Abstract
Left ventricular (LV) function undergoes subtle changes (subclinical left ventricular dysfunction) in a large proportion of patients with type 2 diabetes (T2DM) who develop diabetic cardiomyopathy. This study aimed to quantify LV myocardial strain and synchrony in T2DM by real-time three-dimensional echocardiography (RT-3DE), and to evaluate subclinical LV dysfunction in T2DM at different glycemic control levels. Seventy-two patients with T2DM with an LV ejection fraction (LVEF) ≥ 55% and 45 healthy individuals as controls who underwent RT-3DE were studied. Patients were also subdivided into the DMa group (glycosylated hemoglobin < 7%, n = 38) and the DMb group (glycosylated hemoglobin ≥ 7%, n = 34). Three-dimensional strain and synchronization parameters of the left ventricle were measured by RT-3DE and compared among the three groups. Despite a similar LVEF, global longitudinal strain (GLS), global circumferential strain (GCS), and global area strain (GAS) in the DMb group were lower, and the standard deviation of peak time (Tm-SD) and the maximum difference in peak time (Tm-Dif) in the DMb group higher, than those in the control and DMa groups (all p < 0.05). Multivariable linear regression analysis showed that the duration of diabetes was independently associated with GCS (β = − 0.516, p < 0.001) and GAS (β = − 0.391, p = 0.005). HbA1c levels were independently associated with GLS (β = − 0.675, p < 0.001), Tm-SD (β = 3.363, p < 0.001), and Tm-Dif (β = 3.895, p < 0.001). RT-3DE can detect subclinical myocardial dysfunction in poor glycemic control of T2DM, and myocardial dysfunction is associated with the duration of diabetes and HbA1c.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Diabetes mellitus (DM) is a common risk factor for cardiovascular and cerebrovascular events [1]. The risk of cardiovascular disease in patients with DM is two to three times higher than that of healthy people, and nearly 80% of patients with DM die of cardiovascular complications [2]. Moreover, one of the main causes of increased mortality in DM is diabetic cardiomyopathy (DCM) [3, 4], which refers to cardiac dysfunction and heart failure in DM unrelated to coronary artery disease and hypertension [5]. A large proportion of patients with type 2 DM (T2DM) undergo subtle changes in cardiac function before the diagnosis of DCM. These changes include impaired left ventricular (LV) diastolic function and a reduction in myocardial perfusion, which is termed subclinical LV dysfunction (LVD) [6, 7]. Detection of T2DM subclinical LVD dysfunction at an early stage is important for preventing DCM.
Real-time three-dimensional echocardiography (RT-3DE) speckle-tracking imaging technology can track myocardial trajectory movement in 3D space and acquire 3D myocardial strain, which can be used to quantitatively analyze LV function [8, 9]. The sensitivity and specificity of this technique are significantly better than the traditional ejection fraction. Additionally, subclinical LVD (whereby myocardial strain has been reduced before the LV ejection fraction (LVEF) is preserved) can be determined by this technique [10, 11]. LV synchronous motion, which is an important mechanism, also affects LV function. The time for the systolic period of the left ventricle to reach minimum volume is consistent. When LV motion is not synchronized, the peak contraction time of each segment of the myocardium is different. The RT-3DE full-volume technique can comprehensively and quantitatively obtain the systolic peak time of the left ventricle. The standard deviation of the peak time (Tm-SD) and the maximum peak time difference (Tm-Dif) can then be used to evaluate LV systolic synchrony [12].
This study aimed to analyze LV myocardial strain and synchrony in T2DM patients with a normal LVEF by RT-3DE. We also aimed to apply these strain and synchrony parameters to assess subclinical LVD in T2DM with different degrees of glycemic control.
Methods
Subjects
In this prospective study we recruited 72 patients (age 30–64 years) with T2DM and an LVEF ≥ 55%, which is in line with the 2010 American Diabetes Guidelines diagnostic criteria [13]. According to the control level of glycated hemoglobin (HbA1c), we divided the patients into the two following groups. The DMa group comprised 38 patients with HbA1c levels < 7% (good glycemic control) and the DMb group comprised 34 patients with HbA1c levels ≥ 7% (poor glycemic control) [13]. The duration of diabetes was recorded as reported by the patient. For comparison, 45 age- and sex-matched healthy volunteers with no history of cardiac disease or DM were recruited from the local medical center (control group). The height and weight of all subjects were measured and body mass index was calculated as weight (kg) divided by the square of height (m). Blood pressure was measured in the right arm in a sitting position.
We excluded patients who had hypertension (systolic blood pressure ≥ 140 mmHg), coronary heart disease, heart valve disease, myocardial infarction, cardiomyopathy, severe renal failure (estimated glomerular filtration rate < 30 ml/min), or poor echocardiographic image quality.
All subjects underwent echocardiography, electrocardiography, and biochemical examinations. Our Institutional Review Board approved the study. Informed consent was obtained from all patients.
Laboratory analysis
Biochemical analyses, including measurement of total cholesterol, triglyceride, low-density lipoprotein, high-density lipoprotein, and fasting plasma glucose levels, were performed using standard laboratory techniques in all subjects. In patients with T2DM, HbA1c was measured < 2 weeks before the echocardiographic evaluation.
Echocardiography
Echocardiographic imaging was performed using the Vivid E9 GE Medical Systems commercial scanner (GE Vingmed Ultrasound, Norway), equipped with 5S probe (1–5 MHz) and 4 V probe (1.5–4.0 MHz). The subjects lay on their left side and breathed calmly, and electrocardiograms were recorded simultaneously during the examinations. Two-dimensional (2D) echocardiography with the 5S probe was used to determine the left ventricular mass index (LVMI), early and late mitral valvular blood flow velocity peak (E, A), and LV sidewall mitral annular early peak velocity (Em), then E/A and E/Em were calculated. The LVEF was measured by the Simpson biplane method. We obtained the cardiac isovolumic contraction time (ICT), isovolumic relaxation time (IRT), and ejection time (ET). We then calculated the Tei index as follows:
RT-3DE with the 4 V probe was used to obtain the apical four-chamber view and then entered 4D mode to record the subject's six consecutive cardiac cycles during a breath-hold. The “store” button was then used to store 3D dynamics. It was necessary to avoid occasional premature beats when collecting images with the six consecutive cardiac cycles in sinus rhythm. The images required a heart rate with a frame rate > 40%. The range of the imaging frame rate in this study was 25–49.
Echocardiographic image analysis
Echo PAC analysis software (version: 201) was used for analysis of echocardiographic images. We selected the midpoint of the mitral annulus and the apical endocardium at the end of diastole and end-systole, respectively. The system automatically delineates the endocardium and epicardium and is manually adjusted to correctly track the myocardium. The software automatically calculates 3D myocardial strain and 16-segment peak contraction time of the left ventricle. If ≤ 2 segments of the left ventricle cannot be analyzed, we click the "approve" button in the software, accept the analysis results, and obtain the strain value. If the image quality is too poor for analysis, we do not accept it as valid.
Data from a total of 80 patients were initially collected for this study, 3 of whom were significantly obese and could not hold their breath and 2 of whom had atrial fibrillation, which resulted in failure to acquire 3D full-volume images. In addition, no data were available from 3 patients because of poor image quality. Finally, 72 patients were enrolled.
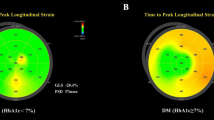
After this analysis, we obtained 3D general longitudinal strain (GLS), general circumferential strain (GCS), general radial strain (GRS), and general area strain (GAS) (Fig. 1). After standardization of the RR interval of the cardiac cycle, the Tm-SD and Tm-Dif were used as parameters for synchrony of LV wall contraction. Image storage and analysis were performed by experienced physicians.
Intra-observer and inter-observer reproducibility
Echocardiographic image analysis was repeated in 10 selected subjects. Intra-observer reliability assessment was performed 1 week apart by the same observer on the same echocardiographic images in a random order. Assessment of inter-observer reproducibility was carried out by another independent observer.
Statistical analysis
All statistical analyses were performed with SPSS 17.0 (SPSS, Chicago, IL, USA). Continuous data are presented as mean ± SD. Frequencies are expressed as percentages. The Student’s t-test or one-way analysis of variance with the post hoc least significant difference test was used as appropriate for comparison of continuous data among the three groups. The chi-square test was used for comparing the variable of sex. Pearson’s correlation was chosen for test correlations among parameters of the three groups. Independent determinants of subclinical LV dysfunction in T2DM were examined using multivariate stepwise linear regression. p values of < 0.05 were considered statistically significant.
The intraclass correlation coefficient (ICC) was used to evaluate inter- and intra-observer variability. Clinical significance was categorized as follows: good, ICC ≥ 0.75; moderate, ICC ≥ 0.4 and < 0.75; poor, ICC < 0.4.
Results
Participants’ characteristics
The characteristics of the three groups of participants are shown in Table 1. There were no differences in sex, age, body mass index, heart rate, and blood pressure among the three groups (all p values > 0.05). Total cholesterol and high-density lipoprotein levels were greater in patients with T2DM than in those in the control group. However, low-density lipoprotein levels were lower in patients with T2DM than in those in the control group. The duration of diabetes and HbA1c levels were higher in the DMb group than in the DMa group.
General echocardiographic parameters
General parameters of echocardiography among the three groups are shown in Table 2. The LVMI in the DMa and DMb groups was higher than that in the control group (both p values < 0.05). The LVMI was not different between the DMb and DMa groups. The E peak in the DMb group was lower than that in the DMa and control groups (both p values < 0.05). The A peak, E/A ratio, and E/Em ratio in the DMb group were higher than those in the DMa and control groups (all p values < 0.05). There were no differences in the E peak, LVEF, and Tei index among the three groups.
Three-dimensional strain and synchronization parameters
The DMb group had decreased peak systolic strains in GLS, GCS, and GAS compared with DMa and control groups (− 16.67 ± 2.44 vs − 19.31 ± 3.91 vs − 20.19 ± 3.23, − 14.80 ± 6.10 vs − 19.76 ± 4.95 vs − 19.30 ± 6.34, and − 26.49 ± 5.42 vs − 30.26 ± 5.50 vs − 32.99 ± 6.60; all p values < 0.05). Nevertheless, Tm-SD and Tm-Dif in the DMb group were higher than those in DMa and control groups (36.98 ± 6.36 vs 26.23 ± 7.87 vs 27.00 ± 6.93, and 45.31 ± 10.67 vs 30.63 ± 7.02 vs 26.82 ± 8.87 l; both p values < 0.05) (Table 2 and Fig. 2).
Associations between general parameters and 3D parameters in T2DM
Duration of diabetes was inversely correlated with 3D strain parameters (GLS, GCS, GAS) and positively associated with 3D synchrony parameters (Tm-SD, Tm-Dif) (r = − 0.229, p = 0.043; r = − 0.412, p < 0.001; r = − 0.327, p = 0.005; r = 0.529, p < 0.001; r = 0.523, p < 0.001, respectively). Similarly, HbA1c values were negatively correlated with 3D strain parameters (GLS, GCS, GAS) and positively correlated with 3D synchrony parameters (Tm-SD, Tm-Dif) (r = − 0.329, p = 0.005; r = − 0.376, p = 0.001; r = − 0.313, p = 0.007; r = 0.646, p < 0.001; r = 0.581, p < 0.001, respectively). Additionally, fasting plasma glucose levels were negatively correlated with 3D strain parameters (GLS, GAS) and the E/A ratio was positively correlated with 3D strain parameters (GLS, GAS) (r = − 0.266, p = 0.004; r = − 0.341, p < 0.001; r = 0.250, p = 0.008; r = 0.236, p = 0.011, respectively) (Table 3).
Analysis of risk factors for preclinical LV myocardial dysfunction
Clinical and echocardiographic parameters were analyzed using multiple stepwise linear regression, and we assessed risk factors for subclinical LV dysfunction. Confounding factors included the duration of diabetes, levels of HbA1c, fasting blood glucose, triglycerides, total cholesterol, and low-density lipoprotein, the LVMI, E/A ratio, and E/Em ratio. Inclusion of these parameters was based on physiological rationality and the results of the aforementioned data. We found that the duration of diabetes was independently associated with GCS (β = − 0.516, p < 0.001) and GAS (β = − 0.391, p = 0.005) (Table 4). Moreover, HbA1c levels were independently associated with GLS (β = − 0.675, p < 0.001), Tm-SD (β = 3.363, p < 0.001), and Tm-Dif (β = 3.895, p < 0.001).
Inter-observer and intra-observer variability
Table 5 shows inter-observer and intra-observer variability for echocardiographic image analysis. The ICCs for inter- and intra-observer variability were 0.835–0.993 and 0.802–0.978, respectively, which suggested that echocardiographic image analysis was consistent.
Discussion
The main findings of our study were as follows. 3D myocardial strain parameters in the DMb group were lower, and 3D synchronization parameters higher, compared with the DMa and control groups. The duration of diabetes and HbA1c levels were independently associated with GLS, GCS, GAS, Tm-SD, and Tm-Dif.
Analysis of 3D myocardial strain and synchrony parameters among the three groups
In the present study, there was no significant difference in the LVEF between controls and patients with T2DM. Determination of the ejection fraction has been shown to be insufficient for detecting minor myocardial abnormalities [11]. Speckle-tracking echocardiography can measure subtle alterations of an impaired myocardium in patients with preclinical T2DM. Subclinical LVD is considered to be an early sign of DCM [14, 15].
Most previous studies measured LV strain by speckle-tracking echocardiography in patients with T2DM for detection of LV systolic and diastolic dysfunction, and consequent impairment of LV function [16,17,18]. For decades, 2D strain was generally used to analyze cardiac function. However, since 2D strain image acquisition is affected by plane motion and perspective, this may misunderstand the true anatomy of the left ventricle [19]. Therefore, 2D speckle-tracking echocardiography (2DSTE) may seriously misunderstand strain data under various pathological conditions. 3D full-volume images contain all-round information of the left ventricular 3D structure, have the potential to overcome the inherent limitations of 2DSTE, and are more accurate and effective than 2DSTE in evaluating LV myocardial function [20, 21]. 3D speckle-tracking echocardiography (3DSTE) promises to simplify the assessment of tissue deformation by replacing the traditional parameters of multidirectional strain assessments [19]. Studies have shown that real-time 3DSTE can comprehensively and rapidly quantitatively evaluate LV systolic function in 3D space [8, 9], and real-time 3D full volume can quantitatively evaluate the synchrony of LV contraction [12]. In our study, these two techniques were used to obtain myocardial strain and synchrony parameters (GLS, GCS, GAS, GRS, Tm-SD, and Tm-Dif). Various studies have shown that these 3D parameters are reproducible and that they can be used to evaluate subtle LV systolic dysfunction with a preserved LVEF [9].
HbA1c reflects the level of terminal glycosylation products in the body. HbA1c can be used to monitor the level of glycemic control in patients with diabetes to determine long-term glycemic control level and stability of patients with T2DM [13]. HbA1c values < 7% reflect good blood sugar control [13]. Our study showed that patients with good blood glucose control (HbA1c values < 7%) showed no difference in 3D myocardial strain parameters and synchronization parameters compared with the control group, which is consistent with previous studies [10, 22]. We also found lower 3D strain parameters (GLS, GCS, and GAS) and higher synchronization parameters (Tm-SD and Tm-Dif) in patients with poor glucose control (HbA1c values ≥ 7%) compared with those with good blood glucose control. These findings suggest that myocardial function is slightly impaired in T2DM patients with poor glucose control, manifesting as subclinical LVD [10, 22].
Correlation and regression analysis
Impaired islet cell function contributes to the pathophysiology of T2DM. Most previous studies have shown that, with a prolonged duration of T2DM, islet cell function gradually declines, leading to worsening of glycemic control [23]. Our study showed that HbA1c values were positively correlated with the duration of T2DM, in accordance with this previous finding. Simultaneously, myocardial strain parameters were negatively correlated with HbA1c values and the duration of diabetes, and synchronous parameters were positively correlated with HbA1c values and the duration of diabetes. Multiple regression analysis showed that HbA1c values and the duration of T2DM were independently associated with GLS, GCS, GAS, Tm-SD, and Tm-Dif. These findings suggest that T2DM with a shorter duration has better control of blood glucose levels [24] and myocardial function, and patients with a longer duration have poor control of blood glucose levels [24] and subclinical myocardial dysfunction. Conversely, observational studies indicated that HbA1c was associated with the risk of heart failure in T2DM patients, independent of blood pressure, obesity, age, and the presence of coronary heart disease [25, 26]. These data support the notion that glycemic control in individuals with T2DM is a critical mechanism in the prevention of LV cardiac dysfunction [27].
Elevated HbA1c levels indicate poor glycemic control in DM. Persistent hyperglycemia can lead to resistance to insulin metabolism in the cardiac tissue, which occurs independently of other cardiac risk factors [27]. The mechanism of myocardial damage caused by diabetes is not fully understood. Possible causes include cardiac insulin resistance, glucotoxicity, mitochondrial dysfunction, oxidative stress, endoplasmic reticulum stress, impaired calcium treatment, systemic and tissue renin–angiotensin–aldosterone system activation, impaired mitochondria and autophagy, coronary microvascular dysfunction, and dysregulation of exosomes [27,28,29,30,31,32]. These factors continuously damage the myocardium, leading to lipid accumulation and disturbance of autonomic nerve and humoral regulation, and may eventually lead to myocardial cell hypertrophy, necrosis, myocardial fibrosis, and increased myocardial weight [33]. We considered that myocardial stiffness is characterized by decreased myocardial strain and poor myocardial synchrony, which is reflected in subclinical LVD.
Conclusions
RT-3DE can detect subclinical LV myocardial dysfunction in poor glycemic control of T2DM. LV myocardial dysfunction is associated with the duration of diabetes and HbA1c levels of T2DM.
Limitations
The main limitation of this trial is that no follow-up studies have been conducted. When glycemic reduction in the poor glycemic control of T2DM (HbA1c values ≥ 7%), whether their LV function is improved has not been studied. Second, the population comprised patients with uncomplicated T2DM without any other significant comorbidities. Therefore, there is limited applicability of our results. The real-world population has a high incidence of comorbidities. Consequently, we could not perform receiver-operating characteristic curve analysis to identify an appropriate level of myocardial strain and synchrony parameters for subclinical myocardial dysfunction. This might have weakened the clinical implications of our study. Future research needs to address this issue.
References
Tadic M, Ilic S, Cuspidi C, Stojcevski B, Ivanovic B, Bukarica L, Jozika L, Celic V (2015) Left ventricular mechanics in untreated normotensive patients with type 2 diabetes mellitus: a two- and three-dimensional speckle tracking study. Echocardiography 32(6):947–955
Amos AF, McCarty DJ, Zimmet P (1997) The rising global burden of diabetes and its complications: estimates and projections to the year 2010. Diabet Med 5:S1–85
Boudina S, Abel ED (2007) Diabetic cardiomyopathy revisited. Circulation 115:3213–3223
Aneja A, Tang WH, Bansilal S, Garcia MJ, Farkouh ME (2008) Diabetic cardiomyopathy: insights into pathogenesis, diagnostic challenges, and therapeutic options. Am J Med 121:748–757
Von Bibra H, St John Sutton M (2010) Diastolic dysfunction in diabetes and the metabolic syndrome: promising potential for diagnosis and prognosis. Diabetologia 53:1033–1045
Larghat AM, Swoboda PP, Biglands JD, Kearney MT, Greenwood JP, Plein S (2014) The microvascular efects of insulin resistance and diabetes on cardiac structure, function, and perfusion: a cardiovascular magnetic resonance study. Eur Heart J Cardiovasc Imaging 15:1368–1376
Di Carli MF, Janisse J, Grunberger G, Ager J (2003) Role of chronic hyperglycemia in the pathogenesis of coronary microvascular dysfunction in diabetes. J Am Coll Cardiol 41:1387–1393
Luis SA, Yamada A, Khandheria BK, Speranza V, Benjamin A, Ischenko M, Platts DG, Hamilton-Craig CR, Haseler L, Burstow D et al (2014) Use of three-dimensional speckle-tracking echocardiography for quantitative assessment of global left ventricular function: a comparative study to three-dimensional echocardiography. J Am Soc Echocardiography 27:285–291
Tops LF, Delgado V, Marsan NA, Bax JJ (2017) Myocardial strain to detect subtle left ventricular systolic dysfunction. Eur J Heart Fail 19(3):307–313
Ernande L, Bergerot C, Rietzschel ER, De Buyzere ML, Thibault H, Pignonblanc PG, Croisille P, Ovize M, Groisne L, Moulin P et al (2011) Diastolic dysfunction in patients with type 2 diabetes mellitus: is it really the first marker of diabetic cardiomyopathy? J Am Soc Echocardiography 24:1268–1275
Smiseth OA, Torp H, Opdahl A, Haugaa KH, Urheim S (2016) Myocardial strain imaging: how useful is it in clinical decision making? Eur Heart J 37:1196–1207
Khan SG, Klettas D, Kapetanakis S, Monaghan MJ (2016) Clinical utility of speckle-tracking echocardiography in cardiac resynchronisation therapy. Echo Res Pract 3(1):R1–R11. https://doi.org/10.1530/ERP-15-0032
American Diabetes Association (2012) Standards of medical care in diabetes—2012. Diabetes Care 35(suppl):S11–63
Nakai H, Takeuchi M, Nishikage T, Lang RM, Otsuji Y (2009) Subclinical left ventricular dysfunction in asymptomatic diabetic patients assessed by two-dimensional speckle tracking echocardiography: correlation with diabetic duration. Eur J Echocardiogr 10:926–932
Ng AC, Delgado V, Bertini M, van der Meer RW, Rijzewijk LJ, Shanks M, Nucifora G, Smit JW, Diamant M, Romijn JA et al (2009) Findings from left ventricular strain and strain rate imaging in asymptomatic patients with type 2 diabetes mellitus. Am J Cardiol 104:1398–1401
Wang Q, Tan K, Xia H, Gao Y (2018) Left ventricular structural alterations are accompanied by subclinical systolic dysfunction in type 2 diabetes mellitus patients with concomitant hyperlipidemia: an analysis based on 3D speckle tracking echocardiography. Echocardiography 35:965–974
Li ZJ, Du LF, Luo XH (2014) Evaluation of ventricular-vascular coupling in patients with type 2 diabetes mellitus using 2-dimensional speckle tracking imaging. Huazhong Univ Sci Technolog Med Sci 34:929–934
Wang Q, Ma W, Xia J (2018) Nonalcoholic fatty liver is associated with further left ventricular abnormalities in patients with type 2 diabetes mellitus: a 3-dimensional speckle-tracking study. J Ultrasound Med 37:1899–1911
Truong VT, Phan HT, Pham KNP, Duong HNH, Ngo TNM, Palmer C et al (2019) Normal ranges of left ventricular strain by three-dimensional speckle-trackingechocardiography in adults: a systematic review and meta-analysis. J Am Soc Echocardiogr 32(12):1586–1597
Seo Y, Ishizu T, Aonuma K (2014) Current status of 3-dimensional speckle tracking echocardiography: a review from our experiences. J Cardiovasc Ultrasound 22:49–57
Xu TY, Sun JP, Lee AP, Yang XS, Qiao Z, Luo X et al (2014) Three-dimensional specklestrain echocardiography is more accurate and efficient than2Dstrain in the evaluation of left ventricular function. Int J Cardiol 176:360–366
Zhang X, Wei X, Liang Y, Liu M, Li C, Tang H (2013) Differential changes of left ventricular myocardial deformation in diabetic patients with controlled and uncontrolled blood glucose: a three-dimensional speckle-tracking echocardiography-based study. J Am Soc Echocardiogra 26:499–506
Yoshifumi S (2015) β-cell dysfunction: its critical role in prevention and management of type 2 diabetes. World J Diabetes 6(1):109–124
Mannucci E, Monami M, Dicembrini I, Piselli A, Porta M (2014) Achieving HbA1c targets in clinical trials and in the real world: a systematic review and meta-analysis. J Endocrinol Invest 37(5):477–495. https://doi.org/10.1007/s40618-014-0069-6
Iribarren C, Karter AJ, Go AS, Ferrara A, Liu JY, Sidney S, Selby JV (2001) Glycemic control and heart failure among adult patients with diabetes. Circulation 103:2668–2673
Stratton IM, Adler AI, Neil HA, Matthews DR, Manley SE, Cull CA, Hadden D, Turner RC, Holman RR (2000) Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study. BMJ 321:405–412
Jia G, Whaley-Connell A, Sowers JR (2018) Diabetic cardiomyopathy: a hyperglycaemia- and insulin-resistance-induced heart disease. Diabetologia 61(1):21–28. https://doi.org/10.1007/s00125-017-4390-4
Frustaci A, Kajstura J, Chimenti C, Jakoniuk I, Leri A, Maseri A, Nadal-Ginard B, Anversa P (2000) Myocardial cell death in human diabetes. Circ Res 87:1123–1132
Cai L, Wang Y, Zhou G, Chen T, Song Y, Li X, Kang YJ (2006) Attenuation by metallothionein of early cardiac cell death via suppression of mitochondrial oxidative stress results in a prevention of diabetic cardiomyopathy. J Am Coll Cardiol 48:1688–1697
Zhao XY, Hu SJ, Li J, Mou Y, Chen BP, Xia Q (2006) Decreased cardiac sarcoplasmic reticulum Ca2+-ATPase activity contributes to cardiac dysfunction in streptozotocin-induced diabetic rats. J Physiol Biochem 62:1–8
Taegtmeyer H, McNulty P, Young ME (2002) Adaptation and maladaptation of the heart in diabetes, part I. Circulation 105:1727–1733
An D, Rodrigues B (2006) Role of changes in cardiac metabolism in development of diabetic cardiomyopathy. Am J Physiol Heart Circ Physiol 291:H1489–H1506
Aon MA, Foster DB (2015) Diabetic cardiomyopathy and the role of mitochondrial dysfunction: novel insights, mechanisms, and therapeutic strategies. Antioxid Redox Signal 22(17):1499–1501. https://doi.org/10.1089/ars.2015.6349
Funding
This research received no external funding.
Author information
Authors and Affiliations
Contributions
Conceptualization, XC; methodology, XC; echocardiographic imaging analysis, XC and QY; data curation, XC and QY; writing-original draft preparation, XC; writing-review and editing, XC, HG, QY, JF and XK.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, X., Guo, H., Yang, Q. et al. Quantitative evaluation of subclinical left ventricular dysfunction in patients with type 2 diabetes mellitus by three-dimensional echocardiography. Int J Cardiovasc Imaging 36, 1311–1319 (2020). https://doi.org/10.1007/s10554-020-01833-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-020-01833-5