Abstract
To investigate the correlation the correlation between residual cholesterol (RC) and increased carotid intima-media thickness(cIMT) in non-diabetic individuals. This study included 1786 non-diabetic individuals who underwent carotid ultrasound. RC was calculated based on total cholesterol (TC), LDL-C, and high density lipoprotein cholesterol (HDL-C). The subjects were divided into the cIMT thickening group (cIMT ≥ 0.1 cm) and non-thickening group (cIMT < 0.1 cm) groups based on cIMT, binary logistic regression with different models and receiver operating characteristic (ROC) curves were adopted to evaluate the predictive ability of RC in cIMT. Of the research participants , their median age was 55 (49–51) years, 1121 (63%) were male, and 209 (12%) had hypertension, and people in the cIMT thickening group (925) were more likely to be older and male than those in the non-thickening group (843). Across the different RC subgroups, there was an increasing trend in maximum cIMT (P < 0.001) as RC levels increased within quartiles. RC was found to be an independent risk predictor for cIMT thickening (all P < in models 1–3); and this result persisted in the LDL-C normal subgroup (P = 0.002). The results suggested that RC was an independent predictor of cIMT thickening in non-diabetic individuals and had a strong atherogenic effect.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
LDL-C is known to be a major contributor to AS and the target of current treatment and primary prevention strategies for atherosclerotic cardiovascular disease (ASCVD) [1, 2]. Analyses have shown that individuals receiving statins have a significant residual cardiovascular risk, even those with normal LDL-C levels [3]; however, some studies have suggested that adding medication targeting triglycerides (TG) and remnant cholesterol (RC) to statin therapy may be more effective [3,4,5]. Recent epidemiological studies have shown that elevated RC, an independent factor in the development of cardiovascular events, is associated with ASCVD risk. RC has better ability to predict cardiovascular disease risk compared to LDL-C [6,7,8,9,10,11].
RC is the cholesterol content of TG-rich lipoproteins, including intermediate-density lipoproteins (IDLs), very low-density lipoproteins (VLDLs), and remnants of celiac particles in the non-fasting state. Quispe et al. [6] reported that residual particles have similar atherogenic capacity to LDLs but carry 40 times more cholesterol than VLDLs and can pass through the endothelium. The unique physicochemical characteristics of remnant particles make them highly atherogenic and important risk factors of cardiovascular events.
Carotid arteries are a high−prevalence site for atherosclerosis, and increased cIMT and carotid plaques, especially unstable atherosclerotic plaques, are high-risk factors for ischemic stroke [12, 13]. cIMT is a relatively simple measure of large artery atherosclerosis and is widely used to assess cardiovascular events due to its noninvasive and convenient nature [14]. Many researchers have analyzed conventional lipid parameters such as LDL-C or TG in relation to cIMT thickening [15, 16]; however, there are few reports on the association between RC and cIMT thickening. Therefore, this work was performed to evaluate the predictive ability of RC in cIMT thickening in a non-diabetic population.
Methods
Study subjects
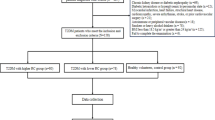
A amount of 2216 non-diabetic participants who underwent carotid ultrasound from January 2018 to October 2021 at the Drum Tower Hospital Affiliated with Nanjing University (Nanjing, Jiangsu,China) were initially enrolled in this research. All the study participants were screened, and patients were excluded if they were aged > 80 or < 18, had cancer or severe chronic illness, or had incomplete data. Patients with an incomplete clinical history (n = 234), missing fasting glucose levels (n = 36), and incomplete carotid ultrasound values (n = 178) were also excluded (Fig. 1). This study was approved by the Ethics Committee of Drum Tower Hospital Affiliated to Nanjing University (ethics code 2022-333-01, approval date 01-May-2022), all participants signed an informed consent to participate in the study.
Data collection and measurement
Basic clinical data were retrospectively collected from the non-diabetic population as follows: (1) demographic characteristics: sex and age; (2) past history: hypertension, coronary heart disease (CAD), stroke; (3) laboratory tests: red blood cell distribution width (RDW), fasting glucose, TC, TG, LDL-C and HDL-C; fasting glucose, TC, TG, LDL-C, and HDL-C levels were assayed with an automated biochemical analyzer, while RDW was measured with an automated hematology analyzer. There is no standard assay for RC; thus, a formula commonly used in previous studies was used to calculate RC values (RC = TC-LDL-HDL) [17, 18]. The atherosclerosis index (AI), a non-conventional lipid parameter, was also calculated (AI = non-HDLC/HDL-C).
Carotid intima-media thickness measurement
All participants underwent carotid Doppler scanning(Philips HD5G, GE, USA, 5 ~ 12 MHz), in which the ultrasound probe was moved cephalad from the root of the neck in a transverse sweep, assessing the distal common carotid artery, the carotid bifurcation, and the proximal internal carotid artery, respectively. Multi-point and multiple measurements were performed on both sides of the carotid intima, and the maximum and average values of the left and right sides were calculated for subsequent analysis (Fig. 2). Two qualified physicians in the ultrasound department performed the carotid ultrasound examinations and reported the results. cIMT ≥ 1 mm was defined as thickening [19].
Other covariates
Hypertension was defined as systolic blood pressure (BP) ≥ 140 mmHg or diastolic BP ≥ 90 measured three times on different days, self-reported physician diagnosis or taking blood pressure-lowering medication [20]. Diabetes was diagnosed as a fasting blood glucose ≥ 11.1 mmol/l or non-fasting blood glucose ≥ 7.0 mmol/l, a reported history of diabetes or taking glucose-lowering medication [21]. CAD referred to a heart disease caused by ischemia and hypoxia of the myocardium due to atherosclerosis of the coronary arteries [22]. It is generally believed that ischemic stroke is caused by severe narrowing or occlusion of cerebral blood vessels due to impaired cerebral blood circulation and consequent ischemia and hypoxia leading to brain tissue necrosis in the blood supply area, often confirmed by cranial CT or MRI images [23].
Statistical analysis
The enrolled population was grouped according to the cIMT as well as RC quartiles, respectively. The measurement data were expressed as the mean ± standard deviation (x̄ ± S), and the count data were expressed as frequency and percentage (%); independent samples t-tests or chi-square (χ2) tests were used for comparison between the two groups. One-way ANOVA was adopt for comparisons between multiple groups; non-normally distributed measures were expressed in terms of medians and quartiles, and a non-parametric U test was used. The Spearman correlation between RC and each variable was determined, and the relationship between cIMT and each variable was analyzed by binary logistic regression. Multi-model logistic regression was used to analyze the predictive ability of RC for cIMT thickening and to calculate intergroup trends after correction for confounders. The patients with normal LDL-C levels were screened and grouped according to RC quartiles, and the subgroups were evaluated using ANOVA to explore whether the observed correlation was independent of LDL-C.
Results
Baseline characteristics of participants according to cIMT
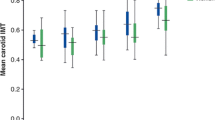
Among the 2216 non-diabetic individuals who underwent carotid ultrasound, 1786 were selected in the ultimate analysis. Of these individuals, 925 in the cIMT thickening group and 843 in the non-thickening group. Figure 1 showed the specific screening process, while Table 1 depicted the baseline characteristics of the subjects. Of the research participants, their median age was 55 (49–51) years, 1121 (63%) were male, and 209 (12%) had hypertension. Those with cIMT ≥ 0.1 cm were in the cIMT thickening group, and those with patients had higher fasting glucose, systolic BP, LDL-C, TC, TG, non-HDL, and RC levels and lower HDL-C levels (Table 1; Fig. 3).
Baseline characteristics of participants according to RC quartiles
The 1786 non-diabetic individuals were divided by RC quartiles, 427 in group Q1, 437 in group Q2, 456 in group Q3, and 443 in group Q4. The comparison of the baseline characteristics among the groups showed that patients with higher RC levels were more possible to be older and male than those with lower RC levels; those with high RC levels also had higher LDL-C, TC, TG, non-HDL-C, fasting glucose, and BP levels. The prevalence of hypertension, CAD, and stroke was mostly balanced among the four groups. There was an increasing trend in maximum cIMT (P < 0.001) as RC levels increased within quartiles (Table 2).
Correlation analysis and logistic regression
The Spearman’s correlation analysis showed that RC was positively correlated with age,fasting glucose, RDW, BP parameters, TG, TC, LDL-C, non-HDL-C, and AI levels and negatively correlated to HDL-C levels (Table 3). Intergroup trends in RC quartiles were assessed by calculating the median within each quartile. Three models (models 1−3) were developed, including statistically significant covariates and clinically significant models to evaluate the predictive ability of RC in cIMT thickening. cIMT increased over the interquartile range of RC as the variables in the models were continuously adjusted. In model 1, the OR for the highest quartile of RC was 2.175 (95% CI = 1.593−2.969, P < 0.001) compared with the lowest quartile. The adjustment of model 2 did not change anything significantly. In model 3, with further adjustment for statistically significant variables and exclusion of confounders affecting RC, the corresponding ORs for the highest and lowest quartiles of RC were 2.376 (95% CI = 1.508−3.745) and 1.658 (95% CI = 1.18−2.33). Additionally, RC was entered as a categorical variable into the model for analysis, and P-values were obtained for the 3 comparisons. Larger RC values were an independent risk predictor for cIMT (P < 0.001) (Table 4). Further research was applied to explore the correlation between cIMT and each variable; the results revealed RC level (OR = 1.466, CI = 1.149−1.87, P = 0.002) to be an independent risk factor in cIMT thickening (Fig. 4).
Logistic regression analysis evaluating predictive implication of RC in various stratifications. Abbreviations: HDL-C high-density lipoprotein cholesterol,LDL-C low-density lipoprotein cholesterol,RC remnant cholesterol,TC total cholesterol,TG triglycerides,BP blood pressure,AI atherosclerosis index,CI=confidence interval
Analysis after LDL-C adjustment
Notably, RC was still related to cIMT thickening after adjustment for LDL-C values. People with normal levels of LDL-C were selected into the subgroup analysis and grouped by RC quartiles to analyze the differences in cIMT among these groups. The RC quartiles were calculated as 0.43, 0.58, and 0.78 mmol/l, respectively, and evaluated using ANOVA. The distribution of Max-cIMT was significantly different between the four groups (F = 4.805, P = 0.002). Tukey’s test showed that the mean Max-cIMT score increased by 0.006 and 0.008 (95% CI: 0.0002−0.012, 0.0024−0.014) from group Q1 to group Q3, respectively, and the difference was statistically significant (P = 0.04, P = 0.002; Table 5).
ROC Curves.
Figure 5 showed the comparisons of RC and other common lipid parameters predicting cIMT thickening (cIMT ≥ 1.0 mm). The ROC results showed the AUC of RC is the largest (AUC = 0.586), revealing that RC has better ability to predict cIMT compared to conventional lipid parameters such as LDL-C or TG.

Fig. 5 ROC curve evaluating predictive effect of RC and other non-conventional lipid parameters for cIMT thickening. (a) RC versus Non-HDL-C or TG; (b) RC versus LDL-C or TC. Abbreviations: HDL-C high-density lipoprotein cholesterol,LDL-C low-density lipoprotein cholesterol,RC remnant cholesterol,TC total cholesterol,TG triglycerides
Discussion
This retrospective study of 1786 non-diabetic participants explored RC’s predictive ability in cIMT thickening. RC correlated with cIMT, a subclinical marker of AS. Higher RC levels were found to have stronger predictive power for cIMT thickening; additionally, RC was related with many risk factors for cardiovascular diseases. This correlation persisted in subgroups with normal LDL-C levels. The findings suggest that RC is a good predictor of cIMT thickening events in non-diabetic populations.
Numerous studies have shown that AS is a causative factor and an important predictor of ASCVD events [24, 25]. Percutaneous angiography is the currently accepted standard for the diagnosis of AS, but it is not suitable for clinical application because of its invasiveness and high cost; therefore, we recommend cIMT measurement as a more noninvasive and convenient index for AS evaluation. cIMT is a widely used subclinical indicator of atherosclerosis worldwide and can be measured simply, non-invasively, and reproducibly by ultrasound. Several studies have revealed cIMT measurements to be of great value for assessing the risk of cardiovascular disease events; thus, cIMT was chosen in this study [26,27,28].
The present study were in agreement with the previous researches that have shown RC to be independently connected with cardiovascular events and a significant predictor of ASCVD events [29,30,31]. An analysis of the PREDIMED trial indicated that TG and RC (but not LDL-C) levels were independently related to cardiovascular events in a Mediterranean population with a high prevalence of diabetes and obesity [3]. The Copenhagen studies reported an relationship between RC levels and an increased risk of ischemic stroke, with atherosclerosis as the main presumed mechanism [10]. Similarly, Lin et al. [32] found a obvious correlation between RC levels and total coronary atherosclerotic burden, which persisted in normal LDL levels, independent of HDL-C and traditional ASCVD risk factors. The study provides stronger evidence for the atherogenicity of RC by analyzing the relationship between RC and CIMT, which is subclinical evidence of atherosclerosis.
RC, the cholesterol content of all TG-rich lipoproteins, consists of VLDLs, IDLs, and chylomicron residues in the non-fasting state. Some TG-rich lipoproteins are cholesterol-rich in circulation due to delayed metabolism and are highly atherosclerotic [3, 33]. The exact mechanism by which RC causes atherosclerosis is unknown, and several potential mechanisms exist. One of the possible explanations is that residual lipoproteins can readily penetrate the arterial wall [34, 35], which is necessary for atherosclerosis to occur. Remnant lipoproteins are readily retained after entering the arterial intima and can be absorbed by macrophages without oxidative metabolism [36]. This accumulation could enhance plaque formation. Another possible explanation is that elevated RC is related to inflammation and is another mechanism that promotes atherosclerosis. One study revealed increased expression of integrins on the surface of pro-inflammatory monocytes in participants with elevated RC levels, which induces foam cell production and promotes atherosclerosis [37]. Nordestgaard et al. [35] found 37% increase C-reactive protein per 1 mmol/l increase in RC levels and a causal association with 28% higher levels, suggesting a causal relationship between elevated RC and low-grade inflammation. In addition, remnant lipoproteins are hydrolyzed by lipoprotein lipase, and their hydrolysis products induce the formation of cytokines (TNF-α, IL-6, IL-8) and cell adhesion molecules; the upregulation of pro-inflammatory factors causes a cascade of inflammatory responses. RC could also activate some relevant cellular pathways. Some remnant lipoproteins upregulate the expression of ICAM-1, TF, and VCAM-1 (preatherogenic thrombogenic molecules) in vascular endothelial cells through a redox mechanism [38] and induce the formation of superoxide, leading to endothelial cell apoptosis [39]. All these factors play major roles in atherogenesis. TG-rich lipoproteins are believed to activate platelets and coagulation pathways and support the assembly of coagulation complexes, amplifying the coagulation cascade and, ultimately, dysregulating coagulation and impairing fibrinolysis, predisposing patients to atherosclerotic embolism [40].
LDL-C is the most frequently analyzed lipid parameter and is currently the main clinical target for treating patients in danger of dyslipidemia and cardiovascular events. However, a significant residual cardiovascular risk exists in individuals receiving statins, even those with optimal LDL-C levels [3]. The study are associated with the aforementioned observation. Würtz et al. [41] found that statin treatment is relevant to significant reductions in IDL and VLDL particle concentrations and that lowering RC or TG was more beneficial in patients eligible for statin treatment [42]. Meanwhile, some studies have shown that strengthening lipid-lowering treatment for patients with high RC levels can significantly lower the incidence of ASCVD events [43]; thus, RC is a potential future treatment target. Large randomized clinical trials are still lacking, and future studies require substantial clinical data to show that treatments that lower RC levels are more effective.
The research has several differences from other studies. First, previous studies usually pay attention to the relationships between RC and cardiovascular events, while the scope of present study was narrowed to focus on the predictive ability of RC in cIMT thickening. Second, while many studies have explored the risk of cardiovascular events with conventional lipid markers such as LDL-C or TG, this study focused on RC, a non-conventional assessment. Finally, this study was conducted in a non-diabetic population, eliminating major confounders and improving the reliability of the results. This work better explained the main cause of cardiovascular disease, namely the atherogenic effect of RC, which has been shown in numerous studies to be an important risk factor for ASCVD events.
This retrospective study provides strong evidence for the correlation between RC and cIMT thickening in the non-diabetic individuals. However, limitations should also be noted. First, the subjects in this research were few, and more data are still being collected. Second, as a retrospective study, the sample included in the analysis was non-randomized, which impacts on the wider application of our results. Third, the team needs to explore the relationship between RC and cIMT thickening through prospective studies with larger samples. Finally, the use of lipid-lowering medications in the study population was not considered, which may impact the results.
There is no standard testing methods for RC, and it is recognized to use lipid calculations to obtain residual cholesterol values at no additional cost, which is expected to be widely used in clinical practice in the future. We report a new relationship between RC and increased carotid intima-media thickness in non-diabetic individuals. This population-based finding provides a better explanation for remnant cholesterol-induced cIMT thickening, that is, RC has a strong atherogenic capacity, while our study has clinical implications in exploring ASCVD events.
Conclusions
The results suggested that RC was an independent predictor of cIMT thickening in non-diabetic individuals and had a strong atherogenic effect. This result expanded the field of assessing cIMT indicators, and highlighted RC as a new potential therapeutic target. In clinical practice, patients with normal conventional lipid indices could be monitored for RC, and targeted therapy for patients with high RC could prevent cardiovascular events; however, the findings need to be confirmed in prospective studies and clinical trials. It is expected that RC is included in routine lipid screens in the future and that targeted treatments will be developed for future clinical applications.
Abbreviations
- AI:
-
Atherosclerosis index
- AS:
-
Atherosclerosis
- ASCVD:
-
Atherosclerotic cardiovascular disease
- AUC:
-
Area under the curve
- BP:
-
Blood pressure
- CAD:
-
Coronary heart disease
- cIMT:
-
Carotid intima-media thickness
- HDL-C:
-
High-density lipoprotein cholesterol
- IDLs:
-
Intermediate-density lipoproteins
- LDL-C:
-
Low-density lipoprotein cholesterol
- RC:
-
Remnant cholesterol
- RDW:
-
Red cell distribution width
- ROC:
-
Receiver operating characteristic
- TC:
-
Total cholesterol
- TG:
-
Triglycerides
- VLDLs:
-
Very low-density lipoproteins
References
Grundy SM, Stone NJ, Bailey AL, Beam C, Birtcher KK, Blumenthal RS, AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA et al (2018) /ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;139(25):e1082-e143
Herrington W, Lacey B, Sherliker P, Armitage J, Lewington S (2016) Epidemiology of atherosclerosis and the potential to reduce the global burden of Atherothrombotic Disease. Circul Res 118(4):535–546
Castañer O, Pintó X, Subirana I, Amor AJ, Ros E, Hernáez Á et al (2020) Remnant cholesterol, not LDL cholesterol, is Associated With Incident Cardiovascular Disease. J Am Coll Cardiol 76(23):2712–2724
Silverman MG, Ference BA, Im K, Wiviott SD, Giugliano RP, Grundy SM et al (2016) Association between lowering LDL-C and Cardiovascular Risk Reduction among different therapeutic interventions: a systematic review and Meta-analysis. JAMA 316(12):1289–1297
Amarenco P, Labreuche J (2009) Lipid management in the prevention of stroke: review and updated meta-analysis of statins for stroke prevention. Lancet Neurol 8(5):453–463
Quispe R, Martin SS, Michos ED, Lamba I, Blumenthal RS, Saeed A et al (2021) Remnant cholesterol predicts cardiovascular disease beyond LDL and ApoB: a primary prevention study. Eur Heart J 42(42):4324–4332
Varbo A, Benn M, Tybjærg-Hansen A, Jørgensen AB, Frikke-Schmidt R, Nordestgaard BG (2013) Remnant cholesterol as a causal risk factor for ischemic heart disease. J Am Coll Cardiol 61(4):427–436
Kexin W, Yaodong D, Wen G, Rui W, Jiaxin Y, Xiaoli L et al (2021) Association of increased remnant cholesterol and the risk of coronary artery disease: a retrospective study. Front Cardiovasc Med 8:740596
Langsted A, Madsen CM, Nordestgaard BG (2020) Contribution of remnant cholesterol to cardiovascular risk. J Intern Med 288(1):116–127
Varbo A, Nordestgaard BG (2019) Remnant cholesterol and risk of ischemic stroke in 112,512 individuals from the general population. Ann Neurol 85(4):550–559
Qian S, You S, Sun Y, Wu Q, Wang X, Tang W et al (2021) Remnant cholesterol and common carotid artery intima-media thickness in patients with ischemic stroke. Circulation Cardiovasc Imaging 14(4):e010953
Bos D, Arshi B, van den Bouwhuijsen QJA, Ikram MK, Selwaness M, Vernooij MW et al (2021) Atherosclerotic carotid plaque composition and incident stroke and coronary events. J Am Coll Cardiol 77(11):1426–1435
Cao X, Yang Q, Tang Y, Pan L, Lai M, Yu Z et al (2020) Normalized wall index, intraplaque hemorrhage and ulceration of carotid plaques correlate with the severity of ischemic stroke. Atherosclerosis 315:138–144
SahBandar IN, Ndhlovu LC, Saiki K, Kohorn LB, Peterson MM, D’Antoni ML et al (2020) Relationship between circulating inflammatory Monocytes and Cardiovascular Disease Measures of Carotid Intimal Thickness. J Atheroscler Thromb 27(5):441–448
Fernández-Friera L, Fuster V, López-Melgar B, Oliva B, García-Ruiz JM, Mendiguren J et al (2017) Normal LDL-Cholesterol levels are Associated with subclinical atherosclerosis in the absence of risk factors. J Am Coll Cardiol 70(24):2979–2991
Raposeiras-Roubin S, Rosselló X, Oliva B, Fernández-Friera L, Mendiguren JM, Andrés V et al (2021) Triglycerides and residual atherosclerotic risk. J Am Coll Cardiol 77(24):3031–3041
Nordestgaard BG, Benn M, Schnohr P, Tybjaerg-Hansen A (2007) Nonfasting triglycerides and risk of myocardial infarction, ischemic heart disease, and death in men and women. JAMA 298(3):299–308
Joshi PH, Khokhar AA, Massaro JM, Lirette ST, Griswold ME, Martin SS et al (2016) Remnant lipoprotein cholesterol and Incident Coronary Heart Disease: the Jackson Heart and Framingham offspring Cohort Studies. J Am Heart Association. ;5(5)
Bauer M, Caviezel S, Teynor A, Erbel R, Mahabadi AA, Schmidt-Trucksäss A (2012) Carotid intima-media thickness as a biomarker of subclinical atherosclerosis. Swiss Med Wkly 142:w13705
Wang Z, Chen Z, Zhang L, Wang X, Hao G, Zhang Z et al (2018) Status of hypertension in China: results from the China Hypertension Survey, 2012–2015. Circulation 137(22):2344–2356
Williams B, Mancia G, Spiering W, Agabiti Rosei E, Azizi M, Burnier M et al (2018) 2018 ESC/ESH guidelines for the management of arterial hypertension. Eur Heart J 39(33):3021–3104
Kim HL, Lim WH, Seo JB, Kim SH, Zo ZH, Kim MA (2020) Prediction of cardiovascular events using brachial-ankle pulse wave velocity in hypertensive patients. J Clin Hypertens (Greenwich Conn) 22(9):1659–1665
Feske SK (2021) Ischemic stroke. Am J Med 134(12):1457–1464
Weber C, Noels H (2011) Atherosclerosis: current pathogenesis and therapeutic options. Nat Med 17(11):1410–1422
Frostegård J (2013) Immunity, atherosclerosis and cardiovascular disease. BMC Med 11:117
Nezu T, Hosomi N, Aoki S, Matsumoto M (2016) Carotid intima-media thickness for atherosclerosis. J Atheroscler Thromb 23(1):18–31
Sun P, Liu L, Liu C, Zhang Y, Yang Y, Qin X et al (2020) Carotid intima-media thickness and the risk of first stroke in patients with hypertension. Stroke 51(2):379–386
Parish S, Arnold M, Clarke R, Du H, Wan E, Kurmi O et al (2019) Assessment of the role of Carotid Atherosclerosis in the Association between Major Cardiovascular Risk factors and ischemic stroke subtypes. JAMA Netw open 2(5):e194873
Varbo A, Freiberg JJ, Nordestgaard BG (2015) Extreme nonfasting remnant cholesterol vs extreme LDL cholesterol as contributors to cardiovascular disease and all-cause mortality in 90000 individuals from the general population. Clin Chem 61(3):533–543
Jepsen AM, Langsted A, Varbo A, Bang LE, Kamstrup PR, Nordestgaard BG (2016) Increased remnant cholesterol explains part of residual risk of all-cause mortality in 5414 patients with ischemic heart disease. Clin Chem 62(4):593–604
Jørgensen AB, Frikke-Schmidt R, West AS, Grande P, Nordestgaard BG, Tybjærg-Hansen A (2013) Genetically elevated non-fasting triglycerides and calculated remnant cholesterol as causal risk factors for myocardial infarction. Eur Heart J 34(24):1826–1833
Lin A, Nerlekar N, Rajagopalan A, Yuvaraj J, Modi R, Mirzaee S et al (2019) Remnant cholesterol and coronary atherosclerotic plaque burden assessed by computed tomography coronary angiography. Atherosclerosis 284:24–30
Cao YX, Zhang HW, Jin JL, Liu HH, Zhang Y, Xu RX et al (2020) Prognostic utility of triglyceride-rich lipoprotein-related markers in patients with coronary artery disease. J Lipid Res 61(9):1254–1262
Varbo A, Nordestgaard BG (2017) Remnant lipoproteins. Curr Opin Lipidol 28(4):300–307
Nordestgaard BG (2016) Triglyceride-Rich Lipoproteins and Atherosclerotic Cardiovascular Disease: New Insights from Epidemiology, Genetics, and Biology. Circul Res 118(4):547–563
Varbo A, Benn M, Tybjærg-Hansen A, Nordestgaard BG (2013) Elevated remnant cholesterol causes both low-grade inflammation and ischemic heart disease, whereas elevated low-density lipoprotein cholesterol causes ischemic heart disease without inflammation. Circulation 128(12):1298–1309
Bernelot Moens SJ, Verweij SL, Schnitzler JG, Stiekema LCA, Bos M, Langsted A et al (2017) Remnant cholesterol elicits arterial wall inflammation and a Multilevel Cellular Immune response in humans. Arteriosclerosis, thrombosis, and vascular biology. 37(5):969–975
Doi H, Kugiyama K, Oka H, Sugiyama S, Ogata N, Koide SI et al (2000) Remnant lipoproteins induce proatherothrombogenic molecules in endothelial cells through a redox-sensitive mechanism. Circulation 102(6):670–676
Shin HK, Kim YK, Kim KY, Lee JH, Hong KW (2004) Remnant lipoprotein particles induce apoptosis in endothelial cells by NAD(P)H oxidase-mediated production of superoxide and cytokines via lectin-like oxidized low-density lipoprotein receptor-1 activation: prevention by cilostazol. Circulation 109(8):1022–1028
Olufadi R, Byrne CD (2006) Effects of VLDL and remnant particles on platelets. Pathophysiol Haemost Thromb 35(3–4):281–291
Würtz P, Wang Q, Soininen P, Kangas AJ, Fatemifar G, Tynkkynen T et al (2016) Metabolomic profiling of statin use and genetic inhibition of HMG-CoA reductase. J Am Coll Cardiol 67(10):1200–1210
Schwartz GG, Abt M, Bao W, DeMicco D, Kallend D, Miller M et al (2015) Fasting triglycerides predict recurrent ischemic events in patients with acute coronary syndrome treated with statins. J Am Coll Cardiol 65(21):2267–2275
Vallejo-Vaz AJ, Fayyad R, Boekholdt SM, Hovingh GK, Kastelein JJ, Melamed S et al (2018) Triglyceride-rich lipoprotein cholesterol and risk of Cardiovascular events among patients receiving statin therapy in the TNT Trial. Circulation 138(8):770–781
Acknowledgements
We thank all participants and clinical staff for their support and contributions to this study.We thank LetPub (www.letpub.com) for its linguistic assistance during the preparation of this manuscript.
Funding
This work was funded by the National Natural Science Foundation of China (81571122), and the Health Institute of Nanjing (ZKX19013, ZKX21025).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Dujuan Sha designed and drafted the manuscript. Material preparation, data collection and analysis were performed by Xiaoqiong Du, Jie Ding, Xinchen Ma, Ruijie Yang and Luna Wang.The first draft of the manuscript was written by Xiaoqiong Du and all authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of Drum Tower Hospital Affiliated to Nanjing University (ethics code 2022-333-01, approval date 01-May-2022), all participants signed an informed consent form.
Consent for publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Du, X., Ding, J., Ma, X. et al. Remnant cholesterol has an important impact on increased carotid intima-media thickness in non-diabetic individuals. Int J Cardiovasc Imaging 39, 2487–2496 (2023). https://doi.org/10.1007/s10554-023-02957-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-023-02957-0