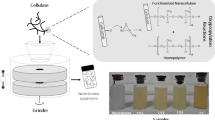
Abstract
Hemp bast (α-cellulose 79.4%, Klason lignin 4.9%) was directly oxidized by 2,2,6,6-tetramethylpiperidine-1-oxyl radical (TEMPO)-mediated oxidation in water at pH 10 and room temperature for 2 h. The level of added NaClO in oxidation varied from 5 to 30 mmol/g (based on dry weight of hemp bast). Weight recovery ratios of the TEMPO-oxidized hemp bast celluloses were in the range of 81–91%, and their carboxylate contents increased up to 1.2 mmol/g with the increased NaClO addition level. The lignin contents decreased to 0.5–0.9% after oxidation, and the viscosity-average degrees of polymerization decreased from 1100 to 560 because of depolymerization during oxidation. Thus, direct TEMPO-mediated oxidation of hemp bast introduced a significant number of carboxylate groups and simultaneously achieved sufficient delignification. Small amounts of xylose, mannose, and rhamnose originating from hemicelluloses remained in the TEMPO-oxidized hemp bast samples prepared by oxidation with 5–20 mmol/g NaClO. However, oxidation with 30 mmol/g NaClO completely removed these hemicellulose-originating sugars, and produced almost pure TEMPO-oxidized cellulose. When TEMPO-oxidized hemp bast samples were mechanically disintegrated in water, their nanofibrillation yields were 58–65%. After removal of unfibrillated fractions by centrifugation, transparent dispersions showed birefringence when observed between cross-polarizers, while atomic force microscopy images showed near-individually dispersed nanofibril elements with widths of ~2 nm.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The production of cellulose nanomaterials from renewable plant cellulose, which is the most abundant biopolymer on earth, has gained increasing attention in recent years. Plant cellulose fibers prepared by pulping and bleaching with or without pretreatment can be converted to nanofibrillated and microfibrillated celluloses by disintegration in water (Kalia et al. 2011; Klemm et al. 2011). Various pretreatments of wood celluloses or bleached chemical pulps (such as kraft and sulfite pulps) before mechanical disintegration have been reported to reduce energy consumption in the following mechanical fibrillation process in water. Mild endo-type cellulase treatment (Pääkkö et al. 2007; Henriksson et al. 2007), carboxymethylation (Wågberg et al. 2008), phosphorylation (Ghanadpour et al. 2015; Noguchi et al. 2017), periodate treatment (Sirviö et al. 2014; Yang et al. 2015), cationization (Horvath et al. 2008), and others are included in the pretreatments reported so far.
The catalytic oxidation of wood celluloses with 2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO) radical has been reported as one of the most efficient methods to prepare TEMPO-oxidized cellulose nanofibrils (TOCNs). The fibrils are typically 3–4 nm wide, several micromters long, and are dispersed in water with good homogeneity (Saito et al. 2006, 2007, 2009; Isogai et al. 2011; Isogai 2013). The cast and dried films prepared from aqueous TOCN dispersions are transparent, have high mechanical strengths, and high oxygen barrier properties under dry conditions, which are caused by the dense nanolayered structures in the film cross sections (Fukuzumi et al. 2009, 2011; Saito et al. 2011). When aerogels and cryogels are prepared from aqueous TOCN dispersions, they have unique thermal conductivities, filtration behaviors, and mechanical properties (Kobayashi et al. 2014; Nemoto et al. 2015; Sakai et al. 2016).
Softwood bleached kraft pulps (papermaking grade), in which glucomannan is present at 10–15% (w/w) as the main hemicellulose component, have been primarily used as the original wood cellulose to prepare and characterize TOCNs. When hardwood bleached kraft pulps (papermaking grade), which have xylan as the main hemicellulose component, are used to prepare TOCNs, some xylan tends to remain in the product because xylan has no C6–OH groups and is resistant to oxidative degradation (Rodionova et al. 2012; Tanaka et al. 2012). TOCNs prepared from hardwood bleached kraft pulp by mechanical disintegration under the same conditions therefore have lower light transparency or lower nano-dispersibility because hydrophobic xylan molecules remain present. Non-wood cellulose fibers such as cotton (de Morais et al. 2010; Soni et al. 2015), hemp, bamboo, and bagasse pulps (Puangsin et al. 2013a), hemp holocellulose (Puangsin et al. 2013b), and rice straw and kenaf holocelluloses (Kuramae et al. 2014) have been used as resources to prepare TOCNs. Because most of these non-wood cellulose fibers are delignified or bleached beforehand, they contain almost no lignin but do contain various hemicelluloses. Kuramae et al. (2014) reported that TEMPO-oxidized celluloses prepared from various plant holocelluloses by the TEMPO/NaBr/NaClO system in water at pH 10 contained almost no xylose or mannose, when suitable oxidation conditions were adopted. Thus, TOCNs prepared from various plant holocelluloses by using the TEMPO/NaBr/NaClO system have similar sugar compositions and nanofibril widths, although their lengths and length distributions tend to vary. In contrast, TEMPO-oxidized celluloses prepared from non-wood cellulose fibers contain significant amounts of mannose and xylose because of the presence of glucomannan and xylan (Kuramae et al. 2014; TEMPO/NaClO/NaClO2 system in water at pH ~7).
All plant celluloses, including agricultural wastes, have the potential to produce TOCNs with similar morphologies and properties. When the TEMPO/NaBr/NaClO oxidation is applied under suitable conditions (Kuramae et al. 2014), such non-wood resources may provide effective alternatives to paper pulp sources for TOCNs, particularly in developing countries. In our previous study, hemp holocellulose fibers were subjected to TEMPO/NaBr/NaClO oxidation to prepare TOCNs (Puangsin et al. 2013a, b). The TOCNs prepared from hemp bast holocellulose fibers had morphologies, chemical compositions, and film properties similar to those prepared from wood celluloses. However, in this case, the original hemp bast was first delignified and bleached by the Wise method (Puangsin et al. 2013a, b) to prepare hemp bast holocellulose fibers. Such pulping and bleaching processes, even for non-wood plants, require well-controlled and large-scale plant facilities to minimize any environmental burden caused by effluents and air emissions. Furthermore, the need for sequential process facilities for pulping, bleaching, and TEMPO-mediated oxidation make it difficult for local areas to use agricultural wastes as starting resources for TOCN production.
Because most agricultural wastes and some grass plants contain less lignin than wood resources, it may be possible for such lignin-containing non-wood cellulose resources to be pulped, bleached, and TEMPO-oxidized in one step to prepare TEMPO-oxidized celluloses with properties similar to those prepared from wood pulps. This one-step preparation process would be advantageous for local areas because it would require the installation of relatively small-scale facilities for TOCN production. Okita et al. (2009) and Ma and Zhai (2013) reported TEMPO-mediated oxidation of lignin-rich softwood thermomechanical pulps (TMPs) for preparation of TOCNs. They found that oxidation using TEMPO/NaBr/NaClO in water at pH 10 removed lignin and hemicelluloses in TMPs, and oxidized C6–OH groups present on the crystalline cellulose microfibril surfaces simultaneously. Moreover, the TEMPO/NaBr/NaClO oxidation appears to be a more efficient delignification process for TMPs than oxidation with NaClO alone (Okita et al. 2009).
In this study, TEMPO/NaBr/NaClO oxidation was directly applied to lignin-containing stick-like hemp bast (Cannabis sativa L. ssp. sativa) in water at pH 10 with various conditions. The prepared TEMPO-oxidized hemp bast samples were subjected to chemical analyses, including carboxylate, aldehyde, and lignin contents, sugar compositions, and viscosity-average degrees of polymerization (DPv) values. The prepared TEMPO-oxidized hemp bast samples were mechanically disintegrated in water under the same conditions, and the nanofibrillation behaviors of the samples were characterized by atomic force microscopy (AFM) and by measurements of dispersion transparency.
Experimental
Materials
Hemp bast was kindly supplied by Queen Sirikit Botanic Garden (QSBG) Project in Chiang Mai Province, Northern Thailand. The hemp bast was air-dried and cut into sticks (5–10 mm long) using scissors. The stick-like hemp bast sample was soaked in water for 24 h with continuous stirring to swell the material. The resulting sample was washed thoroughly with water several times by filtration on a grass filter. It was stored wet at 4 °C before further treatment or analysis. The original hemp bast sample had α-cellulose and Klason lignin contents of 79.4 and 4.9%, respectively. TEMPO, sodium bromide, sodium chlorite, 13% sodium hypochlorite solution, sodium hydroxide, and other chemicals and solvents were of laboratory grade (Wako Pure Chemicals, Osaka, Japan) and were used without further purification.
TEMPO-mediated oxidation of hemp bast
Hemp bast sample (1 g based on dry weight) was suspended in water (100 mL), containing TEMPO (0.016 g) and sodium bromide (0.1 g). TEMPO-mediated oxidation was initiated by addition of a designated amount of NaClO (5–30 mmol/g) to the slurry at room temperature. The mixture was stirred at pH 10 with continuous addition of 0.5 M NaOH using a pH stat for 2 h. The TEMPO-oxidized hemp bast cellulose (TOHBC) thus obtained was washed thoroughly with water by filtration and was stored wet at 4 °C until further analysis. Weight recovery ratios of TOHBCs were calculated from their dry weights before and after the TEMPO-mediated oxidation. Carboxylate and aldehyde contents of TOHBCs were determined by conductometric titration (Saito and Isogai 2004; Shinoda et al. 2012). TOHBCs were suspended in water, gently agitated, and converted to homogeneous handsheets by filtration on a Buchner funnel. ISO brightness values were measured according to TAPPI Test Method T452 om-08 (2011). Lignin contents of TOBHCs were evaluated as micro-Kappa numbers according to a previously reported method (Okita et al. 2009). The neutral sugar compositions were determined for TOBHCs by high-performance liquid chromatography (HPLC) after acid hydrolysis (Puangsin et al. 2013a).
Preparation of TEMPO-oxidized cellulose nanofibrils
TOHBC sample (never dried) was suspended in water (25 mL) at 0.1% (w/v) and then disintegrated using a double-cylinder-type homogenizer and an ultrasonic homogenizer according to the previously reported method (Puangsin et al. 2013a) to prepare an aqueous TOCN dispersion. Unfibrillated or partly fibrillated fraction was removed from the dispersion by centrifugation at 12,000×g for 20 min and the nanofibrillation yield was measured by the dry weight of the residual fraction (Shinoda et al. 2012). The morphology of the nanofibrils was observed by tapping-mode AFM (Nanoscope III Multimode, Digital Instruments, Massachusetts, USA), and TOCN fibril widths were measured from AFM height images.
Results and discussion
TEMPO-mediated oxidation of hemp bast
The original hemp bast was cut into small sticks, and these were directly treated with NaClO (0–30 mmol/g) as part of the TEMPO/NaBr/NaClO system in water at pH 10 and room temperature for 2 h. The hemp bast became swollen and partly turned to fibers or fiber bundles as the oxidation and delignification proceeded. Figure 1 shows carboxylate contents, DPv values, and weight recovery ratios of the TOHBCs. The carboxylate contents increased up to ~1.2 mmol/g and the DPv values decreased from 1100 to 560 as the amount of added NaClO increased to 30 mmol/g. Low aldehyde contents (0–0.12 mmol/g) were detected for the TOHBCs prepared under various conditions. The weight recovery ratio decreased from 92 to 81% as the amount of added NaClO increased from 5 to 30 mmol/g, respectively. Given that the α-cellulose content of the hemp bast was 79%, the weight recovery ratios shown in Fig. 1 indicate that almost all lignin and hemicelluloses present in the original hemp bast were degraded and removed in the water-soluble fraction during the oxidation and washing processes, as described later. Thus, direct oxidation of the hemp bast with NaClO (15–30 mmol/g) gave TEMPO-oxidized celluloses in high yields, with carboxylate contents and DPv values similar to those prepared from wood pulps.
Photographs of the prepared TOHBCs are shown in Fig. 2, together with their lignin contents and ISO brightness values. More than 80% of lignin present in the original hemp bast was removed by oxidation with 5 mmol/g NaClO, and then the lignin content gradually decreased to ~0.5% (i.e., ~90% removal of the original lignin), as the oxidation was performed with the increased amount of NaClO (20–30 mmol/g). Correspondingly, the ISO brightness of the TOHBC sheets increased to 80%. Thus, delignification and introduction of carboxylate groups (and partial conversion from stick morphologies to fibers and fiber bundles) can be achieved by direct TEMPO-mediated oxidation of stick-like hemp bast under suitable oxidation conditions.
Direct TEMPO-mediated oxidations of lignin-rich and wood-based TMPs were studied previously for simultaneous delignification and introduction of carboxylate groups in the preparation of TOCNs (Okita et al. 2009; Sbiai et al. 2011; Ma et al. 2012). However, in these previous studies, longer oxidation times were required to remove most of the lignin in the TMPs, and consequently the weight recovery ratios decreased to ~40%. Because the lignin content of hemp bast is much lower than that of wood, the direct TEMPO-mediated oxidation is well suited to the treatment of hemp bast to prepare TEMPO-oxidized celluloses in high yield with little or no lignin. Because the NaClO treatment in water at pH 10 did not efficiently remove lignin from TMP (Okita et al. 2009), the combination system of TEMPO/NaBr/NaClO in water at pH 10 was likely to efficiently oxidize and degrade the lignin component in the hemp bast and removed it from the water-insoluble TOHBC fractions during oxidation and washing processes.
The crystal structure and crystal sizes of the original cellulose I of hemp bast are probably unchanged for TOHBCs prepared under various conditions (Puangsin et al. 2013a, b). Thermal degradation points of TOHBCs prepared in this study are likely to be ~200 °C as in the case of other TEMPO-oxidized celluloses prepared from various plant celluloses (Fukuzumi et al. 2010). Decarboxylation of abundant sodium carboxylate groups present on crystalline cellulose microfibril surfaces in TEMPO-oxidized celluloses starts to occur at ~200 °C.
Sugar composition analysis of TOHBCs
The neutral sugar compositions of the original hemp bast and the TOHBCs prepared under various conditions are listed in Table 1. The original hemp bast sample contained 3.9% mannose and 2.7% xylose, as hemicellulose-originating neutral sugars. However, these values are lower than mannose content in softwood or xylose content in hardwood, because the α-cellulose content of hemp bast is as high as 79%. The weight ratios of these hemicellulose-originating neutral sugars decreased as the amount of added NaClO increased in the TEMPO-mediated oxidation. Because hemicelluloses have more disordered structures and lower molecular weights than cellulose, they are susceptible to degradation during oxidation and become water-soluble (Isogai et al. 2009). Unlike glucomannan in hemp bast hemicelluloses, xylan has no C6–OH groups and are therefore more resistant to degradation during oxidation. The ‘others’ fractions (see Table 1) are likely to consist of glucuronic acid, its dimers, and other compounds (Puangsin et al. 2013a, b). When 20 mmol/g NaClO was added, the TOHBC contained no mannose or xylose, resulting in the formation of pure TEMPO-oxidized cellulose from the hemp bast.
Characterization of TOHBC nanofibrils
The TOHBCs prepared under various conditions were mechanically disintegrated in water under the same moderate conditions. Before centrifugation, the TOHBC dispersions contained significant amounts of unfibrillated materials, and consequently their light transmittances were lower than 60% at 600 nm (Fig. 3). Correspondingly, the nanofibrillation yields ranged from 35 to 65% as the amount of added NaClO was increased from 5 to 30 mmol/g in the TEMPO-mediated oxidation. The photographs of the dispersions also suggested low nanofibrillation yields. Although almost pure TOCN can be prepared from hemp bast by TEMPO-mediated oxidation with 30 mmol/g NaClO (based on neutral sugar composition; see Table 1), the TOHBC dispersion had a low nanofibrillation yield. When hemp bast holocellulose was used as the starting material, the nanofibrillation yield reached ~80%, with a carboxylate content of ~1.2 mmol/g (Dufresne 2012; Puangsin et al. 2013a). Thus, the small amounts of residual lignin present in the TOHBCs may have caused the low nanofibrillation yields, which should be taken into account (Fujisawa et al. 2010; Chen et al. 2011).
After centrifugation to remove unfibrillated material, all the TOHBC dispersions had high transparencies of ~90% at 600 nm (Fig. 4). Moreover, the dispersions showed birefringence when observed between cross-polarizers, which suggests the presence of nearly individualized TOCNs in dispersion (photographs in Fig. 3) (De Souza Lima and Borsali 2004). The base-line shift at 720–740 nm in Fig. 4 is caused by a change of light source in the UV–vis spectrophotometer. The results in Figs. 3 and 4 show that it is difficult to prepare transparent dispersions consisting of mostly individualized nanofibrils in high yield from lignin-containing hemp bast by the direct TEMPO-mediated oxidation.
Photographs and UV–vis transmission spectra of aqueous nanofibril dispersions prepared from TEMPO-oxidized hemp bast celluloses by disintegration in water under the same conditions (original solid content was 0.1% w/v), after removal of unfibrillated fraction by centrifugation. Photographs were taken between cross-polarizers
Figure 5 shows AFM images of TOHBC nanofibrils prepared from hemp bast under different oxidation conditions. The width distributions of the TOHBC nanofibrils were measured from their AFM height images. The average widths of TOHBC nanofibrils were in the range of 1.6–2.1 nm. The TOHBC nanofibrils prepared by TEMPO-mediated oxidation using 5–20 mmol/g NaClO had relatively large width distributions and showed some network structures in the AFM images. In contrast, the TOHBC nanofibrils prepared with 20 mmol/g NaClO were mostly individualized and had a narrower width distribution. All the TOHBC nanofibrils were several microns in length. Thus, TOCNs with ~2 nm widths and high aspect ratios can be prepared from hemp bast by TEMPO-mediated oxidation when suitable oxidation conditions are adopted, although the nanofibrillation yields are low compared with TOCNs prepared from wood celluloses.
Conclusions
Direct TEMPO-mediated oxidation of lignin-containing hemp bast efficiently achieves simultaneous delignification (down to ~0.5%) and introduces carboxylate groups (up to ~1.2 mmol/g) sufficient for conversion to TOCNs by mechanical disintegration in water. When using 30 mmol/g NaClO in the TEMPO/NaBr/NaClO oxidation system, the lignin content decreased to ~0.5% and almost no mannose or xylose remained in the oxidized product. This indicates that almost all hemicelluloses were degraded and removed from the oxidized product. The weight recovery ratio and DPv value decreased to 81% and 560, respectively, and these values were higher than those of nanofibrils prepared by direct TEMPO-mediated oxidation of softwood TMPs. However, the TOHBC dispersion prepared from hemp bast had a low nanofibrillation yield of 65% and was not transparent. After removal of the unfibrillated fraction by centrifugation, the dispersion showed birefringence and contained mostly individualized TOCNs with widths of ~2 nm and high aspect ratios.
References
Chen W, Yu H, Liu Y, Hai Y, Zhang M, Chen P (2011) Isolation and characterization of cellulose nanofibers from four plant cellulose fibers using a chemical-ultrasonic process. Cellulose 18:433–442
de Morais Teixeira E, Corrêa AC, Manzoli A, de Lima Leite F, de Oliveira C, Mattoso L (2010) Cellulose nanofibers from white and naturally colored cotton fibers. Cellulose 17:595–606
De Souza Lima MM, Borsali R (2004) Rodlike cellulose microcrystals: structure, properties, and applications. Macromol Rapid Commun 25:771–787
Dufresne A (2012) Nanocellulose. Walter De Gruyter, Berlin, p 460
Fujisawa S, Saito T, Isogai A (2010) Temperature and pH stability of cellouronic acid. Cellulose 17:607–615
Fukuzumi H, Saito T, Iwata T, Kumamoto Y, Isogai A (2009) Transparent and high gas barrier films of cellulose nanofibers prepared by TEMPO-mediated oxidation. Biomacromolecules 10:162–165
Fukuzumi H, Saito T, Okita Y, Isogai A (2010) Thermal stabilization of TEMPO-oxidized cellulose. Polym Degrad Stab 95:1502–1508
Fukuzumi H, Saito T, Iwamoto S, Kumamoto Y, Ohdaira T, Suzuki R, Isogai A (2011) Pore size determination of TEMPO-oxidized cellulose nanofibril films by positron annihilation lifetime spectroscopy. Biomacromolecules 12:4057–4062
Ghanadpour M, Carosio F, Larsson TP, Wågberg L (2015) Phosphorylated cellulose nanofibrils: a renewable nanomaterial for the preparation of intrinsically flame-retardant materials. Biomacromolecules 16:3399–3410
Henriksson M, Henriksson G, Berglund LA, Lindström T (2007) An environmentally friendly method for enzyme-assisted preparation of microfibrillated cellulose (MFC) nanofibers. Eur Polym J 43:3434–3441
Horvath AT, Horvath AE, Lindström T, Wågberg L (2008) Diffusion of cationic polyelectrolytes into cellulosic fibers. Langmuir 24:10797–10806
Isogai A (2013) Wood nanocelluloses: fundamentals and applications as new bio-based nanomaterials. J Wood Sci 59:449–459
Isogai T, Yanagisawa M, Isogai A (2009) Degrees of polymerization (DP) and DP distribution of cellouronic acid prepared from alkali-treated celluloses and ball-milled celluloses by TEMPO-mediated oxidation. Cellulose 16:117–127
Isogai A, Saito T, Fukuzumi H (2011) TEMPO-oxidized cellulose nanofibers. Nanoscale 3:71–85
Kalia S, Kaith BS, Kaur I (2011) Cellulose fibers: bio- and nano-polymer composites. Springer, Berlin, p 737
Klemm D, Kramer F, Moritz S, Lindström T, Ankerfors M, Gray D, Dorris A (2011) Nanocelluloses: a new family of nature—based materials. Angew Chem Int Ed 50:5438–5466
Kobayashi Y, Saito T, Isogai A (2014) Aerogels with 3D ordered nanofiber skeletons of liquid-crystalline nanocellulose derivatives as tough and transparent insulators. Angew Chem Int Ed 53:10394–10397
Kuramae R, Saito T, Isogai A (2014) TEMPO-oxidized cellulose nanofibrils prepared from various plant holocelluloses. React Funct Polym 85:126–133
Ma P, Zhai H (2013) Selective TEMPO-oxidation of thermomechanical pulp. Bioresources 8:4396–4405
Ma P, Fu S, Zhai HM, Law KN, Daneault C (2012) Influence of TEMPO-mediated oxidation on the lignin of thermomechanical pulp. Bioresour Technol 118:607–610
Nemoto J, Saito T, Isogai A (2015) Simple freeze-drying procedure for producing nanocellulose aerogel-containing, high-performance air filters. ACS Appl Mater Interfaces 7:19809–19815
Noguchi Y, Homma I, Matsubara Y (2017) Complete nanofibrillation of cellulose prepared by phosphorylation. Cellulose 24:1295–1305
Okita Y, Saito T, Isogai A (2009) TEMPO-mediated oxidation of softwood thermomechanical pulp. Holzforschung 63:529–535
Pääkkö M, Ankerfors M, Kosonen H, Nykanen A, Ahola S, Osterberg M, Ruokolainen J, Laine J, Larsson PT, Ikkala O, Lindström T (2007) Enzymatic hydrolysis combined with mechanical shearing and high-pressure homogenization for nanoscale cellulose fibrils and strong gels. Biomacromolecules 8:1934–1941
Puangsin B, Fujisawa S, Kuramae R, Saito T, Isogai A (2013a) TEMPO-mediated oxidation of hemp bast holocellulose to prepare cellulose nanofibrils dispersed in water. J Polym Environ 21:555–563
Puangsin B, Yang Q, Saito T, Isogai A (2013b) Comparative characterization of TEMPO-oxidized cellulose nanofibril films prepared from non-wood resources. J Biol Macromol 59:208–213
Rodionova G, Saito T, Lenes M, Eriksen Ø, Gregersen Ø, Fukuzumi H, Isogai A (2012) Mechanical and oxygen barrier properties of films prepared from fibrillated dispersions of TEMPO-oxidized Norway spruce and eucalyptus pulps. Cellulose 19:705–711
Saito T, Isogai A (2004) TEMPO-mediated oxidation of native cellulose. The effect of oxidation conditions on chemical and crystal structures of the water-insoluble fractions. Biomacromolecules 5:1983–1989
Saito T, Nishiyama Y, Putaux JL, Vignon M, Isogai A (2006) Homogeneous suspensions of individualized microfibrils from TEMPO-catalyzed oxidation of native cellulose. Biomacromolecules 7:1687–1691
Saito T, Kimura S, Nishiyama Y, Isogai A (2007) Cellulose nanofibers prepared by TEMPO-mediated oxidation of native cellulose. Biomacromolecules 8:2485–2491
Saito T, Hirota M, Tamura N, Kimura S, Fukuzumi H, Heux L, Isogai A (2009) Individualization of nano-sized plant cellulose fibrils by direct surface carboxylation using TEMPO catalyst under neutral conditions. Biomacromolecules 10:1992–1996
Saito T, Uematsu T, Kimura S, Enomae T, Isogai A (2011) Self-aligned integration of native cellulose nanofibrils towards producing diverse bulk materials. Soft Matter 7:8804–8809
Sakai K, Kobayashi Y, Saito T, Isogai A (2016) Partitioned airs at microscale and nanoscale: thermal diffusivity in ultrahigh porosity solids of nanocellulose. Sci Rep 6:20434
Sbiai A, Sautereat H, Maazouz H, Fleury E (2011) TEMPO-mediated oxidation of lignocellulosic fibers from date palm leaves. Carbohydr Polym 86:1445–1450
Shinoda R, Saito T, Okita Y, Isogai A (2012) Relationship between length and degree of polymerization of TEMPO-oxidized cellulose nanofibrils. Biomacromolecules 13:842–849
Sirviö JA, Kolehmainen A, Visanko M, Liimatainen H, Niinimäki J, Hormi OEO (2014) Strong, self-standing oxygen barrier films from nanocelluloses modified with regioselective oxidative treatments. ACS Appl Mater Interfaces 6:14384–14390
Soni B, Hassan EB, Mahmoud B (2015) Chemical isolation and characterization of different cellulose nanofibers from cotton stalks. Carbohydr Polym 134:581–589
Tanaka R, Saito T, Isogai A (2012) Cellulose nanofibrils prepared from softwood cellulose by TEMPO/NaClO/NaClO2 systems in water at pH 4.8 or 6.8. Int J Biol Macromol 51:228–234
Wågberg L, Decher G, Norgren M, Lindström T, Ankerfors M, Axnas K (2008) The build-up of polyelectrolyte multilayers of microfibrillated cellulose and cationic polyelectrolytes. Langmuir 24:784–795
Yang H, Chen D, van de Ven TGM (2015) Preparation and characterization of sterically stabilized nanocrystalline cellulose obtained by periodate oxidation of cellulose fibers. Cellulose 22:1743–1752
Acknowledgments
The authors thank Dr. Suphat Kamthai of Chaing Mai University and Queen Sirikit Botanic Garden, Thailand, who kindly provided the hemp bast sample. This research was supported by Core Research for Evolutional Science and Technology (CREST, Grant Number JPMJCR13B2) of the Japan Science and Technology Agency (JST).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Puangsin, B., Soeta, H., Saito, T. et al. Characterization of cellulose nanofibrils prepared by direct TEMPO-mediated oxidation of hemp bast. Cellulose 24, 3767–3775 (2017). https://doi.org/10.1007/s10570-017-1390-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-017-1390-y