Abstract
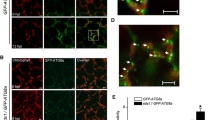
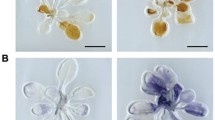
Autophagy has been implicated as a cellular protein degradation process that is used to recycle cytoplasmic components under biotic and abiotic stresses and so restrict programmed cell death (PCD). In this study, we report a novel regulatory mechanism by which NADPH oxidase respiratory burst oxidase homolog D (RBOHD) regulated pathogen-induced autophagy and hypersensitive (HR) cell death. We found that the Pseudomonas syringae pv tomato bacteria DC3000 expressing avrRps4 (Pst-avrRps4) induction of RBOHD-dependent reactive oxygen species (ROS) production promoted the onset of autophagy, whereas a pretreatment with an NADPH oxidase RBOHD inhibitor reversed this trend. The inhibitor significantly blocked pathogen-induced autophagosome formation and ROS increase. Moreover, we also show that in the wild-type and atrbohF mutant, Pst-avrRps4-induced cell death was limited, whereas in the case of the atrbohD mutant, the infection triggered a spreading-type necrosis. Our results demonstrate that the RBOHD-dependent ROS accumulation stimulated autophagosome formation and limited HR cell death.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- ATG :
-
autophagy-related genes
- BTH:
-
benzothiadiazole
- DAB:
-
3,3′-diaminobenzidine
- DCF:
-
2′,7′-dichlorofluorescein diacetate
- DPI:
-
diphenylene iodinium
- HR:
-
hypersensitive response
- PCD:
-
programmed cell death
- PR genes:
-
pathogen-related genes
- RBOHD:
-
respiratory burst oxidase homolog D
- RPS4:
-
resistance to Pseudomonas syringae 4
- SA:
-
salicylic acid
- WT:
-
wild type
References
Alvarez, M.E., Pennell, R.I., Meijer, P.J., Ishikawa, A., Dixon, R.A., Lamb, C.: Reactive oxygen intermediates mediate a systemic signal network in the establishment of plant immunity. — Cell 92: 773–784, 1998.
Bi, Y., Chen, W., Zhang, W., Zhou, Q., Yun, L., Xing, D.: Production of reactive oxygen species, impairment of photosynthetic function and dynamic changes in mitochondria are early events in cadmium induced cell death in Arabidopsis thaliana. — Biol. Cell 101: 629–643, 2009.
Bolwell, G.P., Davies, D.R., Gerrish, C., Auh, C.K., Murphy, T.M.: Comparative biochemistry of the oxidative burst produced by rose and French bean cells reveals two distinct mechanisms. — Plant Physiol. 116: 1379–1385, 1998.
Doelling, J.H., Walker, J.M., Friedman, E.M., Thompson, A.R., Vierstra, R.D.: The APG8/12-activating enzyme APG7 is required for proper nutrient recycling and senescence in Arabidopsis thaliana. — J. biol. Chem. 277: 33105–33114, 2002.
Gao, X., Chen, X., Lin, W., Chen, S., Lu, D., Niu, Y., Li, L., Cheng, C., McCormack, M., Sheen, J., Shan, L., He, P.: Bifurcation of Arabidopsis NLR immune signaling via Ca2+-dependent protein kinases. — PLoS Patholog. 9: e1003127, 2013.
Hammond-Kosack, K.E., Jones, J.D.: Resistance genedependent plant defense responses. — Plant Cell 8: 1773–1791, 1996.
Hanaoka, H., Noda, T., Shirano, Y., Kato, T., Hayashi, H., Shibata, D., Tabata, S., Ohsumi, Y.: Leaf senescence and starvation-induced chlorosis are accelerated by the disruption of an Arabidopsis autophagy gene. — Plant Physiol. 129: 1181–1193, 2002.
Hofius, D., Schultz-Larsen, T., Joensen, J., Tsitsigiannis, D.I., Petersen, N.H., Mattsson, O., Jorgensen, L.B., Jones, J.D., Mundy, J., Petersen, M.: Autophagic components contribute to hypersensitive cell death in Arabidopsis. — Cell 137: 773–783, 2009.
Iavicoli, A., Boutet, E., Buchala, A., Métraux, J.P.: Induced systemic resistance in Arabidopsis thaliana in response to root inoculation with Pseudomonas fluorescens CHA0. — MPMI 16: 851–858, 2003.
Inoue, Y., Suzuki, T., Hattori, M., Yoshimoto, K., Ohsumi, Y., Moriyasu, Y.: AtATG genes, homologs of yeast autophagy genes, are involved in constitutive autophagy in Arabidopsis root tip cells. — Plant Cell Physiol. 47: 1641–1652, 2006.
Jiang, M., Zhang, J.: Involvement of plasma-membrane NADPH oxidase in abscisic acid- and water stress-induced antioxidant defense in leaves of maize seedlings. — Planta 215: 1022–1030, 2002.
Lai, Z., Wang, F., Zheng, Z., Fan, B., Chen, Z.: A critical role of autophagy in plant resistance to necrotrophic fungal pathogens. — Plant J. 66: 953–968, 2011.
Lam, E.: Controlled cell death, plant survival and development. — J. mol. cell. Biol. 5: 305–315, 2004.
Levine, A., Tenhaken, R., Dixon, R., Lamb, C.: H2O2 from the oxidative burst orchestrates the plant hypersensitive disease resistance response. — Cell 79: 583–593, 1994.
Liu, J., Zhou, J., Xing, D.: Phosphatidylinositol 3-kinase plays a vital role in regulation of rice seed vigor via altering NADPH oxidase activity. — PLoS One 7: e33817, 2012.
Liu, Y., Bassham D.C.: TOR is a negative regulator of autophagy in Arabidopsis thaliana. — PLoS One 5: e11883, 2010.
Liu, Y., Schiff, M., Czymmek, K., Tallóczy, Z., Levine, B., Dinesh-Kumar, S.P.: Autophagy regulates programmed cell death during the plant innate immune response. — Cell 121: 567–577, 2005.
Liu, Y., Xiong, Y., Bassham, D.C.: Autophagy is required for tolerance of drought and salt stress in plants. — Autophagy 5: 954–963, 2009.
Mackey, D., Belkhadir, Y., Alonso, J.M., Ecker, J.R., Dangl, J.L.: Arabidopsis RIN4 is a target of the type III virulence effector AvrRpt2 and modulates RPS2-mediated resistance. — Cell 112: 379–389, 2003.
McDowell, J.M., Dangl, J.L.: Signal transduction in the plant immune response. — Trends Biochem. Sci. 25: 79–82, 2000.
Millet, Y.A., Danna, C.H., Clay, N.K., Songnuan, W., Simon, M.D., Werck-Reichhart, D., Ausubel, F.M.: Innate immune responses activated in Arabidopsis roots by microbeassociated molecular patterns. — Plant Cell 22: 973–990, 2010.
Ogasawara, Y., Kaya, H., Hiraoka, G., Yumoto, F., Kimura, S., Kadota, Y., Hishinuma, H., Senzaki, E., Yamagoe, S., Nagata, K., Nara, M., Suzuki, K., Tanokura, M., Kuchitsu, K.: Synergistic activation of the Arabidopsis NADPH oxidase AtrbohD by Ca2+ and phosphorylation. — J. biol. Chem. 283: 8885–8892, 2008.
Patel, S., Dinesh-Kumar, S.P.: Arabidopsis ATG6 is required to limit the pathogen-associated cell death response. — Autophagy 4: 20–27, 2008.
Pogány, M., Von Rad, U., Grün, S., Dongó, A., Pintye, A., Simoneau, P., Bahnweg, G., Kiss, L., Barna, B., Durner, J.: Dual roles of reactive oxygen species and NADPH oxidase RBOHD in an Arabidopsis-Alternaria pathosystem. — Plant Physiol. 151: 1459–1475, 2009.
Rose, T.L., Bonneau, L., Der, C., Marty-Mazars, D., Marty, F.: Starvation-induced expression of autophagy-related genes in Arabidopsis. — Biol. Cell 98: 53–67, 2006.
Scherz-Shouval, R., Shvets, E., Fass, E., Shorer, H., Gil, L., Elazar, Z.: Reactive oxygen species are essential for autophagy and specifically regulate the activity of Atg4. — EMBO J. 26: 1749–1760, 2007.
Seay, M., Hayward, A.P., Tsao, J., Dinesh-Kumar, S.P.: Something old, something new: plant innate immunity and autophagy. — Curr. Topics Microbiol. 335: 287–306, 2009.
Seay, M., Patel, S., Dinesh-Kumar, S.P.: Autophagy and plant innate immunity. — Cell Microbiol. 8: 899–906, 2006.
Slavíková, S., Shy, G., Yao, Y., Glozman, R., Levanony, H., Pietrokovski, S., Elazar, Z., Galili, G.: The autophagyassociated Atg8 gene family operates both under favourable growth conditions and under starvation stresses in Arabidopsis plants. — J. exp. Bot. 56: 2839–2849, 2005.
Thompson, A.R., Doelling, J.H., Suttangkakul, A., Vierstra, R.D.: Autophagic nutrient recycling in Arabidopsis directed by the ATG8 and ATG12 conjugation pathways. — Plant Physiol. 138: 2097–2110, 2005.
Torres, M.A., Dangl, J.L.: Functions of the respiratory burst oxidase in biotic interactions, abiotic stress and development. — Curr. Opin. Plant Biol. 8: 397–403, 2005.
Torres, M.A., Dangl, J.L., Jones, J.D.: Arabidopsis gp91phox homologues AtrbohD and AtrbohF are required for accumulation of reactive oxygen intermediates in the plant defense response. — Proc. nat. Acad. Sci. USA 99: 517–522, 2002.
Torres, M.A., Jones, J.D., Dangl, J.L.: Reactive oxygen species signaling in response to pathogens. — Plant Physiol. 141: 373–378, 2006.
Van Loon, L.C., Bakker, P., Pieterse, C.M.J.: Systemic resistance induced by rhizosphere bacteria. — Annu. Rev. Phytopathol. 36: 453–483, 1998.
Wang, Y., Nishimura, M.T., Zhao, T., Tang, D.: ATG2, an autophagy-related protein, negatively affects powdery mildew resistance and mildew-induced cell death in Arabidopsis. — Plant J. 68: 74–87, 2011.
Wojtaszek, P.: Oxidative burst: an early plant response to pathogen infection. — Biochem. J. 322: 681–692, 1997.
Xiong, Y., Contento, A.L., Bassham, D.C.: AtATG18a is required for the formation of autophagosomes during nutrient stress and senescence in Arabidopsis thaliana. — Plant J. 42: 535–546, 2005.
Xiong, Y., Contento, A.L., Nguyen, P.Q., Bassham, D.C.: Degradation of oxidized proteins by autophagy during oxidative stress in Arabidopsis. — Plant Physiol. 143: 291–299, 2007.
Yoshimoto, K., Jikumaru, Y., Kamiya, Y., Kusano, M., Consonni, C., Panstruga, R., Ohsumi, Y., Shirasu, K.: autophagy negatively regulates cell death by controlling NPR1-dependent salicylic acid signaling during senescence and the innate immune response in Arabidopsis. — Plant Cell 21: 2914–2927, 2009.
Yun, L.J., Chen, W.L.: SA and ROS are involved in methyl salicylate-induced programmed cell death in Arabidopsis thaliana. — Plant Cell Rep. 30: 1231–1239, 2011.
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgments: This study was supported by the National Natural Science Foundation of China (31170250) and Natural science Foundation of Guangdong Province (2014A030313420). We are grateful to Richard D. Vierstra (the Department of Genetics, the University of Wisconsin, Madison, USA) for kindly providing the Arabidopsis thaliana seeds stably expressing 35S:GFP-ATG8a.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Liu, H.B., Wang, X.D., Zhang, Y.Y. et al. NADPH oxidase RBOHD contributes to autophagy and hypersensitive cell death during the plant defense response in Arabidopsis thaliana . Biol Plant 59, 570–580 (2015). https://doi.org/10.1007/s10535-015-0519-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10535-015-0519-9