Abstract
Constructive Neutral Evolution (CNE) is an evolutionary mechanism that can explain much molecular inter-dependence and organismal complexity without assuming positive selection favoring such dependency or complexity, either directly or as a byproduct of adaptation. It differs from but complements other non-selective explanations for complexity, such as genetic drift and the Zero Force Evolutionary Law, by being ratchet-like in character. With CNE, purifying selection maintains dependencies or complexities that were neutrally evolved. Preliminary treatments use it to explain specific genetic and molecular structures or processes, such as retained gene duplications, the spliceosome, and RNA editing. Here we aim to expand the scope of such explanation beyond the molecular level, integrating CNE with Multi-Level Selection theory, and arguing that several popular higher-level selection scenarios are in fact instances of CNE. Suitably contextualized, CNE occurs at any level in the biological hierarchy at which natural selection as normally construed occurs. As examples, we focus on modularity in protein–protein interaction networks or “interactomes,” the origin of eukaryotic cells and the evolution of co-dependence in microbial communities—a variant of the “Black Queen Hypothesis” which we call the “Gray Queen Hypothesis”.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
[A]lthough complexity in biology is generally regarded as evidence of “fine tuning” or “sophistication,” large biological conglomerates might be better interpreted as the consequences of runaway bureaucracy—as biological parallels of nonsensically complex Rube Goldberg machines that are over-engineered to perform a single task. – Gray et al. (2010, 921)
Explanations for the origin and maintenance of complexFootnote 1 traits usually rely on attributing to such traits a “finely tuned,” or “sophisticated” character. Such explanations comprise adaptationist stories about how successive beneficial mutations were selected for their positive effect on fitness, usually at the level of organisms. Of course, constraints are admitted. For instance, pleiotropic interactions limit the evolution of many genes, complex structures can emerge as fortuitous byproducts (spandrels) of adaptive ones (Gould and Lewontin 1979), and seemingly adaptive structures can exhibit beneficial effects (exaptations) other than those for which they arose (Gould and Vrba 1982). For complex (e.g. multi-component) structures and processes, the default belief is that the above well-understood constraints are the only factors to be excluded before adaptation is the favored explanation.
At the molecular level, the neutrality of many small changes in sequences of the individual genes that affect complex features is now widely accepted. As in Kimura’s Neutral Theory of Molecular Evolution (1983), a mutation with no effect on protein sequence (synonymous), and nonsynonymous mutations with equal effects on fitness can spread through a population without being differentially affected by selection. Neutral processes can also generate complexity, and three answers as to how have been offered. The first and most general is the “Zero-Force Evolutionary Law” (ZFEL) of McShea and Brandon (2010). The mechanism here is simple: replication and reproduction are not error-free and when variants generated by them are effectively neutral, they accumulate. Variance is equivalent to complexity and diversity, both within and between individuals and species, McShea and Brandon (2010) argue.
Secondly, effective neutrality is related to population size, as Michael Lynch and colleagues have reminded us (Lynch and Conery 2000; Lynch and Force 2000). Many of the more byzantine aspects of animal and plant genomes might be seen as the result of fixation by drift of slightly deleterious extra DNA, more likely to occur in comparatively small populations, in which selection is a less reliable force. Contrast multicellular eukaryotes with prokaryotes (Bacteria and Archaea), which are thought to have much larger population sizes and characteristically boast stripped-down genomes, taken as paragons of efficiency.
A third neutral creative process, which we highlight here (and define by examples in the next section), was termed ‘Constructive Neutral Evolution’ (CNE) by Stoltzfus (Stoltzfus 1999; see also Covello and Gray 1993). Receiving renewed attention from molecular and cell biologists (Omer et al. 2017; Cohen and Steger 2017), CNE denotes the effectively irreversible retention of molecular interactions that initially arise neutrally, and differs from both ZFEL and the population-size-dependent drift envisioned by Lynch in its irreversibility. Selection is involved, but only against the loss of complexity, not for its origin.
Here we aim to expand CNE as an explanatory process, uniting it with Multi-Level Selection (MLS) theory. MLS theory is also an alternative to adaptationism, especially when organismal-level fitness is the focus of the latter. Traits that appear neutral or even slightly deleterious at any level N might, according to MLS theory, be products of selection (adaptations) at level N + 1 or N −1, and thus byproducts at N. At lower and higher levels than organism-centric biology usually attends to, the debates about selfish and junk-DNA (Orgel and Crick 1980; Doolittle and Sapienza 1980), about sexual reproduction (Michod 1997) and more recently about species and group selection (Jablonski 2008; Brunet and Doolittle 2015) fall within the purview of MLS. Both selection (with all its constraints) and drift can occur at all levels of the biological hierarchy. Our purpose here is to explain how CNE too might contribute to complexification at higher levels than that of molecular machinery, to which it has so far been restricted.
As with neutral models generally, the clear value of CNE is in generating null hypotheses to be contrasted with adaptation-based hypotheses that are often presumed or taken as the default in evolutionary biology. Explanations employing CNE as a null are attractive on three grounds: (1) they are often parsimonious, not requiring the assumption of many sequential adaptive steps and benefits, (2) they shift the burden of proof onto the adaptationism that pervades evolutionary and especially genomic biology (Gould and Lewontin 1979; Doolittle 2013; Koonin 2016), and (3) they can explain directional or biased changes in complexity without assuming directional or teleological biases in the nature or frequency of occurrence of mutations. When conjoined with MLS, these attractions are increased by their further applicability at higher levels of organization. Our goal here is to show that evolutionary scenarios proposed for important processes at supra-individual levels are instances of CNE.
CNE exemplified by three examples at the molecular level
Gene duplication (Fig. 1a)
The simplest sort of case,Footnote 2 as described by Stoltzfus (1999) and by Lynch and Conery (2000) may be that involving duplication of a protein-coding gene whose product carries out a cellular function at a level of activity L, a minimum required to maintain fitness (Fig. 1a). Such duplications might often be neutral (a doubling of L being harmless). Imagine, next, that each duplicate suffers a mutation resulting in its producing less than L-level functionality, a likely scenario since most mutations reduce activity. If the total activity from both genes must be at least L, neither gene can now be fully inactivated or deleted. Two genes are required (are maintained by purifying selection) where one was once enough.
Three generalized examples of Constructive Neutral Evolution (CNE). a Gene duplication and subfunctionalization; b pre-suppression via fortuitous prior interaction (stabilization); c RNA-editing (illustrated by the arguably simplest form in which specific C’s in an initial transcript (pre-mRNA) are converted to U’s by an editing machinery. See text for details
This process of degenerating mutation, leading to either ‘dosage compensation’ (Hughes et al. 2007) where duplicates suffer reduced activity or expression or ‘subfunctionalization’ where each duplicate takes on a subset of the original functions of the gene, can be sharply contrasted with a selection-based scenario wherein partitioning or “dividing labour” between a number of genes results in an increase in fitness. In the CNE case the organism with duplicated genes is not more fit.Footnote 3
Intermolecular interaction (Fig. 1b)
Next, consider CNE via the fortuitous interactions of two molecular components, A and B. Imagine that A has cellular function or activity, FA, while B has either no function or one, FB, that is irrelevant to its ability to interact with A. Fortuitous interactions of greater or lesser duration are inevitable and observable in cells, typically crowdedFootnote 4 with proteins, RNA molecules and metabolites (Omer et al. 2017; Golding and Cox 2006; Yang et al. 2012). Although experimental methods may give inflated values for “false positives” (components that interact but have not been selected to do so), this must sometimes occur (Launay et al. 2017), and is likely even more prevalent for protein RNA-interactions (Majumder et al. 2016).
Some of these interactions will also inevitably have a stabilizing effect on one or another partner. For instance, B might stabilize A so that mutations that would otherwise weaken A’s structure and reduce its ability to perform FA (indicated by black dots in Fig. 1a) are now permitted—are effectively neutral. Any component(s) having a stabilizing effect on A is to that extent a potential B in such a scenario. Since FB is assumed to have nothing to do with FA until the point when B fortuitously suppresses mutations in A, suppression is an exaptation (Gould and Vrba 1982). If several structure-weakening mutations in A occur, its return to wild-type independence from B is unlikely, just as is the return to initial conditions of simplicity in the “drunkard’s walk” metaphor for evolving complexity and diversity overall employed by Gould in Wonderful Life (1989). The presence of B, which need have no other “function,” comes under stabilizing selection to maintain FA, although positive selection on B for the performance of FA was never involved.
As a source of indiscriminately stabilizing proteins, the molecular chaperone system is an especially clear target for a CNE explanation. Chaperone proteins bind to, or encapsulate, unfolded or yet-to-be folded peptides to facilitate the folding process. While their mechanisms are diverse, the ultimate effect is clear: proteins that would not have folded properly without a chaperone may fold correctly with them. There are advantages to having such a system in place, but CNE can here operate in tandem with positive selection. Once the chaperone system exists, certain amino acid sites are free to mutate to a state that would otherwise have resulted in a misfolded and thus non-functional protein: misfolding mutations are pre-suppressed (Rutherford 2003). Further missense mutations causing misfolding, and thus dependence on chaperones, are surely more likely than mutations returning to self-sufficient folding. A ratchet drives towards a chaperone-dependent proteome. Moreover, CNE was likely involved in the evolution of the structure of chaperonins themselves (Archibald et al. 1999, 2000).
A and B might thus be two proteins, but some of the more obvious examples involve a protein molecule and an RNA molecule. Some RNA molecules have catalytic activities: generally these depend on intramolecular base-pairing stabilizing a specific three-dimensional RNA structure. But some base pairs might be dispensed with by mutation if a fortuitously binding protein also stabilizes the RNA, as must sometimes happen. With enough such potentially destabilizing mutations, the dependence of the RNA’s activity on the protein would be hard to reverse (a ratchet). Initially postulated as such an example, the evolved dependence of a once “self-splicing” catalytic intron RNA on a fortuitously binding protein seems now well documented as CNE (Fig. 1b). As Lamech et al. (2014) write,
Our results suggest a scenario of constructive neutral (i.e. pre-adaptive) evolution in which an initial non-specific interaction between the CTD [C-terminal domain] of an ancestral fungal mtTyrRS [the protein] and a self-splicing group I intron [the RNA] was “fixed” by an intron RNA mutation that resulted in protein-dependent splicing. Once fixed, this interaction could be elaborated by further adaptive mutations in both the catalytic domain and CTD that enabled specific binding of group I introns. [ibid, 4]
As noted here, CNE and adaptive (positive) selection may often go hand-in-hand. A neutrally-arising interaction maintained by purifying selection might be further “improved” (increasing organismal fitness) through subsequent alterations, while selected-for interactions might be more irrevocably “locked in” by CNE. Indeed, CNE may produce essentialized molecular interactions that enable subsequent rounds of positive selection, yet caution should be taken not to see the interactions produced by CNE as teleologically explained by their future selective potential. Many multi-subunit complexes such as the ribosome and the messenger RNA splicing apparatus (spliceosome) of eukaryotes would likely have such a mixed heritage: among many features now maintained by purifying selection (against loss) only some would owe their initial existence to positive selection. For the spliceosome, a complex machine with five interacting RNAs and scores of associated proteins, such a scenario is especially appealing (Gray et al. 2010).
Lamech et al. (2014) describe CNE as “pre-adaptive,” a notion some might find tainted by teleology (Gould and Vrba 1982), and would replace with “exaptive”. But these and the term “pre-suppression” are indeed appropriate here. Too often, we contend, scenarios like that described by Lamech et al. (2014) are presented in reverse (Simpson et al. 2000; Bernstein et al. 1984; Martin and Koonin 2006). First, it is postulated, a modestly maladaptive mutation occurs in the A gene, and is fixed by drift (or as a spandrel). Then a second (suppressor) mutation in the B gene that restores fitness is selected for positively, improving fitness vis-à-vis the mutated state. Many “error-correcting” processes are supposed to have arisen in this way (see Grosche et al. (2012) for such a view of chloroplast RNA editing). But pre-suppression (as opposed to “error-corrective post-suppression”) seems at least equally likely: the suppressive activity already exists and no disadvantaged intermediate waiting for salvation by suppression needs to be imagined. The suppressive activity of B—as noted above—might well have been a by-product (spandrel) of some previously unrelated activity FB that is selected for. The B gene becomes a suppressor and is adaptive as such only in retrospect, after mutations in the A gene creating dependence on it have occurred (Fig. 2).
Typical error-correction evolutionary scenario (a) and its CNE alternative (b). Many evolutionary stories, for instance describing the origin of RNA editing (Simpson et al. 2000), the eukaryotic nucleus (Martin and Koonin 2006) or eukaryotic sexuality (Bernstein et al. 1984) assume that error-correcting machinery was selected for, that is, positively selected from an error-prone and thus disadvantaged background (Hartwell et al. 1999). CNE imagines that error-correction, at least in a nascent form, pre-exists, permits error accumulation, and subsequently becomes “locked in” by purifying selection
Messenger RNA editing (Fig. 1c)
In most well-studied systems protein-coding sequences are transcribed base-for-base into a corresponding sequence in a messenger RNA, and this is translated according to the triplet (three-base) code into a linear sequence of amino acids in a protein. Some genes, however, produce “precursor” messenger RNAs that must be “edited”—some nucleotides added, deleted, or changed—into “mature” messenger RNAs. Only these can be translated into functional proteins.Footnote 5 Editing may involve hundreds of sites and/or dozens of genes (Simpson et al. 2000; Grosche et al. 2012).
Editing systems are not all the same mechanistically, and in some it may well be that the initial activity arose to correct for a previously occurring mutational accident, though our concern about disadvantaged intermediates remains. But the spread of edited sites seems unarguably the result of CNE: because an editing process already exists, mutations that require its existence will not be selected against and will accumulate (Fig. 1c). The more such mutations, the more turns of the irreversibility ratchet. The gain of editing-requiring sites during trypanosome evolution is indeed well documented by comparative studies (Simpson et al. 2000).
Uniting these three examples at the molecular level are the following features:
-
1.
Presuppression Otherwise fitness-reducing changes in gene arrangement or sequence are rendered innocuous because a pre-existing condition (gene duplication, interacting components or processes) fortuitously corrects for (genetically suppresses) them, this condition being “pre-suppressive”.
-
2.
Ratcheting Such dependencies will increase whenever further mutations reparable by the same “pre-suppressor” are possible. That is, whenever there are more potential dependency-increasing than dependency-decreasing mutations in component A, a ratchet analogous to the “Drunkard’s Walk” to complexity cited by Gould (1989) is in play.
-
3.
Biased increase in complexity It is pre-suppression and its ratchet-like character that distinguishes CNE from other neutral processes. Often CNE will increase complexity insofar as a single function comes to require more component structures or processes. But it is dependency, not complexity increase that CNE necessarily entails. The evolved integration of mitochondrial and nuclear genomes is in our view quintessential CNE, but the initial state (endosymbiosis) was arguably more complex in terms of the number of participating components than the end-product (the eukaryotic cell).
-
4.
Raw material The initial pre-suppressive interactions may have been long-standing or evolutionarily transient. Even transient interactions can be “locked in” by the ratchet.
-
5.
Mixed history Many complex systems will involve inter- (and intra-) component interactions first created by CNE and then advanced by “positive” selection, and vice versa, both now maintained by “purifying selection”. It will be in any specific case, and surely for organismal complexity overall (however this term is construed), very difficult to sort out which process has dominated.
While CNE provides a useful framework in theories of molecular evolution, as an explanatory strategy it is applicable to any evolving system where pre-suppression and ratcheting can occur, and thus to any level of a traditional biological hierarchy, like those generally discussed in Multi-Level Selection theory (Okasha 2005). In the next section we are concerned with identifying processes that irreversibly increase the complexity of protein–protein interaction networks (PPIs) or interactomes (networks of all components) generally, without any significant change to the fitness of organisms. We argue that the “interactome” is a higher level of organization than the individual molecular machines (traditional targets of CNE explanations) that make it up. In other words, we are concerned with the emergence of Rube Goldberg-like molecular systems independently of any “engineering to perform a single task.”
…consideration of protein–protein interactions exposes a vast array of intricate cellular processes to a new way of thinking about how they might have originated as a multitude of drunkards walking through complexity space. –Lukeš et al. (2011, 535)
In “Explaining transitions between levels: constructive neutral eukaryogenesis” section we address the role of CNE in eukaryogenesis, as a specific example of a “major transition in evolution”. In “Neutral evolution across levels: the Gray Queen Hypothesis” section we show how CNE could drive the emergence of complex interdependencies in microbial communities. Beyond the extent to which neutral models can explain species distributions (Bell 2001), we see a role for CNE in explaining metabolic integration between community members in a manner similar to the selection-driven Black Queen Hypothesis (BQH) first formulated by Morris et al. (2012).Footnote 6
A level above macromolecules: the modularity of PPIs, “interactomes” and toxin-antitoxin evolution
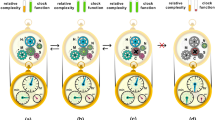
While there is general agreement that the biological world is hierarchically organized and indeed MLS theory requires some such organization, there is persistent disagreement about exactly how levels are to be delimited, and whether each really constitutes a “level of selection.” It is important in any case to distinguish levels of what might be called biological organization from levels of selection. A common hierarchy of selection is as follows: gene, genome, cell/organism, group, species. Each level contains entities showing heritable variation in fitness, and entities at higher levels contain lower-level entities as their parts. Nonetheless, an organizational discontinuity exists between the first two levels and higher ones: while genomes are mereological sums of genes (broadly defined), cells are sums of much more than just their genomes (see Varzi 2016). The “macromolecular conglomerates” and their multitude of interactions (the “interactome”) of which cells are mereological sums are absent from popular hierarchies. At precisely this level, biological organization and levels of selection come apart.Footnote 7 This problematic situation is depicted in our Fig. 3.
Hierarchies of selection and organization. Eliminating the organizational discontinuity in the commonly proposed hierarchy of selection (leftmost column) would require replacing the level below cell with the things that it is a mereological sum (Σ) of, i.e. macromolecular machines. Unfortunately macromolecular machines and interactomes are not reproducers, and thus do not serve the same role as units of selection as other levels of the hierarchy. If we accept such discontinuity, we can still make room for the interactome as the collection spanning from macromolecular conglomerates to cells, but then the interactome serves the same role as cell, contrary to common usage
A variety of terms are used to describe cellular constituents: ‘module’, ‘part’ (McShea and Venit 2001), and ‘organelle’ (perhaps including less technically delimited collective terms like ‘system’, ‘conglomerate” and ‘interactome’). All seem to converge on some criteria for comparing internal to external connections. Likewise, each implies a level of organization and co-ordination, and thus a target for evolutionary explanations, above that of genes and occasionally even above physically integrated molecular machines, such as the ribosome or splicing apparatus. It seems that the choice of terms depends on the research program at hand: molecular and cell biologists talk of organelles, philosophers prefer parts, and bioinformaticians and modelers use the term module, yet explaining how such entities emerge is of central interest to all. We argue that complex modularity (whether conceived as “parts” or “organelles”) can arise within PPIs or interactomes (not restricted to proteins) independently of both functional specificity and positive selection. A generalized theory of CNE shows how.
Modularity in networks has been given a precise definition (Newman 2006), although for our purposes it will suffice to define modularity informally as non-randomness in the distribution of intra- and inter-module connections favoring internal connections. A collection of nodes (proteins, RNAs, etc.) in a biological network will be modular to the extent that there are a greater number of connections between those nodes than expected by chance. Connections are determined in a variety of ways: physical interactions between proteins, co-expression and co-occurrence of cognate genes or epistatic interactions between them, for instance (Cafarelli et al. 2017). However a network model of the proteome (or “interactome”) is constructed, modules will be an object of interest in the project of understanding cellular complexity, and thereby deserving of explanation.
The complexity of parts that perform specific functions and exhibit modularity in PPI networks may, of course, often be correctly explained by positive selection. Through selection, proteins involved in specific biochemical processes can be separated from superfluous connections with irrelevant partners (Launay et al. 2017) and coaxed towards beneficial interactions, while systems lacking essential interactions are purified away. However, it would be excessively adaptationist to propose that positive selection is responsible for modularity wherever it might be observed. Similarly, it is unreasonable to assume that all modular portions of PPIs are functionally specific. Indeed, Wang and Zhang (2007) conclude that,
…nonrandom patterns are often interpreted as having functional significance and having been particularly favored by natural selection. While this may be true in many cases, a nonrandom network structure can also originate as a byproduct of other processes without having its own function. [ibid, 1019], emphasis added
Several recent studies bear directly on this possibility.Footnote 8 Atay and Skotheim (2014), emphasizing stochastic cell-to-cell variations in gene expression, argue that “most gene expression stochasticity and pathway interconnectivity is nonfunctional,” while Landry et al. (2013) stress the evolutionarily ephemeral nature of many PPIs and “view PPI data as they exist today as capturing a snapshot of evolutionary wiring and rewiring of a PPI network in which much of the information may be superfluous to the contemporary function of an organism.” Most recently, Sorrels and Johnson (2015, 715) argue that many common features of transcriptional regulatory networks (including feed-forward loops, redundancy of regulators, cooperative binding and network size), “particularly those that seem overly complex and counter-intuitive, can be understood as relatively crude products of high-probability evolutionary trajectories rather than as highly optimized, specific solutions.”
Although some fraction of modularity surely owes its presence to selection, the question is “how much?” and “when can we reject neutrality and randomness?” Zuckerkandl (1992) would have called neutral systems interactions “polite,” and Stoltzfus (1999) writes,
Given inevitable purifying selection, any novel attribute that arises in this manner is likely to be coordinated, rather than in conflict, with its biological milieu: it will be an “aptation” in the sense of Gould and Vrba (1982), and “polite” in the sense of Zuckerkandl (1992). – Stoltzfus (1999, 6), emphasis added
Computational modeling and experiments both suggest that neutral modularity can arise merely from duplication and divergence (Wang and Zhang 2007), although any divergence will presumably also need to be sufficiently apt and polite. This is because duplicates initially form the same connections as their parent and can later diverge and contribute to modularity by subfunctionalization (Lynch and Force 2000). Thus the functional specificity of a module and the adaptiveness of its degree of modularity are independent notions, i.e. a module can be functionally specific and modular, yet not have its modularity explained by positive selection. As many authors have argued, a module might accomplish a quite specific function, yet its degree of modularity might be best explained by neutral processes such as CNE (e.g. splicing, the ribosome) (Covello and Gray 1993; Stoltzfus 1999; Gray et al. 2010; Lukeš et al. 2011; Fernandez and Lynch 2011; Finnigan et al. 2012). Moreover, a function could be diffuse (Lukeš et al. 2011)—it could be accomplished by the joint action of independent proteins.
CNE might also generate modularity through potentially damaging interactions that it prevents, that is, coupling co-dependency with something like toxicity. Instead of the new dependency of positively-selected function FA on the presence of component B, it could be that their interaction is required to prevent some negative consequence of A acting alone. That is to say, the A:B pair might be acting as toxin and antitoxin. As Omer et al. (2017, 16) point out,
…purifying selection may be due to removing harmful mutations from the population, that is, the gene and its encoded protein become harmful after a mutation occurred, possibly because the mutated protein interferes with the translation machinery, or because of toxicity of the misfolded protein. Finding a signature of purifying selection should not automatically be considered proof of the gene’s selectable function. –Omer et al. (2017, 11), emphasis added
There are many toxin-antitoxin systems now known, especially for prokaryotes where genetic analysis is easiest (van Melderen 2010). When encoded on plasmid genomes, toxin-antitoxin systems are often called “addiction modules.” One gene encodes a product that, expressed alone, kills the plasmid-containing cell through interference with some vital process (hence is a toxin). The other encodes an antitoxin that prevents this, but is less stable, or less effective at low concentration. If the encoding plasmid is lost, both gene products are diluted out by cell division. Residual toxin kills such cells when concentrations of antitoxin fall below necessary preventative levels. Thus cells free of plasmids, even though they might otherwise be fitter, are punished by death for their infidelity: they are addicted to the plasmid.
Surprisingly, though, many addiction modules (pairs of genes) are found on chromosomes, which no cell would want to lose. One model for how toxin-antitoxin system unrelated to plasmid maintenance might arise is shown in Fig. 4. Components A, B and C are initially independently functional and A and B fortuitously (neutrally) interact in such a way that A can become dependent on B. In so doing (or subsequently), it (accidentally) acquires toxicity, in particular by negatively affecting the activity of C (or of cellular activities generally, as with many improperly folded proteins (Hartl 2017)). In our earlier examples it was the fitness-maintaining function of gene duplicates, A:B complexes or editing-like processes that were overseen by purifying selection, while in this case that function might even have been lost and still the complex is maintained as an “addictive” cellular component.
Abstract model of CNE where function of AB complexes are not under direct purifying selection. (Top) In pre-suppression stage, B (transiently or constantly) binds to or interacts with A, and possesses an excess capacity to detoxify mutations arising in A during the suppression stage. Finally, the accumulation of C-inactivating mutations (toxicity) in A makes the interaction with B irreversible in the ratcheting stage. (Bottom) Initially, C is active and may or may not interact with AB complex. During suppression stage A obtains a mutation that would inactivate C, if B were absent or dysfunctional. In this variant of CNE, the functional specificity of A:B complexes is not the trait under purifying selection, the activity of C is. An example would be the accumulation of mutations that would preclude proper folding of proteins without the assistance of pre-existing chaperone proteins. Improperly folded proteins are toxic to many cellular activities, represented here by C (Hartl 2017)
Such a scenario may seem farfetched if A, B and C are each proteins with separately evolved functions, but again chaperonins provide an example. By facilitating folding of a protein which in an unfolded state would hinder cellular activity, chaperonins are antitoxins. Moreover, many toxin-antitoxin systems involve noncoding RNAs that can interact with each other more-or-less transiently through more-or-less accurately-matched base pairs. Much of eukaryotic gene regulation is now thought to involve interactions between proteins and such RNAs, and between RNAs themselves, sometimes as complementary and inhibitory “antisense” to specific messenger RNAs. In a recent review, Kapusta and Feschotte (2014, 450) write that “the rapid turnover of lncRNA [long non-coding RNA] repertoires raises fascinating questions with regard to their significance in speciation, adaptation, and trait variation between and within species.” It could be that speciation and adaptive evolution are as often the result of neutrally “toxic” interactions (as in Fig. 4) that establish new gene regulatory regimes as by direct selection for the creation of such networks.
So CNE has a more general scope of applicability than just functionally specific “large biological conglomerates,” but might apply generally across biochemical networks. Indeed, given the sensitivity of many biochemical interactions, it would be unsurprising if many modularity-increasing interactions served only to prevent one binding partner from disturbing—“toxifying”—a number of other processes.
To conceptually disentangle modularity from functional specificity, organelles (as paradigm candidates for macromolecular evolutionary explanations) might be best conceived as biological modules of the highest degree. This view of organelles is in keeping with McShea and Venit’s conception of a biological part generally, “a part is a system that is both integrated internally and isolated from its surrounds” (McShea and Venit 2001). Notably, this view differs from both standard individuations of organelles by function (see Mendoza 2013) and from the energetic perspective offered by Frey-Wyssling (1978, 547), “a definition of the organelle must include the capacity of energy transfer of the structure under consideration.” While functional specificity and energy intake might be paradigmatic characteristics of organelles, they are neither necessary nor sufficient for modularity. With a McShea and Venit (2001) style view in mind, we can then note that modularity is the result of selective processes and neutral ratchets. Investigations should thus address the extent to which the complex structure of organelles is functionally specific, including in its energetic demands, instead of presuming specificity.
Explaining transitions between levels: constructive neutral eukaryogenesis
The evolutionary transitions in individuality that accompany Eörs Szathmáry’s and John Maynard Smith’s “Major Transitions in Evolution” (1995) are often understandable as resulting from CNE: the loss of lower-level independence following aggregation (neutral or selected for other reasons). Several independent acquisitions of multicellularity and a whole spectrum of increasingly interdependent endosymbiotic bacteria-eukaryotic host pairs are available for study (Keeling et al. 2015). Positive selection for the aggregate might explain some cases of aggregation, but purifying selection against disaggregation of selectively neutrally ratcheted aggregates could be of equal importance. Maynard Smith and Szathmáry recognize this as “contingent irreversibility,” writing early in their 1995 book that,
If an entity has replicated as part of a larger whole for a long time, it may have lost the capacity for independent replication it once had, for accidental reasons that have little to do with the selective forces that led to the evolution of the higher-level entity in the first place. –Smith and Szathmáry (1995, 9)
The most-discussed example of such a transition is eukaryogenesis, and in particular the evolved interdependence of the nucleus and the mitochondrion, widely believed to have descended from an endosymbiotic bacterium. We focus on that here. The loss of mitochondrial genes and/or their transfer to the nucleus do result in complete interdependence and have traditionally been seen as the consequence of the operation of neutral ratchets (Muller’s and another described in Doolittle 1998). It is not clear that these alone make eukaryotic cells more complex than their precursor,Footnote 9 if that is taken to be one autonomous cell maintained as endosymbiont within another (Booth and Doolittle 2015). But the typical eukaryotic cell does have more genes and more complex regulatory mechanisms than the typical prokaryotic cell, and this may have been enabled by some broadly-effective pre-suppression mechanism.
Stoltzfus’ (1999) expansion of the concept of CNE describes the initial conditions for the formation of irreversible dependencies as follows,
[C]omplex and intricate traits arise, not by the classical model of beneficial refinements, but instead by a repetition of neutral steps. The fundamental sequence of events is that a novel attribute appears initially as an excess capacity and later becomes a contributor to fitness, due to a neutral change at some other locus that creates a dependency on it. –Stoltzfus (1999, 6), emphasis added
The presuppression of mutations (Lukeš et al. 2011) is a kind of excess capacity at the genetic level. Other examples, as described above, include the stabilization of self-splicing introns, the duplication of a gene with currently sufficient expression levels or functional profiles (Bridgham et al. 2009; Finnigan et al. 2012), or the capacity to edit precursor RNAs so as to compensate for mutations in the genes encoding them (Covello and Gray 1993; Grosche et al. 2012). All can be construed as pre-existing “excess capacities,” subsequently used. But recent debates about eukaryogenesis, and specifically the importance of the acquisition of mitochondria (Lane and Martin 2010, 2015; Booth and Doolittle 2015; Lynch and Marinov 2017) have raised the possibility of a different (albeit less specific) kind of excess: the availability of high-energy intermediates.
Lane and Martin (2010) claim that …
Prokaryotic genome size is constrained by bioenergetics. The endosymbiosis that gave rise to mitochondria restructured the distribution of DNA in relation to bioenergetic membranes, permitting a remarkable 200,000-fold expansion in the number of genes expressed. This vast leap in genomic capacity was strictly dependent on mitochondrial power, and prerequisite to eukaryote complexity: the key innovation en route to multicellular life. [ibid, 929]
This notion is so far empirically unverified, and the more traditional alternative—that the evolution of phagocytosis is what made eukaryotes complex and that mitochondria were acquired later—remains very much alive (Embley and Williams 2015). But we take Lane and Martin’s conjecture as an imaginative invocation of CNE, breaking it down as follows. (1) Pre-suppression: the acquisition of the mitochondrion created an excess capacity: the availability of energy. (2) Suppression: this excess allowed gene duplications and protein expression to occur at a higher rate, “permitting eukaryotes to explore protein space” (Lane and Martin 2015, E4823) without the previously deleterious cost of excess expression. (3) Ratcheting: this neutral walk though protein space subsequently resulted in a variety of irreversible genomic changes, such as essentialization of new processes, or subfunctionalization of duplicates with high energetic costs of expression (Lynch and Force 2000), thus creating a dependency on the mitochondrion and putting its loss (or just reductions in energy output) at a selective disadvantage. The organelle’s origin (acquisition) might have been neutral or even mildly disadvantageous. A CNE reinterpretation of Lane and Martin (2010, 2015) allows us to keep the energetic exploration of protein space in the narrative for eukaryogenesis without needing to rely on sequential adaptations as the cause of increased complexity.
We only maintain that, if eukaryote radiation did indeed stem from an excess energetic capacity, constructive explanations can be framed under neutral or near-neutral conditions. This principle established, that the eukaryotic condition enjoys an excess energetic capacity is arguable in the first place. Lynch and Marinov (2017) recently argued that, after scaling for cell volume, eukaryotes may on average be no more energetically capable than prokaryotes, and may even be less capable. They offer a near-neutral and non-energetic account of eukaryogenesis that also fits happily into CNE,
…it is also possible that one member of the original consortium was a parasite rather than a benevolent partner (made plausible by the fact that many of the alpha-protobacteria to which mitochondria are most closely related are intracellular parasites). Despite its disadvantages, such a system could be rendered stable if one member of the pair (the primordial mitochondrion) experienced relocation of just a single self-essential gene to the other member’s genome, while the other lost a key function that was complemented by the presence of the endosymbiont. –Lynch and Marinov (2017, 9), emphasis added
Clearly this is a “process of subfunctionalization” [ibid, 10]. Moreover, the reciprocal loss of self-essential and other-complemented functions is all the more likely in the case of tight symbiosis between entire genomes than in the ordinary subfunctionalization of duplicate genes—there is more that could go wrong on either side when more parts must cooperate. Of course, the host could have lost or experienced reductions in energy capacity, but there are plenty of other functional losses that might have served to lock in the endosymbiotic aspects of the eukaryotic condition.
A third and independent CNE scenario for complexification accompanying eukaryogenesis has been offered by Fedoroff (2012) and colleagues (see also Kidwell 2002), who propose that the early evolution of effective epigenetic silencing mechanisms rendered the further expansion of transposable element (TE) families innocuous, or at least less damaging. Copies of TEs comprise the majority of many eukaryotic genomes, including our own (Brunet and Doolittle 2015). Although silencing mechanisms might initially have been positively selected for (to reduce the damage initially inflicted by TEs or viruses) they also permitted the subsequent enormous expansion in the number of such selfish elements: an example of CNE where regulation serves as the presuppressor.
While it is always tempting to draw distinctions between eukaryotes and prokaryotes that are more than phylogenetic, the availability of neutral explanations for eukaryotic complexity should make us skeptical of the adaptive significance of the complexity distinguishing eukaryotes from prokaryotes. One of us has elsewhere challenged this and other aspects of typical euakryote-triumphalist rhetoric (Booth and Doolittle 2015). Similarly, consider Sancar’s (2008) perspective on molecular clocks.
[I]n observing our understanding of the molecular mechanisms of circadian clocks of cyanobacteria and humans unfold over the last decade, one cannot help but notice the elegant simplicity of the former and the ever-increasing complexity of the latter, and ask why humans need such a Rube Goldbergesque design for a task that can be accomplished with a much simpler, more economical, and perhaps more reliable design. –Sancar (2008, 24)
The canonical retort would be to point out that eukaryotes may require more complex regulation since they have to regulate a more complex and differentiated cellular environment. But similarly to what Kidwell (2002) and Fedoroff (2012) have pointed out, pre-existing excess regulation might also be the cause or enabler of complexity and not a consequence. Moreover, circadian rhythms are hardly the only “Goldbergesque” difference revealed by comparative molecular studies, and we doubt an “argument from regulatory complexity” will hold true generally across eukaryotic complexity space.
Neutral evolution across levels: the Gray Queen Hypothesis
The fact that many microbes cannot be cultured in isolation and appear to function in Nature as multi-species communities is of growing interest to microbiologists and philosophers (Pande and Kost 2017; Clarke 2016; Doolittle and Booth 2017). Many microbes have lost key enzymes and pathways needed to survive on known growth media: they have become too simple to be self-sufficient. More such microbes, living in apparently cooperative communities, are being discovered as culture-independent community genomic analyses become cheaper and more widely applied (Brown et al. 2015). In addition to the problem of devising experimental procedures to culture such microbes, there is renewed interest within microbiology to explain how simplicity can evolve, and why it might be so common (Wolf and Koonin 2013; O’Malley et al. 2016).
Evolution by simplification often occurs in systems where some function of an organism has been externalized, e.g. where the production of a specific compound is outsourced to another organism in an ecological network. This kind of “reductive genomic evolution” can lead to dependencies, and has been embodied in the Black Queen Hypothesis (BQH) of Morris et al. (2012). In short, when many members of a population perform a function in a leaky way, some members can enjoy the benefits of abundance without possessing the requisite gene(s). “Cheaters” arise, and there is a selection-driven “race to the bottom” in genome size.
As Morris et al. (2012) put it …
Gene loss can provide a selective advantage by conserving an organism’s limiting resources, provided the gene’s function is dispensable. Many vital genetic functions are leaky, thereby unavoidably producing public goods that are available to the entire community. The BQH predicts that the loss of a costly, leaky function is selectively favored at the individual level and will proceed until the production of public goods is just sufficient to support the equilibrium community; at that point, the benefit of any further loss would be offset by the cost. [ibid, 1]
Instead of the Black Queen’s “race to the bottom” we propose a Gray Queen’s “drunkards walk towards community interdependence,” and a corresponding Gray Queen Hypothesis (GQH). What manifests as simplicity in the genomic architecture of individuals within a population might better be described as complex interdependency at the community level, neutrally-occurring gene losses being locked in by the relative irreversibility of such loss. Arguably, selected-for gene regain by lateral transfer does occasionally mitigate community collapse, restoring what Morris et al. (2012) call “equilibrium” when some particular public good is in too short supply, and may partially explain the apparent positive correlation between gene number and strength of selection (Koonin 2016). As public goods dwindle the selection coefficient for regain by transfer will inevitably rise and compensate for its rare occurrence.
Thus, while interdependencies can emerge from selection for genomic reduction, they can also emerge from neutral changes that are biased more towards interdependency than to self-sufficiency (i.e. biased towards self-insufficiency). A similar duality of interpretation can be seen in the literatures on genome reduction in endosymbionts, parasites or marine cyanobacteria (Giovannoni et al. 2014). At stake is whether genome reduction is selected for (as “streamlining”) or is simply not selected against (as in Muller’s ratchet).
Fullmer et al. (2015) entertain a “strong” BQH, going still further than Morris et al. (2012), to suggest that …
… we may need to more seriously consider populations as the operative units in which genes are selected in [sic] rather than exclusively individual organisms. Similar to how Richard Dawkins advocated thinking of an organism as a collection of generally agreeable, but selfish, genes perhaps we should be thinking of lineages and populations as the collection of genes, i.e. pan-genomes, rather than the individual cells. –Fullmer et al. (2015, 4)
Such “meta-organism” thinking is not easily reconciled with standard Darwinian models of evolution by natural selection, although modelers (e.g. Hagstrom and Levin 2017) are comfortable with it and one of us (Doolittle 2017; Doolittle and Booth 2017) has recently suggested recasting the network of interactions uniting a community, not the collection of individuals and species responsible for its maintenance, as the “unit of selection.” Clearly some level of community integration is required for such meta-organism and interaction-level accounts of selectable units, as well for more organism-centric issues of culturability.
In any case, when combined with a view of functions as having variable modularity, the GQH highlights some constraints on functional loss and leads to testable predictions about genome architecture. For example, Kuo et al. (2009) have shown that the degree of genome-wide drift (measured in terms of the ratio of non-synonymous to synonymous mutation rates) coupled to deletion bias are negatively correlated with genome size, supporting a “drift (rather than race) to the bottom” explanation of reduced bacterial genomes. While much of the correlation observed by Kuo et al. (2009) can be attributed to lower effective population size, some contribution is surely made by “relaxed selection.” Selection can reliably be relaxed by constant environments within host cells for endosymbionts or parasites, but also by community-level leaky functions. In the latter case, we would expect stronger signals of drift in, or total deletion of, genes whose function has been externalized.
A problem for both the selection-driven BQH and the neutralist GQH is that when functions are accomplished by a collective of genes with epistatic dependencies beyond their “primary” function, such genes might not be shed without significant, costly ramifications. This is one interpretation of the interesting results of Martiny et al. (2013). What they find is that the more genes required for the performance of a complex trait, the more it is phylogenetically conserved. Extremes would be, at the high end, oxygenic photosynthesis (found in all and only members of the phylum Cyanobacteria and requiring scores of genes) and, at the low end, sugar utilization or phosphate uptake (variable within species on a strain-by-strain basis and requiring one or a few genes). Indeed, that complex functions are more integrated functionally with the rest of a cell’s metabolism (are connected to many other modules) seems the only interpretation of such a result.
Complex functions can nonetheless be externalized under the right conditions. Once an organism has externalized one of its functions, large parts of the interaction network could be lost or irreparably damaged without significant cost to the organism just in case that function is specific to a sufficiently modular portion of the protein interaction network. Indeed, intra-module selection is more easily relaxed than inter-module selection. This is because most of the connections severed by the loss of gene products in such a module (organelle, part) will be to other gene products in the same module—function loss can be epistatically isolated. The function of these connections will already have been externalized (i.e. made non-essential) and thus mutations or deletions within modules made selectively neutral or nearly so. Indeed, once a module has become non-functional, further losses are neutral with respect to the present state. In short, deleterious epistatic ramifications and dependencies can be isolated within modules while fitness consequences are neutralized by externalization.
This mechanism places a CNE-style ratchet at the level of the ecological community, where existence of a leaky function presuppresses epistatically isolated mutations in the functional pathways of a community member. The “leaky functions” of Morris et al. (2012) become a community-level case of the “excess capacity” proposed by Stoltzfus (1999). Once pathway activity has been lost or significantly reduced, mutations leading to further disruption or deletion are more likely than function-restoring mutations, producing a neutral ratchet towards genomic non-functionality and further community-level interdependence.
The emergence of such community dependencies by the GQH generates modularity in ecological networks in much the same way that proteomic CNE generates modularity in PPIs: a community with highly interdependent members will have more dependencies internal to the community than external to it. The GQH is formally similar to the abstract model of CNE offered by Gray et al. (2010), Lukeš et al. (2011) and Doolittle et al. (2011), except functionally-redundant microbial “guilds” take the place of a class of proteins and cohabitation takes the role served by binding in the molecular case. Instead of a presuppressing protein, B is whatever member or guild in a community performs the leaky function. The presence of B allows member(s) A to accumulate mutations that, absent B, would be lethal or deleterious to A. Once A has lost or reduced its capacity to preform the function, the cohabitation of A and B becomes irreversible. A community-level toxin-antitoxin version of the GQH is equally viable: for guilds A, B, and C which are already mutually dependent or cohabitating, B might detoxify A to the benefit of C, allowing a cycle of increased toxicity for A to ratchet a tighter relationship with B.
One could test the extent to which this process explains cases of evolution of simplification by looking at the functional specificity of modules that performed the functions externalized and subsequently lost in organisms with genomes reduced by deletion. We would predict that organisms subject to a BQH-style CNE will have lost whole modules, not parts thereof. Alternatively, the above model would be supported if the PPI of a genomically reduced organism showed an overrepresentation of modules that lack functional specificity (indicating their resistance to being lost in a G/BQH manner). Finally, prime candidates for a GQH explanation would occur in populations too small to be subject to strong selection. Indeed, a GQH model of genome reduction can happily accommodate recent reports that the strength of selection is positively correlated with number of genes (Koonin 2016; Novichkov et al. 2009).
Conclusion
As an addition to the analogy of evolution as a “tinkerer” (Jacob 1977) who designs things by minor modifications to and re-purposing of pre-existing components, McShea and Brandon (2010) note that ZFEL is something like an “assistant” in the tinkerer’s workshop whose contributions tend to add complexity.
…the tinkerer is not alone. There is also an assistant present, an odd character whose main job seems to be to attach parts to the evolving machine, not just parts but novel part types. Occasionally, rarely, the assistant removes a part type, but mostly he adds them. Now most of the new part types that the assistant adds have no conceivable function, and the tinkerer rejects them, removing them as fast as he can. Sadly, the main effect of the assistant’s activity is to discombobulate the machine and distract the tinkerer…Luckily, some of the parts are useless but benign, and the tinkerer can ignore them, allowing them to remain in place. [ibid, 121]
Clearly a limited tinkerer cannot remove all the useless or even mildly detrimental additions of the assistant—selection is not always strong enough to overcome the ZFEL creep towards complexity. Yet the assistant is presented mostly as benign or ignorant, and the limitations on removing parts attributed just to the efficiency of the tinkerer. We add that it is possible for the random addition of parts (or interactions between parts) to create dependencies between previously independent and useless ones, such that the tinkerer is no longer free to tinker by removal. Furthermore, once a new dependency has been introduced, it can be reinforced by further additions to it, ratcheting up neutral dependencies through either further tinkering or (benign or mildly detrimental) “assistance.” The assistant is sometimes perverse.
While complexification without positive selection and the retention of complexity by purifying selection has been extolled elsewhere, it is rarely stated explicitly in CNE terms. Moreover, from the fundamental tenant of MLS, that selection can in principle apply at multiple levels, it is a short step to the conclusion that drift applies there too. Fitting CNE into MLS then demands recognizing that pre-suppression, or excess capacities, and ratcheting can obtain at levels above the molecular. Indeed, many “major transitions in evolution” can be recast in these terms.
Recognizing that many traits maintained by purifying selection did not arise through positive selection also challenges “selected effect” definitions of function. For instance, in his “final attempt at a definition of function” Godfrey-Smith (1994) requires that functional traits “were successful under selection in the recent past, through positively contributing to the fitness of systems…”. Our model of CNE involves no stage in the distant or recent past in which this could be said to be true. Complex features might, in part or in whole, have originated “accidentally” and have no or only causal role functions at relevant levels in the biological hierarchy. In the case of toxin-antitoxin-like systems (Fig. 4) purifying selection only addresses removal of a part but not the whole complex. Increasingly, instances in which components of toxin-antitoxin systems masquerade as essential are coming to light, as in the very recent report that the pha-1 locus, long thought important for pharynx development in nematodes, is proven to be essential only because it counteracts the embryo-killing activity of a maternal-effect toxin sup-35 (Ben-David et al. 2017). In such cases, members of the toxin-antitoxin pair are falsely ascribed functions based on the disease phenotype that results from their removal, when a double deletion of the pair shows that neither had this function—indeed, neither may even have been physiologically involved under normal conditions. We have no idea of how many cellular, organismal or community components or activities exist simply to counteract the baleful effects of others, nor how many of these owe their ameliorative role to CNE. Sophisticated genetic tests involving extensive phenotypic characterization of single, double and multiple deletions or synthetic biology reconstructions of simplified genomes (for instance Hutchison et al. 2016) seem a credible way to assess this.
CNE has thus far been confined to explanations much like those in “Introduction” section: creation of dependencies and subsequent evolutionary ratchets in molecular machines affecting cell viability. The full scope and utility of the CNE explanatory strategy is much wider than this. CNE explanations can be deployed in a variety of cases, explicitly or implicitly, both as counters to adaptationism and evolutionary hypotheses sui generis. In the former case, if the CNE null cannot be rejected as the origin of complexity, then so much worse for the (pan-) adaptationist just-so (Koonin 2016). In the latter, the scope of applicability of CNE style evolutionary mechanisms has widened significantly—from hypothesis, to mechanism, to framework. What first appeared as a clever solution to a chicken-and-egg problem in the origin of RNA editing (Covello and Gray 1993) can be extended into a general theory of the neutral emergence of proteome complexity (Stoltzfus 1999; Gray et al. 2010; Lukeš et al. 2011; Doolittle et al. 2011), and generalized into a function-independent and level-transcendent evolutionary framework.
Notes
Complexity is of course an onerous notion, highly dependent on context. Increases or decreases in its value might nevertheless be assessed—in terms of the interdependency of component parts or some product of the number of parts compared to part-types—with less ambiguity than assigning definite values to particular entities.
Indeed, Koonin (2016) describes subfunctionalization as the clearest cases of the “evolutionary modality” of CNE.
Gene duplication is also, of course, a source of “new genes” as when one duplicate acquires a novel function (“neofunctionalization”; Lynch and Conery 2000; Lynch and Force 2000) or when each of the duplicate pairs retains a subset of the functions of a their multi-functional common ancestor. Such “subfunctionalization” (Lynch and Conery 2000) comprises CNE as we define it below (pre-suppression followed by ratcheting).
In the case of bacterial cells, Golding and Cox (2006) report that on time scales longer than a second, “the motion of the RNA molecule is dominated by interactions with obstacles in the medium surrounding it”—interactions we expect only to be amplified in more complexly compartmentalized eukaryotes.
That such proteins are functional can be verified biochemically or by amino acid sequence similarity to homologous proteins in closely related organisms that do not have RNA editing.
The Black Queen gets its name from the game of Hearts, where the goal of the game is to avoid being stuck with the queen of spades; likewise the goal is to avoid being stuck with certain costly genes. We term the CNE variant of the BQH the Gray Queen Hypothesis, to emphasize the likeness of both views, the absence of an all-or-nothing approach to selection, and since gray is a neutral colour.
Something important is neglected thereby, and accepting CNE as a co-equal force to selection might help us recognize this level. It is after all this level that is the focal point of contemporary “systems biology” (Ideker et al. 2001).
One could also cite so-called “moonlighting proteins”. These have well-known and likely more ancient functions, but have been “recruited” to serve additional important roles in basic transcriptional or translational processes (or as crystallins in the vertebrate eye and as tumor suppressors). Presumably these associations (even if subsequently “improved by positive selection”) began as fortuitous interactions with pre-existing cellular entities (Jeffery 2003).
Moreover, as discussed in the next section, it may be a multi-species microbial community rather than a single prokaryotic cell that is appropriately compared to a eukaryotic cell.
Abbreviations
- CNE:
-
Constructive Neutral Evolution
- PPI:
-
Protein–protein interaction networks
- ZFEL:
-
Zero-force evolutionary law
- BQH:
-
Black Queen Hypothesis
- GQH:
-
Gray Queen Hypothesis
References
Archibald JM, Logsdon JM, Doolittle WF (1999) Recurrent paralogy in the evolution of archaeal chaperonins. Curr Biol 9(18):1053–S6
Archibald JM, Logsdon JM Jr, Doolittle WF (2000) Origin and evolution of eukaryotic chaperonins: phylogenetic evidence for ancient duplications in CCT genes. Mol Biol Evol 17(10):1456–1466
Atay O, Skotheim JM (2014) Modularity and predictability in cell signaling and decision making. Mol Biol of the Cell 25(22):3445–3450
Bell G (2001) Neutral macroecology. Science 293(5539):2413–2418
Ben-David E, Burga A, Krugkyak L (2017) A maternal-effect selfish element in Caenorhabditis elegans. Science 356(6342):1051–1055
Bernstein H, Byerly HC, Hopf FA, Michod RE (1984) Origin of sex. J Theor Biol 110(3):323–351
Booth A, Doolittle WF (2015) Eukaryogenesis, how special really? Proc Natl Acad Sci USA 112(33):10278–10285
Bridgham JT, Ortlund EA, Thornton JW (2009) An epistatic ratchet constrains the direction of glucocorticoid receptor evolution. Nature 461(7263):515–519
Brown CT, Hug LA, Thomas BC et al (2015) Unusual biology across a group comprising more than 15% of domain Bacteria. Nature 523:208–211
Brunet TDP, Doolittle WF (2015) Multi-Level Selection Theory and the evolutionary functions of transposable elements. Genome Biol Evol 7(8):2445–2457
Cafarelli TM, Desbuleux A, Wang Y et al (2017) Mapping, modeling, and characterization of protein-protein interactions on a proteomic scale. Curr Opin Struct Biol 44:201–210
Clarke E (2016) Levels of selection in biofilms: multispecies biofilms are not evolutionary individuals. Biol Philos 31(20):191–212
Cohen DM, Steger DJ (2017) Nuclear receptor function through genomics: lessons from the glucocorticoid receptor. Trends Endocrinol Metab 28(7):531–540
Covello P, Gray M (1993) On the evolution of RNA editing. Trends Genet 9(8):265–268
Doolittle WF (1998) You are what you eat: a gene transfer ratchet could account for bacterial genes in eukaryotic nuclear genomes. Trends Genet 14(8):307–311
Doolittle WF (2013) Is junk DNA bunk? A critique of ENCODE. Proc Natl Acad Sci USA 110(14):5294–5300
Doolittle WF (2017) Darwinizing Gaia. J Theor Biol 434:11–19
Doolittle WF, Booth A (2017) It’s the song, not the singer: an exploration of holobiosis and evolutionary theory. Biol Philos 32(1):5–24
Doolittle WF, Sapienza C (1980) Selfish genes, the phenotype paradigm and genome evolution. Nature 284(5757):601–603
Doolittle WF, Luke J, Archibald JM, Keeling PJ, Gray MW (2011) Comment on “Does constructive neutral evolution play an important role in the origin of cellular complexity?”. BioEssays 33(6):427–429
Embley TM, Williams TA (2015) Evolution: steps on the road to eukaryotes. Nature 521(7551):169–170
Fedoroff NV (2012) Transposable elements, epigenetics, and genome evolution. Science 338(6108):758–767
Fernandez A, Lynch M (2011) Non-adaptive origins of interactome complexity. Nature 474:502–505
Finnigan GC, Hanson-Smith V, Stevens TH, Thornton JW (2012) Evolution of increased complexity in a molecular machine. Nature 481(7381):360–364
Frey-Wyssling A (1978) Concerning the concept ‘organelle’. Experientia 34(4):547
Fullmer MS, Soucy SM, Gogarten JP (2015) The pan-genome as a shared genomic resource: mutual cheating, cooperation and the black queen hypothesis. Front Microbiol 6:728
Giovannoni SJ, Thrash JC, Temperton B (2014) Implications of streamlining theory for microbial ecology. ISME J 8:1553–1565
Godfrey-Smith P (1994) A modern history theory of functions. Nous 28(3):344–362
Golding I, Cox EC (2006) Physical nature of bacterial cytoplasm. Phys Rev Lett 96(9):09810
Gould SJ (1989) Wonderful life: the Burgess shale and the nature of life. Norton, New York
Gould SJ, Lewontin RC (1979) The spandrels of San Marco and the Panglossian paradigm: a critique of the adaptationist programme. Proc R Soc Lond B Biol Sci 205(1161):581–598
Gould SJ, Vrba ES (1982) Exaptation—a missing term in the science of form. Paleobiol 8(01):4–15
Gray MW, Lukeš J, Archibald JM, Keeling PJ, Doolittle WF (2010) Irremediable complexity? Science 330(6006):920–921
Grosche C, Funk HT, Maier UG, Zauner S (2012) The chloroplast genome of Pellia endiviifolia: gene content, RNA-editing pattern, and the origin of chloroplast editing. Genome Biol Evol 4(12):1349–1357
Hagstrom GI, Levin SA (2017) Marine ecosystems as complex adaptive systems: emergent patterns, critical transitions, and public goods. Ecosystems 20(3):458–476
Hartl F (2017) Protein misfolding diseases. In press, Ann Rev Biochem
Hartwell LH, Hopfield JJ, Leibler S, Murray AW (1999) From molecular to modular cell biology. Nature 402(6761 Suppl):C47–C52
Hughes T, Ekman D, Ardawatia H, Elofsson A, Liberles DA (2007) Evaluating dosage compensation as a cause of duplicate gene retention in Paramecium tetraurelia. Genome Biol 8(5):213
Hutchison CA, Chuang R-Y, Noskov VN et al (2016) Design and synthesis of a minimal bacterial genome. Science 351(6280):aad6253
Ideker T, Galitski T, Hood L (2001) A new approach to decoding life: systems biology. Ann Rev Genomics Hum Genet 2:343–372
Jablonski D (2008) Species selection: theory and data. Ann Rev Ecol Evol Syst 39:501–524
Jacob F (1977) Evolution and tinkering. Science 196(4295):1161–1166
Jeffery CJ (2003) Moonlighting proteins: old proteins learning new tricks. Trends Genet 19(8):415–417
Kapusta A, Feschotte C (2014) Volatile evolution of long noncoding RNA repertoires: mechanisms and biological implications. Trends Genet 30(10):439–452
Keeling PJ, McCutcheon JP, Doolittle WF (2015) Symbiosis becoming permanent: survival of the luckiest. Proc Natl Acad Sci 112(33):10101–10103
Kidwell MG (2002) Transposable elements and the evolution of genome size in eukaryotes. Genetica 115(1):49–63
Kimura M (1983) The neutral theory of molecular evolution. Cambridge University Press, Cambridge
Koonin EV (2016) Splendor and misery of adaptation, or the importance of neutral null for understanding evolution. BMC Biol 14(1):114
Kuo CH, Moran NA, Ochman H (2009) The consequences of genetic drift for bacterial genome complexity. Genome Res 19(8):1450–1454
Lamech LT, Mallam AL, Lambowitz AM (2014) Evolution of RNA-protein interactions: non-specific binding led to RNA splicing activity of fungal mitochondrial tyrosyl-tRNA sythetases. PLoS Biol 12(12):e1002028
Landry CR, Levy ED, Rabbo DA, Tarassov K, Michnick SW (2013) Extracting insight from noisy cellular networks. Cell 155(5):983–989
Lane N, Martin W (2010) The energetics of genome complexity. Nature 467(7318):929–934
Lane N, Martin WF (2015) Eukaryotes really are special, and mitochondria are why. Proc Natl Acad Sci 112(35):E4823
Launay G, Ceres N, Martin J (2017) Non-interacting proteins may resemble interacting proteins: prevalence and implications. Sci Rep 7:40419
Lukeš J, Archibald JM, Keeling PJ, Doolittle WF, Gray MW (2011) How a neutral evolutionary ratchet can build cellular complexity. IUBMB Life 63(7):528–537
Lynch M, Conery JS (2000) The evolutionary fate and consequence of duplicate genes. Science 290:1151–1155
Lynch M, Force A (2000) The probability of duplicate gene preservation by subfunctionalization. Genetics 154(1):459–473
Lynch M, Marinov GK (2017) Membranes, energetics, and evolution across the prokaryote-eukaryote divide. eLife 6:e20437
Majumder S, DeMott CM, Reverdatto S, Burz DS, Shektman A (2016) Total cellular RNA modulates protein activity. Biochemistry 55(32):4568–4573
Martin WF, Koonin EV (2006) Introns and the origin of nucleus-cytosol compartmentalization. Nature 440:41–45
Martiny AC, Treseder K, Pusch G (2013) Phylogenetic conservation of functional traits in microorganisms. ISME J 7:830–838
McShea DW, Brandon RN (2010) Biology’s first law: the tendency for diversity and complexity to increase in evolutionary systems. University of Chicago Press, Chicago
McShea DW, Venit EP (2001) What is a part. In: Wagner GP (ed) The character concept in evolutionary biology. Academic Press, London, pp 259–284
Mendoza E (2013) Organelle and functional module resources. In: Dubitzky W, Wolkenhauer O, Yokota H, Cho K-H (eds) Encyclopedia of systems biology. Springer, New York, pp. 1609–1611
Michod RE (1997) Cooperation and conflict in the evolution of individuality. I. Multilevel selection of the organism. Am Nat 149(4):607–645
Morris JJ, Lenski RE, Zinser ER (2012) The Black Queen Hypothesis: evolution of dependencies through adaptive gene loss. MBio 3(2):e00036–12
Newman ME (2006) Modularity and community structure in networks. Proc Natl Acad Sci 103(23):8577–8582
Novichkov PS, Wolf YI, Dubchak I, Koonin EV (2009) Trends in prokaryotic evolution revealed by comparison of closely related bacterial and archaeal genomes. J Bacteriol 191(1):65–73
O’Malley MA, Wideman JG, Ruiz-Trillo I (2016) Losing Complexity: the Role of Simplification in Macroevolution. Trends Ecol Evol 31(8):608–621
Okasha S (2005) Multilevel selection and the major transitions in evolution. Philos Sci 72(5):1013–1025
Omer S, Harlow TJ, Gogarten JP (2017) Does sequence conservation provide evidence for biological function? Trends Microbiol 25(1):11–18
Orgel LE, Crick FHC (1980) Selfish DNA: the ultimate parasite. Nature 284:604–607
Pande S, Kost C (2017) Bacterial unculturability and the formation of intercellular metabolic networks. Trends Microbiol 25(5):349–361
Rutherford SL (2003) Between genotype and phenotype: protein chaperones and evolvability. Nat Rev Genet 4:263–274
Sancar A (2008) The intelligent clock and the Rube Goldberg clock. Nat Struct Mol Biol 15(1):23–24
Simpson L, Thiemabb OH, Savill NJ, Alfonzo JD, Maslov DA (2000) Evolution of RNA editing in trypanosome mitochondria. Proc Natl Acad Sci USA 97:6986–6993
Sorrells TR, Johnson AD (2015) Making sense of transcription networks. Cell 161(4):714–723
Stoltzfus A (1999) On the possibility of constructive neutral evolution. J Mol Evol 49(2):169–181
Szathmary E, Smith JM (1995) Major transitions in evolution. Oxford University Press, Oxford
Van Melderen L (2010) Toxin-antitoxin systems: why so many, what for? Curr Opin Microbiol 13(6):781–785
Varzi A (2016) Mereology. The Stanford encyclopedia of philosophy. Zalta EN (ed). https://plato.stanford.edu/archives/win2016/entries/mereology/. Accessed 7 Nov 2017
Wang Z, Zhang J (2007) In search of the biological significance of modular structures in protein networks. PLoS Comput Biol 3(6):e107
Wolf YI, Koonin EV (2013) Genome reduction as the dominant mode of evolution. BioEssays 35(9):829–837
Yang JR, Liao BY, Zhuang SM, Zhang J (2012) Protein misinteraction avoidance causes highly expressed proteins to evolve slowly. Proc Natl Acad Sci 109(14):E831–E840
Zuckerkandl E (1992) Revisiting junk DNA. J Mol Evol 34(3):259–271
Acknowledgements
We thank Dr. S. A. Inkpen, and the Dalhousie-Kings STS reading group for helpful comments, and NSERC Discovery Grant GLDSU 447989 for support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brunet, T.D.P., Doolittle, W.F. The generality of Constructive Neutral Evolution. Biol Philos 33, 2 (2018). https://doi.org/10.1007/s10539-018-9614-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10539-018-9614-6