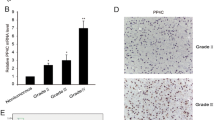
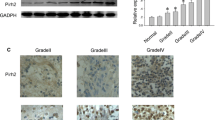
Abstract
Glioma is the most common type of primary central nervous system tumor. Ser/Thr protein phosphatase 5 (PP5) has been shown to regulate multiple signaling cascades that suppress growth and facilitate apoptosis in several human cancer cells. However, the role of PP5 in human gliomas remains unclear. Herein, the relationship between PP5 expression and glioma cell growth was investigated, and the therapeutic value of PP5 in glioma was further evaluated. We employed a short hairpin RNA targeting PPP5C gene to knock down PP5 expression in human glioma cell lines U251 and U373. Depletion of PPP5C via RNAi remarkably inhibited glioma cell proliferation and colony formation, and arrested cell cycle in the G0/G1 phase. Moreover, knockdown of PP5 markedly suppressed glioma cell migration, as determined by Transwell assay. Our findings suggest that PPP5C could be essential for glioma cell growth and serve as a promising therapeutic target in human gliomas.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The reversible protein phosphorylation (phosphorylation and dephosphorylation) plays an important role in most aspects of cell life, including cell growth regulation, differentiation, senescence, and apoptosis (Cohen 2001). Protein phosphatase catalyzes dephosphorylation in vivo, and there are three major types of protein phosphatases in humans, tyrosine phosphatase (PTPase) (Zhang 2002), serine/threonine phosphatase (PPase) (Mumby and Walter 1993) and dual specificity phosphatase (DSPase) (Camps et al. 2000). Serine/threonine protein phosphatase-5 (PP5, PPP5C) is one member of the protein serine/threonine phosphatase gene family which is broadly expressed in mammalian tissues. It is confirmed that PP5 comprises a regulatory tetratricopeptide repeat (TPR) domain and functions in signaling pathways in many cellular responses. Currently, the biological function of PP5 is not clear, and its elevated expression level is associated with the cancer development in breast, liver, and possibly other forms (Golden et al. 2004, 2008a, b; Ghobrial et al. 2005; Shirato et al. 2000; Fukuda et al. 2007). High levels of PP5 have been observed in human cancers, and constitutive PP5 overexpression aids tumor progression in mouse models of tumor development (Golden et al. 2004, 2008a, b). PP5 is found to be encountered in several proteins that regulate intra-cellular signaling cascades by hormones (i.e., glucocorticoids) or cellular stress. In glucocorticoids signaling, PP5 is associated with heat shock protein-90 (Hsp-90) which is glucocorticoids receptor (GR) (Chen et al. 1996; Silverstein et al. 1997; Davies et al. 2005), and can induce phosphorylation of p53 tumor suppressor protein (Zuo et al. 1998, 1999; Urban et al. 2003). In response to genomic cellular stress, especially when PP5 is down-regulated by siRNA or antisense oligonucleotides, several stress-responsive proteins such as ASK1, PKcs, p53, Cdc37, Raf1, and GRs show rich phosphorylation (Vaughan et al. 2008; Mkaddem et al. 2009; Huang et al. 2004; Wechsler et al. 2004; Morita et al. 2001; Kutuzov et al. 2005). Phosphorylation of these responsive proteins can trigger apoptosis but apparently PP5 negatively regulates the processes (Morita et al. 2001; Kutuzov et al. 2005; Mkaddem et al. 2009; Huang et al. 2004). Taken together, previous studies have indicated that PP5 is a potentially important regulator of cellular signaling networks.
Glioma is the most common primary malignancy in the human central nervous system (CNS). Approximately, 13,000 deaths and 18,000 new cases of primary malignant brain and CNS tumors occur annually in the US (Ostrom et al. 2013). Four grades of gliomas are classified by histological progression: low-grade astrocytomas (Grade I–II), anaplastic astrocytomas (Grade III), and glioblastoma (GBM, Grade IV) (Kleihues et al. 1993, 2002). The five-year survival in low-grade gliomas is around 30–70 % depending on histology (Louis et al. 2007), but the patients with the most aggressive type of glioma, GBM, have the worst prognosis with a median survival of 9–12 months even after surgical resection, radiation therapy, and chemotherapy (Stupp et al. 2005). As a result of that, early diagnostics with treatment can provide comprehensive and preventive health care on glioma patients. Scientists are in aggressive needs to figure out new therapeutic target against human gliomas.
RNA interference (RNAi) technique is a powerful tool to carry out loss-of-function assays. It provides a new approach to investigate cancer gene therapy (Kim et al. 2005; Guo et al. 2011). In this study, we employed a lentiviral vector-mediated RNAi system to achieve highly stable silencing of PP5. In this study, we are aiming to evaluate the biological function of PP5 protein (gene: PPP5C) and try to reveal its contribution to glioma progression. In order to verify that PPP5C could be an effective therapeutic target for glioma, we suppressed PP5 expression via RNAi in human glioma cell lines U251 and U373. To our knowledge, this is the first time that we provided evidence to demonstrate knockdown of endogenous PP5 expression could suppress the oncogenic properties of glioma cells and their invasion capacity in vitro. It is suggesting that PPP5C could be a promising therapeutic target for human gliomas.
Materials and Methods
Cell Culture
Human embryonic kidney cell line 293T (HEK293T) and human glioblastoma cell lines U251, U87MG, U373, A172, and U-118MG were obtained from Shanghai Institute of Cell Biology, the Chinese Academy of Sciences. U87MG cells were cultured in EMEM (Hyclone, Logan, UT, USA) supplemented with 10 % fetal bovine serum (FBS), 1 mM sodium pyruvate and 1 % non-essential amino acid at 37 °C in a humidified atmosphere of 5 % CO2. U251, U373, A172, U-118MG, and HEK293T cells were maintained in DMEM (Hyclone, Logan, UT, USA) supplemented with 10 % FBS at 37 °C in a humidified atmosphere of 5 % CO2.
Construction of Recombinant Lentivirus
The following oligonucleotides were synthesized. Two of short hairpin RNA (shRNA) sequences (5′-GAGAC AGAGA AGATT ACAGT ACTCG AGTAC TGTAA TCTTC TCTGT CTCTT TTT-3′, sequence 1) and (5′-CCACG AGACA GACAA CATGA ACTCG AGTTC ATGTT GTCTG TCTCG TGGTT TTT-3′, sequence 2) for human PPP5C gene (NM_001204284.1) were screened and validated to be candidate shRNAs. And the negative control shRNA was 5′-GCGGA GGGTT TGAAA GAATA TCTCG AGATA TTCTT TCAAA CCCTC CGCTT TTTT-3′. The stem-loop-stem oligos (shRNAs) were synthesized, annealed, and ligated into the AgeI/EcoRI-linearized pFH-L vector (Shanghai Hollybio, China). The lentiviral-based shRNA-expressing vectors were confirmed by DNA sequencing. The generated plasmids were named as pFH-L-shPPP5C-S1, pFH-L-shPPP5C-S2, and pFH-L-shCon.
HEK293T cells (1.0 × 106) were seeded into 10-cm dishes and cultured for 24 h to reach 70–80 % confluence. Two hours before transfection, the medium was replaced with serum-free DMEM. The plasmids including 10 µg of pFH-L-shPPP5C-S1/S2 or pFH-L-shCon, 7.5 µg of packaging vector pHelper-1.0, and 5 µg of expression plasmid pHelper-2.0 were added to 0.95 ml Opti-MEM and 50 µl of Lipofectamine 2000. The mixture was added to the cells and incubated for 8 h before replacing the medium with 10 ml of complete DMEM medium (supplemented with 10 % PBS). Lentiviral particles were harvested at 48 h after transfection. As the lentivirus carries green fluorescence protein (GFP), the viral titer was determined by end-point dilution assay through counting GFP-expressing cells under fluorescence microscope 96 h after infection.
RNA Extraction and Real-Time PCR
Human glioblastoma cells (U251 and U373) were pre-cultured and infected with recombinant lentivirus for 5 days. Total RNA was prepared using Trizol reagent (Gibco RL, Grand Island, NY, USA) according to the manufacturer’s instruction. 5 mg of total RNA was used to synthesize the first strand of cDNA using SuperScript II RT 200 U/ml (Invitrogen, Carlsbad, CA, USA). PPP5C mRNA expression was evaluated by real-time PCR on BioRad Connet Real-Time PCR platform (BioRad, Hercules, CA, USA) with SYBR Green PCR core reagents. The qPCR reaction system contains 2× SYBR premix ex taq 10 µl, plus forward and reverse primers (2.5 µM) 0.8 µl, cDNA template 5 µl, and ddH2O 4.2 µl. β-Actin was applied as the internal reference. The following primers were synthesized and applied: PPP5C: 5′-CCCAACTACTGCGACCAGAT-3′ as forward and 5′-CCCGTCACCTCACATCATTC-3′ as reverse; β-actin: 5′-GTGGACATCCGCAAAGAC-3′ as forward and 5′-AAAGGGTGTAACGCAACTA-3′ as reverse. The reaction procedure was initiated with denaturation at 95 °C for 1 min and followed by 40 repeated cycles (denaturation at 95 °C for 5 s and annealing extension at 60 °C for 20 s). Results were presented as CT values, defined as the threshold PCR cycle number at which an amplified product was first detected. The average CT was calculated for both PPP5C and β-actin, and ΔCT was determined as the mean of the triplicate CT values for PPP5C minus the mean of the triplicate CT values for β-actin.
Western Blot Analysis
U251 and U373 cells were cultured and infected with recombinant lentivirus for 5 days. Cells were washed twice with ice-cold PBS and lysed in 2× SDS sample buffer (100 mM Tris–HCl (pH 6.8), 10 mM EDTA, 4 % SDS, 10 % Glycine). Equal amounts of proteins (30 µg) were loaded and separated on 10 % SDS-PAGE gels and transferred to polyvinylidene difluoride (PVDF) membranes (Millipore, Bedford, MA, USA) at 300 mA for 1.5 h. Membranes were blocked and then probed with primary antibodies, mouse anti-PPP5C (1:3,000 dilution; Abcam, Cambridge, UK) or rabbit anti-GAPDH (1:60,000 dilution; Proteintech Group Inc., Chicago, IL, USA) overnight at 4 °C. After washing, the blots were incubated with horseradish peroxidase-conjugated secondary antibodies (Santa Cruz, Dallas, Texas, USA) for 2 h at room temperature and then visualized by super ECL detection reagent (Applygen, Beijing, China).
MTT Viability Assay
Both U251 and U373 cells were cultured in 6-well plates and inoculated with recombinant lentiviruses. After 72 h of infection, cells were washed and re-cultured in 96-well plates with 2.0 × 103 cells per well (U251) or 2.5 × 103 cells per well (U373). MTT solution was added to each well and incubated at 37 °C for 4 h at different time points after lentivirus infection (1, 2, 3, 4, 5 days). Then the converted dye solubilized in acidic isopropanol (10 % SDS, 5 % isopropanol and 0.01 M HCl) was used and incubated at 37 °C for 10 min. The optical density was measured using microplate reader at the wavelength of 595 nm. The experiment was repeated at least three times.
Colony Formation Assay
Human glioblastoma cells U251 and U373 were cultured in 6-well plates and treated with recombinant lentiviruses. After 96 h of incubation, infected cells were washed, re-cultured in the prepared 6-well plates at a density of 800 cells per well (U251) or 500 cells per well (U373) and allowed to form natural colonies. After 13 days of incubation (U251) or 9 days of incubation (U373), treated glioblastoma cells were subjected to crystals violet staining. Subsequently, cells were washed and fixed by paraformaldehyde. Fixed cells were washed twice with PBS solution, treated with crystals violet for 10 min, washed 3 times by ddH2O, and then photographed with a digital camera. The number of colonies (>50 cells/colony) was counted.
Flow Cytometry Analysis
U251 cells were cultured in 6-well plates and inoculated with recombinant lentiviruses at a MOI of 10. After 72 h of infection, cells were inoculated into 6-cm dishes at a density of 2.0 × 105 cells per dish. After 40 h incubation period, cells in each well were harvested and cell cycle was determined by propidium iodide (PI) staining method before cell density reached 80 % confluency. Tests were performed in triplicate for each sample, and analyses were performed by FAC Scan flow cytometer (Becton–Dickinson, San Jose, CA, USA) in accordance with the manufacturer’s guidelines.
Cell Migration Assay
U251 and U373 cells infected with Lv-shPPP5C or Lv-shCon for 96 h and uninfected cells were harvested, and their ability to migrate in vitro was determined using a Transwell chamber (Corning, NY, USA). Cells were seeded into the upper chamber (3.0 × 104 cells/well of U251, 5.0 × 104 cells/well of U373) in 100 µl serum-free medium. Medium (1 ml) containing 20 % FBS was added to the lower chamber as a chemo-attractant. After incubation for 24 h at 37 °C in 5 % CO2, the surface of the upper chamber was swabbed with cotton-tipped applicators to remove the cells that did not migrate, including dead cells. The viable cells that moved to the lower surface of the filter were fixed in 4 % paraformaldehyde and then stained with crystal violet. The migrated cells were counted in five random fields (100×) per filter under a light microscope, and the amount of dissolved crystal violet was detected by the spectrometric absorbance at 570 nm.
Statistical Analysis
All statistical analyses were performed using SPSS13.0 software. The differences between groups were compared using Student’s t test, and data were expressed as mean ± SD of three independent experiments. Statistical significant difference was accepted at p < 0.05.
Results
Efficacy of Lentivirus-Mediated RNAi Targeting of PPP5C
PP5 protein (gene: PPP5C) is broadly expressed in various human glioma cell lines. Before we could move forward with RNAi study, we tried to screen and select the most appropriate cell line with highest intra-cellular PP5 expression. qPCR assay was carried out to better study the relative expression levels of PPP5C in glioma cell lines. As we could see from Fig. 1, we examined five glioma cell lines including U251, U87MG, U373, A172, and U-118MNG, whereas the highest expression level of PPP5C was observed in U251 cells, followed by U373 cells. Therefore, we employed U251 cells along with U373 cells for future investigation of the role of PPP5C in glioma progression.
To determine the silencing effect of lentivirus-mediated PPP5C RNAi on PP5 expression in U251 and U373 cells, we constructed both control lentivirus (Lv-shCon) and specific PPP5C-targeting lentivirus (Lv-shPPP5C-S1). We further constructed one more lentivirus (Lv-shPPP5C-S2) targeting PPP5C to avoid off-target effect. Both U251 and U373 cells were cultured and infected with Lv-shCon and Lv-shPPP5C, respectively. Non-infected parent cells were deemed as negative control (Con). To demonstrate the infection efficiency, we involved GFP tag which was embedded in lentivirus to provide visualized confirmation. As demonstrated in Fig. 2a, b, over 90 % of U251 and U373 cells were GFP positive after lentiviral infection, indicating that the transfection rate was satisfying. To further investigate the knockdown efficiency of PPP5C, we carried out real-time PCR and western blotting to study the expression levels of PPP5C in glioma cancer cells after infection. The non-silencing lentivirus encoded with irrelevant sequence had negligible effect on PPP5C expression, but PPP5C-silencing lentivirus (Lv-shPPP5C-S1) could remarkably down-regulate both mRNA and protein levels of PPP5C, by 67.9 and 78.0 % reduction in U251 and U373 cells, respectively (Fig. 2c–f). The other specific lentivirus, Lv-shPPP5C-S2, also had good performance in down-regulating the expression level of PPP5C by 81.8 % (Fig. S1a, b). Taken together, the lentiviruses we constructed ensured us the most efficient tool to knock down PPP5C expression in glioma cells.
Knockdown efficacy of PPP5C by lentivirus infection in glioma cells. a, b Microscopic images of U251 and U373 cells infected with lentivirus at MOI of 10, respectively (scale bar 100 µm). Visible GFP proteins proved that over 90 % of cells were successfully infected. qPCR analysis of PPP5C knockdown efficiency in U251 (c) and U373 cells (e). The transcription of PPP5C mRNA was significantly suppressed when infected with Lv-shPPP5C-S1. Western blot analysis of PPP5C knockdown efficiency in U251 (d) and U373 cells (f). ***p < 0.001
Deprivation of PP5 Remarkably Inhibited the Proliferation Rate of Glioma Cells
To better understand the role of PPP5C in glioma cancer tumorigenesis, we examined the variation tendency of cell proliferation after lentivirus infection. We involved MTT assay due to its wonder testing sensitivity and dynamic range to examine the cell proliferation rate after 3-day incubation. Line chart in Fig. 3 indicated that non-silencing cells had no obvious difference against control cells (Lv-shCon vs. Con), but meanwhile significant proliferation alteration was observed in the PPP5C-silenced cells (Lv-shPPP5C-S1 vs. Lv-shCon, p < 0.001). The similar result was observed in U251 cells after Lv-shPPP5C-S2 infection (Fig. S1c).
We subsequently cross evaluated the colony formation capacity in both U251 and U373 cells. As shown in Fig. 4, down-regulation of PP5 could lead to a significant reduction in colony formation capacity. The colony was remarkably smaller, and colony numbers were statistically fewer than control cells (p < 0.001), whereas there was no noticeable difference between non-silencing cells and control cells. The conformity of cell proliferation rate and colony formation capacity was surprising good when we treated U251 cells with Lv-shPPP5C-S1 or Lv-shPPP5C-S2. When U251 cells were infected with Lv-shPPP5C-S2, the cell proliferation rate was stalled and also the colony formation ability was mitigated (Fig. S1d, e). Collectively, knockdown of PPP5C by RNAi could markedly suppress the proliferation and colony formation ability of glioma cells.
Down-regulated expression of PP5 suppresses the colony formation capacity in glioma cells. Microscopic images of colonies formed in U251 (a) and U373 cells (b). Single colony was stained by crystal violet (scale bar 250 µm). The full vision of six-well plate under microscope showed significant colony formation inhibition in Lv-shPPP5C-S1-treated culture plate. Statistical analysis of colonies numbers utilizing crystal violet staining in U251 (c) and U373 cells (d). ***p < 0.001
Cell Cycle Arrest was Induced by Down-Regulation of PP5
To explore the potential mechanism of cell growth inhibition, we determined the cell cycle regulation alteration when U251 glioma cells were infected with Lv-shPPP5C. As we could see from Fig. 5a, cell distribution in cell cycles (G0/G1 phase, S phase, and G2/M phase) was significantly different in three groups (Con, Lv-shCon, and Lv-shPPP5C-S1). By contrast to control group, cells infected with Lv-shPPP5C-S1 were mostly distributed in the G0/G1 phase (61.4 %) and less distributed in the S phase (30.9 %) and G2/M phase (7.7 %) (Fig. 5b). Our results demonstrated that treatment with Lv-shPPP5C could remarkably induce G0/G1 cell cycle arrest (p < 0.001), but the cells infected with non-silencing lentivirus did not show any significant difference against non-infected cells. These findings are in agreement with cell growth inhibition, which suggest that PPP5C could modulate glioma cell growth via cell cycle control.
Impact of Down-Regulation of PP5 Expression on Cell Migration
To determine the role of PPP5C in glioma cell migration, we employed transwell chamber assay after 72 h lentiviral infection. As we could see from Fig. 6, the migrative ability of Lv-shCon-infected cells did not significantly differ from non-infected cells. But meanwhile, the migration ability of glioma cells infected with Lv-shPPP5C-S1 was remarkably lower than those of non-infected and Lv-shCon-infected cells (p < 0.001 in U251 cells and p < 0.01 in U373 cells, respectively, Lv-shCon vs. Lv-shPPP5C-S1). Therefore, we proved that down-regulation of PPP5C could mitigate the migration of glioma cells.
Knockdown of PP5 mitigated glioma cell migration by lentivirus infection. Microscopic images of migrated U251 (a) and U373 cells (b) (scale bar 100 µm). Cell counting of migrated U251 (c) and U373 cells (e). Quantitative analysis of migrated U251 (d) and U373 cells (f) by destaining and reading optical density at 570 nm. *p < 0.05, **p < 0.01, ***p < 0.001
Discussion
Glioma is the most common malignant tumor over the world. It has a worse prognosis especially when it turns to Grade IV due to its aggression and metastasis. Therefore, developing novel therapeutic method for gliomas has attracted great attention. In the current study, we firstly found that PPP5C is widely expressed in multiple human glioma cell lines. Thus two shRNA vectors for PPP5C were constructed to suppress PP5 expression in glioma cell lines U251 and U373 so as to determine its role in glioma malignancy progress.
Our results indicated that transfection with PPP5C-targeting shRNA significantly suppressed cell proliferation and colony formation capacity. Moreover, down-regulation of PPP5C in U251 cells significantly increased the cell percentage in G0/G1 phase, but concomitantly decreased the cell percentage in S phase. Therefore, knockdown of PPP5C inhibited glioma cell proliferation possibly via G0/G1 phase cell cycle arrest. Alternatively, when PP5 is suppressed, the cells could die in S phase and/or G2 M phase. However, the molecular mechanism of whether PP5 regulates cell cycle progression and apoptosis in glioma is poorly understood. Previous studies with siRNA and antisense oligonucleotides demonstrate that a decrease of PP5 expression can also result in G1-growth arrest, GR/p53-dependent increase in p21 expression (Zuo et al. 1998, 1999), altered DNA-PKcs signaling (Wechsler et al. 2004), DNA-damage checkpoint failure (Zhang et al. 2005) and prolonged activation of ASK1 signaling (Zhou et al. 2004; Morita et al. 2001). In particular, PP5 has been shown to act as a suppressor of ASK1 (Matsuzawa et al. 2002; Ferguson et al. 2003; Morita et al. 2001; Zhou et al. 2004; Huang et al. 2003, 2004), p53 (Zuo et al. 1998; Urban et al. 2003), and DNA-PKcs (Wechsler et al. 2004). Further investigation should be carried out to elucidate the regulation mechanisms of PP5 in cell cycle control and apoptosis.
Furthermore, the effect of PP5 on glioma cell invasion was investigated in this study. We proved that down-regulation of PPP5C remarkably mitigated cell migration in both U251 and U373 cell lines, which suggested that PP5 could also play an essential role in glioma metastasis.
To the best of our knowledge, this is the first study to demonstrate the correlation between PPP5C and glioma progression. Our data provided evidence that reduced PP5 expression was correlated with reduced proliferation and impeded migration of glioma cells, underlying a connection between down-regulation of PP5 and stalled glioma progression. This study extends our knowledge of the basic biological mechanism of PPP5C in glioma in vitro. As a result, we speculate that PPP5C could be an potential therapeutic target against human gliomas.
References
Camps M, Nichols A, Arkinstall S (2000) Dual specificity phosphatases: a gene family for control of MAP kinase function. FASEB J 14(1):6–16
Chen MS, Silverstein AM, Pratt WB, Chinkers M (1996) The tetratricopeptide repeat domain of protein phosphatase 5 mediates binding to glucocorticoid receptor heterocomplexes and acts as a dominant negative mutant. J Biol Chem 271(50):32315–32320
Cohen P (2001) The role of protein phosphorylation in human health and disease. The Sir Hans Krebs Medal Lecture. Eur J Biochem 268(19):5001–5010
Davies TH, Ning YM, Sanchez ER (2005) Differential control of glucocorticoid receptor hormone-binding function by tetratricopeptide repeat (TPR) proteins and the immunosuppressive ligand FK506. Biochemistry 44(6):2030–2038. doi:10.1021/bi048503v
Ferguson HA, Marietta PM, Van Den Berg CL (2003) UV-induced apoptosis is mediated independent of caspase-9 in MCF-7 cells: a model for cytochrome c resistance. J Biol Chem 278(46):45793–45800. doi:10.1074/jbc.M307979200
Fukuda H, Tsuchiya N, Hara-Fujita K, Takagi S, Nagao M, Nakagama H (2007) Induction of abnormal nuclear shapes in two distinct modes by overexpression of serine/threonine protein phosphatase 5 in Hela cells. J Cell Biochem 101(2):321–330. doi:10.1002/jcb.21178
Ghobrial IM, McCormick DJ, Kaufmann SH, Leontovich AA, Loegering DA, Dai NT, Krajnik KL, Stenson MJ, Melhem MF, Novak AJ, Ansell SM, Witzig TE (2005) Proteomic analysis of mantle-cell lymphoma by protein microarray. Blood 105(9):3722–3730. doi:10.1182/blood-2004-10-3999
Golden T, Aragon IV, Zhou G, Cooper SR, Dean NM, Honkanen RE (2004) Constitutive over expression of serine/threonine protein phosphatase 5 (PP5) augments estrogen-dependent tumor growth in mice. Cancer Lett 215(1):95–100. doi:10.1016/j.canlet.2004.03.027
Golden T, Aragon IV, Rutland B, Tucker JA, Shevde LA, Samant RS, Zhou G, Amable L, Skarra D, Honkanen RE (2008a) Elevated levels of Ser/Thr protein phosphatase 5 (PP5) in human breast cancer. Biochim Biophys Acta 1782(4):259–270. doi:10.1016/j.bbadis.2008.01.004
Golden T, Swingle M, Honkanen RE (2008b) The role of serine/threonine protein phosphatase type 5 (PP5) in the regulation of stress-induced signaling networks and cancer. Cancer Metastasis Rev 27(2):169–178. doi:10.1007/s10555-008-9125-z
Guo W, Zhang Y, Chen T, Wang Y, Xue J, Zhang Y, Xiao W, Mo X, Lu Y (2011) Efficacy of RNAi targeting of pyruvate kinase M2 combined with cisplatin in a lung cancer model. J Cancer Res Clin Oncol 137(1):65–72. doi:10.1007/s00432-010-0860-5
Huang S, Shu L, Dilling MB, Easton J, Harwood FC, Ichijo H, Houghton PJ (2003) Sustained activation of the JNK cascade and rapamycin-induced apoptosis are suppressed by p53/p21(Cip1). Mol Cell 11(6):1491–1501
Huang S, Shu L, Easton J, Harwood FC, Germain GS, Ichijo H, Houghton PJ (2004) Inhibition of mammalian target of rapamycin activates apoptosis signal-regulating kinase 1 signaling by suppressing protein phosphatase 5 activity. J Biol Chem 279(35):36490–36496. doi:10.1074/jbc.M401208200
Kim DH, Behlke MA, Rose SD, Chang MS, Choi S, Rossi JJ (2005) Synthetic dsRNA Dicer substrates enhance RNAi potency and efficacy. Nat Biotechnol 23(2):222–226. doi:10.1038/nbt1051
Kleihues P, Burger PC, Scheithauer BW (1993) The new WHO classification of brain tumours. Brain Pathol 3(3):255–268
Kleihues P, Louis DN, Scheithauer BW, Rorke LB, Reifenberger G, Burger PC, Cavenee WK (2002) The WHO classification of tumors of the nervous system. J Neuropathol Exp Neurol 61(3):215–225 discussion 226–219
Kutuzov MA, Andreeva AV, Voyno-Yasenetskaya TA (2005) Regulation of apoptosis signal-regulating kinase 1 (ASK1) by polyamine levels via protein phosphatase 5. J Biol Chem 280(27):25388–25395. doi:10.1074/jbc.M413202200
Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, Scheithauer BW, Kleihues P (2007) The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol 114(2):97–109. doi:10.1007/s00401-007-0243-4
Matsuzawa A, Nishitoh H, Tobiume K, Takeda K, Ichijo H (2002) Physiological roles of ASK1-mediated signal transduction in oxidative stress- and endoplasmic reticulum stress-induced apoptosis: advanced findings from ASK1 knockout mice. Antioxid Redox Signal 4(3):415–425. doi:10.1089/15230860260196218
Mkaddem SB, Werts C, Goujon JM, Bens M, Pedruzzi E, Ogier-Denis E, Vandewalle A (2009) Heat shock protein gp96 interacts with protein phosphatase 5 and controls toll-like receptor 2 (TLR2)-mediated activation of extracellular signal-regulated kinase (ERK) 1/2 in post-hypoxic kidney cells. J Biol Chem 284(18):12541–12549. doi:10.1074/jbc.M808376200
Morita K, Saitoh M, Tobiume K, Matsuura H, Enomoto S, Nishitoh H, Ichijo H (2001) Negative feedback regulation of ASK1 by protein phosphatase 5 (PP5) in response to oxidative stress. EMBO J 20(21):6028–6036. doi:10.1093/emboj/20.21.6028
Mumby MC, Walter G (1993) Protein serine/threonine phosphatases: structure, regulation, and functions in cell growth. Physiol Rev 73(4):673–699
Ostrom QT, Gittleman H, Farah P, Ondracek A, Chen Y, Wolinsky Y, Stroup NE, Kruchko C, Barnholtz-Sloan JS (2013) CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2006–2010. Neuro Oncol 15(Suppl 2):ii1–ii56. doi:10.1093/neuonc/not151
Shirato H, Shima H, Nakagama H, Fukuda H, Watanabe Y, Ogawa K, Matsuda Y, Kikuchi K (2000) Expression in hepatomas and chromosomal localization of rat protein phosphatase 5 gene. Int J Oncol 17(5):909–912
Silverstein AM, Galigniana MD, Chen MS, Owens-Grillo JK, Chinkers M, Pratt WB (1997) Protein phosphatase 5 is a major component of glucocorticoid receptor.hsp90 complexes with properties of an FK506-binding immunophilin. J Biol Chem 272(26):16224–16230
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO, European Organisation for R, Treatment of Cancer Brain T, Radiotherapy G, National Cancer Institute of Canada Clinical Trials G (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352(10):987–996. doi:10.1056/NEJMoa043330
Urban G, Golden T, Aragon IV, Cowsert L, Cooper SR, Dean NM, Honkanen RE (2003) Identification of a functional link for the p53 tumor suppressor protein in dexamethasone-induced growth suppression. J Biol Chem 278(11):9747–9753. doi:10.1074/jbc.M210993200
Vaughan CK, Mollapour M, Smith JR, Truman A, Hu B, Good VM, Panaretou B, Neckers L, Clarke PA, Workman P, Piper PW, Prodromou C, Pearl LH (2008) Hsp90-dependent activation of protein kinases is regulated by chaperone-targeted dephosphorylation of Cdc37. Mol Cell 31(6):886–895. doi:10.1016/j.molcel.2008.07.021
Wechsler T, Chen BP, Harper R, Morotomi-Yano K, Huang BC, Meek K, Cleaver JE, Chen DJ, Wabl M (2004) DNA-PKcs function regulated specifically by protein phosphatase 5. Proc Natl Acad Sci USA 101(5):1247–1252. doi:10.1073/pnas.0307765100
Zhang ZY (2002) Protein tyrosine phosphatases: structure and function, substrate specificity, and inhibitor development. Annu Rev Pharmacol Toxicol 42:209–234. doi:10.1146/annurev.pharmtox.42.083001.144616
Zhang J, Bao S, Furumai R, Kucera KS, Ali A, Dean NM, Wang XF (2005) Protein phosphatase 5 is required for ATR-mediated checkpoint activation. Mol Cell Biol 25(22):9910–9919. doi:10.1128/MCB.25.22.9910-9919.2005
Zhou G, Golden T, Aragon IV, Honkanen RE (2004) Ser/Thr protein phosphatase 5 inactivates hypoxia-induced activation of an apoptosis signal-regulating kinase 1/MKK-4/JNK signaling cascade. J Biol Chem 279(45):46595–46605. doi:10.1074/jbc.M408320200
Zuo Z, Dean NM, Honkanen RE (1998) Serine/threonine protein phosphatase type 5 acts upstream of p53 to regulate the induction of p21(WAF1/Cip1) and mediate growth arrest. J Biol Chem 273(20):12250–12258
Zuo Z, Urban G, Scammell JG, Dean NM, McLean TK, Aragon I, Honkanen RE (1999) Ser/Thr protein phosphatase type 5 (PP5) is a negative regulator of glucocorticoid receptor-mediated growth arrest. Biochemistry 38(28):8849–8857. doi:10.1021/bi990842e
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Xinglong Zhi and Hongqi Zhang have equally contributed to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
Off-target exclusion assay of PPP5C using Lv-shPPP5C-S2 in U251 cells. a qPCR analysis of PPP5C knockdown efficiency by Lv-shPPP5C-S2 in U251 cells. b Western blot analysis of PPP5C knockdown efficiency by Lv-shPPP5C-S2 in U251 cells. c Growth curve of U251 cells after Lv-shPPP5C-S2 infection by MTT assay. d Representative images of colonies formed in Lv-shPPP5C-S2 infected U251 cells (scale bar: 250 µm). e Statistical analysis of colonies numbers in Lv-shPPP5C-S2 infected U251 cells. **: p < 0.01, ***: p < 0.001. Supplementary material 1 (TIFF 772 kb)
Rights and permissions
About this article
Cite this article
Zhi, X., Zhang, H., He, C. et al. Serine/Threonine Protein Phosphatase-5 Accelerates Cell Growth and Migration in Human Glioma. Cell Mol Neurobiol 35, 669–677 (2015). https://doi.org/10.1007/s10571-015-0162-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-015-0162-1