Abstract
Background
The role of G-protein-coupled estrogen receptor 1 (GPER-1) in the development of tamoxifen resistance in breast cancer is a highly controversial issue. The aim of this study was to determine the expression of GPER-1 in the clinical routine under conditions of endocrine treatment.
Patients and methods
GPER-1 expression was analyzed in 442 patients with primary invasive breast cancer. GPER-1 score of > 3 was determined as positive. Expression data were correlated with clinical and pathological characteristics and patient survival.
Results
GPER-1 expression was observed in 352 (80.9%) cases, and positively correlated with estrogen and progesterone receptor status (p = 0.0001). GPER-1 positivity was associated with an increased grade of differentiation (p = 0.0001) and with a low level of Ki-67 expression (p = 0.0001). High GPER-1 expression was associated with a decreased level upon systemic treatment (p = 0.011). In the whole cohort, GPER-1 expression was associated with prolonged disease-free survival (DFS). DFS between tamoxifen- and aromatase inhibitor-treated GPER-1-positive patients was similar (p = 0.090). Notably, after matching the analysis for the most important prognostic factors, DFS for tamoxifen-treated GPER-1-positive patients was 69.1%, which is a percentage that is significantly lower compared to DFS for GPER-1-positive patients treated with aromatase inhibitors (92.7%) (p = 0.005).
Conclusion
GPER-1 expression is a favorable prognostic factor in breast cancer patients. Its predictive role for poor benefit form tamoxifen treatment should be investigated in further studies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
G-protein-coupled estrogen receptor 1 (GPER-1), also referred to as GPR30, belongs to the family of G-protein-coupled receptors (GPCRs) and is involved in the rapid non-genomic action of estrogen [1]. In addition, tamoxifen possesses agonistic activity toward GPER-1, suggesting a potential role of this receptor in the development of tamoxifen resistance. Conducting a large retrospective study with breast cancer patients, we found that GPER-1 expression is an unfavorable prognostic factor regarding treatment response to tamoxifen [2]. In accordance, Sjöström et al. found that absence of plasma membrane expression of GPER-1 is associated with improved long-term prognosis of tamoxifen-treated patients [3]. In vitro data support the hypothesis that GPER-1 might be involved in tamoxifen resistance. In estrogen receptor-positive breast cancer cell lines, we observed increased expression of GPER-1 on the cell surface, and a cross-talk between GPER-1 and epidermal growth factor receptor 1 (EGFR-1). This may be responsible for the increased agonistic activity of tamoxifen [4]. In agreement, Mo and co-workers found that GPER-1 expression is facilitated by tamoxifen treatment, and that blockade of EGFR-1 and GPER-1 signaling pathways is an option to overcome tamoxifen resistance [5]. Nevertheless, the role of GPER-1 in tamoxifen resistance is still poorly understood, and further studies, especially prospective trials, are needed.
In this context, we investigated the expression of GPER-1 in breast cancer tissue and compared it with established clinicopathological parameters and clinical outcome.
Patients and methods
Immunohistochemistry
GPER-1 expression was analyzed as previously described [2]. Sections of formalin-fixed and paraffin-embedded breast cancer specimens or corresponding recurrent lesions (3.0 µm thick) were mounted on SuperFrost Plus glass slides (Menzel, Braunschweig, Germany) and dried overnight. A Benchmark XT (Ventana, Unterhaching, Germany) conducted the immunostaining. The slides were incubated with affinity-purified rabbit antibody against GPER-1 (SP4677P; Acris antibodies, Herford, Germany) diluted 1:500 for 32 min at 37 °C, after antigen retrieval with Protease I (Ventana) for 10 min. The reactions were visualized by DAB detection. The slides were counterstained with hematoxylin and cover slipped after being embedded in mounting medium.
GPER-1 expression analysis
GPER-1 expression was analyzed and expression score was obtained as previously described [6, 7]. Briefly, staining extensity was categorized as 0 (no positive cells), 1 (< 10% positive cells), 2 (10–50% positive cells), or 3 (> 50% positive cells), whereas staining intensity was categorized as 0 (negative), 1 (weak), 2 (moderate), or 3 (strong). The individual categories were multiplied to give a total immunohistochemical score (IHC). IHC score ranged between 0 and 9. The GPER-1 cut-off was determined using the GPER-1 expression in normal breast tissue surrounding the invasive breast cancer [6]. Tumors with an IHC of ≥ 3 were categorized as GPER-1 positive. GPER-1 expression was observed predominantly on the plasma membrane and in the cell cytoplasm cytosol (Fig. 1). GPER-1 expression in nucleus was not evaluated. Representative examples of different GPER-1 expression are shown in Fig. 1: negative staining (Fig. 1a), slightly positive staining (Fig. 1b), moderately positive (Fig. 1c) and strongly positive (Fig. 1d) cytoplasmic immunostaining. The specificity of the GPR30 peptide antibody was tested in cell lysates prepared from human embryonic kidney HEK-293 cells, human breast cancer MDA-MB231 and MCF-7 cells as described [2].
Patients and tissue samples
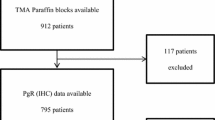
The data of 442 patients with primary invasive non-metastatic breast cancer, who had been admitted to the Department of Obstetrics and Gynecology, Otto-von-Guericke University, Magdeburg, Germany from 2014 to 2017, were selected by retrospective analysis. Since January 2014, we evaluated GPER-1 expression routinely after the primary diagnosis of breast cancer. In seven cases, GPER-1 expression was not determined, and they were excluded from further analysis (Fig. 2). Four hundred and thirty-five patients were eligible for analysis. Concerning the evaluation of survival in terms of endocrine treatment of GPER-1-positive patients, 184 patients were excluded (Fig. 2), including patients with negative GPER-1 expression (n = 83), in cases of not specified (n = 48) or not performed (n = 44) endocrine treatment, and in cases of bilateral breast cancer (n = 9). For survival analysis, 251 cases were available. These patients were divided into two groups depending on their endocrine treatment: tamoxifen or aromatase inhibitor.
To avoid a selection bias between both groups, a matching analysis was performed. The matching process was based on four prognostic criteria: tumor stage (≤ 2, or > 2 cm), tumor histology, tumor grading (grade 1, 2 or 3), and patient treatment. The matching procedure was conducted at random and without any information about the patients’ outcome. The rate of chemotherapy was performed in 41.7% and 34.3% in tamoxifen-treated and aromatase inhibitor-treated patients, respectively. This difference was not significant.
The main prognostic factors assessed in this study were patient age, expression of estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2), tumor size, tumor grading, histological type, and lymph node status. Histological grade, Ki-67 expression and ER, PR, and HER2 status were assessed at the time of diagnosis. The Ki-67 score was defined as percentage of positively stained tumor cells among the total number of malignant cells assessed.
The primary outcome was DFS defined as the time from the date of diagnosis and the date of loco-regional and/or distant relapse and/or cancer-specific death. Loco-regional relapse included the recurrence in ipsilateral breast, chest wall or regional lymph nodes. Distant recurrences consist of distant lymph node metastases (beyond the ipsilateral axillary, infra- and/or supraclavicular, internal mammary area), as well as of metastases in bone (including bone marrow), brain, liver, lung (including pleura and lymphangitic carcinomatosis), and other organs (including peritoneum, other organs not elsewhere classified, and skin tumors not affecting the breast and chest wall). The follow-up ended with the patient’s death, the latest available information, or the last follow-up on March 1st, 2018. The median follow-up was 33 months (range 1–64 months). The manuscript was prepared in accordance with the STROBE statement criteria [8]. According to the statement of Research and Ethical Committee, Otto-von-Guericke University, Magdeburg, Germany, informed consent was obtained from all individual participants included in the study before treatment.
Statistical analysis
The statistical calculations were performed using SPSS Version 22.0 (SPSS, Chicago, IL, USA). An association between GPER-1 expression and the tumor and patients’ variables was evaluated using the χ2 test or Fisher’s exact test. DFS survival was calculated using the Kaplan–Meier method. The equality of survival curves was tested by the log rank test. The statistical analyses were two-sided, and p values < 0.05 were considered statistically significant.
Results
GPER-1 expression and clinical and pathological characteristics
GPER-1 expression was analyzed in 435 patients with primary non-metastatic breast cancer. Patients were considered positive for GPER-1 if they had an IHC score of at least 3. GPER-1 expression was observed in 352 (80.9%) of 435 cases (Table 1). All of the surrounding non-tumor mammary tissues included in the tissue specimens were GPR30-positive. GPER-1 immunostaining correlated significantly with ER (p = 0.0001) and PR (p = 0.0001) status. No significant association was observed between GPER-1 and HER2 receptor status (Table 1). GPER-1 expression was inversely correlated to tumor grading. GPER-1 positivity correlated with low histologic grade of tumor (p = 0.0001), confirmed by the finding that GPER-1 expression was associated with a low level of Ki-67 expression. The mean level of Ki-67 expression was 38.3% (range 5–95%) and 24.2% (range 2–82%) for GPER-1 negative and positive cases, respectively. With regard to patient treatment, GPER-1 expression was associated with a decreased rate of systematic treatment (p = 0.011). There was no correlation between the expression of GPER-1 and other clinical and pathological characteristics (Table 1).
GPER-1 expression and patient outcome
For survival analysis, only patients with positive GPER-1, ER and/or PR expression and known endocrine therapy were included (Fig. 2). Two hundred and fifty-one patients were eligible. During the follow-up time, there were 40 (15.9%) breast cancer relapses and or cancer-specific deaths. Local recurrence, loco-regional recurrence, distant metastases, and death occurred in 16 (6.4%) cases, 3 (1.2%) cases, 12 (4.8%) cases, and 11 (4.4%) cases, respectively. In 2 cases, both local and loco-regional disease was observed. Bone was the site most frequently affected by distant metastases (n = 10), followed by liver (n = 4), lung (n = 3), other organs (n = 2), and distant lymph nodes (n = 1).
Furthermore, we evaluated the influence of endocrine treatment on patient survival. GPER-1 expression was associated with prolonged disease-free survival (data not shown). In the entire cohort of GPER-1-positive patients, the survival between tamoxifen and aromatase inhibitor-treated patients was similar (Fig. 3a). The survival for tamoxifen-treated and aromatase inhibitor-treated patients was 79.6% and 89.5%, respectively (p = 0.090). To avoid a selection bias between patients treated with tamoxifen and aromatase inhibitor, a matching analysis of the most important prognostic factors was performed. Notably, DFS was 69.1% for patients treated with tamoxifen, which is significantly lower than DFS for patients treated with aromatase inhibitors (92.7%). Thus, tamoxifen treatment was associated with a significantly poorer DFS compared to the individuals who received aromatase inhibitors in this group of GPER-1-positive patients (p = 0.005; Fig. 3b).
Kaplan–Meier analysis of DFS breast cancer patients. a DFS depends on treatment with tamoxifen or aromatase inhibitor in GPER-1-positive patients b DFS depends on treatment with tamoxifen or aromatase inhibitor after matching for tumor stage, tumor histology, tumor grading, and patient treatment in GPER-1-positive patients c and d DFS of patients treated with tamoxifen and aromatase inhibitor, respectively, depends of GPER-1 expression. The log rank test was used to calculate the p value. DFS disease-free survival, GPER-1 G-protein-coupled estrogen receptor 1
Discussion
In this study, we prospectively investigated the expression of GPER-1 and compared it with clinical and pathological characteristics, as well as with DFS in a cohort of 435 patients with invasive non-metastatic breast cancer. GPER-1 expression was associated with decreased DFS in patients treated with tamoxifen.
GPER-1 was expressed in 80.9% of the studied breast cancer specimens. This result is in accordance with our previously published data and with the results obtained by others [2, 3, 6, 9, 10]. Nevertheless, a direct comparison between these studies regarding GPER-1 expression is not easy because different evaluation methods and scores were used. It is clear that GPER-1 expression significantly correlates with ER and PR expression, as confirmed by us. In our cohort, both ER-positive tumors (87.2%) and PR-tumors (79.8%) expressed GPER-1. Regarding HER2 receptor expression, we did not observe any correlation, which is in contrast to our recent results [2], and the data reported by Sjöström et al. and Samartzis et al. [3, 10]. This discrepancy could be due to the different scores used or the way GPER-1 expression was analyzed. In the present study, GPER-1 expression was prospectively evaluated, whereas the evaluation was retrospective in all other studies. As conformation, Broselid et al. also reported that there exists a significant correlation between GPER-1 and HER2 expression [9]. Interestingly, the localization of GPER-1 expression, nuclear or cytoplasmic, correlated differently with clinical and pathological characteristics, including HER2 status, and should be considered [10].
Interestingly, most of the cited studies have clearly demonstrated that GPER-1 expression is associated with improved survival of patients with breast and other cancers [3, 6, 9, 11,12,13]. This finding is in line with ours, namely that GPER-1 is a potential tumor suppressor [12, 14, 15]. Similar data were reported by other studies investigating cancer cells of different origins [9, 13, 16, 17]. The fact that in our cohort, GPER-1 expression is associated with favorable pathological characteristics and improved outcome indirectly supports a tumor-suppressive action of GPER-1.
Of note, we found that GPER-1 expression was associated with a worse median DFS in the group of patients treated with tamoxifen, and these results are in agreement with previous investigations conducted in retrospective breast cancer cohorts [2, 3]. Tamoxifen is an agonist for GPER-1 [4, 5, 18, 19]. Activation of GPER-1 with tamoxifen is associated with increased expression of GPER on cell surface, leading to an increased cross-talk with growth factor receptors and resulting in increased cell proliferation [4, 5]. Continuous treatment with tamoxifen was associated with reduced DFS of 69.1% as compared with DFS of 92.7% in the group of patients treated with aromatase inhibitors. These results are consistent with our previous retrospective investigation [2]. The data of Sjöström et al. support our findings. They found that lack of GPER-1 expressions identified tamoxifen-treated patients with an extremely low risk of distant metastases in a long follow-up period [3]. By contrast, Broselid et al. found no correlation between tamoxifen treatment and GPER expression [9]. The discrepancy between these results could be due to several reasons. First, the retrospective nature of the cohort was investigated. Second, in the cohort of Broselid and co-workers, tamoxifen treatment continued for only 2 years. We know from in vitro data that tamoxifen resistance is associated with long-term treatment of tamoxifen [4, 5], and 2 years of treatment might be insufficient. Third, GPER-1 expression was evaluated using different scores, which resulted in different rates of GPER-1 expression. Furthermore, we found that GPER-1 expression is epigenetically regulated, and promoter methylation of GPER-1 is a prognostic factor [7]. These data were recently confirmed for colorectal cancer [13]. Thus, the influence of GPER-1 promoter methylation might be used as co-founder in the interpretation of survival outcome under treatment with tamoxifen. In this context, the value of GPER as predictor of reduced benefit of tamoxifen should be evaluated in prospective trials.
However, our study has some limitations because the subcellular localization of GPER-1 was not investigated. In this regard, the supposed distinct biological features of GPER-1, which depend on its localization [10], were not evaluated. Another limitation of our investigation lies in the fact that the patients treated with tamoxifen and aromatase inhibitor were not randomized. However, the matching analysis particularly decreased the selection bias. The strength of our study lies in its prospective character as far as GPER-1 expression analysis is concerned. The clinical data on hormonal treatment and survival were collected prospectively but analyzed retrospectively, which limits our results. A further strength is the validation of our GPER-1 score obtained in a retrospective study [2].
In conclusion, the expression of GPER-1 is strong in breast cancer tissue, and correlates with ER and PR expression. GPER-1 possesses tumor-suppressive characteristics; its expression is associated with improved patient outcome and reduced benefit of tamoxifen treatment. To prove this hypothesis, prospective randomized clinical trials are necessary.
References
Molina L, Figueroa CD, Bhoola KD, Ehrenfeld P (2017) GPER-1/GPR30 a novel estrogen receptor sited in the cell membrane: therapeutic coupling to breast cancer. Expert Opin Ther Targets 21:755–766
Ignatov A, Ignatov T, Weissenborn C et al (2011) G-protein-coupled estrogen receptor GPR30 and tamoxifen resistance in breast cancer. Breast Cancer Res Treat 128:457–466
Sjostrom M, Hartman L, Grabau D et al (2014) Lack of G protein-coupled estrogen receptor (GPER) in the plasma membrane is associated with excellent long-term prognosis in breast cancer. Breast Cancer Res Treat 145:61–71
Ignatov A, Ignatov T, Roessner A, Costa SD, Kalinski T (2010) Role of GPR30 in the mechanisms of tamoxifen resistance in breast cancer MCF-7 cells. Breast Cancer Res Treat 123:87–96
Mo Z, Liu M, Yang F et al (2013) GPR30 as an initiator of tamoxifen resistance in hormone-dependent breast cancer. Breast Cancer Res 15:R114
Ignatov T, Weissenborn C, Poehlmann A et al (2013) GPER-1 expression decreases during breast cancer tumorigenesis. Cancer Invest 31:309–315
Weissenborn C, Ignatov T, Nass N et al (2017) GPER promoter methylation controls GPER expression in breast cancer patients. Cancer Invest 35:100–107
von Elm E, Altman DG, Egger M et al (2007) The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet 370:1453–1457
Broselid S, Cheng B, Sjostrom M et al (2013) G protein-coupled estrogen receptor is apoptotic and correlates with increased distant disease-free survival of estrogen receptor-positive breast cancer patients. Clin Cancer Res 19:1681–1692
Samartzis EP, Noske A, Meisel A, Varga Z, Fink D, Imesch P (2014) The G protein-coupled estrogen receptor (GPER) is expressed in two different subcellular localizations reflecting distinct tumor properties in breast cancer. PLoS ONE 9:e83296
Ariazi EA, Brailoiu E, Yerrum S et al (2010) The G protein-coupled receptor GPR30 inhibits proliferation of estrogen receptor-positive breast cancer cells. Cancer Res 70:1184–1194
Ignatov T, Modl S, Thulig M et al (2013) GPER-1 acts as a tumor suppressor in ovarian cancer. J Ovarian Res 6:51
Liu Q, Chen Z, Jiang G et al (2017) Epigenetic down regulation of G protein-coupled estrogen receptor (GPER) functions as a tumor suppressor in colorectal cancer. Mol Cancer 16:87
Weissenborn C, Ignatov T, Ochel HJ et al (2014) GPER functions as a tumor suppressor in triple-negative breast cancer cells. J Cancer Res Clin Oncol 140:713–723
Weissenborn C, Ignatov T, Poehlmann A et al (2014) GPER functions as a tumor suppressor in MCF-7 and SK-BR-3 breast cancer cells. J Cancer Res Clin Oncol 140:663–671
Chan QK, Lam HM, Ng CF et al (2010) Activation of GPR30 inhibits the growth of prostate cancer cells through sustained activation of Erk1/2, c-jun/c-fos-dependent upregulation of p21, and induction of G(2) cell-cycle arrest. Cell Death Differ 17:1511–1523
Ribeiro MPC, Santos AE, Custodio JBA (2017) The activation of the G protein-coupled estrogen receptor (GPER) inhibits the proliferation of mouse melanoma K1735-M2 cells. Chem Biol Interact 277:176–184
Ignatov T, Eggemann H, Semczuk A et al (2010) Role of GPR30 in endometrial pathology after tamoxifen for breast cancer. Am J Obstet Gynecol 203:595 e9–e16
Vivacqua A, Bonofiglio D, Recchia AG et al (2006) The G protein-coupled receptor GPR30 mediates the proliferative effects induced by 17beta-estradiol and hydroxytamoxifen in endometrial cancer cells. Mol Endocrinol 20:631–646
Funding
This research did not receive any specific Grant from any funding agency in the public, commercial, or not-for-profit sector.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest.
Ethical standards
The experiments comply with the current laws of Germany and were performed according to the good clinical practice (GCP) guidelines.
Rights and permissions
About this article
Cite this article
Ignatov, T., Claus, M., Nass, N. et al. G-protein-coupled estrogen receptor GPER-1 expression in hormone receptor-positive breast cancer is associated with poor benefit of tamoxifen. Breast Cancer Res Treat 174, 121–127 (2019). https://doi.org/10.1007/s10549-018-5064-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-018-5064-8