Abstract
Purpose
The therapeutic effect of systemic treatment for breast cancer (BC) generally depends on its intrinsic subtypes. In addition, tumor infiltrating lymphocytes (TILs) are considered to be an independent factor for tumor shrinkage and disease prognosis. High TILs at baseline or after primary systemic chemotherapy are reported to be associated with better survival in triple-negative or human epithelial growth factor receptor 2 (HER2)-positive BCs. However, the prognostic value of TILs in estrogen receptor (ER)-positive and HER2-negative (ER+/HER2−) BC is still controversial.
Methods
We assessed TIL score (low, intermediate, and high) before and after primary systemic chemotherapy in every subtype of BC, and compared the clinical outcomes. Biopsy specimens of 47 triple-negative, 58 HER2+ and 91 ER+/HER2− BCs were used to assess TILs before treatment. To assess TILs after treatment, we examined residual invasive carcinoma in surgically resected samples of 28 triple-negative, 30 HER2+ and 80 ER+/HER2− BCs.
Results
A high TIL score in triple-negative BC before treatment resulted in a significantly higher proportion of pathological complete response (pCR). In contrast, ER+/HER2− BC exhibited fewer instances of pCR than other subtypes. Although not statistically significant, ER+/HER2− cases with a high TIL score also tended to achieve pCR (p = 0.088). Moreover, we revealed that low TIL BCs after chemotherapy, but not at baseline, had significantly better relapse-free survival in ER+/HER2− BC (p = 0.034).
Conclusion
Pathological examination of TILs after treatment may be a surrogate marker for prognosis in ER+/HER2− BC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Breast cancer (BC) is the most common malignant neoplasm in women worldwide. In the United States, approximately 230,000 new cases of BC occurred in 2015. In the same year, more than 40,000 women died as a result of BC, making it the leading cause of cancer death in females in the US [1]. Among Japanese females, BC is also the most frequently diagnosed cancer and the fifth highest cause of cancer death [2].
Strategies for treating BC depend on the intrinsic subtypes [3, 4]. In clinical practice, the intrinsic subtypes are determined using immunohistochemical staining for estrogen receptor (ER), progesterone receptor (PgR), human epithelial growth factor receptor 2 (HER2), and Ki-67 as surrogate markers for the classification originally defined by a microarray assay [5]. ER-positive and HER2-negative breast cancers (ER +/HER2− BCs) are usually treated by surgical resection followed by hormonal therapies. On the other hand, trastuzumab-based systemic chemotherapy is used in the treatment of HER2-positive breast cancers (HER2 + BCs). No specific targeted therapy exists for the treatment of triple-negative breast cancers (TNBCs), i.e., BCs without ER, PgR and HER2 expression, and this subtype has the worst prognosis when compared to others [6]. Thus, the evidence indicates that the therapeutic effect and prognosis could be influenced by the intrinsic subtype of the BC. However, the role of host immunity against cancer cells should be considered when evaluating the anti-tumor effect in all BC subtypes [7].
The relationship between cancer cell elimination during immune surveillance and escape from the host immune system during immunoediting has been studied in various malignant neoplasms, including melanoma and non-small cell lung cancer [8,9,10,11]. In BC, tumor infiltrating lymphocytes (TILs) are considered to be representative of the host immune response against cancer cells. Abundant TILs are usually reported to be associated with good prognosis [12]. Higher TILs are favorably associated with both relapse-free survival (RFS) and overall survival (OS) among TNBC and HER2 + BC in the post-surgical chemotherapy setting [13,14,15].
Recently, TILs were investigated not only as a prognostic factor, but also as a predictive factor for chemotherapy. Sensitivity for chemotherapy in early BCs is usually judged by complete disappearance of cancer cells defined as pathological complete response (pCR) with or without lymph node involvement in preoperative chemotherapy, because pCR is closely associated with longer RFS and OS [16]. A higher probability of pCR is predicted by increased TILs in the setting of primary systemic chemotherapy [17, 18]. Thus, high TILs now serve as a new surrogate marker to predict a higher rate of pCR resulting in better prognosis. Nevertheless, in ER +/HER2- BC the prognostic value of TILs has remained controversial. Previous reports [19,20,21] revealed that ER+/HER2− BC with abundant TILs were inclined to achieve pCR after primary systemic chemotherapy, but these results were not statistically verified. The role of abundant TILs after primary systemic chemotherapy on surgically removed samples was reported to predict better prognosis [22]; however, to our knowledge, an association between TILs after chemotherapy and prognosis for ER+/HER2− BC has never been reported.
In this study, we assessed TILs in all BC subtypes before and after primary systemic chemotherapy, and analyzed the clinicopathological features and prognosis. We aimed to clarify the predictive and prognostic value of TILs in BC as well as to identify different uses of TILs in each subtype.
Methods
Eligibility of patients
We retrospectively retrieved 197 invasive BCs treated with primary systemic chemotherapy followed by surgical resection at Hyogo College of Medicine Hospital, Nishinomiya, Hyogo, Japan. Consecutive cases between July 2008 and October 2016 were included. The study was approved by the ethics committee of the Hyogo College of Medicine (approval number #1886). Regimens were administered as follows: preoperative anthracycline-containing regimen to 9 patients; taxane-based to 46 patients; sequential use of anthracycline-containing and taxane-based to 127 patients; and unspecified regimens to 15 patients. All patients were female and 10 patients had distant metastases at diagnosis (Stage IV). HER2+ BC patients received chemotherapy, including anti-HER2 agents. We categorized these cases into three subtypes: TNBCs, HER2+ BCs, and ER+/HER2− BCs, according to the pathological reports of the biopsy specimens obtained before systemic therapy. Forty-seven cases were classified as TNBCs, 58 cases as HER2+ BCs, and 91 cases as ER+/HER2− BCs. One case was not categorized because HER2 status was unavailable in the records of the other institution. The nuclear grade and therapeutic effect of systemic chemotherapy were determined according to the guidelines of the Japanese Breast Cancer Society [23]. pCR was defined as the absence of residual invasive carcinoma in the breast of surgically resected specimens, regardless of residual intraductal carcinoma or lymph node metastasis. In previous studies, pCR was defined as the absence of invasive carcinoma in both the breast and lymph nodes [16, 17, 24] or in the breast regardless of lymph node metastasis [19, 23]. In order to analyze correlations between TILs and pCR in the breast, we used the definition of pCR regardless of lymph node metastasis.
Immunohistochemical procedure
Immunohistochemical staining for ER, HER2 and Ki-67 was performed on formalin-fixed, paraffin-embedded tumor tissues obtained before and after chemotherapy. Anti-ER antibody (clone 6F11) was purchased from Leica Biosystems (Wetzler, Germany) and anti-Ki-67 antibody (clone MIB1) from Dako (Glostrup, Denmark). The Hercep Test (Dako) or the BOND Oracle HER2 IHC System (Leica Biosystems) was used for HER2 staining. For ER and Ki-67, the percentages of positive cancer cells in nuclei were counted. The cut-off value for ER positivity was set at 1%. HER2 status was evaluated according to the ASCO/CAP HER2 Test Guideline Recommendations [25]. Briefly, BCs were defined as HER2 positive when immunohistochemical score 3 + or when 2 + with amplification of the HER2 gene by fluorescence in situ hybridization yielded.
Assessment of tumor infiltrating lymphocytes
We evaluated TILs on 197 biopsy specimens (pre-TILs) and 139 surgical specimens with residual invasive carcinoma (post-TILs) according to previous reports [26, 27]. Five cases containing minimum residual carcinoma were excluded. Briefly, a representative slide containing relatively high amounts of both invasive cancer and lymphocytic infiltration were selected. We then screened the slide using a low-power field (×40), and defined a small region with the highest lymphocytic infiltration within the tumor (Supplementary Fig. 1a). On this hot spot, TILs were assessed in a medium-power field (×100) (Supplementary Fig. 1b–d). Neutrophils, eosinophils, and macrophages were disregarded, and lymphocytes and plasma cells within the tumor border were counted as TILs, as instructed by the International TILs Working Group [28]. We estimated the proportion of the area infiltrated by lymphocytes to the area of the entire tumor plus adjacent stroma, and classified the TIL scores as low (< 10%), intermediate (10–50%), or high (≥ 50%). Two pathologists (TW and AIH) assessed the pre- and post-TIL scores without knowledge of the other’s score or the patient’s information. When the scores were discordant, the slides were reviewed and discussed until a final score was agreed upon.
Statistical analysis
We compared the pre- and post-TIL scores with various clinicopathological parameters or comparision between pre-TILs and pCR was studied by using Chi square or Fisher’s exact test. Univariate analysis and multivariate logistic regression of various clinicopathological factors for pCR were performed. RFS was indicated using the Kaplan–Meier method and analyzed by log-rank tests. Univariate and multivariate analyses of the clinical factors and TIL scores for RFS and OS were performed using a Cox proportional-hazards model to obtain the hazard ratio (HR) and 95% confidence interval (95% CI). Differences were considered statistically significant when p values were less than 0.05. We used the JMP Pro 10 software (SAS Institute Inc., Cary, NC, USA) for all statistical analyses.
Results
Relationship between tumor infiltrating lymphocytes and clinicopathological factors
Table 1 shows the relationship between TIL scores and clinicopathological factors. Node-negative BCs showed significantly higher pre-TILs (p = 0.021). Low-grade BCs had significantly lower pre-TILs (p < 0.001) and post-TILs (p = 0.041). ER+ BCs showed significantly lower pre-TILs (p < 0.001) and post-TILs (p < 0.001). When comparing subtypes, TNBCs had a significantly higher pre-TIL score (p < 0.001), and ER+/HER2− BCs significantly lower pre-TIL and post-TIL scores (p < 0.001, p = 0.004). BCs with a higher Ki-67 labeling index (≥ 20%) exhibited significantly higher pre-TILs (p < 0.001). There were no significant differences in scores of pre- and post-TILs between other clinicopathological factors.
Relationship between tumor infiltrating lymphocytes and pathological complete response
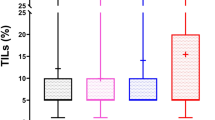
We examined the relationship between pre-TIL score and pCR rate in each BC subtype (Fig. 1). In total, 53 cases achieved pCR among 197 BCs (pCR rate: 26.9%). Nineteen cases achieved pCR in 47 TNBCs (pCR rate: 40.4%), 26 cases in 58 HER2 + BCs (pCR rate: 44.8%), and 8 cases in 91 ER +/HER2- BCs (pCR rate: 9.9%). A higher pre-TIL score was significantly associated with a higher pCR rate in the total number of BCs (Fig. 1a, p < 0.001) and in the TN subtype, (Fig. 1b, p = 0.002). There was no significant correlation between higher pre-TIL score and higher pCR rate (p = 0.74, Fig. 1c) in HER2+ BCs. The difference was also not significant in ER+/HER2− BC (p = 0.088); however, there was a positive correlation (Fig. 1d).
Association between tumor infiltrating lymphocytes before primary systemic chemotherapy (pre-TILs) and pathological complete response (pCR). Score of pre-TILs was evaluated as low, intermediate, and high in all breast cancer (BC) cases (a), triple-negative (TN) BCs (b), HER2+ BCs (c), and estrogen receptor (ER)+/HER2− BCs (d)
Univariate and multivariate analyses for pathological complete response
Table 2 shows univariate analysis and multivariate logistic regression for pCR. We selected pre-TILs (intermediate/low versus high), ER (negative versus positive), HER2 (negative versus positive), T stage (T1 vs. T2-T4), nuclear grade (1 vs. 2 and 3), and Ki-67 labeling index (< 20% vs. ≥ 20%) as objective variables and pCR as a predictor variable. All objective variables showed significant differences in univariate analysis. In contrast, multivariate analysis revealed that high pre-TILs (odds ratio [OR] 4.97; 95% CI 1.94–12.80; p < 0.001), positive ER (OR 0.32; 95% CI 0.13–0.77; p = 0.011), and positive HER2 (OR 5.90; 95% CI 2.33–14.90; p < 0.001) were independently associated with pCR.
Changes in numbers of tumor infiltrating lymphocytes between pre- and post-primary systemic chemotherapy
Among 139 BC cases without pCR, changes in pre- and post-TIL scores were examined (Fig. 2). In total, the proportion of BCs with a low TIL score increased after chemotherapy (49.6% to 64.7%, p = 0.006). In HER2+ BCs and ER+/HER2− BCs, the proportion of BCs with low TIL scores increased marginally (36.7–63.3% and 57.5–73.8%, respectively), but the difference was not statistically significant (HER2+ BCs, p = 0.12; ER+/HER2− BCs, p = 0.088). In TNBCs, there were no cases with high post-TILs and the proportion of cases with intermediate TIL scores increased significantly (39.3–60.7%, p = 0.03).
Changes in numbers of tumor infiltrating lymphocytes before primary systemic chemotherapy (pre-TILs) and after primary systemic chemotherapy (post-TILs) in BCs which did not achieve pCR after primary systemic chemotherapy. Pre-TILs were evaluated using the biopsy specimens for diagnosis of BCs. Post-TILs were evaluated using the representative slides of surgical specimens. Score of TILs was assessed as low, intermediate, and high in all breast cancer (BC) cases (a), triple-negative (TN)BCs (b), HER2+ BCs (c), and estrogen receptor (ER)+/HER2− BCs (d)
Prognostic value of pre- and post-tumor infiltrating lymphocytes
Among 187 cases (excluding ten cases of stage IV at the first diagnosis), RFS was analyzed according to pre-TIL scores (Fig. 3). In 22 cases, recurrence was observed and in 10 cases, the patients died during the 24.5 month median follow-up period (Interquartile range [IQR] 13.0–45.6 months). In total, pre-TILs score did not predict relapse (p = 0.41, Fig. 3a). There was no significant difference between pre-TIL scores and RFS in TNBCs and ER +/HER2- BCs. In contrast, in HER2+ BCs, pre-TILs were significantly associated with RFS (p = 0.048, Fig. 3c).
Relapse-free survival (RFS) of different scores of tumor infiltrating lymphocytes (TILs) before primary systemic chemotherapy (pre-TILs). Pre-TILs were evaluated using the biopsy specimens for diagnosis of BCs. Score of TILs was divided into three scales (low, intermediate, high) and evaluated in all breast cancer (BC) cases (a), triple-negative (TN)BCs (b), HER2+ BCs (c), and estrogen receptor (ER)+/HER2− BCs (d)
RFS was analyzed according to post-TIL scores in 129 cases that did not achieve pCR or were not minimum residual carcinoma after chemotherapy (Fig. 4). Recurrence was observed in 19 cases and in 10 cases the patients died during the 26.1 month median follow-up period (IQR 13.5–48.8 months). Post-TIL scores did not predict the risk of relapse in total (p = 0.23, Fig. 4a) and in HER2 + subtypes (p = 0.71, Fig. 4c). There were not enough TNBC cases to analyze as post-TIL because of the absence of high post-TIL score (Fig. 4b). In contrast, low post-TIL score was significantly associated with better RFS in ER +/HER2- BC cases (p = 0.034, Fig. 4d). The risk of relapse after 36 months was 0.95 in low post-TILs, 0.69 in intermediate post-TILs, and 0.75 in high post-TILs.
Relapse-free survival (RFS) of different scores of tumor infiltrating lymphocytes (TILs) after primary systemic chemotherapy (post-TILs) in patients with non-pCR and without minimum residual carcinoma after primary systemic chemotherapy. Post-TILs were evaluated using the representative slides of residual BCs after primary systemic chemotherapy in all breast cancer (BC) cases (a), triple-negative (TN) BCs (b), HER2+ BCs (c), and estrogen receptor (ER) +/HER2− BCs (d)
Univariate and multivariate analyses for RFS is shown in Supplementary Table 1. Tumor size and lymph node metastasis, but not pre- and post-TIL scores were significantly associated with RFS by univariate and multivariate analyses in all BCs. In ER +/HER2- BCs, only post-TIL scores (low vs intermediate and high) were a significant prognostic factor for RFS (HR 5.17, 95% CI 1.27–25.20, p = 0.023, Supplementary Table 3), further multivariate analysis was not performed. Univariate and multivariate analyses for OS is shown in Supplementary Table 2. Similarly to RFS, tumor size and lymph node metastasis, but not pre- and post-TIL scores were significantly associated with OS by univariate analysis in all BCs. Lymph node metastasis was an independent prognostic factor for OS by multivariate analysis. In ER +/HER2- BCs, lymph node metastasis but not post-TIL scores was a significant prognostic factor for OS (Supplementary Table 3), further multivariate analysis was not performed.
Discussion
In this study, we assessed TIL scores, before and after primary systemic chemotherapy in all subtypes of BC, and compared the clinical outcomes. We were specifically interested in the results of the ER +/HER2- subtype, because of the controversial role of TILs in predicting prognosis in this subtype. ER+/HER2− BCs showed a lower pCR rate than other types of BCs. At the same time, higher pre-TIL score in ER+/HER2− BCs was inclined to be associated with higher pCR rate; however, the result was not significant. In this subtype, RFS of low post-TIL BCs after chemotherapy, but not at baseline, was significantly better. This suggests that pathological examination of TILs after treatment can be used as a surrogate marker for prognosis in ER+/HER2− BCs (Fig. 5).
Schematic representation of the mechanisms of the tumor infiltrating lymphocytes (TILs) in relation to pathological complete response (pCR) and prognosis in breast cancer (BC) by focusing on estrogen receptor (ER)+/HER2− BC. Patients with high pre-TILs tended to achieve pCR that resulted in a favorable prognosis. In contrast, in patients without pCR, prognoses with high and low post-TILs seemed to be poor and favorable, respectively in ER+/HER2− BCs
We also showed that BCs with higher T stages (T2-T4) tended to have lower pre-TIL scores (p = 0.079), and that in BCs with lymph node metastases, the pre-TIL score was significantly lower (p = 0.021) (Table 1). These results suggest that TILs in primary BC sites had an inhibitory effect both on the progression of the BC itself, and on metastases, to regional lymph nodes. In contrast, no significant correlation could be established between BCs with distant metastases and pre-TIL scores (p = 0.85). Therefore, TILs in primary BC may not inhibit hematogenous spreading.
The sensitivity and specificity of high TIL score for predicting pCR were 43.4 and 88.2%, respectively, in all BCs. Although specificities were similarly high in both ER +/HER2- and TN (90.4% vs 78.6%), the sensitivity of ER+/HER2− (37.5%) was low when compared to TN (73.7%). Thus, the sensitivity of TIL score seemed to differ depending on subtypes. In previous reports [20, 29, 30], cases with a higher histological grade, negative ER expression, and higher Ki-67 labeling index were shown to have high pre-TILs, and our study confirms these results. TNBCs with high pre-TILs showed higher pCR rates (70%) than those with low and intermediate pre-TILs (15 and 21%, respectively). Similar results have been reported by other groups [13, 14, 17, 20]. The consistency of these results indicates that our method of TIL assessment is appropriate. Although HER2+ BCs with a high pre-TIL score were inclined to result in good pCR rates (67%), there was no statistical difference when compared to those with low and intermediate TIL scores. Higher pCR rates seen in HER2+ BCs with lower pre-TILs could be explained by trastuzumab-based treatment of HER2+ BCs. Trastuzumab in combination with chemotherapy exhibits strong cytotoxicity against cancer cells, or modulates an immunoreaction through antibody-dependent cell-mediated cytotoxicity instead of through TILs [31, 32]. In this study, pre-TIL scores in ER+/HER2− BCs were not statistically correlated to pCR rates (p = 0.088), which is consistent with results reported previously [19,20,21]. However, ER+/HER2− BC cases with high pre-TIL scores tended to achieve higher pCR rates than those with intermediate and low pre-TIL scores (27% versus 7% versus 6%). These results suggest that pre-TILs could be a surrogate marker to predict pCR, not only in TNBCs and HER2+ BCs, but also in ER +/HER2- BCs.
Our results indicated that TILs before primary systemic chemotherapy tended to decrease in HER2+ BCs and ER+/HER2− BCs. Similarly, in TNBCs, high TILs before chemotherapy (21.4%) decreased to 0, and intermediate TILs increased from 39.3 to 60.7% after chemotherapy. An increase of TILs after primary systemic chemotherapy in TNBCs was reported previously by Dieci et al. [22]. Dieci et al. [22] and Loi et al. [24] also reported that high levels of TILs after primary systemic chemotherapy in TNBCs significantly predicted improved RFS and OS. In contrast, similar to our results, Hamy et al. [33] reported decreased TILs after chemotherapy in 78% of HER2-positive breast cancers. The reason for this discrepancy in terms of TILs after chemotherapy remains unknown. Hamy et al. [33] reported that high levels of TILs after treatment, but not before treatment were significantly associated with adverse disease-free survival (DFS) by multivariate analysis. In addition, infiltration of immune cells after chemotherapy in residual tumor was associated with a worse DFS [34]. Our results are consistent with the latter study that identified an association between high TILs and poor prognosis, despite including all BC subtypes. Interestingly, levels of TILs after chemotherapy were higher in residual cancers which display aggressive patterns such as high residual cancer burden, high mitotic index, and high tumor cellularity [33]. Haricharan et al. reported that ER+ tumors with high somatic mutation load were significantly associated with poor OS [35]. Since immune reactions are likely to occur in tumors with high mutation load, poor prognosis of tumors with high TILs may be explained by this high mutation load. Based on these reports, it was speculated that the high incidence of TILs after chemotherapy was linked to poor prognosis due to the unfavorable characteristics of the tumor in the ER +/HER2- BCs. This association between TIL and prognosis may also depend on breast cancer subtypes. Montagna et al. [30] recently reported that higher TILs in inoperable ER+/HER2− BCs with metastasis treated with metronomic chemotherapy predicted significantly poorer progression-free survival (p = 0.011). They also verified that those inoperable ER+/HER2− BCs with higher TILs were significantly associated with higher Ki-67 labeling index (p = 0.03). These results also suggest that TILs after primary systemic chemotherapy is indicative of host immunity against cancer cells as well as the proliferative nature of the tumor. Since there was no significant association between TILs before chemotherapy and RFS, the prognostic significance of TILs in the ER+/HER2− subset seems to be enhanced after chemotherapy. The role and prognostic value of post-TILs might differ in each subtype. More research is needed to clarify the mechanism of decrease of post-TILs in ER+/HER2− BCs with good prognosis.
Our study has some limitations. The number of cases evaluated was relatively small; therefore, results obtained by subgroup analyses may contain errors. The follow-up period was also relatively short. We did not investigate the subsets of TILs using immunohistochemistry for CD4, CD8, and FOXP3. Detailed analysis of these subsets of TILs including a larger number of samples with longer follow-up period is necessary to acquire further insight into the role and prognostic value of TILs.
In conclusion, we established that the pathological evaluation of TILs after primary systemic chemotherapy predicts prognosis of ER+/HER2− BC patients.
References
Siegel RL, Miller KD, Jemal A (2015) Cancer statistics, 2015. CA Cancer J Clin 65(1):5–29. https://doi.org/10.3322/caac.21254
Center for Cancer Control and Information Services, National Cancer Center, Japan. http://ganjoho.jp/en/public/statistics/short_pred.html. Accessed 18 Aug 2016
Perou CM, Sørlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA, Pollack JR, Ross DT, Johnsen H, Akslen LA et al (2000) Molecular portraits of human breast tumours. Nature 406(6797):747–752. https://doi.org/10.1038/35021093
Sørlie T, Perou CM, Tibshirani R, Aas T, Geisler S, Johnsen H, Hastie T, Eisen MB, van de Rijn M, Jeffrey SS et al (2001) Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA 98(19):10869–10874. https://doi.org/10.1073/pnas.191367098
Coates AS, Winer EP, Goldhirsch A, Gelber RD, Gnant M, Piccart-Gebhart M, Thürlimann B, Senn HJ, Members P: Tailoring therapies–improving the management of early breast cancer (2015) St Gallen International Expert Consensus on the primary therapy of early breast cancer 2015. Ann Oncol 26(8):1533–1546. https://doi.org/10.1093/annonc/mdv221
Carey LA, Dees EC, Sawyer L, Gatti L, Moore DT, Collichio F, Ollila DW, Sartor CI, Graham ML, Perou CM (2007) The triple negative paradox: primary tumor chemosensitivity of breast cancer subtypes. Clin Cancer Res 13(8):2329–2334. https://doi.org/10.1158/1078-0432.CCR-06-1109
Okazaki T, Chikuma S, Iwai Y, Fagarasan S, Honjo T (2013) A rheostat for immune responses: the unique properties of PD-1 and their advantages for clinical application. Nat Immunol 14(12):1212–1218. https://doi.org/10.1038/ni.2762
Dunn GP, Old LJ, Schreiber RD (2004) The three Es of cancer immunoediting. Annu Rev Immunol 22:329–360. https://doi.org/10.1146/annurev.immunol.22.012703.104803
Schreiber RD, Old LJ, Smyth MJ (2011) Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science 331(6024):1565–1570. https://doi.org/10.1126/science.1203486
Qin A, Coffey DG, Warren EH, Ramnath N (2016) Mechanisms of immune evasion and current status of checkpoint inhibitors in non-small cell lung cancer. Cancer Med 5(9):2567–2578. https://doi.org/10.1002/cam4.819
Hodi FS, O’Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, Gonzalez R, Robert C, Schadendorf D, Hassel JC et al (2010) Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med 363(8):711–723. https://doi.org/10.1056/NEJMoa1003466
Aaltomaa S, Lipponen P, Eskelinen M, Kosma VM, Marin S, Alhava E, Syrjänen K (1992) Lymphocyte infiltrates as a prognostic variable in female breast cancer. Eur J Cancer 28A(4–5):859–864. https://doi.org/10.1016/0959-8049(92)90134-N
Adams S, Gray RJ, Demaria S, Goldstein L, Perez EA, Shulman LN, Martino S, Wang M, Jones VE, Saphner TJ et al (2014) Prognostic value of tumor-infiltrating lymphocytes in triple-negative breast cancers from two phase III randomized adjuvant breast cancer trials: ECOG 2197 and ECOG 1199. J Clin Oncol 32(27):2959–2966. https://doi.org/10.1200/JCO.2013.55.0491
Loi S, Michiels S, Salgado R, Sirtaine N, Jose V, Fumagalli D, Kellokumpu-Lehtinen PL, Bono P, Kataja V, Desmedt C et al (2014) Tumor infiltrating lymphocytes are prognostic in triple negative breast cancer and predictive for trastuzumab benefit in early breast cancer: results from the FinHER trial. Ann Oncol 25(8):1544–1550. https://doi.org/10.1093/annonc/mdu112
Ibrahim EM, Al-Foheidi ME, Al-Mansour MM, Kazkaz GA (2014) The prognostic value of tumor-infiltrating lymphocytes in triple-negative breast cancer: a meta-analysis. Breast Cancer Res Treat 148(3):467–476. https://doi.org/10.1007/s10549-014-3185-2
Kaufmann M, Hortobagyi GN, Goldhirsch A, Scholl S, Makris A, Valagussa P, Blohmer JU, Eiermann W, Jackesz R, Jonat W et al (2006) Recommendations from an international expert panel on the use of neoadjuvant (primary) systemic treatment of operable breast cancer: an update. J Clin Oncol 24(12):1940–1949. https://doi.org/10.1200/JCO.2005.02.6187
Denkert C, Loibl S, Noske A, Roller M, Müller BM, Komor M, Budczies J, Darb-Esfahani S, Kronenwett R, Hanusch C et al (2010) Tumor-associated lymphocytes as an independent predictor of response to neoadjuvant chemotherapy in breast cancer. J Clin Oncol 28(1):105–113. https://doi.org/10.1200/JCO.2009.23.7370
West NR, Milne K, Truong PT, Macpherson N, Nelson BH, Watson PH (2011) Tumor-infiltrating lymphocytes predict response to anthracycline-based chemotherapy in estrogen receptor-negative breast cancer. Breast Cancer Res 13(6):R126. https://doi.org/10.1186/bcr3072
Yamaguchi R, Tanaka M, Yano A, Tse GM, Yamaguchi M, Koura K, Kanomata N, Kawaguchi A, Akiba J, Naito Y et al (2012) Tumor-infiltrating lymphocytes are important pathologic predictors for neoadjuvant chemotherapy in patients with breast cancer. Hum Pathol 43(10):1688–1694. https://doi.org/10.1016/j.humpath.2011.12.013
Ono M, Tsuda H, Shimizu C, Yamamoto S, Shibata T, Yamamoto H, Hirata T, Yonemori K, Ando M, Tamura K et al (2012) Tumor-infiltrating lymphocytes are correlated with response to neoadjuvant chemotherapy in triple-negative breast cancer. Breast Cancer Res Treat 132(3):793–805. https://doi.org/10.1007/s10549-011-1554-7
Mao Y, Qu Q, Zhang Y, Liu J, Chen X, Shen K (2014) The value of tumor infiltrating lymphocytes (TILs) for predicting response to neoadjuvant chemotherapy in breast cancer: a systematic review and meta-analysis. PLoS ONE 9(12):e115103. https://doi.org/10.1371/journal.pone.0115103
Dieci MV, Criscitiello C, Goubar A, Viale G, Conte P, Guarneri V, Ficarra G, Mathieu MC, Delaloge S, Curigliano G et al (2014) Prognostic value of tumor-infiltrating lymphocytes on residual disease after primary chemotherapy for triple-negative breast cancer: a retrospective multicenter study. Ann Oncol 25(3):611–618. https://doi.org/10.1093/annonc/mdt556
Japanese Breast Cancer Society (2012) General rules for clinical and pathological recording of breast cancer, 17th edn. Kanehara & Co., Ltd, Tokyo
Loi S, Dushyanthen S, Beavis PA, Salgado R, Denkert C, Savas P et al (2016) RAS/MAPK activation is associated with reduced tumor-infiltrating lymphocytes in triple-negative breast cancer: therapeutic cooperation between MEK and PD-1/PD-L1 immune checkpoint inhibitors. Clin Cancer Res 22(6):1499–1509. https://doi.org/10.1158/1078-0432.CCR-15-1125
Wolff AC, Hammond ME, Hicks DG, Dowsett M, McShane LM, Allison KH, Allred DC, Bartlett JM, Bilous M, Fitzgibbons P et al (2013) Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. J Clin Oncol 31(31):3997–4013. https://doi.org/10.1200/JCO.2013.50.9984
Hida AI, Sagara Y, Yotsumoto D, Kanemitsu S, Kawano J, Baba S, Rai Y, Oshiro Y, Aogi K, Ohi Y (2016) Prognostic and predictive impacts of tumor-infiltrating lymphocytes differ between Triple-negative and HER2-positive breast cancers treated with standard systemic therapies. Breast Cancer Res Treat 158(1):1–9. https://doi.org/10.1007/s10549-016-3848-2
Hida AI, Ohi Y (2015) Evaluation of tumor-infiltrating lymphocytes in breast cancer; proposal of a simpler method. Ann Oncol 26(11):2351. https://doi.org/10.1093/annonc/mdv363
Salgado R, Denkert C, Demaria S, Sirtaine N, Klauschen F, Pruneri G, Wienert S, Van den Eynden G, Baehner FL, Penault-Llorca F et al (2015) The evaluation of tumor-infiltrating lymphocytes (TILs) in breast cancer: recommendations by an International TILs Working Group 2014. Ann Oncol 26(2):259–271. https://doi.org/10.1093/annonc/mdu450
Huszno J, Nożyńska EZ, Lange D, Kołosza Z, Nowara E (2017) The association of tumor lymphocyte infiltration with clinicopathological factors and survival in breast cancer. Pol J Pathol 68(1):26–32. https://doi.org/10.5114/pjp.2017.67612
Montagna E, Vingiani A, Maisonneuve P, Cancello G, Contaldo F, Pruneri G, Colleoni M (2017) Unfavorable prognostic role of tumor-infiltrating lymphocytes in hormone-receptor positive, HER2 negative metastatic breast cancer treated with metronomic chemotherapy. Breast 34:83–88. https://doi.org/10.1016/j.breast.2017.05.009
Park S, Jiang Z, Mortenson ED, Deng L, Radkevich-Brown O, Yang X, Sattar H, Wang Y, Brown NK, Greene M et al (2010) The therapeutic effect of anti-HER2/neu antibody depends on both innate and adaptive immunity. Cancer Cell 18(2):160–170. https://doi.org/10.1016/j.ccr.2010.06.014
Bianchini G, Gianni L (2014) The immune system and response to HER2-targeted treatment in breast cancer. Lancet Oncol 15(2):e58–e68. https://doi.org/10.1016/S1470-2045(13)70477-7
Hamy AS, Pierga JY, Sabaila A, Laas E, Bonsang-Kitzis H, Laurent C, Vincent-Salomon A, Cottu P, Lerebours F, Rouzier R, Lae M, Reyal F (2017) Stromal lymphocyte infiltration after neoadjuvant chemotherapy is associated with aggressive residual disease and lower disease-free survival in HER2-positive breast cancer. Ann Oncol 28(9):2233–2240. https://doi.org/10.1093/annonc/mdx309
García-Martínez E, Gil GL, Benito AC, González-Billalabeitia E, Conesa MA, García García T, García-Garre E, Vicente V, de la Peña FA (2017) Tumor-infiltrating immune cell profiles and their change after neoadjuvant chemotherapy predict response and prognosis of breast cancer. Breast Cancer Res 16(6):488. https://doi.org/10.1186/s13058-014-0488-5
Haricharan S, Bainbridge MN, Scheet P, Brown PH (2014) Somatic mutation load of estrogen receptor-positive breast tumors predicts overall survival: an analysis of genome sequence data. Breast Cancer Res Treat 146(1):211–220. https://doi.org/10.1007/s10549-014-2991-x
Acknowledgements
We would like to thank Editage (www.editage.jp) for English language editing. This study was supported by a Grant-in-Aid from the Ministry of Education, Culture, Sports, Science, and Technology of Japan (No. 15K10077).
Funding
We do not have a financial relationship with the organization that sponsored the research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study was approved by the ethics committee of the Hyogo College of Medicine (approval number #1886).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Watanabe, T., Hida, A.I., Inoue, N. et al. Abundant tumor infiltrating lymphocytes after primary systemic chemotherapy predicts poor prognosis in estrogen receptor-positive/HER2-negative breast cancers. Breast Cancer Res Treat 168, 135–145 (2018). https://doi.org/10.1007/s10549-017-4575-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-017-4575-z