Abstract
Objectives
Human epidermal growth factor receptor 2 (HER2, ERBB2) is a valuable prognostic and predictive biomarker in breast cancer. Accurate assessment of HER2 status is essential in selecting the patients with invasive breast cancer who will likely response to HER2-targeted therapies. Some major modifications in the diagnostic recommendation for fluorescence in situ hybridization (FISH) have been made in the updated 2018 American Society of Clinical Oncology (ASCO)/College of American Pathologist (CAP) guideline. According to the revised guideline, concomitant IHC assays are required to arrive at the most accurate HER2 status designation after HER2 FISH equivocal results; however, little is known about its influence on the clinical practice of pathologist. The purpose of this study was to evaluate the impact of the revised 2018 ASCO/CAP guidelines on the HER2 status designation.
Methods
We retrospectively reviewed the HER2 FISH testing results from 2233 cases of invasive breast cancer between January 2014 and December 2017. Concomitant immunohistochemistry (IHC) were performed on the same tissue blocks that were used for the FISH testing.
Results
Compared to the 2013 guidelines, the HER2 status in 183 (8.2%) cases were re-defined when reassessed by the 2018 guidelines. Among these 183 cases, 175 equivocal cases according to the 2013 guideline were re-defined as HER2 negative (n = 173) or HER2 positive (n = 2). Eight previously classified as HER2 positive cases were converted to negative in the 2018 scheme, all of which were with HER2 IHC scores of 1+ or 2+. The number of cases in the negative category was 1705 according to the 2018 guidelines as opposed to 1524 by the 2013 guidelines.
Conclusions
The updated 2018 ASCO/CAP guidelines eliminated the FISH equivocal category, which can be attributed to reflex HER2 IHC, and partly ease the dilemma for clinical practice. Reflex IHC for FISH equivocal cases is of prime importance; furthermore, HER2 FISH results were converted from positivity to negativity based on the concomitant IHC results in a small percentage of cases. In all, implementation of the 2018 ASCO/CAP guidelines provides much clearer instructions and recommendations for the HER2 status designation, and thus reduces the risk of misdiagnosis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Breast cancer is the most common cancer in women worldwide [1]. Human epidermal growth factor receptor 2 (HER2), also called ERBB2 and encoded by the ERBB2 (v-erb-b2 avian erythroblastic leukemia viral oncogene homolog 2) gene, is amplified or overexpressed in about 15–20% of invasive breast cancers [2]. Patients with HER2 positive are associated with poor prognosis and tend to have early relapse and distant metastasis in the absence of systemic therapy [3,4,5,6,7,8]. HER2 positivity enables patient selection for HER2- targeted therapies, such as trastuzumab, pertuzumab, and other anti-HER2 agents, which target HER2 or its downstream pathways [9,10,11,12,13,14]. Therefore, accurate assessment of HER status becomes more and more critical. Evaluation of HER2 protein expression by immunohistochemistry (IHC) and gene amplification via fluorescence in situ hybridization (FISH) are the two technologies most commonly applied to determined HER2 status [7, 15, 16].
The clinical importance of HER2 evaluation led to the need of improving the accuracy and reproducibility of HER2 testing. Thus, the American Society of Clinical Oncology (ASCO)/College of American Pathologists (CAP) have initially issued detailed guideline recommendations for conducting and interpreting HER2 testing in clinical practice in 2007, and first revised them in 2013. Later in 2018, ASCO/CAP updated the guidelines again [17, 18]. The updated 2018 guidelines have made some major modification in the diagnostic recommendations for FISH, including the following: (1) cases with a HER2/CEP17 ratio less than 2.0 as well as a average HER2 copy number ≥ 4.0 and < 6.0/cell can now be diagnosed as negative or even positive if they qualify based on HER2 IHC scores of 0/1+/2+ or 3+, respectively; (2) cases with a HER2/CEP17 ratio less than 2.0 and a average HER2 copy number ≥ 6.0/cell are diagnosed as negative if IHC score was 0/1+; (3) cases with a HER2/CEP17 ratio less than 2.0 and a average HER2 copy number ≥ 6.0/cell can be still diagnosed as positive if they had 2+ or 3+ IHC score; (4) cases with HER/CEP17 ratio of 2.0 or more are re-classified as negative, given the fact that they have low HER2 copy number (less than 4.0) and IHC score (less than 3+).
To investigate how the updated guidelines affected the final HER2 designation, we retrospectively examined the HER2 status of 2233 patients with invasive breast cancer who were enrolled in our study for FISH testing between January 2014 and December 2017, and compared the difference in HER2 status interpretation based on the 2013 and 2018 ASCO/CAP Guidelines.
Materials and methods
Case cohort
We retrospectively reviewed the HER2 FISH testing results from 2233 cases of invasive breast cancer between January 2014 and December 2017. In our institution, both HER2 IHC and HER2 FISH testing are routinely performed for all patients who are diagnosed as invasive or metastatic breast cancer. Specimens were composed of core needle biopsy samples, surgical excisions, and biopsy samples from metastatic sites.
The study was approved by the Ethics Committee of Guangdong General Hospital, Guangdong Academy of Medical Sciences, Guangzhou, China.
Fluorescence in situ hybridization (FISH)
HER2 FISH testing was performed on a 4-µm formalin-fixed paraffin-embedded tissue specimens using the FDA-approved PathVysion HER2 DNA probe kit (Abbott Molecular, Des Plaines, IL, USA) according to the manufacturer’s recommended protocols, which has been described in detail previously [19, 20]. HER2 FISH signals were independently analyzed by one certified pathologist (ZHL) and one certified technologist (DYL). Each counted 30 cells from two non-overlapping areas. When there was a conflict between their scores, another pathologist (FPX) who was the most experienced in FISH would review the slide and reached the final result. For investigational purpose, all cases were reclassified using both 2013 and 2018 CAP/ASCO guidelines [17, 18].
Immunohistochemistry (IHC)
The expression of HER protein was evaluated by IHC staining using standardized automated methodology (Ventana Medical Systems, Oro Valley, AZ). IHC for HER2 was performed using the c-erb-B2 antibody (4B5, Ventana). Standardized immunohistochemical protocols were followed with control slides as appropriate. IHC slides were scored by two pathologists according to the guidelines already available at the time of testing.
Statistical analyses
Data analysis was done by use of IBM SPSS statistic software version 22.0 (v22.0; IBM, Chicago, IL) or Graphad (GraphPad Software Inc, La Jolla, CA). The Chi square test was used to compare these two groups (with and without HER2 status changes) with respect to the proportion with CEP17 signals ≥ 3. Statistical significance was assumed if P < 0.05.
Results
2240 patients with primary or metastatic invasive breast cancer were tested for HER2 status with FISH between 2014 and 2017. Seven cases were excluded from this study for a lack of HER2 and CEP17 signal details. We re-classified the remaining 2233 cases based on their HER2 FISH results and IHC scores using both 2013 and 2018 guidelines. The distribution of HER2 FISH results is summarized in Table 1. Meanwhile, we also found that the interpretations of HER2 status for a total of 183 (8.2%) cases were changed when reassessed by 2018 guidelines (Table 2). Changes are described in detail as follows.
Cases remaining HER2 negative according to both 2013 and 2018 ASCO/CAP guidelines
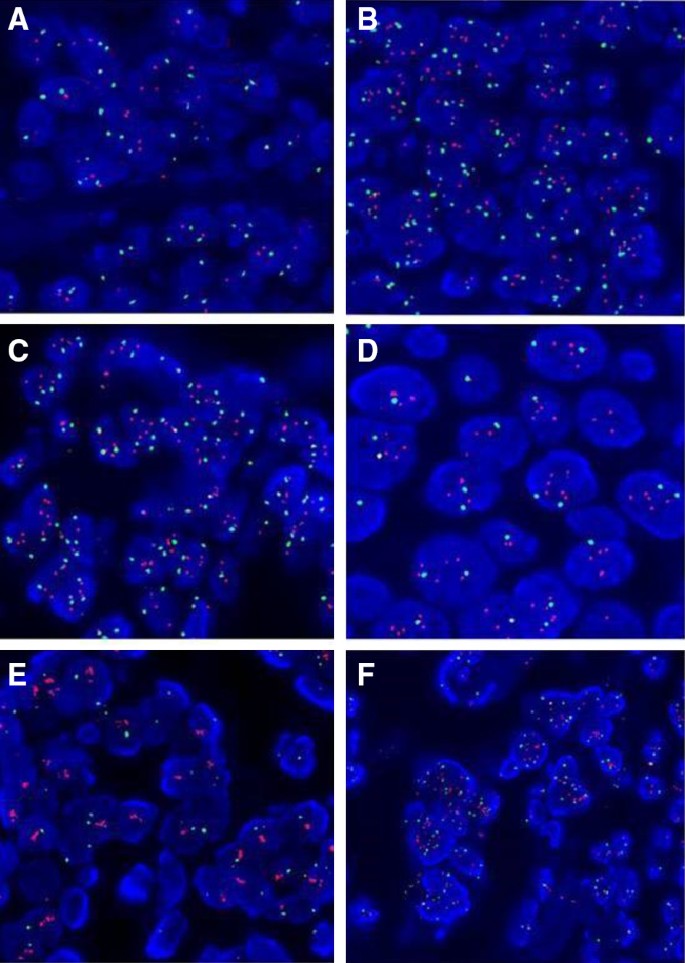
In total, 1524 (68.2%) patients were diagnosed as negative for HER2 according to both guidelines. These were cases with HER2/CEP17 ratio less than 2.0 and HER2 gene copy number less than 4.0. A representative FISH image is shown in Fig. 1a.
Representative HER2 fluorescence in situ hybridization signal (red signals = HER2, green = CEP17). a This case was scored as negative under both 2013 and 2018 scoring criteria.HER2/CEP17 ratio = 1.048; HER2/nucleus ratio = 2.167. b A case was scored as HER2 equivocal with 2013 criteria but became negative with the 2018 scoring criteria.HER2/CEP17 ratio = 1.77; HER2/nucleus = 4.75; IHC HER2 2+. c One case was scored as HER2 equivocal with 2013 criteria but became positive for HER2 amplification with the 2018 scoring criteria.HER2/CEP17 ratio = 1.861; HER2/nucleus = 4.9; IHC HER2 3+. d One case scored as positive by the 2013 criteria was scored as negative per 2018 criteria. HER2/CEP17ratio = 2.367; HER2/nucleus = 4.733;IHC HER2 1+. e This case was scored as positive under both scoring schema.HER2/CEP17 ratio = 10.467; HER2/nucleus = 26.167; IHC HER2 3+. f An example of the fourteen cases with HER2/nucleus ratio greater than 6.0 but a HER2/CEP17 ration less than 2.0 was scored as positive under both scoring schema.HER2/CEP17 ratio = 1.59; HER2/nucleus = 6.467;CEP17/nucleus = 4.067; IHC HER2 2+
Cases diagnosed as HER2 equivocal according to 2013 ASCO/CAP guidelines but HER2 negative when 2018 ASCO/CAP guidelines was applied
173 (7.7%) cases initially classified as HER2 equivocal in 2013 scoring schema were re-diagnosed as negative according to the revised 2018 guidelines, representing cases with HER2/CEP17 ratio less than 2.0 and average HER2 copy number ≥ 4 and < 6/cell. An example is illustrated in Fig. 1b. Same tissue samples used for FISH were used for the concomitant IHC testing. 19 of these 173 cases had HER2 IHC scored as 0/1+, and the remaining cases were assessed as 2+ HER2 IHC. Based on the revised 2018 guidelines, another certified pathologist recounted at least 30 cells included the invasive area with IHC 2+ staining, and generated similar HER2 FISH results. Thus, all of the 173 cases in the original cohort that were reported as equivocal with a comment recommending repeat testing on the same specimen using additional chromosome 17 probe, were reassessed as negative.
Cases diagnosed as HER2 equivocal according to 2013 ASCO/CAP guidelines but HER2 positive when 2018 ASCO/CAP guidelines was applied
Only 2 (0.1%) cases considered as equivocal in the 2013 schema were re-diagnosed as positive under the 2018 guidelines. One of the two cases is presented in Fig. 1c. The same biopsy specimens for FISH underwent IHC testing for HRE2 expression, and the results showed that the two cases had 3+ HER2 IHC score.
Cases diagnosed as HER2 positive according to 2013 ASCO/CAP guidelines but HER2 negative when 2018 ASCO/CAP guidelines was applied
Eight (0.4%) cases categorized as positive under the 2013 guidelines were re-classified in the negative category by 2018 guidelines, given the low HER2/nucleus ratio less than 4.0 and the IHC score less than 3+. According to the 2018 scoring schema, the HER2/nucleus ratio less than 4.0 would not warrant a positive interpretation on its own, were it not for the condition with HER2/CEP17 ratio just slightly over threshold and the IHC score less than 3+. An example is illustrated in Fig. 1d. In this category only one case had 1+ IHC, and seven had 2+ equivocal IHC score. All 7 equivocal cases were recounted additional nuclei to define the final HER2 FISH classification.
Cases remaining HER2 positive according to both 2013 and 2018 ASCO/CAP guidelines
526 (23.5%) cases that were scored positive in the 2013 schema remained positive with the revised scoring system. These were cases with HER2/nucleus counts of 4.0 or greater and HER2 IHC scored 2+or 3+. A representative FISH image is shown in Fig. 1e.
This group included fourteen cases with HER2/nucleus greater than 6.0 but a HER2/CEP17 ratio less than 2.0, given an elevated CEP17 count (“polysomy”). An example is illustrated in Fig. 1f and detailed data for these cases is provided in Table 3, including interpretation based on both scoring schemes. In fact, polysomy 17 can lead to a low HER2/CEP17 ratio despite a high HER2/nucleus count, but the relationship between chromosome 17 polysomy and the response to HER2-targeted therapy is not entirely clear, partially due to inadequate clinical reports in published studies [21,22,23].
To sum up, the final HER2 FISH classification of 183 cases in our study cohort were significantly changed when the updated 2018 guidelines was applied. The frequency of chromosome 17 polysomy (defined as a mean of ≥ 3 CEP17 signals/cell) was re-classified in the new recommendations (p < 0.001; Chi square test). Based on the 2018 guidelines, HER2 status reassessment was present in 86/183 (47.0%) cases with chromosome 17 polysomy, as compared to 324/2050 (15.8%) cases without HER2 status reassignment (Table 4). Overall, 410 of the 2233 cases (18.4%) are considered to have chromosome 17 polysomy.
Discussion
With the development of anti-HER2- targeted agents, accurate assessment of HER2 status is the most important factor in selecting patients who will likely respond to the therapy, and avoiding the use of these costly drugs in patients with HER2 negative [10, 12, 24]. Thus, the accuracy and reproducibility of HER2 testing is critical. Large numbers of literatures have been published to compare the advantages and disadvantages of different techniques developed so far for the assessment of HER2 status, and found that FISH assay demonstrated greater accuracy compared with IHC [25,26,27,28]. However, studies have also shown that IHC and FISH had the highest concordance rates in tumors with HER2 IHC 0/1+ and 3+, and lowest for cases with HER2 IHC 2+ equivocal [26, 29]. Due to the bias between HER2 IHC and FISH results and the fact that modification of guideline recommendations could affect the final HER2 classification, it is important to understand the impact of the updated guideline on the determination of HER2 gene status.
ASCO/CAP updated the recommendations of the testing guidelines for HER2 and the revised guidelines were published in 2018 [18]. One of the significant modifications in the updated guidelines was redefining the interpretation criteria for arriving at the most accurate HER2 status designation (positive or negative) based on the interpretation of IHC and FISH results. Data from our retrospective single-institution comparative study has shown that 183 out of the reviewed 2233 invasive breast cancer cases had re-classified HER2 status, among which 175 cases were changed from HER2 FISH equivocal to negative (n = 173) or positive (n = 2) using the 2018 ASCO/CAP guidelines (Tables 1, 2). The findings were concordant with previous reports and showed that group 4 (HER2 equivocal using 2013 guideline) and group 5 (HER2 positive but the HER2 copy numberless than 4.0) were strongly correlated with immunohistochemistry 0/1 + status.
The subsequent changes implied that the guideline updates were effective in identifying more patients with HER2 negative who may avoid the use of costly anti -HER2-targeted agents, along with their harmful side effects.
According to our data, the most significant change in the HER2 status assignment was noticed in tumors with CEP17 signals ≥ 3, also called chromosome 17 polysomy group (Tables 3, 4). Prevalence rate of chromosome 17 polysomy in breast cancer was reported ranging from 3 to 46% [30,31,32,33]. Several studies and our previous study also showed that the frequency of chromosome polysomy 17 was significantly higher in the HER2-amplified or HER2 equivocal (using 2013 ASCO/CAP guidelines) breast cancers than those in the non-amplified ones [32, 34]. Since the re-categorized cases mostly occurred in HER2-amplified or HER2 equivocal group using the 2013 ASCO/CAP guidelines in the study, this may be the explanation for why the change in the HER2 status assignment was significantly associated with chromosome 17 polysomy. However, most of (173/175) the previous HER2 equivocal cases were re-diagnosed as negative after using the new guidelines in our cohort, and the vast majority of (167/173) them were IHC 2+. Whether these cases were truly HER2-negative requires further investigations. In addition, fourteen cases with HER2/nucleus greater than 6.0 but a HER2/CEP17 ratio less than 2.0 due to polysomy were all classified as positive by both 2013 and 2018 guidelines. A very recent study, which was conducted based on the largest number of breast cancer cases with average HER2 copy number ≥ 6 HER2 copy but HER2/CEP17 ratio < 2.0, showed that all of these patients did not benefit from trastuzumab therapy [35]. More studies are needed to evaluate whether the subgroup within our cohort is worth treating with HER2-targeted therapy. Nevertheless, some studies have demonstrated that true chromosome 17 polysomy was a rare event in breast cancers [36].
To conclude, our single-center retrospective study is the first to evaluate the impact of the 2018 ASCO/CAP updated guidelines on the final HER2 FISH classification based on a large number of invasive breast cancers cases. Our analysis has demonstrated that a substantial number of patients with breast cancer would be classified differently when interpreted their initial FISH results under the 2018 guidelines. During the revision of our manuscript, a study including 331 breast cancer cases had also showed that the 2018 guideline would increase the negative rate of HER2 FISH for IHC 2+ patients [37]. Our study has shown that interpretation of HER2 status for breast cancer with chromosome 17 polysomy was most affected by the updated 2018 guidelines. Although the new guidelines are helpful, and can more accurately identify HER2 gene status in breast cancer, diagnostic judgment and long-term outcome data remains the touchstone of the evaluation of the new criterion.
Abbreviations
- HER2:
-
Human epidermal growth factor 2
- IHC:
-
Immunohistochemistry
- FISH:
-
Fluorescence in situ hybridization
- ASCO:
-
American Society of Clinical Oncology
- CAP:
-
College of American Pathologists
References
McPherson K, Steel CM, Dixon JM (2000) ABC of breast diseases. Breast cancer-epidemiology, risk factors, and genetics. BMJ 321:624–628
Wolff AC, Hammond ME, Schwartz JN, Hagerty KL, Allred DC, Cote RJ, Dowsett M, Fitzgibbons PL, Hanna WM, Langer A, McShane LM, Paik S, Pegram MD, Perez EA, Press MF, Rhodes A, Sturgeon C, Taube SE, Tubbs R, Vance GH, van de Vijver M, Wheeler TM, Hayes DF, American Society of Clinical Oncology/College of American P (2007) American Society of Clinical Oncology/College of American Pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. Arch Pathol Lab Med 131:18–43. https://doi.org/10.1043/1543-2165(2007)131%5B18:ASOCCO%5D2.0.CO;2
Adamczyk A, Kruczak A, Harazin-Lechowska A, Ambicka A, Grela-Wojewoda A, Domagala-Haduch M, Janecka-Widla A, Majchrzyk K, Cichocka A, Rys J, Niemiec J (2018) Relationship between HER2 gene status and selected potential biological features related to trastuzumab resistance and its influence on survival of breast cancer patients undergoing trastuzumab adjuvant treatment. Onco Targets Ther 11:4525–4535. https://doi.org/10.2147/OTT.S166983
Cooke T, Reeves J, Lannigan A, Stanton P (2001) The value of the human epidermal growth factor receptor-2 (HER2) as a prognostic marker. Eur J Cancer 37(Suppl 1):S3–S10
Curigliano G, Viale G, Bagnardi V, Fumagalli L, Locatelli M, Rotmensz N, Ghisini R, Colleoni M, Munzone E, Veronesi P, Zurrida S, Nole F, Goldhirsch A (2009) Clinical relevance of HER2 overexpression/amplification in patients with small tumor size and node-negative breast cancer. J Clin Oncol 27:5693–5699. https://doi.org/10.1200/JCO.2009.22.0962
Press MF, Bernstein L, Thomas PA, Meisner LF, Zhou JY, Ma Y, Hung G, Robinson RA, Harris C, El-Naggar A, Slamon DJ, Phillips RN, Ross JS, Wolman SR, Flom KJ (1997) HER-2/neu gene amplification characterized by fluorescence in situ hybridization: poor prognosis in node-negative breast carcinomas. J Clin Oncol 15:2894–2904. https://doi.org/10.1200/JCO.1997.15.8.2894
Rosen PP, Lesser ML, Arroyo CD, Cranor M, Borgen P, Norton L (1995) Immunohistochemical detection of HER2/neu in patients with axillary lymph node negative breast carcinoma. A study of epidemiologic risk factors, histologic features, and prognosis. Cancer 75:1320–1326
Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL (1987) Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 235:177–182
Dang C, Iyengar N, Datko F, D’Andrea G, Theodoulou M, Dickler M, Goldfarb S, Lake D, Fasano J, Fornier M, Gilewski T, Modi S, Gajria D, Moynahan ME, Hamilton N, Patil S, Jochelson M, Norton L, Baselga J, Hudis C (2015) Phase II study of paclitaxel given once per week along with trastuzumab and pertuzumab in patients with human epidermal growth factor receptor 2-positive metastatic breast cancer. J Clin Oncol 33:442–447. https://doi.org/10.1200/JCO.2014.57.1745
Frenel JS, Bourbouloux E, Berton-Rigaud D, Sadot-Lebouvier S, Zanetti A, Campone M (2009) Lapatinib in metastatic breast cancer. Womens Health (Lond) 5:603–612. https://doi.org/10.2217/whe.09.54
Ji G, Zhang C, Guan S, Yao X (2018) Erlotinib for Progressive Brain and Leptomeningeal Metastases From HER2-positive Breast Cancer After Treatment Failure With Trastuzumab and Lapatinib: Experience and Review of Literature. Clin Breast Cancer. https://doi.org/10.1016/j.clbc.2018.07.022
Rastogi P (2010) Targeting alternative pathways in HER2-positive breast cancer. Oncology (Williston Park) 24:415–416
Schoffski P, Cresta S, Mayer IA, Wildiers H, Damian S, Gendreau S, Rooney I, Morrissey KM, Spoerke JM, Ng VW, Singel SM, Winer E (2018) A phase Ib study of pictilisib (GDC-0941) in combination with paclitaxel, with and without bevacizumab or trastuzumab, and with letrozole in advanced breast cancer. Breast Cancer Res 20:109. https://doi.org/10.1186/s13058-018-1015-x
Urruticoechea A, Rizwanullah M, Im SA, Ruiz ACS, Lang I, Tomasello G, Douthwaite H, Badovinac Crnjevic T, Heeson S, Eng-Wong J, Munoz M (2017) Randomized Phase III Trial of Trastuzumab Plus Capecitabine With or Without Pertuzumab in Patients With Human Epidermal Growth Factor Receptor 2-Positive Metastatic Breast Cancer Who Experienced Disease Progression During or After Trastuzumab-Based Therapy. J Clin Oncol 35:3030–3038. https://doi.org/10.1200/JCO.2016.70.6267
Pathmanathan N, Bilous AM (2012) HER2 testing in breast cancer: an overview of current techniques and recent developments. Pathology 44:587–595. https://doi.org/10.1097/PAT.0b013e328359cf9a
Tubbs RR, Hicks DG, Cook J, Downs-Kelly E, Pettay J, Hartke MB, Hood L, Neelon R, Myles J, Budd GT, Moore HC, Andresen S, Crowe JP (2007) Fluorescence in situ hybridization (FISH) as primary methodology for the assessment of HER2 Status in adenocarcinoma of the breast: a single institution experience. Diagn Mol Pathol 16:207–210. https://doi.org/10.1097/PDM.0b013e318064c72a
Ramakrishna N, Temin S, Chandarlapaty S, Crews JR, Davidson NE, Esteva FJ, Giordano SH, Gonzalez-Angulo AM, Kirshner JJ, Krop I, Levinson J, Modi S, Patt DA, Perez EA, Perlmutter J, Winer EP, Lin NU (2014) Recommendations on disease management for patients with advanced human epidermal growth factor receptor 2-positive breast cancer and brain metastases: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol 32:2100–2108. https://doi.org/10.1200/JCO.2013.54.0955
Wolff AC, Hammond MEH, Allison KH, Harvey BE, Mangu PB, Bartlett JMS, Bilous M, Ellis IO, Fitzgibbons P, Hanna W, Jenkins RB, Press MF, Spears PA, Vance GH, Viale G, McShane LM, Dowsett M (2018) Human Epidermal Growth Factor Receptor 2 Testing in Breast Cancer: American Society of Clinical Oncology/College of American Pathologists Clinical Practice Guideline Focused Update. J Clin Oncol 36:2105–2122. https://doi.org/10.1200/JCO.2018.77.8738
Liu YH, Xu FP, Rao JY, Zhuang HG, Luo XL, Li L, Luo DL, Zhang F, Xu J (2009) Justification of the change from 10 to 30% for the immunohistochemical HER2 Scoring criterion in breast cancer. Am J Clin Pathol 132:74–79. https://doi.org/10.1309/AJCPG3LCVKPNFJ6R
Xu FP, Wang K, Xu J, Chen J, Zhang YF, Wu HM, Zhang MH, Long XX, Luo XL, Zhang KP, Lin DY, Liu YH (2017) Impact of repeat HER2 testing after initial equivocal HER2 FISH results using 2013 ASCO/CAP guidelines. Breast Cancer Res Treat 166:757–764. https://doi.org/10.1007/s10549-017-4479-y
Bartlett JM, Munro AF, Dunn JA, McConkey C, Jordan S, Twelves CJ, Cameron DA, Thomas J, Campbell FM, Rea DW, Provenzano E, Caldas C, Pharoah P, Hiller L, Earl H, Poole CJ (2010) Predictive markers of anthracycline benefit: a prospectively planned analysis of the UK National Epirubicin Adjuvant Trial (NEAT/BR9601). Lancet Oncol 11:266–274. https://doi.org/10.1016/S1470-2045(10)70006-1
Gaiser T, Ruschoff J, Moll R (2012) [In situ hybridization in clinical pathology. Significance of polysomy 17 for HER2 determination and genetic tumor heterogeneity in breast cancer]. Pathologe 33(Suppl 2):307–310. https://doi.org/10.1007/s00292-012-1663-z
Hofmann M, Stoss O, Gaiser T, Kneitz H, Heinmoller P, Gutjahr T, Kaufmann M, Henkel T, Ruschoff J (2008) Central HER2 IHC and FISH analysis in a trastuzumab (Herceptin) phase II monotherapy study: assessment of test sensitivity and impact of chromosome 17 polysomy. J Clin Pathol 61:89–94. https://doi.org/10.1136/jcp.2006.043562
Dent S, Verma S, Latreille J, Rayson D, Clemons M, Mackey J, Verma S, Lemieux J, Provencher L, Chia S, Wang B, Pritchard K (2009) The role of HER2-targeted therapies in women with HER2-overexpressing metastatic breast cancer. Curr Oncol 16:25–35
Ellis IO, Dowsett M, Bartlett J, Walker R, Cooke T, Gullick W, Gusterson B, Mallon E, Lee PB (2000) Recommendations for HER2 testing in the UK. J Clin Pathol 53:890–892
Furrer D, Jacob S, Caron C, Sanschagrin F, Provencher L, Diorio C (2017) Concordance of HER2 immunohistochemistry and fluorescence in situ hybridization using tissue microarray in breast cancer. Anticancer Res 37:3323–3329. https://doi.org/10.21873/anticanres.11701
Hyeon J, Cho SY, Hong ME, Kang SY, Do I, Im YH, Cho EY (2017) NanoString nCounter(R) approach in breast cancer: a comparative analysis with quantitative real-time polymerase chain reaction, in situ hybridization, and immunohistochemistry. J Breast Cancer 20:286–296. https://doi.org/10.4048/jbc.2017.20.3.286
Varga Z, Noske A, Ramach C, Padberg B, Moch H (2013) Assessment of HER2 status in breast cancer: overall positivity rate and accuracy by fluorescence in situ hybridization and immunohistochemistry in a single institution over 12 years: a quality control study. BMC Cancer 13:615. https://doi.org/10.1186/1471-2407-13-615
Jimenez RE, Wallis T, Tabasczka P, Visscher DW (2000) Determination of Her-2/Neu status in breast carcinoma: comparative analysis of immunohistochemistry and fluorescent in situ hybridization. Mod Pathol 13:37–45. https://doi.org/10.1038/modpathol.3880007
Hanna WM, Ruschoff J, Bilous M, Coudry RA, Dowsett M, Osamura RY, Penault-Llorca F, van de Vijver M, Viale G (2014) HER2 in situ hybridization in breast cancer: clinical implications of polysomy 17 and genetic heterogeneity. Mod Pathol 27:4–18. https://doi.org/10.1038/modpathol.2013.103
Marchio C, Lambros MB, Gugliotta P, Di Cantogno LV, Botta C, Pasini B, Tan DS, Mackay A, Fenwick K, Tamber N, Bussolati G, Ashworth A, Reis-Filho JS, Sapino A (2009) Does chromosome 17 centromere copy number predict polysomy in breast cancer? A fluorescence in situ hybridization and microarray-based CGH analysis. J Pathol 219:16–24. https://doi.org/10.1002/path.2574
Singh K, Tantravahi U, Lomme MM, Pasquariello T, Steinhoff M, Sung CJ (2016) Updated 2013 College of American Pathologists/American Society of Clinical Oncology (CAP/ASCO) guideline recommendations for human epidermal growth factor receptor 2 (HER2) fluorescent in situ hybridization (FISH) testing increase HER2 positive and HER2 equivocal breast cancer cases; retrospective study of HER2 FISH results of 836 invasive breast cancers. Breast Cancer Res Treat 157:405–411. https://doi.org/10.1007/s10549-016-3824-x
Vanden Bempt I, Van Loo P, Drijkoningen M, Neven P, Smeets A, Christiaens MR, Paridaens R, De Wolf-Peeters C (2008) Polysomy 17 in breast cancer: clinicopathologic significance and impact on HER-2 testing. J Clin Oncol 26:4869–4874. https://doi.org/10.1200/JCO.2007.13.4296
Persons DL, Tubbs RR, Cooley LD, Dewald GW, Dowling PK, Du E, Mascarello JT, Rao KW, Wilson KS, Wolff DJ, Habegger-Vance G (2006) HER-2 fluorescence in situ hybridization: results from the survey program of the College of American Pathologists. Arch Pathol Lab Med 130:325–331. https://doi.org/10.1043/1543-2165(2006)130%5B325:HFISHR%5D2.0.CO;2
Press MF, Sauter G, Buyse M, Fourmanoir H, Quinaux E, Tsao-Wei DD, Eiermann W, Robert N, Pienkowski T, Crown J, Martin M, Valero V, Mackey JR, Bee V, Ma Y, Villalobos I, Campeau A, Mirlacher M, Lindsay MA, Slamon DJ (2016) HER2 gene amplification testing by fluorescent in situ hybridization (FISH): comparison of the ASCO-College of american pathologists guidelines With FISH scores used for enrollment in breast cancer International Research Group Clinical Trials. J Clin Oncol 34:3518–3528. https://doi.org/10.1200/JCO.2016.66.6693
Koudelakova V, Trojanec R, Vrbkova J, Donevska S, Bouchalova K, Kolar Z, Varanasi L, Hajduch M (2016) Frequency of chromosome 17 polysomy in relation to CEP17 copy number in a large breast cancer cohort. Genes Chromosom Cancer 55(5):409–417. https://doi.org/10.1002/gcc.22337
Xu B, Shen J, Guo W, Zhao W, Zhuang Y, Wang L (2018) Impact of the 2018 ASCO/CAP HER2 guidelines update for HER2 testing by FISH in breast cancer. Pathol Res Pract. https://doi.org/10.1016/j.prp.2018.10.035
Acknowledgements
We thank Hui–Hui Jiang for outstanding technical support and Zhi Li for constructive comments on the manuscript.
Funding
This study is supported by a grant from National Clinical Key Subject Construction Project Fund of China, and grant from Guangdong Medical Science and Technology Research Fund (No. A2016086).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
All procedures performed involving human participants were in accordance with the ethical standards of the Institutional Review Board of Guangdong General Hospital, Guangdong Academy of Medical Sciences and with the 1964 Helsinki declaration and its later amendments.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Zhi-Hua Liu and Kun Wang have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Liu, ZH., Wang, K., Lin, DY. et al. Impact of the updated 2018 ASCO/CAP guidelines on HER2 FISH testing in invasive breast cancer: a retrospective study of HER2 fish results of 2233 cases. Breast Cancer Res Treat 175, 51–57 (2019). https://doi.org/10.1007/s10549-019-05148-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-019-05148-5