Abstract
Few assessments of species vulnerability to climate change used to inform conservation management consider the intrinsic traits that shape species’ capacity to respond to climate change. This omission is problematic as it may result in management actions that are not optimised for the long-term persistence of species as climates shift. We present a tool for explicitly linking data on plant species’ life history traits and range characteristics to appropriate management actions that maximise their capacity to respond to climate change. We deliberately target data on easily measured and widely available traits (e.g. dispersal syndrome, height, longevity) and range characteristics (e.g. range size, climatic/soil niche breadth), to allow for rapid comparison across many species. We test this framework on 1237 plants, categorising species on the basis of their potential climate change risk as related to four factors affecting their response capacity: reproduction, movement capability, abiotic niche specialisation and spatial coverage. Based on these four factors, species were allocated risk scores, and these were used to test the hypothesis that the current protection status under national legislation and related management actions capture species response capacity to climate change. Our results indicate that 20% of the plant species analysed (242 species) are likely to have a low capacity to respond to climate change based on the traits assessed, and are therefore at high risk. Of the 242 high risk species, only 10% (24 species) are currently listed for protection under conservation legislation. Importantly, many management plans for these listed species fail to address the capacity of species to respond to climate change with appropriate actions: 70% of approved management plans do not include crucial actions which may improve species’ ability to adapt to climate change. We illustrate how the use of easily attainable traits associated with ecological and evolutionary responses to changing environmental conditions can inform conservation actions for plant species globally.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
We are amidst a global biodiversity crisis, with threatening processes and species extinctions both increasing (Butchart et al. 2010; WWF 2016). Biodiversity is threatened by a wide array of factors, including climate change, habitat loss, degradation and fragmentation, and changing fire regimes (Hoekstra et al. 2005; Hooper et al. 2012). Climate change, in addition to its direct impact on biodiversity, can drive shifts in species distributions and interactions, resulting in novel ecosystems and species’ ranges (Chen et al. 2011), which has implications for species’ conservation, biosecurity and food security.
A fundamental failure in conservation planning and assessment, however, is that conservation resources are frequently allocated solely based on exposure of a species to threats (extrinsic factors) (Butt et al. 2016). This focus on exposure underestimates the problem, as vulnerability is also profoundly affected by two other intrinsic factors (Dawson et al. 2011). First, the sensitivity to a threat, which describes how strongly a species will be affected by a threat; for example, a tree species living at the edge of its upper thermal tolerance range may be sensitive to an increase in temperature (Araújo et al. 2013; Butt et al. 2013). Second, the adaptive capacity of the species, which represents its ability to adapt to or recover from exposure to a threat, as constrained by its traits. For example, climate change will have a larger impact on a species with limited mobility and dispersal than one that has a greater capacity for movement, given the assumption that species will undergo range shifts in response to climate change (Urban 2015; Pecl et al. 2017). The capacity of species to respond to climate change is therefore governed by intrinsic factors, evolutionary responses and phenotypic plasticity (Bellard et al. 2012; Nicotra et al. 2010; Nicotra et al. 2015; Christmas et al. 2016).
It is essential that conservation programmes that seek “future-smart” solutions explicitly consider not only species’ exposure, but also their capacity to respond when quantifying climate change risk, which may be in the form of assessment of vulnerability (Nicotra et al. 2015; Butt et al. 2016; Dawson et al. 2011; Beever et al. 2016): failure to do so diminishes our ability to successfully manage threats. Here we define response capacity as a species’ ability to respond to climate change as a function of the combination of its sensitivity and adaptive capacity, as underpinned by traits. Traits have become a common tool for generalising species’ responses to a wide range of ecological and environmental phenomenon, including invasive species (Stohlgren and Schnase 2006; Pyšek et al. 2012; Hejda et al. 2017), and climate change (Angert et al. 2011; Cabrelli et al. 2014; Foden et al. 2013), and are a useful option for preliminary screening and prioritisation (e.g. Cabrelli et al. 2014; Fortuni and Schubert 2017; Pearson et al. 2014; Pacifici et al. 2015). Their appeal is an ability to provide generalisable approximations of how species may respond to ecological and evolutionary processes. An ability to generalise is essential in conservation science as prioritisation often requires knowledge of the comparative effect of a threatening process across many species.
As exposure to climate change is generally fixed, management actions can only act on intrinsic factors (sensitivity and adaptive capacity), which govern response capacity. However, we lack operational frameworks for linking these intrinsic factors to appropriate management actions, particularly for plants (e.g. only one of the ten trait-based frameworks identified in a recent review included plants; Willis et al. 2015). Vulnerability assessments that only consider exposure (e.g. Beever et al. 2016; Segan et al. 2015; Thorne et al. 2013) are not useful as they do not identify which interventions are likely to be effective for a species. Existing trait-based vulnerability frameworks for climate change are not explicitly linked to specific management actions (e.g. Angert et al. 2011; Foden et al. 2013; Cabrelli et al. 2014; Garcia et al. 2014), which may reduce their capacity to be implemented by practitioners and agencies (Willis et al. 2015). Effective assessments of vulnerability should include both extrinsic and intrinsic factors (cf. Chessman 2013; Foden et al. 2013), to inform conservation planning.
Trait-based tools for assessing vulnerability in terms of climate change risk may be particularly useful in species with long-generation times. Longer-lived species, such as trees and shrubs, have lower capacity for genetic recombination and less opportunity to accumulate potentially beneficial mutations—particularly where effective population sizes are low—which may influence their ability to adapt (Skelly et al. 2007). For these species, focusing on the intrinsic traits involved in response capacity when evaluating management options may be more informative. In shorter-lived groups, such as insects, there is clear evidence that genetic adaptation to rapidly changing climate can occur (Hoffmann and Sgrò 2011). Whilst a recent review showed that although plasticity and evolutionary change are already occurring in plants exposed to climate change, the rate of climate change may be too fast to allow these processes to effectively act upon populations (Franks et al. 2014). Without accounting for the intrinsic elements of vulnerability that can be captured by traits, and using the implications to link to appropriate management actions, conservation planning is unlikely to succeed in the medium and long term.
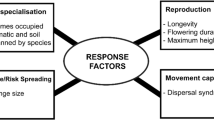
In this study, we outline a new approach, which integrates climate change risk and conservation actions by incorporating information on the traits and range metrics of species, using plants as an example. We combine data on morphological and phenological traits (dispersal mode, maximum height, flowering duration, longevity) with metrics of species’ range (range size, climate and soil niche breadth, habitat breadth, biomes occupied) to classify 1237 Australian plant species by their potential capacity to respond to climate change. Our overarching goal is to transform the way in which we manage species by explicitly considering how traits and range characteristics limit species’ capacity to respond to changing climate, and recommend management actions based on these characteristics which may be most appropriate, and ultimately successful, for conservation. We consider traits and range metrics hypothesised to be factors in determining Australian plant species’ capacity to respond to climate change, and explicitly relate them to a suite of commonly proposed conservation actions. By thus linking potential management actions and the factors on which they can act to maximise response capacity, we demonstrate a more effective way of managing for species’ persistence (Fig. 1). We also cross-reference our list of high risk species with lists of nationally protected species to highlight inadequacies in current management approaches for effectively planning for climate change.
Vulnerability by factors for response capacity (as driven by traits and range metrics, Table 1). In-situ and ex-situ management actions grouped as: Habitat; Landscape; Interspecific interaction; Species-specific and Translocation. The factors are linked to the actions that can act to maximise response capacity/resilience
2 Methods
2.1 Traits and metrics
We targeted four traits and six range metrics hypothesised to be important factors in determining species response capacity to climate change (Bush et al. 2016; Damschen et al. 2012; Fordham et al. 2012; Franks et al. 2014; Slatyer et al. 2013; Urban 2015). The traits and metrics were: longevity, flowering duration, dispersal mode, maximum height, range size, niche breadths for rainfall, temperature, soil fertility [clay content, total phosphorus], and the number of biomes occupied (Table 1). Using available information for these traits, this enabled us to score climate change risk for 1237 Australian plant species using a trait-based tool designed to assess how species reproduction, movement, abiotic niche specialisation and spatial coverage may limit their ability to respond (factors) (see Fig. 2 for examples).
Our dataset includes a broad selection of plant species native to Australia that are drawn from 21% of genera (410 of the 1933) in the Australian Plant Census, within 116 of the 198 higher plant families (see Supplementary Information for full list of taxa). Species occur across all the biomes present in Australia according to Olsen et al. (2011) and our dataset contains several representatives of the two largest and most ecologically dominant Australian genera, Acacia (n = 146 species) and Eucalyptus (n = 34 species). Species taxonomy was standardised to The Plant List (www.theplantlist.org) using the R package Taxonstand (Cayuela et al. 2012).
All trait data were sourced from published floras held within the Austraits database—a compilation of trait measurements for the Australian flora. All range metrics were calculated by matching cleaned occurrence records from vouchered herbarium specimens used in Gallagher (2016) to gridded climatologies for current conditions (1950–2000) from the ANUCLIM 1.0 dataset (http://www.emast.org.au/our-infrastructure/observations/anuclimate_data/) at a 5-arc-minute resolution, biome boundaries in Olsen et al. (2011), and soil conditions in the National Soil Attribute Maps (www.clw.csiro.au/aclep/soilandlandscapegrid/index.html). Range size (km2) was calculated as the area inside a modified convex-hull encompassing all cleaned occurrences (alpha-hull; Burgman and Fox 2003; Gallagher 2016). Niche breadths for climate and soil conditions were calculated as the difference between the upper (98th percentile) and lower (2nd percentile) values for each variable across the species range. Using traits and range metric values, climate change risk was quantitatively scored within in each factor (see “Reproduction”, “Movement capability”, “Abiotic niche specialisation”, “Spatial coverage”) and then assigned to a category (low, medium, high). Below we outline the justification for trait selection and scoring scheme for each factor.
2.1.1 Reproduction
Species’ ability to respond and adapt to changing climatic conditions in situ will be determined, in part, by the rate of population turnover, which is linked to an intrinsic rate of increase (Fordham et al. 2012). Species with shorter generation lengths (time to maturity) are expected to have a faster population turnover, and as a result benefit from greater opportunities for evolutionary or epigenetic change in response to rapid climate change (Bush et al. 2016; Hughes 2000; Franks et al. 2014). We used plant longevity (annual, biennial, perennial or combinations thereof), and maximum height (m) as proxies for generation length. On average, taller species have longer generation times than do shorter species (Moles et al. 2009). We also used duration of the flowering period (months) as a measure of the opportunity for reproductive success, with increasing flowering duration being associated with greater seed set and rate of population increase (Gibson et al. 2011). Species were assigned numerical scores for each trait (Table 1) which were then used to calculate a geometric mean for ranking species by their reproductive capacity, with lower scores conferring lower climate change risk. Geometric means for reproduction ranged between 1.26 and 4.16. We split geometric mean scores across all species into three equal-sized categories (low, medium, and high vulnerability) using the 33rd and 66th percentiles (low < 3.1, medium 3.1–3.6, high > 3.6).
2.1.2 Movement capability
Species’ ability to shift distributional range to track optimal conditions for growth and survival is a fundamental factor for their capacity to respond to climate change. Species whose capacity to range shift is low are at a greater risk of extinction, particularly where their exposure to the effects of climate change is high (Fordham et al. 2012). We scored species’ potential capability for movement based on their dispersal syndrome, assuming that species capable of moving longer distances are more likely to be able to spread to suitable or novel habitats (corresponding to lower vulnerability). The connection between seed dispersal syndromes and species’ movement capability has been established across multiple disciplines operating at different temporal and spatial domains, including paleobotany (Eriksson et al. 2000), biogeography (Seidler and Plotkin 2006), and trait ecology (Willson and Traveset 2000). We allocated species into four categories according to seed dispersal mechanism ((1) wind, (2) vertebrate, (3) invertebrate and (4) localised (seeds with no apparent dispersal appendage which rely on gravity or explosive dehiscence for dispersal); Table 1). Species were then assigned to low, medium and high movement capability (based on scores 1 = low, 2 = medium, 3–4 = high), with higher scores indicating a greater climate change risk. Species with multiple dispersal syndromes were assigned their lowest potential score (e.g. Themeda triandra Forsk. has adaptations for dispersal both by wind and in the fur of vertebrates and so received a score of 1).
2.1.3 Abiotic niche specialisation
Plant species adapted to a narrow suite of abiotic conditions across their realised niche (e.g. infertile soils, ephemeral rainfall) are more likely to be ecological specialists with a lower inherent capacity to respond changing conditions (Ackerly 2003; Slatyer et al. 2013). For these species, the combination of rapidly changing climate and relatively stable soil conditions may lead to a mismatch in suitable conditions for population persistence (Damschen et al. 2012). Species with wider realised niche breadths are expected to have greater ability to cope with diverse abiotic conditions, relative to those species which occupy narrow abiotic niches. To assess the role of abiotic niche specialisation in climate change risk we used data on the breadth of rainfall (annual precipitation (AP); mm), temperature (mean annual temperature (MAT); °C), soil P content (total P; %) and soil clay content (clay %) in the top 5 cm of the soil profile. The two climate variables are commonly used to summarise broad-scale niche requirements and influence vegetation structure across the strong north-south temperature and east-west rainfall gradients in Australia (Groves 1994). Similarly, low soil fertility—in particular low phosphorus contents—has driven key ecological adaptations in the Australian flora (e.g. cluster roots, sclerophylly; Beadle 1966; Lambers et al. 2006). In addition, we also assessed how many of Australia’s biomes each species occupies as a measure of specialisation, based on the biome classification in Olsen et al. (2011). Species were assigned numerical scores based on the width of their niche and number of biomes occupied (Table 1). These scores were then used to calculate a geometric mean across all scores for ranking species by their specialisation and assigning them to low, medium and high climate change risk categories. Geometric means, used to allow for combination of values across disparate ranges and units, ranged between 1.0 and 5.1 and were split into three equal-sized categories at the 33rd and 66th percentiles, as with the reproduction category.
2.1.4 Spatial coverage
Range size is a well-established surrogate for extinction risk, commonly used in conservation assessments and declarations (Mace et al. 2008). Species with small range sizes are at an increased risk of extinction primarily because singular deterministic or stochastic events are more likely to affect their entire population (IUCN 2016). To assign species into low-, medium- and high-risk categories based on their range size across Australia, we used a combination of the IUCN thresholds for listing species under the “restricted geographic distribution” clause, and the percent coverage of the species across Australia (Table 1). Specifically, species with a range size small enough to be listed under the threshold for critically endangered, endangered or vulnerable in the IUCN Red List criteria (i.e. 20,000 km2, for extent of occurrence, EOO) were scored as high-risk. Medium risk species were those which had range sizes greater than the IUCN threshold, but occupied less than 1% of the Australian continent by land area (i.e. 70,000 km2). All other species were assigned as low-risk for the spatial coverage category.
2.2 Categorical risk framework
We used the categorisation of each factor as low-, medium- or high-risk for each species to assign an overall climate change risk for each species. The overall risk (LOW, MEDIUM or HIGH), was determined using three methods considering different possible combinations of categories: consensus, equality or mixed combinations (see Fig. 2 for examples). Where three or more factors shared the same risk category (n = 555 species), consensus was considered to be reached and that category was assigned (e.g. HIGH overall risk for Beilschmiedia peninsularis B.P.M. Hyland). Where an equal split of categories occurred across factors (n = 246 species) the higher of the two categories was assigned (e.g. two medium, two high = HIGH [e.g. Polyalthia patinata Jessup.]; or two medium, two low = MEDIUM [e.g. Carpobrotus glaucescens (Haw.) Schwantes]). For all other mixed combinations of risk categories (n = 991 species) species were assigned as MEDIUM if one high category was present (e.g. Persoonia oxycoccoides Sieber ex Spreng.).
2.3 Management streams based on climate change risk
We used species’ capacity to respond to climate change—as captured by their risk scores for each factor (reproduction, movement capability, abiotic niche specialisation, spatial coverage), to link them with appropriate management actions. We divided conservation management actions into five groupings accounting for scale, location and biotic interactions: habitat, landscape, interspecific interaction and species-specific translocation (Fig. 1). Plant conservation management actions were collated from management plans and the conservation literature, allocated to one of these five broad groupings. Conservation actions for plants can be broadly characterised as in-situ or ex-situ, and a mix of these strategies is represented in the groupings (Fig. 1).
We investigated how the results of our analysis mapped to national regulations for listing species as threatened, using the Environmental Biodiversity Protection (EPBC) Act (1999) (http://www.environment.gov.au/epbc). The EPBC Act lists species under three categories associated with increasing threat levels; vulnerable, endangered and critically endangered, which are broadly aligned with the listing guidelines for the IUCN Red List (IUCN 2016). We used chi-square analysis to test for equality between our risk scores and the EPBC list categories. Whilst anthropogenic climate change is listed as a threatening process under the Act, its interaction with species’ response traits is not incorporated in assessments of species’ vulnerability to extinction, whereas our framework explicitly accounts for these factors.
To compare the types of conservation actions currently recommended for species listed on the EPBC Act, we extracted information on proposed management options for species, ranked as HIGH in our dataset and with EPBC Act schedules, from their approved conservation advice or recovery plan (www.environment.gov.au/biodiversity/threatened/conservation-advices). We grouped the recommended management options into our five categories (as above: habitat; landscape; interspecific interaction; species-specific; translocation) and assessed where current management recommendations fail to recognise traits related to response capacity to climate change, and thus missed recommending particular management actions.
3 Results
3.1 Risk scores
Of the 1237 species assessed for climate change risk, 242 (20%), 707 (57%) and 288 (23%) were categorised as HIGH, MEDIUM and LOW, respectively. Species were more likely to be at high-risk due to reproduction or specialisation factors (n = 554 species and 490 species, respectively), and least likely to be at high-risk due to spatial limitations (n = 229 species; Fig. 3). Overall, most species (n = 707 species) fell under the MEDIUM risk category, which was also the case for movement capability as a factor (n = 673 species).
Numbers of 1237 Australian plant species classified into HIGH vulnerability (black), MEDIUM vulnerability (dark grey) and LOW vulnerability (pale grey) categories, on the basis of traits affecting four factors: specialisation, movement capability, spatial coverage and reproduction, and overall vulnerability
3.2 Legislative protection status of assessed species
A small fraction of the 1237 species assessed (n = 29 species; 2%) are already formally listed as threatened in Australia under the EPBC Act (Table 2). Of these 29 species, the majority were categorised as HIGH risk in our framework (n = 24); ten of these were listed in the endangered category, and 14 in the vulnerable category. The remaining five species were categorised as MEDIUM risk and listed as vulnerable. No species with a LOW risk as identified by our framework were listed under the EPBC Act. Chi-square analysis indicated no equality between species’ climate change risk and their EPBC list groups (X2 = 78.9; df = 4; P < 0.0001): species that were at HIGH risk due to low response capacity in our framework were not necessarily listed as threatened (vulnerable, endangered or critically endangered) by the EPBC.
3.3 Adequacy of conservation actions for management of response capacity
To compare the types of conservation actions currently recommended for species listed in the EPBC Act, we extracted information on proposed management options for species, ranked as HIGH risk in our dataset and with EPBC Act schedules, from their approved conservation advice or recovery plan.
Of the 24 species in the HIGH risk category in our analysis, only seven have actions recommended in their conservation advice under the EPBC Act that effectively incorporate the management of their response capacity (i.e. Acacia flocktoniae, Acacia gordonii, Acacia handonis, Acacia macnuttiana, Allocasuarina fibrosa, Cryptocarya foetida, Persoonia marginata; Table 3). Therefore, ~ 70% of species classified as at high climate change risk, and already listed in the EPBC Act, lack management plans that include appropriate actions based on their intrinsic traits. Overall, all but two species had recommended actions under the management action stream Habitat; all save three under Species-specific; all but four under Translocation; ten under Landscape and one under Interspecific interaction. Additional action streams suggested by our analysis are largely landscape-related, and focus on improving connectivity between populations and patches, in many cases at local and regional scale (Table 3).
4 Discussion
This study quantifies the inadequacy of current management approaches for maximising the response capacity of plant species to climate change. We show how using information on plant traits and range characteristics, which govern species’ sensitivity and adaptive capacity, and thus capacity to respond to climate change, to both assess risk and inform conservation actions could improve management and persistence outcomes for species. Our trait-based analyses, based on universal plant traits (e.g. longevity, dispersal syndrome, niche breadth), show that 20% of the flora examined (n = 1237 species), is potentially at high climate change risk. Of the 242 high-risk species, only a small fraction (10%; n = 24 species) are currently listed for protection under national conservation legislation for Australia (EPBC Act). Importantly, many management plans for these listed species fail to detail explicit actions which may preserve, or increase, the capacity of threatened species to respond to climate change. That is, 70% of management plans assessed (i.e. approved conservation advice, species recovery plans) did not make a clear link between proposed conservation actions and improvements in species responses to climate change. This omission of targeted management actions, coupled with the large numbers of vulnerable but currently unprotected species, may be problematic given the rapid pace of global change.
The 24 EPBC Act listed species have a conservation status of vulnerable or endangered, whereas our analysis suggested that they could justifiably be listed as critically endangered, given their high-risk due to their low capacity to respond to climate change. These species are at high-risk due to specialisation and spatial scale factors, and most of them due to reproduction factors. However, the management actions given for their conservation do not necessarily reflect this. For example, Syzygium moorei, a tree listed as vulnerable by the EPBC Act, is HIGH in our analysis, suggesting that there is more urgency required for conservation actions. It is at risk due to reproduction, specialisation, and spatial scale factors, and whilst the EPBC Act advice sets out several effective measures for addressing the reproduction and specialisation factors, such as seed germination and propagation, site protection and ex-situ translocation, it does not include maintenance or restoration of habitat quality, or, in relation to the spatial scale factor, the creation of corridors or promotion of landscape connectivity or buffer zones (Fig. 1). In addition, EPBC plans are not necessarily implemented when they reach the funding stage. For example, several species scored in the ‘Translocation’ action group due to the statement: “Implement national translocation protocols (Vallee et al. 2004) if establishing additional populations is considered necessary and feasible” in their EPBC Conservation Advice document. However, if the trait factors are not taken into account when their vulnerability is assessed, this may not be “considered necessary” when action is actually taken.
Species will not be equally exposed to the effects of climate change due to spatial variation in the magnitude and pace of temperature and precipitation changes (IPCC 2013). In Australia, climate is projected to change in terms of significant decreases in seasonal rainfall and increases in temperature as a result of the subtropical zone expanding southwards (Cai et al. 2012), which will especially threaten tree and forest species. Drought incidence has increased and will continue to do so (Nicholls 2004), whilst in some areas monsoon rainfall has intensified (Smith 2004). These changes will substantially increase the risk to species with narrow thermal or precipitation niches, for example, species in the southwest of Western Australia or the Australian alpine zone. Further, these climatic changes may also drive phenological shifts in flowering duration, which may mean that species may become more vulnerable, for example, where flowering periods are reduced. Future climates must therefore be integrated into on-the-ground management planning, for example, by using downscaling of climate variables (Fowler and Wilby 2007).
4.1 Trait-based approaches to allocating management actions
Traits and range characteristics are useful tools for screening and prioritising taxa for placement into particular management streams. However, we caution against traits being viewed as a panacea for addressing the ever-growing number of species that require conservation attention. We have shown here that traits can be useful in prioritising the climate change risk of species and guiding management actions. Equally, recommended management actions should not be implemented without deeper consideration of the biology and ecological or environmental context of the target species—a process which often requires more nuanced experimental work or field surveys to ensure adequate data analysis is possible (Gosper et al. 2016). In short, prioritisation schemes and risk or vulnerability frameworks are the headwaters of the management stream, intended to complement—not overshadow—on-ground knowledge of biodiversity.
Explicitly linking species’ response capacity to climate change to appropriate management actions has the potential to achieve more effective long-term conservation actions and reduce extinction risks (Fortuni and Schubert 2017; Urban 2015). Species sensitivity and adaptive capacity are increasingly being quantified (e.g. Foden et al. 2013; Gardali et al. 2012; Lee et al. 2015; Summers et al. 2012; Wade et al. 2017), but few frameworks exist, to our knowledge, for connecting this information to appropriate on-ground actions (see Christmas et al. 2016 for perspectives on management of adaptive genetic variation). To make this connection effective, management decisions should be further prefaced by consideration of key factors which may limit success, such as species biology and the cost-benefit trade-off of investment in particular conservation actions (Murdoch et al. 2007; McDonald-Madden et al. 2008). Many conservation actions proposed here to accommodate species response capacity may positively affect co-occurring species, including landscape and habitat actions (e.g. restoration, invasive species management, increasing connectivity between populations). However, some actions—like translocation—are likely to be highly species-specific, reducing the net benefit of conservation actions on wider suites of species (Summers et al. 2015). Similarly, management actions may have perverse outcomes; for instance, increased connectivity providing a conduit for the movement of invasive species and facilitating undesirable shifts in vegetation composition (Leishman and Gallagher 2015).
Where the interaction of climate change with species’ response capacity is not incorporated in assessments of species’ risk, some key opportunities for more targeted interventions, likely to be more successful, are being missed. An important consideration is whether the action actually addresses the trait driving risk. For example, if a management action for a species is indicated to be translocation, as its risk is related to spatial scale, sourcing individuals from more widely distributed populations, as well as from its local distribution, can mimic dispersal (composite provenancing), ensuring that a range of adaptations is captured in the new population, which may mean it will not be so spatially restricted (Gillson et al. 2013). Similarly, where movement is the factor, enhanced connectivity between suitable habitats may be indicated as an effective management action. If niche specialisation is also a factor for the species, it is not enough to create a corridor across the landscape, as the quality of the habitat within the corridor will also need to match the species’ requirements.
5 Conclusion
Changes in species and community composition, ecosystem function (and service provision) may be rapid in response to global change, and species’ capacity to respond to new environments, habitats or ecosystems will determine whether they can persist (Moritz and Agudo 2013; Dawson et al. 2011). Given limited resources to address conservation problems, how do we best allocate them to maximise the long-term integrity and persistence of species under threats such as climate change? Investing in conservation where the probability of success is low is a lose-lose situation: not only does it not achieve persistence of the target species; it diverts scarce resources from other species and ecosystems that could have been helped.
There are many established protocols for prioritising species based on their extinction risk; however, they may not adequately capture the potential effects of climate change in their current form (Hannah et al. 2012; Thomas et al. 2004). Species with a low capacity to respond to climate change are often not being allocated to the highest level of threat, and are therefore not well-placed for long-term protection. Incorporating life history trait information could be a relatively straightforward way of ensuring that the best decisions can be made for supporting species’ persistence under climate change.
References
Ackerly DD (2003) Community assembly, niche conservatism, and adaptive evolution in changing environments. Int J Plant Sci 164:165–184
Angert AL, Crozier LG, Rissler LJ et al (2011) Do species’ traits predict recent shifts at expanding range edges? Ecol Lett 14:677–689
Araújo MB, Ferri-Yáñez F, Bozinovic F et al (2013) Heat freezes niche evolution. Ecol Lett 16:1206–1219
Beadle NCW (1966) Soil phosphate and its role in molding segments of the Australian flora and vegetation, with special reference to xeromorphy and sclerophylly. Ecology 47:992–1007
Beever EA, O'Leary J, Mengelt C et al (2016) Improving conservation outcomes with a new paradigm for understanding species’ fundamental and realized adaptive capacity. Conserv Lett 9:131–137
Bellard C, Bertelsmeier C, Leadley P et al (2012) Impacts of climate change on the future of biodiversity. Ecol Lett 15:365–377
Burgman MA, Fox JC (2003) Bias in species range estimates from minimum convex polygons: implications for conservation and options for improved planning. In Anim Conserv forum (Vol. 6, No. 1, pp. 19–28). Cambridge University Press
Butchart SHM, Walpole M, Collen B et al (2010) Global biodiversity: indicators of recent declines. Science 328:1164–1168
Bush A, Mokany K, Catullo R et al (2016) Incorporating evolutionary adaptation in species distribution modelling reduces projected vulnerability to climate change. Ecol Lett 19:1468–1478
Butt N, Pollock L, McAlpine C (2013) Eucalypts face increasing climate stress. Ecology and Evolution 3:5011–5022. https://doi.org/10.1002/ece3.873
Butt N, Possingham HP, De Los Rios C et al (2016) Challenges in assessing the vulnerability of species to climate change to inform conservation actions. Biol Conserv 199:10–15
Cabrelli AL, Stow AJ, Hughes L (2014) A framework for assessing the vulnerability of species to climate change: a case study of the Australian elapid snakes. Biodivers Conserv 23:3019
Cai WJ, Cowan T, Thatcher M (2012) Rainfall reductions over southern hemisphere semi-arid regions: the role of subtropical dry zone expansion. Sci Rep 2:702. https://doi.org/10.1038/srep00702
Cayuela L, Granzow-de la Cerda I, Albuquerque FS, Golicher DJ (2012) Taxonstand: an R package for species names standardisation in vegetation databases. Methods Ecol Evol 3:1078–1083
Chen IC, Hill JK, Ohlemüller R (2011) Rapid range shifts of species associated with high levels of climate warming. Science 333:1024–1026
Chessman BC (2013) Identifying species at risk from climate change: Traits predict the vulnerability of freshwater fishes. Biol Conserv 160:40–49
Christmas MJ, Breed MF, Lowe AJ (2016) Constraints to and conservation implications for climate change adaptation in plants. Conserv Genet 17:305
Dawson TP, Jackson SY, House JI et al (2011) Beyond predictions: biodiversity conservation in a changing climate. Science 332:53–58
Damschen EI, Harrison S, Ackerly DD et al (2012) Endemic plant communities on special soils: early victims or hardy survivors of climate change? J Ecol 100:1122–1130
Eriksson O, Friis EM, Löfgren P (2000) Seed size, fruit size, and dispersal systems in angiosperms from the early cretaceous to the late tertiary. Am Nat 156:47–58
Foden WB, Butchart SHM, Stuart SN et al (2013) Identifying the world’s most climate change vulnerable species: a systematic trait-based assessment of all birds, amphibians and corals. PLoS One 8(6):e65427. https://doi.org/10.1371/journal.pone.0065427
Fordham DA, Resit Akçakaya H, Araújo MB et al (2012) Plant extinction risk under climate change: are forecast range shifts alone a good indicator of species vulnerability to global warming? Glob Chang Biol 18:1357–1371. https://doi.org/10.1111/j.1365-2486.2011.02614.x
Fortuni L, Schubert O (2017) Beyond exposure, sensitivity and adaptive capacity: a response based ecological framework to assess species climate change vulnerability. Climate Change Responses 4:2. https://doi.org/10.1186/s40665-017-0030-y
Fowler HJ, Wilby RL (2007) Beyond the downscaling comparison study. Int J Climatol 27:1543–1545. https://doi.org/10.1002/joc.1616
Franks SJ, Weber JJ, Aitken SN (2014) Evolutionary and plastic responses to climate change in terrestrial plant populations. Evol Appl 7:123–139
Gallagher RV (2016) Correlates of range size variation in the Australian seed-plant flora. J Biogeogr 43:1287–1298
Gardali T, Seavy NE, DiGaudio RT, Comrack LA (2012) A climate change vulnerability assessment of California’s at-risk birds. PLoS One 7:e29507
Garcia RA, Araújo MB, Burgess ND et al (2014) Matching species traits to projected threats and opportunities from climate change. J Biogeogr 41:724–735
Gibson MR, Richardson DM, Marchante E et al (2011) Reproductive biology of Australian acacias: important mediator of invasiveness? Divers Distrib 17:911–933
Gillson L, Dawson TP, Jack S, McGeoch MA (2013) Accommodating climate change contingencies in conservation strategy. TREE 28:135–142
Gosper CR, O'Donnell AJ, Yates CJ et al (2016) Better planning outcomes require adequate data and ecological understanding to be successful and credible: a reply to Evans et al. 2015. Biol Conserv 200:240–241
Groves RH (1994) Australian vegetation. Cambridge University Press, Cambridge
Hejda M, Hanzelka J, Kadlec T, Štrobl M, Pyšek P, Reif J (2017) Impacts of an invasive tree across trophic levels: species richness, community composition and resident species’ traits. Divers Distrib 23:997–1007. https://doi.org/10.1111/ddi.12596
Hoekstra JM, Boucher TM, Ricketts TH, Roberts C (2005) Confronting a biome crisis: global disparities of habitat loss and protection. Ecol Lett 8:23–29
Hoffmann AA, Sgrò CM (2011) Climate change and evolutionary adaptation. Nature 470:479–485
Hooper DU, Adair EC, Cardinale BJ et al (2012) A global synthesis reveals biodiversity loss as a major driver of ecosystem change. Nature 486:105–108
Hughes L (2000) Biological consequences of global warming: is the signal already apparent? TREE 15:56–61
IPCC (2013) Climate change 2013: the physical science basis. Contribution of working group I to the fifth assessment report of the intergovernmental panel on climate change (eds Stocker TF, Qin D, Plattner G-K, et al.), IPCC, Cambridge
IUCN Standards and Petitions Subcommittee (2016) Guidelines for using the IUCN red list categories and criteria. Version 12. Prepared by the Standards and Petitions Subcommittee http://www.iucnredlist.org/documents/RedListGuidelines.pdf
Lambers H, Shane MW, Cramer MD et al (2006) Root structure and functioning for efficient acquisition of phosphorus: matching morphological and physiological traits. Ann Bot 98:693–713
Lee JR, Maggini R, Taylor MFJ, Fuller RA (2015) Mapping the drivers of climate change vulnerability for Australia's threatened species. PLoS One 10:e0124766
Leishman MR, Gallagher RV (2015) Will there be a shift to alien-dominated vegetation assemblages under climate change? Divers Distrib 21:848–852
McDonald-Madden E, Baxter PW, Possingham HP (2008) Making robust decisions for conservation with restricted money and knowledge. J Appl Ecol 45:1630–1638
Mace GM, Collar NJ, Gaston KJ et al (2008) Quantification of extinction risk: IUCN's system for classifying threatened species. Conserv Biol 22:1424–1442
Moles AT, Warton DI, Warman L et al (2009) Global patterns in plant height. J Ecol 97:923–932
Moritz C, Agudo R (2013) The future of species under climate change: resilience or decline? Science 341:504–508
Murdoch W, Polasky S, Wilson KA et al (2007) Maximizing return on investment in conservation. Biol Conserv 139:375–388
Nicholls N (2004) The changing nature of Australian droughts. Clim Chang 63:323–336
Nicotra AB, Atkin OK, Bonser SP et al (2010) Plant phenotypic plasticity in a changing climate. Trends Plant Sci 15:684–692
Nicotra AB, Beever EA, Robertson AL et al (2015) Assessing the components of adaptive capacity to improve conservation and management efforts under global change. Conserv Biol 29:1268–1278
Olsen DM, Dinerstein E, Wikramanayake ED et al (2011) Terrestrial ecoregions of the world: a new map of life on earth: a new global map of terrestrial ecoregions provides an innovative tool for conserving biodiversity. BioScience 51:933–938
Pacifici M, Foden WB, Visconti P et al (2015) Assessing species vulnerability to climate change. Nat Clim Chang 5:215–224
Pearson RG, Stanton JC, Shoemaker KT et al (2014) Life history and spatial traits predict extinction risk due to climate change. Nat Clim Chang 4:217–221
Pecl GT, Araújo MB, Bell JD et al (2017) Biodiversity redistribution under climate change: impacts on ecosystems and human well-being. Science 355:eaai9214
Pyšek P, Jarošík V, Hulme PE, Pergl J, Hejda M, Schaffner U, Vilà M (2012) A global assessment of invasive plant impacts on resident species, communities and ecosystems: the interaction of impact measures, invading species' traits and environment. Glob Chang Biol 18:1725–1737. https://doi.org/10.1111/j.1365-2486.2011.02636.x
Segan DB, Hole DG, Donatti CI et al (2015) Considering the impact of climate change on human communities significantly alters the outcome of species and site-based vulnerability assessments. Divers Distrib 21:1101–1111
Seidler TG, Plotkin JB (2006) Seed dispersal and spatial pattern in tropical trees. PLoS Biol 4:e344
Skelly DK, Joseph LN, Possingham HP et al (2007) Evolutionary responses to climate change. Conserv Biol 21:1353–1355
Slatyer RA, Hirst M, Sexton JP (2013) Niche breadth predicts geographical range size: a general ecological pattern. Ecol Lett 16:1104–1114
Smith IN (2004) Trends in Australian rainfall: are they unusual? Aust Meteorol Mag 53:11
Stohlgren TJ, Schnase JL (2006) Risk analysis for biological hazards: what we need to know about invasive species. Risk Anal 26:163–173. https://doi.org/10.1111/j.1539-6924.2006.00707.x
Summers DM, Bryan BA, Crossman ND, Meyer WS (2012) Species vulnerability to climate change: impacts on spatial conservation priorities and species representation. Glob Chang Biol 18:2335–2348
Thomas CD, Cameron A, Green RE et al (2004) Extinction risk from climate change. Nature 427:145–147
Thorne JH, Seo C, Basabose M, et al. (2013) Alternative biological assumptions strongly influence models of climate change effects on mountain gorillas. Ecosphere 4
Urban MC (2015) Accelerating extinction risk from climate change. Science 348:571–573
Vallee L, Hogbin T, Monks L et al (2004) Guidelines for the translocation of threatened plants in Australia, Second edn. Australian Network for Plant Conservation, Canberra
Wade AA, Hand BK, Kovach RP et al (2017) accounting for adaptive capacity and uncertainty in assessments of species’ climate-change vulnerability. Conserv Biol 31:136–149
Willis SG, Foden W, Baker DJ et al (2015) Integrating climate change vulnerability assessments from species distribution models and trait-based approaches. Biol Conserv 190:167–178
Willson MF, Traveset A (2000) The ecology of seed dispersal. Seeds 2:85–110
WWF (2016) Living planet report 2016. Risk and resilience in a new era. WWF International, Gland, Switzerland
Acknowledgements
Both authors are supported by Australian Research Council Discovery Early Career Awards (DE150101552 to N. Butt and DE170100208 to R. Gallagher). We are grateful to Lesley Hughes and Tony Auld for helpful discussions, and thank Jessica O’Donnell for her assistance with figures.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
Dataset of response capacity for 1237 plant species. (XLSX 170 kb)
Rights and permissions
About this article
Cite this article
Butt, N., Gallagher, R. Using species traits to guide conservation actions under climate change. Climatic Change 151, 317–332 (2018). https://doi.org/10.1007/s10584-018-2294-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10584-018-2294-z