Abstract
To prospectively compare image quality and myocardial T1 relaxation times of modified Look-Locker inversion recovery (MOLLI) imaging at 3.0 T (T) acquired with patient-adaptive dual-source (DS) and conventional single-source (SS) radiofrequency (RF) transmission. Pre- and post-contrast MOLLI T1 mapping using SS and DS was acquired in 27 patients. Patient wise and segment wise analysis of T1 times was performed. The correlation of DS MOLLI measurements with a reference spin echo sequence was analysed in phantom experiments. DS MOLLI imaging reduced T1 standard deviation in 14 out of 16 myocardial segments (87.5%). Significant reduction of T1 variance could be obtained in 7 segments (43.8%). DS significantly reduced myocardial T1 variance in 16 out of 25 patients (64.0%). With conventional RF transmission, dielectric shading artefacts occurred in six patients causing diagnostic uncertainty. No according artefacts were found on DS images. DS image findings were in accordance with conventional T1 mapping and late gadolinium enhancement (LGE) imaging. Phantom experiments demonstrated good correlation of myocardial T1 time between DS MOLLI and spin echo imaging. Dual-source RF transmission enhances myocardial T1 homogeneity in MOLLI imaging at 3.0 T. The reduction of signal inhomogeneities and artefacts due to dielectric shading is likely to enhance diagnostic confidence.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
T1 relaxation time mapping is an emerging technique for quantitative characterization of the myocardial structure. In contrast to conventional late gadolinium enhancement (LGE) imaging, T1 mapping allows for detection and quantification of rather subtle or diffuse myocardial disease such as fibrosis or amyloid depletion [1–4]. Quantitative T1 measurements are usually obtained by serial acquisition of inversion-recovery (IR) sequences with varying inversion times. The prototype of this technique is the so-called Look-Locker sequence, which has been widely implemented in MR devices of the newer generation [5, 6]. It is commonly used for calculation of the optimal myocardial inversion time prior to IR LGE imaging (“TI Scout”). An important drawback of this technique is its inherent susceptibility to cardiac motion thus allowing only for regional-wise T1 estimation. The modified Look-Locker inversion recovery sequence (MOLLI) has been developed to overcome these limitations [7, 8]. ECG-gated image acquisition has significantly reduced heart-rate dependency compared to conventional Look-Locker T1 measurement.
MOLLI imaging can be performed at 1.5 T (T) and 3.0 T [9]. Pre-contrast myocardial T1 relaxation time at 3.0 T was found to be 22–33% greater than at 1.5 T [10]. Cardiac MRI at 3.0 T generally offers an improved signal-to-noise ratio (SNR) compared to 1.5 T. Nonetheless, high field strength imaging is accompanied by new challenges. Image quality is quite susceptible to inhomogeneities in the B1-field with consequent local peaks of energy deposition (SAR) and flip angle nonuniformities [11, 12]. Moreover, the actual average flip angle is typically lower in the heart region than the desired flip angle [13].
At 3.0 T, small variations introduced by off-resonance and local variation of flip angle may lead to significant biases in measured myocardial T1 [14, 15]. Local artefactual T1 variations might be falsely confused with real pathology.
Independent parallel transmission of the radiofrequency (RF) pulse (dual-source parallel RF transmission) has been implemented in MR scanners of the latest generation to reduce the abovementioned artefacts by enhancing B1-field homogeneity at 3.0 T. Patient-adaptive local RF shimming improves signal homogeneity across the heart via a significant reduction of flip angle variation [16]. Enhanced signal uniformity and contrast-to-noise ratio with dual-source RF transmission has been demonstrated for abdominal, spinal and cardiac workups at 3.0 T [16–19].
The purpose of the present investigation was to prospectively compare image quality and myocardial T1 relaxation time homogeneity of 3.0 T MOLLI imaging using patient adaptive dual-source RF transmission with conventional RF transmission.
Materials and methods
Study population
27 adult patients (7 women, 20 men; age range 24–85 years; mean age 53.0 ± 16.6 years) were enrolled in this study between September 2015 and April 2016. Written informed consent was obtained prior to the examination. The study has been approved by the local ethics committee of the Medical Faculty of the Technische Universität München. All patients underwent conventional and dual-source RF transmission MOLLI as well as LGE imaging within the same session. Clinical indication for cardiac MRI were suspected or known myocarditis (11 patients), suspected or known ischemia (8 patients) and suspected cardiac manifestation of systemic diseases (sarcoidosis, thalassaemia, amyloidosis, lupus erythematodes, M. Erdheim-Chester, 8 patients). Patients with contraindications for MRI (pacemaker etc.) were excluded.
Imaging
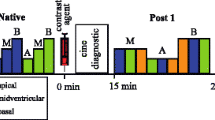
Cardiac imaging was performed at 3.0 T using a clinical dual-source RF transmission MR system (max. gradient amplitude 45 mT/m; slew rate 200 T/m/s; rise time 0.2 ms) with local RF transmission (Ingenia, Philips Healthcare, Best, the Netherlands). Standard 12-element posterior and 16-element anterior coils were used for signal reception with the ‘SmartSelect’ option for the automated selection of coil elements with sufficient SNR. ECG-gated breath-hold 2D MOLLI sequences were performed prior to and 10–15 min after intravenous application of gadopentetate dimeglumine (Magnevist®) at a dose of 0.2 mmol/kg body weight with the following pulse sequence parameters: TR 2.4 ms, TE 1.08 ms, flip angle 20°. Acquired voxel size was 1.9 × 2.0 × 10.0 mm, FOV was 363 × 300 mm. The MOLLI protocol used a 5(3 s) three sampling scheme with a steady-state free precession (SSFP) readout. Three slices in short-axis orientation were obtained covering the basal, midventricular and apical myocardial segments. For acquisition of “single-source” images, the RF transmit system was operated in conventional quadrature mode with a fixed phase difference of 90° between the two transmit sources. The transmit power was identical for both sources. Using dual-source RF transmission, the RF power was distributed to the coil system via two transmission sources with independent control of the amplitude and phase of the RF waveforms. Prior to imaging, a B1-calibration scan for adjustment of dual-source patient adaptive RF transmission was performed. The shim volume was planned to include the entire heart. Parallel imaging (SENSE [20], factor 2) was enabled. Besides MOLLI imaging, all patients received a clinical cardiac MR protocol adjusted to the respective clinical issue. CINE SSFP imaging, T2 TSE dark-blood imaging and LGE imaging (3D IR GRE or 2D PSIR) was performed in all subjects according to the institutional standard protocol.
Phantom imaging
A T1 phantom with 11 cylindrical test tubes was built. Each 50 ml tube had a dimension of 115 × 30 mm. The tubes were filled with distilled water and gadopentetate dimeglumine at concentrations varying from 0.0 to 1.0 mmol/l. The respective T1 values ranged from 200 to 3125 ms. For reference values, the T1 phantom was scanned using a 3D inversion-recovery spin echo sequence with the following pulse sequence parameters: TR 15,000 ms, TE 11.5 ms, matrix 256 × 256, FOV 160 × 160 mm, slice thickness 3 mm, 25 inversion times from 25 to 5000 ms. Then single-source and dual-source RF transmission MOLLI imaging was performed in a perpendicular plane to the long axis of the phantom (TR 2.4 ms, TE 1.08 ms, flip angle 20°). T1 times were then measured by manually drawn ROIs on the automatically generated T1 maps using a dedicated post-processing software (Philips IntelliSpace Portal®). Measurements were repeated three times for both single-source and dual-source RF transmission.
Image analysis
For assessment of myocardial T1 times, regions-of-interest (ROI) were manually drawn into the automatically generated pre- and post-contrast T1 maps using a dedicated post-processing software (Philips IntelliSpace Portal®). Therefore, the three short-axis slices obtained in each subject (basal, midventricular and apical) were divided into 16 segments modified to the AHA recommendations [21]. ROIs were of comparable size and did not include adjacent anatomical structures. Areas with pathologic findings on T2w dark-blood images or LGE images such as myocardial edema or infarction were carefully excluded. An AHA segment was excluded from further analysis if it was in total affected by pathology. The reader was blinded to the type of RF transmission. For determination of signal-to-noise ratio (SNR), signal intensity (SI) was measured by three regions-of-interest placed in the interventricular septum in a midpapillary short-axis plane in each patient. The SI was measured in the image with the longest inversion time. To perform SNR calculation, the signal deviation of the noise (SD) was measured by a ROI placed in the air outside the body in the respective slice. SNR was defined as SNR = SISeptum/SDNoise.
Statistical analysis
Correlation between phantom T1 times measured with single- and dual-source RF transmission MOLLI and reference values acquired with a 3D IR SE sequence was analysed using linear regression analysis. The Wilcoxon test was used to assess differences of T1 measurement between the methods. Homogeneity of myocardial T1 was evaluated patient wise, plane wise and segment wise. Differences in myocardial T1 time between images acquired with single-source and dual-source RF transmission were assessed using a two-sided paired student’s t test. A p value less than 0.05 was considered significant. For assessment of myocardial T1 variance, standard deviation, standard error and coefficient of variation were analysed. The F-test was used to test for statistical significance of variance differences. Statistical analyses were performed with Prism (Version 5, GraphPad, La Jolla, USA).
Results
Phantom experiments
Figure 1 shows the automatically generated T1 maps of the T1 phantom acquired with single-source and dual-source RF transmission. Mean reference T1 values obtained with a 3D IR SE sequence ranged between 200 ms (Gd concentration 1.0 mmol/l) and 2970 ms for the water tube. Regression analysis showed a good correlation between IR SE T1 values, single-source RF MOLLI (y = 0.9769x − 8.550, R2 = 0.9990) and dual-source RF MOLLI (y = 0.9477x − 18.8, R2 = 0.9937), Fig. 2. T1 times measured with single-source and dual-source RF transmit MOLLI did not significantly differ from IR SE acquisition (p = 1.0 for single-source RF transmission and p = 0.43 for dual-source RF transmission, Wilcoxon test). In our phantom, neither absolute T1 values nor standard deviation (SD) and standard error of mean (SEM) did significantly differ between both transmission modes.
T1 maps of the T1 phantom acquired with single-source (a) and dual-source RF transmission (b). c Mean reference T1 values obtained with a 3D IR SE sequence ranged between 200 ms (Gd concentration 1.0 mmol/l) and 2970 ms for the water tube (hatched bars). T1 values of the ten test tubes measured with single-source RF MOLLI (white bars) and dual-source RF MOLLI (grey bars), and the water tube (W). Error bars represent standard error of mean (SEM)
Patient experiments
Segment wise analysis
Single-source and dual-source RF transmit MOLLI imaging was performed in 27 subjects. In two patients, significant motion artefacts occurred in all three slices due to patient movement and insufficient breath hold. These patients were excluded from further analysis.
Segment wise T1 analysis was exclusively performed for pre-contrast MOLLI images to foreclose myocardial T1 inhomogeneities due to interindividually varying gadolinium washout dynamics. 16 AHA-segments in 25 patients were analysed. 30 AHA segments were excluded from analysis as these were completely affected by pathology, resulting in a total of 370 myocardial segments analysed. Patient-adaptive dual-source RF transmission resulted in a reduction of T1 standard deviation and coefficient of variation in 14 out of 16 myocardial segments (87.5%). Significant reduction of T1 variance could be obtained in 7 segments (43.8%). For two segments (segment 10 and 13), T1 variance was higher for dual-source RF transmission without reaching statistical significance. No significant differences in absolute myocardial T1 time could be observed for both techniques. SNR measured in the interventricular septum was 20.3 ± 1.5 for single-source RF transmission and 34.9 ± 1.7 for dual-source transmission with patient adaptive shimming resulting in an improvement of 71.9% (p < 0.0001). Results of segment wise analysis are detailed in Table 1.
Slice wise analysis
T1 variation in three short-axis slices (basal, midventricular and apical) was analysed in 25 patients. The use of dual-source RF transmission resulted in a significant reduction of T1 variance in all three planes. F-Test p values were 0.001 for the basal slice, 0.03 for the midventricular slice and 0.0003 for the apical slice. For the basal slice, absolute T1 was significantly higher on images acquired with conventional RF transmission (1279 ± 4.9 ms vs. 1260 ± 2.9 ms, p < 0.0001). No significant difference in myocardial T1 relaxation time was observed in the midventricular and apical slice.
Patient wise analysis
Myocardial T1 was assessed in 16 AHA segments in each patient before and after administration of gadolinium-based contrast agent. For pre-contrast MOLLI imaging, dual-source RF transmission resulted in a significant reduction of overall T1 variance in 12 out of 25 patients (48%). In another ten patients, T1 standard deviation and standard error were reduced without reaching statistical significance. In three patients, T1 variance was higher with dual-source RF transmission, in one case significantly. For post-contrast T1 maps, dual-source RF transmission resulted in a significant reduction of T1 variance in 16 out of 25 patients (64%). In three patients T1 variance was significantly higher with DS RF transmission. Results of patient wise analysis are detailed in Tables 1 and 2 of the electronic supplementary material.
In six patients (24%), artefacts due to dielectric shading were seen on MOLLI images and the respective T1 maps acquired with conventional RF transmission. The artefacts were in all cases located in the area of the right ventricle and the interventricular septum. No such artefacts occurred in T1 maps acquired with dual-source RF transmission (Figs. 3, 4). In one patient, inhomogeneous T1 alterations appeared in the anterolateral wall of the left ventricle with conventional RF transmission suggesting myocardial pathology. A correlate for this was neither found in images acquired with patient-adaptive RF shimming nor in conventional T2w and LGE sequences (Fig. 5).
67 year-old male patient with suspected myocarditis. On pre-contrast single-source RF MOLLI images (SS) and the respective T1 map, signal drop-off artefacts are seen in the right ventricular cavity partially affecting the interventricular septum. Dual-source RF transmission with local RF shimming (DS) results in a more homogeneous signal over the whole heart. No correlation was found on T2 TSE and LGE images (not shown)
33 year-old male patient with suspected myocarditis. Post-contrast single-source (SS) and dual-source (DS) RF MOLLI images and the respective T1 maps. On single-source RF MOLLI images, signal drop-off artefacts are seen in the right ventricular cavity and over the anterolateral septum with reduced diagnostic confidence. Dual-source RF transmission (DS) significantly reduces these artefacts. No correlation was found on T2 TSE and LGE images (not shown)
55 year-old male patient with suspected myocarditis. Post-contrast single-source (SS) and dual-source (DS) RF MOLLI images and the respective T1 maps. Artifactual signal alterations of the anterolateral left ventricular wall on single-source MOLLI images (arrows). These might falsely be interpreted as myocardial pathology. A correlate was neither found in the dual-source RF MOLLI imaging nor in the 3D IR images after gadolinium application (LGE)
In our cohort, ten patients had pathologic findings on cardiac MRI. Findings suggestive of myocarditis were seen in five patients. In these subjects, pre-contrast T1 difference between the affected and the remote myocardium was 136.2 ± 48.5 ms for single-source RF MOLLI and 185.6 ± 43.5 ms for dual-source RF MOLLI (p = 0.47). Post-contrast T1 differences were 89.0 ± 30.4 and 87.4 ± 20.8 ms (p = 0.97), respectively. Five patients had findings suggestive of focal fibrosis or infarction. Pre-contrast T1 difference between the affected and the remote myocardium was 160.6 ± 45.1 ms for single-source RF MOLLI and 166.6 ± 51.6 ms for dual-source RF MOLLI (p = 0.93). Post-contrast T1 differences were 125.6 ± 24.2 and 141.4 ± 23.2 ms (p = 0.65), respectively. In all patients there was good accordance of imaging findings between single-source MOLLI, dual-source MOLLI and LGE imaging. (Fig. 6).
Upper row 31 year-old female patient with suspected myocarditis. Post-contrast MOLLI T1 maps acquired with single-source (SS) and dual-source (DS) RF transmission. In accordance with LGE-imaging, a band-like subepicardial area with reduced T1 relaxation time is seen in the anterior aspect of the left ventricular wall suggesting myocardial inflammation (arrows). Myocardial edema was seen on T2w images (not shown). Lower row 52 year-old male patient with acute embolic infarction of the inferior left ventricular wall. In accordance with LGE imaging, an area of prolonged T1 relaxation time is found on pre-contrast single-source and dual-source RF MOLLI T1 maps (arrow). Note the signal drop-off over the right ventricular cavity on images acquired with conventional RF transmission
Discussion
Differences of T1 relaxation times between pathologically altered myocardium and the remote myocardium can be subtle, even in case of acute myocardial infarction [22]. Hence, accurate measurement of myocardial T1 is a prerequisite for diagnostic reliability of MOLLI imaging. Artefactual T1 variations may easily be misinterpreted as myocardial pathology.
This investigation assessed the performance of cardiac MOLLI T1 mapping at 3.0 T using dual-source RF transmission with patient adaptive RF shimming. Compared to conventional RF transmission, this technique resulted in a significant reduction of pre-contrast myocardial T1 variance in 7 of 16 AHA segments assessed in 25 patients (43.8%). Dual-source RF transmission significantly reduced overall pre-contrast T1 variance in 12 out of 25 patients (48.0%). For post-contrast imaging, a significant reduction of T1 variance could be obtained in 16 out of 25 patients (64.0%). Distinct dielectric shading occurred in six patients examined with single-source RF transmission causing a diagnostic uncertainty in the clinical setting. No according artefacts were seen in MOLLI images acquired with dual-source RF transmission. In all patients there was good accordance of imaging findings between single-source MOLLI, dual-source MOLLI and LGE imaging.
Magnetic resonance imaging at 3.0 T holds the promise for higher SNR compared to 1.5 T [11, 23]. However, adoption to cardiac imaging has long been hampered by inhomogeneities of both the static magnetic field (B0) and the transmit radiofrequency field (B1). At 3.0 T, these magnetic field inconsistencies have a greater impact on image quality compared to 1.5 T. Variations in local off-resonance as well as signal loss due to dielectric shading are more likely to occur at 3.0 T. There is a complicated relationship between the variation of off-resonance, B0 homogeneity and flip angle variations due to local B1 field inconsistencies [14]. Flip angle variations across the scan volume are the main cause for higher T1 variation using single-source MOLLI imaging. Flip angle variation has been shown to reach up to 63% of the desired flip angle across the entire LV volume [23, 24]. Significant T1 bias due to flip angle nonuniformity has been noticed by several groups. Sung et al. reported a 52% T1 estimation bias in breast MRI at 3.0 T without B1 correction [25]. As suggested by Kellman et al., a reduced flip angle of 20° was used for single-source and dual-source RF transmission MOLLI in the present investigation. This can be regarded as a tradeoff between dielectric shading at the cost of decreased SNR.
Patient adaptive parallel transmission of the radiofrequency (RF) pulse has been implemented in MR scanners of the latest generation to enhance B1 field homogeneity at 3.0 T. Prior to image acquisition, the B1 field generated by the two radiofrequency sources is analysed. The amplitude and the phase of the RF waves are then tailored in a patient adaptive manner. Enhanced B1 homogeneity resulting in improved image quality has been documented for abdominal, spinal and cardiac workups [16–19]. Flip angle homogenization across the cardiac scan volume has been demonstrated [16]. Dual-source RF transmission improves B1 homogeneity with a consequent reduction of local RF energy absorption (SAR) peaks. This allows for an optimization of the local and whole-body SAR [26, 27]. Consequently, TR can be shortened with reduced examination time. This is especially favorable in patients with problems to lay still during imaging. To our best knowledge, there is no data comparing image quality and diagnostic confidence of 3.0 T MOLLI T1 mapping with patient adaptive dual-source and conventional RF transmission.
RF field inhomogeneity inside the patient is highly dependent on the patient’s individual anatomy and structural abnormalities. This might explain why a significant improvement of myocardial T1 homogeneity due to dual-source RF transmission could not be obtained in every patient in this study. Hence, this technique might be more beneficial in patients with higher BMI, pleural effusions or sternal wires.
The precision of the T1 estimates is also a function of SNR [28]. Segment-wise analysis of our data resulted in T1 standard deviation of 60.3 ms for single-source RF and 42.4 ms for dual-source RF transmission resulting in a reduction of 42.2%, while improvement of SNR was 71.9%. It seems that higher SNR does not fully translate into reduction of T1 standard deviation. This phenomenon might be due other sources of error like uncompensated heart or respiratory motion.
In our cohort, average myocardial T1 relaxation times for both methods were in line with previously published data [10, 29]. As described before, pre-contrast T1 times slightly increased from base to apex [29]. Phantom experiments demonstrated good correlation of myocardial T1 relaxation time measured with dual-source RF transmission MOLLI and the IR spin echo reference sequence. In our phantom, neither absolute T1 values nor SD and SEM did significantly differ between both transmission modes. This is likely owing to the small volume of the test tubes (50 ml) since flip angle variances with consequent signal fluctuation at 3.0 T increase with the scan volume [16, 19].
We found notable inter-patient variation in absolute pre-contrast myocardial T1 relaxation times. This observation is in line with previously published data reporting a coefficient of variation of up to 4.5% for pre-contrast MOLLI imaging at 3.0 T [30]. In eight patients (32.0%), average pre-contrast myocardial T1 relaxation time was significantly different for dual-source and single-source RF transmission. There is no sufficient explanation for this phenomenon. After administration of contrast agent, significant differences in myocardial T1 values were observed between both imaging protocols. This is attributable to the sequential acquisition of single-source and dual-source MOLLI sequences after Gd-application. The myocardial concentration of Gd is largely dependent on the exchange rate with the blood pool and the renal clearance with washout dynamics differing interindividually [29].
Our study had several limitations. First, it was conducted as a single centre study with only 27 patients included. Myocardial T1 homogeneity using dual-source RF transmission has only been tested against the results obtained from the conventional MOLLI sequence. No histopathologic workup is available to confirm imaging findings since no patient had a clinical indication for myocardial biopsy.
Artefacts occurring with single source transmitting are most likely a result of this specific method due to local appearance. However, since imaging with dual source and single source transmitting was performed subsequently the observed artefacts may theoretically result from motion or respiration.
Ten patients in our cohort had pathologic findings on cardiac MRI. Five patients showed focal myocardial scar or focal fibrosis, five patients had findings suggestive of myocardial inflammation. Areas of recognizable myocardial disease were carefully excluded from T1 measurement. However, we cannot exclude structural alterations of the remote myocardium with altered T1 relaxation time, especially in case of inflammatory disease. Dedicated analysis of sensitivity and specificity of dual-source RF MOLLI for the detection of cardiac disease was beyond the scope of the present investigation. However, there was good accordance between dual-source MOLLI and conventional imaging sequences in all patients with myocardial pathology.
In conclusion, dual-source RF transmission with patient-adaptive local shimming enhances myocardial T1 homogeneity in MOLLI imaging at 3.0 T. The reduction of signal inhomogeneity and dielectric effects is likely to enhance diagnostic confidence.
Abbreviations
- DS:
-
Dual-source
- ECG:
-
Electrocardiography
- FOV:
-
Field of view
- GRE:
-
Gradient echo
- IR:
-
Inversion recovery
- LGE:
-
Late gadolinium enhancement
- MOLLI:
-
Modified look-locker inversion recovery
- MR:
-
Magnetic resonance
- PSIR:
-
Phase-sensitive inversion recovery
- RF:
-
Radiofrequency
- ROI:
-
Region of interest
- SE:
-
Spin echo
- SNR:
-
Signal-to-noise ratio
- SAR:
-
Specific absorption rate
- SS:
-
Single-source
- SSFP:
-
Steady state free precession
- T:
-
Tesla
- TSE:
-
Turbo spin echo
References
Iles L, Pfluger H, Phrommintikul A et al (2008) Evaluation of diffuse myocardial fibrosis in heart failure with cardiac magnetic resonance contrast-enhanced T1 mapping. J Am Coll Cardiol 52:1574–1580
Sparrow P, Messroghli DR, Reid S, Ridgway JP, Bainbridge G, Sivananthan MU (2006) Myocardial T1 mapping for detection of left ventricular myocardial fibrosis in chronic aortic regurgitation: pilot study. AJR Am J Roentgenol 187:W630–W635
Maceira AM, Joshi J, Prasad SK et al (2005) Cardiovascular magnetic resonance in cardiac amyloidosis. Circulation 111:186–193
Pennell DJ, Maceira AM (2009) Magnetic resonance imaging in cardiac amyloidosis. JACC Cardiovasc Imaging 2:1378–1380
Amano Y, Takayama M, Kumita S (2009) Contrast-enhanced myocardial T1-weighted scout (look-locker) imaging for the detection of myocardial damages in hypertrophic cardiomyopathy. J Magn Reson Imaging 30:778–784
Karlsson M, Nordell B (2000) Analysis of the look-locker T(1) mapping sequence in dynamic contrast uptake studies: simulation and in vivo validation. Magn Reson Imaging 18:947–954
Messroghli DR, Greiser A, Frohlich M, Dietz R, Schulz-Menger J (2007) Optimization and validation of a fully-integrated pulse sequence for modified look-locker inversion-recovery (MOLLI) T1 mapping of the heart. J Magn Reson Imaging 26:1081–1086
Messroghli DR, Radjenovic A, Kozerke S, Higgins DM, Sivananthan MU, Ridgway JP (2004) Modified look-locker inversion recovery (MOLLI) for high-resolution T1 mapping of the heart. Magn Reson Med 52:141–146
Raman FS, Kawel-Boehm N, Gai N et al (2013) Modified look-locker inversion recovery T1 mapping indices: assessment of accuracy and reproducibility between magnetic resonance scanners. J Cardiovasc Magn Reson 15:64
Lee JJ, Liu S, Nacif MS et al (2011) Myocardial T1 and extracellular volume fraction mapping at 3 T. J Cardiovasc Magn Reson 13:75
Gutberlet M, Noeske R, Schwinge K, Freyhardt P, Felix R, Niendorf T (2006) Comprehensive cardiac magnetic resonance imaging at 3.0 Tesla: feasibility and implications for clinical applications. Invest Radiol 41:154–167
Wieben O, Francois C, Reeder SB (2008) Cardiac MRI of ischemic heart disease at 3 T: potential and challenges. Eur J Radiol 65:15–28
Dietrich O, Reiser MF, Schoenberg SO (2008) Artifacts in 3-T MRI: physical background and reduction strategies. Eur J Radiol 65:29–35
Kellman P, Herzka DA, Arai AE, Hansen MS (2013) Influence of off-resonance in myocardial T1-mapping using SSFP based MOLLI method. J Cardiovasc Magn Reson 15:63
Kellman P, Hansen MS (2014) T1-mapping in the heart: accuracy and precision. J Cardiovasc Magn Reson 16:2
Mueller A, Kouwenhoven M, Naehle CP et al (2012) Dual-source radiofrequency transmission with patient-adaptive local radiofrequency shimming for 3.0-T cardiac MR imaging: initial experience. Radiology 263:77–85
Jia H, Wang C, Wang G et al (2013) Impact of 3.0 T cardiac MR imaging using dual-source parallel radiofrequency transmission with patient-adaptive B1 shimming. PLoS One 8:e66946
Nelles M, Konig RS, Gieseke J et al (2010) Dual-source parallel RF transmission for clinical MR imaging of the spine at 3.0 T: intraindividual comparison with conventional single-source transmission. Radiology 257:743–753
Willinek WA, Gieseke J, Kukuk GM et al (2010) Dual-source parallel radiofrequency excitation body MR imaging compared with standard MR imaging at 3.0 T: initial clinical experience. Radiology 256:966–975
Pruessmann KP, Weiger M, Scheidegger MB, Boesiger P (1999) SENSE: sensitivity encoding for fast MRI. Magn Reson Med 42:952–962
Cerqueira MD, Weissman NJ, Dilsizian V et al (2002) Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart. A statement for healthcare professionals from the Cardiac Imaging Committee of the Council on Clinical Cardiology of the American Heart Association. Circulation 105:539–542
Dall’Armellina E, Piechnik SK, Ferreira VM et al (2012) Cardiovascular magnetic resonance by non contrast T1-mapping allows assessment of severity of injury in acute myocardial infarction. J Cardiovasc Magn Reson 14:15
Greenman RL, Shirosky JE, Mulkern RV, Rofsky NM (2003) Double inversion black-blood fast spin-echo imaging of the human heart: a comparison between 1.5 and 3.0 T. J Magn Reson Imaging 17:648–655
Sung K, Nayak KS (2008) Measurement and characterization of RF nonuniformity over the heart at 3 T using body coil transmission. J Magn Reson Imaging 27:643–648
Sung K, Daniel BL, Hargreaves BA (2013) Transmit B1+ field inhomogeneity and T1 estimation errors in breast DCE-MRI at 3 T. J Magn Reson Imaging 38:454–459
Katscher U, Bornert P (2006) Parallel RF transmission in MRI. NMR Biomed 19:393–400
Homann H, Graesslin I, Eggers H et al (2012) Local SAR management by RF shimming: a simulation study with multiple human body models. MAGMA 25:193–204
Kellman P, Arai AE, Xue H (2013) T1 and extracellular volume mapping in the heart: estimation of error maps and the influence of noise on precision. J Cardiovasc Magn Reson 15:56
von Knobelsdorff-Brenkenhoff F, Prothmann M, Dieringer MA et al (2013) Myocardial T1 and T2 mapping at 3 T: reference values, influencing factors and implications. J Cardiovasc Magn Reson 15:53
Kawel N, Nacif M, Zavodni A et al (2012) T1 mapping of the myocardium: intra-individual assessment of post-contrast T1 time evolution and extracellular volume fraction at 3 T for Gd-DTPA and Gd-BOPTA. J Cardiovasc Magn Reson 14:26
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
Informed consent was obtained from all individual participants included in the study. All procedures performed involving human participants were in accordance with the ethical standards of the institutional research committee. The study has been approved by the local ethics committee of the Medical Faculty of the Technische Universität München.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rasper, M., Nadjiri, J., Sträter, A.S. et al. Cardiac MOLLI T1 mapping at 3.0 T: comparison of patient-adaptive dual-source RF and conventional RF transmission. Int J Cardiovasc Imaging 33, 889–897 (2017). https://doi.org/10.1007/s10554-017-1072-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-017-1072-8