Abstract
The Apolipoprotein E ε4 (ApoE ε4) allele, encoding ApoE4, is the strongest genetic risk factor for late-onset Alzheimer’s disease (LOAD). Emerging epidemiological evidence indicated that ApoE4 contributes to AD through influencing β-amyloid (Aβ) deposition and clearance. However, the molecular mechanisms of ApoE4 involved in AD pathogenesis remains unclear. Here, we introduced the structure and functions of ApoE isoforms, and then we reviewed the potential mechanisms of ApoE4 in the AD pathogenesis, including the effect of ApoE4 on Aβ pathology, and tau phosphorylation, oxidative stress; synaptic function, cholesterol transport, and mitochondrial dysfunction; sleep disturbances and cerebrovascular integrity in the AD brains. Furthermore, we discussed the available strategies for AD treatments that target to ApoE4. In general, this review overviews the potential roles of ApoE4 in the AD development and suggests some therapeutic approaches for AD.
Graphical Abstract
ApoE4 is genetic risk of AD. ApoE4 is involved in the AD pathogenesis. Aβ deposition, NFT, oxidative stress, abnormal cholesterol, mitochondrial dysfunction and neuroinflammation could be observed in the brains with ApoE4. Targeting the interaction of ApoE4 with the AD pathology is available strategy for AD treatments.

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Alzheimer’s disease (AD) is a progressive neurodegenerative disease affecting people over the age of 65. AD is mainly characterized by senile plaques (SPs) (Jack et al. 2010) and neurofibrillary tangles (NFTs) (Muralidar et al. 2020), constituting two major hypotheses in the pathogenesis of AD; the β-amyloid cascade hypothesis and tau protein hyperphosphorylation, respectively. Additionally, synaptic loss and synaptic damage (John and Reddy 2021) can be observed in the early stage of AD, as well as abnormal cholesterol transport (Kao et al. 2020).
Apolipoprotein E (ApoE) interacts with β-amyloid (Aβ) and regulates its aggregation and clearance in the AD brain. Furthermore, ApoE contributes to AD pathogenesis by modulating brain synaptic plasticity (Kim et al. 2014), glucose metabolism (Jiang et al. 2020), neuronal signaling (Huang et al. 2019), oxidative stress (Jofre-Monseny et al. 2008), neuroinflammation (Lanfranco et al. 2021), mitochondrial dysfunction (Yin et al. 2020) and cholesterol transportation (Jeong et al. 2019). The ApoE ε4 allele, encoding ApoE4, is the strongest risk factor for late-onset AD (LOAD); more than 65% of patients suffering from LOAD are found to carry at least one ε4 allele at clinical diagnosis (Saunders et al. 1993). The frequency of the ε4 allele in the population is about 15–20%, and there are some variations of this frequency among different ethnic groups. Carriers with one ε4 allele have a threefold increasing risk of AD, and carriers of both ε4 alleles have up to 90% risk of AD (Liu et al. 2013). Reversely, carriers with ε2 allele could slow the pathological process of AD. ApoE ε3 allele is a commonly occurring subtype in the population with a neutral risk of AD prevalence. ApoE4 has been found to be involved in AD development by mediating Aβ pathology (Tachibana et al. 2019), NFT load (Sabbagh et al. 2013), synaptic impairment (Sun et al. 2017) and abnormal cholesterol transport (Dunk and Driscoll 2022). In addition, ApoE4 mice always showed high level of reactive oxygen species (ROS) (Lauderback et al. 2002). ApoE4 is also associated with mitochondrial functions by regulating its biogenesis and dynamics (Yin et al. 2020). Furthermore, ApoE4 increases the expression of inflammatory factors and induces neuroinflammation (Iannucci et al. 2021). Currently, the therapies targeting to ApoE4 mainly concentrate on the interaction of ApoE with Aβ (Kuszczyk et al. 2013), ApoE receptors (Shi et al. 2021), ApoE4 genotype correction (Wang et al. 2018), ApoE antibody (Liao et al. 2018), and nonpharmacological therapy (Liu et al. 2014b). Therefore, understanding the link between ApoE4 and AD pathological mechanisms will help develop more strategies on diagnostic and therapy for AD patients.
Biology of ApoE
The Structure and Localization of ApoE
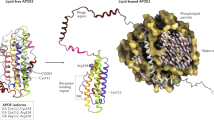
ApoE is a secreted glycoprotein with 299 amino acid residues (Mr = 34kd). The C-terminal domain of ApoE is responsible for binging to cholesterol and phospholipids, and the N-terminal domain comprises some receptors-binding regions (Jones et al. 2011). The three common isoforms of ApoE are ApoE2, ApoE3 and ApoE4 (Zannis et al. 1981) encoded by three allelic variants ɛ2, ɛ3 and ɛ4, respectively. Genetic variations of the three isoforms are induced by single nucleotide polymorphisms (SNP) that differ by two amino acid residues of ApoE at positions 112 and 158, labeled rs429358 (C > T) and rs7412 (C > T) (Weisgraber et al. 1981). ApoE3 is cysteine (Cys) at position 112 and arginine (Arg) at position 158. ApoE2 is Cys at the both positions and ApoE4 is Arg at the both positions (Fig. 1). The Arg at position 112 of ApoE4 contributes to Arg61-Glu255 and Arg112-Glu109 salt bridges, leading to interactions of domains between the N-terminal and C-terminal. Furthermore, salt bridge structure is also observed in ApoE3, showing at Arg61-Asp65. Differences in salt bridges could be one of the reasons affecting the ability of ApoE isoforms to form complexes with Aβ, and mutating ApoE4 to Ala or Lys at position 112 could eliminate complex formation (Bentley et al. 2002). In general, compared with ApoE2 and ApoE3, ApoE4 is more likely to form molten globules and aggregate at 37 °C (Morrow et al. 2002), which could be implicated abnormal physiological functions.
In periphery, ApoE is secreted from many cells, for example, hepatic parenchymal cells, muscle cells, adipocytes, monocytes and macrophages (Kockx et al. 2008). In the central nervous system, ApoE is synthesized mainly in astrocytes, partially in microglia, vascular wall cells and choroid plexus cells. ApoE is also expressed when neurons are stimulated or impaired (Xu et al. 2006; Polazzi et al. 2015; Huang et al. 2004). ApoE in the CSF is independent of peripheral pool, which in turn cannot cross the blood–brain barrier (BBB) (Linton et al. 1991).
Physiology Functions of ApoE
ApoE is a multifunctional protein, primarily as a ligand of cell surface receptors for the low-density lipoprotein (LDL) receptor family. ApoE is involved in lipid storage, transport and metabolism (Mahley 1988; Hauser et al. 2011); for example, clearance of cholesterol in the plasma. ApoE deficiency has been related to type III hyperlipoproteinemia, which causes early atherosclerosis and raises the level of cholesterol in plasma (Ghiselli et al. 1981). Indeed, ApoE mutations and abnormalities are associated with a wide range of illnesses, including atherosclerosis (Venegas-Pino et al. 2016), type 2 diabetes 2 (Chen et al. 2019), lipoprotein glomerulopathy (Yang et al. 2020), and heighten the infectious diseases susceptibility to infection, for example, AIDS, HIV and COVID-19 (Burt et al. 2008; Gkouskou et al. 2021). As a crucial apolipoprotein in the CNS, ApoE is essential for sustaining peripheral lipid metabolism, as well as lipid transport and metabolism in the brain. Cholesterol participates in maintaining neuronal plasticity, synapse growth and synaptogenesis (Mauch et al. 2001; de Chaves et al. 1997). In normal brain, ApoE is a component of lipoprotein particles, and they can transport cholesterol to neurons and increase the formation of synapse (Mauch et al. 2001). Overexpression of ApoE in astrocytes and macrophages can regulate the level of cholesterol or remove lipid debris (Poirier et al. 1991). Since the ApoE ɛ4 allele was reported as an AD risk gene, studies on the function of ApoE in the brain have focused on its genetic polymorphisms linked with AD pathology. Increasing evidence showed that ApoE2 is protective against AD. ApoE3 is risk-neutral in AD development and it is the most common isoform in the population (Lane-Donovan and Herz 2017). However, when compared to ApoE3 and ApoE2, ApoE4 is observed to increase the risk of AD development by impeding the clearance of Aβ (Castellano et al. 2011).
Although ApoE ɛ2 is considered as protective in the nervous system, individuals expressing homozygous ApoE2/2 were found glomerulopathy (Saito et al. 2020). Therefore, more studies and cases are needed to confirm the functions and polymorphism-based gene therapy of ApoE.
ApoE Receptors
LDL receptor family is main class of ApoE receptors, including LDLR, very-low density lipoprotein receptor (VLDLR), low-density lipoprotein receptor-related protein (LRP)1, LRP1B, LRP2, LRP4, LRP5, LRP6, and LRP8 (ApoER2) (Carlo 2013). LDLR family is a class of transmembrane receptors with many functions, for example, lipid metabolism and cardiovascular homeostasis (Calvier et al. 2022). In the central system, LDLR is primarily associated with cholesterol regulation through binding and internalization of cholesterol-containing LDL, which in turn affects cholesterol-related BBB functions and cognitive deficits (Hong et al. 2022). The binding affinity of LDLR family depends on isomers and lipidation status of ApoE (Bu 2009). LDLR preferentially binds to lipid ApoE particles, while VLDLR preferentially binds to lipid-free ApoE. Moreover, ApoE2 has a lower affinity for LDLR compared to ApoE3 and ApoE4 (Zhao et al. 2018). LRP1 is closely associated with AD. LRP1 not only regulates the endocytosis and spread of tau (Rauch et al. 2020) but also contributes to APP processing (Ulery et al. 2000). In addition, LRP1, VLDLR and ApoER2 identified in the postsynaptic density interacts with glutamate (Nakajima et al. 2013) and Reelin signaling (Strasser et al. 2004), in turn affecting synaptic function.
In neurons, there is a novel ApoE receptor called sortilin. It has a high binding affinity with ApoE. The expression of sortilin in the CNS is restricted to neurons and it might be a major neuronal ApoE receptor for catabolism of Aβ in the brain (Carlo et al. 2013). Sortilin can help neurons to take up essential lipids by binding ApoE and by facilitating the transfer of lipids from extracellular to intracellular transport particles. In the presence of ApoE3, Sortilin and ApoE3 cycle to the cell surface properly, allowing the lipid-rich brain fatty acid binding protein 7 (FABP7, a carrier of ω3-polyunsaturated fatty acids) to be released. However, sortilin recycling would be blocked in the presence of ApoE4, which impairs FABP7 release and affects intracellular lipid transport (Asaro et al. 2021).
The triggering receptor expressed on myeloid cells 2 (TREM2) is one of the major receptors of ApoE (Yeh et al. 2016). TREM2 is associated with inflammatory response of microglia, including proliferation, transport, and phagocytosis (Wang et al. 2020). The affinity of TREM2 with ApoE shows isoforms specific: ApoE4 > ApoE3 > ApoE2 (Mai et al. 2022). This higher affinity of TREM2 with ApoE4 might be a reason for ApoE4 risk in AD. The increased affinity of TREM2 for ApoE4 may contribute to the increased risk of ApoE4 in AD by amplifying TREM2 signaling and leading to microglia overactivation. Studies in vitro have shown that a lack of TREM2 would decrease Aβ uptake by ApoE4-treated microglia (Fitz et al. 2021). Furthermore, TREM2 variants, for example R47H, are associate with AD risk (Sayed et al. 2021). Besides, the signaling pathways triggered by the interaction between ApoE and TREM2 in AD pathogenesis are important.
In addition to the above receptors, ApoE4 can interact specifically with leukocyte immunoglobulin-like receptor B3 (LilrB3). ApoE4 can activate human microglia (HMC3) into a pro-inflammatory state, which is dependent on LilrB3 (Zhou et al. 2023). Generally, ApoE receptors play important role in the physiology function of ApoE.
Mechanism of ApoE4 in the AD Pathogenesis
Aβ Pathology
ApoE is one of the components of amyloid plaques in the brain observed by imaging. Early research studies on the roles of ApoE4 in AD mostly focused on the interaction of ApoE with Aβ. To date, Aβ pathology is the most well-defined part of ApoE in AD pathogenesis. Aβ is produced from the transmembrane protein amyloid precursor protein (APP) by the hydrolysis of β- and γ-secretase, mainly including Aβ1-40 and Aβ1-42 (2020 Alzheimer's disease facts and Figs. 2020). Aβ monomers polymerize into different types of assemblies, including oligomers, protofibrils and amyloid fibrils. Amyloid fibrils are the main components of SPs in AD pathology. Aβ oligomers are soluble, so they are easy to spread throughout the brain (Chen et al. 2017). In normal brain, ApoE associated with Aβ is involved in the metabolism and clearance of Aβ. However, fragments of ApoE4 combined with Aβ could aggravate the accumulation of Aβ. Mouchard et al. showed that ApoE fragments of 18 and 16 kD are the partners of Aβ1-42 in the brains of AD patients. These fragments of ApoE lack of C-terminus, and ApoE4 genotype can generate more ApoE fragment of 18 kD than those with ApoE ɛ2 or ɛ3 (Mouchard et al. 2019). Furthermore, lower plasma Aβ42/Aβ40 in ApoE4 carriers might predict higher cortical Aβ deposition, hippocampal atrophy and the decline of cognitive abilities (Shi et al. 2022). In normal brains, sotilin-related receptor (SORL1) can bind Aβ and target it to lysosomes for degradation. However, neural stem cells from patients carrying two ApoE4 allele showed reduced SORL1 expression and increased levels of Aβ. This may be one of the ways in which ApoE4 affects Aβ clearance (Zollo et al. 2017).
APP transport and Aβ production can be regulated by ApoE, which correlates with the isoform specificity of ApoE. From analysis of 18F-florbetapir-PET imaging, cognitively intact ApoE4 carriers become positive on amyloid imaging at around 56 years of age, and ApoE4 non-carriers become positive at around 76 years of age (Fleisher et al. 2013). These observations suggested that ApoE4 might increase AD risk by initiating and accelerating the accumulation, aggregation, and deposition of Aβ in the brain. However, compared with ApoE3 and ApoE4 carriers, individuals with ApoE2 tend to slow cognitive decline and less pronounced Aβ pathology and NFT (Serrano-Pozo et al. 2015). ApoE4 can increase Aβ production by accelerating APP endocytosis transport mediated by low-density lipoprotein receptor-related protein (LRP) (Pietrzik et al. 2002; Tachibana et al. 2019). LRP1 plays an important role in Aβ clearance. BBB-associated pericytes clear Aβ aggregates through LRP1/ApoE interactions, affecting by ApoE4 (Ma et al. 2018). LRP6 is an essential receptor of ApoE for Wnt signaling. LRP6 can reduce endocytic transport of APP and Aβ production. Knocking down LRP6 in neurons could increase amyloid deposition and neuroinflammation (Liu et al. 2014a). Furthermore, ApoE can act as a signaling molecule of Aβ depending on its subtype-specific. Compared with ApoE2 and ApoE3, ApoE4 binding to ApoE receptors easily leads to combination between dual leucine-zipper kinase (DLK) and ApoE receptors, activates ERK1/2 MAP kinase and induces cFos phosphorylation, which stimulates transcription factor AP-1, thereby increasing APP transcription and Aβ production (Huang et al. 2017), as shown in Fig. 2.
Roles of ApoE4 in Aβ pathology. ApoE4 binds to Aβ and form a complex that affects Aβ clearance and accelerates its accumulation and deposition in the brain. ApoE4 makes the ApoE receptor more susceptible to binding to dual leucine-zipper kinase (DLK), activates ERK1/2 MAP kinase and induces cFos phosphorylation, stimulates transcription factor AP-1, resulting in enhanced APP transcription, increased APP expression, and consequently increased Aβ
ApoE4 contributes to Aβ oligomers in the brain. ApoE ε4/ε4 carriers have higher levels of Aβ oligomers compared to ApoE ε3/ε3 carriers. The individuals expressing ApoE4 may increase dendritic spine loss and accelerate memory impairment, leading to early cognitive decline in AD (Hashimoto et al. 2012). Kara et al. (2018) found, compared with ApoE2 and ApoE3, there is a significant interaction between ApoE4 and Aβ in primary immortalized astrocytes measured by flow cytometry. This interaction makes more likely to form protein deposition complexes. Although ApoE4 affects the production, aggregation and transport of Aβ, whether APP processing correlates with ApoE subtype specificity remains unclear. In fact, a study using ApoE targeted-replacement mice suggested that ApoE isoform specificity didn’t affect APP expression, full protein levels, or its processing in the brains (Novy et al. 2022). However, the different expression and processing of APP in brain regions were not detected. Therefore, the relationship between ApoE4 and APP is needed to further investigate.
Tau Phosphorylation
Hyperphosphorylated tau is a major constituent of NFTs. Studies have shown that AD patients expressing ApoE4 may possess relatively more NFTs in their medial temporal lobe, most notably in the entorhinal cortex (Emrani et al. 2020). The pathophysiology of NFTs in AD patients with the ε4 allele in the cortex is worsened compared to those who do not have the ε4 allele. Overexpression of ApoE4 in astrocytes can increase the phosphorylation and aggregation of tau within neurons (Jablonski et al. 2021). A study about chimeric human cerebral organoids (chCOs) also confirmed that ApoE4 astrocytes may accelerate the phosphorylation of tau in neurons, and neuron-astrocyte transport of p-tau may be impaired by neuronal ApoE4 (Huang et al. 2022). Furthermore, the extent of NFTs is greater in ε4 homozygous compared to heterozygous carriers (Sabbagh et al. 2013). Salami et al. observed a longitudinal increase of plasma p-tau level in ɛ4 carriers, which is related to the elevated local hippocampal connectivity at resting-state and hippocampus connectivity (Salami et al. 2022).
The correlation of ApoE and tau is likely independent of Aβ pathology. Farfel et al. (2016) identified by autopsy staining of volunteers in several regions in the internal olfactory cortex, hippocampus at CA1, entorhinal cortex, and inferior parietal cortex and found that ApoE ε4 and ε2 were associated with NFTs independent of Aβ. ApoE4 may have an influence on tau directly without going through Aβ. Shi et al. (2017) showed that tau-mediated neurodegeneration was exacerbated in P301S tau transgenic mice transfected with the human ApoE4 gene, but it was not associated with Aβ. The levels of tau and p-tau in CSF levels are positively associated with the number of ApoE4 allele (Benson et al. 2022). Kang et al. revealed that ApoE4 inhibits the vesicular monoamine transporter2 (VMAT2) in the locus coeruleus and facilitate tau pathology (Kang et al. 2021). However, there is insufficient evidence that ApoE4 can act directly on tau, as few AD patients with ApoE4 have no Aβ deposition in the brain.
NFT pathology in ApoE4-positive AD patients may be linked to tau phosphorylation. Neuron-specific proteolytic cleavage of ApoE4 is associated with increased phosphorylation of tau. ApoE fragments might play vital role in the development of neuronal deficits. ApoE fragments, extracted from brain tissue homogenates of transgenic mouse with ApoE4 were transfected into neurons or astrocytes, study showed that ApoE hydrolysis fragments increased phosphorylation of tau (Brecht et al. 2004). In addition, astrocyte-secreted protein glypican-4 (GPC-4), as a partner of ApoE4, could contribute to tau hyperphosphorylation (Saroja et al. 2022). LPR1 may be associated with tau propagation. Hyperphosphorylated tau binds inefficiently to LRP1, making tau internalization inefficient. ApoE4 would inhibit LRP1-mediated tau uptake, possibly because ApoE4 has a higher affinity for LRP1 (Cooper et al. 2021). Studies on ApoE4 and tau pathology are still in the preliminary stages, and evidence on whether ApoE4 bypasses Aβ and directly affects tau needs more investigations (Fig. 3).
Synaptic Impairment
In the normal brain, ApoE transports cholesterol-rich lipoproteins to neurons, promoting synaptogenesis and maintaining synaptic connections. However, ApoE ε4 carriers may contribute to AD risk through synaptic damage. Neurogranin (Ng) is a postsynaptic protein that is highly expressed in the hippocampus. Ng is usually involved in memory consolidation. Compared with ApoE4 non-carriers with mild cognitive impairment, the levels of Ng in the CSF expressed ApoE4 were significantly higher (Sun et al. 2016). This data showed that ApoE4 contributes to early cognitive impairment and synaptic damage in AD patients. ApoE4 retains ApoER2 and glutamate receptors in endosomal compartments. Synaptic transmission mediated by Reelin is inhibited and ApoE on the surface of neurons is decreased (Chen et al. 2010). Sun et.al. (Sun et al. 2017) established model by transferring human-derived ApoE4 and ApoE3. They found, compared with mice expressing ApoE3, mice with ApoE4 showed shortened dendrite length, reduced dendritic spine density, and enhanced facilitation of basic synaptic transmission and paired pulses in hippocampal CA1 region neurons. This result suggested that the effects of ApoE4 on synaptic function possibly prior to AD occurrence.
ApoE4 can contribute to neuronal and synaptic dysfunction by activation of Cyclophilin-A matrix metalloproteinase-9 (CypA-MMP9) pathway (Anderson et al. 2022). In addition, Aβ burden has toxic effects on synapses. Aβ/ApoE4 complexes would lead to the accumulation of Aβ in the intercellular space of neurons rather than its clearance (Bilousova et al. 2019). In contrast, Aβ/ApoE2 complexes may contribute to Aβ clearance and protect synaptic function of ApoE transgenic (TR) mouse (Arold et al. 2012). Synaptic dysfunction is a central mechanism of cognitive impairment in AD. It is helpful for diagnosis to recognize the effect of ApoE ε4 on synaptic function in the early stage of AD development (Fig. 4).
Roles of ApoE4 in synaptic impairment. ApoE4 retains ApoER2 and glutamate receptors in endosomal compartments. Synaptic transmission mediated by reelin is inhibited and ApoE on the surface of neurons is decreased. ApoE4 can contribute to neuronal and synaptic dysfunction by activation of cyclophilin-A matrix metalloproteinase-9 (CypA-MMP9) pathway. ApoE4 would lead to the accumulation of Aβ, inducing synaptic impairment
Cholesterol Transportation
The brain has the highest content of cholesterol, accounting for approximately 20% of whole cholesterol in the body. Approximately 70% of cholesterol in the brain is found in myelin. The rest about 30% of cholesterol is found in the membranes of glial cells and neurons, where it is primarily circulated for neuronal repair and remodeling (Dietschy and Turley 2001, 2004). ApoE initiates the formation of high-density lipoprotein (HDL)-like particles by accepting cholesterol and phospholipids via ABCA1 and ABCG1, activating members of the ATP-binding cassette (ABC) family of transport proteins (Horiuchi et al. 2019). Lipoproteins and lipid complexes combined with ApoE interact with cell surface heparan sulfate proteoglycans and cell membrane-associated receptors to promote cellular uptake and redistribution and storage of cholesterol (Lanfranco et al. 2020).
Generally, AD brain shows abnormal cholesterol. In cultured neurons, transient increases of cholesterol in membrane are associated with early AD pathological features, for example, excessive Aβ1-42 production, enlarged endosomes, and abnormal axonal transport (Marquer et al. 2014). ApoE4 has a low affinity and binding capacity for lipid transport (Hatters et al. 2006). Inadequate ApoE levels or impaired ApoE function in ApoE ε4 carriers may lead to imbalance of cholesterol homeostasis in the CNS and affect neuronal health. A study showed that the level of total cholesterol was higher in ApoE4 carriers compared to those ApoE3 and ApoE2 (Dunk and Driscoll 2022). Generally, higher total cholesterol may be a risk factor for AD pathogenesis. Previous study connected cholesterol with APP processing mediated by ApoE (Howland et al. 1998). Cholesterol clearance depends on its catabolic derivative, 24S-hydroxycholesterol (24-OHC). Elevated 27-OHC can activate C/EBPβ at the presence ApoE4, which subsequently increases Aβ production and tau hyperphosphorylation in ApoE4 TR mice (Wang et al. 2021). ABCA1 is involved in cholesterol transport. ApoE4 increases ABCA1 aggregation by promoting ADP-ribosylation factor 6 (ARF6) expression and reduces ABCA1 cell membrane recycling in the primary astrocytes obtained from ApoE4 TR mice (Rawat et al. 2019). A study showed that 27-OHC might affect cholesterol transport through ABCA1-ApoE-LDLR/LRP1 proteins in the brains of ApoE4 TR mice (Wang et al. 2022b). Lee et al. (2021) found that astrocytes expressing ApoE4 could lead to excessive cholesterol accumulation, which in turn increases the level of APP in lipid rafts, as detailed in Fig. 5. Additionally, cholesterol abnormalities induced by ApoE4 might further abnormalize glucose metabolism (Wu et al. 2018), aggregating AD process. Therefore, ApoE4 plays profound roles in cholesterol metabolism, transport, and AD development.
Regulation of cholesterol in brain mediated by ApoE4. ApoE4 decreases ABCA1 cycling by promoting increased expression of ADP-ribosylation factor 6 (ARF6), which in turn decreases ABCA1 cycling to the cell membrane in astrocytes and affects cholesterol distribution. astrocytes with ApoE4 accumulate excess cholesterol and increase the level of APP in lipid rafts. Cholesterol clearance depends on its catabolic derivative, 24S-hydroxycholesterol (24-OHC). Elevated 27-OHC can activate C/EBPβ at the presence ApoE4, which subsequently increases Aβ production
Oxidative Stress
When the redox balance in the brain is disrupted, oxidative stress occurs, leading to neuronal death. AD brain in a state of high oxidative stress. Nevertheless, oxidative stress is a common side effect of aging. ApoE has antioxidative properties demonstrated by ApoE KO mice. ApoE deficient mice may have poor learning and memory ability as a result of higher levels of diet-induced oxidation (Evola et al. 2010). Increasing evidence connected the AD induced by ApoE genotype with the oxidative stress. The degree of oxidative stress in AD brain is closely related to the genotype of ApoE: ApoE2 < ApoE3 < ApoE4 (Miyata and Smith 1996). The level of reactive oxygen species (ROS) and the oxidation of protein and lipid are always increased in synaptosomes isolated from ApoE4 mice (Lauderback et al. 2002). Compared with AD patients without ApoE4, AD patients carrying ApoE4 showed higher hydroxyl radical levels in the blood (Ihara et al. 2000) and lower cerebral oxygen utilization (Robb et al. 2022). Individuals with ApoE4 is associated with increased levels of oxidative stress, which may contribute to the earlier onset of AD. Caberlotto et.al found ApoE4 is involved in oxidative stress possibly through the Notch signaling pathway suggested by transcriptomic data (Caberlotto et al. 2016).
In fact, it is still unclear how ApoE4 aggravates oxidative stress in the brain. One possible explanation related to the ApoE structure. This different amino acid composition of the three ApoE phenotype affects the binding with lipid peroxides; and the levels of lipid peroxides usually are used as markers of oxidative stress. A lipid peroxide, named 4-hydroxynonenal (HNE), can bind to Cys residue on ApoE2 and ApoE3 to reduce the damage. However, ApoE4 is lack of Cys residue that can clear the toxic of lipid peroxide (Butterfield and Mattson 2020). Another, ApoE4 aggravates oxidative stress and damages cerebral cortical neuron by triggering Ca2+ overload and CaMK II phosphorylation abnormity (Xu and Peng 2017). Although oxidative stress is thought to be a common pathomechanism involved in AD pathogenesis, it is often associated with aging and inflammation. In other word, oxidative stress is an intermediary process.
Mitochondrial Dysfunction
Mitochondrial dysfunction usually drives the early stage of AD pathogenesis. ApoE isoforms affect mitochondria-related proteins, and consequently altered mitochondrial function aggravates the AD pathological process (Fig. 6). ApoE4 could aggravate the progression of AD by impairing the respiration and increase glycolysis. When the neurons are stressed, the expression of ApoE4 will reduce the generation of ATP (Orr et al. 2019). In ApoE4 carriers, activated receptor-gamma coactivator 1α (PGC-1α), deacetylase (Sirtuin 3, SIRT3), Mitofusin 1 (MFN1), MFN2 and dynamin-like protein 1 (DLP1) levels were lower than those in noncarriers (Yin et al. 2020). It suggested that ApoE4 affects physiological functions, for example, mitochondrial biogenesis and dynamics. ApoE4 may be involved in mitochondrial dysfunction by damaging autophagy and mitophagy via the repression of FoxO3a in the brains of ApoE4 carriers (Sohn et al. 2021). ApoE4 could be hydrolyzed by neuron-specific proteins to produce a neurotoxic fragment (12–29 kDa). The link of the lipid and receptor-binding regions in the ApoE4 fragments leads to mitochondrial dysfunction and neurotoxicity of the Neuro-2a (N2a) cells transfected with ApoE4 (Chang et al. 2005). In N2a cells expressing ApoE4, the levels of mitochondrial respiratory complexes I, IV, and V were all deregulated. However, compared to N2a cells expressed ApoE4, the gene transcripts of all respiratory complexes in ApoE3 N2a cells were all downregulated (Chen et al. 2011). A recent study found that postmortem tissues from ApoE4 carriers showed a deficiency of cytochrome oxidase (COX), complex II and III (Troutwine et al. 2022). In addition, ApoE4 was revealed to bind to α/β subunits of F1 mitochondrial ATP synthase in liver (Mahley et al. 1989). The results from proteomic analysis indicated that there was a remarkable reduction in the levels of 50% of detected F1/F0 subunits of mitochondrial respiratory complexes V (ATP synthase) (Orr et al. 2019). However, more investigates are needed to clarify the mechanism of ApoE4-driven dysfunction of respiratory complexes.
Influence of ApoE4 in mitochondrial dysfunction. When the neurons are stressed, ApoE4 expression in neuron cells would inhibit the generation of ATP, increase the reactive oxygen species (ROS) level and promotes calcium overload. Some proteins about mitochondrial biogenesis and dynamics could be reduced when ApoE4 presents. ApoE4 fragments also lead to mitochondrial dysfunction and endoplasmic reticulum (ER) stress, as well as the formation of mitochondrial-associated membrane (MAM)
Increasing studies focus on the mitochondrial dysfunction and endoplasmic reticulum (ER) related to high level of ROS. Astrocyte‐conditioned media (ACM) containing ApoE4 showed increases of mitochondrial-associated membrane (MAM) function compared to ApoE3 (Tambini et al. 2016). In N2a cells and female ApoE4 (Δ272–299) TR mice, ApoE4 (Δ272–299) significantly induces mitochondrial dysfunction by triggering ER stress and increasing the expression of GRP75, which promotes the formation of mitochondrial-associated membrane (MAM) and mitochondrial calcium overload (Liang et al. 2021). ApoE4 (Δ1-272) could induce the activities of mitochondrial respiratory complex III and IV, which triggered mitochondrial dysfunction (Nakamura et al. 2009). In fact, MAM activity mediated by ApoE4 on AD development is still unclear, but it can be determined that ApoE4 may contribute to the development of AD by affecting mitochondria-related functions.
Neuroinflammation
ApoE4 may be involved in neuroinflammation. The inflammatory factors in the brain are mainly produced from microglia and astrocytes. Similarly, ApoE protein is normally synthesized by microglia and astrocytes. Lynch et al. (2003) used human-derived ApoE4 and ApoE3 transgenic mice and administered lipopolysaccharide intravenously. They found a significant increase in TNF-α and IL-6 present in the brains of ApoE4 mice compared to ApoE3 mice. These results also found in the cells that ApoE4 increases the expression of inflammatory factors in human astrocytes (Iannucci et al. 2021). Moreover, ApoE4 genotype also alters immunometabolism of microglia. A study indicated that aerobic glycolysis and level of hypoxia inducible factor 1α (Hif1α) are increased in microglia expressing ApoE4. Furthermore, ApoE4 could aggravate plaque-induced microglial reactivity and lipid metabolism (Lee et al. 2023).
Apparently, ApoE4 exacerbates neuroinflammation. ApoE is a ligand for triggering receptor expressed on myeloid cells 2 (TREM2). TREM2, expressed on microglia, is an immune receptor. TREM2 is important in microglia-related functions. A study showed that microglia could be activated by TREM2 through spleen tyrosine kinase (SYK) or DAP10-dependent pathways. SYK could guild signaling and effector functions downstream of TREM2. Phagocytosis of Aβ by microglia is also associated with SYK. Moreover, Akt-GSK-3β signaling could be affected by the combination between DAP10 and TREM2. Deficiency in SYK can lead to the development of disease-associated microglia and the onset of the prodromal stage of ApoE expression, which occurs via DAP10-dependent pathways (Ennerfelt et al. 2022; Wang et al. 2022a).
TREM2-mediated signaling pathways for neuroprotection can be affected by ApoE (Ulrich and Holtzman 2016). In fact, TREM2 is also a major genetic risk factor for AD development. ApoE-TREM2 interaction is related to AD. The interaction between ApoE and TREM2 is affected by ApoE polymorphisms, and ApoE4 shows the highest affinity with TREM2 compared to ApoE2 and ApoE3 (Kober et al. 2020). Krasemann et al. reported that ApoE-TREM2 pathway could be activated when neuronal apoptosis happened, inducing microglia homeostatic imbalance (Krasemann et al. 2017). However, this study was conducted in mice and could not account for genetic subtype differences of ApoE in relation to TREM2. In the microglia expressing ApoE4, lacking of TREM2 would reduce Aβ uptake by microglia (Fitz et al. 2021) (Fig. 7). A study showed that ApoE4 induces neuroinflammation through activating the proinflammatory PGE2 pathway or inhibiting the TREM2 pathway (Li et al. 2015). Furthermore, the molecular mechanism of interaction between ApoE4 and TREM2 deserves to be further investigated.
Neuroinflammation effects of ApoE4 in AD pathogenesis. ApoE4 induces the increase of inflammatory factors, including TNF-α and IL-6. ApoE4 could exacerbate neuroinflammation through activating the proinflammatory PGE2 pathway or inhibiting the anti-inflammatory TREM2 pathway. ApoE4 also can contribute to neuroinflammation through inducing Ca2+ dependent phospholipase A2 (cPLA2) activations and changes on arachidonic acid (AA) signaling cascades. Further, ApoE4 affects Aβ clearance mediated by the interaction between ApoE4 and TREM2
In addition, ApoE4 can induce Ca2+ dependent phospholipase A2 (cPLA2) activations and contributes to the changes on arachidonic acid (AA) signaling cascades. AA changes are generally related to the chronic brain inflammation (Duro et al. 2022).
Sleep Disturbances
ApoE4 may also be associated with sleep disturbances in AD patients. The older with cognitive impairment and expressing ApoE4 generally showed long sleep duration (Basta et al. 2021). Carriers containing two ɛ4 alleles showed more sleep abnormalities compared to ɛ4 noncarriers or AD patients with one ɛ4 allele (Koo et al. 2019). A study of 698 community dwelling older adults without dementia showed that improving sleep quality reduced the negative effects of ApoE4 on NFTs (Lim et al. 2013). Furthermore, ApoE4 effects on sleep might be independent of Aβ and tau stages and ApoE4 might regulate the level of melatonin. AD patients with two ɛ4 alleles was associated with significantly reduced post-mortem CSF melatonin compared to AD patients with one ɛ4 allele (Blackman et al. 2022). Furthermore, sleep disorders could activate microglial and even contribute to neuroinflammation (Hu et al. 2021).
Cerebrovascular Integrity
The contribution of cerebrovascular integrity to AD is increasingly recognized. ApoE4 can exacerbate the deposition of Aβ in the brain to form cerebral amyloid angiopathy (CAA), which can severely damage vascular integrity and BBB functions. Although CAA is not clinically identical to AD pathology, it has a similar molecular basis with AD. (Safieh et al. 2019). Reducing ApoE4 in astrocytes from 5XFAD ApoE4 knock-in mice could decrease overall Aβ-mediated gliosis and increase cerebrovascular integrity in CAA-containing vessels (Xiong et al. 2023). Cerebrovascular integrity and function are also regulated by fibrinogen, which is also dependent on the subtype specificity of ApoE. AD patients with ApoE ɛ4/ɛ4 genotype exhibit increased fibrinogen deposition in CAA and oligomeric Aβ-positive vessels (Hultman et al. 2013). Data from recent years suggested that ApoE4 expression could exert detrimental effects on the cerebrovascular system, including BBB impairments. BBB integrity is critical in the pathology of neurodegeneration and cognitive impairment. Compared with ApoE3, ApoE4 could accelerate BBB catabolism, neuronal loss and behavioral deficits, which can be independent of Aβ (Montagne et al. 2021). ApoE4 activates the CypA-MMP9 pathway in the cerebrospinal fluid and thereby accelerates BBB catabolism, leading to neuronal and synaptic dysfunction (Montagne et al. 2020).
Others
ApoE4 plays an important role in many aspects of AD pathogenesis. In addition to the mechanisms described above, intracerebral glucose metabolism, transactive response DNA binding protein (TDP-43) pathology, and the relationship with other non-AD diseases still require more investigations. In addition, astrocytes expressing ApoE4 showed high glycolytic activity, low oxygen consumption, and reduced rate of glucose oxidation in the presence of lactate (Farmer et al. 2021). Diabetes is a risk factor for AD (Shinohara et al. 2020). Atherosclerosis is related to LDLR and ApoE (Zhao et al. 2022). Therefore, studies related to these two diseases may provide the investigation of the mechanisms and treatment for AD. Furthermore, the AD risk for ApoE4 carriers might differ between sexes. The activity of BACE1 is associated with ApoE4, which female showed higher BACE1 expression than male, using the mice with ApoE4 or AD risk factors (APPSwe, PS1M146V, tauP301L; 3xTg) (Hou et al. 2015). Astrocyte coverage of plaques was the poorest in ApoE4 females (Stephen et al. 2022). Compared with primary microglia in ApoE4 males, the levels of IL1b, TNFa, IL6, and NOS2 were higher in ApoE4 female primary microglia (Mhatre-Winters et al. 2022). The interaction between sex and ApoE4 in AD pathogenesis remains unclear. In addition, AD brains have another pathological deposition, that is TDP-43 pathology. TDP-43 is a 43 kDa heterogeneous nuclear ribonuclear protein, which regulates gene expression and RNA processing (Stover et al. 2004; Higashi et al. 2013). Some evidence showed an association between ApoE4 and increased TDP-43 pathology. Two community-based cohort studies of ageing and dementia indicated that ApoE4 seemed to interact with increased TDP-43 burden, and high levels of TDP-43 might induce hippocampal atrophy (Yang et al. 2018). A study on 738 older adults with AD also showed that TDP-43 are associated with ApoE4 directly (mediated by Aβ and tau) or indirectly (Wennberg et al. 2018). Furthermore, brains with limbic-predominant TDP-43 pathology were more likely to carry ε4 allele and had higher possibility of AD (Teylan et al. 2021). In addition, TREM2 deficiency could damage phagocytic clearance of pathological TDP-43 by microglia, inducing neuronal damage (Xie et al. 2022).
Therapeutic Approaches Targeting to ApoE
To date, AD remains far from cure, and there are a few classes of drugs approved for the AD treatment; for example, cholinesterase inhibitors (Tacrine, Donepezil, Rivastigmine, Galanthaminone) NMDA receptor antagonists (Memantine), and Aβ monoclonal antibody (Aducanumab). The genotype of ApoE determines the degree of LOAD risk, and ApoE plays a crucial role in the brain. Therefore, targeting ApoE4 is a highly promising therapy for AD. Considering the roles of ApoE4 in the LOAD pathogenesis, the current research studies on ApoE4-targeted AD therapy can be divided into the following categories: targeting the interaction of ApoE with Aβ, targeting ApoE receptors, correcting the ApoE4 genotype or its function by gene editing, ApoE antibody, and nonpharmacological therapy (Fig. 8). The research on ApoE-targeted therapy can be helpful in the early intervention and treatment of LOAD. Furthermore, there are several drugs targeting ApoE to improve AD pathology in trials are shown in Tab. 1.
AD therapy targeting to ApoE. Targeting ApoE4 is highly promising for AD treatment and improvement. Considering the roles of ApoE4 in the LOAD pathogenesis, the therapeutic approaches can be subdivided into the following categories: targeting the interaction of ApoE with Aβ, targeting ApoE receptors, correcting the ApoE4 genotype by gene editing (CRISPR/Cas9), ApoE antibody, and nonpharmacological therapy
Targeting the Interaction of ApoE with Aβ
In ApoE4 carriers, ApoE-Aβ interaction contributes to Aβ more susceptible to deposition into amyloid plaques. Blocking the interaction between ApoE and Aβ could reduce intraneuronal accumulation of Aβ and inhibit synaptic degeneration. Aβ12-28P is mimics of Aβ. Aβ12-28P can reduce neurotoxicity induced by Aβ through blocking the binding between ApoE and Aβ at residues 12–28. Aβ12-28P can also attenuate Aβ deposition and insoluble tau accumulation and inhibit synaptic degeneration in AD mice model (Sadowski et al. 2004; Kuszczyk et al. 2013; Liu et al. 2014b). The enoxaparin molecule has a strong energetic affinity for ApoE4 and disrupt the interaction between ApoE4 and Aβ (Aguilar-Pineda et al. 2022). Therefore, amyloid load can be reduced by regulating ApoE levels in the brain.
Targeting ApoE Receptors
ApoE participates in different physiological process mediated by its receptors. ApoE4 will reduce the expression of LRP8 (ApoER2) (Chen et al. 2010); this receptor can prevent the loss of corticospinal neurons with aging (Beffert et al. 2006). Overexpression of ApoER2 may reduce the risk of ApoE4. LRP1 also plays an important role in mediating the involvement of ApoE4 in AD pathogenesis, mainly associated with Aβ signaling (Pietrzik et al. 2002). Regulating the expression of ApoE receptors can restore lipid homeostasis and synaptic plasticity and increase Aβ clearance. In addition, overexpression of LDLR in microglia can inhibit ApoE expression and reduce tau-associated neurodegeneration mediated by ApoE (Shi et al. 2021).
Gene Editing for ApoE4 Gene
The function of ApoE is dependent of its heterodimer. Therefore, it might be effective for suppressing the risk of ApoE4 to modify ApoE from ε4 to ε3 or ε2 by gene editing. When iPSC-derived neurons were converted from ApoE ε4 to ε3 using zinc finger nucleases, there were reductions in ApoE fragments, Aβ production, and tau phosphorylation. This research study suggested gene editing can eliminate the deleterious effects of ApoE4 (Wang et al. 2018). CRISPR/Cas9, the third-generation gene editing technology, has shown great potential in modifying ApoE genotypes. The technology has proven successful at the cellular level, where cells from healthy ApoE3/E4 individuals can be transformed into ApoE2/E2, ApoE3/E3, ApoE4/E4 or ApoE KO (Schmid et al. 2019). In recent studies, CRISPR/Cas9 gene editing technology is mostly used to establish models with ApoE4 to study the effects of this gene on synaptic function and lipid metabolism (Lin et al. 2018). However, whether CRISPR/Cas9 gene editing technology can be used for gene therapy of ApoE4 remains to be investigated. Notably, there are ethical issues and risks associated with gene editing therapies, so whether they can be administered to AD patients remains highly controversial.
ApoE Antibody
An anti-human ApoE antibody (HAE-4) which recognizes human ApoE4 and ApoE3. It preferentially binds to nonlipidated forms of ApoE and ApoE in plaques. HAE-4 has been reported to suppress Aβ plaques (Liao et al. 2018) and tau spreading driven by Aβ. This clearance abilities of HAE-4 might depend on microglial activation. HAE-4 could increase microglial activation (Gratuze et al. 2022). Furthermore, HAE-4 not only reduces Aβ deposition including CAA but also inhibits reactive microglia, astrocytes and pro-inflammatory related genes in the cortex (Xiong et al. 2021a). HJ6.3, a monoclonal antibody against ApoE, could block the interaction of ApoE with Aβ. Liao et al. treated 7-month-old APP/PS1 mice with HJ6.3 for 21 weeks. They found HJ6.3 could reduce the formation and aggregation of Aβ and improve the spatial learning abilities of mice (Liao et al. 2014). Although ApoE antibodies are effective, more investigations are needed to determine whether these antibodies have side effects.
Nonpharmacological Therapy
ApoE4 carriers have a high probability of AD development, but early intervention can still delay the onset of AD or even prevent it from occurring. The individuals carrying ApoE4 can prevent AD through diet, such as using a low glycemic index and low carbohydrate diet structure. This diet could be helpful in preventing the glycation of ApoE and brain lipid metabolism for AD patients. Nevertheless, this diet could prevent the effects of ApoE4 on the insulin cascade (Norwitz et al. 2021). Ketogenic diet is an option of low glycemic index and low carbohydrate diet structure for patients. A ketogenic diet for ApoE4 carriers can make their metabolism dependent on ketones rather than glucose (Wu et al. 2018), thereby reducing the metabolic burden and improving cognition of ApoE4 carriers with AD (Morrill and Gibas 2019). Long-term omega-3 supplementation improved the cognition abilities and lowered Aβ burden in ApoE4 carriers (Li et al. 2022). DHA-containing fish oil supplementation improved novel object recognition memory, increased BDNF protein, and improved abnormal Erβ, Cldn1 and Glut-5 expression (Pontifex et al. 2022) in ApoE4 mice treated with VCD (4-vinylcyclohexene diepoxide). Additionally, a poor lifestyle in ApoE4 carriers, such as sedentary lifestyle, might increase the risk of amyloid deposition; active physical activity might prevent the AD risk associated with ApoE4 (Liu et al. 2014b). Notably, improvement on sleep disturbance might reduce the risk of probable AD patients for individuals carrying ApoE4 allele.
Summary and Prospect
AD is a neurodegenerative disease influenced by genetic and the environmental factors, but there is still no cure for AD patients. Given the discovery of ApoE4 as an AD risk gene, it is expected that more insights can be gained into the pathogenesis and treatment of AD by investigating the mechanism followed by the role of this allele in AD pathogenesis. ApoE plays an important role in the AD brain. Although ApoE4 is not inevitable in the induction of AD, it remains possible for this genetic isoform to accelerate AD progression. Mechanistically, ApoE4 could increase the risk of cognitive decline by initiating and accelerating the accumulation, aggregation, and deposition of Aβ in the brain, as well as increasing the level of Aβ oligomers in the brain. Additionally, ApoE4 could affect tau phosphorylation and the density of NFTs in the brain directly or indirectly. Compared with ApoE3 and ApoE2, ApoE4 shows a lower level of efficiency not only in providing cholesterol but also in maintaining synaptic integrity and plasticity. These reviews are similar to the previous reviews, for example Koutsodendris et al. (2022) and Serrano-Pozo et al. (2021). In addition, we further reviewed that ApoE4 exacerbates the level of oxidative stress in the AD brain. Definitely, it is also suspected that ApoE4 may also be associated with mitochondrial dysfunction, neuroinflammation, and sleep disturbances in AD pathogenesis. Notably, the fragments generated by hydrolysis of ApoE4 are linked to several pathological features of AD, such as tau. Therefore, understanding the involvement of ApoE ε4 allele in the AD pathogenesis is not only conducive to preventing or delaying the onset of AD, but also contributory to developing the therapeutic strategies for AD targeting ApoE4. The current studies targeting to ApoE4 focus on ApoE-Aβ interactions, ApoE receptors, correcting the ApoE4 genotype or its function through gene editing. ApoE antibody might be a possible treatment for AD. Furthermore, nonpharmacological therapy could be effective in prevention. However, ApoE4-targeted therapy can only slow down the progression of AD and reduce the probability of AD occurrence in populations with ApoE4. Therefore, ApoE4-related therapies are recommended for early treatment of AD in individuals who carry the ApoE4 gene or for preventing AD pathology before it occurs. Furthermore, although recent studies have reviewed about ApoE degradation and structural correctors (Safieh et al. 2019), our review do not cover these topics. Because we believe that preserving the physiological function of ApoE is crucial and that relevant therapies should not be utilized in a manner that compromises its original function. The functions performed by ApoE4 vary in different cells. In addition to microglia and astrocytes, it is also worthwhile to study the expression of ApoE4 in vascular wall cells. The expression of ApoE4 in blood vessels not only reduces the blood flow in small arteries but also impairs the ability of spatial learning (Yamazaki et al. 2021). Therefore, to reveal the different roles of ApoE4 in different cells is beneficial to understand AD pathogenesis. Furthermore, ApoE4 is related to COVID-19 (Xiong et al. 2021b), dementia with lewy bodies (Zhao et al. 2020), and diabetes mellitus type, and all of which have a close link to AD. It is suggested that the connection between ApoE4 and other pathological proteins characteristic of these disease can be found in the pathogenesis of AD. AD pathogenesis can be understood from different perspectives. Notably, ApoE4 can exacerbate or induce inflammation, which may be a commonality between these diseases. In conclusion, most studies are still focused on the connection between ApoE4 and Aβ, and we believe that this study about ApoE4-Aβ interval is a promising target for AD treatment. In addition, diet improvement might be a better treatment for ApoE4 carrier without AD. Furthermore, there are some new perspectives, for example, tau propagation, vascular dysfunction. Lipid homeostasis related to the function of ApoE is also well worth an in-depth discussion.
Data Availability
Enquiries about data availability should be directed to the authors
References
Aguilar-Pineda JA, Paco-Coralla SG, Febres-Molina C, Gamero-Begazo PL, Shrivastava P, Vera-López KJ, Davila-Del-Carpio G, López CP, Gómez B, Lino Cardenas CL (2022) In silico analysis of the antagonist effect of enoxaparin on the ApoE4-amyloid-beta (Aβ) complex at different pH conditions. Biomolecules. https://doi.org/10.3390/biom12040499
Anderson EL, Williams DM, Walker VM, Davies NM (2022) Little genomic support for Cyclophilin A-matrix metalloproteinase-9 pathway as a therapeutic target for cognitive impairment in APOE4 carriers. Sci Rep 12 (1):1057. https://doi.org/10.1038/s41598-022-05225-8
Arold S, Sullivan P, Bilousova T, Teng E, Miller CA, Poon WW, Vinters HV, Cornwell LB, Saing T, Cole GM, Gylys KH (2012) Apolipoprotein E level and cholesterol are associated with reduced synaptic amyloid beta in Alzheimer’s disease and apoE TR mouse cortex. Acta Neuropathol 123 (1):39–52. https://doi.org/10.1007/s00401-011-0892-1
As Association (2020) 2020 Alzheimer's disease facts and figures. Alzheimer's Dementia. https://doi.org/10.1002/alz.12068
Asaro A, Sinha R, Bakun M, Kalnytska O, Carlo-Spiewok AS, Rubel T, Rozeboom A, Dadlez M, Kaminska B, Aronica E, Malik AR, Willnow TE (2021) ApoE4 disrupts interaction of sortilin with fatty acid-binding protein 7 essential to promote lipid signaling. J Cell Sci. https://doi.org/10.1242/jcs.258894
Basta M, Zaganas I, Simos P, Koutentaki E, Dimovasili C, Mathioudakis L, Bourbouli M, Panagiotakis S, Kapetanaki S, Vgontzas A (2021) Apolipoprotein E ɛ4 (APOE ɛ4) allele is associated with long sleep duration among elderly with cognitive impairment. J Alzheimer’s Disease 79 (2):763–771. https://doi.org/10.3233/jad-200958
Beffert U, Nematollah Farsian F, Masiulis I, Hammer RE, Yoon SO, Giehl KM, Herz J (2006) ApoE receptor 2 controls neuronal survival in the adult brain. Curr Biol 16 (24):2446–2452. https://doi.org/10.1016/j.cub.2006.10.029
Benson GS, Bauer C, Hausner L, Couturier S, Lewczuk P, Peters O, Hüll M, Jahn H, Jessen F, Pantel J, Teipel SJ, Wagner M, Schuchhardt J, Wiltfang J, Kornhuber J, Frölich L (2022) Don't forget about tau: the effects of ApoE4 genotype on Alzheimer's disease cerebrospinal fluid biomarkers in subjects with mild cognitive impairment-data from the Dementia Competence Network. J Neural Transm (Vienna, Austria : 1996) 129 (5–6):477–486. https://doi.org/10.1007/s00702-022-02461-0
Bentley NM, Ladu MJ, Rajan C, Getz GS, Reardon CA (2002) Apolipoprotein E structural requirements for the formation of SDS-stable complexes with beta-amyloid- (1–40): the role of salt bridges. Biochem J 366 (Pt 1):273–279. https://doi.org/10.1042/bj20020207
Bilousova T, Melnik M, Miyoshi E, Gonzalez BL, Poon WW, Vinters HV, Miller CA, Corrada MM, Kawas C, Hatami A, Albay R 3rd, Glabe C, Gylys KH (2019) Apolipoprotein E/amyloid-β complex accumulates in Alzheimer disease cortical synapses via Apolipoprotein E receptors and is enhanced by APOE4. Am J Pathol 189 (8):1621–1636. https://doi.org/10.1016/j.ajpath.2019.04.010
Blackman J, Love S, Sinclair L, Cain R, Coulthard E (2022) APOE ε4, Alzheimer’s disease neuropathology and sleep disturbance, in individuals with and without dementia. Alzheimer’s Res Therapy 14 (1):47. https://doi.org/10.1186/s13195-022-00992-y
Brecht WJ, Harris FM, Chang S, Tesseur I, Yu GQ, Xu Q, Dee Fish J, Wyss-Coray T, Buttini M, Mucke L, Mahley RW, Huang Y (2004) Neuron-specific Apolipoprotein E4 proteolysis is associated with increased tau phosphorylation in brains of transgenic mice. J Neurosci 24 (10):2527–2534. https://doi.org/10.1523/jneurosci.4315-03.2004
Bu G (2009) Apolipoprotein E and its receptors in Alzheimer’s disease: pathways, pathogenesis and therapy. Nat Rev Neurosci 10 (5):333–344. https://doi.org/10.1038/nrn2620
Burt TD, Agan BK, Marconi VC, He W, Kulkarni H, Mold JE, Cavrois M, Huang Y, Mahley RW, Dolan MJ, McCune JM, Ahuja SK (2008) Apolipoprotein (apo) E4 enhances HIV-1 cell entry in vitro, and the APOE epsilon4/epsilon4 genotype accelerates HIV disease progression. Proc Natl Acad Sci USA 105 (25):8718–8723. https://doi.org/10.1073/pnas.0803526105
Butterfield DA, Mattson MP (2020) Apolipoprotein E and oxidative stress in brain with relevance to Alzheimer’s disease. Neurobiol Disease 138:104795. https://doi.org/10.1016/j.nbd.2020.104795
Caberlotto L, Marchetti L, Lauria M, Scotti M, Parolo S (2016) Integration of transcriptomic and genomic data suggests candidate mechanisms for APOE4-mediated pathogenic action in Alzheimer’s disease. Sci Rep 6:32583. https://doi.org/10.1038/srep32583
Calvier L, Herz J, Hansmann G (2022) Interplay of low-density lipoprotein receptors, LRPs, and lipoproteins in pulmonary hypertension. JACC Basic Transl Sci 7 (2):164–180. https://doi.org/10.1016/j.jacbts.2021.09.011
Carlo AS (2013) Sortilin, a novel APOE receptor implicated in Alzheimer disease. Prion 7 (5):378–382. https://doi.org/10.4161/pri.26746
Carlo AS, Gustafsen C, Mastrobuoni G, Nielsen MS, Burgert T, Hartl D, Rohe M, Nykjaer A, Herz J, Heeren J, Kempa S, Petersen CM, Willnow TE (2013) The pro-neurotrophin receptor sortilin is a major neuronal Apolipoprotein E receptor for catabolism of amyloid-β peptide in the brain. J Neurosci 33 (1):358–370. https://doi.org/10.1523/jneurosci.2425-12.2013
Castellano JM, Kim J, Stewart FR, Jiang H, DeMattos RB, Patterson BW, Fagan AM, Morris JC, Mawuenyega KG, Cruchaga C, Goate AM, Bales KR, Paul SM, Bateman RJ, Holtzman DM (2011) Human apoE isoforms differentially regulate brain amyloid-β peptide clearance. Sci Transl Med 3 (89):89ra57. https://doi.org/10.1126/scitranslmed.3002156
Chang S, ran Ma T, Miranda RD, Balestra ME, Mahley RW, Huang Y (2005) Lipid- and receptor-binding regions of Apolipoprotein E4 fragments act in concert to cause mitochondrial dysfunction and neurotoxicity. Proc Natl Acad Sci USA 102 (51):18694–18699. https://doi.org/10.1073/pnas.0508254102
Chen Y, Durakoglugil MS, Xian X, Herz J (2010) ApoE4 reduces glutamate receptor function and synaptic plasticity by selectively impairing ApoE receptor recycling. Proc Natl Acad Sci USA 107 (26):12011–12016. https://doi.org/10.1073/pnas.0914984107
Chen HK, Ji ZS, Dodson SE, Miranda RD, Rosenblum CI, Reynolds IJ, Freedman SB, Weisgraber KH, Huang Y, Mahley RW (2011) Apolipoprotein E4 domain interaction mediates detrimental effects on mitochondria and is a potential therapeutic target for Alzheimer disease. J Biol Chem 286 (7):5215–5221. https://doi.org/10.1074/jbc.M110.151084
Chen GF, Xu TH, Yan Y, Zhou YR, Jiang Y, Melcher K, Xu HE (2017) Amyloid beta: structure, biology and structure-based therapeutic development. Acta Pharmacol Sin 38 (9):1205–1235. https://doi.org/10.1038/aps.2017.28
Chen DW, Shi JK, Li Y, Yang Y, Ren SP (2019) Association between ApoE polymorphism and type 2 diabetes: a meta-analysis of 59 studies. Biomed Environ Sci 32 (11):823–838. https://doi.org/10.3967/bes2019.104
Cooper JM, Lathuiliere A, Migliorini M, Arai AL, Wani MM, Dujardin S, Muratoglu SC, Hyman BT, Strickland DK (2021) Regulation of tau internalization, degradation, and seeding by LRP1 reveals multiple pathways for tau catabolism. J Biol Chem 296:100715. https://doi.org/10.1016/j.jbc.2021.100715
Cramer PE, Cirrito JR, Wesson DW, Lee CY, Karlo JC, Zinn AE, Casali BT, Restivo JL, Goebel WD, James MJ, Brunden KR, Wilson DA, Landreth GE (2012) ApoE-directed therapeutics rapidly clear β-amyloid and reverse deficits in AD mouse models. Science (new York, NY) 335 (6075):1503–1506. https://doi.org/10.1126/science.1217697
de Chaves EI, Rusiñol AE, Vance DE, Campenot RB, Vance JE (1997) Role of lipoproteins in the delivery of lipids to axons during axonal regeneration. J Biol Chem 272 (49):30766–30773. https://doi.org/10.1074/jbc.272.49.30766
Dietschy JM, Turley SD (2001) Cholesterol metabolism in the brain. Curr Opin Lipidol 12 (2):105–112. https://doi.org/10.1097/00041433-200104000-00003
Dietschy JM, Turley SD (2004) Thematic review series: brain Lipids. Cholesterol metabolism in the central nervous system during early development and in the mature animal. J Lipid Res 45 (8):1375–1397. https://doi.org/10.1194/jlr.R400004-JLR200
Dunk MM, Driscoll I (2022) Total cholesterol and APOE-related risk for Alzheimer’s disease in the Alzheimer’s disease neuroimaging initiative. J Alzheimer’s Disease 85 (4):1519–1528. https://doi.org/10.3233/jad-215091
Duro MV, Ebright B, Yassine HN (2022) Lipids and brain inflammation in APOE4-associated dementia. Curr Opin Lipidol 33 (1):16–24. https://doi.org/10.1097/mol.0000000000000801
Emrani S, Arain HA, DeMarshall C, Nuriel T (2020) APOE4 is associated with cognitive and pathological heterogeneity in patients with Alzheimer’s disease: a systematic review. Alzheimer’s Res Therapy 12 (1):141. https://doi.org/10.1186/s13195-020-00712-4
Ennerfelt H, Frost EL, Shapiro DA, Holliday C, Zengeler KE, Voithofer G, Bolte AC, Lammert CR, Kulas JA, Ulland TK, Lukens JR (2022) SYK coordinates neuroprotective microglial responses in neurodegenerative disease. Cell 185 (22):4135-4152.e4122. https://doi.org/10.1016/j.cell.2022.09.030
Evola M, Hall A, Wall T, Young A, Grammas P (2010) Oxidative stress impairs learning and memory in apoE knockout mice. Pharmacol Biochem Behav 96 (2):181–186. https://doi.org/10.1016/j.pbb.2010.05.003
Farfel JM, Yu L, De Jager PL, Schneider JA, Bennett DA (2016) Association of APOE with tau-tangle pathology with and without β-amyloid. Neurobiol Aging 37:19–25. https://doi.org/10.1016/j.neurobiolaging.2015.09.011
Farmer BC, Williams HC, Devanney NA, Piron MA, Nation GK, Carter DJ, Walsh AE, Khanal R, Young LEA, Kluemper JC, Hernandez G, Allenger EJ, Mooney R, Golden LR, Smith CT, Brandon JA, Gupta VA, Kern PA, Gentry MS, Morganti JM, Sun RC, Johnson LA (2021) APOΕ4 lowers energy expenditure in females and impairs glucose oxidation by increasing flux through aerobic glycolysis. Mol Neurodegener 16 (1):62. https://doi.org/10.1186/s13024-021-00483-y
Fitz NF, Nam KN, Wolfe CM, Letronne F, Playso BE, Iordanova BE, Kozai TDY, Biedrzycki RJ, Kagan VE, Tyurina YY, Han X, Lefterov I, Koldamova R (2021) Phospholipids of APOE lipoproteins activate microglia in an isoform-specific manner in preclinical models of Alzheimer’s disease. Nat Commun 12 (1):3416. https://doi.org/10.1038/s41467-021-23762-0
Fleisher AS, Chen K, Liu X, Ayutyanont N, Roontiva A, Thiyyagura P, Protas H, Joshi AD, Sabbagh M, Sadowsky CH, Sperling RA, Clark CM, Mintun MA, Pontecorvo MJ, Coleman RE, Doraiswamy PM, Johnson KA, Carpenter AP, Skovronsky DM, Reiman EM (2013) Apolipoprotein E ε4 and age effects on florbetapir positron emission tomography in healthy aging and Alzheimer disease. Neurobiol Aging 34 (1):1–12. https://doi.org/10.1016/j.neurobiolaging.2012.04.017
Ghiselli G, Schaefer EJ, Gascon P, Breser HB Jr (1981) Type III hyperlipoproteinemia associated with Apolipoprotein E deficiency. Science (new York, NY) 214 (4526):1239–1241. https://doi.org/10.1126/science.6795720
Gkouskou K, Vasilogiannakopoulou T, Andreakos E, Davanos N, Gazouli M, Sanoudou D, Eliopoulos AG (2021) COVID-19 enters the expanding network of Apolipoprotein E4-related pathologies. Redox Biol 41:101938. https://doi.org/10.1016/j.redox.2021.101938
Gratuze M, Jiang H, Wang C, Xiong M, Bao X, Holtzman DM (2022) APOE antibody inhibits Aβ-associated tau seeding and spreading in a mouse model. Ann Neurol 91 (6):847–852. https://doi.org/10.1002/ana.26351
Hashimoto T, Serrano-Pozo A, Hori Y, Adams KW, Takeda S, Banerji AO, Mitani A, Joyner D, Thyssen DH, Bacskai BJ, Frosch MP, Spires-Jones TL, Finn MB, Holtzman DM, Hyman BT (2012) Apolipoprotein E, especially Apolipoprotein E4, increases the oligomerization of amyloid β peptide. J Neurosci 32 (43):15181–15192. https://doi.org/10.1523/jneurosci.1542-12.2012
Hatters DM, Peters-Libeu CA, Weisgraber KH (2006) Apolipoprotein E structure: insights into function. Trends Biochem Sci 31 (8):445–454. https://doi.org/10.1016/j.tibs.2006.06.008
Hauser PS, Narayanaswami V, Ryan RO (2011) Apolipoprotein E: from lipid transport to neurobiology. Prog Lipid Res 50 (1):62–74. https://doi.org/10.1016/j.plipres.2010.09.001
Higashi S, Kabuta T, Nagai Y, Tsuchiya Y, Akiyama H, Wada K (2013) TDP-43 associates with stalled ribosomes and contributes to cell survival during cellular stress. J Neurochem 126 (2):288–300. https://doi.org/10.1111/jnc.12194
Hong DY, Lee DH, Lee JY, Lee EC, Park SW, Lee MR, Oh JS (2022) Relationship between brain metabolic disorders and cognitive impairment: LDL receptor defect. Int J Mol Sci. https://doi.org/10.3390/ijms23158384
Horiuchi Y, Ohkawa R, Lai SJ, Yamazaki A, Ikoma H, Yano K, Kameda T, Tozuka M (2019) Characterization of the cholesterol efflux of Apolipoprotein E-containing high-density lipoprotein in THP-1 cells. Biol Chem 400 (2):209–218. https://doi.org/10.1515/hsz-2018-0284
Hou X, Adeosun SO, Zhang Q, Barlow B, Brents M, Zheng B, Wang J (2015) Differential contributions of ApoE4 and female sex to BACE1 activity and expression mediate Aβ deposition and learning and memory in mouse models of Alzheimer’s disease. Front Aging Neurosci 7:207. https://doi.org/10.3389/fnagi.2015.00207
Howland DS, Trusko SP, Savage MJ, Reaume AG, Lang DM, Hirsch JD, Maeda N, Siman R, Greenberg BD, Scott RW, Flood DG (1998) Modulation of secreted beta-amyloid precursor protein and amyloid beta-peptide in brain by cholesterol. J Biol Chem 273 (26):16576–16582. https://doi.org/10.1074/jbc.273.26.16576
Hu HY, Ma LZ, Hu H, Bi YL, Ma YH, Shen XN, Ou YN, Dong Q, Tan L, Yu JT (2021) Associations of sleep characteristics with cerebrospinal fluid sTREM2 in cognitively normal older adults: the CABLE study. Neurotox Res 39 (4):1372–1380. https://doi.org/10.1007/s12640-021-00383-5
Huang Y, Weisgraber KH, Mucke L, Mahley RW (2004) Apolipoprotein E: diversity of cellular origins, structural and biophysical properties, and effects in Alzheimer’s disease. J Mol Neurosci 23 (3):189–204. https://doi.org/10.1385/jmn:23:3:189
Huang YA, Zhou B, Wernig M, Südhof TC (2017) ApoE2, ApoE3, and ApoE4 differentially stimulate APP transcription and Aβ secretion. Cell 168 (3):427-441.e421. https://doi.org/10.1016/j.cell.2016.12.044
Huang YA, Zhou B, Nabet AM, Wernig M, Südhof TC (2019) Differential signaling mediated by ApoE2, ApoE3, and ApoE4 in human neurons parallels Alzheimer’s disease risk. J Neurosci 39 (37):7408–7427. https://doi.org/10.1523/jneurosci.2994-18.2019
Huang S, Zhang Z, Cao J, Yu Y, Pei G (2022) Chimeric cerebral organoids reveal the essentials of neuronal and astrocytic APOE4 for Alzheimer’s tau pathology. Signal Transduct Target Ther 7 (1):176. https://doi.org/10.1038/s41392-022-01006-x
Hultman K, Strickland S, Norris EH (2013) The APOE ɛ4/ɛ4 genotype potentiates vascular fibrin (ogen) deposition in amyloid-laden vessels in the brains of Alzheimer’s disease patients. J Cerebral Blood Flow Metab 33 (8):1251–1258. https://doi.org/10.1038/jcbfm.2013.76
Iannucci J, Sen A, Grammas P (2021) Isoform-specific effects of Apolipoprotein E on markers of inflammation and toxicity in brain glia and neuronal cells in vitro. Curr Issues Mol Biol 43 (1):215–225. https://doi.org/10.3390/cimb43010018
Ihara Y, Hayabara T, Sasaki K, Kawada R, Nakashima Y, Kuroda S (2000) Relationship between oxidative stress and apoE phenotype in Alzheimer’s disease. Acta Neurol Scand 102 (6):346–349. https://doi.org/10.1034/j.1600-0404.2000.102006346.x
Jablonski AM, Warren L, Usenovic M, Zhou H, Sugam J, Parmentier-Batteur S, Voleti B (2021) Astrocytic expression of the Alzheimer’s disease risk allele, ApoEε4, potentiates neuronal tau pathology in multiple preclinical models. Sci Rep 11 (1):3438. https://doi.org/10.1038/s41598-021-82901-1
Jack CR Jr, Knopman DS, Jagust WJ, Shaw LM, Aisen PS, Weiner MW, Petersen RC, Trojanowski JQ (2010) Hypothetical model of dynamic biomarkers of the Alzheimer’s pathological cascade. Lancet Neurol 9 (1):119–128. https://doi.org/10.1016/s1474-4422(09)70299-6
Jeong W, Lee H, Cho S, Seo J (2019) ApoE4-induced cholesterol dysregulation and its brain cell type-specific implications in the pathogenesis of Alzheimer’s disease. Mol Cells 42 (11):739–746. https://doi.org/10.14348/molcells.2019.0200
Jiang L, Lin H, Alzheimer’s Disease Neuroimaging I, Chen Y (2020) Sex difference in the association of APOE4 with cerebral glucose metabolism in older adults reporting significant memory concern. Neurosci Lett 722:134824. https://doi.org/10.1016/j.neulet.2020.134824
Jofre-Monseny L, Minihane AM, Rimbach G (2008) Impact of apoE genotype on oxidative stress, inflammation and disease risk. Mol Nutr Food Res 52 (1):131–145. https://doi.org/10.1002/mnfr.200700322
John A, Reddy PH (2021) Synaptic basis of Alzheimer’s disease: focus on synaptic amyloid beta, P-tau and mitochondria. Ageing Res Rev 65:101208. https://doi.org/10.1016/j.arr.2020.101208
Johnson NR, Wang AC, Coughlan C, Sillau S, Lucero E, Viltz L, Markham N, Allen C, Dhanasekaran AR, Chial HJ, Potter H (2022) Imipramine and olanzapine block apoE4-catalyzed polymerization of Aβ and show evidence of improving Alzheimer’s disease cognition. Alzheimer’s Res Therapy 14 (1):88. https://doi.org/10.1186/s13195-022-01020-9
Jones PB, Adams KW, Rozkalne A, Spires-Jones TL, Hshieh TT, Hashimoto T, von Armin CA, Mielke M, Bacskai BJ, Hyman BT (2011) Apolipoprotein E: isoform specific differences in tertiary structure and interaction with amyloid-β in human Alzheimer brain. PLoS ONE 6 (1):e14586. https://doi.org/10.1371/journal.pone.0014586
Kang SS, Ahn EH, Liu X, Bryson M, Miller GW, Weinshenker D, Ye K (2021) ApoE4 inhibition of VMAT2 in the locus coeruleus exacerbates Tau pathology in Alzheimer’s disease. Acta Neuropathol 142 (1):139–158. https://doi.org/10.1007/s00401-021-02315-1
Kao YC, Ho PC, Tu YK, Jou IM, Tsai KJ (2020) Lipids and Alzheimer’s disease. Int J Mol Sci. https://doi.org/10.3390/ijms21041505
Kara E, Marks JD, Roe AD, Commins C, Fan Z, Calvo-Rodriguez M, Wegmann S, Hudry E, Hyman BT (2018) A flow cytometry-based in vitro assay reveals that formation of Apolipoprotein E (ApoE)-amyloid beta complexes depends on ApoE isoform and cell type. J Biol Chem 293 (34):13247–13256. https://doi.org/10.1074/jbc.RA117.001388
Kim J, Yoon H, Basak J, Kim J (2014) Apolipoprotein E in synaptic plasticity and Alzheimer’s disease: potential cellular and molecular mechanisms. Mol Cells 37 (11):767–776. https://doi.org/10.14348/molcells.2014.0248
Kockx M, Jessup W, Kritharides L (2008) Regulation of endogenous Apolipoprotein E secretion by macrophages. Arterioscler Thromb Vasc Biol 28 (6):1060–1067. https://doi.org/10.1161/atvbaha.108.164350
Koo KYG, Schweizer TA, Fischer CE, Munoz DG (2019) Abnormal sleep behaviours across the spectrum of Alzheimer’s disease severity: influence of APOE genotypes and Lewy bodies. Curr Alzheimer Res 16 (3):243–250. https://doi.org/10.2174/1567205016666190103161034
Koutsodendris N, Nelson MR, Rao A, Huang Y (2022) Apolipoprotein E and Alzheimer’s disease: findings, hypotheses, and potential mechanisms. Annu Rev Pathol 17:73–99. https://doi.org/10.1146/annurev-pathmechdis-030421-112756
Krasemann S, Madore C, Cialic R, Baufeld C, Calcagno N, El Fatimy R, Beckers L, O’Loughlin E, Xu Y, Fanek Z, Greco DJ, Smith ST, Tweet G, Humulock Z, Zrzavy T, Conde-Sanroman P, Gacias M, Weng Z, Chen H, Tjon E, Mazaheri F, Hartmann K, Madi A, Ulrich JD, Glatzel M, Worthmann A, Heeren J, Budnik B, Lemere C, Ikezu T, Heppner FL, Litvak V, Holtzman DM, Lassmann H, Weiner HL, Ochando J, Haass C, Butovsky O (2017) The TREM2-APOE pathway drives the transcriptional phenotype of dysfunctional microglia in neurodegenerative diseases. Immunity 47 (3):566-581.e569. https://doi.org/10.1016/j.immuni.2017.08.008
Krishnamurthy K, Cantillana V, Wang H, Sullivan PM, Kolls BJ, Ge X, Lin Y, Mace B, Laskowitz DT (2020) ApoE mimetic improves pathology and memory in a model of Alzheimer’s disease. Brain Res 1733:146685. https://doi.org/10.1016/j.brainres.2020.146685
Kuszczyk MA, Sanchez S, Pankiewicz J, Kim J, Duszczyk M, Guridi M, Asuni AA, Sullivan PM, Holtzman DM, Sadowski MJ (2013) Blocking the interaction between Apolipoprotein E and Aβ reduces intraneuronal accumulation of Aβ and inhibits synaptic degeneration. Am J Pathol 182 (5):1750–1768. https://doi.org/10.1016/j.ajpath.2013.01.034
Lane-Donovan C, Herz J (2017) ApoE, ApoE receptors, and the synapse in Alzheimer’s disease. Trends Endocrinol Metab 28 (4):273–284. https://doi.org/10.1016/j.tem.2016.12.001
Lanfranco MF, Ng CA, Rebeck GW (2020) ApoE lipidation as a therapeutic target in Alzheimer's disease. Int J Mol Sci. https://doi.org/10.3390/ijms21176336
Lanfranco MF, Sepulveda J, Kopetsky G, Rebeck GW (2021) Expression and secretion of apoE isoforms in astrocytes and microglia during inflammation. Glia 69 (6):1478–1493. https://doi.org/10.1002/glia.23974
Lauderback CM, Kanski J, Hackett JM, Maeda N, Kindy MS, Butterfield DA (2002) Apolipoprotein E modulates Alzheimer’s Abeta (1–42)-induced oxidative damage to synaptosomes in an allele-specific manner. Brain Res 924 (1):90–97. https://doi.org/10.1016/s0006-8993(01)03228-0
Lee SI, Jeong W, Lim H, Cho S, Lee H, Jang Y, Cho J, Bae S, Lin YT, Tsai LH, Moon DW, Seo J (2021) APOE4-carrying human astrocytes oversupply cholesterol to promote neuronal lipid raft expansion and Aβ generation. Stem Cell Rep 16 (9):2128–2137. https://doi.org/10.1016/j.stemcr.2021.07.017
Lee S, Devanney NA, Golden LR, Smith CT, Schwartz JL, Walsh AE, Clarke HA, Goulding DS, Allenger EJ, Morillo-Segovia G, Friday CM, Gorman AA, Hawkinson TR, MacLean SM, Williams HC, Sun RC, Morganti JM, Johnson LA (2023) APOE modulates microglial immunometabolism in response to age, amyloid pathology, and inflammatory challenge. Cell Rep 42 (3):112196. https://doi.org/10.1016/j.celrep.2023.112196
Li X, Montine KS, Keene CD, Montine TJ (2015) Different mechanisms of Apolipoprotein E isoform-dependent modulation of prostaglandin E2 production and triggering receptor expressed on myeloid cells 2 (TREM2) expression after innate immune activation of microglia. FASEB J 29 (5):1754–1762. https://doi.org/10.1096/fj.14-262683
Li L, Xu W, Tan CC, Cao XP, Wei BZ, Dong CW, Tan L (2022) A gene-environment interplay between omega-3 supplementation and APOE ε4 provides insights for Alzheimer’s disease precise prevention amongst high-genetic-risk population. Eur J Neurol 29 (2):422–431. https://doi.org/10.1111/ene.15160
Liang T, Hang W, Chen J, Wu Y, Wen B, Xu K, Ding B, Chen J (2021) ApoE4 (Δ272-299) induces mitochondrial-associated membrane formation and mitochondrial impairment by enhancing GRP75-modulated mitochondrial calcium overload in neuron. Cell Biosci 11 (1):50. https://doi.org/10.1186/s13578-021-00563-y
Liao F, Hori Y, Hudry E, Bauer AQ, Jiang H, Mahan TE, Lefton KB, Zhang TJ, Dearborn JT, Kim J, Culver JP, Betensky R, Wozniak DF, Hyman BT, Holtzman DM (2014) Anti-ApoE antibody given after plaque onset decreases Aβ accumulation and improves brain function in a mouse model of Aβ amyloidosis. J Neurosci 34 (21):7281–7292. https://doi.org/10.1523/jneurosci.0646-14.2014
Liao F, Li A, Xiong M, Bien-Ly N, Jiang H, Zhang Y, Finn MB, Hoyle R, Keyser J, Lefton KB, Robinson GO, Serrano JR, Silverman AP, Guo JL, Getz J, Henne K, Leyns CE, Gallardo G, Ulrich JD, Sullivan PM, Lerner EP, Hudry E, Sweeney ZK, Dennis MS, Hyman BT, Watts RJ, Holtzman DM (2018) Targeting of nonlipidated, aggregated apoE with antibodies inhibits amyloid accumulation. J Clin Investig 128 (5):2144–2155. https://doi.org/10.1172/jci96429
Lim AS, Yu L, Kowgier M, Schneider JA, Buchman AS, Bennett DA (2013) Modification of the relationship of the Apolipoprotein E ε4 allele to the risk of Alzheimer disease and neurofibrillary tangle density by sleep. JAMA Neurol 70 (12):1544–1551. https://doi.org/10.1001/jamaneurol.2013.4215
Lin YT, Seo J, Gao F, Feldman HM, Wen HL, Penney J, Cam HP, Gjoneska E, Raja WK, Cheng J, Rueda R, Kritskiy O, Abdurrob F, Peng Z, Milo B, Yu CJ, Elmsaouri S, Dey D, Ko T, Yankner BA, Tsai LH (2018) APOE4 causes widespread molecular and cellular alterations associated with Alzheimer’s disease phenotypes in human iPSC-derived brain cell types. Neuron 98 (6):1141-1154.e1147. https://doi.org/10.1016/j.neuron.2018.05.008
Linton MF, Gish R, Hubl ST, Bütler E, Esquivel C, Bry WI, Boyles JK, Wardell MR, Young SG (1991) Phenotypes of apolipoprotein B and Apolipoprotein E after liver transplantation. J Clin Investig 88 (1):270–281. https://doi.org/10.1172/jci115288
Liu CC, Liu CC, Kanekiyo T, Xu H, Bu G (2013) Apolipoprotein E and Alzheimer disease: risk, mechanisms and therapy. Nat Rev Neurol 9 (2):106–118. https://doi.org/10.1038/nrneurol.2012.263
Liu CC, Tsai CW, Deak F, Rogers J, Penuliar M, Sung YM, Maher JN, Fu Y, Li X, Xu H, Estus S, Hoe HS, Fryer JD, Kanekiyo T, Bu G (2014a) Deficiency in LRP6-mediated Wnt signaling contributes to synaptic abnormalities and amyloid pathology in Alzheimer’s disease. Neuron 84 (1):63–77. https://doi.org/10.1016/j.neuron.2014.08.048
Liu S, Breitbart A, Sun Y, Mehta PD, Boutajangout A, Scholtzova H, Wisniewski T (2014b) Blocking the Apolipoprotein E/amyloid β interaction in triple transgenic mice ameliorates Alzheimer’s disease related amyloid β and tau pathology. J Neurochem 128 (4):577–591. https://doi.org/10.1111/jnc.12484
Lynch JR, Tang W, Wang H, Vitek MP, Bennett ER, Sullivan PM, Warner DS, Laskowitz DT (2003) APOE genotype and an ApoE-mimetic peptide modify the systemic and central nervous system inflammatory response. J Biol Chem 278 (49):48529–48533. https://doi.org/10.1074/jbc.M306923200
Ma Q, Zhao Z, Sagare AP, Wu Y, Wang M, Owens NC, Verghese PB, Herz J, Holtzman DM, Zlokovic BV (2018) Blood-brain barrier-associated pericytes internalize and clear aggregated amyloid-β42 by LRP1-dependent Apolipoprotein E isoform-specific mechanism. Mol Neurodegener 13 (1):57. https://doi.org/10.1186/s13024-018-0286-0
Mahley RW (1988) Apolipoprotein E: cholesterol transport protein with expanding role in cell biology. Science (new York, NY) 240 (4852):622–630. https://doi.org/10.1126/science.3283935
Mahley RW, Hui DY, Innerarity TL, Beisiegel U (1989) Chylomicron remnant metabolism. Role of hepatic lipoprotein receptors in mediating uptake. Arteriosclerosis (Dallas, Tex) 9 (1 Suppl):I14–18
Mai Z, Wei W, Yu H, Chen Y, Wang Y, Ding Y (2022) Molecular recognition of the interaction between ApoE and the TREM2 protein. Transl Neurosci 13 (1):93–103. https://doi.org/10.1515/tnsci-2022-0218
Marquer C, Laine J, Dauphinot L, Hanbouch L, Lemercier-Neuillet C, Pierrot N, Bossers K, Le M, Corlier F, Benstaali C, Saudou F, Thinakaran G, Cartier N, Octave JN, Duyckaerts C, Potier MC (2014) Increasing membrane cholesterol of neurons in culture recapitulates Alzheimer’s disease early phenotypes. Mol Neurodegener 9:60. https://doi.org/10.1186/1750-1326-9-60
Mauch DH, Nägler K, Schumacher S, Göritz C, Müller EC, Otto A, Pfrieger FW (2001) CNS synaptogenesis promoted by glia-derived cholesterol. Science (new York, NY) 294 (5545):1354–1357. https://doi.org/10.1126/science.294.5545.1354
Mhatre-Winters I, Eid A, Han Y, Tieu K, Richardson JR (2022) Sex and APOE genotype alter the basal and induced inflammatory states of primary microglia from APOE targeted replacement mice. Int J Mol Sci. https://doi.org/10.3390/ijms23179829
Miyata M, Smith JD (1996) Apolipoprotein E allele-specific antioxidant activity and effects on cytotoxicity by oxidative insults and beta-amyloid peptides. Nat Genet 14 (1):55–61. https://doi.org/10.1038/ng0996-55
Montagne A, Nation DA, Sagare AP, Barisano G, Sweeney MD, Chakhoyan A, Pachicano M, Joe E, Nelson AR, D’Orazio LM, Buennagel DP, Harrington MG, Benzinger TLS, Fagan AM, Ringman JM, Schneider LS, Morris JC, Reiman EM, Caselli RJ, Chui HC, Tcw J, Chen Y, Pa J, Conti PS, Law M, Toga AW, Zlokovic BV (2020) APOE4 leads to blood-brain barrier dysfunction predicting cognitive decline. Nature 581 (7806):71–76. https://doi.org/10.1038/s41586-020-2247-3
Montagne A, Nikolakopoulou AM, Huuskonen MT, Sagare AP, Lawson EJ, Lazic D, Rege SV, Grond A, Zuniga E, Barnes SR, Prince J, Sagare M, Hsu CJ, LaDu MJ, Jacobs RE, Zlokovic BV (2021) APOE4 accelerates advanced-stage vascular and neurodegenerative disorder in old Alzheimer’s mice via cyclophilin A independently of amyloid-β. Nat Aging 1 (6):506–520. https://doi.org/10.1038/s43587-021-00073-z
Morrill SJ, Gibas KJ (2019) Ketogenic diet rescues cognition in ApoE4+ patient with mild Alzheimer’s disease: a case study. Diabetes Metabolic Syndrome 13 (2):1187–1191. https://doi.org/10.1016/j.dsx.2019.01.035
Morrow JA, Hatters DM, Lu B, Hochtl P, Oberg KA, Rupp B, Weisgraber KH (2002) Apolipoprotein E4 forms a molten globule. A potential basis for its association with disease. J Biol Chem 277 (52):50380–50385. https://doi.org/10.1074/jbc.M204898200
Mouchard A, Boutonnet MC, Mazzocco C, Biendon N, Macrez N (2019) ApoE-fragment/Aβ heteromers in the brain of patients with Alzheimer’s disease. Sci Rep 9 (1):3989. https://doi.org/10.1038/s41598-019-40438-4
Muralidar S, Ambi SV, Sekaran S, Thirumalai D, Palaniappan B (2020) Role of tau protein in Alzheimer’s disease: The prime pathological player. Int J Biol Macromol 163:1599–1617. https://doi.org/10.1016/j.ijbiomac.2020.07.327
Nakajima C, Kulik A, Frotscher M, Herz J, Schäfer M, Bock HH, May P (2013) Low density lipoprotein receptor-related protein 1 (LRP1) modulates N-methyl-D-aspartate (NMDA) receptor-dependent intracellular signaling and NMDA-induced regulation of postsynaptic protein complexes. J Biol Chem 288 (30):21909–21923. https://doi.org/10.1074/jbc.M112.444364
Nakamura T, Watanabe A, Fujino T, Hosono T, Michikawa M (2009) Apolipoprotein E4 (1–272) fragment is associated with mitochondrial proteins and affects mitochondrial function in neuronal cells. Mol Neurodegener 4:35. https://doi.org/10.1186/1750-1326-4-35
Norwitz NG, Saif N, Ariza IE, Isaacson RS (2021) Precision nutrition for Alzheimer's prevention in ApoE4 Carriers. Nutrients https://doi.org/10.3390/nu13041362
Novy MJ, Newbury SF, Liemisa B, Morales-Corraliza J, Alldred MJ, Ginsberg SD, Mathews PM (2022) Expression and proteolytic processing of the amyloid precursor protein is unaffected by the expression of the three human Apolipoprotein E alleles in the brains of mice. Neurobiol Aging 110:73–76. https://doi.org/10.1016/j.neurobiolaging.2021.10.015
Orr AL, Kim C, Jimenez-Morales D, Newton BW, Johnson JR, Krogan NJ, Swaney DL, Mahley RW (2019) Neuronal Apolipoprotein E4 expression results in proteome-wide alterations and compromises bioenergetic capacity by disrupting mitochondrial function. J Alzheimer’s Disease 68 (3):991–1011. https://doi.org/10.3233/jad-181184
Pietrzik CU, Busse T, Merriam DE, Weggen S, Koo EH (2002) The cytoplasmic domain of the LDL receptor-related protein regulates multiple steps in APP processing. EMBO J 21 (21):5691–5700. https://doi.org/10.1093/emboj/cdf568
Poirier J, Hess M, May PC, Finch CE (1991) Astrocytic Apolipoprotein E mRNA and GFAP mRNA in hippocampus after entorhinal cortex lesioning. Brain Res Mol Brain Res 11 (2):97–106. https://doi.org/10.1016/0169-328x(91)90111-a
Polazzi E, Mengoni I, Peña-Altamira E, Massenzio F, Virgili M, Petralla S, Monti B (2015) Neuronal regulation of neuroprotective microglial Apolipoprotein E secretion in rat in vitro models of brain pathophysiology. J Neuropathol Exp Neurol 74 (8):818–834. https://doi.org/10.1097/nen.0000000000000222
Pontifex MG, Martinsen A, Saleh RNM, Harden G, Fox C, Muller M, Vauzour D, Minihane AM (2022) DHA-enriched fish oil ameliorates deficits in cognition associated with menopause and the APOE4 genotype in rodents. Nutrients. https://doi.org/10.3390/nu14091698
Rauch JN, Luna G, Guzman E, Audouard M, Challis C, Sibih YE, Leshuk C, Hernandez I, Wegmann S, Hyman BT, Gradinaru V, Kampmann M, Kosik KS (2020) LRP1 is a master regulator of tau uptake and spread. Nature 580 (7803):381–385. https://doi.org/10.1038/s41586-020-2156-5
Rawat V, Wang S, Sima J, Bar R, Liraz O, Gundimeda U, Parekh T, Chan J, Johansson JO, Tang C, Chui HC, Harrington MG, Michaelson DM, Yassine HN (2019) ApoE4 alters ABCA1 membrane trafficking in astrocytes. J Neurosci 39 (48):9611–9622. https://doi.org/10.1523/jneurosci.1400-19.2019
Robb WH, Khan OA, Ahmed HA, Li J, Moore EE, Cambronero FE, Pechman KR, Liu D, Gifford KA, Landman BA, Donahue MJ, Hohman TJ, Jefferson AL (2022) Lower cerebral oxygen utilization is associated with Alzheimer’s disease-related neurodegeneration and poorer cognitive performance among Apolipoprotein E ε4 carriers. J Cerebral Blood Flow Metab 42 (4):642–655. https://doi.org/10.1177/0271678x211056393
Sabbagh MN, Malek-Ahmadi M, Dugger BN, Lee K, Sue LI, Serrano G, Walker DG, Davis K, Jacobson SA, Beach TG (2013) The influence of Apolipoprotein E genotype on regional pathology in Alzheimer’s disease. BMC Neurol 13:44. https://doi.org/10.1186/1471-2377-13-44
Sadowski M, Pankiewicz J, Scholtzova H, Ripellino JA, Li Y, Schmidt SD, Mathews PM, Fryer JD, Holtzman DM, Sigurdsson EM, Wisniewski T (2004) A synthetic peptide blocking the Apolipoprotein E/beta-amyloid binding mitigates beta-amyloid toxicity and fibril formation in vitro and reduces beta-amyloid plaques in transgenic mice. Am J Pathol 165 (3):937–948. https://doi.org/10.1016/s0002-9440(10)63355-x
Safieh M, Korczyn AD, Michaelson DM (2019) ApoE4: an emerging therapeutic target for Alzheimer’s disease. BMC Med 17 (1):64. https://doi.org/10.1186/s12916-019-1299-4
Saito T, Matsunaga A, Fukunaga M, Nagahama K, Hara S, Muso E (2020) Apolipoprotein E-related glomerular disorders. Kidney Int 97 (2):279–288. https://doi.org/10.1016/j.kint.2019.10.031
Salami A, Adolfsson R, Andersson M, Blennow K, Lundquist A, Adolfsson AN, Schöll M, Zetterberg H, Nyberg L (2022) Association of APOE ɛ4 and plasma p-tau181 with preclinical Alzheimer’s disease and longitudinal change in hippocampus function. J Alzheimer’s Dis 85 (3):1309–1320. https://doi.org/10.3233/jad-210673
Santos DB, Peres KC, Ribeiro RP, Colle D, dos Santos AA, Moreira EL, Souza DO, Figueiredo CP, Farina M (2012) Probucol, a lipid-lowering drug, prevents cognitive and hippocampal synaptic impairments induced by amyloid β peptide in mice. Exp Neurol 233 (2):767–775. https://doi.org/10.1016/j.expneurol.2011.11.036
Saroja SR, Gorbachev K, Julia T, Goate AM, Pereira AC (2022) Astrocyte-secreted glypican-4 drives APOE4-dependent tau hyperphosphorylation. Proc Natl Acad Sci USA 119 (34):e2108870119. https://doi.org/10.1073/pnas.2108870119
Saunders AM, Strittmatter WJ, Schmechel D, George-Hyslop PH, Pericak-Vance MA, Joo SH, Rosi BL, Gusella JF, Crapper-MacLachlan DR, Alberts MJ et al (1993) Association of Apolipoprotein E allele epsilon 4 with late-onset familial and sporadic Alzheimer’s disease. Neurology 43 (8):1467–1472. https://doi.org/10.1212/wnl.43.8.1467
Sayed FA, Kodama L, Fan L, Carling GK, Udeochu JC, Le D, Li Q, Zhou L, Wong MY, Horowitz R, Ye P, Mathys H, Wang M, Niu X, Mazutis L, Jiang X, Wang X, Gao F, Brendel M, Telpoukhovskaia M, Tracy TE, Frost G, Zhou Y, Li Y, Qiu Y, Cheng Z, Yu G, Hardy J, Coppola G, Wang F, DeTure MA, Zhang B, Xie L, Trajnowski JQ, Lee VMY, Gong S, Sinha SC, Dickson DW, Luo W, Gan L (2021) AD-linked R47H-TREM2 mutation induces disease-enhancing microglial states via AKT hyperactivation. Sci Transl Med 13 (622):eabe3947. https://doi.org/10.1126/scitranslmed.abe3947
Scheinman SB, Zaldua S, Dada A, Krochmaliuk K, Dye K, Marottoli FM, Thatcher GRJ, Tai LM (2021) Systemic candesartan treatment modulates behavior, synaptic protein levels, and neuroinflammation in female mice that express human APOE4. Front Neurosci 15:628403. https://doi.org/10.3389/fnins.2021.628403
Schmid B, Prehn KR, Nimsanor N, Garcia BIA, Poulsen U, Jørring I, Rasmussen MA, Clausen C, Mau-Holzmann UA, Ramakrishna S, Muddashetty R, Steeg R, Bruce K, Mackintosh P, Ebneth A, Holst B, Cabrera-Socorro A (2019) Generation of a set of isogenic, gene-edited iPSC lines homozygous for all main APOE variants and an APOE knock-out line. Stem Cell Res 34:101349. https://doi.org/10.1016/j.scr.2018.11.010
Serrano-Pozo A, Qian J, Monsell SE, Betensky RA, Hyman BT (2015) APOEε2 is associated with milder clinical and pathological Alzheimer disease. Ann Neurol 77 (6):917–929. https://doi.org/10.1002/ana.24369
Serrano-Pozo A, Das S, Hyman BT (2021) APOE and Alzheimer’s disease: advances in genetics, pathophysiology, and therapeutic approaches. Lancet Neurol 20 (1):68–80. https://doi.org/10.1016/s1474-4422(20)30412-9
Shi Y, Yamada K, Liddelow SA, Smith ST, Zhao L, Luo W, Tsai RM, Spina S, Grinberg LT, Rojas JC, Gallardo G, Wang K, Roh J, Robinson G, Finn MB, Jiang H, Sullivan PM, Baufeld C, Wood MW, Sutphen C, McCue L, Xiong C, Del-Aguila JL, Morris JC, Cruchaga C, Fagan AM, Miller BL, Boxer AL, Seeley WW, Butovsky O, Barres BA, Paul SM, Holtzman DM (2017) ApoE4 markedly exacerbates tau-mediated neurodegeneration in a mouse model of tauopathy. Nature 549 (7673):523–527. https://doi.org/10.1038/nature24016
Shi Y, Andhey PS, Ising C, Wang K, Snipes LL, Boyer K, Lawson S, Yamada K, Qin W, Manis M, Serrano JR, Benitez BA, Schmidt RE, Artyomov M, Ulrich JD, Holtzman DM (2021) Overexpressing low-density lipoprotein receptor reduces tau-associated neurodegeneration in relation to apoE-linked mechanisms. Neuron 109 (15):2413-2426.e2417. https://doi.org/10.1016/j.neuron.2021.05.034
Shi D, Xie S, Li A, Wang Q, Guo H, Han Y, Xu H, Gan WB, Zhang L, Guo T (2022) APOE-ε4 modulates the association among plasma Aβ (42)/Aβ (40), vascular diseases, neurodegeneration and cognitive decline in non-demented elderly adults. Transl Psychiatry 12 (1):128. https://doi.org/10.1038/s41398-022-01899-w
Shinohara M, Shinohara M, Zhao J, Fu Y, Liu CC, Kanekiyo T, Bu G (2019) 5-HT3 antagonist ondansetron increases ApoE secretion by modulating the LXR-ABCA1 pathway. Int J Mol Sci. https://doi.org/10.3390/ijms20061488
Shinohara M, Tashiro Y, Suzuki K, Fukumori A, Bu G, Sato N (2020) Interaction between APOE genotype and diabetes in cognitive decline. Alzheimer’s Dementia (amsterdam, Netherlands) 12 (1):e12006. https://doi.org/10.1002/dad2.12006
Sohn HY, Kim SI, Park JY, Park SH, Koh YH, Kim J, Jo C (2021) ApoE4 attenuates autophagy via FoxO3a repression in the brain. Sci Rep 11 (1):17604. https://doi.org/10.1038/s41598-021-97117-6
Stephen TL, Breningstall B, Suresh S, McGill CJ, Pike CJ (2022) APOE genotype and biological sex regulate astroglial interactions with amyloid plaques in Alzheimer’s disease mice. J Neuroinflam 19 (1):286. https://doi.org/10.1186/s12974-022-02650-4
Stover CM, Lynch NJ, Hanson SJ, Windbichler M, Gregory SG, Schwaeble WJ (2004) Organization of the MASP2 locus and its expression profile in mouse and rat. Mamm Genome 15 (11):887–900. https://doi.org/10.1007/s00335-004-3006-8
Strasser V, Fasching D, Hauser C, Mayer H, Bock HH, Hiesberger T, Herz J, Weeber EJ, Sweatt JD, Pramatarova A, Howell B, Schneider WJ, Nimpf J (2004) Receptor clustering is involved in Reelin signaling. Mol Cell Biol 24 (3):1378–1386. https://doi.org/10.1128/mcb.24.3.1378-1386.2004
Sun X, Dong C, Levin B, Crocco E, Loewenstein D, Zetterberg H, Blennow K, Wright CB (2016) APOE ε4 carriers may undergo synaptic damage conferring risk of Alzheimer’s disease. Alzheimer’s Dementia 12 (11):1159–1166. https://doi.org/10.1016/j.jalz.2016.05.003
Sun GZ, He YC, Ma XK, Li ST, Chen DJ, Gao M, Qiu SF, Yin JX, Shi J, Wu J (2017) Hippocampal synaptic and neural network deficits in young mice carrying the human APOE4 gene. CNS Neurosci Ther 23 (9):748–758. https://doi.org/10.1111/cns.12720
Tachibana M, Holm ML, Liu CC, Shinohara M, Aikawa T, Oue H, Yamazaki Y, Martens YA, Murray ME, Sullivan PM, Weyer K, Glerup S, Dickson DW, Bu G, Kanekiyo T (2019) APOE4-mediated amyloid-β pathology depends on its neuronal receptor LRP1. J Clin Investig 129 (3):1272–1277. https://doi.org/10.1172/jci124853
Tambini MD, Pera M, Kanter E, Yang H, Guardia-Laguarta C, Holtzman D, Sulzer D, Area-Gomez E, Schon EA (2016) ApoE4 upregulates the activity of mitochondria-associated ER membranes. EMBO Rep 17 (1):27–36. https://doi.org/10.15252/embr.201540614
Teylan MA, Mock C, Gauthreaux K, Culhane JE, Jicha G, Chen YC, Chan KCG, Kukull WA, Nelson PT, Katsumata Y (2021) Differences in symptomatic presentation and cognitive performance among participants with LATE-NC compared to FTLD-TDP. J Neuropathol Exp Neurol 80 (11):1024–1032. https://doi.org/10.1093/jnen/nlab098
Troutwine BR, Strope TA, Franczak E, Lysaker CR, Hamid L, Mansel C, Stopperan JA, Gouvion CM, Haeri M, Swerdlow RH, Wilkins HM (2022) Mitochondrial function and Aβ in Alzheimer’s disease postmortem brain. Neurobiol Disease 171:105781. https://doi.org/10.1016/j.nbd.2022.105781
Ulery PG, Beers J, Mikhailenko I, Tanzi RE, Rebeck GW, Hyman BT, Strickland DK (2000) Modulation of beta-amyloid precursor protein processing by the low density lipoprotein receptor-related protein (LRP) Evidence that LRP contributes to the pathogenesis of Alzheimer’s disease. J Biol Chem 275 (10):7410–7415. https://doi.org/10.1074/jbc.275.10.7410
Ulrich JD, Holtzman DM (2016) TREM2 function in Alzheimer’s disease and neurodegeneration. ACS Chem Neurosci 7 (4):420–427. https://doi.org/10.1021/acschemneuro.5b00313
Venegas-Pino DE, Wang PW, Stoute HK, Singh-Pickersgill NA, Hong BY, Khan MI, Shi Y, Werstuck GH (2016) Sex-specific differences in an ApoE (-/-):Ins2 (+/Akita) mouse model of accelerated atherosclerosis. Am J Pathol 186 (1):67–77. https://doi.org/10.1016/j.ajpath.2015.09.009
Wang C, Najm R, Xu Q, Jeong DE, Walker D, Balestra ME, Yoon SY, Yuan H, Li G, Miller ZA, Miller BL, Malloy MJ, Huang Y (2018) Gain of toxic Apolipoprotein E4 effects in human iPSC-derived neurons is ameliorated by a small-molecule structure corrector. Nat Med 24 (5):647–657. https://doi.org/10.1038/s41591-018-0004-z
Wang Y, Lin Y, Wang L, Zhan H, Luo X, Zeng Y, Wu W, Zhang X, Wang F (2020) TREM2 ameliorates neuroinflammatory response and cognitive impairment via PI3K/AKT/FoxO3a signaling pathway in Alzheimer’s disease mice. Aging 12 (20):20862–20879. https://doi.org/10.18632/aging.104104
Wang ZH, Xia Y, Liu P, Liu X, Edgington-Mitchell L, Lei K, Yu SP, Wang XC, Ye K (2021) ApoE4 activates C/EBPβ/δ-secretase with 27-hydroxycholesterol, driving the pathogenesis of Alzheimer’s disease. Progr Neurobiol 202:102032. https://doi.org/10.1016/j.pneurobio.2021.102032
Wang S, Sudan R, Peng V, Zhou Y, Du S, Yuede CM, Lei T, Hou J, Cai Z, Cella M, Nguyen K, Poliani PL, Beatty WL, Chen Y, Cao S, Lin K, Rodrigues C, Ellebedy AH, Gilfillan S, Brown GD, Holtzman DM, Brioschi S, Colonna M (2022a) TREM2 drives microglia response to amyloid-β via SYK-dependent and -independent pathways. Cell 185 (22):4153-4169.e4119. https://doi.org/10.1016/j.cell.2022.09.033
Wang Y, Hao L, Wang T, Liu W, Wang L, Ju M, Feng W, Xiao R (2022b) 27-Hydroxycholesterol-induced dysregulation of cholesterol metabolism impairs learning and memory ability in ApoE ε4 transgenic mice. Int J Mol Sci. https://doi.org/10.3390/ijms231911639
Weisgraber KH, Rall SC, Jr., Mahley RW (1981) Human E apoprotein heterogeneity. Cysteine-arginine interchanges in the amino acid sequence of the apo-E isoforms. J Biol Chem 256 (17):9077–9083
Wennberg AM, Tosakulwong N, Lesnick TG, Murray ME, Whitwell JL, Liesinger AM, Petrucelli L, Boeve BF, Parisi JE, Knopman DS, Petersen RC, Dickson DW, Josephs KA (2018) Association of Apolipoprotein E ε4 with transactive response DNA-binding protein 43. JAMA Neurol 75 (11):1347–1354. https://doi.org/10.1001/jamaneurol.2018.3139
Wu L, Zhang X, Zhao L (2018) Human ApoE isoforms differentially modulate brain glucose and ketone body metabolism: implications for Alzheimer’s disease risk reduction and early intervention. J Neurosci 38 (30):6665–6681. https://doi.org/10.1523/jneurosci.2262-17.2018
Xie M, Liu YU, Zhao S, Zhang L, Bosco DB, Pang YP, Zhong J, Sheth U, Martens YA, Zhao N, Liu CC, Zhuang Y, Wang L, Dickson DW, Mattson MP, Bu G, Wu LJ (2022) TREM2 interacts with TDP-43 and mediates microglial neuroprotection against TDP-43-related neurodegeneration. Nat Neurosci 25 (1):26–38. https://doi.org/10.1038/s41593-021-00975-6
Xiong M, Jiang H, Serrano JR, Gonzales ER, Wang C, Gratuze M, Hoyle R, Bien-Ly N, Silverman AP, Sullivan PM, Watts RJ, Ulrich JD, Zipfel GJ, Holtzman DM (2021a) APOE immunotherapy reduces cerebral amyloid angiopathy and amyloid plaques while improving cerebrovascular function. Sci Transl Med. https://doi.org/10.1126/scitranslmed.abd7522
Xiong N, Schiller MR, Li J, Chen X, Lin Z (2021b) Severe COVID-19 in Alzheimer’s disease: APOE4’s fault again? Alzheimer’s Res Therapy 13 (1):111. https://doi.org/10.1186/s13195-021-00858-9
Xiong M, Wang C, Gratuze M, Saadi F, Bao X, Bosch ME, Lee C, Jiang H, Serrano JR, Gonzales ER, Kipnis M, Holtzman DM (2023) Astrocytic APOE4 removal confers cerebrovascular protection despite increased cerebral amyloid angiopathy. Mol Neurodegener 18 (1):17. https://doi.org/10.1186/s13024-023-00610-x
Xu D, Peng Y (2017) Apolipoprotein E 4 triggers multiple pathway-mediated Ca2+ overload, causes CaMK II phosphorylation abnormity and aggravates oxidative stress caused cerebral cortical neuron damage. Eur Rev Med Pharmacol Sci 21 (24):5717–5728. https://doi.org/10.26355/eurrev_201712_14018
Xu Q, Bernardo A, Walker D, Kanegawa T, Mahley RW, Huang Y (2006) Profile and regulation of Apolipoprotein E (ApoE) expression in the CNS in mice with targeting of green fluorescent protein gene to the ApoE locus. J Neurosci 26 (19):4985–4994. https://doi.org/10.1523/jneurosci.5476-05.2006
Yamazaki Y, Liu CC, Yamazaki A, Shue F, Martens YA, Chen Y, Qiao W, Kurti A, Oue H, Ren Y, Li Y, Aikawa T, Cherukuri Y, Fryer JD, Asmann YW, Kim BYS, Kanekiyo T, Bu G (2021) Vascular ApoE4 impairs behavior by modulating gliovascular function. Neuron 109 (3):438-447.e436. https://doi.org/10.1016/j.neuron.2020.11.019
Yang HS, Yu L, White CC, Chibnik LB, Chhatwal JP, Sperling RA, Bennett DA, Schneider JA, De Jager PL (2018) Evaluation of TDP-43 proteinopathy and hippocampal sclerosis in relation to APOE ε4 haplotype status: a community-based cohort study. Lancet Neurol 17 (9):773–781. https://doi.org/10.1016/s1474-4422(18)30251-5
Yang M, Weng Q, Pan X, Hussain HMJ, Yu S, Xu J, Yu X, Liu Y, Jin Y, Zhang C, Li X, Ren H, Chen N, Xie J (2020) Clinical and genetic analysis of lipoprotein glomerulopathy patients caused by APOE mutations. Mol Genet Genom Med 8 (8):e1281. https://doi.org/10.1002/mgg3.1281
Yeh FL, Wang Y, Tom I, Gonzalez LC, Sheng M (2016) TREM2 binds to apolipoproteins, including APOE and CLU/APOJ, and thereby facilitates uptake of amyloid-beta by microglia. Neuron 91 (2):328–340. https://doi.org/10.1016/j.neuron.2016.06.015
Yin J, Reiman EM, Beach TG, Serrano GE, Sabbagh MN, Nielsen M, Caselli RJ, Shi J (2020) Effect of ApoE isoforms on mitochondria in Alzheimer disease. Neurology 94 (23):e2404–e2411. https://doi.org/10.1212/wnl.0000000000009582
Zannis VI, Just PW, Breslow JL (1981) Human Apolipoprotein E isoprotein subclasses are genetically determined. Am J Hum Genet 33 (1):11–24
Zhao N, Liu CC, Qiao W, Bu G (2018) Apolipoprotein E, receptors, and modulation of Alzheimer’s disease. Biol Psychiatry 83 (4):347–357. https://doi.org/10.1016/j.biopsych.2017.03.003
Zhao N, Attrebi ON, Ren Y, Qiao W, Sonustun B, Martens YA, Meneses AD, Li F, Shue F, Zheng J, Van Ingelgom AJ, Davis MD, Kurti A, Knight JA, Linares C, Chen Y, Delenclos M, Liu CC, Fryer JD, Asmann YW, McLean PJ, Dickson DW, Ross OA, Bu G (2020) APOE4 exacerbates α-synuclein pathology and related toxicity independent of amyloid. Sci Transl Med. https://doi.org/10.1126/scitranslmed.aay1809
Zhao Y, Jia X, Yang X, Bai X, Lu Y, Zhu L, Cheng W, Shu M, Zhu Y, Du X, Wang L, Shu Y, Song Y, Jin S (2022) Deacetylation of Caveolin-1 by Sirt6 induces autophagy and retards high glucose-stimulated LDL transcytosis and atherosclerosis formation. Metabolism 131:155162. https://doi.org/10.1016/j.metabol.2022.155162
Zhou J, Wang Y, Huang G, Yang M, Zhu Y, Jin C, Jing D, Ji K, Shi Y (2023) LilrB3 is a putative cell surface receptor of APOE4. Cell Res 33 (2):116–130. https://doi.org/10.1038/s41422-022-00759-y
Zollo A, Allen Z, Rasmussen HF, Iannuzzi F, Shi Y, Larsen A, Maier TJ, Matrone C (2017) Sortilin-related receptor expression in human neural stem cells derived from Alzheimer’s disease patients carrying the APOE Epsilon 4 Allele. Neural Plast 2017:1892612. https://doi.org/10.1155/2017/1892612
Funding
Funding was provide by Academic Research Projects of Beijing Union University (Grant Numbers JZ10202001, XP202008 and ZK70202101).
Author information
Authors and Affiliations
Contributions
HCH designed the main theme of the manuscript and the guidelines of the sections and the paragraphs. YYS finished the writing of the first draft and revised the manuscript thoroughly; ZW participated in literature collection and revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that there is no potential conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, YY., Wang, Z. & Huang, HC. Roles of ApoE4 on the Pathogenesis in Alzheimer’s Disease and the Potential Therapeutic Approaches. Cell Mol Neurobiol 43, 3115–3136 (2023). https://doi.org/10.1007/s10571-023-01365-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-023-01365-1