Abstract
Purposes
Dietary patterns have been found to be associated with the overall cancer risk and survival. However, the associations of healthy dietary patterns and breast cancer remain unclear. We aimed to conduct a meta-analysis of prospective cohort studies to estimate the pooled results of the association of healthy dietary patterns with breast cancer risk and survival.
Methods
PubMed, EMBASE, and Web of Science were searched for literature published until June 24th, 2018 that examined the associations between healthy dietary patterns and breast cancer risk and survival. Risk ratios (RRs) and 95% confidence intervals (CIs) were calculated by using a random-effects model for meta-analysis.
Results
There were 32 articles retrieved for the meta-analysis, with 27 for breast cancer risk and five for breast cancer survival. There was a statistically significant lower risk of breast cancer associated with healthy dietary patterns (RR = 0.93, 95% CI: 0.88, 0.98). Subgroup analysis results suggested that there was an inverse association between breast cancer risk and posterori-derived healthy patterns, but no statistically significant associations were found in other stratified subgroups (a priori-derived diet, study region, menopausal status, or breast cancer subtypes). Healthy dietary patterns were associated inversely with all-cause mortality (RR = 0.76, 95% CI: 0.63, 0.92); however, no association was found for breast cancer-specific mortality.
Conclusions
The results suggested that healthy dietary patterns might be associated with a reduced risk of breast cancer and all-cause mortality among breast cancer patients. It could be clinically relevant to promote healthy dietary patterns for breast cancer prevention and improve survival among breast cancer patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Breast cancer is the second leading cause of cancer deaths in women worldwide [1]. Also, there are over 3 million breast cancer survivors in the USA [2]. Diet has been investigated in many breast cancer studies mostly focusing on single nutrients or food groups [3,4,5,6,7,8,9,10,11,12]. However, the associations with most nutrients and food groups have been inconclusive [13,14,15,16], except for the consistent and positive associations between alcohol and breast cancer [17]. As foods and nutrients are rarely eaten in isolation, the roles of individual dietary components should be considered within the context of overall dietary quality [18]. Moreover, it is advised by American Cancer Society Guidelines for cancer prevention to consume whole foods following an overall healthy dietary pattern, which is characterized as high intake of vegetables, fruits, whole grains, and limited consumption of processed meat and red meat [19]. Several studies have examined the association between dietary patterns and breast cancer risk [20,21,22,23,24,25], but the evidence is still inconclusive [26]. As results of new research have been published [21, 27,28,29,30,31,32] since the latest reviews [33,34,35], we aimed to conduct a meta-analysis to estimate a pooled association of healthy dietary patterns with breast cancer risk to incorporate recently published prospective cohort studies on breast cancer risk and dietary patterns.
The inconsistency of association between dietary patterns and breast cancer risk might be due to that dietary patterns have different effects for populations with varied characteristics. Previous epidemiological studies have reported heterogeneous results on the association between dietary patterns and breast cancer subtypes and menopausal status [22, 36,37,38,39,40]. However, the most recent meta-analysis has only included evidence of a posterori dietary pattern with breast cancer risk stratified by menopausal status and breast cancer subtypes [35]. Therefore, we would like to include a priori dietary pattern evidence in the meta-analysis [41], and to examine the association of healthy dietary patterns with breast cancer risk stratified by breast cancer subtypes, menopausal status, study region, and methods to derive dietary patterns. It would be beneficial to know the effects of certain dietary patterns on different subgroups of populations.
There is increasing recognition of the potential significance of nutrition in cancer survival and cancer survivors are likely to have special nutrition requirements [26]. However, only a few studies reported relationships between healthy dietary patterns and breast cancer survival with inconsistent results [42,43,44,45,46]. Recent systematic reviews among breast cancer survivors concluded that no factors were convincing for reducing breast cancer mortality partly due to limited number of studies [43, 47, 48]. Thus, we also conducted a meta-analysis to incorporate the up-to-date evidence on healthy dietary patterns and breast cancer survival.
Methods
Search strategy
For meta-analysis, the PRISMA (Preferred Reporting Items for Systematic reviews and Meta-Analyses) guideline was followed [49]. Cohort studies that examined diet and breast cancer risk and/or breast cancer survival were systematically searched for in PubMed, EMBASE, and Web of Science until June 24th, 2018. The search terms used in each database are shown in Supplemental Table 1. Additionally, we manually searched for studies listed in review papers, as there may be studies not included in the database. The language of the articles was limited to English.
Data selection and extraction
Study selection was conducted in three steps. First, the titles and abstracts of studies identified in our literature search were independently reviewed by two reviewers (RH, JW). Second, the full texts of studies retrieved in the initial screening were independently reviewed by two reviewers (RH, JW). Any disagreement between the researchers during the study selection period was resolved through discussion with a third researcher (YH). Third, data from studies that met inclusion and exclusion criteria (Supplemental Table 2) were extracted independently by three reviewers (RH, JW, YH), including the first author, year of publication, study name, study’s country, sample size, number of cases, diet assessment methods, dietary pattern development methods, labels of the identified dietary patterns and the food components, follow-up years, means and standard deviations of the outcomes, and covariates adjusted in the analysis. If there were multiple articles based on the same cohort, the article with the largest sample size was used for the overall meta-analysis and other articles within the same cohort may be used for subgroup analysis. Corresponding authors of articles with missing information were contacted.
The classification of healthy dietary patterns was based on the authors’ description from selected published articles. When two or more healthy dietary patterns were presented, researchers generally considered a healthy pattern that mostly include protective foods, such as vegetables, fruits, fish, legumes, whole grains, and low-fat dairy products. When multiple dietary guideline indices were used to derive dietary patterns, we chose Mediterranean diet for European studies as it might be more appropriate for the European population, and Dietary Approaches to Stop Hypertension (DASH) diet for U.S. studies as it ranks as top overall healthy diets by U.S. News and World Report compared to Alternative Healthy Eating Index (AHEI) or Healthy Eating Index (HEI) [50, 51]. All discrepancies were resolved through discussion by three reviewers (RH, JW, YH) for the choices of healthy dietary patterns into the meta-analysis.
Quality assessment
The Newcastle–Ottawa Scale was used for quality assessment [52]. The Newcastle–Ottawa Scale evaluates quality of cohort studies in three domains: selection of exposed and unexposed cohorts (representativeness of the exposed cohort, selection of the unexposed cohort, ascertainment of exposure, and demonstration of absence of outcome at the beginning of studies), comparability of exposed and unexposed cohorts (analysis appropriately adjusted for potential confounding factors, including the most important and additional factors), and outcome ascertainment (adequacy of outcome assessment, length of follow-up, and adequacy of follow-up). A study was given a maximum of 1 point for selection and outcome domains and a maximum of two points for comparability for a possible maximum total score of 9, with higher scores indicating higher quality. Three independent reviewers (RH, JW, YH) assessed the quality of all included studies. All reviewers agreed on the score of quality assessment.
Statistical analysis
Estimates of associations such as hazard ratios (HRs), odds ratios (ORs), and risk ratios (RRs) of breast cancer and their corresponding 95% CIs were derived from included studies after full adjustment. HR and OR were considered equivalent to RR. To combine the results, a meta-analysis was conducted where we evaluated breast cancer risk or survival by comparing the highest category with lowest category of healthy dietary pattern. Random-effects meta-analysis models were used for included studies. The pooled RR is considered statistically significant if 95% CI did not contain 1. Publication bias was evaluated by Egger’s test [53]. Heterogeneity between studies was assessed using I2 tests. I2 statistic was calculated to quantify the proportion of between-study heterogeneity attributable to variability in the association rather than sampling variation. An I2 of > 50% was considered an indicator of substantial heterogeneity across studies. If results showed significant heterogeneity, potential sources of heterogeneity were explored by sensitivity analysis. Sensitivity analysis was conducted by excluding one study each time from the analysis, if evidence of significant heterogeneity across studies existed.
Subgroup analysis was also undertaken using random-effects model based on menopausal status, breast cancer subtypes (estrogen receptor negative (ER-) and ER-/progesterone receptor negative (PR-)), methods to identify dietary patterns (a priori and a posteriori), and regions (Europe and North America). We also conducted analysis for Mediterranean diet as five studies used this diet to derived healthy pattern. No subgroup analysis based on the type of a posteriori methods used was conducted as most studies used factor analysis [including principal component analysis (PCA)]. Also, other breast cancer subtypes was not analyzed due to limited number of studies (< 5). No subgroup analysis was conducted for survival due to limited total number of studies.
Stata 14 (Stata Corp, College Station, TX) was used to conduct meta-analysis and Egger’s test for publication bias.
Results
Baseline characteristics for included studies
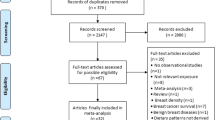
Initial database search yielded a total of 5130 records as shown in the flow chart (Supplemental Fig. 1). After excluding letters or reviews and eliminating duplicates, 3197 remained. After the abstract screening, 2813 studies were not relevant and therefore excluded. The 384 remaining reports were retrieved in full-text. Finally, a total of 32 articles were included in the meta-analysis, including 27 for risk (19 cohorts) and 5 for survival (5 cohorts), and ten of them were published after 2012. In total, the 19 cohorts for overall breast cancer risk meta-analysis included 1,143,395 women with 38,098 incident cases of breast cancer (Supplemental Table 3) and the five studies for breast cancer survival meta-analysis included 13,443 women diagnosed with breast cancer (Supplemental Table 4).
Among the 27 included articles for breast cancer risk, there were 19 cohorts in total for overall meta-analysis. Out of the 27 published literature (19 cohorts), 11 cohorts were evaluated for pre-menopausal and 11 cohorts were for post- menopausal women; 7 were identified as priori and 15 as posterior dietary patterns; six were from Europe, eight from North America, and the other five cohorts were not grouped together for analysis due to the heterogeneity in food culture and diet; five reported ER- breast cancer and five reported ER-/PR-breast cancer.
Quality
The quality of each study regarding selection, comparability, and outcome is summarized in Supplemental Table 5. Overall quality of included studies was adequate, with a range of score from 7 to 9. For selection, eight studies were scored “0” for cohort representation, with 6 of them using Nurses’ Health Study (NHS) [31, 38, 54,55,56] or NHS-II [57], one recruited teachers in California [58] and another one study only recruited participants from one province in Italy [59]. Five studies did not specifically mention that breast cancer was not present at the start of the study [21, 24, 55, 59, 60]. The overall comparability for all studies was good with only two studies scores “1” for adjusting only major covariates [21, 61]. For outcome, three studies have not reported statement about their follow-up adequacy but all studies have appropriate length of follow-up and outcome assessment methods [32, 62, 63].
Overall breast cancer risk
The overall results of 19 cohorts from quality-effects model suggested that there was a statistically significant lower risk of breast cancer associated with healthy dietary patterns (RR = 0.93, 95% CI: 0.88, 0.98) (Fig. 1). There was no apparent evidence of heterogeneity and no indication of publication bias for the meta-analysis (Egger’s p = 0.90).
Forest plot of the highest compared with the lowest categories of intake of healthy dietary patterns and risk of breast cancer. AHS-2 Adventist Health Study-2, BCDDP Breast Cancer Detection Demonstrati on Project, BWHS Black Women’s Health Study, CI Confidence Interval, CSDLH Canadian Study of Diet, Lifestyle and Health, CTS California Teachers Study, EPIC European Prospective Investigation into Cancer and Nutrition, ES Effect Size, JACC Japan Collaborative Cohort Study, JPHC Japan Public Health Center based on Prospective Study, MCCS Melbourne Collaborative Cohort Study, NBSS National Breast Screening Study, NHS Nurses’ Health Study, NHS-II Nurse’ Health Study II, NLCS Netherlands Cohort Study on diet and cancer, ORDET Ormoni e Dieta nella Eziologia dei Tumori in Italy, SCHS Singapore Chinese Healthy Study, SMC Swedish Mammography Cohort, SWLH Swedish Women’s Lifestyle and Health, UKWCS UK Women’s Cohort Study
Breast cancer survival
All-cause mortality
The meta-analysis of 5 cohort studies indicated that there was an inverse association between healthy dietary patterns and all-cause mortality (RR = 0.76, 95% CI: 0.63, 0.92), with no evidence of heterogeneity (Fig. 2). Egger’s test suggested that possible publication bias for all-cause mortality existed among studies (Egger’s p = 0.04).
Forest plot of the highest compared with the lowest categories of intake of healthy dietary patterns and all-cause mortality. CPS-II Cancer Prevention Study–II Nutrition Cohort, ES Effect Size, HEAL Health, Eating, Activity and Lifestyle, LACE Life After Cancer Epidemiology Study, NHS Nurses’ Health Study
Breast cancer mortality
No statistically significant association was found between dietary patterns and breast cancer mortality (RR = 0.91, 95% CI: 0.65, 1.28) and there was evidence of high heterogeneity (Fig. 3). With sensitivity analysis, the heterogeneity is still high (Supplemental Table 6). It might suggest that limited number of research is available and more studies are needed. Egger’s test suggested no potential publication bias for breast cancer mortality (Egger’s p = 0.86).
Forest plot of the highest compared with the lowest categories of intake of healthy dietary patterns and breast cancer mortality. CPS-II Cancer Prevention Study–II Nutrition Cohort, ES Effect Size, HEAL Health, Eating, Activity and Lifestyle, LACE Life After Cancer Epidemiology Study, NHS Nurses’ Health Study
Subgroup analysis
Subgroup analysis results are shown in Table 1. We found an inverse association between healthy dietary patterns and breast cancer risk in cohorts using posteriori methods to derive dietary patterns. However, no statistically significant associations were found between the risk of breast cancer and a priori healthy dietary patterns or Mediterranean diet. Also, no significant association was found between healthy dietary patterns and risk of breast cancer by study region, breast cancer subtypes (including ER- subgroup or ER-/PR- subgroup), or menopausal status (Figs. 4, 5).
Forest plot of the highest compared with the lowest categories of intake of healthy dietary patterns and risk of breast cancer in postmenopausal women. AHS-2 Adventist Health Study-2, BWHS Black Women’s Health Study, CI Confidence Interval, CSDLH Canadian Study of Diet, Lifestyle and Health, EPIC European Prospective Investigation into Cancer and Nutrition, ES Effect Size, JACC Japan Collaborative Cohort Study, JPHC Japan Public Health Center based on Prospective Study, NBSS National Breast Screening Study, NHS Nurses’ Health Study, SCHS Singapore Chinese Healthy Study, SWLH Swedish Women’s Lifestyle and Health, UKWCS UK Women’s Cohort Study
Forest plot of the highest compared with the lowest categories of intake of healthy dietary patterns and risk of breast cancer in pre-menopausal women. AHS-2 Adventist Health Study-2, BWHS Black Women’s Health Study, CI Confidence Interval, CSDLH Canadian Study of Diet, Lifestyle and Health, EPIC European Prospective Investigation into Cancer and Nutrition, ES Effect Size, JACC Japan Collaborative Cohort Study, JPHC Japan Public Health Center based on Prospective Study, NBSS National Breast Screening Study, NHS-II Nurse’ Health Study II, SCHS Singapore Chinese Healthy Study
Discussion
Our updated meta-analysis indicated that healthy dietary patterns may decrease breast cancer risk, which is in agreement with previously published systematic reviews [33, 34, 64]. Moreover, there was an inverse association for posteriori-derived dietary patterns, but no modification effects were found by menopausal status, study regions, and breast cancer subtypes. Among breast cancer patients, an inverse association was found between healthy dietary patterns and all-cause mortality, but no association was observed between the healthy dietary pattern and breast cancer mortality.
The healthy dietary patterns, characterized by high intake of vegetables, fruit, and food with a low fat content, are promoted by the World Cancer Research Fund (WCRF) to reduce cancer risk [26] and align with American Cancer Society Guidelines [19]. These foods contain substances with anti-carcinogenic and anti-inflammation properties, which could have protective functions through antioxidant effects on estrogen metabolism and cell proliferation reduction [65, 66]. One epidemiological study suggested an inverse association between high fruits and vegetables consumption with breast cancer risk [67]. Higher dietary inflammatory potential score was also found to be associated with greater breast cancer, suggesting that modifying inflammatory characteristics of diet can reduce the risk of breast cancer [68]. Other plausible reason that diet plays a role in breast cancer might be due to its contribution to obesity pathways or epigenetic alterations affecting carcinogenesis [69,70,71].
In a posteriori subgroup analysis, the inverse association between a healthy dietary pattern and breast cancer risk was found, while no association was found in a priori subgroup. It has been found that dietary patterns derived by a posteriori analysis have reasonable reproducibility and validity using data from food frequency questionnaire (FFQ), which could minimize the risk of bias and probably results in the inverse association found [41]. It is also possible that we found no association in a priori subgroup due to the heterogeneity in dietary patterns used across studies and limited number of studies, which suggests the need of a more specific dietary guideline created for breast cancer prevention.
Although some studies reported modification effects by menopausal status between breast cancer and foods [36], no significant effects were found in our study, which is consistent with the latest review on dietary pattern and cancer risk [72]. Moreover, no significant associations were found in our analysis for ER- group or ER-/PR- group even though different breast cancers subtypes have different molecular, pathologic, and clinical features [73]. It is possible that we are unable to identify the association due to limited number of studies and the high heterogeneity between studies. These studies were conducted in different regions with different dietary patterns influenced by culture and in different FFQ. Moreover, it is found that different techniques to measure ER and PR have limitations and it is likely to have different techniques in cohorts of different regions [73].
As dietary pattern is strongly related to cultural habits and differs by country or ethnicity and variables like cooking methods may not be captured, subgroup analysis based on the study region was conducted as surrogate since no point estimates were provided by ethnic group in the original articles. However, no association between healthy dietary patterns and breast cancer risk was found in North America or Europe. It is possible that we are underpowered within each region with six studies in Europe and eight studies in North America. We did not conduct analysis in different regions of Asia due to limited number of studies and diverse dietary patterns of various cultures.
For breast cancer survival, an inverse association was found between healthy dietary patterns and all-cause mortality, but no association was shown with breast cancer-specific mortality in our analysis. Our findings are consistent to a recent systematic review, where better overall dietary intake was found to be associated with decreased risk of overall mortality but insufficient to draw conclusions regarding breast cancer-specific survival [48]. One previous study has also shown that healthy diet was not associated with breast cancer-specific mortality but reduces risk of overall mortality in women who had been previously treated for early-stage breast cancer [43]. It could indicate that although dietary habits may not influence breast cancer-related outcomes for breast cancer patients, they could still play an important role in etiology of overall health [74,75,76]. Moreover, better diet quality is associated with improved physical functioning and vitality in cancer survivors [77]. Furthermore, sample size, follow-up duration, measurement errors in dietary assessment, and stage of breast cancer may also affect the results [78,79,80,81]. Our results are consistent with dietary guidelines directed towards the general population for overall chronic disease or cancer prevention [40, 82, 83], which indicates that women diagnosed with breast cancer may benefit from healthy dietary patterns and improve their survival rates. Five cohorts were used between healthy dietary patterns and breast cancer survival meta-analysis, and the small number of studies underscores the limited publications on the research topic. Therefore, representativeness of the findings cannot be ensured.
Our meta-analysis has several strengths. First, we only included evidence from prospective cohort studies as case–control studies are more likely to be influenced by recall bias and less adept at showing a causal relationship compared to prospective cohort studies [84]. Second, we examined the association based on menopausal status, breast cancer subtypes, methods to derive dietary and region, which could provide novel knowledge to the relationship between dietary patterns and breast cancer. There are also some limitations. First, the findings are directly driven by the included studies, and those studies have their own strengths and limitations in terms of study design, e.g., measurement errors for individual intake or no dietary pattern data in childhood which could miss a critical period for breast cancer prevention. Also, not including alternate studies in the same cohort (those with smaller sample size than the included ones) may lose some information and affect the results. Second, we only compared the risk estimates or mortality between the highest and lowest categories of healthy dietary patterns. The middle categories were not considered in the current analysis and the highest category of one study could be considered as low in another study; therefore, we cannot recommend an absolute intake/category to achieve the beneficial effects of healthy diet. However, the relative categories within the same study could still provide useful information about relative effects of healthy dietary patterns. Moreover, the dietary assessment methods were different among studies although the majority of studies used FFQ, which could introduce heterogeneity. This tool is known to be subject to substantial amounts of random and systematic variability [85, 86]. It is possible that the associations shown in this analysis was attenuated to the null due to non-differential misclassification. However, FFQs are commonly used in nutrition studies and mostly validated in the studies. Additionally, only single time-point measurements of dietary patterns were examined in the included studies, and these do not account for changes in eating habits over time, which might be especially relevant to cancer development. More studies with multiple measurements of dietary patterns are needed to better address the effects of diet on breast cancer risk. Lastly, we have only included published studies and studies published in English.
In conclusion, our results provide evidence of an overall inverse association between a healthy dietary pattern and breast cancer risk. An inverse association between a healthy dietary pattern and all-cause mortality was found among breast cancer patients. It could be clinically relevant to promote healthy dietary patterns for breast cancer prevention and provide guidance for nutritional care of breast cancer patients. Further investigation is needed to better understand the mechanism between dietary patterns and breast cancer and how dietary patterns affect people differently by menopausal status and breast cancer subtypes. Future studies involving large scale randomized controlled trials or carefully designed observational studies are required to get more definitive conclusions.
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D (2011) Global cancer statistics. Cancer J Clin 61(2):69–90
Miller KD, Siegel RL, Lin CC, Mariotto AB, Kramer JL, Rowland JH, Stein KD, Alteri R, Jemal A (2016) Cancer treatment and survivorship statistics. Cancer J Clin 66(4):271–289. https://doi.org/10.3322/caac.21349
Hauner H, Hauner D (2010) The impact of nutrition on the development and prognosis of breast cancer. Breast Care 5(6):377–381
Wakai K, Tamakoshi K, Fukui M, Suzuki S, Lin Y, Niwa Y, Nishio K, Yatsuya H, Kondo T, Tokudome S (2005) Dietary intakes of fat and fatty acids and risk of breast cancer: a prospective study in Japan. Cancer Sci 96(9):590–599
Smith-Warner SA, Spiegelman D, Yaun S-S, Adami H-O, Beeson WL, Van Den Brandt PA, Folsom AR, Fraser GE, Freudenheim JL, Goldbohm RA (2001) Intake of fruits and vegetables and risk of breast cancer: a pooled analysis of cohort studies. JAMA 285(6):769–776
Rohan T, McMichael A, Baghurst P (1988) A population-based case–control study of diet and breast cancer in Australia. Am J Epidemiol 128(3):478–489
Löf M, Sandin S, Lagiou P, Hilakivi-Clarke L, Trichopoulos D, Adami H, Weiderpass E (2007) Dietary fat and breast cancer risk in the Swedish women’s lifestyle and health cohort. Br J Cancer 97(11):1570–1576
Lima FELd, Latorre MdRDd, Costa MJdC, Fisberg RM (2008) Diet and cancer in Northeast Brazil: evaluation of eating habits and food group consumption in relation to breast cancer. Cadernos de Saúde Pública 24(4):820–828
Howe GR, Hirohata T, Hislop TG, Iscovich JM, Yuan J-M, Katsouyanni K, Lubin F, Marubini E, Modan B, Rohan T (1990) Dietary factors and risk of breast cancer: combined analysis of 12 case—control studies. J Natl Cancer Inst 82(7):561–569
Gandini S, Merzenich H, Robertson C, Boyle P (2000) Meta-analysis of studies on breast cancer risk and diet: the role of fruit and vegetable consumption and the intake of associated micronutrients. Eur J Cancer 36(5):636–646
Freudenheim JL, Marshall JR, Vena JE, Laughlin R, Brasure JR, Swanson MK, Nemoto T, Graham S (1996) Premenopausal breast cancer risk and intake of vegetables, fruits, and related nutrients. J Natl Cancer Inst 88(6):340–348
Franceschi S, Favero A, Russo A, Decarli A, La Vecchia C, Ferraroni M, Negri E, Amadori D, Conti E, Montella M (1996) Intake of macronutrients and risk of breast cancer. The Lancet 347(9012):1351–1356
Van Gils CH, Peeters PH, Bueno-de-Mesquita HB, Boshuizen HC, Lahmann PH, Clavel-Chapelon F, Thiébaut A, Kesse E, Sieri S, Palli D (2005) Consumption of vegetables and fruits and risk of breast cancer. JAMA 293(2):183–193
Michels KB, Mohllajee AP, Roset-Bahmanyar E, Beehler GP, Moysich KB (2007) Diet and breast cancer. Cancer 109(S12):2712–2749
Jung S, Spiegelman D, Baglietto L, Bernstein L, Boggs DA, Van Den Brandt PA, Buring JE, Cerhan JR, Gaudet MM, Giles GG (2013) Fruit and vegetable intake and risk of breast cancer by hormone receptor status. J Natl Cancer Inst 105(3):219–236
Hunter DJ, Spiegelman D, Adami H-O, Beeson L, Van Den Brandt PA, Folsom AR, Fraser GE, Goldbohm RA, Graham S, Howe GR (1996) Cohort studies of fat intake and the risk of breast cancer—a pooled analysis. N Engl J Med 334(6):356–361
Smith-Warner SA, Spiegelman D, Yaun S-S, Van Den Brandt PA, Folsom AR, Goldbohm RA, Graham S, Holmberg L, Howe GR, Marshall JR (1998) Alcohol and breast cancer in women: a pooled analysis of cohort studies. JAMA 279(7):535–540
Rock CL, Doyle C, Demark-Wahnefried W, Meyerhardt J, Courneya KS, Schwartz AL, Bandera EV, Hamilton KK, Grant B, McCullough M (2012) Nutrition and physical activity guidelines for cancer survivors. Cancer J Clin 62(4):242–274
Kushi LH, Doyle C, McCullough M, Rock CL, Demark-Wahnefried W, Bandera EV, Gapstur S, Patel AV, Andrews K, Gansler T (2012) American Cancer Society Guidelines on nutrition and physical activity for cancer prevention: reducing the risk of cancer with healthy food choices and physical activity. Cancer J Clin 62(1):30–67. https://doi.org/10.3322/caac.20140
Couto E, Sandin S, Lof M, Ursin G, Adami HO, Weiderpass E (2013) Mediterranean dietary pattern and risk of breast cancer. PLoS ONE 8(2):e55374. https://doi.org/10.1371/journal.pone.0055374
Catsburg C, Kim RS, Kirsh VA, Soskolne CL, Kreiger N, Rohan TE (2015) Dietary patterns and breast cancer risk: a study in 2 cohorts. Am J Clin Nutr 101(4):817–823. https://doi.org/10.3945/ajcn.114.097659
Cottet V, Touvier M, Fournier A, Touillaud MS, Lafay L, Clavel-Chapelon F, Boutron-Ruault M-C (2009) Postmenopausal breast cancer risk and dietary patterns in the E3 N-EPIC prospective cohort study. Am J Epidemiol 170(10):1257–1267
Cade J, Taylor E, Burley V, Greenwood D (2011) Does the mediterranean dietary pattern or the healthy diet index influence the risk of breast cancer in a large British cohort of women? Eur J Clin Nutr 65(8):920–928
Männistö S, Dixon LB, Balder HF, Virtanen MJ, Krogh V, Khani BR, Berrino F, Van Den Brandt PA, Hartman AM, Pietinen P (2005) Dietary patterns and breast cancer risk: results from three cohort studies in the DIETSCAN project. Cancer Causes Control 16(6):725–733
Baglietto L, Krishnan K, Severi G, Hodge A, Brinkman M, English D, McLean C, Hopper J, Giles G (2011) Dietary patterns and risk of breast cancer. Br J Cancer 104(3):524–531
World Cancer Research Fund AIfCR (2018) Diet, nutrition, physical activity and cancer: a global perspective. www.dietandcancerreport.org. Accessed 31 Dec 2018
Li YJ, Roswall N, Sandin S, Strom P, Adami HO, Weiderpass E (2015) Adherence to a healthy nordic food index and breast cancer risk: results from a Swedish cohort study. Cancer Causes Control 26(6):893–902. https://doi.org/10.1007/s10552-015-0564-x
Kojima R, Okada E, Ukawa S, Mori M, Wakai K, Date C, Iso H, Tamakoshi A (2017) Dietary patterns and breast cancer risk in a prospective Japanese study. Breast Cancer 24(1):152–160. https://doi.org/10.1007/s12282-016-0689-0
van den Brandt PA, Schulpen M (2017) Mediterranean diet adherence and risk of postmenopausal breast cancer: results of a cohort study and meta-analysis. Int J Cancer 140(10):2220–2231. https://doi.org/10.1002/ijc.30654
Shin S, Saito E, Inoue M, Sawada N, Ishihara J, Takachi R, Nanri A, Shimazu T, Yamaji T, Iwasaki M, Sasazuki S, Tsugane S (2016) Dietary pattern and breast cancer risk in Japanese women: the Japan Public Health Center-based Prospective study (JPHC Study). The British journal of nutrition 115(10):1769–1779. https://doi.org/10.1017/s0007114516000684
Hirko KA, Willett WC, Hankinson SE, Rosner BA, Beck AH, Tamimi RM, Eliassen AH (2016) Healthy dietary patterns and risk of breast cancer by molecular subtype. Breast Cancer Res Treat 155(3):579–588. https://doi.org/10.1007/s10549-016-3706-2
Penniecook-Sawyers JA, Jaceldo-Siegl K, Fan J, Beeson L, Knutsen S, Herring P, Fraser GE (2016) Vegetarian dietary patterns and the risk of breast cancer in a low-risk population. Br J Nutr 115(10):1790–1797. https://doi.org/10.1017/s0007114516000751
Brennan SF, Cantwell MM, Cardwell CR, Velentzis LS, Woodside JV (2010) Dietary patterns and breast cancer risk: a systematic review and meta-analysis. Am J Clin Nutr 91(5):1294–1302. https://doi.org/10.3945/ajcn.2009.28796
Albuquerque RC, Baltar VT, Marchioni DM (2014) Breast cancer and dietary patterns: a systematic review. Nutr Rev 72(1):1–17. https://doi.org/10.1111/nure.12083
Grosso G, Bella F, Godos J, Sciacca S, Del Rio D, Ray S, Galvano F, Giovannucci EL (2017) Possible role of diet in cancer: systematic review and multiple meta-analyses of dietary patterns, lifestyle factors, and cancer risk. Nutr Rev 75(6):405–419. https://doi.org/10.1093/nutrit/nux012
Gaudet MM, Britton JA, Kabat GC, Steck-Scott S, Eng SM, Teitelbaum SL, Terry MB, Neugut AI, Gammon MD (2004) Fruits, vegetables, and micronutrients in relation to breast cancer modified by menopause and hormone receptor status. Cancer Epidemiol Prev Biomark 13(9):1485–1494
Fung TT, Hu FB, Holmes MD, Rosner BA, Hunter DJ, Colditz GA, Willett WC (2005) Dietary patterns and the risk of postmenopausal breast cancer. Int J Cancer 116(1):116–121
Fung TT, Hu FB, McCullough ML, Newby P, Willett WC, Holmes MD (2006) Diet quality is associated with the risk of estrogen receptor–negative breast cancer in postmenopausal women. J Nutr 136(2):466–472
Olsen A, Tjønneland A, Thomsen BL, Loft S, Stripp C, Overvad K, Møller S, Olsen JH (2003) Fruits and vegetables intake differentially affects estrogen receptor negative and positive breast cancer incidence rates. J Nutr 133(7):2342–2347
Marmot M, Atinmo T, Byers T, Chen J, Hirohata T, Jackson A, James W, Kolonel L, Kumanyika S, Leitzmann C (2007) Food, nutrition, physical activity, and the prevention of cancer: a global perspective. World Cancer Research Fund / American Institute for Cancer Research, Washington DC
Hu FB (2002) Dietary pattern analysis: a new direction in nutritional epidemiology. Curr Opin Lipidol 13(1):3–9
McCullough ML, Gapstur SM, Shah R, Campbell PT, Wang Y, Doyle C, Gaudet MM (2016) Pre-and postdiagnostic diet in relation to mortality among breast cancer survivors in the CPS-II Nutrition Cohort. Cancer Causes Control 27(11):1303–1314
Kwan ML, Weltzien E, Kushi LH, Castillo A, Slattery ML, Caan BJ (2009) Dietary patterns and breast cancer recurrence and survival among women with early-stage breast cancer. J Clin Oncol 27(6):919–926. https://doi.org/10.1200/jco.2008.19.4035
George SM, Irwin ML, Smith AW, Neuhouser ML, Reedy J, McTiernan A, Alfano CM, Bernstein L, Ulrich CM, Baumgartner KB (2011) Postdiagnosis diet quality, the combination of diet quality and recreational physical activity, and prognosis after early-stage breast cancer. Cancer Causes Control 22(4):589–598
Vrieling A, Buck K, Seibold P, Heinz J, Obi N, Flesch-Janys D, Chang-Claude J (2013) Dietary patterns and survival in German postmenopausal breast cancer survivors. Br J Cancer 108(1):188–192
Kroenke CH, Fung TT, Hu FB, Holmes MD (2005) Dietary patterns and survival after breast cancer diagnosis. J Clin Oncol 23(36):9295–9303
World Cancer Research Fund International AIfCR (2014) Continuous update project report: diet, nutrition, physical activity, and breast cancer survivors. World Cancer Research Fund International, London
Terranova CO, Protani MM, Reeves MM (2018) Overall dietary intake and prognosis after breast cancer: a systematic review. Nutr Cancer 70(2):153–163. https://doi.org/10.1080/01635581.2018.1412478
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J, Moher D (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med 6(7):e1000100
Giacosa A, Barale R, Bavaresco L, Gatenby P, Gerbi V, Janssens J, Johnston B, Kas K, La Vecchia C, Mainguet P, Morazzoni P, Negri E, Pelucchi C, Pezzotti M, Rondanelli M (2013) Cancer prevention in Europe: the Mediterranean diet as a protective choice. Eur J Cancer Prev 22(1):90–95. https://doi.org/10.1097/CEJ.0b013e328354d2d7
Onvani S, Haghighatdoost F, Azadbakht L (2015) Dietary approach to stop hypertension (DASH): diet components may be related to lower prevalence of different kinds of cancer: a review on the related documents. J Res Med Sci 20(7):707–713. https://doi.org/10.4103/1735-1995.166233
Peterson J, Welch V, Losos M, Tugwell P (2011) The Newcastle-Ottawa scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Ottawa Hospital Research Institute, Ottawa
Egger M, Smith GD, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315(7109):629–634
Fung TT, Hu FB, Holmes MD, Rosner BA, Hunter DJ, Colditz GA, Willett WC (2005) Dietary patterns and the risk of postmenopausal breast cancer. Int J Cancer 116(1):116–121. https://doi.org/10.1002/ijc.20999
Fung TT, Schulze MB, Hu FB, Hankinson SE, Holmes MD (2012) A dietary pattern derived to correlate with estrogens and risk of postmenopausal breast cancer. Breast Cancer Res Treat 132(3):1157–1162. https://doi.org/10.1007/s10549-011-1942-z
Izano MA, Fung TT, Chiuve SS, Hu FB, Holmes MD (2013) Are diet quality scores after breast cancer diagnosis associated with improved breast cancer survival? Nutr Cancer 65(6):820–826. https://doi.org/10.1080/01635581.2013.804939
Adebamowo CA, Hu FB, Cho E, Spiegelman D, Holmes MD, Willett WC (2005) Dietary patterns and the risk of breast cancer. Ann Epidemiol 15(10):789–795. https://doi.org/10.1016/j.annepidem.2005.01.008
Link LB, Canchola AJ, Bernstein L, Clarke CA, Stram DO, Ursin G, Horn-Ross PL (2013) Dietary patterns and breast cancer risk in the California teachers study cohort. Am J Clin Nutr 98(6):1524–1532. https://doi.org/10.3945/ajcn.113.061184
Sant M, Allemani C, Sieri S, Krogh V, Menard S, Tagliabue E, Nardini E, Micheli A, Crosignani P, Muti P, Berrino F (2007) Salad vegetables dietary pattern protects against HER-2-positive breast cancer: a prospective Italian study. Int J Cancer 121(4):911–914. https://doi.org/10.1002/ijc.22714
Sieri S, Krogh V, Pala V, Muti P, Micheli A, Evangelista A, Tagliabue G, Berrino F (2004) Dietary patterns and risk of breast cancer in the ORDET cohort. Cancer epidemiol Biomark Prev 13(4):567–572
Terry P, Suzuki R, Hu FB, Wolk A (2001) A prospective study of major dietary patterns and the risk of breast cancer. Cancer Epidemiol Biomark Prev 10(12):1281–1285
Butler LM, Wu AH, Wang R, Koh WP, Yuan JM, Yu MC (2010) A vegetable-fruit-soy dietary pattern protects against breast cancer among postmenopausal Singapore Chinese women. Am J Clin Nutr 91(4):1013–1019. https://doi.org/10.3945/ajcn.2009.28572
Cade JE, Taylor EF, Burley VJ, Greenwood DC (2011) Does the Mediterranean dietary pattern or the healthy diet index influence the risk of breast cancer in a large British cohort of women? Eur J Clin Nutr 65(8):920–928. https://doi.org/10.1038/ejcn.2011.69
Edefonti V, Randi G, La Vecchia C, Ferraroni M, Decarli A (2009) Dietary patterns and breast cancer: a review with focus on methodological issues. Nutr Rev 67(6):297–314. https://doi.org/10.1111/j.1753-4887.2009.00203.x
Steinmetz KA, Potter JD (1991) Vegetables, fruit, and cancer II. Mechanisms. Cancer Causes Control 2(6):427–442
Maskarinec G, Morimoto Y, Takata Y, Murphy SP, Stanczyk FZ (2006) Alcohol and dietary fibre intakes affect circulating sex hormones among premenopausal women. Public health nutrition 9(7):875–881
Aune D, Chan D, Vieira A, Rosenblatt DN, Vieira R, Greenwood D, Norat T (2012) Fruits, vegetables and breast cancer risk: a systematic review and meta-analysis of prospective studies. Breast Cancer Res Treat 134(2):479–493
Zahedi H, Djalalinia S, Sadeghi O, Asayesh H, Noroozi M, Gorabi AM, Mohammadi R, Qorbani M (2018) Dietary inflammatory potential score and risk of breast cancer: systematic review and meta-analysis. Clin Breast Cancer 18:e561–e570. https://doi.org/10.1016/j.clbc.2018.01.007
Ostan R, Lanzarini C, Pini E, Scurti M, Vianello D, Bertarelli C, Fabbri C, Izzi M, Palmas G, Biondi F, Martucci M, Bellavista E, Salvioli S, Capri M, Franceschi C, Santoro A (2015) Inflammaging and cancer: a challenge for the mediterranean diet. Nutrients 7(4):2589–2621. https://doi.org/10.3390/nu7042589
Cappellani A, Di Vita M, Zanghi A, Cavallaro A, Piccolo G, Veroux M, Berretta M, Malaguarnera M, Canzonieri V, Lo Menzo E (2012) Diet, obesity and breast cancer: an update. Front Biosci 4:90–108
Nystrom M, Mutanen M (2009) Diet and epigenetics in colon cancer. World J Gastroenterol 15(3):257–263
Trichopoulou A, Bamia C, Lagiou P, Trichopoulos D (2010) Conformity to traditional mediterranean diet and breast cancer risk in the Greek EPIC (European Prospective Investigation into Cancer and Nutrition) cohort. Am J Clin Nutr 92(3):620–625. https://doi.org/10.3945/ajcn.2010.29619
Althuis MD, Fergenbaum JH, Garcia-Closas M, Brinton LA, Madigan MP, Sherman ME (2004) Etiology of hormone receptor-defined breast cancer: a systematic review of the literature. Cancer Epidemiol Biomark Prev 13(10):1558–1568
Harris EE, Correa C, Hwang W-T, Liao J, Litt HI, Ferrari VA, Solin LJ (2006) Late cardiac mortality and morbidity in early-stage breast cancer patients after breast-conservation treatment. J Clin Oncol 24(25):4100–4106
Brunner EJ, Mosdøl A, Witte DR, Martikainen P, Stafford M, Shipley MJ, Marmot MG (2008) Dietary patterns and 15-y risks of major coronary events, diabetes, and mortality. Am J Clin Nutr 87(5):1414–1421
Heidemann C, Schulze MB, Franco OH, van Dam RM, Mantzoros CS, Hu FB (2008) Dietary patterns and risk of mortality from cardiovascular disease, cancer, and all causes in a prospective cohort of women. Circulation 118(3):230–237
Mosher CE, Sloane R, Morey MC, Snyder DC, Cohen HJ, Miller PE, Demark-Wahnefried W (2009) Associations between lifestyle factors and quality of life among older long-term breast, prostate, and colorectal cancer survivors. Cancer 115(17):4001–4009
Thompson FE, Subar AF (2008) Dietary assessment methodology. Nutr Prev Treat Dis 2:3–39
Izano MA, Fung TT, Chiuve SS, Hu FB, Holmes MD (2013) Are diet quality scores after breast cancer diagnosis associated with improved breast cancer survival? Nutr Cancer 65(6):820–826
George SM, Ballard-Barbash R, Shikany JM, Caan BJ, Freudenheim JL, Kroenke CH, Vitolins MZ, Beresford SA, Neuhouser ML (2014) Better postdiagnosis diet quality is associated with reduced risk of death among postmenopausal women with invasive breast cancer in the Women’s Health Initiative. Cancer Epidemiol Prev Biomark 23:575–583
Kim EH, Willett WC, Fung T, Rosner B, Holmes MD (2011) Diet quality indices and postmenopausal breast cancer survival. Nutr Cancer 63(3):381–388
Doyle C, Kushi LH, Byers T, Courneya KS, Demark-Wahnefried W, Grant B, McTiernan A, Rock CL, Thompson C, Gansler T (2006) Nutrition and physical activity during and after cancer treatment: an American Cancer Society guide for informed choices. Cancer J Clin 56(6):323–353
US Department of Health and Human Services and US Department of Agriculture (2011) Dietary guidelines for Americans. US Gov Print Office, Washington, DC
Song JW, Chung KC (2010) Observational studies: cohort and case-control studies. Plast Reconstr Surg 126(6):2234–2242. https://doi.org/10.1097/PRS.0b013e3181f44abc
Schatzkin A, Kipnis V, Carroll RJ, Midthune D, Subar AF, Bingham S, Schoeller DA, Troiano RP, Freedman LS (2003) A comparison of a food frequency questionnaire with a 24-hour recall for use in an epidemiological cohort study: results from the biomarker-based Observing Protein and Energy Nutrition (OPEN) study. Int J Epidemiol 32(6):1054–1062
Kroke A, Klipstein-Grobusch K, Voss S, Moseneder J, Thielecke F, Noack R, Boeing H (1999) Validation of a self-administered food-frequency questionnaire administered in the European Prospective Investigation into Cancer and Nutrition (EPIC) Study: comparison of energy, protein, and macronutrient intakes estimated with the doubly labeled water, urinary nitrogen, and repeated 24-h dietary recall methods. Am J Clin Nutr 70(4):439–447. https://doi.org/10.1093/ajcn/70.4.439
Agurs-Collins T, Rosenberg L, Makambi K, Palmer JR, Adams-Campbell L (2009) Dietary patterns and breast cancer risk in women participating in the Black Women’s Health Study. Am J Clin Nutr 90(3):621–628. https://doi.org/10.3945/ajcn.2009.27666
Buckland G, Travier N, Cottet V, Gonzalez CA, Lujan-Barroso L, Agudo A, Trichopoulou A, Lagiou P, Trichopoulos D, Peeters PH, May A, Bueno-de-Mesquita HB, Bvan Duijnhoven FJ, Key TJ, Allen N, Khaw KT, Wareham N, Romieu I, McCormack V, Boutron-Ruault M, Clavel-Chapelon F, Panico S, Agnoli C, Palli D, Tumino R, Vineis P, Amiano P, Barricarte A, Rodriguez L, Sanchez MJ, Chirlaque MD, Kaaks R, Teucher B, Boeing H, Bergmann MM, Overvad K, Dahm CC, Tjonneland A, Olsen A, Manjer J, Wirfalt E, Hallmans G, Johansson I, Lund E, Hjartaker A, Skeie G, Vergnaud AC, Norat T, Romaguera D, Riboli E (2013) Adherence to the mediterranean diet and risk of breast cancer in the European prospective investigation into cancer and nutrition cohort study. Int J Cancer 132(12):2918–2927. https://doi.org/10.1002/ijc.27958
Fung TT, Hu FB, McCullough ML, Newby PK, Willett WC, Holmes MD (2006) Diet quality is associated with the risk of estrogen receptor-negative breast cancer in postmenopausal women. J Nutr 136(2):466–472. https://doi.org/10.1093/jn/136.2.466
Cottet V, Touvier M, Fournier A, Touillaud MS, Lafay L, Clavel-Chapelon F, Boutron-Ruault MC (2009) Postmenopausal breast cancer risk and dietary patterns in the E3 N-EPIC prospective cohort study. Am J Epidemiol 170(10):1257–1267. https://doi.org/10.1093/aje/kwp257
Velie EM, Schairer C, Flood A, He JP, Khattree R, Schatzkin A (2005) Empirically derived dietary patterns and risk of postmenopausal breast cancer in a large prospective cohort study. Am J Clin Nutr 82(6):1308–1319. https://doi.org/10.1093/ajcn/82.6.1308
Mai V, Kant AK, Flood A, Lacey JV Jr, Schairer C, Schatzkin A (2005) Diet quality and subsequent cancer incidence and mortality in a prospective cohort of women. Int J Epidemiol 34(1):54–60. https://doi.org/10.1093/ije/dyh388
Mannisto S, Dixon LB, Balder HF, Virtanen MJ, Krogh V, Khani BR, Berrino F, van den Brandt PA, Hartman AM, Pietinen P, Tan F, Wolk A, Goldbohm RA (2005) Dietary patterns and breast cancer risk: results from three cohort studies in the DIETSCAN project. Cancer Causes Control 16(6):725–733. https://doi.org/10.1007/s10552-005-1763-7
Baglietto L, Krishnan K, Severi G, Hodge A, Brinkman M, English DR, McLean C, Hopper JL, Giles GG (2011) Dietary patterns and risk of breast cancer. Br J Cancer 104(3):524–531. https://doi.org/10.1038/sj.bjc.6606044
Schulz M, Hoffmann K, Weikert C, Nothlings U, Schulze MB, Boeing H (2008) Identification of a dietary pattern characterized by high-fat food choices associated with increased risk of breast cancer: the European Prospective Investigation into Cancer and Nutrition (EPIC)-Potsdam Study. Br J Nutr 100(5):942–946. https://doi.org/10.1017/s0007114508966149
Harris HR, Bergkvist L, Wolk A (2015) An estrogen-associated dietary pattern and breast cancer risk in the Swedish mammography cohort. Int J Cancer 137(9):2149–2154. https://doi.org/10.1002/ijc.29586
McCullough ML, Gapstur SM, Shah R, Campbell PT, Wang Y, Doyle C, Gaudet MM (2016) Pre- and postdiagnostic diet in relation to mortality among breast cancer survivors in the CPS-II Nutrition Cohort. Cancer Causes Control 27(11):1303–1314. https://doi.org/10.1007/s10552-016-0802-x
Acknowledgment
The authors appreciate the help of authors of included articles for providing additional information needed for the analysis. The authors’ responsibilities were as follows—RH: data selection, data extraction, quality assessment of literature, wrote the paper, and contributed to the writing, editing, and proofreading of the final version of the article; JW: conducted the literature search, data selection, data extraction, quality assessment of literature, conducted the statistical analysis, and contributed to the writing, editing, and proofreading of the final version of the article; YH: data selection, data extraction, quality assessment of literature, conducted the statistical analysis, and contributed to the writing, editing, and proofreading of the final version of the article; XZ, SX, EKC, VSV: contributed to the writing, editing, and proofreading of the final version of the article.
Funding
Xiaotao Zhang was funded (in part) by a Research Training Award for Cancer Prevention Post-Graduate Training Program in Integrative Epidemiology from the Cancer Prevention & Research Institute of Texas, Grant Number RP160097 (PI: M. Spitz).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hou, R., Wei, J., Hu, Y. et al. Healthy dietary patterns and risk and survival of breast cancer: a meta-analysis of cohort studies. Cancer Causes Control 30, 835–846 (2019). https://doi.org/10.1007/s10552-019-01193-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10552-019-01193-z