Abstract
Objectives
Especially patients with aortic aneurysms and multiple computed tomography angiographies (CTA) might show medical conditions which oppose the use of iodine-based contrast agents. CTA using monoenergetic reconstructions from dual layer CT and gadolinium (Gd-)based contrast agents might be a feasible alternative in these patients. Therefore, the purpose of this study was to evaluate the feasibility of clinical spectral CTA with a Gd-based contrast agent in patients with aortic aneurysms.
Methods
Twenty-one consecutive scans in 15 patients with and without endovascular aneurysm repair showing contraindications for iodine-based contrast agents were examined using clinical routine doses (0.2 mmol/kg) of Gd-based contrast agent with spectral CT. Monoenergetic reconstructions of the spectral data set were computed.
Results
There was a significant increase in the intravascular attenuation of the aorta between pre- and post-contrast images for the MonoE40 images in the thoracic and the abdominal aorta (p < 0.001 for both). Additionally, the ratio between pre- and post-contrast images was significantly higher in the MonoE40 images as compared to the conventional images with a factor of 6.5 ± 4.5 vs. 2.4 ± 0.5 in the thoracic aorta (p = 0.003) and 4.1 ± 1.8 vs. 1.9 ± 0.5 in the abdominal aorta (p < 0.001).
Conclusions
To conclude, our study showed that Gd-CTA is a valid and reliable alternative for diagnostic imaging of the aorta for clinical applications. Monoenergetic reconstructions of computed tomography angiographies using gadolinium based contrast agents may be a useful alternative in patients with aortic aneurysms and contraindications for iodine based contrast agents.
Key points
-
Patients with aortic aneurysms may show contraindications for iodine based contrast agents but need multiple follow-up examinations.
-
Monoenergetic reconstructions of computed tomography angiography using gadolinium based contrast agents is feasible in patients with aortic aneurysms.
-
Dual-layer spectral computed tomography may therefore be a useful tool in patients with aortic aneurysms needing follow-up scans.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
List of Abbreviations.
Abbreviation | Definition |
|---|---|
CTA | computed tomography angiographies |
EVAR | endovascular aneurysm repair |
Gd | gadolinium |
HU | Hounsfield units |
iDose[i4]) | Iterative image reconstructions |
MRI | magnetic resonance imaging |
ROI | regions of interest |
SPCT | spectral detector CT |
MonoE | virtual monoenergetic |
MonoE40 | virtual monoenergetic images with 40 keV |
Introduction
There has been an increase in the incidence of aortic aneurysms both, in the thorax and the abdomen, as a result of an aging population, the introduction of screening programs, an increasing number of smokers and improved diagnostic tools [1]. Depending on the aneurysm diameter and the risk factors there are conservative therapeutic approaches such as risk factor modification and betablockers or surgical approaches through endovascular aneurysm repair (EVAR) [2, 3]. In both cases patients need regular follow up examinations through CT scans using iodine-based contrast agents [2]. Nevertheless, these patients may have contraindications to iodine-based contrast agents and not seldomly, especially in an aging population, patients present with an impaired kidney function due to application of high amounts of contrast agents during the EVAR procedure [4].
Gadolinium (Gd)-based contrast agents as used in magnetic resonance imaging (MRI) on the other hand show a more favorable safety profile. Previous studies have shown that compared to iodine-based contrast agents, Gd-based contrast agents cause substantial, albeit lower radiation attenuation [5, 6]. Additionally, severe allergic reactions are reported less often and there is less affection of kidney function when Gd-based contrast agents are applied [7]. Because of lower attenuation properties, older studies applied high doses of Gd (0.3 to 0.4 mmol/kg) to achieve adequate contrast [8]. A pilot-study by Nadjiri et al. showed that using spectral CT, Gd-based contrast agent thoracic angiography with clinical doses of Gd is technically feasible [9].
Spectral CT is capable of overcoming limitations of conventional CT by offering the possibility to extrapolate virtual monoenergetic (MonoE) data [10,11,12]. The novel technique to detect a multienergy spectrum is based on a dual-layer detector system [13, 14]: Lower energy photons are attenuated in the first layer so that the second layer detects a harder spectrum of the same radiation, which then results in the registration of two different energy spectra. These two spectra can be detected simultaneously and with the same amount of radiation exposure. Virtual monoenergetic images are generated from this spectral data. Using a phantom model and a dose of 0.5 mmol/kg bodyweight, Bongers et al. showed that virtual monoenergetic images with 40 keV (MonoE40), which are generated from this spectral data are especially favorable for Gd-based contrast agents [15]. Nadjiri et al. showed that using spectral CT and reconstruction of these monoenergetic images, Gd-based contrast agent angiography with clinical doses of Gd (0.2 mmol/kg bodyweight) is technically feasible in patients in a clinical off-label use [9]. Nevertheless, no study has proven the usability spectral CT imaging using Gd-based contrast agents in a well circumscribed disease or a specific application, respectively.
Therefore, the goal of this study was to evaluate feasibility of using spectral CT for angiography using Gd-based contrast agent in patients with aortic disease or post-interventional follow-up examinations after interventional aortic repair.
Material and methods (454)
Study poplulation Between October 2015 and December 2021 all consecutive patients with clinical indication for angiography of the aorta but contraindications for iodine-based contrast agents were included in this retrospective study. Indication for Gd-agent-based CTA was approved as an individual healing trial in interdisciplinary consensus in each subject for clinical reasons. All patients gave written informed consent for Gd-enhanced CTA in which potential complications and “off-label-use” of Gd was discussed. Study analysis was carried out retrospectively from the registry data. This study has been approved by the local ethics committee.
21 scans in 15 patients were included in this study. The majority of patients included were male (n = 13). Average age at scanning time was at 72.1 ± 9.3 years (IQR: 70.0–78.8 years; range: 55–85years). Two patients showed a severe allergoid reaction after application of an iodine-based contrast agent in a previous examination and therefore declined application of iodine. The other 13 patients had a severely reduced renal function with a GFR between 30 and 45 mL/min and declined application of an iodine-based contrast agent.
Computed tomography scans
A 64-slice single source dual-layer spectral CT system was used for imaging in all cases (IQon; Philips Healthcare). Slice thickness was set to 0.9 mm with a collimation of 0.625 mm. All images were acquired with 120 kVp using a spiral scanning technique. For each patient, 0.2 mmol/kg body weight of Gd-DOTA (Dotagraf, Berlis AG, Switzerland) was used, with a 1:1 saline dilution for bolus prolongation. For timing of the contrast phase the test-bolus technique was used: 10 mL of the prepared Gd-DOTA-saline solution mixture was applied without use of spectral data to detect the time point of peak attenuation. For the test bolus, conventional technique was used, as MonoE reconstruction is not yet available for this application. No additional delay was added. The contrast-enhanced scan was then obtained at an injection rate of 4 to 6mL/s of the above-mentioned dilution followed by a 50 mL of saline chaser bolus. Iterative image reconstructions (iDose[i4]) were obtained with 0.9 and 3 mm slice thickness using the XCA (smooth) kernel. All patients included received an enhanced scan of the thoracic or the abdominal aorta with at least about half of the abdomen / thorax additionally included in the field of view. A subset of patients also received a pre-contrast scan.
Post processing
After acquisition, multiplanar virtual MonoE40 reconstructions were calculated from the spectral CT data set using a dedicated postprocessing software (Philips Intellispace Portal 8.0; Philips Healthcare). To evaluate changes in extrapolated attenuation, regions of interest (ROI) were placed within the thoracic and abdominal aorta in pre- and post-contrast images (ROI > 20 mm2). Mean attenuation values were extracted from the ROIs in both the thoracic and abdominal aorta.
Statistical analysis
All statistical analyses were performed by F.G.G using the statistical package R version 3.2.4 (R Foundation for Statistical Computing, Vienna, Austria). Categorical variables are expressed as frequencies and percentages, continuous variables are expressed as mean ± standard deviation. The tested data have visually been evaluated for normal distribution. Two-sided t test was applied. A P-Value < 0.05 was considered statistically significant.
Results
Imaging and image quality
Twelve of the 21 scans were indicated as a follow-up examination after TEVAR for evaluation of the graft position or endoleakage. Nine patients were scanned due to an aortic aneurysm without any previous intervention. 15 scans included the thorax, abdomen and pelvis region, three scans the thorax only and three scans the abdomen and pelvis region. In 9 patients a previous scan of the same region using iodine-based contrast agent was available for comparison. All 21 cases showed a successful contrast-phase timing and all images were free from relevant motion artifacts. All Gd-angiographies were independently considered to be diagnostic by two different cardiovascular radiologists, each with more than 7 years of experience in reading cardiovascular cases. No disagreements on diagnostic quality occurred. Exemplary images of conventional and MonoE40 reconstructions are shown in Fig. 1.
Comparison of pre- and post-contrast images
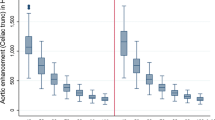
There was a significant increase in the intravascular attenuation of the aorta between pre- and post-contrast images for the MonoE40 images from 33.7 ± 15.6 to 162.3 ± 29.9 Hounsfield units (HU) in the thoracic aorta and from 45.1 ± 16.9 to 164.0 ± 35.9 HU in the abdominal aorta (p < 0.001 for both) as shown in Fig. 2.
The conventional images also showed a significant increase in attenuation between pre- and post-contrast images from 36.6 ± 5.8 to 84.4 ± 11.4 in the thoracic aorta and from 44.8 ± 10.9 to 81.6 ± 13.3 HU in the abdominal aorta (p < 0.001 for both).
Comparison of attenuation increase between conventional and MonoE40 images
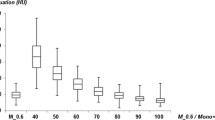
The ratio between pre- and post-contrast images was significantly higher in the MonoE40 images as compared to the conventional images with a factor of 2.4 ± 0.5 vs. 6.5 ± 4.5 in the thoracic aorta (p = 0.003) and 1.9 ± 0.5 vs. 4.1 ± 1.8 in the abdominal aorta (p < 0.001) as shown in Fig. 3.
The absolute difference in attenuation between pre- and post-contrast images was also significantly higher in the MonoE40 images as compared to the conventional images with 49.5 ± 13.3 vs. 134.6 ± 34.0 HU in the thoracic aorta and 39.1 ± 20.0 vs. 116.8 ± 40.7 HU in the abdominal aorta (p < 0.001 for both). Figure 4 shows a conventional and MonoE40 reconstruction of a Gd-enhanced CTA (arterial phase) comparing it to an iodine-enhanced CTA (arterial phase) of the same patient.
A shows a conventional reconstruction of a Gd-enhanced CTA (arterial phase); mean attenuation of the aorta is 73 HU. In B MonoE40 reconstruction of Gd-enhanced CTA demonstrates the aorta in arterial phase, mean attenuation of the aorta is 191 HU. C illustrates an iodine-enhanced CTA (arterial phase) of the same patient; mean attenuation of the aorta is 153HU.
Figure 5 shows example images of a post-procedural endoleak which is clearly visible in MonoE40 reconstructions and specific iodine maps, but cannot be discovered on the conventional images.
In this figure for image A-C windowing was identical (centre: 100 HU; widths: 600 HU). A shows a conventional reconstruction of a Gd-enhanced CTA (arterial phase); mean attenuation of the aorta is 95 HU. In B MonoE40 reconstruction of the aorta is demonstrated in a venous phase; mean attenuation of the aorta is 80 HU. C illustrates MonoE40 reconstruction of a Gd-enhanced CTA (arterial phase); mean attenuation of the aorta is 220 HU; the white arrow marks the early endoleak. In D a fusion of an arterial MonoE40 with a contrast dye map is shown; the white arrow marks the early endoleak [Note: The contrast dye map is an algorithm developed to highlight iodine. However, Gd can be isolated to a certain extent, too]
Discussion
This is the first study evaluating Gd-CTA utilizing dual energy for the specific diagnostic purpose of aortic assessment for treatment indications. The main findings are (i) the here proposed technique is feasible for a specific application namely diagnostic imaging of the aorta; (ii) MonoE40 reconstructions allow for a relevant and significant increase of attenuation and reach diagnostically recommended thresholds in the aorta; and (iii) Gd-CTA with dual layer CT allows for diagnostic results with high reliability.
Especially in an aging population the number of patients with contraindications to iodine based contrast agents is constantly increasing. High amounts of contrast agents during the EVAR procedure often decrease kidney function limiting the use of CTA for follow examinations [4]. As compared to other theoretical alternatives to iodine based contrast agents such as gold, gadolinium containing contrast agent seems to be a more suitable alternative, due to its broad experience and favorable safety profile in human use, including a lower nephrotoxic potential as well as less allergic reactions reported in literature [15,16,17]. Follow-up of patients with aortic aneurysms with and without EVAR requires CT scans, which in a routine clinical setting are performed using iodine based contrast agents.
For Gd-based examinations previous studies have shown a low contrast to noise ratio achieved at clinically reasonable contrast agent concentrations when using conventional CT imaging [15, 18]. Despite studies showing feasibility of gadolinium-based CTA in general, very high doses of gadolinium had to be administered to achieve a reasonable contrast and no technique was available to overcome this limitation [8, 18]. Studies by Zhang et al. in rabbits demonstrated a high accuracy for gadolinium-enhanced dual-energy CT pulmonary angiography to detect pulmonary embolism while Gabbai et al. reported high required intravenous doses of gadolinium contrast agent at 1.5 and 2.5 mmol/kg bodyweight [19, 20].
A study by Hamersvelt et al. has shown that dual-layer spectral detector CT (SDCT) allows for overall accurate quantification of gadolinium at both, 120 and 140 kVp in an anthropomorphic thoracic phantom, which is in line with results of this study, acquiring images at 120 kVp [21]. They have proven, that clinically encountered low concentrations of gadolinium, down to 0.5 mg/mL, can be accurately quantified using SDCT under these conditions.
To our knowledge, Nadjiri et al. were the first who were able to achieve a diagnostic attenuation in gadolinium-based CT angiography using MonoE-40 reconstructions at gadolinium concentrations of 0.2 mmol/kg bodyweight in humans [9]. MonoE40 reconstructions reach best results although this method is based on extrapolation of two energy spectra. Otherwise the maximum absorption would be expected at the k-edge of Gd which is 50.2 kEV [22]. However physical characteristics of atoms cannot be simulated by this method. Extrapolation further downwards might even yield better results for the here proposed method.
While the previous publication by Nadjiri et al. comprised several clinical indications including coronary and pulmonary angiography, the present study demonstrates the feasibility of gadolinium based angiography in a well circumscribed clinical application namely CTA of the aorta in patients with aortic aneurysms and follow up after aortic repair. In our clinic the technique has been accepted by clinical partners and is regularly requested for aortic imaging either for follow up after repair or for evaluation. The results from this technique are regarded as sufficient for the purpose by the referring vascular surgeons at our facility.
This study examines SDCT. As alimitation, results might not be transferable on other dual source techniques. While single-source rapid kilovoltage switching might be too slow regarding its offset between the scans dual source dual energy CT also might suffer due to its angular offset. Photon counting CT using dual contrast of iodine and gadolinium based contrast agents or gadolinium based contrast agents alone might become of interest in the future [23, 24].
As a further limitation, our retrospective study evaluates increase in attenuation between conventional and MonoE40 images only and does not evaluate the gold standard of iodine enhanced CTA in a head to head comparison. Future studies in e.g. animal models might consider a direct comparison between these two contrast agents.
Moreover, the use of Gd-based contrast agents in patients with renal failure carries a risk for the development of nephrogenic systemic fibrosis (NSF). According to recent findings, the risk of NSF in patients with Stage IV and V of chronic kidney disease with application of a group II Gd-based contrast agent is 0.07%, suggesting that the diagnostic benefit may often justify the risk [25]. However, each individual case must be evaluated.
To conclude, our study showed that Gd-CTA is a valid and reliable alternative for diagnostic imaging of the aorta for clinical applications. In case of contraindications for iodine-based contrast agent Gd-spectral-CTA should be considered whenever available.
Data availability
The data presented in this study are available upon reasonable request from the corresponding author.
References
Lederle FA, Johnson GR, Wilson SE, Chute EP, Hye RJ, Makaroun MS, Barone GW, Bandyk D, Moneta GL, Makhoul RG (2000) The aneurysm detection and management study screening program: validation cohort and final results. Aneurysm Detection and Management Veterans affairs Cooperative Study investigators. Arch Intern Med 160(10):1425–1430
Li B, Khan S, Salata K, Hussain MA, de Mestral C, Greco E, Aljabri BA, Forbes TL, Verma S, Al-Omran M (2019) A systematic review and meta-analysis of the long-term outcomes of endovascular versus open repair of abdominal aortic aneurysm. J Vasc Surg 70(3):954–969e930
Calero A, Illig KA (2016) Overview of aortic aneurysm management in the endovascular era. Semin Vasc Surg 29(1–2):3–17
Saratzis A, Bath MF, Harrison S, Sayers RD, Mahmood A, Sarafidis P, Bown MJ (2015) Long-term renal function after endovascular aneurysm repair. Clin J Am Soc Nephrol 10(11):1930–1936
Fraum TJ, Ludwig DR, Bashir MR, Fowler KJ (2017) Gadolinium-based contrast agents: a comprehensive risk assessment. J Magn Reson Imaging 46(2):338–353
Thomsen HS (2007) ESUR guideline: gadolinium-based contrast media and nephrogenic systemic fibrosis. Eur Radiol 17(10):2692–2696
Kane GC, Stanson AW, Kalnicka D, Rosenthal DW, Lee CU, Textor SC, Garovic VD (2008) Comparison between gadolinium and iodine contrast for percutaneous intervention in atherosclerotic renal artery stenosis: clinical outcomes. Nephrol Dial Transpl 23(4):1233–1240
Remy-Jardin M, Dequiedt P, Ertzbischoff O, Tillie-Leblond I, Bruzzi J, Duhamel A, Remy J (2005) Safety and effectiveness of gadolinium-enhanced multi-detector row spiral CT angiography of the chest: preliminary results in 37 patients with contraindications to iodinated contrast agents. Radiology 235(3):819–826
Nadjiri J, Pfeiffer D, Straeter AS, Noël PB, Fingerle A, Eckstein HH, Laugwitz KL, Rummeny EJ, Braren R, Rasper M (2018) Spectral computed tomography angiography with a Gadolinium-based contrast Agent: First Clinical Imaging results in Cardiovascular Applications. J Thorac Imaging 33(4):246–253
Grant KL, Flohr TG, Krauss B, Sedlmair M, Thomas C, Schmidt B (2014) Assessment of an advanced image-based technique to calculate virtual monoenergetic computed tomographic images from a dual-energy examination to improve contrast-to-noise ratio in examinations using iodinated contrast media. Invest Radiol 49(9):586–592
Lenga L, Albrecht MH, Othman AE, Martin SS, Leithner D, D’Angelo T, Arendt C, Scholtz JE, De Cecco CN, Schoepf UJ et al (2017) Monoenergetic Dual-energy computed Tomographic Imaging: cardiothoracic applications. J Thorac Imaging 32(3):151–158
Tabari A, Lo Gullo R, Murugan V, Otrakji A, Digumarthy S, Kalra M (2017) Recent advances in computed Tomographic Technology: cardiopulmonary imaging applications. J Thorac Imaging 32(2):89–100
McCollough CH, Leng S, Yu L, Fletcher JG (2015) Dual- and multi-energy CT: principles, Technical Approaches, and clinical applications. Radiology 276(3):637–653
Altman A, Carmi R (2009) TU-E‐210A‐03: a double‐layer detector, dual‐energy CT — principles, advantages and applications. Med Phys 36:2750–2750
Bongers MN, Schabel C, Krauss B, Claussen CD, Nikolaou K, Thomas C (2017) Potential of gadolinium as contrast material in second generation dual energy computed tomography - an ex vivo phantom study. Clin Imaging 43:74–79
Prince MR, Arnoldus C, Frisoli JK (1996) Nephrotoxicity of high-dose gadolinium compared with iodinated contrast. J Magn Reson Imaging 6(1):162–166
Wack C, Steger-Hartmann T, Mylecraine L, Hofmeister R (2012) Toxicological safety evaluation of gadobutrol. Invest Radiol 47(11):611–623
Bae KT, McDermott R, Gierada DS, Heiken JP, Nolte MA, Takahashi N, Hong C (2004) Gadolinium-enhanced computed tomography angiography in multi-detector row computed tomography: initial observations. Acad Radiol 11(1):61–68
Zhang LJ, Peng J, Wu SY, Yeh BM, Zhou CS, Lu GM (2010) Dual source dual-energy computed tomography of acute myocardial infarction: correlation with histopathologic findings in a canine model. Invest Radiol 45(6):290–297
Gabbai M, Leichter I, Mahgerefteh S, Sosna J (2015) Spectral material characterization with dual-energy CT: comparison of commercial and investigative technologies in phantoms. Acta Radiol 56(8):960–969
van Hamersvelt RW, Willemink MJ, de Jong PA, Milles J, Vlassenbroek A, Schilham AMR, Leiner T (2017) Feasibility and accuracy of dual-layer spectral detector computed tomography for quantification of gadolinium: a phantom study. Eur Radiol 27(9):3677–3686
Meng B, Cong W, Xi Y, De Man B, Wang G (2016) Energy Window optimization for X-Ray K-Edge Tomographic Imaging. IEEE Trans Biomed Eng 63(8):1623–1630
Ren L, Huber N, Rajendran K, Fletcher JG, McCollough CH, Yu L (2022) Dual-contrast Biphasic Liver Imaging with Iodine and Gadolinium using photon-counting detector computed Tomography: an exploratory animal study. Invest Radiol 57(2):122–129
Noda Y, Goshima S, Nakashima Y, Miyoshi T, Kawai N, Kambadakone A, Matsuo M (2020) Iodine dose optimization in portal venous phase virtual monochromatic images of the abdomen: prospective study on rapid kVp switching dual energy CT. Eur J Radiol 122:108746
Woolen SA, Shankar PR, Gagnier JJ, MacEachern MP, Singer L, Davenport MS (2020) Risk of nephrogenic systemic fibrosis in patients with stage 4 or 5 chronic kidney Disease receiving a Group II Gadolinium-based contrast Agent: a systematic review and Meta-analysis. JAMA Intern Med 180(2):223–230
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
The manuscript was conceptualized by F.G.G. and M.G., with the methodology developed by J.N. Software was provided by F.G.G. and M.G., while verification was performed by J.N., M.R.M., and M.K. F.G.G. performed the in-depth analysis, and the investigation was performed by M.G., S.Z., and A.W.M. Resources were provided by M.R.M., and data management was managed by M.G. and F.G.G, The original draft was prepared by F.G.G. and M.G., and the manuscript was revised and edited by J.N., A.W.M., F.T.G., M.R.M., S.Z., and M.K. Visualization was contributed by F.G.G., and the project was supervised by J.N., with project management by M.G. All authors read and approved the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Graf, M., Gassert, F.G., Marka, A.W. et al. Spectral computed tomography angiography using a gadolinium-based contrast agent for imaging of pathologies of the aorta. Int J Cardiovasc Imaging 40, 1059–1066 (2024). https://doi.org/10.1007/s10554-024-03074-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-024-03074-2