Abstract
Purpose
Several clinical trials have investigated the prognostic and predictive usefulness of molecular markers. With limited predictive value, molecular markers have mainly been used to identify prognostic subgroups in which the indication for chemotherapy is doubtful and the prognosis is favorable enough for chemotherapy to be avoided. However, limited information is available about which groups of patients may benefit from additional therapy. This study aimed to describe the prognostic effects of Ki-67 in several common subgroups of patients with early breast cancer.
Methods
This retrospective study analyzed a single-center cohort of 3140 patients with HER2−, hormone receptor-positive breast cancer. Five-year disease-free survival (DFS) rates were calculated for low (< 10%), intermediate (10–19%), and high (≥ 20%) Ki-67 expression levels, as assessed by immunohistochemistry, and for subgroups relative to age, body mass index, disease stage, tumor grade, and (neo-)adjuvant chemotherapy. It was also investigated whether Ki-67 had different effects on DFS in these subgroups.
Results
The 5-year DFS rates for patients with low, intermediate, and high levels of Ki-67 expression were 0.90, 0.89, and 0.77, respectively. Ki-67 was able to further differentiate patients with an intermediate prognosis into different prognostic groups relative to common clinical parameters. Patients with stage II breast cancer had 5-year DFS rates of 0.84, 0.88, and 0.79 for low, intermediate, and high levels of Ki-67 expression. Ki-67 had different prognostic effects in subgroups defined by age and tumor grade.
Conclusions
Ki-67 may help identify patients in intermediate prognostic groups with an unfavorable prognosis who may benefit from further therapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cell proliferation is a central process that is connected to pathogenesis and progression across cancer histologies [1, 12, 28, 30, 45]. Particularly in patients with HER2− and hormone receptor-positive breast cancer, the proliferation index is able to identify women with early and advanced stages of the disease who have an unfavorable prognosis [7, 12, 17, 29, 35, 36, 46]. Proliferation has also been included in most multigene assays for patients with early breast cancer, as one of the major driving factors that determine the prognostic value of these tests.
Patients with breast cancer who have a high proliferation index and an unfavorable prognosis usually receive chemotherapy. However, especially in the neoadjuvant setting, it has been found that the prognosis-improving effect of a pathological complete response (pCR) after chemotherapy is stronger in some subgroups than in others [11, 17, 18, 43]. pCR is an indicator of a very good prognosis both in patients with HER2+ breast cancer and in those with triple-negative breast cancer, whereas in patients with HER2−, hormone receptor-positive cancer, the effect is much smaller. However, it has been shown that Ki-67 is a very good predictor of the prognosis in the latter subgroup [17]. It therefore appears to be a promising marker to use in decision-making for or against chemotherapy in this group of patients [23], and has been included in national and international guidelines and current therapeutic studies [40, 47, 48].
CDK4/6 inhibitors (CDK4/6is) in combination with antiendocrine treatments have recently been introduced for the treatment of advanced breast cancer [19,20,21, 24, 33, 34, 41]. They interfere with the cell cycle and have a direct effect on proliferation. It has been shown that they lead to consistent improvement in prolonging the progression-free survival (PFS), with hazard ratios (HRs) of around 0.5 to 0.6 [19, 21, 24, 33, 34, 41]. CDK4/6is may possibly even lead to an overall survival benefit [42]. The safety profile also allows for long treatment periods [14]. The treatment might, therefore, be suitable for improving the prognosis in the adjuvant setting as well, and studies in this therapy setting are underway [8,9,10]. Some of these trials use Ki-67 as an inclusion criterion, and it would, therefore, be beneficial to understand the prognostic effects of Ki-67 in the various commonly used subgroups of patients with early breast cancer.
Patients and methods
Patient selection
The patients included in this retrospective study were selected from all patients with invasive breast cancer treated at the University Breast Center for Franconia, Germany, since 2001. For inclusion, the patients had to have a HER2− and hormone receptor-positive tumor, along with information available regarding staging and other covariates. Figure 1 shows a flow diagram for patient selection. Approval for carrying out the planned analysis was obtained from the Ethics Committee of the Medical Faculty of Friedrich Alexander University, Erlangen.
Data collection
As part of a continuous quality-assurance process, each certified breast cancer center in Germany is required to document all primary diagnoses, patient and tumor characteristics, tumor board decisions, and treatment characteristics, as well as follow-up data for disease-free survival (DFS) and overall survival [3, 47]. These data have to be collected prospectively and are audited on an annual basis by the German Cancer Society (Deutsche Krebsgesellschaft) and German Society for Breast Diseases (Deutsche Gesellschaft für Senologie). As part of this process, all histopathological data have to be documented, including tumor size; axillary lymph-node status; tumor grade; and estrogen receptor, progesterone receptor, and HER2 status, among other items. The data have to be documented from the original pathology reports. All of the data used in this retrospective analysis were obtained from this database.
Histopathological data
All histopathological information used in this analysis was directly documented from the original pathological reports, which were reviewed by two investigators. Grading, tumor type, estrogen receptor status, progesterone receptor status, HER2 status, and proliferation status as assessed by Ki-67 staining have been routinely recorded at the Breast Center since 1995. The pretreatment core biopsies were stained in clinical routine as follows: estrogen receptor-alpha (clone 1D5, 1:200 dilution or clone EP1, dilution 1:40; DAKO, Denmark); progesterone receptor (clone pgR636, 1:200 dilution; DAKO, Denmark), Ki-67 (clone MIB-1, 1:200 or 1:100 dilution; DAKO, Denmark). The percentage of positively stained cells was stated in the pathological reports.
The tumors were considered to be positive for the estrogen and progesterone receptors if a percentage of at least 10% (up to 2009) or at least 1% (since 2010) more cells stained positive.
To assess HER2 status, a polyclonal antibody against HER2 (1:200 or a 1:1000 dilution, DAKO, Denmark) was used and HER2 status was stated as negative, 0, 1+, 2+, or 3+ in accordance with Sauter et al. [32]. Tumors with a score of 0 or 1+ were considered HER2−, and 3+ tumors were considered positive. Tumors with 2+ staining were tested for gene copy numbers using chromogene in situ hybridization. Using a kit with two probes of different colors (ZytoDot, 2C SPEC HER2(ERBB2)/CEN17; Zyto Vision Ltd., Bremerhaven, Germany), the gene copy numbers of HER2 and centromeres of the corresponding chromosome 17 were retrieved. A HER2/CEN17 ratio of ≥ 2.2 up to 2013 and ≥ 2 thereafter was considered to represent amplification of HER2.
With regard to Ki-67 assessment, the overall tumor area was reviewed including the invasion front. In case there were regions with the higher percentage of Ki-67+ nuclei (“hot spot”) this area determined the Ki-67 assessment.
Statistical analysis
The primary objective of this study was to analyze the effect of Ki-67 levels on DFS in commonly analyzed subgroups of patients. DFS was defined as the time from diagnosis to the earliest date of disease progression (distant metastasis, local recurrence, death from any cause) or the date of censoring, after 10 years at the latest. Survival rates, and specifically 5-year survival rates with 95% confidence intervals (CIs), were estimated using the Kaplan–Meier product limit method.
P values for interactions were calculated to assess whether the effect of Ki-67 on DFS varied between patient subgroups, using Cox regression models (one for each patient or tumor characteristic), with patient/tumor characteristic, Ki-67, and the interaction between the patient/tumor characteristic and Ki-67 as predictors. All patient and tumor characteristics were used as categorical or ordinal variables: age at diagnosis (< 45 vs. 45–54 vs. 55–64 ≥ 65 years), body mass index (BMI; < 25 vs. 25–25.99 vs. ≥ 30 kg/m2), pT (1 vs. 2 vs. 3 vs. 4), pN (negative vs. positive), American Joint Committee on Cancer, AJCC stage (I vs. II vs. III), tumor grade (G1 vs. G2 vs. G3), adjuvant or neoadjuvant chemotherapy administered (no vs. yes). Ki-67 was categorized into three groups, < 10%, 10–19%, and ≥ 20%, to obtain information about subgroups similar to those used in other reports [49].
In addition, the prognostic effect of all predictors together was described using a multivariate Cox model with all patient and tumor characteristics of interest as predictors. Information about tumor size and nodal status was used, such as the AJCC stage.
All of the tests were two-sided, and a P value of < 0.05 was regarded as statistically significant. Calculations were carried out using the R system for statistical computing (version 3.0.1; R Development Core Team, Vienna, Austria, 2013).
Results
A total of 3140 patients with HER2−, hormone receptor-positive breast cancer were available for the analysis. Of these, 1095 had Ki-67 levels < 10% (low), 1117 were assessed as having a Ki-67 level of 10–19% (intermediate), and 1195 cases had a Ki-67 level ≥ 20% (high).
Associations between these groups of Ki-67 and patient and tumor characteristics are shown in Table 1. Most of the tumors with Ki-67 < 10% were in stage I (n = 693, 63.3%). Tumors with Ki-67 levels of 10–19% and Ki-67 levels ≥ 20% represented stage I disease in 51.5% and 44.8% of the patients, respectively. Higher proliferation rates as assessed by Ki-67 were strongly correlated with larger tumors with a higher grade. In addition, patients with higher Ki-67 levels received adjuvant or neoadjuvant chemotherapy more often (Table 1).
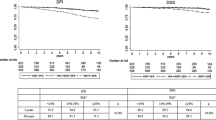
There were 166, 161, and 259 events in the groups of patients with low, intermediate, and high levels of Ki-67 expression, respectively. The 5-year DFS rates for those groups were 0.90 (95% CI 0.88 to 0.92), 0.89 (95% CI 0.87 to 0.91), and 0.77 (95% CI 0.74 to 0.80). Figure 2 shows Kaplan–Meier curves for these Ki-67 categories.
Disease-free survival relative to Ki-67 categories (corresponding hazard ratios are shown in Table 3)
The 5-year DFS rates for Ki-67 relative to tumor stage and other patient and tumor characteristics are shown in Table 2. Analysis of the subgroups defined by Ki-67 and disease stage (as the strongest prognostic factor) differentiated groups of patients who have a 5-year DFS rate of 0.93 (95% CI 0.91 to 0.95) in patients with stage I tumors and Ki-67 < 10%; and patients with a 5-year DFS rate of 0.49 (95% CI 0.41 to 0.58) in patients with stage III tumors and Ki-67 ≥ 20%. In patients with stage II tumors, Ki-67 separated the patients into groups with a 5-year DFS of 0.86 (95% CI 0.82 to 0.90) in patients with Ki-67 < 10%; 0.90 (95% CI 0.86 to 0.93) in patients with a Ki-67 of 10–19%; and 0.77 (95% CI 0.72 to 0.91) in patients with Ki-67 ≥ 20%. Figure 3 shows Kaplan–Meier curves for the subgroups defined by disease stage and Ki-67.
The effect of Ki-67 on DFS varied in patient groups defined by age (interaction P = 0.01) and grade (interaction P < 0.01). In younger patients (< 45 years), there were survival differences between patients with low Ki-67 expression and those with intermediate Ki-67 expression. This difference was not observed in other age classes (Supplementary Fig. 1). The survival curves for patients with G1, G2, and G3 tumors differed in those with intermediate or high Ki-67 values, but there were hardly any differences between patients with G1 tumors and those with G2 tumors if Ki-67 was low (Supplementary Fig. 5). Different prognostic effects of Ki-67 were not observed in any other patient subgroups.
The Cox regression model, including age, BMI, disease stage, and Ki-67 as predictors of DFS showed that each of these variables was associated with the DFS. HRs are shown in Table 3.
Discussion
This analysis presents 5-year DFS rates for patients with HER2− and hormone receptor-positive early breast cancer in relation to Ki-67 expression and common tumor and patient characteristics. It was found that the prognostic effect of Ki-67 differs in patient subgroups defined by age and grade. In patients with stage I disease, the absolute effects were small. In patients with stage II disease, patients with low and intermediate Ki-67 expression had a similar 5-year DFS, while patients with Ki-67 expression > 20% had a more unfavorable 5-year DFS rate.
Discussions regarding Ki-67 have so far focused on whether it can be used to assist decision-making for or against adjuvant chemotherapy. Although adjuvant chemotherapy is considered to improve the 10-year overall survival of breast cancer patients by 6–8% [16], this benefit remains questionable in view of the associated toxicity, particularly since new diagnostic methods are now able to identify patients with a prognosis that is favorable enough for chemotherapy to be avoided. Although a few multigene assays have been developed to assist decision-making [6, 37, 38], simpler models have also been established that include Ki-67 expression in the prognostic prediction [12]. In addition, it has been shown in various trials that Ki-67 also has a prognostic effect in patients who do not receive chemotherapy [49]. Another effect of Ki-67 expression that has been described is its value for predicting pCR following neoadjuvant chemotherapy [17, 31, 39]. However, not only because of its limited analytical validity, the clinical utility of Ki-67 expression in decision-making for or against chemotherapy is still under discussion [2, 5, 49]. It has been argued that it may not be able to identify a group of patients who have an excellent prognosis, in whom a decision in favor of chemotherapy is questionable [2].
On the one hand, avoiding chemotherapy in a group of patients with a favorable prognosis is one clinical question. On the other, additional therapy with an acceptable toxicity profile might be beneficial if it was possible to use a relevant marker to identify a group of patients with an unfavorable prognosis. CDK4/6i might be an example of such a treatment. Although it has only been approved for patients with advanced breast cancer, several clinical trials are ongoing to investigate its efficacy in the adjuvant setting [8,9,10]. Ki-67 expression might be a useful marker for decision-making here, since it is easier to identify women with a poorer prognosis than to identify those with an excellent prognosis. The present study shows that the previously reported cut-off value of 20% identifies a group of patients who have a 5-year DFS of less than 80% in patients with stage II disease. As a matter of fact, this group of patients seems to be a group with yet unmet medical need. Such a patient group might, therefore, benefit from additional therapy like but not restricted to CDK4/6i.
This study has several strengths and limitations. Some of these limitations are associated with Ki-67 staining and assessment and relate to previously identified and discussed concerns regarding the usage of Ki-67 in clinical routine and across institutions [15]. First, with regard to the staining we used MIB-1 as an antibody. MIB-1 is well established for the evaluation of Ki-67 in breast cancer [15]. Therefore, we chose this antibody as a standard for clinical routine assessments. In Germany, the quality of those assessments is validated annually in round robin tests. However, there are further antibodies like SP6. We did not compare Ki-67 assessments with different antibodies which might be a weakness, but other studies showed an extremely high concordance when comparing proliferation assessment with several antibodies [44, 50]. Another issue, which is associated with Ki-67 assessment is the intra-tumor heterogeneity. It is well known that Ki-67 is prone to a relevant intra-tumor heterogeneity [4, 22, 26]. This raises the question how Ki-67 values are assessed to optimize the prediction of prognosis or therapy responsiveness. We used a method, which is also referred to as “hot-spot” method, taking the area into account, which has the highest frequency of positively stained cells. Comparing the hot-spot method versus the average, both methods correlate highly with each other and predict prognosis similarly well [27].
Another concern with regard to Ki-67 and our analysis could be its influence on therapy decisions. While Ki-67 was not used at our site as a standard marker for therapeutic decisions, the physicians attending the tumor board were aware of the Ki-67 value when making those decisions. Therefore, it cannot be excluded that some therapeutic bias was introduced by Ki-67 in our cohort. With regard to the Ki-67 cut-off we pre-defined 10% and 20% as cut-offs for categories, based on clinical studies published with these cut-offs [49]. Furthermore, a cut-off of 20% was considered as being the most clinically relevant cut-off by the St Gallen Expert Panel [25]. However, neither a cut-off of 10% or 20% represent the best separation between intrinsic subtypes [7]. However, utilizing Ki-67 for clinical decision-making, even other cut-offs are discussed like 35% which might be optimal to predict neoadjuvant chemotherapy effectiveness [13, 17]. One strength of the study is clearly the sample size, which made it possible to assess survival rates in subgroups with different levels of Ki-67 expression.
In conclusion, this study shows that Ki-67 expression is able to distinguish between different prognostic groups in most subgroups of patients and tumors. Particularly in patients with AJCC stage II disease, those with Ki-67 expression over 20% may benefit from additional treatments, if available.
References
Allegra CJ, Paik S, Colangelo LH, Parr AL, Kirsch I, Kim G, Klein P, Johnston PG, Wolmark N, Wieand HS (2003) Prognostic value of thymidylate synthase, Ki-67, and p53 in patients with Dukes’ B and C colon cancer: a National Cancer Institute-National Surgical Adjuvant Breast and Bowel Project collaborative study. J Clin Oncol 21:241–250. https://doi.org/10.1200/JCO.2003.05.044
Andre F, Arnedos M, Goubar A, Ghouadni A, Delaloge S (2015) Ki67—no evidence for its use in node-positive breast cancer. Nat Rev Clin Oncol 12:296–301. https://doi.org/10.1038/nrclinonc.2015.46
Beckmann MW, Brucker C, Hanf V, Rauh C, Bani MR, Knob S, Petsch S, Schick S, Fasching PA, Hartmann A, Lux MP, Haberle L (2011) Quality assured health care in certified breast centers and improvement of the prognosis of breast cancer patients. Onkologie 34:362–367. https://doi.org/10.1159/000329601
Boros M, Moncea D, Moldovan C, Podoleanu C, Georgescu R, Stolnicu S (2017) Intratumoral heterogeneity for Ki-67 index in invasive breast carcinoma: a study on 131 consecutive cases. Appl Immunohistochem Mol Morphol 25:338–340. https://doi.org/10.1097/PAI.0000000000000315
Caldarella A, Crocetti E, Paci E (2014) Ki67 in breast cancer: a useful prognostic marker? Ann Oncol 25:542. https://doi.org/10.1093/annonc/mdt561
Cardoso F, van’t Veer LJ, Bogaerts J, Slaets L, Viale G, Delaloge S, Pierga JY, Brain E, Causeret S, DeLorenzi M, Glas AM, Golfinopoulos V, Goulioti T, Knox S, Matos E, Meulemans B, Neijenhuis PA, Nitz U, Passalacqua R, Ravdin P, Rubio IT, Saghatchian M, Smilde TJ, Sotiriou C, Stork L, Straehle C, Thomas G, Thompson AM, van der Hoeven JM, Vuylsteke P, Bernards R, Tryfonidis K, Rutgers E, Piccart M, MINDACT Investigators (2016) 70-Gene signature as an aid to treatment decisions in early-stage breast cancer. N Engl J Med 375:717–729. https://doi.org/10.1056/NEJMoa1602253
Cheang MC, Chia SK, Voduc D, Gao D, Leung S, Snider J, Watson M, Davies S, Bernard PS, Parker JS, Perou CM, Ellis MJ, Nielsen TO (2009) Ki67 index, HER2 status, and prognosis of patients with luminal B breast cancer. J Natl Cancer Inst 101:736–750. https://doi.org/10.1093/jnci/djp082
NCT02513394 (2015) PALbociclib CoLlaborative Adjuvant Study: a randomized Phase III Trial of palbociclib with standard adjuvant endocrine therapy versus standard adjuvant endocrine therapy alone for hormone receptor positive (HR+)/human epidermal growth factor receptor 2 (HER2)-negative early breast cancer (PALLAS). NIH U.S. National Library of Medicine. http://clinicaltrials.gov. Accessed 17 Nov 2018
NCT03155997 (2017) Endocrine therapy with or without abemaciclib (LY2835219) following surgery in participants with breast cancer (monarchE). NIH U.S. National Library of Medicine. http://clinicaltrials.gov. Accessed 17 Nov 2018
NCT03701334 (2018) A trial to evaluate efficacy and safety of ribociclib with endocrine therapy as adjuvant treatment in patients with HR+/HER2− early breast cancer (NATALEE). NIH U.S. National Library of Medicine. http://clinicaltrials.gov. Accessed 17 Nov 2018
Cortazar P, Zhang L, Untch M, Mehta K, Costantino JP, Wolmark N, Bonnefoi H, Cameron D, Gianni L, Valagussa P, Swain SM, Prowell T, Loibl S, Wickerham DL, Bogaerts J, Baselga J, Perou C, Blumenthal G, Blohmer J, Mamounas EP, Bergh J, Semiglazov V, Justice R, Eidtmann H, Paik S, Piccart M, Sridhara R, Fasching PA, Slaets L, Tang S, Gerber B, Geyer CE Jr, Pazdur R, Ditsch N, Rastogi P, Eiermann W, von Minckwitz G (2014) Pathological complete response and long-term clinical benefit in breast cancer: the CTNeoBC pooled analysis. Lancet 384:164–172. https://doi.org/10.1016/S0140-6736(13)62422-8
Cuzick J, Dowsett M, Pineda S, Wale C, Salter J, Quinn E, Zabaglo L, Mallon E, Green AR, Ellis IO, Howell A, Buzdar AU, Forbes JF (2011) Prognostic value of a combined estrogen receptor, progesterone receptor, Ki-67, and human epidermal growth factor receptor 2 immunohistochemical score and comparison with the Genomic Health recurrence score in early breast cancer. J Clin Oncol 29:4273–4278. https://doi.org/10.1200/JCO.2010.31.2835
Denkert C, Loibl S, Muller BM, Eidtmann H, Schmitt WD, Eiermann W, Gerber B, Tesch H, Hilfrich J, Huober J, Fehm T, Barinoff J, Jackisch C, Prinzler J, Rudiger T, Erbstosser E, Blohmer JU, Budczies J, Mehta KM, von Minckwitz G (2013) Ki67 levels as predictive and prognostic parameters in pretherapeutic breast cancer core biopsies: a translational investigation in the neoadjuvant GeparTrio trial. Ann Oncol 24:2786–2793. https://doi.org/10.1093/annonc/mdt350
Dieras V, Rugo HS, Schnell P, Gelmon K, Cristofanilli M, Loi S, Colleoni M, Lu DR, Mori A, Gauthier E, Huang Bartlett C, Slamon DJ, Turner NC, Finn RS (2018) Long-term pooled safety analysis of palbociclib in combination with endocrine therapy for HR+/HER2− advanced breast cancer. J Natl Cancer Inst. https://doi.org/10.1093/jnci/djy109
Dowsett M, Nielsen TO, A’Hern R, Bartlett J, Coombes RC, Cuzick J, Ellis M, Henry NL, Hugh JC, Lively T, McShane L, Paik S, Penault-Llorca F, Prudkin L, Regan M, Salter J, Sotiriou C, Smith IE, Viale G, Zujewski JA, Hayes DF, International Ki-67 in Breast Cancer Working Group (2011) Assessment of Ki67 in breast cancer: recommendations from the International Ki67 in Breast Cancer Working Group. J Natl Cancer Inst 103:1656–1664. https://doi.org/10.1093/jnci/djr393
Early Breast Cancer Trialists’ Collaborative Group, Peto R, Davies C, Godwin J, Gray R, Pan HC, Clarke M, Cutter D, Darby S, McGale P, Taylor C, Wang YC, Bergh J, Di Leo A, Albain K, Swain S, Piccart M, Pritchard K (2012) Comparisons between different polychemotherapy regimens for early breast cancer: meta-analyses of long-term outcome among 100,000 women in 123 randomised trials. Lancet 379:432–444. https://doi.org/10.1016/S0140-6736(11)61625-5
Fasching PA, Heusinger K, Haeberle L, Niklos M, Hein A, Bayer CM, Rauh C, Schulz-Wendtland R, Bani MR, Schrauder M, Kahmann L, Lux MP, Strehl JD, Hartmann A, Dimmler A, Beckmann MW, Wachter DL (2011) Ki67, chemotherapy response, and prognosis in breast cancer patients receiving neoadjuvant treatment. BMC Cancer 11:486. https://doi.org/10.1186/1471-2407-11-486
Fasching PA, Loibl S, Hu C, Hart SN, Shimelis H, Moore R, Schem C, Tesch H, Untch M, Hilfrich J, Rezai M, Gerber B, Costa SD, Blohmer JU, Fehm T, Huober J, Liedtke C, Weinshilboum RM, Wang L, Ingle JN, Muller V, Nekljudova V, Weber KE, Rack B, Rubner M, von Minckwitz G, Couch FJ (2018) BRCA1/2 mutations and bevacizumab in the neoadjuvant treatment of breast cancer: response and prognosis results in patients with triple-negative breast cancer from the GeparQuinto Study. J Clin Oncol 36:2281–2287. https://doi.org/10.1200/JCO.2017.77.2285
Finn RS, Crown JP, Lang I, Boer K, Bondarenko IM, Kulyk SO, Ettl J, Patel R, Pinter T, Schmidt M, Shparyk Y, Thummala AR, Voytko NL, Fowst C, Huang X, Kim ST, Randolph S, Slamon DJ (2015) The cyclin-dependent kinase 4/6 inhibitor palbociclib in combination with letrozole versus letrozole alone as first-line treatment of oestrogen receptor-positive, HER2-negative, advanced breast cancer (PALOMA-1/TRIO-18): a randomised phase 2 study. Lancet Oncol 16:25–35. https://doi.org/10.1016/S1470-2045(14)71159-3
Finn RS, Dering J, Conklin D, Kalous O, Cohen DJ, Desai AJ, Ginther C, Atefi M, Chen I, Fowst C, Los G, Slamon DJ (2009) PD 0332991, a selective cyclin D kinase 4/6 inhibitor, preferentially inhibits proliferation of luminal estrogen receptor-positive human breast cancer cell lines in vitro. Breast Cancer Res 11:R77. https://doi.org/10.1186/bcr2419
Finn RS, Martin M, Rugo HS, Jones S, Im SA, Gelmon K, Harbeck N, Lipatov ON, Walshe JM, Moulder S, Gauthier E, Lu DR, Randolph S, Dieras V, Slamon DJ (2016) Palbociclib and letrozole in advanced breast cancer. N Engl J Med 375:1925–1936. https://doi.org/10.1056/NEJMoa1607303
Focke CM, Decker T, van Diest PJ (2016) Intratumoral heterogeneity of Ki67 expression in early breast cancers exceeds variability between individual tumours. Histopathology 69:849–861. https://doi.org/10.1111/his.13007
Gass P, Lux MP, Rauh C, Hein A, Bani MR, Fiessler C, Hartmann A, Haberle L, Pretscher J, Erber R, Wachter DL, Schulz-Wendtland R, Beckmann MW, Fasching PA, Wunderle M (2018) Prediction of pathological complete response and prognosis in patients with neoadjuvant treatment for triple-negative breast cancer. BMC Cancer 18:1051. https://doi.org/10.1186/s12885-018-4925-1
Goetz MP, Toi M, Campone M, Sohn J, Paluch-Shimon S, Huober J, Park IH, Tredan O, Chen SC, Manso L, Freedman OC, Garnica Jaliffe G, Forrester T, Frenzel M, Barriga S, Smith IC, Bourayou N, Di Leo A (2017) MONARCH 3: abemaciclib as initial therapy for advanced breast cancer. J Clin Oncol 35:3638–3646. https://doi.org/10.1200/JCO.2017.75.6155
Goldhirsch A, Winer EP, Coates AS, Gelber RD, Piccart-Gebhart M, Thurlimann B, Senn HJ, Panel m (2013) Personalizing the treatment of women with early breast cancer: highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann Oncol 24:2206–2223. https://doi.org/10.1093/annonc/mdt303
Himuro T, Horimoto Y, Arakawa A, Tanabe M, Saito M (2016) Ki67 heterogeneity in estrogen receptor-positive breast cancers: which tumor type has the most heterogeneity? Int J Surg Pathol 24:103–107. https://doi.org/10.1177/1066896915605179
Jang MH, Kim HJ, Chung YR, Lee Y, Park SY (2017) A comparison of Ki-67 counting methods in luminal breast cancer: the average method vs. the hot spot method. PLoS ONE 12:e0172031. https://doi.org/10.1371/journal.pone.0172031
Khor LY, Bae K, Paulus R, Al-Saleem T, Hammond ME, Grignon DJ, Che M, Venkatesan V, Byhardt RW, Rotman M, Hanks GE, Sandler HM, Pollack A (2009) MDM2 and Ki-67 predict for distant metastasis and mortality in men treated with radiotherapy and androgen deprivation for prostate cancer: RTOG 92-02. J Clin Oncol 27:3177–3184. https://doi.org/10.1200/JCO.2008.19.8267
Loehberg CR, Almstedt K, Jud SM, Haeberle L, Fasching PA, Hack CC, Lux MP, Thiel FC, Schrauder MG, Brunner M, Bayer CM, Hein A, Heusinger K, Heimrich J, Bani MR, Renner SP, Hartmann A, Beckmann MW, Wachter DL (2013) Prognostic relevance of Ki-67 in the primary tumor for survival after a diagnosis of distant metastasis. Breast Cancer Res Treat 138:899–908. https://doi.org/10.1007/s10549-013-2460-y
Margulis V, Lotan Y, Karakiewicz PI, Fradet Y, Ashfaq R, Capitanio U, Montorsi F, Bastian PJ, Nielsen ME, Muller SC, Rigaud J, Heukamp LC, Netto G, Lerner SP, Sagalowsky AI, Shariat SF (2009) Multi-institutional validation of the predictive value of Ki-67 labeling index in patients with urinary bladder cancer. J Natl Cancer Inst 101:114–119. https://doi.org/10.1093/jnci/djn451
Ohno S, Chow LW, Sato N, Masuda N, Sasano H, Takahashi F, Bando H, Iwata H, Morimoto T, Kamigaki S, Nakayama T, Nakamura S, Kuroi K, Aogi K, Kashiwaba M, Yamashita H, Hisamatsu K, Ito Y, Yamamoto Y, Ueno T, Fakhrejahani E, Yoshida N, Toi M (2013) Randomized trial of preoperative docetaxel with or without capecitabine after 4 cycles of 5-fluorouracil–epirubicin–cyclophosphamide (FEC) in early-stage breast cancer: exploratory analyses identify Ki67 as a predictive biomarker for response to neoadjuvant chemotherapy. Breast Cancer Res Treat 142:69–80. https://doi.org/10.1007/s10549-013-2691-y
Sauter G, Lee J, Bartlett JM, Slamon DJ, Press MF (2009) Guidelines for human epidermal growth factor receptor 2 testing: biologic and methodologic considerations. J Clin Oncol 27:1323–1333. https://doi.org/10.1200/JCO.2007.14.8197
Slamon DJ, Neven P, Chia S, Fasching PA, De Laurentiis M, Im SA, Petrakova K, Bianchi GV, Esteva FJ, Martin M, Nusch A, Sonke GS, De la Cruz-Merino L, Beck JT, Pivot X, Vidam G, Wang Y, Rodriguez Lorenc K, Miller M, Taran T, Jerusalem G (2018) Phase III randomized study of ribociclib and fulvestrant in hormone receptor-positive, human epidermal growth factor receptor 2-negative advanced breast cancer: MONALEESA-3. J Clin Oncol 36:2465–2472. https://doi.org/10.1200/JCO.2018.78.9909
Sledge GW Jr, Toi M, Neven P, Sohn J, Inoue K, Pivot X, Burdaeva O, Okera M, Masuda N, Kaufman PA, Koh H, Grischke EM, Frenzel M, Lin Y, Barriga S, Smith IC, Bourayou N, Llombart-Cussac A (2017) MONARCH 2: abemaciclib in combination with fulvestrant in women with HR+/HER2− advanced breast cancer who had progressed while receiving endocrine therapy. J Clin Oncol 35:2875–2884. https://doi.org/10.1200/JCO.2017.73.7585
Sotiriou C, Pusztai L (2009) Gene-expression signatures in breast cancer. N Engl J Med 360:790–800. https://doi.org/10.1056/NEJMra0801289
Sotiriou C, Wirapati P, Loi S, Harris A, Fox S, Smeds J, Nordgren H, Farmer P, Praz V, Haibe-Kains B, Desmedt C, Larsimont D, Cardoso F, Peterse H, Nuyten D, Buyse M, Van de Vijver MJ, Bergh J, Piccart M, Delorenzi M (2006) Gene expression profiling in breast cancer: understanding the molecular basis of histologic grade to improve prognosis. J Natl Cancer Inst 98:262–272. https://doi.org/10.1093/jnci/djj052
Sparano JA, Gray RJ, Makower DF, Pritchard KI, Albain KS, Hayes DF, Geyer CE Jr, Dees EC, Goetz MP, Olson JA Jr, Lively T, Badve SS, Saphner TJ, Wagner LI, Whelan TJ, Ellis MJ, Paik S, Wood WC, Ravdin PM, Keane MM, Gomez Moreno HL, Reddy PS, Goggins TF, Mayer IA, Brufsky AM, Toppmeyer DL, Kaklamani VG, Berenberg JL, Abrams J, Sledge GW Jr (2018) Adjuvant chemotherapy guided by a 21-gene expression assay in breast cancer. N Engl J Med 379:111–121. https://doi.org/10.1056/NEJMoa1804710
Sparano JA, Gray RJ, Makower DF, Pritchard KI, Albain KS, Hayes DF, Geyer CE Jr, Dees EC, Perez EA, Olson JA Jr, Zujewski J, Lively T, Badve SS, Saphner TJ, Wagner LI, Whelan TJ, Ellis MJ, Paik S, Wood WC, Ravdin P, Keane MM, Gomez Moreno HL, Reddy PS, Goggins TF, Mayer IA, Brufsky AM, Toppmeyer DL, Kaklamani VG, Atkins JN, Berenberg JL, Sledge GW (2015) Prospective validation of a 21-gene expression assay in breast cancer. N Engl J Med 373:2005–2014. https://doi.org/10.1056/NEJMoa1510764
Sueta A, Yamamoto Y, Hayashi M, Yamamoto S, Inao T, Ibusuki M, Murakami K, Iwase H (2014) Clinical significance of pretherapeutic Ki67 as a predictive parameter for response to neoadjuvant chemotherapy in breast cancer: is it equally useful across tumor subtypes? Surgery 155:927–935. https://doi.org/10.1016/j.surg.2014.01.009
Taran FA, Schneeweiss A, Lux MP, Janni W, Hartkopf AD, Nabieva N, Overkamp F, Kolberg HC, Hadji P, Tesch H, Wockel A, Ettl J, Luftner D, Wallwiener M, Muller V, Beckmann MW, Belleville E, Wallwiener D, Brucker SY, Fasching PA, Fehm TN, Schutz F (2018) Update breast cancer 2018 (Part 1)—primary breast cancer and biomarkers. Geburtshilfe Frauenheilkd 78:237–245. https://doi.org/10.1055/s-0044-101613
Tripathy D, Im SA, Colleoni M, Franke F, Bardia A, Harbeck N, Hurvitz SA, Chow L, Sohn J, Lee KS, Campos-Gomez S, Villanueva Vazquez R, Jung KH, Babu KG, Wheatley-Price P, De Laurentiis M, Im YH, Kuemmel S, El-Saghir N, Liu MC, Carlson G, Hughes G, Diaz-Padilla I, Germa C, Hirawat S, Lu YS (2018) Ribociclib plus endocrine therapy for premenopausal women with hormone-receptor-positive, advanced breast cancer (MONALEESA-7): a randomised phase 3 trial. Lancet Oncol 19:904–915. https://doi.org/10.1016/S1470-2045(18)30292-4
Turner NC, Slamon DJ, Ro J, Bondarenko I, Im SA, Masuda N, Colleoni M, DeMichele A, Loi S, Verma S, Iwata H, Harbeck N, Loibl S, Andre F, Puyana Theall K, Huang X, Giorgetti C, Huang Bartlett C, Cristofanilli M (2018) Overall survival with palbociclib and fulvestrant in advanced breast cancer. N Engl J Med. https://doi.org/10.1056/NEJMoa1810527
von Minckwitz G, Untch M, Blohmer JU, Costa SD, Eidtmann H, Fasching PA, Gerber B, Eiermann W, Hilfrich J, Huober J, Jackisch C, Kaufmann M, Konecny GE, Denkert C, Nekljudova V, Mehta K, Loibl S (2012) Definition and impact of pathologic complete response on prognosis after neoadjuvant chemotherapy in various intrinsic breast cancer subtypes. J Clin Oncol 30:1796–1804. https://doi.org/10.1200/JCO.2011.38.8595
Voros A, Csorgo E, Kovari B, Lazar P, Kelemen G, Rusz O, Nyari T, Cserni G (2015) Different methods of pretreatment Ki-67 labeling index evaluation in core biopsies of breast cancer patients treated with neoadjuvant chemotherapy and their relation to response to therapy. Pathol Oncol Res 21:147–155. https://doi.org/10.1007/s12253-014-9800-z
Warth A, Cortis J, Soltermann A, Meister M, Budczies J, Stenzinger A, Goeppert B, Thomas M, Herth FJ, Schirmacher P, Schnabel PA, Hoffmann H, Dienemann H, Muley T, Weichert W (2014) Tumour cell proliferation (Ki-67) in non-small cell lung cancer: a critical reappraisal of its prognostic role. Br J Cancer 111:1222–1229. https://doi.org/10.1038/bjc.2014.402
Wiesner FG, Magener A, Fasching PA, Wesse J, Bani MR, Rauh C, Jud S, Schrauder M, Loehberg CR, Beckmann MW, Hartmann A, Lux MP (2009) Ki-67 as a prognostic molecular marker in routine clinical use in breast cancer patients. Breast 18:135–141. https://doi.org/10.1016/j.breast.2009.02.009
Wockel A, Festl J, Stuber T, Brust K, Stangl S, Heuschmann PU, Albert US, Budach W, Follmann M, Janni W, Kopp I, Kreienberg R, Kuhn T, Langer T, Nothacker M, Scharl A, Schreer I, Link H, Engel J, Fehm T, Weis J, Welt A, Steckelberg A, Feyer P, Konig K, Hahne A, Kreipe HH, Knoefel WT, Denkinger M, Brucker S, Luftner D, Kubisch C, Gerlach C, Lebeau A, Siedentopf F, Petersen C, Bartsch HH, Schulz-Wendtland R, Hahn M, Hanf V, Muller-Schimpfle M, Henscher U, Roncarati R, Katalinic A, Heitmann C, Honegger C, Paradies K, Bjelic-Radisic V, Degenhardt F, Wenz F, Rick O, Holzel D, Zaiss M, Kemper G, Budach V, Denkert C, Gerber B, Tesch H, Hirsmuller S, Sinn HP, Dunst J, Munstedt K, Bick U, Fallenberg E, Tholen R, Hung R, Baumann F, Beckmann MW, Blohmer J, Fasching PA, Lux MP, Harbeck N, Hadji P, Hauner H, Heywang-Kobrunner S, Huober J, Hubner J, Jackisch C, Loibl S, Luck HJ, von Minckwitz G, Mobus V, Muller V, Nothlings U, Schmidt M, Schmutzler R, Schneeweiss A, Schutz F, Stickeler E, Thomssen C, Untch M, Wesselmann S, Bucker A, Krockenberger M (2018) Interdisciplinary screening, diagnosis, therapy and follow-up of breast cancer. Guideline of the DGGG and the DKG (S3-Level, AWMF Registry Number 032/045OL, December 2017)—Part 1 with recommendations for the screening, diagnosis and therapy of breast cancer. Geburtshilfe Frauenheilkd 78:927–948. https://doi.org/10.1055/a-0646-4522
Wockel A, Lux MP, Janni W, Hartkopf AD, Nabieva N, Taran FA, Overkamp F, Hadji P, Tesch H, Ettl J, Luftner D, Muller V, Welslau M, Belleville E, Brucker SY, Schutz F, Fasching PA, Fehm T, Schneeweiss A, Kolberg HC (2018) Update breast cancer 2018 (Part 3)—genomics, individualized medicine and immune therapies—in the middle of a new era: prevention and treatment strategies for early breast cancer. Geburtshilfe Frauenheilkd 78:1110–1118. https://doi.org/10.1055/s-0043-111601
Yerushalmi R, Woods R, Ravdin PM, Hayes MM, Gelmon KA (2010) Ki67 in breast cancer: prognostic and predictive potential. Lancet Oncol 11:174–183. https://doi.org/10.1016/S1470-2045(09)70262-1
Zabaglo L, Salter J, Anderson H, Quinn E, Hills M, Detre S, A’Hern R, Dowsett M (2010) Comparative validation of the SP6 antibody to Ki67 in breast cancer. J Clin Pathol 63:800–804. https://doi.org/10.1136/jcp.2010.077578
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
AH has received honoraria from Roche, Janssen, Merck Sharp Dome, AstraZeneca, Novartis, Cepheid, Abbvie and Boehringer-Ingelheim. His institution performs research with Grants from Biontech, Janssen, Cepheid, Roche and Qiagen. DJS has received honoraria from Novartis, Pfizer and Lilly and owns stock in Pfizer, Amgen and Seattle Genetics. His institution performs research with Grants from Novartis. PAF has received honoraria from Novartis, Amgen, Pfizer, Roche, Teva, Celgene, Eisai, Daiichi-Sankyo and PUMA. His institution performs research with Grants from Biontech and Novartis. PG has received honoraria from Novartis, travel support from Novartis, Roche and PharmaMar. MPL has received honoraria from Novartis, AstraZeneca, Roche, Pfizer, Lilly Eisai, Genomic Health and Tesaro. The remaining authors declare that they have no conflict of interest.
Ethical approval
Approval for this retrospective study was obtained from the Ethics Committee of the Faculty of Medicine at Friedrich Alexander University of Erlangen-Nuremberg. All procedures were in accordance with the Ethical Standards of the Institutional and/or National Research Committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from each individual participant included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fasching, P.A., Gass, P., Häberle, L. et al. Prognostic effect of Ki-67 in common clinical subgroups of patients with HER2-negative, hormone receptor-positive early breast cancer. Breast Cancer Res Treat 175, 617–625 (2019). https://doi.org/10.1007/s10549-019-05198-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-019-05198-9