Abstract
Objectives
The NR5A2 and RYR2 genes are important players in steroid metabolism and play an important role in cancer research. In this research, we want to evaluate the effect of NR5A2 and RYR2 polymorphisms on breast cancer (BC).
Methods
Four single nucleotide polymorphisms on NR5A2 and RYR2 were selected to genotype by Agena MassARRAY in 379 BC patients and 407 healthy controls. Using the PLINK software to calculate the Odds ratio (OR) and 95% confidence intervals (CIs) via the logistic regression analysis to evaluate the risk for BC.
Results
We found that NR5A2 rs2246209 significantly decreased the risk of BC with the AA genotype (OR 0.58, 95%CI 0.34–0.99, p = 0.049), and recessive model (OR 0.59, 95%CI 0.35–0.99, p = 0.046); rs12594 in the RYR2 gene significantly decreased the risk of BC in the GG genotype (OR 0.44, 95%CI 0.22–0.88, p = 0.020), and recessive model (OR 0.43, 95%CI 0.21–0.85, p = 0.016). Further stratification analysis showed that NR5A2 rs2246209 was related to a lower incidence of BC affected by age, lymph nodes metastasis, and tumor stage; RYR2 rs12594 was related to a decreased BC risk restricted by age, estrogen receptor (ER), progesterone receptor (PR), menopausal status, tumor size, and tumor stage. Rs12594 in the RyR2 gene remained significant on the genetic susceptibility of PR-positive BC after Bonferroni correction (p < 0.0125).
Conclusions
This study provides an evidence that NR5A2 rs2246209 and RYR2 rs12594 decreased the risk of breast cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Breast cancer (BC), a malignant tumor that occurs in breast epithelial tissue, is a major public health problem worldwide and one of the most common malignant tumors diagnosed in women [1], seriously affecting women's physical and mental health and even life threatening. In 2017, the global incidence of breast cancer increased to 1,960,681 cases [2]. According to the International Agency for Research on Cancer (IARC, https://www.iarc.fr/), there are 2.1 million new cases worldwide only in 2018, and the death toll exceeds 626 679, and 99% of which occur in women and only 1% in men [3, 4]. We all know that history of breast disease, family history of cancer, frequent abortions, taking contraceptives, and passive smoking are risk factors for breast cancer. Studies have found high inter-individual differences in BC susceptibility, clinical outcomes, and treatment response, highlighting the importance of genetic alterations in BC [5]. Previous studies have shown that some genes are related to breast cancer, such as Liver receptor homolog-1 (LRH-1) [6] and Growth Regulation by Estrogen in Breast Cancer 1 (GREB1) [7].
NR5A2 (Nuclear Receptor Subfamily 5 Group A Member 2), alias LRH-1, belongs to the nuclear receptor NR5A subfamily and is expressed in the development of endothelial origin and adult tissues [8], and acts as a transcription factor in embryogenesis, steroid and cholesterol metabolism, inflammation, and various cancers [9, 10]. A study by Ueno et al. in Japanese patients with pancreatic cancer found that SNPs in the NR5A2 gene were associated with a reduced risk of pancreatic cancer [11]. Zhang et al. [12] found that the polymorphism of NR5A2 gene was significantly associated with regional lymph node metastasis or distant metastasis in patients with gastric cancer. Xiao et al. [13] found that the expression of NR5A2 gene was increased in clinical samples of hepatocellular carcinoma by immunohistochemistry, and that NR5A2 gene was predicted to be a therapeutic target for hepatocellular carcinoma. Li-Yun Chang et al. showed that in the ER (−) and ER (−))/ER (+) mixed group, NR5A2 expression plays an important role in the prognosis of breast cancer [14]. Jiang Zhu et al. verified that miR-27b-3p enhances the role of tamoxifen in breast cancer induction by inhibiting the expression of NR5A2 and cAMP response element binding protein 1 (CREB1). The impact of NR5A2 polymorphism on breast cancer risk in Chinese Han population has not been reported. Therefore, we determined that the NR5A2 gene is worth exploring in BC.
The ryanodine receptor is a Ca-releasing channel protein in Ca cells that are sensitive to caffeine and ryandin. Previous studies have shown that the SR is the primary intracellular Ca2+ store and ryanodine receptors (RyR) are the Ca2+ release channels [15]. Y Ogawa found that three subtypes of RyR have been identified in mammals: RyR1, RyR2, and RyR3 [16]. RYR2 (Ryanodine Receptor 2) is also an important subject in cancer research, and mutations of RYR2 were associated with several cancers. Femi et al. [17] found that mutations in the RYR2 gene affect the prognosis of cervical cancer, and speculate that RYR2 can be used as a target for the treatment of cervical cancer. Cai et al. [18] found that RYR2 gene mutation may affect lung adenocarcinoma immunodiagnosis and immunotherapy. Mansoor Abdul et al. indicate that RYR can be used as a prognostic indicator and / or target for breast cancer [19]. Lina Zhang et al. found that receptor gene 3 (RYR3) is associated with breast cancer risk and calcification [20].
The above findings support the associations between NR5A2/RYR2 genetic polymorphisms and the risk of cancers. However, there were few obvious results in the relationship of NR5A2/RYR2 gene with BC. Therefore, the aim of this study was to validate the genetic association of NR5A2/RYR2 polymorphisms with the risk of BC in Chinese Han population.
Materials and methods
Study participants
Using a case–control design, 379 patients (mean age: 51.50 ± 10.14 years) with BC and 407 controls (mean age: 53.04 ± 9.29 years) were enrolled. All patients were recruited from Shaanxi Provincial Cancer Hospital. Patient inclusion criteria: BC was confirmed by physical examination, imaging (X-ray, color Doppler ultrasound, MRI), and histopathology and cytopathology. Patients with complex blood diseases, autoimmune diseases, trauma, or other tumors were excluded from this study. After that, we investigated and collected clinical indicators of the BC patients, including age, tumor size, tumor stage, and the statuses of estrogen receptor (ER), progesterone receptor (PR), Cerb-B2, lymph node metastasis (LNM), and menopausal.
The controls were healthy volunteers from Shaanxi Provincial Cancer Hospital (Xi´an, Shaanxi, China) recruited during the same period. Inclusion criteria of control group included no medical or family history of cancer or any neurogenic diseases or any breast abnormality.
Data collection
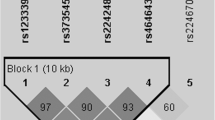
The common methods of selecting SNPs are based on haplotype data or genotype data [21,22,23]. Previous studies have shown that candidate tagging rs12594 and rs16835904 are related to tumor [24]. Four candidate SNPs in the NR5A2 (rs2246209: chr1:200176405, G > A, 3 Prime UTR Variant; rs1056426: chr1:200177275, T > C, 3 Prime UTR Variant) and RYR2 (rs12594: chr1:237833787, A > G, 3 Prime UTR Variant; rs16835904: chr1:237833954, C > T, 3 Prime UTR Variant) genes were selected with a minor allele frequency (MAF) great than 0.05 in global population from the 1000 Genome Projects (https://www.internationalgenome.org/).
5 ml of venous blood was collected, DNA was extracted using the whole blood genomic DNA extraction kit (GOLDMAG, Xi´an, China) [25]; primers of 4 sites (rs2246209, rs1056426, rs12594, and rs16835904) were designed by Agena's online software (https://agenacx.com/online-tools/) and genotyping was performed using MassARRAY time flight mass spectrometry array SNP genotyping platform (Agena Bioscience, San Diego, CA, USA) [26], which as we published in the previous article [27].
Statistical analyses
We used SPSS (version 21.0, IBM Corporation, Armonk, NY, USA) and PLINK software (https://zzz.bwh.harvard.edu/plink/ld.shtml) for statistical analysis. Firstly, the control population was selected to calculate the Hardy Weinberg equilibrium using the Fisher’s exact tests to assess whether we were randomly selected control samples [28]. Secondly, logistic regression analysis was used to evaluate the effects of four polymorphic loci on genetic susceptibility to breast cancer, under five genetic models (allele, genotype, recessive, dominant, and additive model). The Odds ratios (ORs) and 95% confidence intervals (CIs) were used to assess the relative risk of breast cancer [29]. At the same time, according to age, tumor size, clinical stage, lymph node metastasis, and the statuses of ER, PR, Cerb-B2, and menopausal for stratified analysis. Bonferroni correction is one of the most important methods to solve the error discovery rate caused by multiple testing. All p values were Bonferroni corrected, and statistical significance was set at p < 0.0125 (0.05/4).
Results
Characteristics of cases and controls
The basic clinical information of BC patients is shown in Table 1. 379 patients presented with different distribution, according to age (age < 54, 212 cases; age ≥ 54, 167 cases), menopausal status (premenopausal, 135 cases; postmenopausal, 239 cases), tumor size (size ≤ 2 cm, 164 patients; size > 2 cm, 188 patients), tumor stage (I–II stage, 246 patients; III–IV stage, 116 patients), and status of ER (positive, 274 cases; negative, 105 cases), PR (positive, 240 cases; negative, 139 cases), LNM (positive, 186 cases; negative, 175 cases),and Cerb-B2 (positive, 189 patients; negative, 67 patients).
Basic information and allele frequencies of the NR5A2 and RYR2 gene polymorphisms are presented in Table 2. The genotype distribution of all SNPs in control subjects met the HWE (p > 0.05). The correlation between NR5A2 and RYR2 polymorphisms and BC risk under the allele model is shown in Table 2, unfortunately, there were no differences between SNPs in the NR5A2, and RYR2 genes and BC risk (all p > 0.05).
Association between the NR5A2, and RYR2, genes and the risk of BC
Table 3 shows that rs2246209 in the NR5A2 gene significantly decreased the risk of BC with the AA genotype (adjusted OR 0.58, 95%CI 0.34–0.99, p = 0.049), and in the recessive model (adjusted OR 0.59, 95%CI 0.35–0.99, p = 0.046);rs12594 in the RYR2 gene significantly decreased the risk of BC with the GG genotype (adjusted OR 0.44, 95%CI 0.22–0.88, p = 0.020), and in the recessive model (adjusted OR 0.43, 95%CI 0.21–0.85, p = 0.016), no significant difference was found for the other SNPs between cases and controls (all p > 0.05, Table 3).
Stratification analysis of clinical features
We found that rs2246209 in the NR5A2 gene was related to a lower incidence of BC in people aged ≥ 54 in the homozygote model and recessive model (homozygote model: OR 0.39, 95%CI 0.17–0.89, p = 0.025; recessive model: OR 0.39, 95%CI 0.18–0.87, p = 0.021); NR5A2 rs2246209 was associated with a significantly decreased risk of LNM negative BC in the dominant model and log-additive model (dominant model: OR 0.65, 95%CI 0.46–0.94, p = 0.021; log-additive model: OR 0.70, 95%CI 0.53–0.93, p = 0.015); rs2246209 was correlated with a significant reduction in stage I–II BC risk in homozygote model, recessive model, and log-additive model (homozygote model: OR 0.47, 95%CI 0.25–0.90, p = 0.023; recessive model: OR 0.52, 95%CI 0.28–0.98, p = 0.042; log-additive model OR 0.74, 95%CI 0.57–0.95, p = 0.018) (Table 4).
Also, we found that RYR2 rs12594 was related to BC (Table 5). In homozygote (OR 0.29, 95%CI 0.09–0.91, p = 0.034) and recessive model (OR 0.29, 95%CI 0.10–0.90, p = 0.032), rs12594 was associated with a reduced risk of BC in women aged ≥ 54; Rs12594 was a protective site for ER-positive (Homozygote: OR 0.35, 95%CI 0.15–0.83, p = 0.017; Recessive: OR 0.35, 95%CI 0.15–0.83, p = 0.014) and PR-positive (Homozygote: OR 0.23, 95%CI 0.08–0.66, p = 0.007; Recessive: OR 0.23, 95%CI 0.08–0.66, p = 0.006) BC. Rs12594 in the RyR2 gene remained significant on the genetic susceptibility of PR-positive BC after Bonferroni correction (p < 0.0125). And rs12594 was associated with a significant reduction risk of BC in premenopausal women under the homozygote model (OR 0.37, 95%CI 0.16–0.89, p = 0.025) and the recessive model (OR 0.38, 95%CI 0.16–0.88, p = 0.025); we also found that rs12594 mutations significantly reduced the incidence of BC with tumor size ≤ 2 cm (Homozygote: OR 0.34, 95%CI 0.11–0.98, p = 0.045; Recessive: OR 0.33, 95%CI 0.11–0.95, p = 0.040) and tumor stage I–II (Homozygote: OR 0.43, 95%CI 0.19–0.97, p = 0.043; Recessive: OR 0.44, 95%CI 0.20–0.98, p = 0.045).
Discussion
Through this case–control study, we identified that rs2246209 in NR5A2 gene, and rs12594 in RYR2 gene associated with decreased risk of BC.
A number of studies has shown that the occurrence of BC is related to estrogen [30], and the NR5A2 gene plays an important role in steroid metabolism. Garattini et al. [31] showed that NR5A2 may be a nuclear receptor with carcinogenic properties. Nadolny and Dong [32] believed that NR5A2 is a potential BC treatment target. Jiang zhu et al. have shown that by increasing the expression of NR5A2 and CREB1, the low expression of microRNA-2b-3p can enhance the resistance of tamoxifen in breast cancer [33]. Studies have shown that nuclear receptor NR5A2 is involved in the main prognosis of invasive ductal breast cancer [14]. These evidences indicated that there is a relationship between NR5A2 and BC. However, it is still unclear whether the polymorphism of NR5A2 is related to the risk of BC. In our study, we found that mutations of the rs2246209 locus in the NR5A2 gene reduced the risk of BC and clarified the association between NR5A2 gene and BC susceptibility in Chinese Han female. Pang et al. [34] determined that the expression pattern of the NR5A2 gene in BC affects the invasiveness of BC cells. Bianco et al. [35] believed that the NR5A2 gene is involved in the regulation of transcriptional processes in BC cells. We know that non-coding RNA can bind specifically to the 3′UTR region of the target gene. The single nucleotide polymorphism in the target gene 3′UTR region can affect the binding of the target gene to the gene, leading to changes in the expression of the target gene, thus affecting the occurrence of disease. Studies found that the NR5A2 gene was highly expressed in breast cancer tissues, and we obtained the same results by bioinformatics analysis (GEPIA: https://gepia.cancer-pku.cn/detail.php?gene=#boxplot), while the rs2246209 locus is located in the NR5A2 3′UTR region, therefore, we hypothesized that rs2246209 may affect the expression level of NR5A2 in breast cancer and inhibit the occurrence of breast cancer.
Some studies have shown that RyR and RYR3 genes are related to the prognosis, risk and calcification of breast cancer, respectively [19, 20]. Kobylewski et al. [36] found that the invasiveness of female BC was positively correlated with the expression of RYR2 in BC tissues. In the process of epithelial–mesenchymal transition in BC, the most significant change in gene expression is RYR2 [37], indicating that the expression of RYR2 gene is involved in the metastasis of BC. The rs12594 locus is located in the RYR2 3′UTR region, and previous studies have shown that candidate tagging rs12594 and rs16835904 are related to tumor. However, the mechanism of RyR2 polymorphism in breast cancer is still unclear. In our study, we also found that rs12594 on RYR2 gene was significantly associated with BC risk, and it was the first time clearly indicating the relationship between RYR2 gene and BC risk. Therefore, we hypothesized that 12,594 may affect the expression level of RYR2 in breast cancer and inhibit the occurrence of breast cancer.
However, it is not clear whether the mutation of NR5A2 and RYR2 genes really affects expression. After that, it is necessary to analyze the expression of NR5A2 and RYR2 genes in cancer and normal tissues by real-time fluorescence quantitative PCR, and further analyze the influence of different genes on the expression of breast cancer, and further clarify the role of two these genes in the development of breast cancer. Furthermore, our research suggested that age, tumor status, tumor stage, and the statuses of ER, PR, LNM, Cerb-B2, and menopausal play a key role in the susceptibility of BC. After Bonferroni's correction, RyR2 gene rs12594 still had a significant effect on the genetic susceptibility of PR-positive BC (p < 0.0125), while the other loci in our study were not found to be related to the risk of BC. This may be due to our strict SNP screening criteria and small samples. In addition, the Bonferroni correction adjusts the value of alpha according to the number of experiments carried out, so it is conservative; in some cases, due to type II errors, the truly significant differences may be considered insignificant [38]. Huang et al. [39] demonstrated that the risk of BC is affected by clinical features such as ER, PR, LNM, and tumor stage. It is speculated that the cause may be different hormone levels in patients with different clinical phenotypes, and estrogen is essential for BC development [40]. Wang et al. [41] believed that estrogen is a dangerous biomarker for BC development. Suba [42] revealed that estrogen tolerance in BC patients significantly affects treatment outcomes.
This study provides an evidence for NR5A2 rs2246209 and RYR2 rs12594 decreased the risk of breast cancer. However, further studies are warranted on larger patients from other ethnic groups to confirm our results. And, through in vivo and in vitro experiments, explore the molecular mechanism of NR5A2 rs2246209 and RYR2 rs12594 to reduce the risk of breast cancer.
Abbreviations
- BC:
-
Breast cancer
- ER:
-
Estrogen receptor
- PR:
-
Progesterone receptor
- LNM:
-
Lymph node metastasis
- ORs:
-
Odds ratios
- CIs:
-
Confidence intervals
- SNP:
-
Single nucleotide polymorphism
- HWE:
-
Hardy–Weinberg equilibrium
References
Pontikaki A, Sifakis S, Spandidos DA (2016) Endometriosis and breast cancer: a survey of the epidemiological studies. Oncol Lett 11(1):23–30
Li N, Deng Y, Zhou L, Tian T, Yang S, Wu Y, Zheng Y, Zhai Z, Hao Q, Song D et al (2019) Global burden of breast cancer and attributable risk factors in 195 countries and territories, from 1990 to 2017: results from the Global Burden of Disease Study. J Hematol Oncol 12(1):140
Deng Z, Yang H, Liu Q, Wang Z, Feng T, Ouyang Y, Jin T, Ren H (2016) Identification of novel susceptibility markers for the risk of overall breast cancer as well as subtypes defined by hormone receptor status in the Chinese population. J Hum Genet 61(12):1027–1034
Singh K, He X, Kalife ET, Ehdaivand S, Wang Y, Sung CJ (2018) Relationship of histologic grade and histologic subtype with oncotype Dx recurrence score; retrospective review of 863 breast cancer oncotype Dx results. Breast Cancer Res Treat 168(1):29–34
Dai ZJ, Liu XH, Ma YF, Kang HF, Jin TB, Dai ZM, Guan HT, Wang M, Liu K, Dai C et al (2016) Association between single nucleotide polymorphisms in DNA polymerase kappa gene and breast cancer risk in Chinese Han population: a STROBE-Compliant Observational Study. Medicine (Baltimore) 95(2):e2466
Wang S, Zou Z, Luo X, Mi Y, Chang H, Xing D (2018) LRH1 enhances cell resistance to chemotherapy by transcriptionally activating MDC1 expression and attenuating DNA damage in human breast cancer. Oncogene 37(24):3243–3259
Chand AL, Wijayakumara DD, Knower KC, Herridge KA, Howard TL, Lazarus KA, Clyne CD (2012) The orphan nuclear receptor LRH-1 and ERα activate GREB1 expression to induce breast cancer cell proliferation. PLoS ONE 7(2):e31593
Fernandez-Marcos PJ, Auwerx J, Schoonjans K (2011) Emerging actions of the nuclear receptor LRH-1 in the gut. Biochim Biophys Acta 1812(8):947–955
Huang SC, Lee CT, Chung BC (2014) Tumor necrosis factor suppresses NR5A2 activity and intestinal glucocorticoid synthesis to sustain chronic colitis. Sci Signal 7(314):ra20
Sahini N, Borlak J (2016) Genomics of human fatty liver disease reveal mechanistically linked lipid droplet-associated gene regulations in bland steatosis and nonalcoholic steatohepatitis. Transl Res 177:41–69
Ueno M, Ohkawa S, Morimoto M, Ishii H, Matsuyama M, Kuruma S, Egawa N, Nakao H, Mori M, Matsuo K et al (2015) Genome-wide association study-identified SNPs (rs3790844, rs3790843) in the NR5A2 gene and risk of pancreatic cancer in Japanese. Sci Rep 5:17018
Zhang X, Gu D, Du M, Wang M, Cao C, Shen L, Kuang M, Tan Y, Huo X, Gong W et al (2014) Associations of NR5A2 gene polymorphisms with the clinicopathological characteristics and survival of gastric cancer. Int J Mol Sci 15(12):22902–22917
Xiao L, Wang Y, Liang W, Liu L, Pan N, Deng H, Li L, Zou C, Chan FL, Zhou Y (2018) LRH-1 drives hepatocellular carcinoma partially through induction of c-myc and cyclin E1, and suppression of p21. Cancer Manag Res 10:2389–2400
Chang LY, Liu LY, Roth DA, Kuo WH, Hwa HL, Chang KJ, Hsieh FJ (2015) The major prognostic features of nuclear receptor NR5A2 in infiltrating ductal breast carcinomas. Int J Genomics 2015:403576
Laver DR (2018) Regulation of the RyR channel gating by Ca(2+) and Mg(2). Biophys Rev 10(4):1087–1095
Ogawa Y (1994) Role of ryanodine receptors. Crit Rev Biochem Mol Biol 29(4):229–274
Femi OF (2018) Genetic alterations and PIK3CA gene mutations and amplifications analysis in cervical cancer by racial groups in the United States. Int J Health Sci (Qassim) 12(1):28–32
Cai W, Zhou D, Wu W, Tan WL, Wang J, Zhou C, Lou Y (2018) MHC class II restricted neoantigen peptides predicted by clonal mutation analysis in lung adenocarcinoma patients: implications on prognostic immunological biomarker and vaccine design. BMC Genomics 19(1):582
Abdul M, Ramlal S, Hoosein N (2008) Ryanodine receptor expression correlates with tumor grade in breast cancer. Pathol Oncol Res : POR 14(2):157–160
Zhang L, Liu Y, Song F, Zheng H, Hu L, Lu H, Liu P, Hao X, Zhang W, Chen K (2011) Functional SNP in the microRNA-367 binding site in the 3'UTR of the calcium channel ryanodine receptor gene 3 (RYR3) affects breast cancer risk and calcification. Proc Natl Acad Sci USA 108(33):13653–13658
Daly MJ, Rioux JD, Schaffner SF, Hudson TJ, Lander ES (2001) High-resolution haplotype structure in the human genome. Nat Genet 29(2):229–232
Gabriel SB, Schaffner SF, Nguyen H, Moore JM, Roy J, Blumenstiel B, Higgins J, DeFelice M, Lochner A, Faggart M et al (2002) The structure of haplotype blocks in the human genome. Science (New York, NY) 296(5576):2225–2229
Patil N, Berno AJ, Hinds DA, Barrett WA, Doshi JM, Hacker CR, Kautzer CR, Lee DH, Marjoribanks C, McDonough DP et al (2001) Blocks of limited haplotype diversity revealed by high-resolution scanning of human chromosome 21. Science (New York, NY) 294(5547):1719–1723
Chen Q, Sun Y, Wu J, Xiong Z, Niu F, Jin T, Zhao Q (2019) LPP and RYR2 Gene polymorphisms correlate with the risk and the prognosis of astrocytoma. J Mol Neurosci : MN 69(4):628–635
Zhou L, Dong S, Deng Y, Yang P, Zheng Y, Yao L, Zhang M, Yang S, Wu Y, Zhai Z et al (2019) GOLGA7 rs11337, a polymorphism at the microRNA binding site, is associated with glioma prognosis. Mol Ther Nucleic Acids 18:56–65
Deng Y, Zhou L, Li N, Wang M, Yao L, Dong S, Zhang M, Yang P, Hao Q, Wu Y et al (2019) Impact of four lncRNA polymorphisms (rs2151280, rs7763881, rs1136410, and rs3787016) on glioma risk and prognosis: a case-control study. Mol Carcinog 58(12):2218–2229
Wang J, Shi Y, Wang G, Dong S, Yang D, Zuo X (2019) The association between interleukin-1 polymorphisms and their protein expression in Chinese Han patients with breast cancer. Mol Genet Genomic Med 7:e804
Adamec C (1964) Example of the use of the nonparametric test. Test X2 for comparison of 2 independent examples. Ceskoslovenske zdravotnictvi 12:613–619
Bland JM, Altman DG (2000) Statistics notes. The odds ratio. BMJ (Clin Res ed) 320(7247):1468
Christopoulos PF, Corthay A, Koutsilieris M (2018) Aiming for the Insulin-like Growth Factor-1 system in breast cancer therapeutics. Cancer Treat Rev 63:79–95
Garattini E, Bolis M, Gianni M, Paroni G, Fratelli M, Terao M (2016) Lipid-sensors, enigmatic-orphan and orphan nuclear receptors as therapeutic targets in breast-cancer. Oncotarget 7(27):42661–42682
Nadolny C, Dong X (2015) Liver receptor homolog-1 (LRH-1): a potential therapeutic target for cancer. Cancer Biol Ther 16(7):997–1004
Zhu J, Zou Z, Nie P, Kou X, Wu B, Wang S, Song Z, He J (2016) Downregulation of microRNA-27b-3p enhances tamoxifen resistance in breast cancer by increasing NR5A2 and CREB1 expression. Cell Death Dis 7(11):e2454
Pang JB, Molania R, Chand A, Knower K, Takano EA, Byrne DJ, Mikeska T, Millar EKA, Lee CS, O'Toole SA et al (2017) LRH-1 expression patterns in breast cancer tissues are associated with tumour aggressiveness. Oncotarget 8(48):83626–83636
Bianco S, Brunelle M, Jangal M, Magnani L, Gevry N (2014) LRH-1 governs vital transcriptional programs in endocrine-sensitive and -resistant breast cancer cells. Cancer Res 74(7):2015–2025
Kobylewski SE, Henderson KA, Eckhert CD (2012) Identification of ryanodine receptor isoforms in prostate DU-145, LNCaP, and PWR-1E cells. Biochem Biophys Res Commun 425(2):431–435
Davis FM, Parsonage MT, Cabot PJ, Parat MO, Thompson EW, Roberts-Thomson SJ, Monteith GR (2013) Assessment of gene expression of intracellular calcium channels, pumps and exchangers with epidermal growth factor-induced epithelial-mesenchymal transition in a breast cancer cell line. Cancer Cell Int 13(1):76
Perneger TV (1998) What's wrong with Bonferroni adjustments. BMJ 316(7139):1236–1238
Huang W, Nie W, Zhang W, Wang Y, Zhu A, Guan X (2016) The expression status of TRX, AR, and cyclin D1 correlates with clinicopathological characteristics and ER status in breast cancer. Onco Targets Ther 9:4377–4385
Hilton HN, Clarke CL, Graham JD (2018) Estrogen and progesterone signalling in the normal breast and its implications for cancer development. Mol Cell Endocrinol 466:2–14
Wang B, Yuan F (2018) Comment on "Estrogen receptor alpha (ERS1) SNPs c454–397T>C (PvuII) and c454–351A>G (XbaI) are risk biomarkers for breast cancer development". Mol Biol Rep 46:5
Suba Z (2014) Diverse pathomechanisms leading to the breakdown of cellular estrogen surveillance and breast cancer development: new therapeutic strategies. Drug Des Devel Ther 8:1381–1390
Acknowledgements
We thank all authors for their contributions and supports. We are also grateful to all participants for providing blood samples. We also thank the National Natural Science Foundation of China (No. 8170100875) for funding the research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Our present study was approved by the Ethics Committee of Shaanxi Provincial Cancer Hospital. Informed consent forms were signed by all participants.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wei, Y., Wang, X., Zhang, Z. et al. Impact of NR5A2 and RYR2 3′UTR polymorphisms on the risk of breast cancer in a Chinese Han population. Breast Cancer Res Treat 183, 1–8 (2020). https://doi.org/10.1007/s10549-020-05736-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-020-05736-w