC targeting/depletion when administered after completion of adjuvant or neoadjuvant therapy, in a strategy parallel to that reported by Vidula et al. [101]. However, the trial was terminated because of low accrual (NCT01545648). Other strategies to target minimal residual disease (MRD) have also been proposed but have not yet reported their findings or failed to obtain funding or patient recruitment. These include the evaluation of the efficacy of trastuzumab for the elimination of HER2+DCCs (NCT01779050; terminated because of loss of funding); the use of gedatolisib (PI3Kα/γ and mTOR inhibitor), abemaciclib (CDK4/6 inhibitor), and hydroxychloroquine (autophagy inhibitor) to target DCCs in the bone marrow (NCT04523857; no results published yet); and even the mobilization of DCCs out of the bone marrow in prostate cancer by treatment with burixafor hydrobromide (CXCR4 inhibitor), which would render them more susceptible to chemotherapy (NCT02478125; terminated because of low accrual). In 2021, a new phase 2 clinical trial was initiated to target bone marrow DCCs using hydroxychloroquine (autophagy inhibitor) or avelumab (PD-1/PD-L1 blocker), with or without palbociclib (CDK4/6 inhibitor), in patients with early-stage ER + breast cancer (NCT04841148). This clinical trial is ongoing.
Another strategy aims to maintain the dormant state of DCCs. In this case, a phase II trial evaluated whether treatment with a combination of 5-azacitidine (5-AZA) and all-trans-retinoic acid (ATRA), FDA-approved drugs for the treatment of myelodysplastic syndrome [106] and acute promyelocytic leukemia [107], respectively, could inactivate prostate cancer cells and maintain the disease in a dormant state. For this purpose, patients with prostate cancer who presented biochemical recurrence (no evidence of cancer on radiographic scans but with rising prostate-specific antigen (PSA) levels) were recruited and treated with 5-AZA and ATRA. To date, no results have been published (NCT03572387).
Finally, DCC detection could be used as a decision-making tool when monitoring disease response to treatment and identifying biomarkers for outcome determination. In this regard, Naume et al. and others identified high-risk patients with early breast cancer and persistent DCCs after the first round of treatment and applied secondary treatments to eliminate all DCCs, emphasizing their potential as surrogate markers for monitoring treatment efficacy [31, 108]. It is notable that although the presence of DCC predicted recurrence in patients with breast cancer over the following five years [68], 60% of DCC-positive patients remained free of disease during that period. Thus, the identification of biomarkers that can describe the progression of MRD is urgently needed. To this end, Borgen et al. stained bone marrow aspirates from 86 DCC-positive patients for NR2F1, a dormancy inducer, and the Ki67 proliferation marker [109]. The findings revealed that NR2F1 serves as a biomarker for predicting long-term recurrence (within five years), whereas Ki67 staining does not. These findings increase the necessity to perform dormancy marker profiling in MRD. Not only cell intrinsic factors in DCCs may explain why some DCCs take longer to reactivate but also environmental signals could determine DCC fate, such as the presence of inhospitable microenvironments or antimetastatic niches [110]. A clear example of an inhospitable microenvironment for DCCs is the skeletal muscle. Different mechanisms can explain why muscle is arguably the most resistant tissue to metastasis. These include the biomechanical destruction of cancer cells [111], the presence of soluble factors such as adenosine receptor agonists [112], the lineage specification of cancer cells [113], and oxidative stress [114] in skeletal muscle microenvironments. Some of these mechanisms may also explain the low metastatic burden in other organs, such as the spleen, thyroid, and yellow bone marrow [110]. Understanding the factors that make the environment conducive as well as the plasticity of DCCs to adapt to such conditions will open new opportunities for targeting DCCs before they exit dormancy and establish overt metastatic tumors. Further, if we assume that eDCCs derived from early stages of tumor progression have fewer genetic changes, it may be possible that strategies based on the underlying biology of genetically advanced invasive tumors may neglect eDCCs; thus, new therapeutic strategies to eradicate eDCCs are needed. To achieve this, we first need to characterize these patient-derived eDCCs and later be able to identify patients carrying eDCCs before they develop metastasis. These characterization efforts, which have recently started in animal models [12], will translate into the identification of the specific biomarkers that we urgently need to stratify patients at risk and to potentially, develop new therapies.
Detection of circulating cancer cells in early disease
One of the intermediate steps of the metastatic cascade involves the ability of tumor cells to enter and survive in the circulation as circulating cancer cells (CCCs) and extravasate into the surrounding tissues. It is reasonable to assume that CCCs detected in patients diagnosed with primary tumors and metastases are derived from both the primary tumor and metastases, and if surgery has been performed on the primary tumor, then CCCs are derived from metastases. During remission, the detection of CCCs may represent a clinically undetectable recurrence [115]. Interestingly, CCCs have also been detected in patients with benign and early disease, such as pancreatic cystic lesions [116], intestinal polyps [117,118,119], chronic lung diseases [120], and DCIS [121]. However, the metastatic potential of these early CCCs is largely unknown.
Pancreatic ductal adenocarcinoma (PDAC) is associated with high mortality and a poor prognosis [122]. It is generally asymptomatic in its early stages, limiting the efficacy of current therapies. Furthermore, the diagnosis of early disease is rare in the general population and relies heavily on imaging techniques used during the evaluation of unrelated conditions [123]. Thus, the use of CCC detection has been proposed as a tool for screening and early detection of PDAC. Using GEDI (Geometrically Enhanced Differential Immunocapture; a microfluidic device in conjunction with DAPI, CD45, CK19, and Pdx-1 staining) [124], circulating pancreatic cells were detected in 33% of patients diagnosed as cancer-free; however, these patients had intraductal papillary mucinous neoplasm (IPMN), a type of cystic lesion [116] considered a precursor of PDAC [125]. Later, independent groups identified circulating pancreatic cells in patients that were undergoing surgical resection of precursor cystic lesions (mainly IPMN) in up to 88% of cases evaluated [126,127,128] using previously validated CCC detection systems, including ScreenCell® [129], ISET© [130], and CTC-iChip [131]. Interestingly, similar detection rates of CCCs were observed in benign, premalignant, and malignant lesions, suggesting that early evolved cells efficiently disseminated from the primary site [126]. Whether CCCs from benign and/or premalignant pancreatic lesions can be used as a risk factor for future distant relapses remains to be determined.
Using previously validated CCC detection systems, including EPISPOT™ [132] (CK19 +in CD45-depleted cells) and CellSearch [133] (CK+after epithelial cell adhesion molecule (EpCAM) enrichment), positive events that met the criteria for “cancer cells” were detected in a study of 53 patients diagnosed with benign colon diseases (diverticulosis, benign polyps, and Crohn’s disease, among others). Although the metastatic potential of these cells or tumor development in these patients did not occur within three years of follow-up [118], this does not rule these cells out of possessing seeding and/or metastatic potential. In fact, these benign diseases are a risk factor for the development of colorectal cancer [134]. An independent study detected circulating epithelial cells in 33% of preoperative patients with colorectal polyps (compared to only 8% shown in a previous study [118]) by using a more sensitive CMx platform (EpCAM-based capture system and subsequent corroboration by staining against CK20) [119]. Thus, intestinal cells from the earliest type of benign lesion are also capable of dissemination, and whether they can predict the risk of metastatic disease needs to be evaluated.
Although challenging, some clinical trials have shown not only the presence of CCCs in early disease but also their prognostic value for developing tumors. For instance, patients diagnosed with chronic obstructive pulmonary disease (COPD), a risk factor for lung cancer, were monitored for the presence of circulating cells with malignant cytopathologic features. In 3% (5 of 168) of the patients, CCCs were identified and isolated using ISET technology, corroborated by May Grunwald Giemsa cytological analysis and immunostaining for pan-cytokeratin and vimentin. Patients with detectable CCCs did not present lung nodules at the time of COPD diagnosis. However, these were detected at follow-up visits, and all five patients developed tumors within 1–4 years (80% invasive carcinomas and 20% squamous cell carcinoma), demonstrating the predictive value of CCCs in early non-small cell lung carcinoma (NSCLC) [120]. Since NSCLC presents metastatic lineages arising early in tumor development [23], one could ask whether the CCCs detected during COPD could seed future relapses.
Mechanisms of cancer cell dissemination
Partial EMT is linked to dissemination and metastatic colonization
It has been proposed that epithelial-mesenchymal transition (EMT) plays an essential role in metastatic dissemination. The expansion of our understanding of EMT markers and transcriptional profiling led us to arrive at the idea that EMT is not dichotomous but rather a spectrum of phenotypes between epithelial and mesenchymal states that cancer cells exploit for their survival, migration, metastatic capacity, and even resistance to therapy. Although the definition, mechanisms governing these hybrid states, and their relevance have not been explored in detail, some studies have begun to show the association between hybrid states and poorer prognosis [135, 136] and enhanced metastatic capacity [136,137,138].
Transcriptomic analysis of 7180 primary tumors of epithelial origin spanning 25 cancer types from the harmonized version of TCGA using the TCGAbiolinks R package (pan-cancer analysis) reconstructed EMT pseudotime trajectories that suggested three macro-states: epithelial, hybrid, and mesenchymal [139]. The hybrid state was surprisingly frequent among the samples (39%), stable over time, associated with aneuploidy, which is correlated with a poor prognosis [140, 141], and associated with a worse overall survival outcome. Although it is probable that this hybrid state exists within a spectrum of possible states, its stability suggests that this state is energetically favorable, and because increased evidence suggests that hybrid EMT is associated with stemness, chemoresistance, immune evasion, and metastasis, this hybrid state may be relevant in cancer disease [135, 136, 142, 143].
Numerous studies focus on the protein E-cadherin, an adhesion protein in the calcium adhesion superfamily that is mainly expressed in epithelial cells and maintains cell adhesion and epithelial structural integrity. Its loss is a key marker of EMT, which has been linked to an increased risk for metastasis [144,145,146]. However, emerging data on the E-cadherin functions suggests a context-dependent role (i.e. different stages of metastasis, different cancer types) and its downregulation does not always imply upregulation of all well-known EMT mesenchymal traits but perhaps the development of hybrid EMT states. For instance, downregulation of E-cadherin has been linked with a lower metastatic burden and the induction of dormancy. In this regard, Aouad et al. recently demonstrated that E-cadherin reduction in an ER + breast cancer model (MCF7 cells) induced slow proliferation and apoptosis at the primary site and reduced the lung metastatic burden via dormancy induction [147]. Interestingly, ectopic expression of E-cadherin in dormant DCCs was sufficient to drive DCCs out of dormancy. In this study, the reduction of E-cadherin increased zinc finger E-box-binding homeobox 1 (ZEB1) transcripts, whereas the expression of other EMT markers was unaffected. An elegant study using the luminal invasive ductal carcinoma MMTV-PyMT mouse model demonstrated that loss of E-cadherin inhibited metastatic burden [148]. Transcriptional analysis using RNAseq in E-Cadherin + vs. E-cadherin- cells showed no significant changes in the expression of canonical EMT transcripts, suggesting that E-cadherin function does not require a complete loss of epithelial or a complete gain of mesenchymal traits in this model. Similarly, pancreatic dormant DCCs found in human and mouse liver sections became negative for E-cadherin without upregulating EMT markers (Desmin, alpha smooth muscle actin (aSMA), Snail1, or Slug) [53].
Additional studies provide evidence of the enhanced metastatic capacity of hybrid EMT states. For instance, Lui et al. showed that ex vivo cultured CCCs with a hybrid phenotype had a higher metastatic capacity than epithelial or mesenchymal-type CCCs [143]. Interestingly, different grades of hybrid phenotype were identified, with CCCs that were more epithelial than mesenchymal type (higher E-cadherin and EpCAM protein levels) having higher metastatic potential. Using an ER-/PR- breast cancer cell line, Brown et al. [136] isolated three hybrid EMT clones and demonstrated that these intermediate clones had higher tumor-initiating capacity than fully mesenchymal phenotypes. Notably, the hybrid and mesenchymal EMT clones had a latency in growth when compared to parental cells, and this lag was more evident in mesenchymal clones with very low E-cadherin levels. Moreover, Pastushenko et al. [138] showed that hybrid EMT subpopulations (with higher E-cadherin messenger levels) derived from skin tumors had higher metastatic potential than more mesenchymal subpopulations (with lower E-cadherin messenger levels). In line with these results, Cui et al. recently showed that loss of MLL3, a histone methyltransferase, increases metastatic colonization and enriches metastasis with hybrid Vimentin+/E-cadherin + cells when compared to MLL3 wild-type mesenchymal (Vimentin+/E-cadherin-) breast cancer cells [137]. Thus, one hypothesis could be that mesenchymal and hybrid EMT DCCs with low levels of E-cadherin may represent clones more susceptible to a longer dormancy phase, whereas hybrid EMT clones with high E-cadherin levels may immediately (or after a short dormancy period) form metastases. This may not be the case in PanIN lesions where E-cadherin-negative PanIN cells injected into the pancreata of NOD/SCID mice formed tumors faster (2 months post-implantation) than E-cadherin-positive cells (4 months) [38]. Further, PDAC metastatic clones having a more mesenchymal-like hybrid phenotype (lower E-cadherin levels) were more metastatic and disseminated more than epithelial-like hybrid-EMT (higher E-cadherin levels) [149]. Worth mentioning is that PDAC hybrid EMT clones were still more metastatic than the EMT extremes.
Our work in early premalignant lesions from the MMTV-HER2 animal model (see description of the model below and Table 1) described a partial/hybrid EMT at the primary site, downstream of HER2, that mediates in vivo dissemination to the lungs (determined by intravital imaging and quantification of DCCs in the lung at the single cell stage 1–2 weeks post orthotopic injection) [150]. We showed that HER2 downregulated the expression of the NR2F1 nuclear receptor via p38, and loss of NR2F1 triggered a partial/hybrid EMT, characterized by the maintenance of luminal epithelial markers, such as cytokeratin 18, a limited suppression of Gata3 mRNA expression, and downregulation of E-cadherin, while expression of CK14, TWIST, and PRRX1 was induced. No changes were observed in other EMT markers such as VIMENTIN, SNAIL1, and AXIN2. Once in the lungs, these HER2 + eDCCs underwent a period of dormancy (approximately 90% Ki67 negative) [11, 12], and they were enriched in mesenchymal- and hybrid-like EMT signatures, with low E-cadherin mRNA levels [12], nearly undetectable by immunofluorescence (IF) staining [11] (Fig. 1). Interestingly, these HER2 + eDCCs eventually reactivated and were responsible for ~ 80% of lung metastasis [10]. Based on the above-mentioned studies on breast cancer, we can postulate that hybrid dormant HER2 + eDCCs are more sensitive to reactivation signals than the mesenchymal-like type and, therefore, are the first type of eDCCs to transition back to the epithelial phenotype (acquiring high E-cadherin levels), exit dormancy, start proliferating, and form metastases. Undoubtedly, this hypothesis needs to be validated using in vitro models as well as in vivo models such as the ones described below and in Table 1.
Overall, the above studies suggest that in certain types of tumors, cancer cells with hybrid EMT may be clinically more relevant than full mesenchymal states and may rapidly develop into deadly metastases. Further investigations are required to determine the role of hybrid EMT states in all types of dormant DCCs.
Experimental animal models to study early metastatic dissemination
In vitro studies have been essential to address some of the biology of the early dissemination process. For example, 3D Matrigel cultures allows to address questions on motility, invasion, EMT, and co-cultures as we shown in [10,11,12, 150]; scaffolds that mimic different microenvironments are used to explore the behavior of single eDCCs [151,152,153,154]; microfluidics devices to mimic intra- and extravasation processes could also address fundamental questions on eDCCs behavior [155, 156]; and computational models can model the process of metastasis (including tumor cell-bloodstream dynamics) and interactions of tumor cell clones with the secondary microenvironment [157]. However, these methods isolate different aspects of a multi-step process that is difficult to integrate in the absence of in vivo models. Few mouse models have been described for the study of early dissemination and eDCC biology. In the following section, we aim to summarize the details of the transgenic models, the time of cancer cell dissemination, the sites of early dissemination, and the potential role of the immune system in dissemination and dormancy (Table 1). Ultimately, the animal model provides the opportunity to take into account all and every single step of the metastatic process (dissemination, EMT features, survival at new niches, dormancy state, reactivation and metastasis formation) and in a way that closely resembles the spontaneous biology of DCCs.
MMTV-HER2 breast cancer model
Dissemination from early cancer lesions in the absence of invasive histopathology features has been more commonly studied in MMTV-HER2 animal models. These animals develop spontaneous mammary tumors and metastases after overexpression of HER2 (neu/ErbB2) in the mammary gland epithelia, making them a suitable model to study HER2-driven tumorigenesis. The first transgenic mice were developed in the Swiss-Webster background by Muller and colleagues in 1988 [158,159,]. The MMTV/c-neu mouse model carries an activated rat HER2 oncogene with a V664E point mutation in the transmembrane domain that favors the aggregation and activation of the receptor in the absence of a ligand [159,160,161]. In the BALB/c background, activated HER2 is first detected at the onset of puberty (3–4 weeks of age). Hyperplastic lesions are found around 7–9 weeks of age [162], while palpable in situ carcinomas are detected between 14 and 18 weeks of age. Five to ten weeks later, the tumors become invasive, and metastatic foci are detectable macroscopically [13]. In this model, hyperplastic cells express ER/PR, which are later lost at advanced stages of tumor progression [10]. In 1992, the same group generated a second model in the FVB background carrying the wild-type HER2 allele under the MMTV promoter [163]. These animals are also characterized by the development of focal mammary tumors, albeit with a longer latency than in the activated HER2 model. Furthermore, these animals develop spontaneous mammary tumors around 30 weeks of age [163], which progress from hyperplastic lesions that arise around 16–18 weeks of age [11, 12]. Both models offer an accessible time window for the study of early dissemination and therefore, the biology of DCCs in dormant and reactivated states.
In both models (wild-type and activated HER2), at an early age, a subpopulation of HER2 + early cancer cells characterized by low levels of phospho-p38 and its targets p-ATF2, p-MK2, and p-Hsp27, low levels of E-cadherin, high levels of Twist, and a progesterone (PR) signature showed enhanced invasive activity and dissemination capacity when compared to normal mammary epithelium [10, 11, 164]. These early cancer cells activated a program that triggered a partial EMT transition (without complete loss of the epithelial phenotype) that was dependent on HER2, Wnt, and PR signaling [10, 11]. A recent study revealed that the orphan nuclear receptor NR2F1 was negatively regulated by HER2 and positively regulated by p38alpha, and its expression prevented the invasion and dissemination of MMTV-HER2 early cancer cells [150].
The groups of Christoph Klein and Julio Aguirre-Ghiso have shown that HER2 + early cancer cells have metastatic potential [10, 11, 13]. This was achieved by performing several experiments that measured lung metastasis, such as: (a) surgery of the entire milk line after detecting in situ carcinoma in transgenic females [13]; (b) in wild-type females that received temporary orthotopic grafts of mammary glands from donors with in situ carcinoma [13]; (c) injection of early cancer cells into the mammary fat pad of syngeneic and/or nude mice [10,11,12]; and (d) injection of early cancer cells into the tail vein of animals [11, 12].
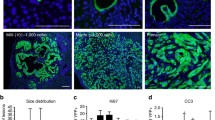
In the HER2 mouse models, eDCCs can be detected by HER2 + staining in the lungs and bone marrow soon after the activation of HER2 during stages of hyperplastic lesions [10, 11, 13]. Single-cell comparative genomic hybridization of the bone marrow eDCCs showed chromosomal aberrations which traced them back to the early hyperplastic lesion [13]. Once in the lungs, eDCCs remained dormant (~ 90% Ki67 negative) for approximately 15–18 weeks and exhibited a hybrid and mesenchymal EMT phenotype [11, 12]. Importantly, genetic profiling using comparative genome hybridization of primary tumors against matched metastases suggested that up to 80% of the metastatic burden in these MMTV-HER2 animals was derived from eDCCs [10]. This result suggests that eDCCs are the main contributors to lung metastases in this model.
Early dissemination events in the MMTV-HER2 animal model also required the participation of macrophages, as their depletion, mediated by CSF1R blockage only during the hyperplastic lesion stage, reduced dissemination, and metastatic burden [33]. Macrophages, normally found in the stroma around the mammary ducts, entered the ductal epithelial layer in early lesions when the epithelium was characterized by hyperplasia and mammary intraepithelial neoplasia [33]. This effect was mediated by CCL2, a potent macrophage chemoattractant produced by HER2 + early cancer cells, among other cells. In response to CCL2 signaling, the presence of macrophages disrupted E-cadherin expression in the surrounding epithelial cells through macrophage-dependent Wnt1 signaling. Furthermore, HER2 + early cancer cells efficiently formed tumor microenvironment of metastasis (TMEM) structures that have been shown to serve as portals for intravasation and have clinical prognostic value in human disease [44, 165]. The detection of intra-lesion macrophages was confirmed in a small cohort of patients with DCIS, and this had no correlation with HER2 status [33].
In summary, these studies have demonstrated HER2 + eDCCs as a new source of metastasis.
MMTV-PyMT breast cancer model
Early dissemination has also been described in an MMTV-PyMT animal model. This animal model is characterized by the expression of the polyoma virus middle T antigen (PyMT) under the mammary tumor virus LTR and is therefore restricted to the mammary gland [45]. Rapid multifocal mammary adenocarcinomas spontaneously develop in these animals and are commonly detected at approximately 14 weeks of age. Hyperplastic early lesions can be detected as early as 4 weeks of age [46]. Although PyMT is not expressed in humans, its oncogenic capacity activates a range of signaling pathways, including Src, Ras, and c-Myc, which are commonly altered in human diseases. In this model, tumors arise from the luminal cells of the mammary gland and resemble human disease as they lose ER and PR, overexpress ErbB2 [46], and exhibit similarity to luminal B tumors according to genetic profiling [47]. Remarkably, despite their rapid progression, eDCCs can be detected by colony formation assays from digested lungs as early as 4 weeks of age, when the histological evaluation of the mammary tree appears normal [48]. Unequivocal identification of eDCCs was demonstrated by Christoph Klein in 2008 by IHC using a GP11 antibody recognizing both CK8 and CK18 staining in bone marrow cytospins and by histology in lung sections [13]. These eDCCs are associated with stem cell markers, which could explain their tumorigenic capacity [48].
Mouse intraductal (MIND) DCIS xenograft models
The intraductal human-in-mouse transplantation model allows the in vivo study of DCIS malignancy. Human DCIS cell lines, primary DCIS, and atypical hyperplasia cultures derived from patient’s biopsies can be implanted in the mammary ducts of immunocompromised mice [50, 51]. Primary DCIS and atypical hyperplasia cells derived from patient’s biopsies recapitulate, in the MIND xenografts, the expression patterns of human cytokeratins, ER, and HER2, which are specific to human cells, pathology, and the heterogeneity of human DCIS disease. The advantage of MIND xenografts of primary human DCIS is that they do not progress to invasive carcinoma for the first 8 weeks, which offers a time window to specifically study DCIS biology. Potentially, the use of MIND DCIS xenografts might provide insight into which patients will progress to invasive breast cancer and reduce overtreatment of patients who are not at risk of progression. However, because the animals are immunocompromised, early lesions and invasive carcinomas will be developed in an environment that will not fully recapitulate human tumor biology or pharmacodynamics, which needs to be carefully considered. Further, this model is compatible with the use of the human cell line SUM-225, which has been shown to resemble HER2 + DCIS lesions, and the MCF10DCIS.COM line which establish basal-like DCIS lesions. The SUM-225 HER2 + DCIS lesions invade the myoepithelial layer 14 weeks after transplantation and the MCF10DCIS.COM DCIS lesions become invasive after 10 weeks [52]. In both cases, a significant time window for the study of eDCCs is available. The molecular characterization of the subpopulations of cells in DCIS lesions that enter the circulation, reach distant sites, and eventually form metastases (eDCCs responsible for DCIS breast cancer mortality [19]) is of utmost importance for the prevention of metastasis not only in patients with DCIS but also in all types of cancers with early spread.
KPC mouse model
The dissemination of pancreatic cancer cells from intraepithelial neoplasia has been described in a Pdx1-Cre-dependent knock-in mouse model for KrasG12D with a conditional null allele of P53, in presence of a YFP as reporter (also known as KPYC for KrasG12D; p53fl/+; RosaYFP; Pdx1-Cre) [38]. The advantage of these animals is that they exhibit histological and molecular resemblances to human disease [38]: they developed pancreatic intraepithelial neoplasia (PanIN) lesions at around 8–10 weeks of age with detection of CCCs in the blood and eDCCs in the liver, before evidence of carcinomas at the primary site. Detection of PDAC (pancreatic ductal adenocarcinoma) occurs at 16 weeks. In this mouse model, inflammation was shown to contribute to the EMT phenotype and early dissemination. The metastatic potential of eDCCs in the KPYC mouse model still needs to be demonstrated, although clinical evidence strongly suggests the relevance of these events: most patients present metastatic disease at time of diagnosis [166] with metastatic lesions having low proliferation rate compared to the primary site [167, 168], but similar sizes at the time of diagnosis, suggestive of parallel evolution and early dissemination. The KPC animal model, carrying knock-in expression of KRASG12D in combination with constitutive p53 deletion mediated by Cre recombinase (under Pdx1, P48, or Nestin promoters), presents PanIN lesions at a young age (8–10 weeks of age), with rapid development of PDAC (10–30 weeks) that requires monitoring by non-invasive imaging techniques or manual palpation [54]. Metastasis to multiple organs are developed with variable latency.
A slower progressor model is the KC model, a knock-in expression of KRAS (KrasG12D) by Cre recombinase under the pancreatic promoter P48, is characterized by rapid development of PanIN lesions in the first weeks of life that resemble human PanINs I–III, with longer latency before PDAC is detected at around 30 weeks of age [55, 61]. The disadvantage of this model is that KRAS mutation alone is rarely found in human disease.
RET.ADD melanoma model
A spontaneous uveal melanoma mouse model called RET. AAD [62] also exhibits early metastatic dissemination [35]. In this model, an activated human RET oncogene is expressed in melanocytes under the mouse metallothionein (MT)-I promoter/enhancer that triggers melanosis and melanocytic tumors [169], in presence of the chimeric MHC class I molecule AAD. The RET.AAD animal model recapitulates melanoma progression, including metastatic stages. In this model, eDCCs are observed in the lung of 6–7-week-old C57BL/6 mice by immunohistochemistry (IHC) targeting the melanocyte marker S100B and in visceral organs as early as 3 weeks of age when determined by qRT-PCR detection of the dopachrome tautomerase (Dct) gene. The dormancy periods between DCC arrival and overt metastasis vary depending on the secondary organ, with metastasis in the lungs becoming evident at 1 year of age on average, offering one of the main advantages of this model for the study of early dissemination and dormancy. Genome wide SNP profiling comparing somatic mutations in primary and metastatic tumors from the same animals suggests that dissemination of tumor cells occurs during the hyperplastic stages [35]. Histopathological analysis of the lesions in the choroids of young animals (2–4 weeks of age) revealed only hyperplastic lesions. Interestingly, small nodules were found in the sclera at 2 weeks of age. Therefore, metastasis latency in the sclera is very brief (~ 2 weeks), but more importantly, cells that locally migrated to the sclera were only derived from hyperplastic lesions found in the choroids. When the RET.ADD model is generated in Balb/c mice there is a more pronounced latency for primary and metastatic disease [63], with DCC presence in secondary organs expected to be delayed as well. Despite it resemblance to human disease, the activated version of the RET oncogene in this animal model is not found in humans, which may limit the translatability of mechanisms and biomarkers of dormant melanoma eDCCs that could be identified in the future. The RET.AAD model also expresses chimeric MHC AAD (alpha1-alpha2 domains of the HLA-A2 linked to the alpha3 domain H2-Dd) that allows the monitoring of CD8 + T cells. The role of CD8 + T cells in maintaining dormancy of eDCCs in visceral and lung metastasis formation has been proposed, as depletion of CD8 + T cells in 6-week-old RET.ADD mice (by administering neutralizing antibodies) led to rapid development of micrometastases [35], arguing that CD8 + T cell depletion may facilitate the exit of eDCCs from dormancy.
Concluding remarks
Secondary organs might accumulate evolutionarily distinct types of DCCs, starting at the early and asymptomatic stages of tumor progression. These multiple waves of DCCs can remain dormant for decades or even the lifetime of patients (e.g. patients diagnosed with only non-invasive early diseases such as DCIS). Considering a niche where different waves of DCCs co-exist, several questions arise. For example, what type of DCC, early or late (or interactions/cooperation between them) initiate the first escape from dormancy. Further, if eDCCs are the first to reactivate, do they switch to a transcriptomic/genomic profile that resemble late DCCs (more epithelial-like/more genetically evolved)? Regarding the remodeling of the microenvironment, which types of DCCs (early or late) regulate the switch from an evasive immune niche to a pro-tumorigenic niche; which types of DCCs induce a pro-metastatic extracellular matrix remodeling. Exploring the use of in vitro, in vivo, and computational models that can recapitulate the behavior of DCC waves—their migration from their primary sites to distant sites, acquisition of different EMT phenotypes in this process, and communication with new niches and each other to survive, remain dormant, and ultimately reawaken—will aid in the design of new therapies to prevent deadly metastatic disease. It is also essential to factor in the contribution of the tumor microenvironment, including its organ- and patient-based diversity, together with the effect of current standard-of-care treatments on the biology of dormant DCCs. For example, different organs may induce different metabolic fitness in DCCs [170] (BOX 1). Further, targeted therapy may exacerbate metabolic vulnerabilities in DCCs, as shown for loco-regional persistent breast cancer cells [171]. Lastly, eDCCs might manifest distinct responses to treatment compared to genetically advanced DCCs. Because our knowledge of the biology, origins, and heterogeneity of DCCs is still limited for most cancer types, research efforts to profile eDCCs and lDCCs are of utmost importance for identifying patients at risk of relapse, reducing overtreatment and designing effective therapeutic strategies.
References
Hanahan D (2022) Hallmarks of cancer: new dimensions. Cancer Discov 12(1):31–46. https://doi.org/10.1158/2159-8290.CD-21-1059
Sosa MS, Bragado P, Aguirre-Ghiso JA (2014) Mechanisms of disseminated cancer cell dormancy: an awakening field. Nat Rev Cancer 14 9:611–622. https://doi.org/10.1038/nrc3793
Carlini MJ, Shrivastava N, Sosa MS (2018) Epigenetic and pluripotency aspects of disseminated Cancer cells during minimal residual disease. Adv Exp Med Biol 1100:1–18. https://doi.org/10.1007/978-3-319-97746-1_1
Giancotti FG (2013) Mechanisms governing metastatic dormancy and reactivation. Cell 155(4):750–764. https://doi.org/10.1016/j.cell.2013.10.029
Ghajar CM (2015) Metastasis prevention by targeting the dormant niche. Nat Rev Cancer 15(4):238–247. https://doi.org/10.1038/nrc3910
Dalla E et al (2023) Dormancy in breast cancer. Cold Spring Harb Perspect Med https://doi.org/10.1101/cshperspect.a041331
Risson E et al (2020) The current paradigm and challenges ahead for the dormancy of disseminated tumor cells. Nat Cancer 1(7):672–680. https://doi.org/10.1038/s43018-020-0088-5
Werner-Klein M et al (2018) Genetic alterations driving metastatic colony formation are acquired outside of the primary tumour in melanoma. Nat Commun 9(1):595. https://doi.org/10.1038/s41467-017-02674-y
Klein CA (2009) Parallel progression of primary tumours and metastases. Nat Rev Cancer 9(4):302–312. https://doi.org/10.1038/nrc2627
Hosseini H et al (2016) Early dissemination seeds metastasis in breast cancer. Nature 540(7634):552–558. https://doi.org/10.1038/nature20785
Harper KL et al (2016) Mechanism of early dissemination and metastasis in Her2(+) mammary cancer. Nature 540(7634):588–592. https://doi.org/10.1038/nature20609
Nobre AR et al (2022) ZFP281 drives a mesenchymal-like dormancy program in early disseminated Breast cancer cells that prevents metastatic outgrowth in the lung. Nat Cancer https://doi.org/10.1038/s43018-022-00424-8
Husemann Y et al (2008) Systemic spread is an early step in breast cancer. Cancer Cell 13:158–68. https://doi.org/10.1016/j.ccr.2007.12.003
Banys M et al (2012) Hematogenous and lymphatic Tumor cell dissemination may be detected in patients diagnosed with ductal carcinoma in situ of the breast. Breast Cancer Res Treat 131(3):801–808. https://doi.org/10.1007/s10549-011-1478-2
Banys M et al (2014) Detection and clinical relevance of hematogenous tumor cell dissemination in patients with ductal carcinoma in situ. Breast Cancer Res Treat 144(3):531–538. https://doi.org/10.1007/s10549-014-2898-6
Gruber IV et al (2016) Relationship between hematogenous tumor cell dissemination and cellular immunity in DCIS patients. Anticancer Res 36(5):2345–2351
Sanger N et al (2011) Disseminated tumor cells in the bone marrow of patients with ductal carcinoma in situ. Int J Cancer 129(10):2522–2526. https://doi.org/10.1002/ijc.25895
Sanger N et al (2014) Molecular markers as prognostic factors in DCIS and small invasive breast cancers. Geburtshilfe Frauenheilkd 74(11):1016–1022. https://doi.org/10.1055/s-0034-1383033
Narod SA et al (2015) Breast cancer mortality after a diagnosis of ductal carcinoma in situ. JAMA Oncol 1(7):888–896. https://doi.org/10.1001/jamaoncol.2015.2510
Giannakeas V, Sopik V, Narod SA (2020) Association of a diagnosis of ductal carcinoma in situ with death from breast cancer. JAMA Netw Open 3(9):e2017124. https://doi.org/10.1001/jamanetworkopen.2020.17124
Hu Z et al (2019) Quantitative evidence for early metastatic seeding in colorectal cancer. Nat Genet 51(7):1113–1122. https://doi.org/10.1038/s41588-019-0423-x
Hu Z et al (2020) Multi-cancer analysis of clonality and the timing of systemic spread in paired primary tumors and metastases. Nat Genet 52(7):701–708. https://doi.org/10.1038/s41588-020-0628-z
Zhao ZM et al (2016) Early and multiple origins of metastatic lineages within primary tumors. Proc Natl Acad Sci USA 113(8)2140–2145. https://doi.org/10.1073/pnas.1525677113
Hudziak RM et al (1989) p185HER2 monoclonal antibody has antiproliferative effects in vitro and sensitizes human breast tumor cells to tumor necrosis factor. Mol Cell Biol 9(3)1165–1172. https://doi.org/10.1128/mcb.9.3.1165-1172.1989
Dillekas H, Rogers MS, Straume O (2019) Are 90% of deaths from cancer caused by metastases? Cancer Med 8(12)5574–5576. https://doi.org/10.1002/cam4.2474
Collins VP, Loeffler RK, Tivey H (1956) Observations on growth rates of human tumors. Am J Roentgenol Radium Ther Nucl Med 76(5):988–1000
Friberg S, Mattson S (1997) On the growth rates of human malignant tumors: implications for medical decision making. J Surg Oncol 65(4):284–297. https://doi.org/10.1002/(SICI)1096-9098(199708)65:4%3C284::AID-JSO11%3E3.0.CO;2-2
Klein CA (2020) Cancer progression and the invisible phase of metastatic colonization. Nat Rev Cancer 20(11):681–694. https://doi.org/10.1038/s41568-020-00300-6
Halsted WS (1894) I. The results of Operations for the cure of Cancer of the breast performed at the Johns Hopkins Hospital from June, 1889, to January, 1894. Ann Surg 20(5):497–555. https://doi.org/10.1097/00000658-189407000-00075
Schardt JA et al (2005) Genomic analysis of single cytokeratin-positive cells from bone marrow reveals early mutational events in breast cancer. Cancer Cell 8(3):227–239. https://doi.org/10.1016/j.ccr.2005.08.003
Naume B et al (2014) Clinical outcome with correlation to disseminated tumor cell (DTC) status after DTC-guided secondary adjuvant treatment with docetaxel in early Breast cancer. J Clin Oncol 32(34):3848–3857. https://doi.org/10.1200/JCO.2014.56.9327
Braun S et al (2000) Cytokeratin-positive cells in the bone marrow and survival of patients with stage I, II, or III Breast cancer. N Engl J Med 342(8):525–533. https://doi.org/10.1056/NEJM200002243420801
Linde N et al (2018) Macrophages orchestrate Breast cancer early dissemination and metastasis. Nat Commun 9(1):21. https://doi.org/10.1038/s41467-017-02481-5
Schmidt-Kittler O et al (2003) From latent disseminated cells to overt Metastasis: genetic analysis of systemic breast cancer progression. Proc Natl Acad Sci USA 100(13):7737–7742. https://doi.org/10.1073/pnas.1331931100
Eyles J et al (2010) Tumor cells disseminate early, but immunosurveillance limits metastatic outgrowth, in a mouse model of melanoma. J Clin Invest 120(6):2030–2039. https://doi.org/10.1172/JCI42002
Shain AH et al (2019) The genetic evolution of metastatic uveal melanoma. Nat Genet 51(7):1123–1130. https://doi.org/10.1038/s41588-019-0440-9
Ulmer A et al (2014) Quantitative measurement of melanoma spread in sentinel lymph nodes and survival. PLoS Med 11(2):e1001604. https://doi.org/10.1371/journal.pmed.1001604
Rhim AD et al (2012) EMT and dissemination precede pancreatic Tumor formation. Cell 148(1–2):349–361. https://doi.org/10.1016/j.cell.2011.11.025
Pantel K et al (1996) Frequency and prognostic significance of isolated tumour cells in bone marrow of patients with non-small-cell lung cancer without overt metastases. Lancet 347(9002):649–653. https://doi.org/10.1016/s0140-6736(96)91203-9
Mederer T et al (2022) EpCAM-positive disseminated cancer cells in bone marrow impact on survival of early-stage NSCLC patients. Lung Cancer 167:73–77. https://doi.org/10.1016/j.lungcan.2022.02.008
Doberstein K et al (2022) L1CAM is required for early dissemination of fallopian tube carcinoma precursors to the ovary. Commun Biol 5(1):1362. https://doi.org/10.1038/s42003-022-04314-8
Hoffmann M et al (2018) Diagnostic pathology of early systemic cancer: ERBB2 gene amplification in single disseminated cancer cells determines patient survival in operable esophageal cancer. Int J Cancer 142(4):833–843. https://doi.org/10.1002/ijc.31108
Kennecke H et al (2010) Metastatic behavior of breast cancer subtypes. J Clin Oncol 28(20):3271–3277. https://doi.org/10.1200/JCO.2009.25.9820
Harney AS et al (2015) Real-time imaging reveals local, transient vascular permeability, and tumor cell intravasation stimulated by TIE2hi macrophage-derived VEGFA. Cancer Discov 5(9):932–943. https://doi.org/10.1158/2159-8290.CD-15-0012
Guy CT, Cardiff RD, Muller WJ (1992) Induction of mammary tumors by expression of polyomavirus middle T oncogene: a transgenic mouse model for metastatic disease. Mol Cell Biol 12(3):954–961. https://doi.org/10.1128/mcb.12.3.954-961.1992
Lin EY et al (2003) Progression to malignancy in the polyoma middle T oncoprotein mouse breast cancer model provides a reliable model for human diseases. Am J Pathol 163 5:2113–2126. https://doi.org/10.1016/S0002-9440(10)63568-7
Pfefferle AD et al (2013) Transcriptomic classification of genetically engineered mouse models of breast cancer identifies human subtype counterparts. Genome Biol 14(11):R125. https://doi.org/10.1186/gb-2013-14-11-r125
Weng D et al (2012) Metastasis is an early event in mouse mammary carcinomas and is associated with cells bearing stem cell markers. Breast Cancer Res 14(1):R18. https://doi.org/10.1186/bcr3102
Lin EY et al (2001) Colony-stimulating factor 1 promotes progression of mammary tumors to malignancy. J Exp Med 193(6):727–740. https://doi.org/10.1084/jem.193.6.727
Valdez KE et al (2011) Human primary ductal carcinoma in situ (DCIS) subtype-specific pathology is preserved in a mouse intraductal (MIND) xenograft model. J Pathol 225(4):565–573. https://doi.org/10.1002/path.2969
Behbod F et al (2009) An intraductal human-in-mouse transplantation model mimics the subtypes of ductal carcinoma in situ. Breast Cancer Res 11(5):R66. https://doi.org/10.1186/bcr2358
Elsarraj HS et al (2015) Expression profiling of in vivo ductal carcinoma in situ progression models identified B cell lymphoma-9 as a molecular driver of Breast cancer invasion. Breast Cancer Res 17:128. https://doi.org/10.1186/s13058-015-0630-z
Pommier A et al (2018) Unresolved endoplasmic reticulum stress engenders immune-resistant, latent Pancreatic cancer metastases. Science. https://doi.org/10.1126/science.aao4908
Gopinathan A et al (2015) GEMMs as preclinical models for testing pancreatic cancer therapies. Dis Model Mech 8(10):1185–1200. https://doi.org/10.1242/dmm.021055
Hingorani SR et al (2003) Preinvasive and invasive ductal Pancreatic cancer and its early detection in the mouse. Cancer Cell 4(6):437–450. https://doi.org/10.1016/s1535-6108(03)00309-x
Lee JW et al (2016) Genetically engineered mouse models of pancreatic cancer: The KPC model (LSL-Kras(G12D/+); LSL-Trp53(R172H/+); Pdx-1-Cre), its variants, and their application in immuno-oncology drug discovery. Curr Protoc Pharmacol 73:14.39.1-14.39.20. https://doi.org/10.1002/cpph.2
Knight JC et al (2020) Early detection in a mouse model of pancreatic cancer by imaging DNA damage response signaling. J Nucl Med 61(7):1006–1013. https://doi.org/10.2967/jnumed.119.234708
Drosos Y et al (2016) Prox1-heterozygosis sensitizes the pancreas to oncogenic Kras-Induced neoplastic transformation. Neoplasia 18(3):172–84. https://doi.org/10.1016/j.neo.2016.02.002
Zhu L et al (2007) Acinar cells contribute to the molecular heterogeneity of pancreatic intraepithelial neoplasia. Am J Pathol 171(1):263–273. https://doi.org/10.2353/ajpath.2007.061176
Drosos Y et al (2017) ATM-deficiency increases genomic instability and metastatic potential in a mouse model of pancreatic cancer. Sci Rep 7(1):11144. https://doi.org/10.1038/s41598-017-11661-8
Aguirre AJ et al (2003) Activated Kras and Ink4a/Arf deficiency cooperate to produce metastatic pancreatic ductal adenocarcinoma. Genes Dev 17(24):3112–3126. https://doi.org/10.1101/gad.1158703
Lengagne R et al (2008) Distinct role for CD8 T cells toward cutaneous tumors and visceral metastases. J Immunol 180(1):130–137. https://doi.org/10.4049/jimmunol.180.1.130
Dragani TA et al (2000) Mapping of melanoma modifier loci in RET transgenic mice. Jpn J Cancer Res 91(11):1142–1147. https://doi.org/10.1111/j.1349-7006.2000.tb00897.x
Fisher B et al (1989) Eight-year results of a randomized clinical trial comparing total mastectomy and lumpectomy with or without irradiation in the treatment of Breast cancer. N Engl J Med 320(13):822–828. https://doi.org/10.1056/NEJM198903303201302
Veronesi U et al (1990) Quadrantectomy versus lumpectomy for small size breast cancer. Eur J Cancer 26(6):671–673. https://doi.org/10.1016/0277-5379(90)90114-9
Rahrmann EP et al (2022) The NALCN channel regulates Metastasis and nonmalignant cell dissemination. Nat Genet 54(12):1827–1838. https://doi.org/10.1038/s41588-022-01182-0
Sopik V, Narod SA (2018) The relationship between tumour size, nodal status and distant metastases: on the origins of breast cancer. Breast Cancer Res Treat 170(3):647–656. https://doi.org/10.1007/s10549-018-4796-9
Braun S et al (2005) A pooled analysis of bone marrow micrometastasis in breast cancer. N Engl J Med 353(8):793–802. https://doi.org/10.1056/NEJMoa050434
Noorani A et al (2020) Genomic evidence supports a clonal diaspora model for metastases of esophageal adenocarcinoma. Nat Genet 52(1):74–83. https://doi.org/10.1038/s41588-019-0551-3
Hafner C et al (2010) Multiple oncogenic mutations and clonal relationship in spatially distinct benign human epidermal tumors. Proc Natl Acad Sci USA 107(48):20780–20785. https://doi.org/10.1073/pnas.1008365107
Lips EH et al (2022) Genomic analysis defines clonal relationships of ductal carcinoma in situ and recurrent invasive Breast cancer. Nat Genet 54(6):850–860. https://doi.org/10.1038/s41588-022-01082-3
Zhang Y et al (2018) Somatic Trp53 mutations differentially drive Breast cancer and evolution of metastases. Nat Commun 9(1):3953. https://doi.org/10.1038/s41467-018-06146-9
Sottoriva A et al (2015) A Big Bang model of human colorectal tumor growth. Nat Genet 47(3):209–216. https://doi.org/10.1038/ng.3214
Kim K et al (2023) Cell competition shapes metastatic latency and relapse. Cancer Discov 13(1):85–97. https://doi.org/10.1158/2159-8290.CD-22-0236
Sproll C et al (2017) Immunohistochemical detection of lymph node-DTCs in patients with node-negative HNSCC. Int J Cancer 140(9):2112–2124. https://doi.org/10.1002/ijc.30617
Gerber B et al (2001) Simultaneous immunohistochemical detection of Tumor cells in lymph nodes and bone marrow aspirates in Breast cancer and its correlation with other prognostic factors. J Clin Oncol 19(4):960–971. https://doi.org/10.1200/JCO.2001.19.4.960
El Hage Chehade H et al (2017) Is sentinel lymph node biopsy indicated in patients with a diagnosis of ductal carcinoma in situ? A systematic literature review and meta-analysis. Am J Surg 213(1):171–180. https://doi.org/10.1016/j.amjsurg.2016.04.019
Sakr R et al (2006) Ductal carcinoma in situ: value of sentinel lymph node biopsy. J Surg Oncol 94(5):426–430. https://doi.org/10.1002/jso.20578
Katz A et al (2006) Sentinel lymph node positivity of patients with ductal carcinoma in situ or microinvasive Breast cancer. Am J Surg 191(6):761–766. https://doi.org/10.1016/j.amjsurg.2006.01.019
Klauber-DeMore N et al (2000) Sentinel lymph node biopsy: is it indicated in patients with high-risk ductal carcinoma-in-situ and ductal carcinoma-in-situ with microinvasion? Ann Surg Oncol 7(9):636–642. https://doi.org/10.1007/s10434-000-0636-2
Lyman GH et al (2017) Sentinel Lymph Node Biopsy for patients with early-stage breast cancer: American society of clinical oncology clinical practice guideline update. J Clin Oncol 35(5):561–564. https://doi.org/10.1200/JCO.2016.71.0947
Kollermann J et al (1999) Comparative immunocytochemical assessment of isolated carcinoma cells in lymph nodes and bone marrow of patients with clinically localized Prostate cancer. Int J Cancer 84(2):145–149. https://doi.org/10.1002/(SICI)1097-0215(19990420)84:2%3C145::AID-IJC9%3E3.0.CO;2-5
Lindemann F et al (1992) Prognostic significance of micrometastatic tumour cells in bone marrow of colorectal cancer patients. Lancet 340(8821):685–689. https://doi.org/10.1016/0140-6736(92)92230-d
Effenberger KE et al (2012) Disseminated Tumor cells in pancreatic cancer-an Independent prognosticator of Disease progression and survival. Int J Cancer 131(4):E475–E483. https://doi.org/10.1002/ijc.26439
Vashist YK et al (2012) Disseminated Tumor cells in bone marrow and the natural course of resected Esophageal cancer. Ann Surg 255(6):1105–1112. https://doi.org/10.1097/SLA.0b013e3182565b0b
Hartkopf AD et al (2021) Disseminated tumour cells from the bone marrow of early breast cancer patients: results from an international pooled analysis. Eur J Cancer 154:128–137. https://doi.org/10.1016/j.ejca.2021.06.028
Bidard FC et al (2009) Disseminated Tumor cells and the risk of locoregional recurrence in nonmetastatic breast cancer. Ann Oncol 20(11):1836–1841. https://doi.org/10.1093/annonc/mdp200
Mansi J et al (2016) Bone marrow micrometastases in early breast cancer-30-year outcome. Br J Cancer 114(3):243–247. https://doi.org/10.1038/bjc.2015.447
Diel IJ et al (1996) Micrometastatic Breast cancer cells in bone marrow at primary surgery: prognostic value in comparison with nodal status. J Natl Cancer Inst 88(22):1652–1658. https://doi.org/10.1093/jnci/88.22.1652
Pierga JY et al (2004) Clinical significance of immunocytochemical detection of tumor cells using digital microscopy in peripheral blood and bone marrow of breast cancer patients. Clin Cancer Res 10(4):1392–1400. https://doi.org/10.1158/1078-0432.ccr-0102-03
Giuliano AE et al (2011) Association of occult metastases in sentinel lymph nodes and bone marrow with survival among women with early-stage invasive Breast cancer. JAMA 306(4):385–393. https://doi.org/10.1001/jama.2011.1034
Gebauer G et al (2001) Epithelial cells in bone marrow of breast cancer patients at time of primary surgery: clinical outcome during long-term follow-up. J Clin Oncol 19(16):3669–3674. https://doi.org/10.1200/JCO.2001.19.16.3669
Wiedswang G et al (2003) Detection of isolated Tumor cells in bone marrow is an independent prognostic factor in Breast cancer. J Clin Oncol 21(18):3469–3478. https://doi.org/10.1200/JCO.2003.02.009
Falck AK et al (2012) Analysis of and prognostic information from disseminated tumour cells in bone marrow in primary breast cancer: a prospective observational study. BMC Cancer 12:403. https://doi.org/10.1186/1471-2407-12-403
Sola M et al (2011) Prognostic value of hematogenous dissemination and biological profile of the Tumor in early breast cancer patients: a prospective observational study. BMC Cancer 11:252. https://doi.org/10.1186/1471-2407-11-252
Domschke C et al (2013) Prognostic value of disseminated tumor cells in the bone marrow of patients with operable primary Breast cancer: a long-term follow-up study. Ann Surg Oncol 20(6):1865–1871. https://doi.org/10.1245/s10434-012-2814-4
Hartkopf AD et al (2015) Disseminated Tumor cells from the bone marrow of patients with nonmetastatic primary breast cancer are predictive of locoregional relapse. Ann Oncol 26(6):1155–1160. https://doi.org/10.1093/annonc/mdv148
Hartkopf AD et al (2014) Prognostic relevance of disseminated tumour cells from the bone marrow of early stage breast cancer patients-results from a large single-centre analysis. Eur J Cancer 50(15):2550–2559. https://doi.org/10.1016/j.ejca.2014.06.025
Coleman R et al (2013) Zoledronic acid (zoledronate) for postmenopausal women with early breast cancer receiving adjuvant letrozole (ZO-FAST study): final 60-month results. Ann Oncol 24(2):398–405. https://doi.org/10.1093/annonc/mds277
Aft R et al (2010) Effect of zoledronic acid on disseminated tumour cells in women with locally advanced breast cancer: an open label, randomised, phase 2 trial. Lancet Oncol 11(5):421–428. https://doi.org/10.1016/S1470-2045(10)70054-1
Vidula N et al (2021) Evaluation of disseminated tumor cells and circulating tumor cells in patients with breast cancer receiving adjuvant zoledronic acid. NPJ Breast Cancer 7(1):113. https://doi.org/10.1038/s41523-021-00323-8
Solomayer EF et al (2012) Influence of zoledronic acid on disseminated tumor cells in primary breast cancer patients. Ann Oncol 23(9):2271–2277. https://doi.org/10.1093/annonc/mdr612
Banys M et al (2013) Influence of zoledronic acid on disseminated tumor cells in bone marrow and survival: results of a prospective clinical trial. BMC Cancer 13:480. https://doi.org/10.1186/1471-2407-13-480
Dai R et al (2022) Osteoblasts and osteoclasts: an important switch of tumour cell dormancy during bone metastasis. J Exp Clin Cancer Res 41(1):316. https://doi.org/10.1186/s13046-022-02520-0
Coleman R et al (2020) Adjuvant denosumab in early breast cancer (D-CARE): an international, multicentre, randomised, controlled, phase 3 trial. Lancet Oncol 21(1):60–72. https://doi.org/10.1016/S1470-2045(19)30687-4
Kaminskas E et al (2005) FDA drug approval summary: azacitidine (5-azacytidine, Vidaza) for injectable suspension. Oncologist 10(3):176–182. https://doi.org/10.1634/theoncologist.10-3-176
Damery E, Solimando DA Jr., Waddell JA (2016) Arsenic trioxide and tretinoin (AsO/ATRA) for acute promyelocytic leukemia (APL). Hosp Pharm 51(8):628–632. https://doi.org/10.1310/hpj5108-628
Synnestvedt M et al (2012) Disseminated Tumor cells as selection marker and monitoring tool for secondary adjuvant treatment in early breast cancer. Descriptive results from an intervention study. BMC Cancer 12:616. https://doi.org/10.1186/1471-2407-12-616
Borgen E et al (2018) NR2F1 stratifies dormant disseminated tumor cells in breast cancer patients. Breast Cancer Res 20(1):120. https://doi.org/10.1186/s13058-018-1049-0
Crist SB, Ghajar CM (2021) When a House is not a home: a survey of antimetastatic niches and potential mechanisms of disseminated tumor cell suppression. Annu Rev Pathol 16:409–432. https://doi.org/10.1146/annurev-pathmechdis-012419-032647
Weiss L (1989) Biomechanical destruction of cancer cells in skeletal muscle: a rate-regulator for hematogenous metastasis. Clin Exp Metastasis 7(5):483–491. https://doi.org/10.1007/BF01753809
Bar-Yehuda S et al (2001) Resistance of muscle to tumor metastases: a role for a3 adenosine receptor agonists. Neoplasia 3(2):125–131. https://doi.org/10.1038/sj.neo.7900138
Parlakian A et al (2010) Skeletal muscle phenotypically converts and selectively inhibits metastatic cells in mice. PLoS ONE 5(2):e9299. https://doi.org/10.1371/journal.pone.0009299
Crist SB et al (2022) Unchecked oxidative stress in skeletal muscle prevents outgrowth of disseminated tumour cells. Nat Cell Biol 24(4):538–553. https://doi.org/10.1038/s41556-022-00881-4
Meng S et al (2004) Circulating tumor cells in patients with Breast cancer dormancy. Clin Cancer Res: Off J Am Association Cancer Res 10(24):8152–8162. https://doi.org/10.1158/1078-0432.CCR-04-1110
Rhim AD et al (2014) Detection of circulating pancreas epithelial cells in patients with pancreatic cystic lesions. Gastroenterology 146(3):647–651. https://doi.org/10.1053/j.gastro.2013.12.007
Yang C et al (2018) Clinical significance of peripheral circulating tumor cell counts in colorectal polyps and non-metastatic colorectal cancer. World J Surg Oncol 16(1):13. https://doi.org/10.1186/s12957-017-1305-2
Pantel K et al (2012) Circulating epithelial cells in patients with benign colon diseases. Clin Chem 58(5):936–940. https://doi.org/10.1373/clinchem.2011.175570
Tsai WS et al (2016) Circulating tumor cell Count correlates with colorectal neoplasm progression and is a prognostic marker for distant Metastasis in non-metastatic patients. Sci Rep 6:24517. https://doi.org/10.1038/srep24517
Ilie M et al (2014) Sentinel circulating tumor cells allow early diagnosis of lung cancer in patients with chronic obstructive pulmonary disease. PLoS ONE 9(10):e111597. https://doi.org/10.1371/journal.pone.0111597
Franken B et al (2012) Circulating Tumor cells, Disease recurrence and survival in newly diagnosed breast cancer. Breast Cancer Res 14(5):R133. https://doi.org/10.1186/bcr3333
Groot VP et al (2019) Defining and predicting early recurrence in 957 patients with resected pancreatic ductal adenocarcinoma. Ann Surg 269 6:1154–1162. https://doi.org/10.1097/SLA.0000000000002734
Kromrey ML et al (2018) Prospective study on the incidence, prevalence and 5-year pancreatic-related mortality of pancreatic cysts in a population-based study. Gut 67(1):138–145. https://doi.org/10.1136/gutjnl-2016-313127
Kirby BJ et al (2012) Functional characterization of circulating tumor cells with a prostate-cancer-specific microfluidic device. PLoS ONE 7(4):e35976. https://doi.org/10.1371/journal.pone.0035976
Becker AE et al (2014) Pancreatic ductal adenocarcinoma: risk factors, screening, and early detection. World J Gastroenterol 20(32):11182–11198. https://doi.org/10.3748/wjg.v20.i32.11182
Cauley CE et al (2015) Circulating epithelial cells in patients with pancreatic lesions: clinical and pathologic findings. J Am Coll Surg 221(3):699–707. https://doi.org/10.1016/j.jamcollsurg.2015.05.014
Poruk KE et al (2017) Circulating epithelial cells in intraductal papillary mucinous neoplasms and cystic pancreatic lesions. Pancreas 46(7):943–947. https://doi.org/10.1097/MPA.0000000000000869
Franses JW et al (2018) Improved detection of circulating epithelial cells in patients with intraductal papillary mucinous neoplasms. Oncologist 23(1):121–7. https://doi.org/10.1634/theoncologist.2017-0234
Desitter I et al (2011) A new device for rapid isolation by size and characterization of rare circulating tumor cells. Anticancer Res 31(2):427–441
Vona G et al (2000) Isolation by size of epithelial tumor cells: a new method for the immunomorphological and molecular characterization of circulatingtumor cells. Am J Pathol 156(1):57–63. https://doi.org/10.1016/S0002-9440(10)64706-2
Sequist LV et al (2009) The CTC-chip: an exciting new tool to detect circulating tumor cells in lung cancer patients. J Thorac Oncol 4(3):281–283. https://doi.org/10.1097/JTO.0b013e3181989565
Alix-Panabieres C (2012) EPISPOT assay: detection of viable DTCs/CTCs in solid tumor patients. Recent Results Cancer Res 195:69–76. https://doi.org/10.1007/978-3-642-28160-0_6
Allard WJ et al (2004) Tumor cells circulate in the peripheral blood of all major carcinomas but not in healthy subjects or patients with nonmalignant diseases. Clin Cancer Res 10(20):6897–6904. https://doi.org/10.1158/1078-0432.CCR-04-0378
Johnson CM et al (2013) Meta-analyses of colorectal cancer risk factors. Cancer Causes Control 24(6):1207–1222. https://doi.org/10.1007/s10552-013-0201-5
Grosse-Wilde A et al (2015) Stemness of the hybrid epithelial/mesenchymal state in breast cancer and its association with poor survival. PLoS ONE 10 5:e0126522. https://doi.org/10.1371/journal.pone.0126522
Brown MS et al (2022) Phenotypic heterogeneity driven by plasticity of the intermediate EMT state governs disease progression and metastasis in breast cancer. Sci Adv 8 31:eabj8002. https://doi.org/10.1126/sciadv.abj8002
Cui J et al (2023) MLL3 loss drives Metastasis by promoting a hybrid epithelial-mesenchymal transition state. Nat Cell Biol 25(1):145–158. https://doi.org/10.1038/s41556-022-01045-0
Pastushenko I et al (2018) Identification of the tumour transition states occurring during EMT. Nature 556(7702):463–468. https://doi.org/10.1038/s41586-018-0040-3
Malagoli Tagliazucchi G et al (2023) Genomic and microenvironmental heterogeneity shaping epithelial-to-mesenchymal trajectories in cancer. Nat Commun 14(1):789. https://doi.org/10.1038/s41467-023-36439-7
Padilla-Nash HM et al (2013) Aneuploidy, oncogene amplification and epithelial to mesenchymal transition define spontaneous transformation of murine epithelial cells. Carcinogenesis 34(8):1929–1939. https://doi.org/10.1093/carcin/bgt138
Ried T et al (1999) Genomic changes defining the genesis, progression, and malignancy potential in solid human tumors: a phenotype/genotype correlation. Genes Chromosomes Cancer 25(3):195–204. https://doi.org/10.1002/(SICI)1098-2264(199907)25:3%3C195::AID-GCC1%3E3.0.CO;2-8
Dongre A, Weinberg RA (2019) New insights into the mechanisms of epithelial-mesenchymal transition and implications for cancer. Nat Rev Mol Cell Biol 20(2):69–84. https://doi.org/10.1038/s41580-018-0080-4
Liu X et al (2019) Epithelial-type systemic breast carcinoma cells with a restricted mesenchymal transition are a major source of metastasis. Sci Adv 5(6):eaav4275. https://doi.org/10.1126/sciadv.aav4275
van Roy F (2014) Beyond E-cadherin: roles of other cadherin superfamily members in cancer. Nat Rev Cancer 14(2):121–134. https://doi.org/10.1038/nrc3647
Wheelock MJ, Johnson KR (2003) Cadherins as modulators of cellular phenotype. Annu Rev Cell Dev Biol 19:207–235. https://doi.org/10.1146/annurev.cellbio.19.011102.111135
Guillot C, Lecuit T (2013) Mechanics of epithelial tissue homeostasis and morphogenesis. Science 340(6137):1185–1189. https://doi.org/10.1126/science.1235249
Aouad P et al (2022) Epithelial-mesenchymal plasticity determines estrogen receptor positive breast cancer dormancy and epithelial reconversion drives recurrence. Nat Commun 13(1):4975. https://doi.org/10.1038/s41467-022-32523-6
Padmanaban V et al (2019) E-cadherin is required for metastasis in multiple models of breast cancer. Nature 573(7774):439–444. https://doi.org/10.1038/s41586-019-1526-3
Simeonov KP et al (2021) Single-cell lineage tracing of metastatic cancer reveals selection of hybrid EMT states. Cancer Cell 39(8):1150–62e9. https://doi.org/10.1016/j.ccell.2021.05.005
Rodriguez-Tirado C et al (2022) NR2F1 is a barrier to dissemination of early stage breast cancer cells. Cancer Res https://doi.org/10.1158/0008-5472.CAN-21-4145
Azarin SM et al (2015) In vivo capture and label-free detection of early metastatic cells. Nat Commun 6:8094. https://doi.org/10.1038/ncomms9094
Rao SS et al (2016) Enhanced survival with Implantable scaffolds that capture metastatic breast cancer cells in vivo. Cancer Res 76(18):5209–5218. https://doi.org/10.1158/0008-5472.CAN-15-2106
Wang J et al (2023) A synthetic metastatic niche reveals antitumor neutrophils drive breast cancer metastatic dormancy in the lungs. Nat Commun 14(1):4790. https://doi.org/10.1038/s41467-023-40478-5
Carpenter RA et al (2018) Implantable pre-metastatic niches for the study of the microenvironmental regulation of disseminated human tumour cells. Nat Biomed Eng 2(12):915–929. https://doi.org/10.1038/s41551-018-0307-x
Haessler U et al (2012) Migration dynamics of breast cancer cells in a tunable 3D interstitial flow chamber. Integr Biol (Camb) 4(4):401–409. https://doi.org/10.1039/c1ib00128k
Kuhlbach C et al (2018) A microfluidic system for the investigation of tumor cell extravasation. Bioeng (Basel). https://doi.org/10.3390/bioengineering5020040
Hirway SU, Weinberg SH (2023) A review of computational modeling, machine learning and image analysis in cancer metastasis dynamics. Comput Syst Oncol. https://doi.org/10.1002/cso2.1044
Muller WJ et al (1988) Single-step induction of mammary adenocarcinoma in transgenic mice bearing the activated c-neu oncogene. Cell 54(1):105–115. https://doi.org/10.1016/0092-8674(88)90184-5
Xie Y, Li K, Hung MC (1995) Tyrosine phosphorylation of shc proteins and formation of Shc/Grb2 complex correlate to the transformation of NIH3T3 cells mediated by the point-mutation activated Neu. Oncogene 10(12):2409–2413
Stern DF, Heffernan PA, Weinberg RA (1986) p185, a product of the Neu proto-oncogene, is a receptorlike protein associated with tyrosine kinase activity. Mol Cell Biol 6(5):1729–1740. https://doi.org/10.1128/mcb.6.5.1729-1740.1986
Bargmann CI, Hung MC, Weinberg RA (1986) Multiple independent activations of the neu oncogene by a point mutation altering the transmembrane domain of p185. Cell 45:649–657. https://doi.org/10.1016/0092-8674(86)90779-8
Di Carlo E et al (1999) Analysis of mammary carcinoma onset and progression in HER-2/neu oncogene transgenic mice reveals a lobular origin. Lab Invest 79(10):1261–1269
Guy CT et al (1992) Expression of the neu protooncogene in the mammary epithelium of transgenic mice induces metastatic disease. Proc Natl Acad Sci USA 89(22):10578–10582. https://doi.org/10.1073/pnas.89.22.10578
Wang J et al (2020) Her2 promotes early dissemination of Breast cancer by suppressing the p38-MK2-Hsp27 pathway that is targetable by Wip1 inhibition. Oncogene 39(40):6313–6326. https://doi.org/10.1038/s41388-020-01437-2
Rohan TE et al (2014) Tumor microenvironment of metastasis and risk of distant metastasis of breast cancer. J Natl Cancer Inst 106:8. https://doi.org/10.1093/jnci/dju136
Konstantinidis IT et al (2013) Pancreatic ductal adenocarcinoma: is there a survival difference for R1 resections versus locally advanced unresectable tumors? What is a true R0 resection? Ann Surg 257(4):731–736. https://doi.org/10.1097/SLA.0b013e318263da2f
Okimura A et al (2009) Immunohistochemical analyses of E-cadherin, beta-catenin, CD44s, and CD44v6 expressions, and Ki-67 labeling index in intraductal papillary mucinous Neoplasms of the pancreas and associated invasive carcinomas. Med Mol Morphol 42(4):222–229. https://doi.org/10.1007/s00795-009-0462-y
Yachida S et al (2010) Distant metastasis occurs late during the genetic evolution of pancreatic cancer. Nature 467(7319):1114–1117. https://doi.org/10.1038/nature09515
Iwamoto T et al (1991) Aberrant melanogenesis and melanocytic tumour development in transgenic mice that carry a metallothionein/ret fusion gene. EMBO J 10(11):3167–3175. https://doi.org/10.1002/j.1460-2075.1991.tb04878.x
Dupuy F et al (2015) PDK1-Dependent metabolic reprogramming dictates metastatic potential in breast cancer. Cell Metab 22(4):577–589. https://doi.org/10.1016/j.cmet.2015.08.007
Fox DB et al (2020) NRF2 activation promotes the recurrence of dormant tumour cells through regulation of redox and nucleotide metabolism. Nat Metab 2(4):318–334. https://doi.org/10.1038/s42255-020-0191-z
Julie Di M et al (2021) A tumor-derived type III collagen-rich ECM niche regulates tumor cell dormancy. Nat Portf https://doi.org/10.21203/rs.3.rs-580847/v1
Aguirre Ghiso JA, Kovalski K, Ossowski L (1999) Tumor dormancy induced by downregulation of urokinase receptor in human carcinoma involves integrin and MAPK signaling. J Cell Biol 147(1):89–104
Malladi S et al (2016) Metastatic latency and immune evasion through autocrine inhibition of WNT. Cell 165(1):45–60. https://doi.org/10.1016/j.cell.2016.02.025
White AC et al (2014) Stem cell quiescence acts as a tumour suppressor in squamous tumours. Nat Cell Biol 16(1):99–107. https://doi.org/10.1038/ncb2889
Liu L et al (2013) Chromatin modifications as determinants of muscle stem cell quiescence and chronological aging. Cell Rep 4(1):189–204. https://doi.org/10.1016/j.celrep.2013.05.043
Lee J et al (2016) Signalling couples hair follicle stem cell quiescence with reduced histone H3 K4/K9/K27me3 for proper tissue homeostasis. Nat Commun 7:11278. https://doi.org/10.1038/ncomms11278
Cui K et al (2009) Chromatin signatures in multipotent human hematopoietic stem cells indicate the fate of bivalent genes during differentiation. Cell Stem Cell 4(1):80–93. https://doi.org/10.1016/j.stem.2008.11.011
Lien WH et al (2011) Genome-wide maps of histone modifications unwind in vivo chromatin states of the hair follicle lineage. Cell Stem Cell 9(3):219–232. https://doi.org/10.1016/j.stem.2011.07.015
Cheung TH et al (2012) Maintenance of muscle stem-cell quiescence by microRNA-489. Nature 482(7386):524–528. https://doi.org/10.1038/nature10834
Lechman ER et al (2012) Attenuation of miR-126 activity expands HSC in vivo without exhaustion. Cell Stem Cell 11(6):799–811. https://doi.org/10.1016/j.stem.2012.09.001
de Morree A et al (2017) Staufen1 inhibits MyoD translation to actively maintain muscle stem cell quiescence. Proc Natl Acad Sci USA 114(43):E8996–E9005. https://doi.org/10.1073/pnas.1708725114
Sosa MS et al (2015) NR2F1 controls tumour cell dormancy via SOX9- and RARbeta-driven quiescence programmes. Nat Commun 6:6170. https://doi.org/10.1038/ncomms7170
Sun D et al (2022) MacroH2A impedes metastatic growth by enforcing a discrete dormancy program in disseminated cancer cells. Sci Adv 8(48):eabo0876. https://doi.org/10.1126/sciadv.abo0876
Shen S et al (2020) Melanoma Persister cells are tolerant to BRAF/MEK inhibitors via ACOX1-Mediated fatty acid oxidation. Cell Rep 33(8):108421. https://doi.org/10.1016/j.celrep.2020.108421
Echeverria GV et al (2019) Resistance to neoadjuvant chemotherapy in triple-negative Breast cancer mediated by a reversible drug-tolerant state. Sci Transl Med. https://doi.org/10.1126/scitranslmed.aav0936
La Belle Flynn A et al (2019) Autophagy inhibition elicits emergence from metastatic dormancy by inducing and stabilizing Pfkfb3 expression. Nat Commun 10(1):3668. https://doi.org/10.1038/s41467-019-11640-9
Aloia A et al (2019) A fatty acid oxidation-dependent metabolic shift regulates the adaptation of BRAF-mutated Melanoma to MAPK inhibitors. Clin cancer Res: Off J Am Assoc Cancer Res 25(22):6852–6867. https://doi.org/10.1158/1078-0432.CCR-19-0253
Schewe DM, Aguirre-Ghiso JA (2008) ATF6alpha-Rheb-mTOR signaling promotes survival of dormant Tumor cells in vivo. Proc Natl Acad Sci USA 105(30):10519–10524
van Velthoven CTJ, Rando TA (2019) Stem cell Quiescence: dynamism, restraint, and cellular idling. Cell Stem Cell 24(2):213–225. https://doi.org/10.1016/j.stem.2019.01.001
Hu J et al (2023) STING inhibits the reactivation of dormant metastasis in lung adenocarcinoma. Nature 616(7958):806–813. https://doi.org/10.1038/s41586-023-05880-5
Correia AL et al (2021) Hepatic stellate cells suppress NK cell-sustained breast cancer dormancy. Nature 594(7864):566–571. https://doi.org/10.1038/s41586-021-03614-z
Muller-Hermelink N et al (2008) TNFR1 signaling and IFN-gamma signaling determine whether T cells induce tumor dormancy or promote multistage carcinogenesis. Cancer Cell 13(6):507–518. https://doi.org/10.1016/j.ccr.2008.04.001
Saudemont A, Quesnel B (2004) In a model of tumor dormancy, long-term persistent leukemic cells have increased B7-H1 and B7.1 expression and resist CTL-mediated lysis. Blood 104(7):2124–2133
Li S et al (2014) Model of tumor dormancy/recurrence after short-term chemotherapy. PLoS ONE 9(5):e98021. https://doi.org/10.1371/journal.pone.0098021
Naumov GN et al (2003) Ineffectiveness of doxorubicin treatment on solitary dormant mammary carcinoma cells or late-developing metastases. Breast Cancer Res Treat 82(3):199–206
Carlson P et al (2019) Targeting the perivascular niche sensitizes disseminated tumour cells to chemotherapy. Nat Cell Biol 21(2):238–250. https://doi.org/10.1038/s41556-018-0267-0
Aguirre-Ghiso JA et al (2003) ERK(MAPK) activity as a determinant of tumor growth and dormancy; regulation by p38(SAPK). Cancer Res 63(7):1684–1695
Ranganathan AC et al (2006) Tumor cell dormancy induced by p38(SAPK) and ER-stress signaling: an adaptive advantage for metastatic cells? Cancer Biol Ther 5(7):729–735
El Touny LH et al (2014) Combined SFK/MEK inhibition prevents metastatic outgrowth of dormant tumor cells. J Clin Investig 124(1):156–168. https://doi.org/10.1172/JCI70259
Ranganathan AC et al (2008) Dual function of pancreatic endoplasmic reticulum kinase in tumor cell growth arrest and survival. Cancer Res 68(9):3260–3268
Ranganathan AC et al (2006) Functional coupling of p38-Induced up-regulation of BiP and activation of RNA-dependent protein kinase-like endoplasmic reticulum kinase to drug resistance of dormant carcinoma cells. Cancer Res 66(3):1702–1711
Lu Z et al (2008) The Tumor suppressor gene ARHI regulates autophagy and Tumor dormancy in human Ovarian cancer cells. J Clin Investig 118(12):3917–3929. https://doi.org/10.1172/JCI35512
Gupta A et al (2010) Autophagy inhibition and antimalarials promote cell death in gastrointestinal stromal Tumor (GIST). Proc Natl Acad Sci USA 107(32):14333–14338. https://doi.org/10.1073/pnas.1000248107
Smith RA et al (2019) Cancer screening in the United States, 2019: a review of current American Cancer Society guidelines and current issues in cancer screening. CA Cancer J Clin 69(3):184–210. https://doi.org/10.3322/caac.21557
Lynge E et al (2003) Reporting of performance indicators of mammography screening in Europe. Eur J Cancer Prev 12(3):213–222. https://doi.org/10.1097/00008469-200306000-00008
Christiansen SR, Autier P, Stovring H (2022) Change in effectiveness of mammography screening with decreasing breast cancer mortality: a population-based study. Eur J Public Health 32(4):630–635. https://doi.org/10.1093/eurpub/ckac047
Tabar L et al (2019) The incidence of fatal breast cancer measures the increased effectiveness of therapy in women participating in mammography screening. Cancer 125(4):515–523. https://doi.org/10.1002/cncr.31840
Mabe K et al (2022) Endoscopic screening for gastric cancer in Japan: current status and future perspectives. Dig Endosc 34(3):412–419. https://doi.org/10.1111/den.14063
Myers ER et al (2015) Benefits and harms of breast cancer screening: a systematic review. JAMA 314(15):1615–1634. https://doi.org/10.1001/jama.2015.13183
Sagara Y et al (2016) Patient prognostic score and associations with survival improvement offered by radiotherapy after breast-conserving surgery for ductal carcinoma in situ: a population-based longitudinal cohort study. J Clin Oncol 34(11):1190–1196. https://doi.org/10.1200/JCO.2015.65.1869
Odle TG (2017) Precision medicine in breast cancer. Radiol Technol 88(4):401M–21M
Strand SH et al (2022) Molecular classification and biomarkers of clinical outcome in breast ductal carcinoma in situ: analysis of TBCRC 038 and RAHBT cohorts. Cancer Cell 40(12):1521–36e7. https://doi.org/10.1016/j.ccell.2022.10.021
Syed YY (2020) Oncotype DX breast recurrence score((R)): a review of its use in early-stage breast cancer. Mol Diagn Ther 24(5):621–632. https://doi.org/10.1007/s40291-020-00482-7
Shah C et al (2021) The clinical utility of DCISionRT((R)) on radiation therapy decision making in patients with ductal carcinoma in situ following breast-conserving surgery. Ann Surg Oncol 28(11):5974–5984. https://doi.org/10.1245/s10434-021-09903-1
Rodriguez-Gonzalez FG et al (2011) MicroRNA-30c expression level is an independent predictor of clinical benefit of endocrine therapy in advanced estrogen receptor positive breast cancer. Breast Cancer Res Treat 127(1):43–51. https://doi.org/10.1007/s10549-010-0940-x
Loudig O et al (2022) MiRNA expression deregulation correlates with the oncotype DX((R)) DCIS score. Breast Cancer Res 24(1):62. https://doi.org/10.1186/s13058-022-01558-4
Xu F et al (2021) Diagnostic accuracy and prognostic value of three-dimensional electrical impedance tomography imaging in patients with Breast cancer. Gland Surg 10(9):2673–2685. https://doi.org/10.21037/gs-21-348
Council IMNR (2005). In: Joy JE, Penhoet EE, Petitti DB (eds) Saving women’s lives: strategies for improving breast cancer detection and diagnosis. The national academies collection: reports funded by national institutes of health. The National Academies Press, Washington
Tucker FL (2012) Imaging-assisted large-format breast pathology: program rationale and development in a nonprofit health system in the United States. Int J Breast Cancer 2012:171792. https://doi.org/10.1155/2012/171792
Kahan Z (2022) Diversity of breast cancers begins at imaging. Eur J Radiol 154:110362. https://doi.org/10.1016/j.ejrad.2022.110362
Michaelson JS et al (2002) Predicting the survival of patients with breast carcinoma using Tumor size. Cancer 95(4):713–723. https://doi.org/10.1002/cncr.10742
Zhou W et al (2017) Ductal breast carcinoma in situ: mammographic features and its relation to prognosis and tumour biology in a population based cohort. Int J Breast Cancer 2017:4351319. https://doi.org/10.1155/2017/4351319
Holmberg L et al (2013) Mammography casting-type calcification and risk of local recurrence in DCIS: analyses from a randomised study. Br J Cancer 108(4):812–819. https://doi.org/10.1038/bjc.2013.26
Tabar L et al (2014) A proposal to unify the classification of breast and prostate cancers based on the anatomic site of cancer origin and on long-term patient outcome. Breast Cancer (Auckl) 8:15–38. https://doi.org/10.4137/BCBCR.S13833
Tarjan M et al (2012) Improved differentiation between ductal and acinar Prostate cancer using three-dimensional histology and biomarkers. Scand J Urol Nephrol 46(4):258–266. https://doi.org/10.3109/00365599.2012.675586
Tabar L et al (2022) Breast cancers originating from the terminal ductal lobular units: in situ and invasive acinar adenocarcinoma of the breast, AAB. Eur J Radiol 152:110323. https://doi.org/10.1016/j.ejrad.2022.110323
Tabár L et al (2022) Imaging biomarkers of breast cancers originating from the major lactiferous ducts: ductal adenocarcinoma of the breast, DAB. Eur J Radiol 154:110394. https://doi.org/10.1016/j.ejrad.2022.110394
Tabár L et al (2022) Breast cancers originating from the major lactiferous ducts and the process of neoductgenesis: ductal adenocarcinoma of the breast, DAB. Eur J Radiol 153:110363. https://doi.org/10.1016/j.ejrad.2022.110363
Tabar L et al (2022) A new approach to breast cancer terminology based on the anatomic site of tumour origin: the importance of radiologic imaging biomarkers. Eur J Radiol 149:110189. https://doi.org/10.1016/j.ejrad.2022.110189
Acknowledgements
We would like to thank all the members of the lab for stimulating scientific discussions on the dormancy aspects of the metastatic cascade. We would also like to thank Editage (www.editage.com) for English language editing. All figures were created using Biorender (www.biorender.com). This work was supported by the Susan G. Komen CCR grant CCR22899137 (M.S.S.), NIH R01 1R01CA266443-01A1 grant (M.S.S.), ASPIRE-Susan G. Komen grant ASP231044859 (M.S.S., C.R.T.), Pershing Square Sohn Prize-PSSCRA (M.S.S.), and Gilead Sciences Research Scholars Program in Solid Tumors grant (M.S.S.).
Author information
Authors and Affiliations
Contributions
CRT and MSS wrote the manuscript and prepared figures. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Presented at the 9th International Congress on Cancer Metastasis through the Lymphovascular System, May 4–6, 2023, in San Francisco, CA. To be published in a Special Issue of Clinical and Experimental Metastasis: Molecular Mechanisms of Cancer Metastasis.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rodriguez-Tirado, C., Sosa, M.S. How much do we know about the metastatic process?. Clin Exp Metastasis 41, 275–299 (2024). https://doi.org/10.1007/s10585-023-10248-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-023-10248-0