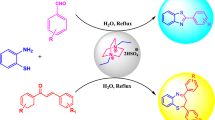
Abstract
This study explains the synthesis of 1,3-dithiolane and benzothiazolo-[2,3-b]-quinazolin-1-one derivatives employing DABCO-based ionic liquids as a facile, sustainable, and recyclable catalyst. FT-IR, NMR, and mass spectrometry were used to characterize synthetic compounds. The current approach displays a number of advantages, including an affordable and environmentally friendly catalyst, green solvent media, a reduced reaction time, good to exceptional product yields, and recyclability of the ionic liquid for subsequent reactions without a significant loss in activity.
Graphical Abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Ionic liquids are currently receiving a lot of interest for their potential use as catalysts in a variety of chemical reactions [1,2,3,4]. Ionic liquids have attracted research and academia, and their catalytic properties have been studied extensively in recent years because of their alluring properties, including low vapour pressure, dissolving power, non-volatility, non-flammability, and high thermal stability [5,6,7]. From an environmental viewpoint, the quest for a variety of ionic liquids that may act as catalysts and solvents is necessary. The popularity of ionic liquids in numerous fields of science can be attributed to their structural flexibility, such as electrochemistry, chemistry, biology, and synthetic and industrial chemistry [8,9,10,11]. The physical and chemical properties of ionic liquids are determined by the types of their cation and anion constituents. This shows that a given ionic liquid can be systematically tailored to have precise features termed as “task specific” and “designer” ionic liquids [12, 13].
The acetalization reaction is a technique that is frequently used in organic synthesis to protect the carbonyl group of aldehydes and ketones and to produce final products, such as enantiomerically pure compounds [14] that are useful in the production of synthetic carbohydrates [15], steroids [16], pharmaceuticals, and fragrances [17]. In the synthesis of natural products and polyfunctional molecules, the protection and deprotection of reactive functional groups is required. In organic synthesis, the carbonyl group is one of the most studied and highly valued functional groups due to its strong reactivity and extensive variety of transformations [18]. The protection of carbonyl functionality as dithioacetals or dithioketals is a common and widely utilized strategy in organic chemistry, and it is regularly employed as a synthetic step for the synthesis of many significant natural and synthetic organic compounds [19, 20]. The last step of the multistep synthesis includes deprotection of the thioacetal to the original carbonyl group. Dethioacetalization of thioacetals into the corresponding carbonyl compounds is a crucial transformation, but it is not a simple procedure. Dithioacetals are commonly prepared by condensation of aldehydes and ketones with dithiols employing protic acids, lewis acids, a number of transition metal complexes, and polymer supported silica reagents.
Protecting carbonyl functional groups from nucleophilic attack until their electrophilic nature is utilized during multistep synthesis is one of the most difficult challenges. Dithioacetals are a key family of compounds among carbonyl protecting groups as acyl anion equivalents or masked methylene in carbon–carbon bond-forming processes [21, 22]. Owing to its efficiency in synthesis and inherent stability under acidic or basic conditions, 1,3-dithioacetal protection is regarded as the most versatile among the current protective groups. Deprotection of acetals [23] and thioacetals [24] to the parent carbonyl group and transthioacetalization of O,O-acetals to S,S-acetals is a synthetically advantageous conversion.
Despite the fact that many studies used traditional catalysts for carbonyl compound protection; there is still a focus on finding alternative catalysts to solve issues like hard reaction conditions and low chemoselectivity. Additionally, environmental and commercial factors drove us to redesign these procedures. As a consequence, there is more scope to investigate mild and effective ways to transthioacetalize substances. Water based organic reactions without the utilization of hazardous chemicals, have generated a lot of interest in recent years because water is widely accessible, relatively inexpensive, and environmentally friendly [25]. Various methods have been reported in the literature to deal with these dithiols because of their significance (Table 1).
Owing to the vast spectrum of biological actions, benzothiazolo-[2,3-b]-quinazolin-1-one derivatives are the most significant class of heterocyclic molecules, and the synthesis of these molecules has attracted a lot of interest in the realm of medicinal chemistry. A review of the literature revealed that pyrimido[2,1-b]benzothiazoles demonstrate a number of biological activities, including anti-inflammatory [26], anti-fertility [27], anticancer [28], antimicrobial [29], anti-allergic [30], anti-HIV [31], antiviral [32], fungicidal [33], and anticonvulsant [34]. Considering the importance of these benzothiazoloquinazolinone molecules, numerous techniques have been documented in the literature (Table 2).
There is a great need to design a new and improved method that employs less hazardous and ecologically friendly materials to limit the amount of toxic waste and byproducts. In continuation of our ongoing framework to support environmentally benign methods in aqueous medium, we report a convenient, mild, and highly chemoselective method for the thioacetalization of aldehydes and ketones and the synthesis of benzothiazolo-[2,3-b]-quinazolin-1-one derivatives utilizing DABCO based acidic ionic liquids using a catalytic amount of DABCO based acidic ionic liquids.
2 Experimental Section
2.1 Materials and Methods
All solvents and chemical materials used in this study were acquired from Spectrochem, Merk, and Sigma Aldrich and used without any purification, while solvents were purified by distillation before being used. Melting points were recorded in capillary tubes by a programmable melting point apparatus, and changes in chemicals were observed visually. FT-IR spectra were recorded with KBr pellets by the Perkin-Elmer spectrometer. 1HNMR and 13CNMR spectra were determined on the BRUKER AVANCE NEO Spectrometer at 500 MHz and 125 MHz respectively, in CDCl3 and DMSO-d6 solvents. Mass spectra were analyzed by a Maldi Synapt XS HD mass spectrometer. TLC was used to examine the purity of the product and monitor the reaction.
2.2 Procedure for Synthesis of 1,4-Diethyl-1,4-diazoniabicyclo[2,2,2]octane Dibromide
In a 100 mL flask, bromoethane (22 mmol) and 1,4-diazabicyclo[2.2.2]octane (DABCO) (10 mmol) in dichloromethane (10 mL) were mixed and stirred at room temperature for 12 h. The reaction mixture was then vacuum evaporated to obtain a white residue, which was subsequently triturated with dichloromethane (3 × 15 mL) to yield a white precipitate, which was later filtered off and vacuum dried.
2.3 Procedure for the Synthesis of 1,4-Diethyl-1,4-diazabicyclo[2,2,2]octane-1,4-diium Hydrogen Sulfate
A stoichiometric amount of sulfuric acid (70%, 12 mmol) was added drop-by-drop at 0 °C to a suspension of 1,4-diethyl-1,4-diazoniabicyclo[2,2,2]octane dibromide (5 mmol) in diethyl ether (10 mL) and the mixture was stirred for 10 h at room temperature. After completion of the reaction, the solvent was decanted, and the resulting yellowish tinted solid was frequently washed with diethyl ether (3 × 10 mL) to obliterate non-ionic residues. Finally, the acquired white solid product was filtered and vacuum dried (Scheme 1) [66].
2.4 Procedure for the Synthesis of 1,4-Diethyl-1,4-diazabicyclo[2,2,2]octane-1,4-diium Perchlorate
A stoichiometric amount of perchloric acid (70%,12 mmol) was added drop-by-drop at 0 °C to a suspension of 1,4-diethyl-1,4-diazoniabicyclo[2,2,2]octane dibromide (5 mmol) in diethyl ether (10 mL) and the mixture was stirred for 10 h at room temperature. After completion of the reaction, the solvent was decanted, and the resulting yellowish tinted solid was frequently washed with diethyl ether (3 × 10 mL) to obliterate non-ionic residues. Finally, the acquired white solid product was filtered and vacuum dried (Scheme 2) [67].
2.5 Procedure for the Preparation of Catalyst [H2-DABCO][H2PO4]2/[H2-DABCO][HSO4]2/[H2-DABCO][ClO4]2
A stoichiometric amount of phosphoric acid/sulfuric acid/perchloric acid (12 mmol) was added drop-by-drop at 0 °C to the solution of 1,4-diazabicyclo[2.2.2]octane (5 mmol) in dichloromethane (10 mL) and the mixture was stirred for 20 h at room temperature. Then the solvent was decanted, and the resultant white solid was frequently washed with diethyl ether (3 × 10 mL) to exclude all non-ionic residues. Finally, the obtained ionic liquid was vacuum dried and employed as a catalyst for further reactions [68, 69].
2.6 General Procedure for the Synthesis of 2-(phenyl)-1,3-dithiolane
In a mixture of carbonyl compound (1 mmol) and 1,2-ethanedithiol (1.2 mmol), the dicationic acidic ionic liquid (4 mol%) catalyst and water (5 mL) is added and left for stirring at the room temperature for the required time (Scheme 3). After completion of the reaction, the reaction mixture was extracted with ethyl acetate. The organic and aqueous layers were separated. The organic layer was concentrated under reduced pressure and purified the crude product by recrystallization with methanol. Using a functional test for aldehydes or ketones, the purified product was screened qualitatively.
2.7 General Procedure for the Synthesis of Benzothiazolo-[2,3-b]-quinazolin-1-one
A mixture of aromatic aldehydes (1 mmol), 1,3-diketones (1 mmol) (Scheme 4) or β-dicarbonyls (Scheme 5), and 2-aminobenzothiazole (1 mmol) in water (5 mL) was refluxed for the required period of time while being exposed to the ionic liquid catalyst (4 mol%). After completion of the reaction, the reaction mixture was cooled to room temperature, and extracted with ethyl acetate. The organic layer and aqueous layers were separated, the organic layer was concentrated under reduced pressure, and the crude product was purified by recrystallization with methanol. The catalyst was dissolved in water and extracted by condensing the aqueous layer under low pressure.
3 Results and Discussion
An ionic liquid that has undergone anionic variations to modify its activity and selectivity is described as a task-specific ionic liquid. In the ongoing work, we examine the feasibility of several acidic ionic liquids based on DABCO, including [H2-DABCO][H2PO4]2, [H2-DABCO][HSO4]2, [H2-DABCO][ClO4]2, [C4H10-DABCO][HSO4]2, and [C4H10-DABCO][ClO4]2.
To optimize the reaction conditions, the synthesis of 2-(4-chlorophenyl)-1,3-dithiolane from 4-Cl benzaldehydes (1 mmol) and 1,2-ethanedithiol (1.2 mmol) was selected as the model reaction, and it was carried out under various reaction parameters, including the solvent and amount of catalysts has been investigated and observations were noted in Table 3. The model reaction was initially performed using a variety of solvents, including ethanol, DMF, acetonitrile, toluene, water, and solvent free.
It was found that among all solvents, water was the most efficient solvent for this reaction rather than other solvents in terms of both reaction time and yield of the required 2-(4-chlorophenyl)-1,3-dithiolane. Investigating the effects of varied ionic liquid concentrations on the model reaction, it was found that 4 mol% of catalyst was adequate to carry out the reaction in high yields regardless of the concentration of the ionic liquid. In order to increase the yield of the desired product, the catalyst was increased to 5 mol%, but no appreciable improvements were seen in the yield of 2-(4-chlorophenyl)-1,3-dithiolane. Therefore, utilizing 4 mol% of the catalyst at room temperature in an aqueous medium is the ideal reaction condition for the synthesis of 2-(4-chlorophenyl)-1,3-dithiolane derivatives (Table 3, row 7).
To provide favourable conditions for the synthesis of benzothiazolo-[2,3-b]-quinazolin-1-ones, the model reaction was selected and ran it under different reaction parameters, as shown in Table 4. Initially, the model reaction was conducted using different solvents like ethanol, methanol, DMF, acetonitrile, water, toluene, and solvent free conditions. It was found that, among all solvents, water was the most effective solvent for this reaction rather than other solvents in terms of reaction time and yield of the desired benzothiazolo-[2,3-b]-quinazolin-1-ones. The effect of the catalyst on the model reaction was investigated in varying amounts. It was found that increasing the ionic liquid concentration by 4 mol% was sufficient to complete the reaction with a high yield. To increase the yield of the desired product, the catalyst concentration was raised to 5 mol%, but this had no noticeable influence on the yield of benzothiazolo-[2,3-b]-quinazolin-1-one. Under the reflux condition, the maximum yield of the end product was noted. As a result, refluxing the reaction with 4 mol% of the catalyst in an aqueous medium provides the optimal reaction condition for the synthesis of benzothiazolo-[2,3-b]-quinazolin-1-one derivative (Table 4 Row 9).
The next step is to test the catalytic ability of ionic liquids in the acceleration of organic transformations in the synthesis of 2-(4-chlorolphenyl)-1,3-dithiolane. The catalytic efficiency of DABCO based ionic liquids was evaluated, and the findings demonstrated that [C4H10-DABCO][ClO4]2 ionic liquid promoted reaction in a shorter time with a higher yield than the other stated DABCO based ionic liquids (Table 5). Then the efficiency of acidic ionic liquids based on DABCO in the synthesis of 12-(4-Hydroxy-phenyl)-2,3,4,12-tetrahydrobenzo[4,5]thiazolo[2,3-b]quinazolin-1-one was examined, and the findings demonstrated that [H2-DABCO][ClO4]2 promoted reaction in a shorter time with a higher yield than the other indicated acidic ionic liquids Table 6).
We explored reaction applicability with several aldehydes and ketones containing different electron withdrawing or electron donating substituents, which gave the expected products, 1,3-dithiolane and benzothiazolo-[2,3-b]-quinazolin-1-one derivatives (Table 7), at good to high yields with high purity in aqueous media. It was observed that aromatic aldehydes with electron withdrawing groups give the desired products faster than those with electron donating groups.
Due to the reactivity difference between aldehydes and ketones, this methodology may be used for chemoselective protection of the aldehydes in the presence of ketones. For this confirmation, an equimolar mixture of benzaldehydes and acetophenone was reacted with 1,2-ethanedithiol using ionic liquid (4 mol%) as a catalyst, yielding only 2-(phenyl)-1,3-dithiolane in a 90% yield and 96% recovery of acetophenone (Scheme 6).
By periodically performing the model reaction under optimum conditions, the recyclability of ionic liquid was examined for the synthesis of 2-(4-chlorophenyl)-1,3-dithiolane (Fig. 1) and 12-(4-Hydroxy-phenyl)-2,3,4,12-tetrahydrobenzo[4,5]thiazolo[2,3-b]quinazolin-1-one (Fig. 2). The product was separated after the completion of the reaction by extracting with ethyl acetate and separating the organic and aqueous phases. The catalyst was recovered by condensing the aqueous layer at reduced pressure, washing it in hot ethanol, and reusing it without losing any reactivity in the subsequent cycles. As a consequence, it can be stated that the recovered catalyst is sufficiently stable after five cycles and can be used in the synthesis of 2-(4-chlorophenyl)-1,3-dithiolane and 12-(4-Hydroxy-phenyl)-2,3,4,12-tetrahydrobenzo[4,5]thiazolo[2,3-b]quinazolin-1-one.
Figure 3 and 4 demonstrates the relationship between the yield of 2-(4-chlorophenyl)-1,3-dithiolane and 12-(4-Hydroxy-phenyl)-2,3,4,12-tetrahydrobenzo[4,5]thiazolo[2,3-b]quinazolin-1-one with reaction time. The yield of 2-(4-chlorophenyl)-1,3-dithiolane increased significantly between 0.1 and 1 h, whereas extending the reaction time had no effect on the reaction rate and was completed in about 1 h. Also the yield of 12-(4-Hydroxy-phenyl)-2,3,4,12-tetrahydrobenzo[4,5]thiazolo[2,3-b]quinazolin-1-one increased between 1 and 2.5 h Contrarily, increasing the reaction time has no impact on the reaction rate.
The probable mechanism for the synthesis of 1,3-dithiolane is shown in Scheme 7. When aldehydes are coordinated with ionic liquid, the carbonyl group of the aldehydes is activated, and the thiol group of the ethylenedithiol attacks the activated carbon of the carbonyl group, resulting in an intermediate I. Finally, the intermediate undergoes intramolecular proton transfer followed by the removal of the water molecule to produce 1,3-dithiolane.
Scheme 8 proposes a potential chemical pathway for the reaction between aldehyde, 1,3-diketone, and 2-aminobenzothiazole. Aldehyde and 1,3-diketone have first been bound to an ionic liquid catalyst and activated for further condensation. The Knoevenagel reaction was used to carry out the condensation of activated aldehyde and 1,3-diketone, producing an intermediate I. Following that, 2-aminobenzothiazole generated an iminium ion by reacting with intermediate I through a Michael addition. Through proton transfer and cyclization, the final product, 4H-pyrimido[2,1-b]benzothiazole, was produced [53, 72].
4 Conclusions
The present study shows the synthesis of 2-(phenyl)-1,3-dithiolane and benzothiazolo-[2,3-b]-quinazolin-1-one derivatives using DABCO based acidic ionic liquids as an eco-friendly, recyclable, non toxic, and thermally stable catalyst in an aqueous medium. FT-IR, NMR, and mass spectrometry were used to characterize the structure of synthesized compounds. In this work, the catalytic applicability of different DABCO based ionic liquids was investigated, and the results revealed that [H2-DABCO][H2PO4]2 ionic liquid had lower catalytic activity, whereas [C4H10-DABCO][ClO4]2 ionic liquid shows higher catalytic activity. The proposed method displayed several notable merits, including an environment friendly and inexpensive catalyst, green solvent media, a shorter reaction time, good to excellent product yield, and recyclability of the ionic liquid without a considerable reduction in activity.
References
Zhu A, Jiang T, Wang D, Han B, Liu L, Huang J, Zhang J, Sun D (2005) Green Chem 7(7):514–517
Lee CW (1999) Tetra Lett 40(13):2461–2464
Ranu BC, Banerjee S (2005) Org Lett 7(14):3049–3052
Hou HL, Qiu FL, Ying AG, Xu SL (2015) Chin Chem Lett 26(3):377–381
Hagiwara R, Ito Y (2000) J Fluor Chem 105(2):221–227
Seddon KR (1997) J Chem Tech Biotech: Inter Res Proce Environ Clean Techn 68(4):351–356
Zhang SJ, Lu XM (2006) Ionic Liq: Fun Res Indust Appli.
Xiong Y, Zhang Z, Wang X, Liu B, Lin J (2014) Chem Eng J 235:349–355
Zang H, Wang M, Cheng BW, Song J (2009) Ultrason Sonochem 16(3):301–303
Hajipour AR, Rafiee F (2009) J Iran Chem Soci 6(4):647–678
Shang Y, Li H, Zhang S, Xu H, Wang Z, Zhang L, Zhang J (2011) Chem Eng J 175:324–329
Gui J, Deng Y, Hu Z, Sun Z (2004) Tetra Lett 45(12):2681–2683
Davis H Jr, J, (2004) Chem Lett 33(9):1072–1077
Mori A, Yamamoto H (1985) J Org Chem 50(25):5444–5446
Clode DM (1979) Chem Rev 79(6):491–513
Bull JR, Floor J, Kruger GJ (1979) J Chem Res Synop 224.
Bruns K, Conrad J, Steigel A (1979) Tetra 35(21):2523–2530
Giovanni S, Roberto B, Franca B (2004) Chem Rev 104(1):199–250
Cordes EH, Bull HG (1974) Chem Rev 74(5):581–603
Oh S, Jeong IH, Ahn CM, Shin WS, Lee S (2004) Bioorg Med Chem 12(14):3783–3790
Utimoto K, Nakamura A, Matsubara S (1990) J Ame Chem Soc 112(22):8189–8190
Corey EJ, Seebach D (1966) J Org Chem 31(12):4097–4099
Burghardt TE (2005) J Sulf Chem 26(4–5):411–427
Carrigan MD, Sarapa D, Smith RC, Wieland LC, Mohan RS (2002) J Org Chem 67(3):1027–1030
Karimi B, Khalkhali M (2007) J Mol Cata A: Chem 271(1–2):75–79
Alagarsamy V, Revathi R, Meena S, Ramaseshu KV, Rajasekaran S, De Clerco E (2004) Ind J Pharma Sci 66(4):459
Hassan AY (2009) Phospho Sulf Sili 184(11):2856–2869
Tanabe Y, Kawai A, Yoshida Y, Ogura M, Okumura H (1997) Hetero 8(45):1579–1588
Singh SP, Misra RS, Parmar SS, Brumleve SJ (1975) J Pharma Sci 64(7):1245–1247
Lebel H, Ladjel C, Brethous L (2007) J Ame Chem Soci 129(43):13321–13326
Atar AB, Jeong YS, Jeong YT (2014) Tetra 70(34):5207–5213
Gajaganti S, Kumari S, Kumar D, Allam BK, Srivastava V, Singh S (2018) J Hetero Chem 55(11):2578–2584
Mazaahir K, Ritika C, Divya B (2012) Sci China Chem 55(10):2154–2160
Heravi MM, Ranjbar L, Derikvand F, Alimadadi B, Oskooie HA, Bamoharram FF (2008) Mol div 12(3):181–185
Kadam KR (2020) J Sulf Chem 41(5):530–541
Kamal A, Chouhan G (2002) Syn 3:0474–0476
De SK (2004) Tetra Lett 45(11):2339–2341
Hajipour AR, Pourmousavi SA, Ruoho AE (2008) Synth Comm 38(15):2548–2566
Perin G, Mello LG, Radatz CS, Savegnago L, Alves D, Jacob RG, Lenardao EJ (2010) Tetra Lett 51(33):4354–4356
Chaiseeda K, Chavasiri W (2017) Phosp Sulf Silic Relat Eleme 192(9):1034–1039
Kamal A, Chouhan G (2003) Tetra Lett 44(16):3337–3340
Adapa SR, Varala R, Nuvula S (2006) Bull Kor Chem Soc 27(7):1079–1082
Dong D, Ouyang Y, Yu H, Liu Q, Liu J, Wang M, Zhu J (2005) J Org Chem 70(11):4535–4537
Hajipour AR, Khazdooz L, Ruoho AE (2009) J Sulf Chem 30(1):46–52
Kamal A, Chouhan G (2004) Adv Synth Cat 346(5):579–582
Karimi B, Vafaeezadeh M (2013) RSC adv 3(45):23207–23211
Gupta N, Kad GL, Singh J (2007) Cata Comm 8(9):1323–1328
Ren YM, Shao JJ, Wu ZC, Zhang S, Tao TX (2014) J Mol Liq 196:392–394
Lenardao EJ, Borges EL, Mendes SR, Perin G, Jacob RG (2008) Tetra Lett 49(12):1919–1921
Habibi D, Rahmani P, Akbaripanah Z (2014) Chem Pape 68(3):417–421
Khaef S, Zolfigol MA, Taherpour AA, Yarie M (2020) RSC Adv 10(73):44946–44957
Sedrpoushan A, Ghazizadeh H (2017) J Sulf Chem 38(1):112–118
Alishahi N, Nasr-Esfahani M, Mohammadpoor-Baltork I, Tangestaninejad S, Mirkhani V, Moghadam M (2020) Appl Organometal Chem 5681.
Kour M, Paul S, Clark JH, Gupta VK, Kant R (2016) J Mol Cata A: Chem 411:299–310
Azad S, Mirjalili BBF (2017) Res on Chem Intermed 43(3):1723–1734
Mirjalili BBF, Aref F (2018) Res on Chem Intermed 44(7):4519–4531
Fazeli-Attar SA, Mirjalili BBF (2018) Res on Chem Intermed 44(10):6419–6430
Shaabani A, Rahmati A, Naderi S (2005) Bioorg Med Chem Lett 15(24):5553–5557
Hosseinikhah SS, Mirjalili BBF (2022) Polycy Aroma Comp 42(4):1013–1022
Khazenipour K, Moeinpour F, Mohseni-Shahri FS (2021) J Chin Chem Soci 68(1):121–130
Talaei R, Olyaei A (2016) Iran J Cata 6(4):339–343
Ziarani GM, Badiei A, Aslani Z, Lashgari N (2015) Arabi J Chem 8(1):54–61
Maleki A, Aghaei M, Ghamari N (2015) Chem Lett 44(3):259–261
Shirini F, Langarudi MSN, Daneshvar N, Mashhadinezhad M, Nabinia N (2017) J Mol Liq 243:302–312
Seyyedi N, Shirini F, Langarudi MSN (2017) J Irani Chem Soci 14(9):1859–1867
Pinate P, Makone S. (2022) Cata Lett 1–18.
Pinate P, Makone S (2023) J Sulf Chem 44(1):20–36
Shirini F, Langarudi MSN, Daneshvar N, Jamasbi N, Irankhah-Khanghah M (2018) J Mol Struct 1161:366–382
Jamasbi N, Irankhah-Khanghah M, Shirini F, Tajik H, Langarudi MSN (2018) New J Chem 42(11):9016–9027
Veisi H, Ghorbani-Vaghei R, Dadamahaleh SA (2011) J Hetero Chem 48(3):699–705
Hajipour AR, Hosseini P, Ruoho AE (2008) Phosp Sulf Silic 183(10):2502–2508
Bhoi MN, Borad MA, Pithawala EA, Patel HD (2019) Arab J Chem 12(8):3799–3813
Zhang J, Bao S, Yang J (2009) Chin Sci Bulle 54(21):3958–3964
Fatemeh C, Fatemeh D, Saeed B (2012) Inter J Org Chem 2012.
Mirjalili BBF, Aref F (2018) Res on Chem Intermed 44:4519–4531
Acknowledgements
The authors are grateful to acknowledge the Department of Science & Technology (DST), New Delhi for the award of Junior Research Fellowship (JRF) and Senior Research fellowship (SRF) [Grant No: DST/INSPIRE Fellowship/IF160600] for the research work.
Funding
This article was funded by Department of Science & Technology (DST), New Delhi, DST/INSPIRE Fellowship/IF160600.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pinate, P., Saifan, A.A.A. & Makone, S. Study of DABCO Based Acidic Ionic Liquids Strategy for the Synthesis of 1,3-Dithiolane and Benzothiazolo-[2,3-b]-quinazolin-1-one Derivatives. Catal Lett 154, 1462–1478 (2024). https://doi.org/10.1007/s10562-023-04411-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-023-04411-1